Abstract
Secretogranin II (SgII) and III (SgIII) function within peptide hormone-producing cells and are involved in secretory granule formation. However, their function in active amine-producing cells is not fully understood. In this study, we analyzed the expression profiles of SgII and SgIII in canine adrenal medulla and pheochromocytomas by immunohistochemical staining. In normal adrenal tissues, the intensity of coexpression of these two secretogranins (Sgs) differed from each chromaffin cell, although a complete match was not observed. The coexpression of vesicular monoamine transporter 2 (VMAT2) with SgIII was similar to that with chromogranin A, but there was a subpopulation of VMAT2-expressing cells that were negative or hardly detectable for SgII. These results are the first to indicate that there are distinct expression patterns for SgII and SgIII in adrenal chromaffin cells. Furthermore, the expression of these two Sgs varied in intensity among pheochromocytomas and did not necessarily correlate with clinical plasma catecholamine levels in patients. However, compared with SgIII, the expression of SgII was shown to be strong at the single-cell level in some tumor tissues. These findings provide a fundamental understanding of the expression differences between SgII and SgIII in normal adrenal chromaffin cells and pheochromocytomas.
Keywords
Introduction
Granins are a family of acidic secretory proteins composed of chromogranin A (CgA), chromogranin B, secretogranin II (SgII), secretogranin III (SgIII), and SgIV-VI, all of which have widespread distribution in endocrine and neuronal cells.1–3 These proteins possess the characteristics of acidic secretory proteins localized inside secretory granules and coaggregate with peptide hormones during the granule formation in endocrine cells. 4 In this process, the role of granins has been identified by their propensity to aggregate in a low-pH, high-calcium environment within the trans-Golgi network, followed by their efficient sorting to the regulated pathway. 5 Thus, granins are considered markers for a regulated pathway and may play a role in secretory granule formation.
SgII possesses two protein domains that bind to cholesterol-rich microdomains of the secretory granule membranes and are involved in the aggregation process of peptide hormones independently of interactions with CgA.6–8 Moreover, SgIII plays a role as a bridging molecule between the peptide hormone/CgA core aggregates and the cholesterol-rich microdomains of the secretory granule membranes.9,10 In addition to being a shared executor for the aggregate formation of peptide hormones, there is a clear difference between SgII and SgIII. While a short peptide fragment, secretoneurin, has been demonstrated to be formed by proteolytic processing of SgII and acts as an active substance,11,12 a corresponding property has not been found in SgIII, despite undergoing similar proteolytic processing. 3 However, recent reports suggest an interesting novel disease-specific function of SgIII as an angiogenic and vascular leakage factor that has a selective binding for diabetic retinal vessels.13,14 On the other hand, secretoneurin derived from SgII has been shown to have a role in developmental and physiological vasculogenesis.15–17
SgII expression has been extensively reported in normal adrenal medullary cells in humans and various animals18–24 and catecholamine-secreting PC12 cells, which are a cell line derived from a rat pheochromocytoma.25,26 These early studies demonstrated that SgIII was significantly expressed in neurons and peptide hormone-secreting cells9,27–30; however, its expression in biogenic active amine-secreting cells has not been sufficiently evaluated.9,27,31,32 Subsequently, the expression of SgIII has been identified in PC12 cells 33 and in mast cells that contain histamine granules. 34 We have recently demonstrated that SgIII is fully expressed in adrenal medullary cells and active amine-secreting gastrointestinal endocrine cells in mice, rats, and chickens.35,36 From a comparative perspective of vertebrate species, expression analysis of these two Sgs in the canine adrenal tissues is limited.21,37
The most compelling function of SgII and SgIII appears to be related to the formation of hormone peptide aggregation in peptide hormone-producing cells; however, the role these granins play in granules containing biogenic amines remains unclear. Compared with peptide hormone-producing cells, synthesized monoamines are transported and stored in dense-core vesicles for exocytosis in adrenal chromaffin cells. The packaging of the monoamine transmitter into dense-core vesicles is mediated by specific transporters that are localized on the vesicular membrane, termed vesicular monoamine transporters 1 and 2 (VMAT1 and VMAT2).38–40 The essential differences between both SgII and SgIII in adrenal chromaffin cells have not been fully evaluated. In this study, we evaluated the expression of SgII and SgIII in the normal canine adrenal medulla and utilized immunohistochemistry to compare their expression patterns. A novel monoclonal antibody against SgIII was also prepared for this approach. In normal adrenal medulla, differential expression of SgII and SgIII has been associated with VMAT2 expression.
In addition to analyzing normal adrenal tissues, we further analyzed adrenal pheochromocytomas. In particular, the evaluation of SgIII expression in pheochromocytomas has thus far been limited to human cases, and there is only one such report detailing this analysis. 41 In addition to humans, spontaneous cases of pheochromocytoma have been observed in dogs, cattle, and rats. 42 Among these animal species, pheochromocytoma occurs spontaneously and most commonly in dogs. 43 Histological investigations of canine pheochromocytoma might be useful as human disease models for analyzing the pathogenesis and mechanism of pheochromocytoma, as well as for further study of novel antineoplastic therapies. Although SgII and SgIII are presumed to be involved in catecholamine granule metabolism, no histological analysis has been reported in canine pheochromocytomas. Therefore, we focused on the relationship between SgII/SgIII expression and clinicopathological findings (blood catecholamine levels and tumor metastasis) in canine pheochromocytomas. The findings indicate that the expression of both SgII and SgIII was inconsistent in normal adrenal tissues and pheochromocytomas.
Materials and Methods
Animals
Two male beagle dogs (Canis lupus familiaris) aged 9–10 months were purchased from Kitayama Labes Co., Ltd. (Nagano, Japan). Tissue samples from 4- to 6-month-old male SgIII gene-trapped mice (SgIII-gtKO, n=6) and control wild-type mice (SgIII-WT, n=6) that had a genetic background of a C57BL/6J strain36,44 were used as controls in the immunohistochemical and biochemical experiments. Following euthanasia by intravenous injection of 50-mg/kg body weight (bw) sodium pentobarbital and 2-mmol/kg/bw potassium chloride solution, the dogs were processed for sample preparation. The mice were anesthetized by intraperitoneal injection of 0.1-mg/g/bw sodium pentobarbital and then subsequently processed for sample preparation. All animal experiments were performed in compliance with both the guidelines of the Care and Use of Laboratory Animals at the Institute of Experimental Animal Science, College of Bioresource Sciences, Nihon University (approval number AP15B051), and the guidelines of the Care and Use of Laboratory Animals of the Research Council of Akita Prefectural University (approval number 17-03 and 18-02), Japan. Written informed consent was obtained from the patient animal owners for the participation of their animals in this study.
Primary Antibodies
The antibodies utilized in this study against granins, secretory granule-associated proteins, and neuroendocrine tumor biomarkers are detailed in Table 1. Rabbit polyclonal anti-SgIII antibodies (SgIII-C#6) were prepared as previously described, 10 and anti-SgIII monoclonal antibodies (SgIII-mAb134) were prepared as described below.
Antibodies Used for the Analysis.
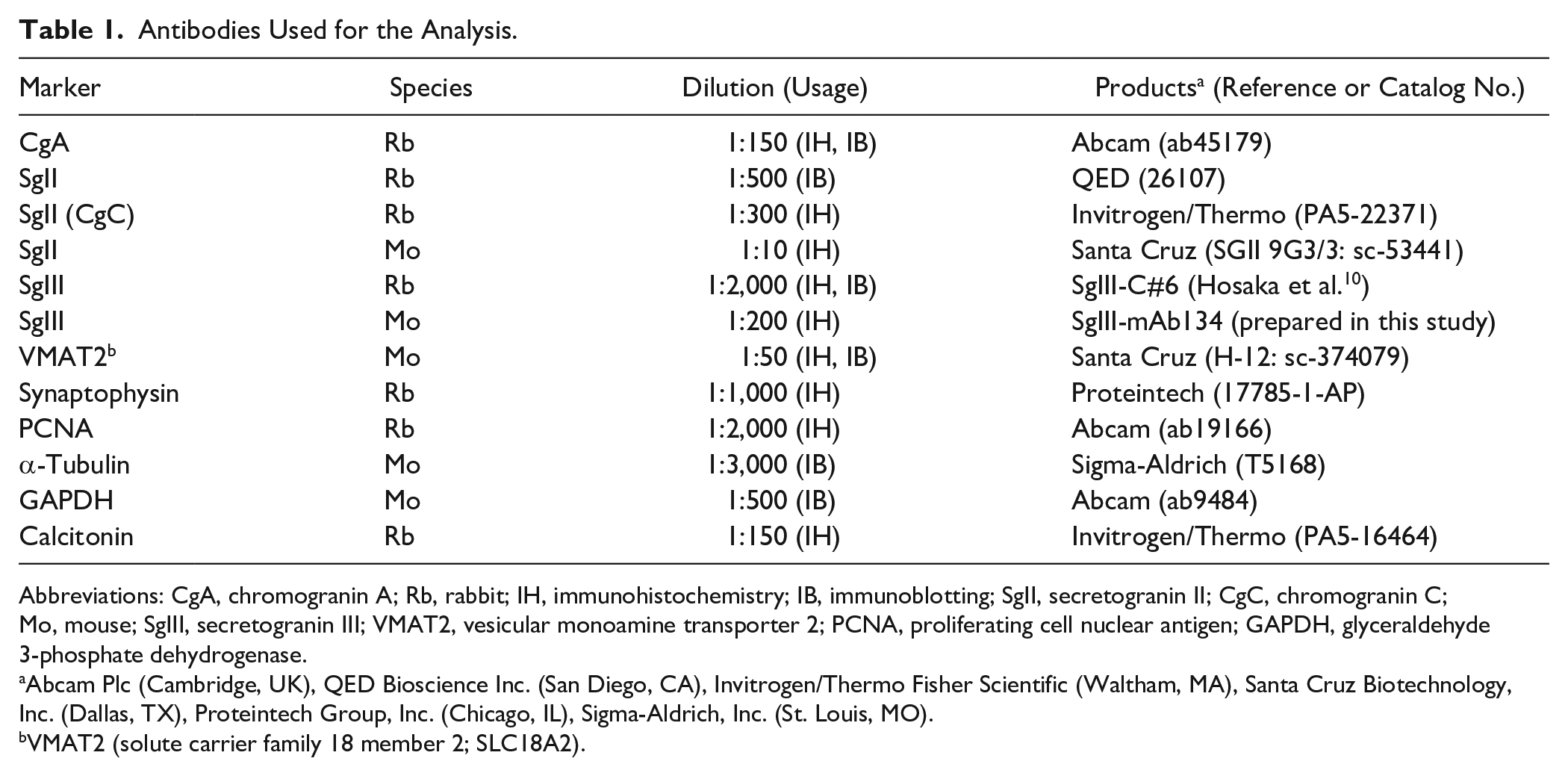
Abbreviations: CgA, chromogranin A; Rb, rabbit; IH, immunohistochemistry; IB, immunoblotting; SgII, secretogranin II; CgC, chromogranin C; Mo, mouse; SgIII, secretogranin III; VMAT2, vesicular monoamine transporter 2; PCNA, proliferating cell nuclear antigen; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Abcam Plc (Cambridge, UK), QED Bioscience Inc. (San Diego, CA), Invitrogen/Thermo Fisher Scientific (Waltham, MA), Santa Cruz Biotechnology, Inc. (Dallas, TX), Proteintech Group, Inc. (Chicago, IL), Sigma-Aldrich, Inc. (St. Louis, MO).
VMAT2 (solute carrier family 18 member 2; SLC18A2).
Preparation of Novel Monoclonal Antibodies Against SgIII
For the immunogen, glutathione-S-transferase (GST)-fused rat SgIII carboxyterminal peptides (amino acids: 373–471) were used. 10 Three 5- to 6-week-old BALB/c female mice were immunized with 20 µg of GST/SgIII-fusion protein conjugated with Freund’s complete adjuvant. Following this, booster immunizations were repeated 3× by injecting 10 µg of fusion protein conjugated with Freund’s incomplete adjuvant at 2-week intervals. The spleens were isolated from immunized mice 7 days after the final booster shot for the preparation of splenocytes. Polyethylene glycol was used for cell fusion of the splenocytes and myeloma cells (P3U1) to prepare hybridomas, according to the conventional protocol in entrustment service (Cosmo Bio Co., Ltd.; Tokyo, Japan). The hybridoma clones were analyzed using an enzyme-linked immunosorbent assay (ELISA), and the hybridoma-produced supernatants were evaluated for their antibody titers. For the ELISA, 50 µL of maltose-binding protein-fused SgIII 373–471 peptides 10 were plated (2-µg/mL concentration), blocked using 1% skim milk/phosphate-buffered saline (PBS), and reacted with 50 µL of supernatant for 1 hr at room temperature (RT). After washing with PBS 3×, horseradish peroxidase (HRP)-labeled anti-mouse IgG (115-035-146) (1:10,000 dilution; Jackson ImmunoResearch Laboratory, West Grove, PA) was added and incubated for 1 hr at RT. The 3,3′,5,5′-tetramethylbenzidine substrate was used to detect HRP activity, and the intensity of the developed color was determined using a spectrophotometer at 450 nm. We further evaluated the hybridoma clones by immunohistochemistry using pituitary and adrenal paraffin sections prepared from mice in the SgIII-WT and SgIII-gtKO groups. Finally, a selected hybridoma clone (1 × 107 cells) was transferred into the peritoneal cavity of BALB/c nude (nu/nu) mice, and the ascites were recovered to prepare a high titer stock using the entrustment service (Immuno-Biological Laboratories Co., Ltd.; Gunma, Japan).
Canine Adrenal Pheochromocytomas
The tissue samples of canine adrenal pheochromocytomas were removed by adrenalectomy surgery conducted at the Animal Medical Center of Nihon University, and all resected tumors were histopathologically diagnosed as pheochromocytomas. 45 In this study, we analyzed five cases of canine pheochromocytomas (Table 2). The clinical information collected included the plasma levels of catecholamines, which were subdivided into two groups: high plasma catecholamine levels (HCL; three cases) and low plasma catecholamine levels (LCL; two cases) plasma levels of catecholamines. In all cases, the adrenal tumors formed tumor emboli in the caudal vena cava. In addition, in one LCL patient, a complication of thyroid medullary carcinoma was observed.
Clinical Observation of Pheochromocytoma Patient Dogs.
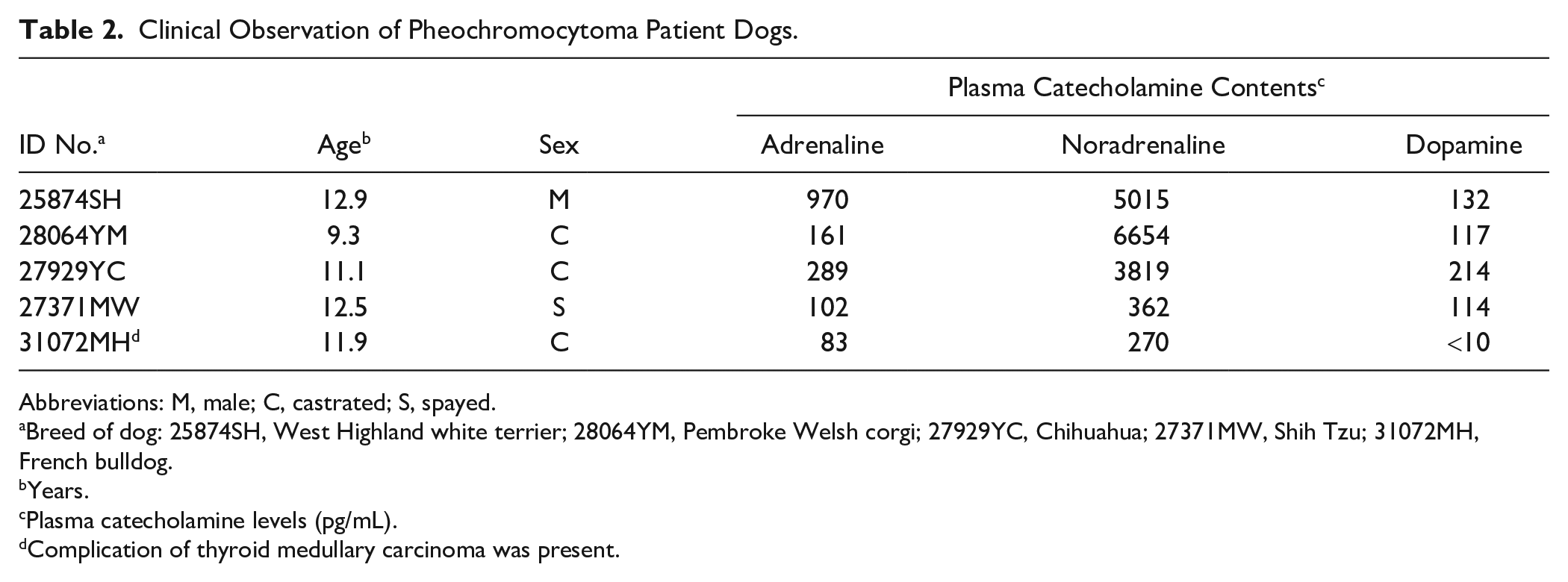
Abbreviations: M, male; C, castrated; S, spayed.
Breed of dog: 25874SH, West Highland white terrier; 28064YM, Pembroke Welsh corgi; 27929YC, Chihuahua; 27371MW, Shih Tzu; 31072MH, French bulldog.
Years.
Plasma catecholamine levels (pg/mL).
Complication of thyroid medullary carcinoma was present.
Tissue Preparation
The normal adrenal glands and pathological pheochromocytoma tissues were fixed in Bouin’s fixative or neutral buffered formalin for histological staining. For histological analysis, the fixed tissue blocks were dehydrated and embedded in paraffin. Tissue samples of the adrenal medulla from healthy beagle dogs and whole adrenal glands from mice were frozen on dry ice and stored at −80C before biochemical analysis.
Histological and Immunohistochemical Analyses
Tissue samples were sectioned with a 5-µm thickness and mounted on silane-coated glass slides (Muto Pure Chemicals; Tokyo, Japan). These sections were deparaffinized in xylene and then rehydrated in a series of graded ethanol and water solutions. Hematoxylin and eosin (HE) staining was applied to observe the gross histological features of the tissues. For immunohistochemical analysis of the enzyme antibody method, the sections were processed for antigen retrieval by heating in a microwave after rehydration as previously described. 35 The sections were treated with Dako REAL Peroxidase-Blocking Solution (Dako; Glostrup, Denmark) and blocked with 5% normal goat serum (Vector Laboratories; Burlingame, CA) in PBS before antibodies were added. After incubation with the primary antibodies overnight at 4C, the sections were incubated with secondary antibodies for 2 hr at RT using the HRP-labeled polymer technique with the EnVision Dual Link System–HRP Kit (Dako). Sections were subsequently visualized using the SK-4100 peroxidase substrate kit (Vector Laboratories) and counterstained with Mayer’s hematoxylin. There was no difference in the stainability of the immunoreaction depending on the fixative solution used. Control immunohistochemistry samples were prepared following the guidelines set by the Histochemical Society. 46 The negative control sections for SgIII immunoreaction were incubated with a mixture of the diluted anti-SgIII antibodies that were pretreated with absorbing SgIII peptides 373–471 (concentration 5 µg/mL) and normal rabbit IgG (sc-2027, 1-µg/mL concentration; Santa Cruz Biotechnology, Santa Cruz, CA), which was in place of the primary antibodies. For the immunoreaction of SgIII monoclonal antibodies, the tissue sections prepared from SgIII-gtKO were stained to evaluate reaction specificity. For the negative controls other than the SgIII immunoreaction, sections were incubated with normal rabbit, mouse, or guinea pig IgG (sc-2027, sc-2025, or sc-2711, 1-µg/mL concentration; Santa Cruz Biotechnology) instead of the primary antibodies. It was confirmed that no significant immunosignals were observed in these samples. Images were acquired using a BX51 microscope equipped with a DP72 CCD camera and digitized with the DP2-BSW software (Olympus; Tokyo, Japan).
Double Fluorescence Immunohistochemical Analyses
To analyze the coexpression of granins and adrenal intrinsic proteins in canine adrenal glands, double immunofluorescence staining was performed as previously described. 35 The sections were first incubated with a mixture of primary antibodies followed by Alexa Fluor-conjugated secondary antibodies (1:2000 dilution; Invitrogen/Thermo Fisher Scientific, Waltham, MA). For the analysis of the coexpression of SgII and CgA, SgIII and CgA, or SgII and SgIII, a combination of either Alexa 555-conjugated anti-mouse IgG or Alexa 488-conjugated anti-rabbit IgG was used. To analyze the coexpression of VMAT2 and CgA, VMAT2 and SgII, or VMAT2 and SgIII, Alexa 488-conjugated anti-mouse IgG and Alexa 555-conjugated anti-rabbit IgG were used. For the negative controls, sections were incubated with a mixture of normal animal IgGs corresponding to the animal species of the primary antibodies. The fluorescence images were acquired using a BX51 microscope equipped with a DP73 CCD camera and digitized with the cellSens Standard 1.8.1 software (Olympus).
Immunoblotting
To analyze the expression of granins in mouse and canine adrenal glands, immunoblotting was performed as previously described.35,36 Frozen adrenal medulla collected from healthy dogs and whole adrenal glands from mice were dethawed and homogenized using a lysis buffer that contained 20-mM Tris-HCl (pH 7.5), 150-mM NaCl, 2.5-mM MgCl2, 1-mM EGTA, 1.0% Triton X-100, and a protease inhibitor cocktail (Complete mini; Roche Diagnostics, Mannheim, Germany). The homogenates were spun at 20,000 × g for 10 min. The supernatant was collected, and the protein concentration was determined using a Coomassie brilliant blue protein assay reagent (Nacalai Tesque; Kyoto, Japan). Protein extracts were normalized to the total protein content (10–20 µg), subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE), and transferred to an Immobilon-P transfer membrane (Millipore; Billerica, MA). After blocking for 1 hr with 5% skim milk in Tris-buffered saline (pH 7.4) that was supplemented with 0.05% Tween-20 (TBS-T), the membrane was incubated with the primary antibodies. Peroxidase-conjugated secondary antibodies (Jackson ImmunoResearch Laboratory), anti-rabbit IgG (cat. no 111-035-144), or anti-goat IgG (705-035-147) were diluted (1:5000) with 5% skim milk/TBS-T and used. For serial reprobing of the same blot membrane to react with another antibody, the membranes were treated with reprobing buffer that contained 6-mM Tris-HCl (pH 6.8), 0.2% SDS, and 0.7% 2-mercaptoethanol (Nacalai Tesque) for 30 min and then washed with TBS-T before incubation with subsequent antibodies. The detection of the protein signal was performed using Amersham ECL Western blotting detection reagents (PRN2106; GE Healthcare Bio-Sciences KK, Tokyo, Japan).
Morphometric Analysis
Morphometric analysis of immunohistochemically labeled pheochromocytoma sections was performed using Fiji ImageJ software (https://imagej.net/software/fiji/). 47 To keep the staining conditions constant, the sections were processed simultaneously for the antibody and 3,3´-Diaminobenzidine tetrahydrochloride hydrate (DAB) chromogenic reactions. Images taken with an objective lens 40× magnification in three non-overlapping fields of different sections were used for measurement. To count the number of proliferating cell nuclear antigen (PCNA)-positive cells (brown nuclei) and PCNA-negative cells (blue nuclei) in the whole field, Cell Counter (plugins → analyze) was used. The immunoreactivity intensity of SgII and SgIII was assessed under the two threshold settings to segregate the strongly or weakly brown-stained areas. The color images were split into red, green, and blue channels (image → color → split channels), and color subtraction (process → image calculator) of the blue channel from the red channel for the weakly brown-stained area or color subtraction of the blue/green channels from the red channel for the strongly brown-stained area was conducted. The thresholds were set at 25–255 in the former group of processed images and set at 1–255 in the latter group of processed images (image → adjust → threshold). Then, the total field area and limit to threshold area (brown-stained area) were measured (analyze → measure) and the percentage of immunoreactive area was calculated.
Results
Expression of Granins in Canine Adrenal Glands
First, the expression of SgII, SgIII, and VMAT2 in mouse and canine adrenal tissues was examined by immunoblotting (Fig. 1). The expression of canine SgII had a major immunoreactive signal at the expected molecular size (86 kDa), which was also observed for the mouse proteins (Fig. 1A). In immunolabeling using an anti-SgII polyclonal antibody (QED, 26107), an identical peptide sequence (human SgII amino acids 31–49, QRNQLLQKEPDLRLENVQK, accession no. AAA36607) as an immunogen was confirmed among humans, mice, and dogs. SgIII expression in mouse and dog tissues was examined using an anti-SgIII polyclonal antibody (SgIII-C#6, against rat SgIII amino acids 373–471). The amino acid sequence similarities of the SgIII C-terminus in these animals were as follows: 98 of the 99 amino acid residues were identical in both rats and mice (98.9% identity), and 93 of the 99 residues were identical in both rats and dogs (93.9% identity). The immunoreactive signals were detected by multiple bands that were considered a precursor and proteolytically processed forms, as previously demonstrated in several animal species,29,35,36,44,48 while no immunoreaction was shown in the SgIII-gtKO sample (Fig. 1B). Therefore, the anti-SgIII antibody cross-reacted with canine SgIII, although there should be a slight difference in the pattern of the detectable signal bands caused by proteolytic processing because an excessive 16 amino acid insertion is shown in the amino-terminal region of canine SgIII (National Center for Biotechnology Information, [NCBI] Reference Sequence: XP_038298264.1). The expression of the adrenal marker protein VMAT2 was examined using an anti-VMAT2 monoclonal antibody (sc-374079, against human VMAT2 amino acids 44–133) at the expected molecular size (63 kDa). The amino acid sequence similarities in these animals were as follows: 67 of the 90 amino acid residues were identical in both humans and mice (74.4% identity), and 65 of the 90 residues were identical in both humans and dogs (72.2% identity), where the cross-reactivity in immunoblotting between human and mouse antigens was shown by the manufacturer’s datasheet, and dog antigen was also predicted to cross-react with anti-VMAT2 antibody. The immunoreactive signals of VMAT2 with an expected molecular size were observed in samples from both mice and dogs (Fig. 1C).
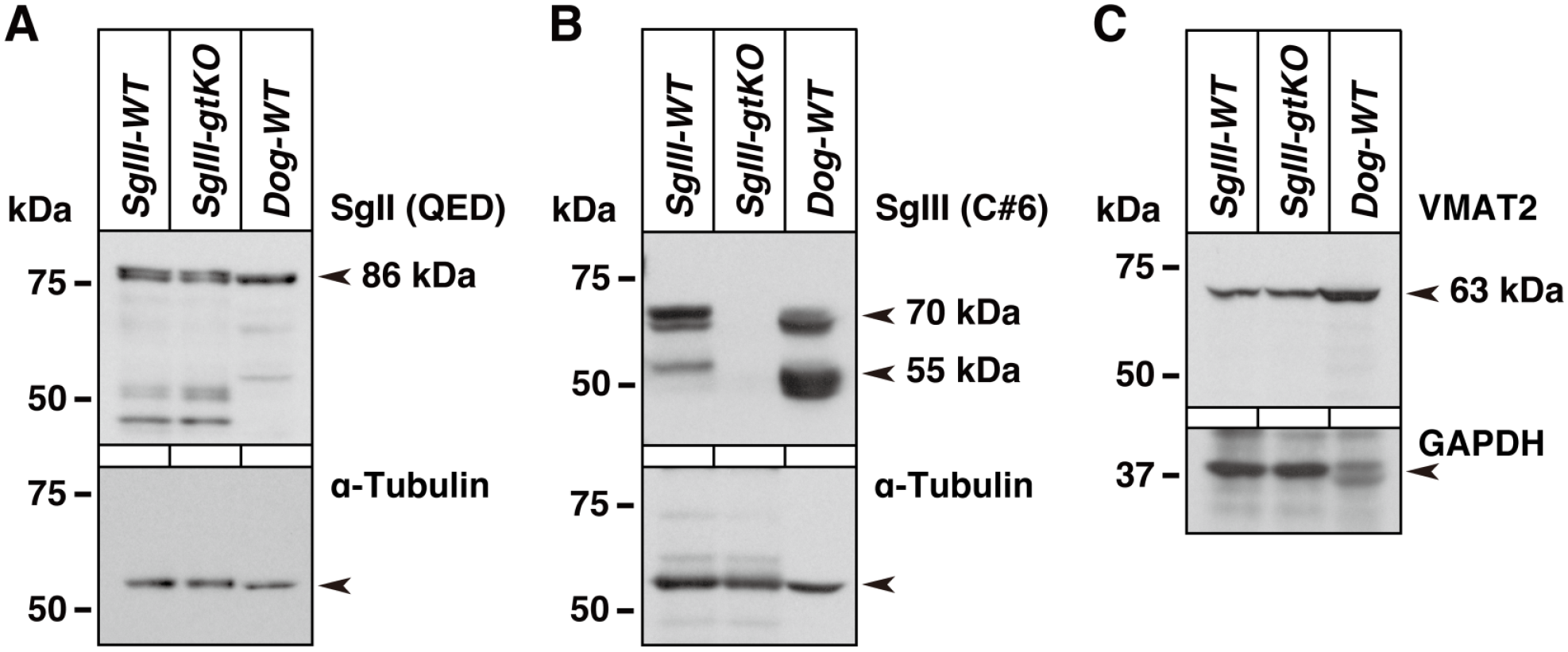
Immunoblot analysis of SgII, SgIII, and VMAT2 in the adrenal glands. Tissue protein extracts (20 µg) from the whole adrenal glands of SgIII-WT, SgIII-gtKO, and the adrenal medulla of beagle dogs (Dog-WT) were used for SDS–PAGE, followed by immunoblotting using anti-SgII (A), anti-SgIII (B), and anti-VMAT2 (C) antibodies. The bottom panels show the reprobed immunoblots using control anti-α-Tubulin or anti-GAPDH antibodies. Abbreviations: SgII, secretogranin II; SgIII, secretogranin III; VMAT2, vesicular monoamine transporter 2; WT, wild-type; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; kDa, kilodalton.
We subsequently examined the expression of granins as well as synaptophysin, a neuroendocrine cell marker, and PCNA, a marker of proliferating cells, in canine adrenal tissues by immunohistochemical analysis using rabbit polyclonal antibodies. The adrenal expression of these granins was observed within the medullary region. While the immunoreaction of CgA was shown to be strong overall in the medullary cells (Fig. 2B), immunoreactions of SgII and SgIII were observed with different staining intensities for each cell, which was more pronounced in the SgII immunoreaction than in the SgIII reaction (Fig. 2C and D). The expression of synaptophysin was also shown in the medullary cells, whereas the expression of PCNA was not observed (Fig. 2E and F). None of the immunosignals were observed in the negative control sections, which were reacted with normal rabbit IgG (Fig. 2G). Immunohistochemical cross-reactivity of anti-SgIII antibody on canine tissue sections was confirmed by incubation with a preadsorbed antibody by the immunogen, where a loss of immunoreactive signal occurred (Fig. 2H).
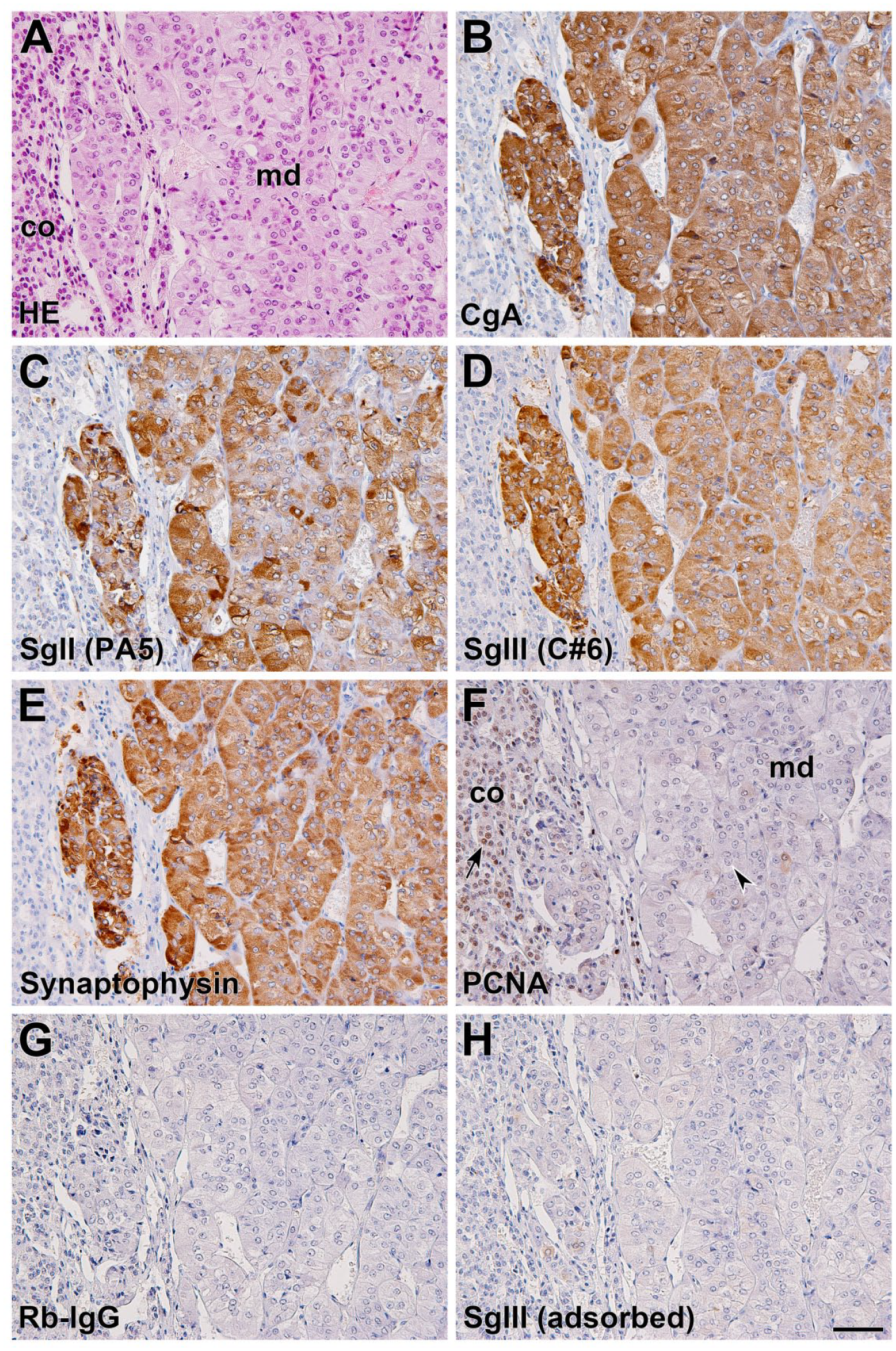
Histological and immunohistochemical analysis of granins, synaptophysin, and PCNA in canine adrenal glands. Histological HE staining of canine adrenal glands (A). Immunohistochemical staining was conducted using anti-CgA (B), anti-SgII (PA5) (C), anti-SgIII (C#6) (D), anti-synaptophysin (E), and anti-PCNA (F) polyclonal antibodies. Note that PCNA is positive for the nuclei of cortical cells (arrow), but not for those of medullary cells (arrowhead) in normal adrenal glands. Negative control sections were treated with normal Rb-IgG instead of a primary antibody (G) or with a preadsorbed anti-SgIII antibody on the purified GST-SgIII fusion protein (rat SgIII amino acids 373–471; 5 µg/mL) (H). Sections were counterstained with hematoxylin. Scale, 50 µm. Abbreviations: PCNA, proliferating cell nuclear antigen; CgA, chromogranin A; SgII, secretogranin II; SgIII, secretogranin III; Rb-IgG, rabbit IgG; GST, glutathione-S-transferase; md, adrenal medulla; co, adrenal cortex.
Preparation of a Novel Monoclonal Antibody Against SgIII, mAb134
To investigate the difference in the innate expression properties between SgII and SgIII in adrenal chromaffin cells, a novel anti-SgIII monoclonal antibody was prepared for use with immunohistochemical double staining. In the standard method using a hybridoma culturing method for the preparation of monoclonal antibodies, 10 of the 288 mixed clones were selected by the initial ELISA screening. After establishing single clones by limiting dilution cultures and expansion cultures, we selected four clones that were validated to be reactive in the screening of immunohistochemical staining. For one of the clones, the mAb134 clone had favorable immunohistochemical reactivities, that is, a specific immunoreaction was observed in the endocrine tissue sections (pancreatic islets, anterior pituitary glands, and adrenal glands) from SgIII-WT but not in those from SgIII-gtKO (Supplemental Fig. S1). In the canine adrenal tissue sections, the immunoreactive signals of mAb134 were detected in the medullary cells, and treatment with a preadsorbed immunogen with mAb134 resulted in a loss of signal (Fig. 3). Thus, a novel mAb134 monoclonal antibody was useful in the immunohistochemical detection of SgIII in canine adrenal tissues.
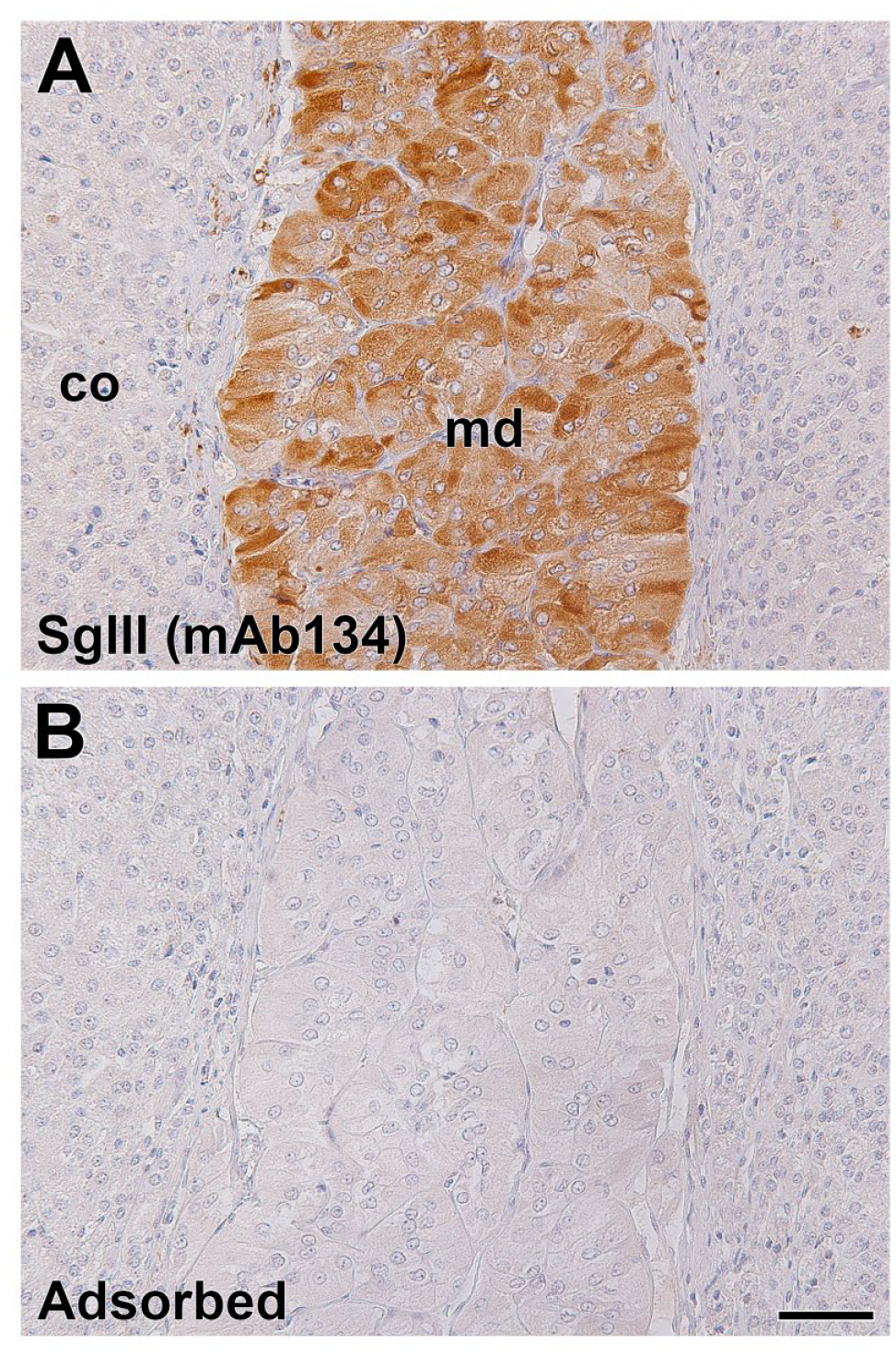
Immunohistochemical staining of canine adrenal glands using a novel mAb134 anti-SgIII monoclonal antibody. Section of canine adrenal glands was reacted with mAb134 monoclonal antibody (A). The negative control section was reacted with mAb134 preadsorbed by a GST-SgIII fusion protein (B). Sections were counterstained with hematoxylin. Scale, 50 µm. Abbreviations: SgIII, secretogranin III; GST, glutathione-S-transferase; md, adrenal medulla; co, adrenal cortex.
Coexpression of Granins in Canine Adrenal Glands
Coexpression of SgII or SgIII with CgA, and SgII with SgIII was analyzed using a double immunofluorescent antibody method. The pattern of the coexpression of SgII with CgA was obviously different from that of SgIII with CgA using monoclonal antibodies against SgII (9G3/3) or SgIII (mAb134) and a polyclonal antibody against CgA. The overall coexpression pattern of SgII and CgA showed the feature of a frequent occurrence of cells that expressed CgA more predominantly, while that of SgIII and CgA was in accordance with their expression intensities (Fig. 4A–F). On the other hand, by using the cross combination of monoclonal and polyclonal antibodies against SgII or SgIII, the coexpression pattern of SgII and SgIII was not completely matched, although many cells were observed to have consistent coexpression patterns. Several cells showed stronger expression of SgIII with very weak or negative expression of SgII (Fig. 4G–L). Conversely, it was also observed that there were a small number of cells that showed stronger expression of SgII than of SgIII (Supplemental Fig. S2). Thus, the expression of SgII in canine adrenal medullary cells was restricted to a specific subpopulation of medullary cells. Negative control sections that were treated with a mixture of normal mouse and rabbit IgG, instead of primary antibodies, are shown in the Supplemental Material (Supplemental Fig. S3A–C).
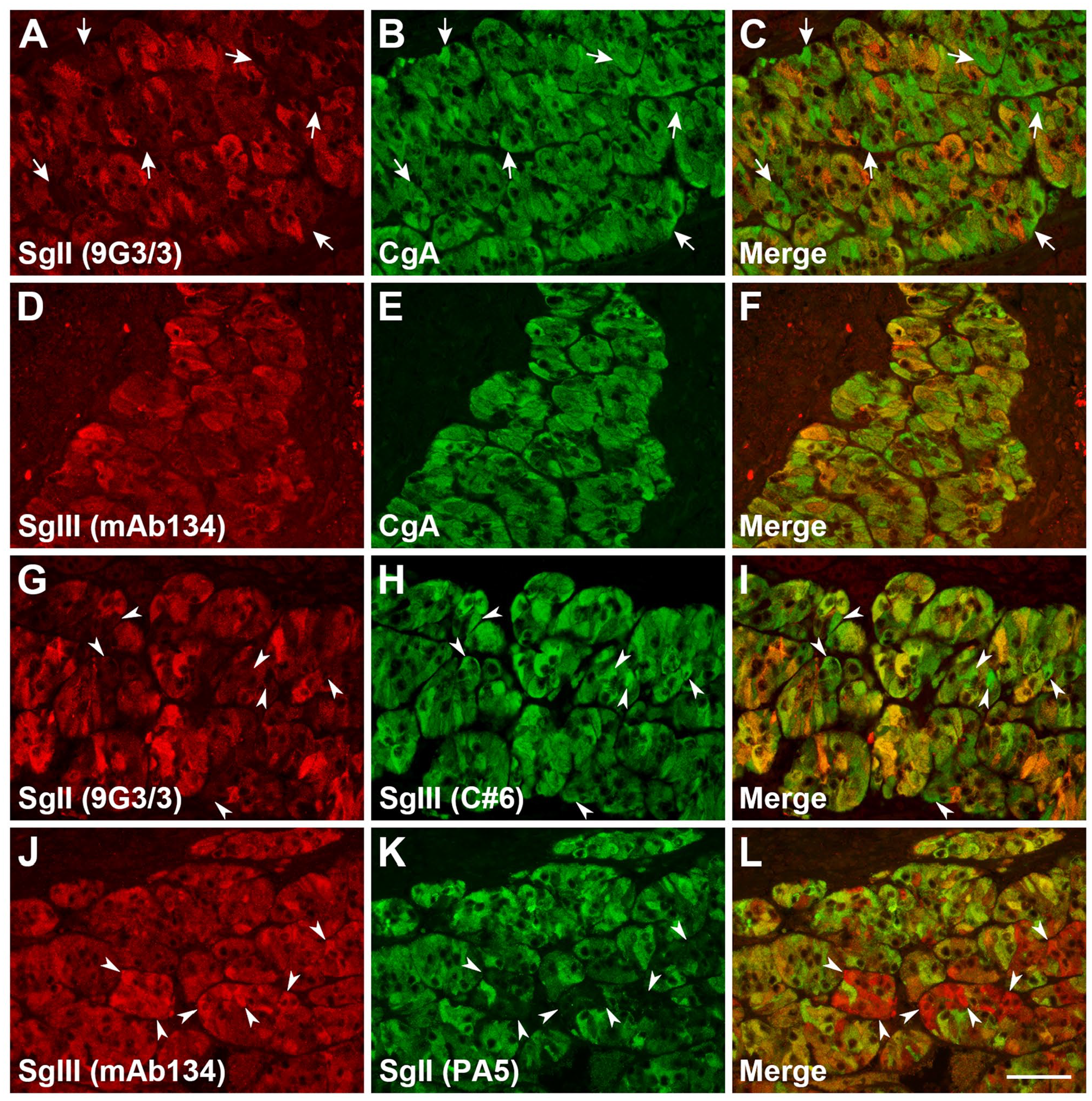
Double immunofluorescence staining for granins in canine adrenal glands. Coexpression of SgII and CgA, (A–C), SgIII and CgA (D–F), and SgII and SgIII (G–L) in the adrenal medulla. For SgII and SgIII immunofluorescence, anti-SgII (9G3/3) and anti-SgIII (mAb134) monoclonal antibodies or anti-SgII (PA5) and anti-SgIII (C#6) polyclonal antibodies were utilized. White arrows indicate the CgA expression dominant but SgII expression negative or very weak cells. White arrowheads indicate SgIII-expressing dominant but SgII-expressing negative or very weak cells. Scale, 50 µm. Abbreviations: SgII, secretogranin II; CgA, chromogranin A; SgIII, secretogranin III.
Coexpression of VMAT2 and Granins in Canine Adrenal Glands
Two VMAT isoforms exist in adrenal chromaffin cells, VMAT1 and VMAT2, and both have different cellular distributions and substrate affinity to monoamines.39,49–51 Therefore, the expression of VMAT2 and its coexpression with granins in the adrenal glands were analyzed, with a cross-reactive antibody against canine VMAT2. The coexpression pattern of VMAT2 and granins demonstrated that it can be subdivided into three subpopulations of medullary cells: (1) consistent expression of both VMAT2 and CgA or SgIII, (2) very weak or negative expression of VMAT2 with stronger expression of CgA or SgIII (Fig. 5A–F), or (3) dominant expression of VMAT2 with very weak or negative expression of SgII (Fig. 5G–I). In VMAT2/CgA- and VMAT2/SgIII-coexpressing cells, a similar expression profile was observed, and cell subpopulations could be classified. In the whole cell population, the cells with stronger expression of VMAT2 were coexpressed CgA or SgIII but not SgII. Thus, the expression patterns of SgII and SgIII were found to be different in adrenal medullary cells in which VMAT2 was predominantly expressed. Negative control sections that were treated with a mixture of normal mouse and rabbit IgG are shown (Supplemental Fig. S3D–F).
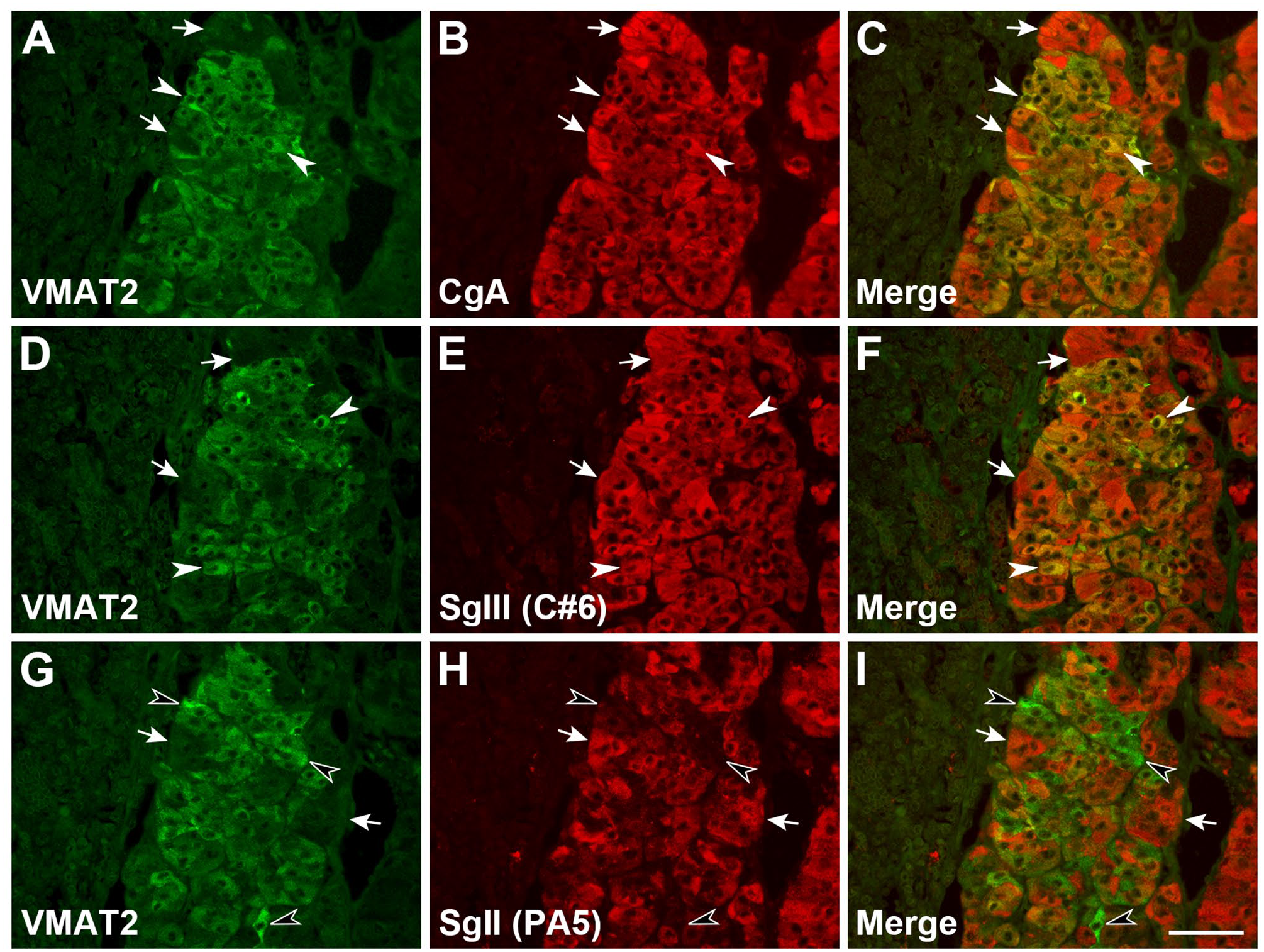
Coexpression of VMAT2 and granins in canine adrenal glands. Coexpression patterns of VMAT2 with CgA (A–C), SgIII (D–F), or SgII (G–I) were analyzed using double immunofluorescence staining. White arrows indicate cells that were shown by each granin expression-dominant but with hardly detectable VMAT2 expression. White arrowheads indicate cells that were dominated by both VMAT2 and granin (CgA and SgIII) expression. Black arrowheads indicate VMAT2 expression-dominant cells with hardly detectable SgII expression. Scale, 50 µm. Abbreviations: VMAT2, vesicular monoamine transporter 2; CgA, chromogranin A; SgIII, secretogranin III; SgII, secretogranin II.
Granin Expression in Canine Adrenal Pheochromocytoma
The histopathological samples of five patient dogs, consisting of three cases with clinical HCL and two cases with LCL, were analyzed. Through general histopathological studies using HE staining, typical angiogenesis and cell proliferation were observed in pheochromocytomas (Fig. 6A–E). Although the degree of angiogenesis seemed to be equal in tissues from both HCL and LCL cases, the cell density tended to be higher in tissues from HCL cases. In the immunohistochemical staining of PCNA, the mitotic figures were remarkably observed in tissues from all cases, and an extremely enlarged nuclear shape was observed in 25874SH and 27371MW cases (Fig. 6F–J). This finding was in contrast to the normal adrenal medulla, where few PCNA-positive cells were observed (Fig. 2F). The intensity of synaptophysin expression was similar among pheochromocytomas (Fig. 6K–O) and comparable to that in normal tissues (Fig. 2E). However, the expression intensity of CgA was different among the pheochromocytomas (Fig. 7A–E). Therefore, the expression properties of these two neuroendocrine tumor markers were different. The expression profile of SgII varied in each case and seemed to be irrespective of the levels of clinical plasma catecholamines (Fig. 7F–J). Although the intensity of SgII expression ranged from moderate to faint, some immunoreactive cells had strong expression in two cases in the HCL group (25874SH and 27929YC) and one case in the LCL group (27371MW) (Fig. 7F, H, and I). On the other hand, the intensity of SgIII expression also varied in each case and showed a similar tendency to that of CgA expression (Fig. 7K–N), except for the single case in the LCL (31072MH) case, where immunoreactive signals were observed in the marginal area of cells or intercellular spaces (Fig. 7O).
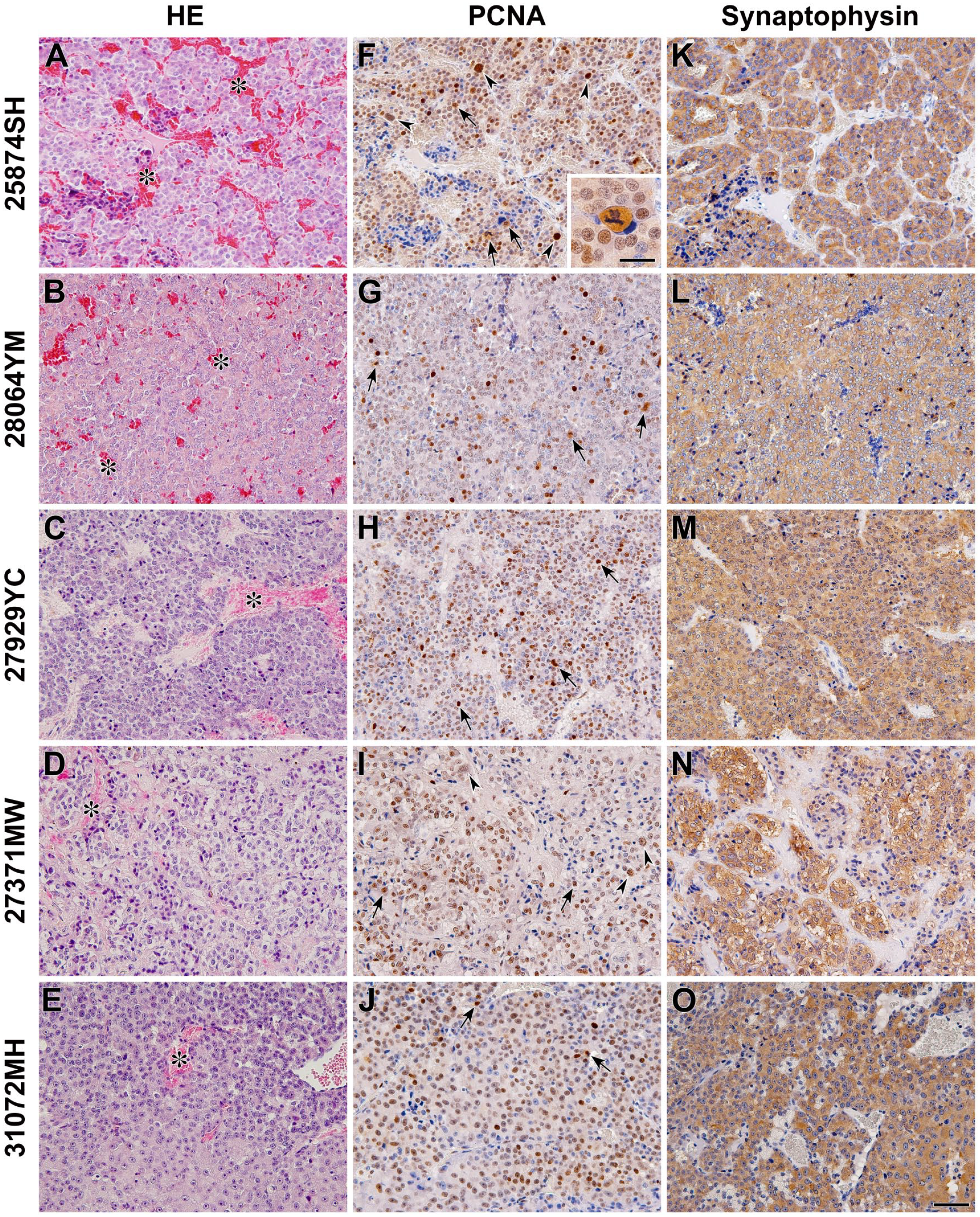
Histological and immunohistochemical analysis of the neuroendocrine tumor markers in canine adrenal pheochromocytomas. Sections were stained with HE (A–E) and immunostained using anti-PCNA (F–J) and anti-synaptophysin (K–O) antibodies. The asterisks indicate the angiogenic vessels. The arrowheads indicate enlarged nuclei, and the arrows indicate the mitotic figures in the panels of PCNA-immunohistochemical staining. The inset panel in PCNA-immunohistochemical staining of 25874SH is an enlarged image of the mitotic figure. Sections were counterstained with hematoxylin. The ID numbers of patient dogs were 25874SH, 28064YM, and 27929YC in the high plasma catecholamine levels (HCL) case and 27371MW and 31072MH in the low plasma catecholamine levels (LCL) group. Scales, 50 and 20 µm (inset). Abbreviation: PCNA, proliferating cell nuclear antigen.
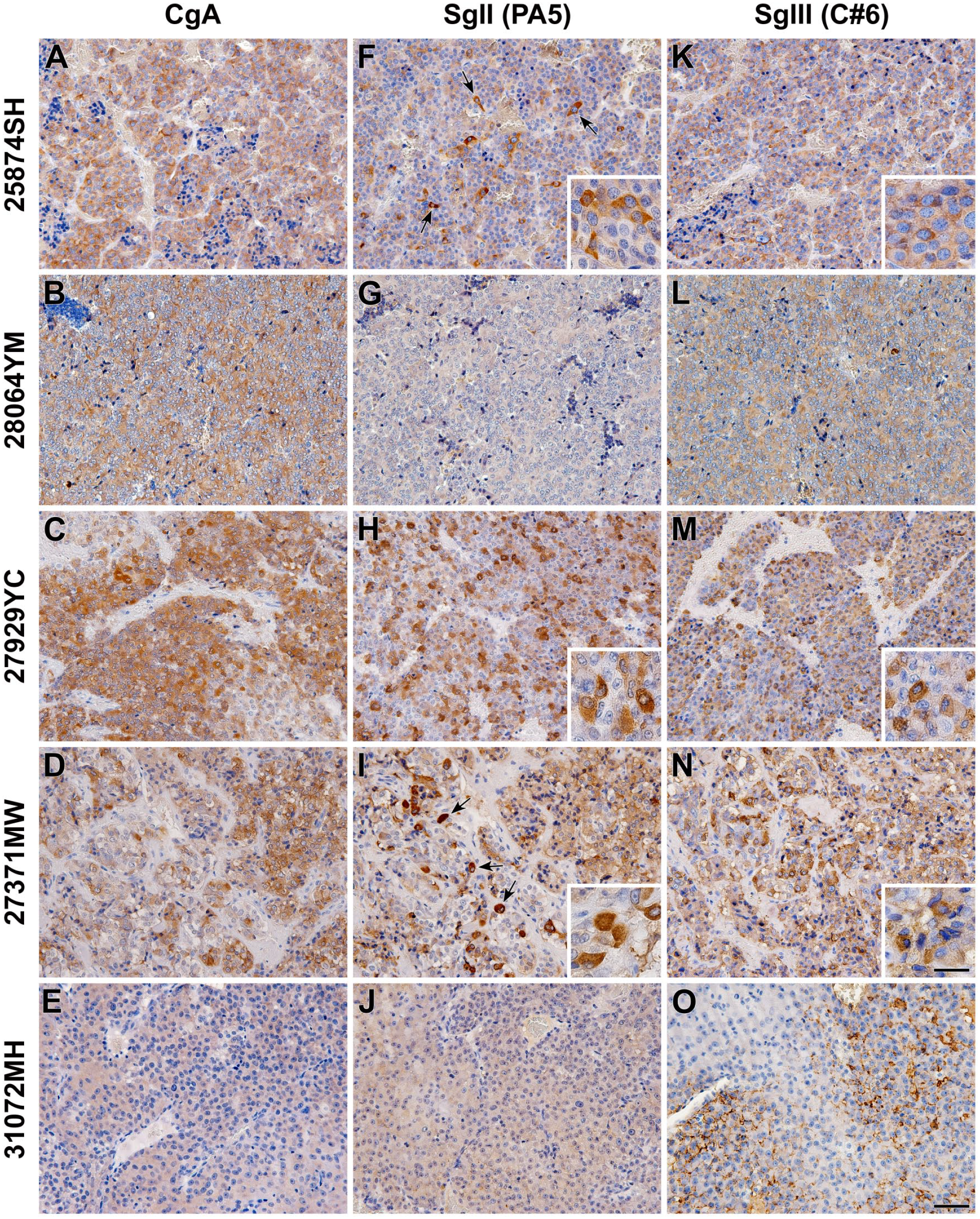
Expression of granins in canine adrenal pheochromocytomas. Sections were immunostained using anti-CgA (A–E), anti-SgII (PA5) (F–J), and anti-SgIII (C#6) (K–O) antibodies. Arrows indicate either spindle- or large round-shaped cells showing SgII immunoreactivity at 25874SH and 27371MW. The inset panels in SgII- and SgIII-immunohistochemical staining are enlarged images. Sections were counterstained with hematoxylin. The ID numbers of patient dogs were 25874SH, 28064YM, and 27929YC in the high plasma catecholamine levels (HCL) case and 27371MW and 31072MH in the low plasma catecholamine levels (LCL) group. Scales, 50 and 20 µm (inset). Abbreviations: CgA, chromogranin A; SgII, secretogranin II; SgIII, secretogranin III.
The morphometric profiles of the expression of PCNA, CgA, and Sgs in pheochromocytomas, that were clinically grouped into HCL and LCL, are shown in Fig. 8. The proliferative potential of tumor cells, as evaluated by immunohistochemical staining for PCNA, demonstrated a higher cell density in the HCL group (74.8% positive) than in the LCL group (70.3% positive) (Fig. 8A). The percentage of positive areas in which CgA was expressed was 48.1% in the HCL group and 26.9% in the LCL group of the total field, and the percentage of positive areas with strong expression was higher in the HCL group (2.7%) than in the LCL group (0.4%) (Fig. 8B). In the case of SgII or SgIII, the percentage of positive areas was expressed as approximately 20.7–22.7% of the total field in the HCL or LCL groups, and the percentage of positive areas with strong expression of SgII (1.5%) was higher than that of SgIII (0.3%) in the HCL group (Fig. 8C). The proliferative tendency, based on PCNA expression, was higher in the HCL group than in the LCL group, and the high expression tendency seen in the HCL group was observed for SgIII, but not for SgII. In addition, the percentage of strongly expressed areas in SgII and SgIII differed between the LCL and HCL groups. Therefore, changes in the expression of SgII and SgIII in pheochromocytoma do not necessarily show the same behavior. The expression profile of CgA showed that its overall expression intensity was consistent with that of SgIII. However, the percentage of strongly expressed regions in CgA and SgIII differed between the LCL and HCL groups. It is speculated that this may reflect the cooperative relationship between SgIII and CgA, but this relationship can be altered in tumor cells. Negative control staining of pheochromocytoma sections is shown (Supplemental Fig. S4).
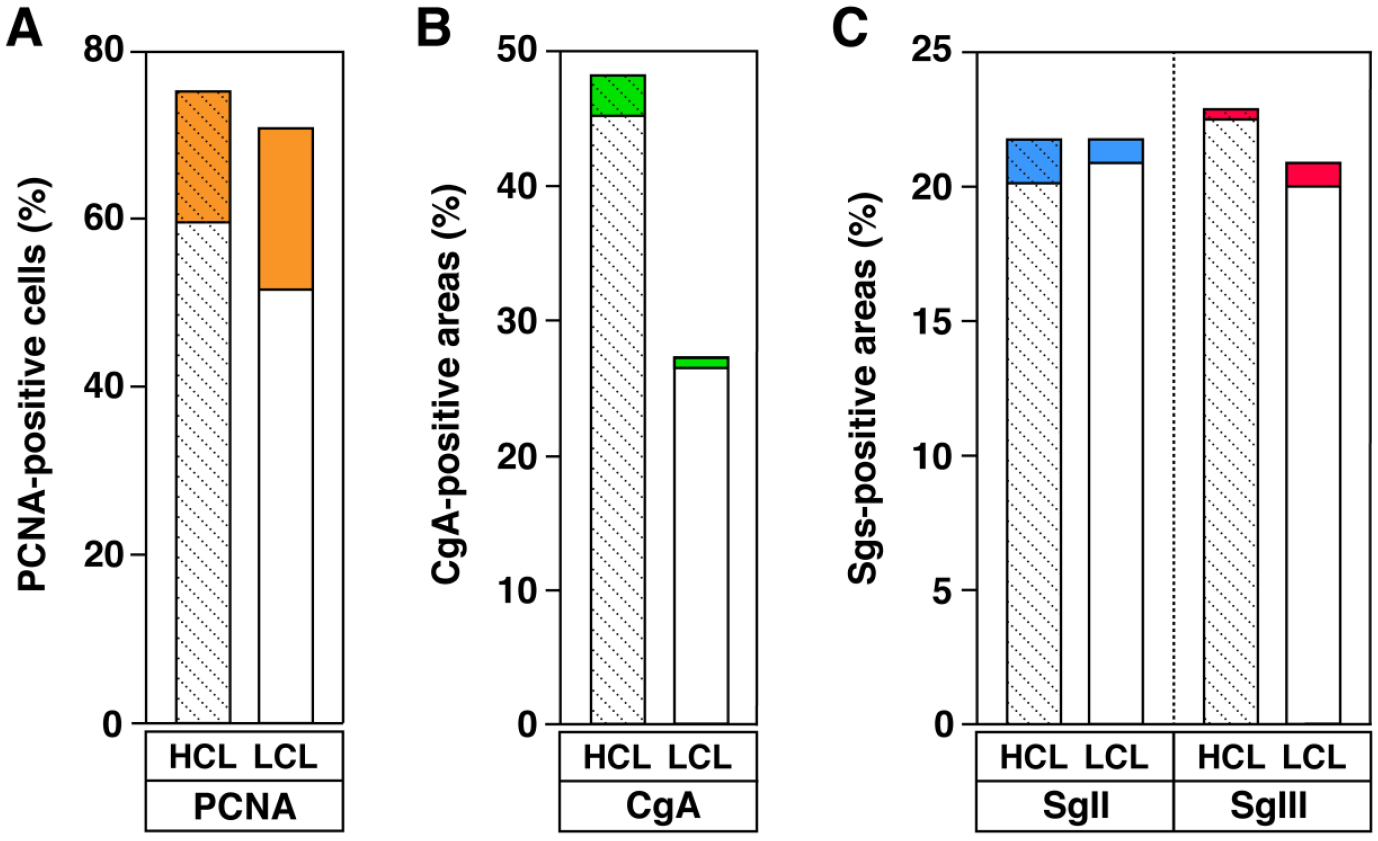
Morphometric profiles of the expression of PCNA, CgA, and Sgs in HCL and LCL pheochromocytoma cases. (A) The percentage of PCNA-immunoreactive cells. The brown areas represent a ratio of the cells that were shown by strong expression in the whole positive cells. (B) The percentage of CgA-immunoreactive cell area. (C) The percentage of SgII- or SgIII-immunoreactive cell area. The green, blue, or red areas represent the ratio of the immunoreactive area with strong expression. Data are expressed as the averages from three HCL cases or two LCL cases. Abbreviations: PCNA, proliferating cell nuclear antigen; CgA, chromogranin A; Sgs, secretogranins; SgII, secretogranin II; SgIII, HCL, high plasma catecholamine levels; LCL, low plasmama catecholamine levels, LCL; secretogranin III.
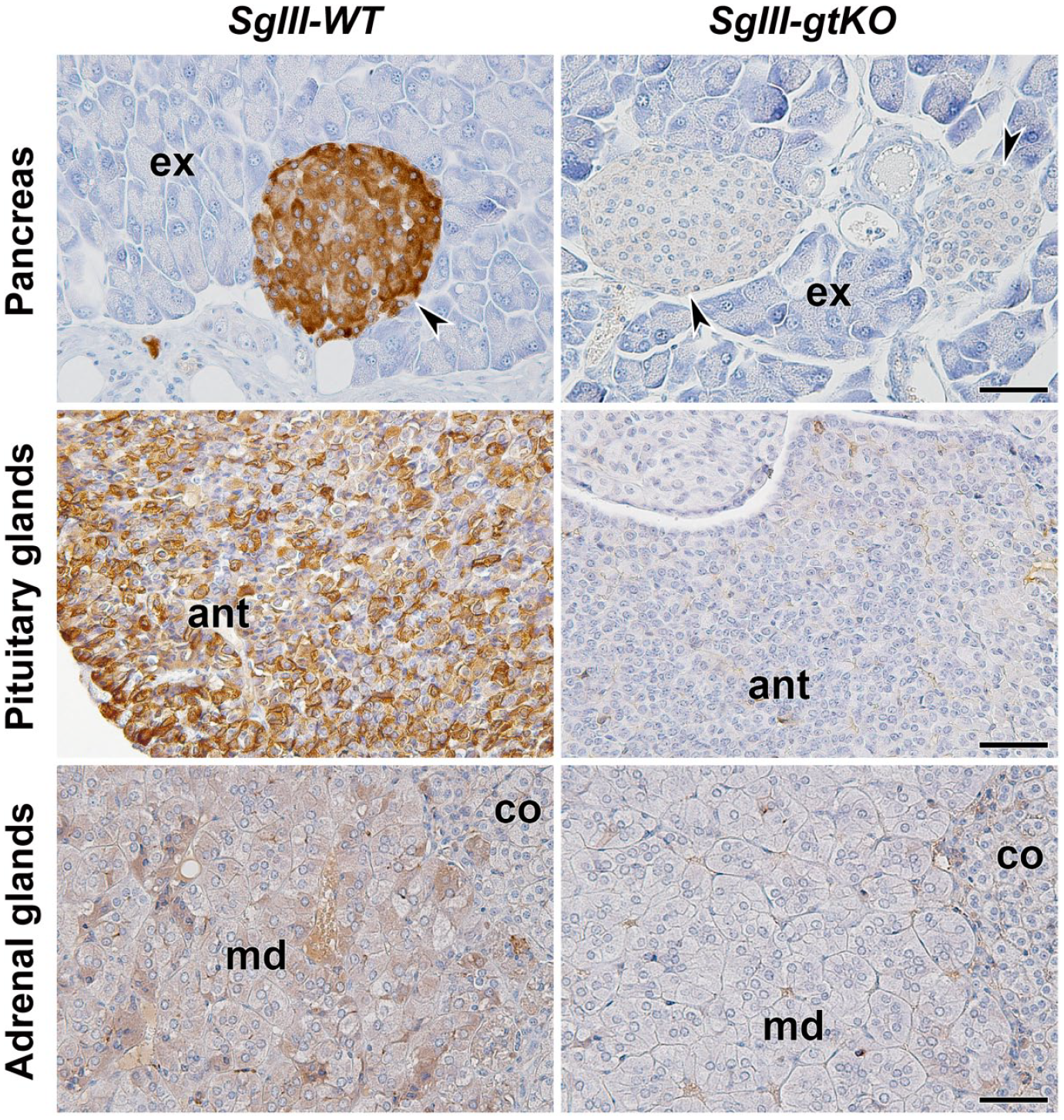
Characterization of a novel anti-SgIII monoclonal antibody, mAb134. Immunohistochemical staining of mouse endocrine tissues. Sections of the pancreas, anterior pituitary glands, and adrenal glands from SgIII-WT and SgIII-gtKO mice were reacted with mAb134. Sections were counterstained with hematoxylin. The arrowheads indicate the pancreatic islets. Scales, 50 μm. Abbreviations: ex, exocrine pancreas; ant, anterior pituitary; md, adrenal medulla; co, adrenal cortex.
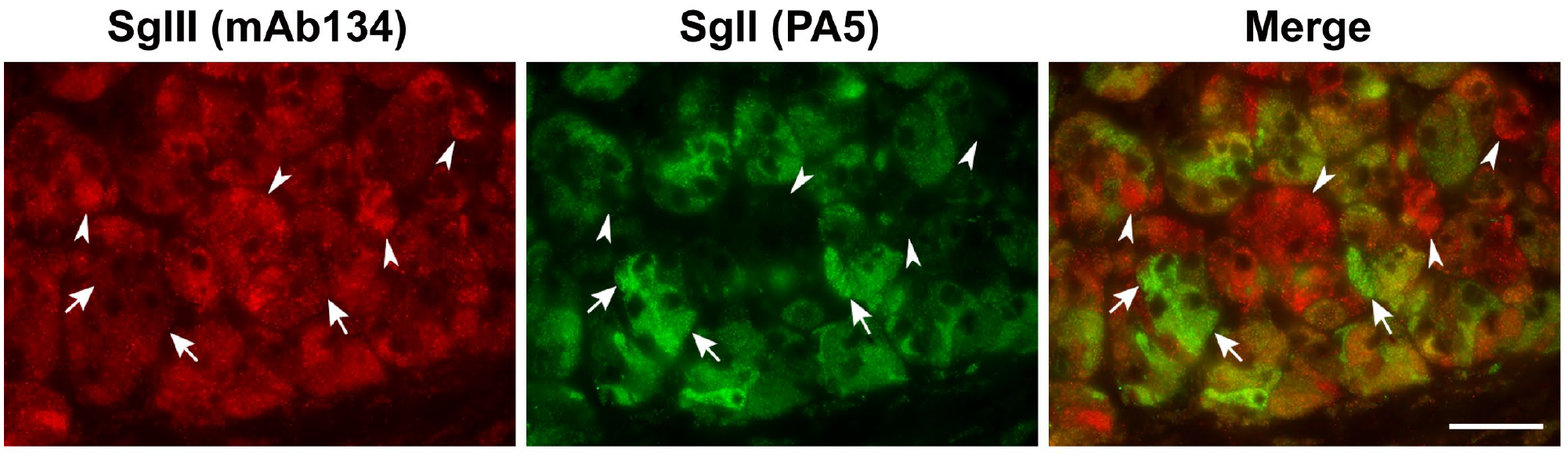
Coexpression pattern of SgII and SgIII in canine adrenal glands. Double immunofluorescence staining using monoclonal anti-SgIII (mAb134) and polyclonal anti-SgII (PA5) antibodies. Three subsets of medullary cells were observed. Many cells coexpressed SgII and SgIII (yellow to orange). White arrows indicate SgII-expressing dominant cells that SgIII-expression are very weak (green). White arrowheads indicate SgIII-expressing dominant cells that SgII-expression are hardly detectable (red). Scales, 75 μm.
Negative control staining of canine adrenal glands in double fluorescence immunohistochemistry. Negative control sections were incubated with a mixture of normal mouse IgG (Mo-IgG, A and D) and normal rabbit IgG (Rb-IgG, B and E) instead of primary antibodies, followed by incubation with Alexa Fluor 488- or 555-conjugated anti-mouse or anti-rabbit secondary antibodies. Merged images of (A) and (B) or (D) and (E) are shown in (C) and (F). Scale, 50 μm. Abbreviations: md, adrenal medulla; co, adrenal cortex.
Negative control staining of canine adrenal glands in enzyme immunohistochemistry. Negative control sections were treated with normal rabbit IgG instead of primary antibodies (Rb-IgG). For the negative detection of SgIII, the sections were treated with the preadsorbed anti-SgIII antibody on the purified GST-SgIII fusion protein (rat amino acids 373–471; 5 μg/mL) (SgIII adsorbed). Sections were counterstained with hematoxylin. The ID numbers of patient dogs were 25874SH, 28064YM, and 27929YC in the HCL group and 27371MW and 31072MH in the LCL group. Scale, 50 μm.
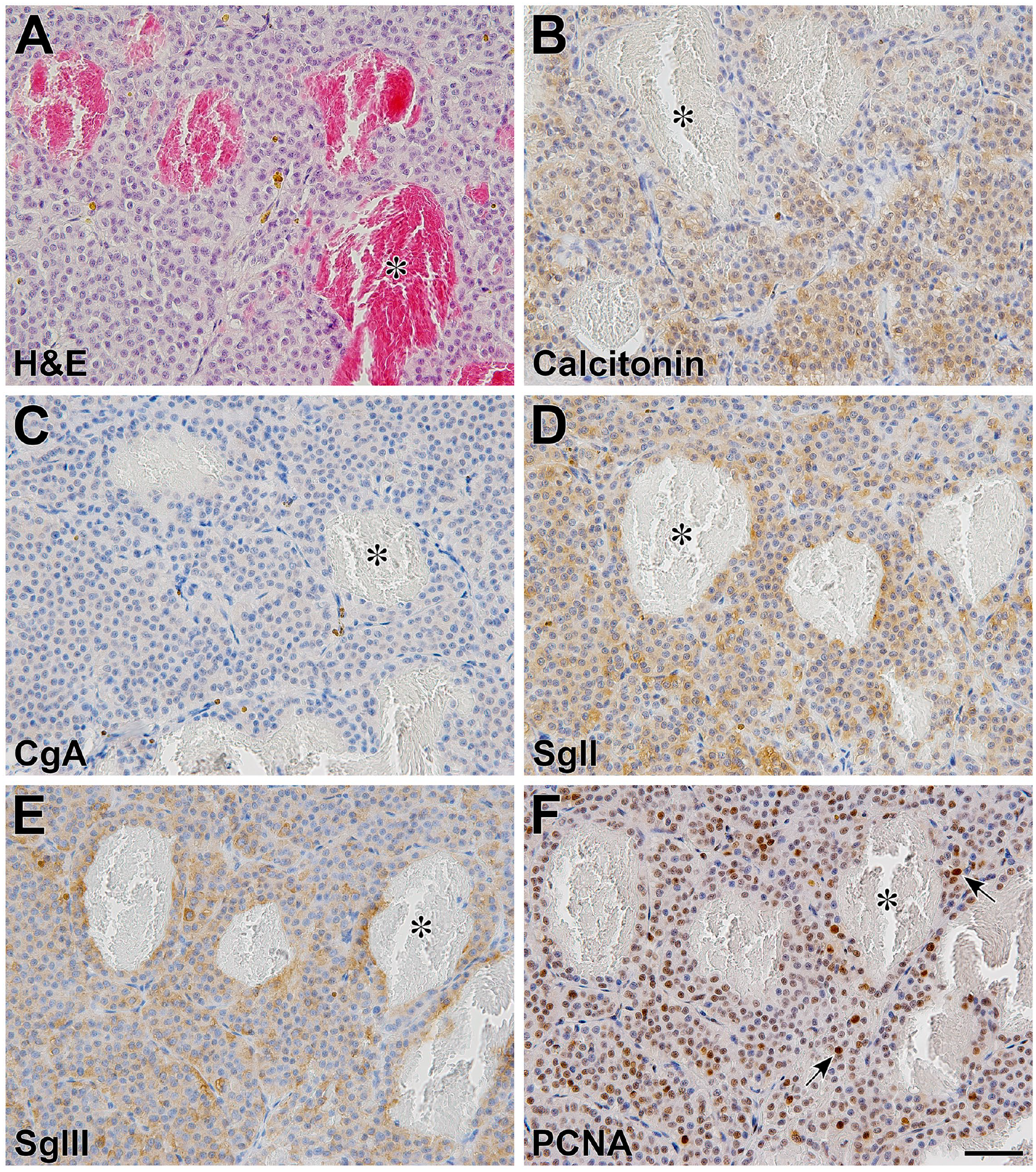
Histological and immunohistochemical analysis of the complicated thyroid medullary carcinoma in patient 31072MH. Histological HE staining of thyroid medullary carcinoma (A). Immunohistochemical staining was conducted using anti-calcitonin (B), anti-CgA (C), anti-SgII (PA5) (D), anti-SgIII (C#6) (E), and anti-PCNA (F) polyclonal antibodies. In this carcinoma, CgA was barely expressed, but calcitonin, SgII, and SgIII were moderately expressed. The asterisks indicate the angiogenic vessels. The arrows indicate PCNA-positive nuclei of proliferating carcinoma cells. Immunohistochemical sections were counterstained with hematoxylin. Scale, 50 μm.
Discussion
The cellular functions of SgII and SgIII in the formation of secretory granules in peptide hormone-producing cells have been widely observed. Both Sgs have common and unique moieties in their molecular structures, which are believed to be responsible for their functional complementarities and differences.44,52–55 On the other hand, the function of these Sgs in active amine-producing cells is not currently fully understood. While SgII expression in the adrenal medulla has been described in previous studies,18–24 the expression of SgIII has only recently been detailed.35,36 In this study, immunohistochemical staining was applied to the canine adrenal medulla to clarify the expression differences between SgII and SgIII. In normal tissues, there are differential expression intensities of SgII and SgIII among chromaffin cells. Coexpression with CgA demonstrated that these two Sgs had a similar expression profile, although the ratio of CgA-expressing cells that coexpressed SgIII was higher than that of cells that coexpressed SgII. Furthermore, the coexpression profile of SgII and SgIII demonstrated that the cells expressing both proteins were not completely matched. In particular, there were distinct cells that had positive SgIII expression but negative SgII expression. These results are the first to show the differential expression of SgII and SgIII in adrenal chromaffin cell subpopulations, and they may be related to the existence of a division of roles in these Sgs.
Interestingly, it has been reported that SgII and SgIII are differentially regulated by the cAMP signaling pathway in astrocytes. 48 Long-term treatment of cultured astrocytes with 8Br-cAMP increased both the synthesis and secretion of SgII, with a decrease in SgIII secretion with no apparent change in synthesis. It is speculated that this difference is due to the presence of a functional cAMP response element in the promoter of the SgII gene. 2 Furthermore, SgII has been known to produce functionally active neurotrophic or angiogenic peptides such as secretoneurin,11,12,15–17,56 EM66,57–59 and manserin60,61 through the processing of its precursor protein. 62 However, the SgIII precursor protein does not produce active peptides, 3 despite undergoing similar proteolytic processing of the precursor protein. 63 Recently, an interesting finding has been made that SgIII acts as an angiogenic vascular leakage factor on retinal vessels in diabetic retinopathy, but it is unclear whether the active peptide produced by processing of the SgIII precursor is involved in this disease-specific phenomenon.13,14
VMATs have been identified in synaptic vesicles of monoamine nerve endings and in the dense-core vesicles of chromaffin cells of the adrenal gland.50,64–66 In mammals, there are two subtypes of VMATs, VMAT1 and VMAT2, and they transport and store synthesized monoamines in vesicles in preparation for exocytotic release.38,49,67,68 VMAT1 is predominantly localized in the membranes of core vesicles of various neuroendocrine cells, whereas VMAT2 is mainly localized in the membranes of synaptic vesicles of monoaminergic nerve endings in the central nervous system and sympathetic nervous system and is also found in the dense-core vesicles of chromaffin cells in the adrenal medulla. 66 In rat adrenal chromaffin cells, VMAT2 colocalizes with tyrosine hydroxylase but not with phenylethanolamine N-methyltransferase (PNMT), suggesting that its expression is in noradrenaline-producing cells. 69 For the functional difference of VMAT1 and VMAT2 in adrenal chromaffin cells, VMAT2 expression was induced by restraint stress in PNMT-positive adrenaline-producing cells, which do not normally express VMAT2 in a non-stressed basal condition. This observation suggests that there is a division of roles between the subpopulations of adrenaline- and noradrenaline-producing cells. 69 Focusing on the involvement of VMATs, we analyzed the coexpression of granins and VMAT2 in adrenal chromaffin cells in this study, although there was a limitation to the expression analysis of VMAT1 because the antibodies that cross-reacted with canine tissues were not available. Differential expression of SgII and SgIII in adrenal chromaffin cells was associated with VMAT2 expression. The coexpression pattern of SgIII and VMAT2 was similar to that of CgA and VMAT2 and showed that the VMAT2 expression-positive cells had positive SgIII expression. However, in the coexpression pattern of SgII and VMAT2, there were distinct cells that had positive VMAT2 expression but negative or hardly detectable SgII expression. These data may suggest that the changes in the expression levels of these Sgs are associated with the cellular properties of adrenal chromaffin cells. In addition, the involvement of VMAT1 also needs to be assessed in future studies because VMAT1 is predominantly localized in the membranes of dense-core vesicles in chromaffin cells. Furthermore, in our study, the beagle dogs were euthanized by intravenous injection of pentobarbital, which is commonly recognized to modulate catecholamine secretion from the adrenals.70,71 Therefore, we did not completely rule out the possibility of secondary effects that were caused by the secretory modulation of catecholamines on the morphological evaluation.
The expression profiles of SgII and SgIII in surgically resected pheochromocytoma tissues were evaluated in this study. Patient dogs were categorized into two groups according to their plasma catecholamine levels, either an HCL or LCL case. Histopathologically, canine pheochromocytomas are characterized by the inclusion of telangiectasia, and islands of tumor cells are interspersed with distended, blood-filled vascular spaces and areas of hemorrhage. 43 The proliferative property of tumor cells was shown as a trend of higher cell density in HCL cases than in LCL cases. For the expression of the neuroendocrine markers, synaptophysin had a relatively strong expression in all pheochromocytomas, whereas different intensities in CgA expression were observed in individual tumors. It was also observed that the expression intensity of SgII and SgIII differed between pheochromocytomas, although there was no strong association with plasma catecholamine levels. However, it was observed that there was a tendency for lower expression of SgIII in HCL cases compared with LCL cases, whereas this was not the case for SgII. Depending on the characteristics of pheochromocytoma, tumor cells with strong expression of SgII were observed to have a scattered distribution. There also seemed to be a tendency for SgII and SgIII to differ in their strong expression patterns at the single-cell level. Thus, SgII and SgIII have their own unique characteristics in their expression in canine pheochromocytomas.
Canine pheochromocytomas are generally regarded as malignant tumors, with a reported metastatic rate of 40% and vascular invasion rate of 82%. 72 The malignancy grade was evaluated based on metastasis to other parts of the body, postoperative recurrence, and bleeding pathology. However, a histopathological staging system or classification based on malignancy has not yet been established for canine pheochromocytoma. All cases analyzed in this study were judged to be highly malignant clinically, regardless of high or low blood catecholamine levels. This histological analysis did not allow us to draw any definite conclusions about the clinical applicability of changes in the intensity of SgII and SgIII expression for the grading and classification of tumor subpopulations. This was because of the variability observed between individual patients and the small number of cases. Furthermore, we discuss the relationship between the characteristic morphological findings of pheochromocytoma (Fig. 7E, J, and O) and complicated medullary thyroid carcinoma in 31072MH patient. In his thyroid medullary tumor tissues, CgA was barely expressed, but SgII and SgIII were moderately expressed, which was different from the expression pattern observed for pheochromocytoma (Supplemental Fig. S5). The morphology of angiogenesis was also different from that in pheochromocytoma. Therefore, we speculated that there may be no direct relationship between these observations. In addition, for human neuroendocrine tumors, it has been shown that the expression pattern of SgIII in pheochromocytoma differs from that in thyroid medullary carcinoma. 41
In human pheochromocytomas, the expression of SgII is associated with malignancy, where malignant tumors have significantly lower SgII expression than benign tumors, as well as reduced production of the SgII-derived peptide fragment EM66.73,74 The processing profile of SgII differs between benign and malignant tumors. 75 Immunohistochemical analysis of tumor tissues has shown a prominent population of large spindle-shaped tumor cells that are characterized by elongated projections within malignant tumors.41,76 In canine pheochromocytomas, the appearance of large spindle-shaped tumor cells with positive SgII expression was found in two out of three cases in the HCL group and one out of two cases in the LCL group. To date, there have been limited studies on SgIII expression in neuroendocrine tumors,41,77,78 with only a single report in human pheochromocytomas. 78 Unlike other neuroendocrine tumors, the number of SgIII-expressing cells was low, their expression intensity was also low, and no coexistence between SgIII and CgA was observed in human pheochromocytomas. The observations in canine pheochromocytoma seem to be consistent with the characteristics of human pheochromocytoma, including the discordant expression patterns of CgA and SgIII and the overall reduction in expression intensity compared with normal adrenal tissues. These may be unique features of pheochromocytoma, in which the cooperative relationship between SgIII and its functional partner, CgA, is lost due to tumorigenesis.
In summary, we demonstrated the histological difference in the expression patterns of SgII and SgIII in physiological and pathological states of adrenal active amine-producing cells. Under physiological conditions, the coexpression of SgII and SgIII was not coincident in adrenal chromaffin cells, indicating that VMAT2 may be responsible for the characteristic differences in the expression of SgII or SgIII. Furthermore, in pathological conditions, there was a clear difference in the expression patterns of SgII and SgIII in adrenal pheochromocytomas, but there was no correlation between expression intensity and blood catecholamine levels in patient animals. These findings add to our understanding of the potential differences in secretogranin family proteins in adrenal active amine-producing cells.
Footnotes
Author Contributions
HG designed and carried out the histological and biochemical experiments, analyzed the data, and wrote the first draft of the manuscript. TN and KA collected and prepared tissue specimens of canine pheochromocytomas and contributed to revise the manuscript. MK performed the histological screening of the anti-secretogranin III (SgIII) monoclonal antibody. TY collected and prepared tissue specimens of normal canine adrenal glands. ST and MH performed antigen preparation and biochemical analysis of the anti-SgIII monoclonal antibody and contributed to the experimental design and data analysis. All authors contributed to manuscript revision and read and approved the submitted version.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid from the Japan Society for the Promotion of Science (JSPS) #16K08078 and #20K06418 (to H.G.). This study was also supported by Nihon University College of Bioresource Sciences Research Grant for 2019–2021 (to H.G. and T.Y.).
