Abstract
Characterization of secretogranin II (SgII) mRNA in various vertebrates has revealed selective conservation of the amino acid sequences of two regions of the protein, i.e., the bioactive peptide secretoneurin and a flanking novel peptide that we named EM66. To help elucidate the possible role of EM66, we examined the occurrence as well as the cellular and subcellular distribution of EM66 in rat pituitary and adrenal glands by using a polyclonal antibody raised against the recombinant human EM66 peptide. High-performance liquid chromatography (HPLC) analysis of rat pituitary and adrenal extracts combined with a radioimmunoassay resolved EM66-immunoreactive material exhibiting the same retention time as recombinant EM66. In the rat pituitary, double-labeling immunohistochemical (IHC) studies showed that EM66 immunoreactivity (IR) was present in gonadotrophs, lactotrophs, thyrotrophs, and melanotrophs, whereas corticotrophs were devoid of labeling. EM66-IR was also observed in nerve endings in the neural lobe. Immunocytochemical staining at the electron microscopic level revealed that EM66-IR is sequestered in the secretory granules within gonadotrophs and lactotrophs. In the adrenal medulla, double IHC labeling showed that EM66-IR occurs exclusively in epinephrine-synthesizing cells. At the ultrastructural level, EM66-IR was seen in chromaffin vesicles of adrenomedullary cells. These results demonstrate that post-translational processing of SgII generates a novel peptide that exhibits a cell-specific distribution in the rat pituitary and adrenal glands where it is stored in secretory granules, supporting the notion that EM66 may play a role in the endocrine system.
Keywords
S
As a further step towards the elucidation of the possible role of EM66, the aim of the present study was to investigate the occurrence of EM66 in the rat pituitary and adrenal glands, where high amounts of the precursor protein SgII and its processing products are found (Anouar and Duval 1991; Lloyd et al. 1992; Fischer-Colbrie et al. 1995; Leitner et al. 1996), and to determinate the precise cellular and subcellular localization of EM66 within these tissues.
Materials and Methods
Animals and Tissue Samples
Three-month-old male Wistar rats weighing 250 to 300 g were maintained under controlled conditions of temperature (22C) and an established photoperiod (lights on from 0700 hr to 1900 hr). Rats had free access to laboratory chow (UAR; Epinay-sur-Orge, France) and water. All manipulations were performed according to the recommendations of the French Ethical Committee and under the supervision of authorized investigators. The animals were sacrificed by decapitation between 0830 hr and 0930 hr, and the adrenal glands and pituitaries were quickly removed and frozen at −80C.
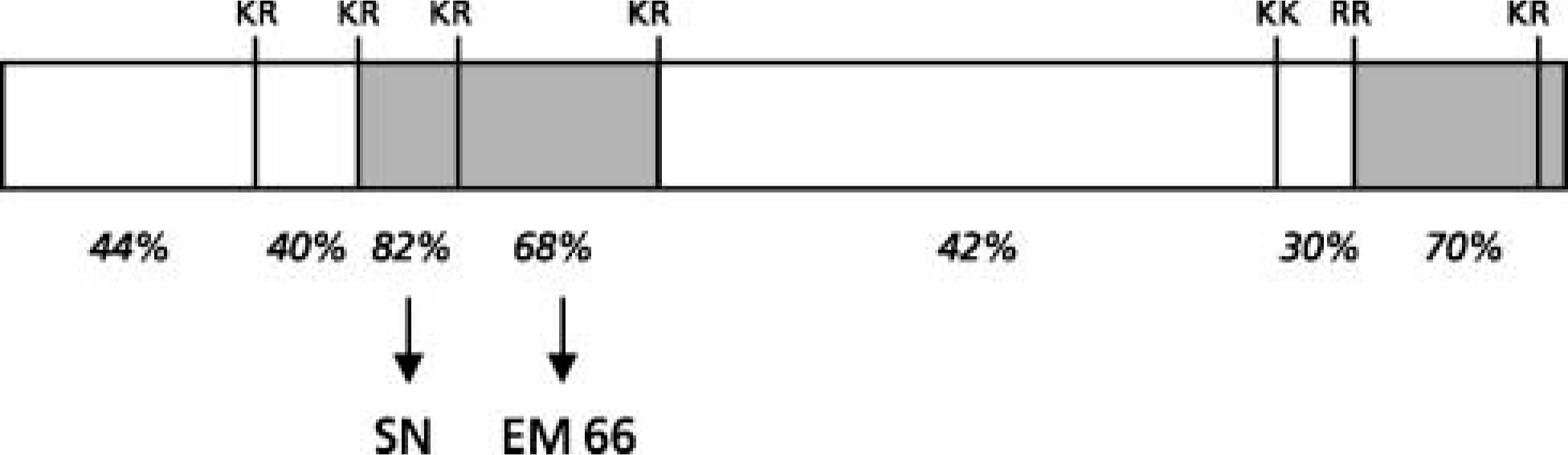
Schematic representation of the structure of SgII showing the location of conserved regions (shaded zones) between the frog and human SgII sequences. Two of these domains correspond to SN and EM66. The conserved dibasic sites and the percentages of amino acid identity between frog and human sequences are indicated.
Production of Recombinant EM66
A fusion protein containing the human EM66 peptide was produced in E. coli as previously described (Anouar et al. 1998). Briefly, the cDNA sequence encoding the EM66 region in human SgII was amplified by PCR, inserted downstream of the maltose-binding protein (MBP) region of the plasmid pMAL-c2 (New England Biolabs; Beverly, MA), and expressed in bacteria. The cleavage of the affinity-purified fusion protein with factor Xa releases EM66 with the native amino acid sequence.
Antibodies
Antibodies against EM66 (code 736–1806) were raised in rabbits against the recombinant fusion protein MBP-EM66, and their specificity has been established by Western blotting and IHC (Anouar et al. 1998). A mouse monoclonal antibody (code 518 B7) against the luteinizing hormone β-subunit (LHβ) was a generous gift from Dr J.F. Roser (Matteri et al. 1987). The adrenocorticotropin (ACTH) antibody (code 105–10) was raised against ACTH 1–24 in sheep and was provided by Dr J. Cote (Jegou et al. 1983). The development of the rabbit antibodies against rat prolactin (PRL) (code 19602), bovine phenylethanolamine N-methyltransferase (PNMT) (code 19886) and human thyroid-stimulating hormone β-subunit (TSHβ) has been previously described (Martinat et al. 1979; Tillet 1988; Uchiyama et al. 2000). The mouse monoclonal antibody directed against tyrosine hydroxylase (TH) was purchased from Chemicon International (Temecula, CA). Fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse (GAM/FITC), FITC-conjugated donkey anti-sheep (DAS/FITC), FITC-conjugated goat anti-rabbit (GAR/FITC), and Texas Red-conjugated donkey anti-rabbit (DAR/TXR) γ-globulins were from Amersham International (Poole, UK).
Immunohistochemical Procedure
For light microscopic IHC the rat adrenal and pituitary glands were dipped overnight in Stefanini's fixative consisting of 4% paraformaldehyde, 0.2% picric acid in 0.1 M phosphate buffer (PB), pH 7.4, rinsed with PB, and frozen at −80C in Tissue-Tek (Leica; Nussloch, Germany). Frozen glands were sliced into 10-μm sections using a cryomicrotome (Frigocut; Reicher-Jung, Nussloch, Germany). After several rinses in PB, tissue sections were processed for indirect immunofluorescence microscopy. Slices were incubated overnight at 4C with one or two (in the case of double labeling) primary antibodies, i.e., the EM66 antiserum diluted 1:500, the TH antibody diluted 1:400, the LHβ antibody diluted 1:300, the ACTH antiserum diluted 1:500, the PRL antiserum diluted 1:2000, the TSHβ antiserum diluted 1:100, or the PNMT antiserum diluted 1:500 in PB containing 0.3% Triton X-100 and 1% bovine serum albumin (BSA; Roche Diagnostics, Mannheim, Germany). Tissue sections were rinsed in PB for 30 min and incubated for 90 min at room temperature (RT) with the appropriate secondary antibody, i.e., GAR/FITC, DAS/FITC, GAM/FITC diluted 1:100, or DAR/TXR diluted 1:50. Finally, slices were rinsed and mounted with PB/glycerol (1:1), coverslipped, and examined using a confocal laser scanning microscope (CLSM; Leica, Heidelberg, Germany) equipped with a diaplan optical system and an argon/krypton ion laser (excitation wavelengths 488/568/647 nm). To verify the specificity of the immunoreaction, various controls were performed: (a) substitution of the primary antisera/antibodies with PB; (b) incubation with non-immune serum instead of the primary antisera; (c) liquid-phase preabsorption of the EM66 antiserum (diluted 1:500) with purified recombinant peptide (10−6 M).
Immunoelectron Microscopy
Three-month-old adult male Sprague-Dawley rats (225–250 g) were perfused transcardially with 200 ml of 2.5% glutaraldehyde in 0.1 M PB. Pituitaries and adrenal glands were excised and postfixed in the same fixative for 6 hr at 4C. Then the anterior lobes of the pituitaries and the adrenals were cut into small fragments (∼1 mm3). The tissues were rinsed overnight in the same buffer and postfixed for 1 hr in 0.2% OsO4. After rinsing for 2 hr in distilled water, the tissues were dehydrated in ethanol and embedded in Araldite. Semithin sections were first performed for selection of the areas of interest in each gland. Ultrathin sections from the anterior lobe of the pituitary, and medullary and cortical zones of the adrenal gland, were then cut with a diamond knife and collected on nickel grids. Tissue sections were immunostained using the protein A-gold complex (10 nm; British Biocell, Cardiff, UK), as described (Roth et al. 1978). The EM66 antiserum was used at a 1:200 dilution. Control experiments were performed by substituting non-immunized rabbit serum (1:200) or the antiserum (1:200) preabsorbed with an excess of antigen (10−6 M). After the immunostaining procedures, the sections were counterstained with 1% uranyl acetate and lead citrate.
Tissue Extraction
Frozen rat pituitary and adrenal glands were immersed for 10 min in a cooled solution of 75% ethanol/18% hydrochloric acid (10 N). The tissue samples were sonicated and centrifuged at 10,000 × g for 30 min at 4C. The supernatants were collected and dried by vacuum centrifugation (Speed-Vac Concentrator; Savant AES 2000, Hicksville, NY). The samples were reconstituted in 0.1% TFA in water and loaded onto three Sep-Pak C18 cartridges (Waters Associates; Milford, MA) connected in series. The bound material was eluted with a solution of acetonitrile/water/TFA (49.9:50:0.1, v/v/v). The solvent was evaporated in the Speed-Vac Concentrator and the samples were kept dry until chromatographic analysis.
High-performance Liquid Chromatography (HPLC) Analysis
Dried samples were reconstituted in 1 ml of 0.1% TFA and centrifuged at 10,000 × g for 10 min at 4C. The supernatants were injected onto a 0.45 × 25-cm Vydac C18 column equilibrated with a solution of acetonitrile/water/TFA (9.9:90:0.1, v/v/v) at a flow rate of 1 ml/min. The concentration of acetonitrile in the eluting solvent was raised to 60% over 25 min using a linear gradient. HPLC standard consisted of 1 μg purified recombinant EM66. Fractions of 1 ml were collected, evaporated, and kept dry until RIA.
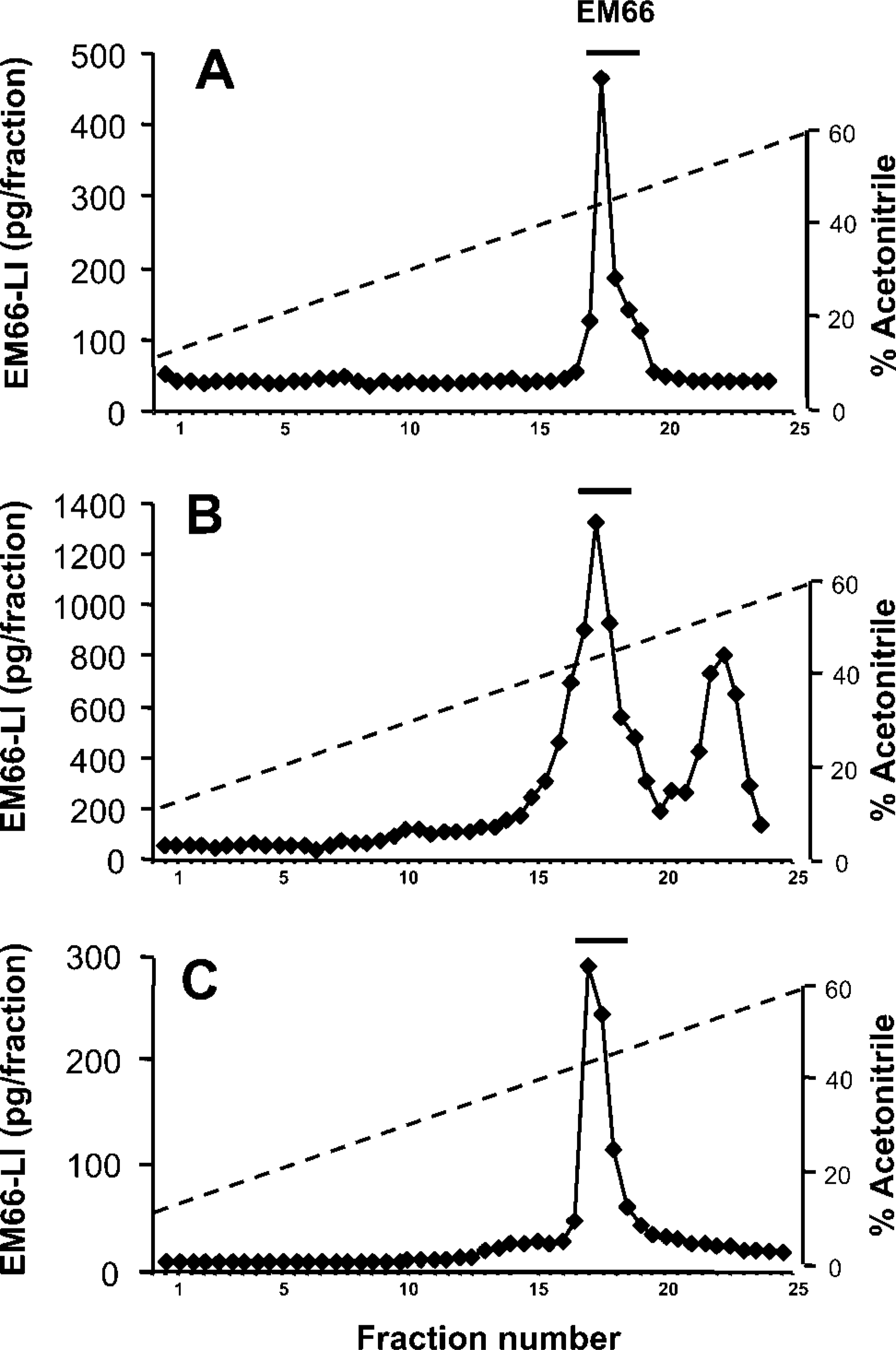
RP-HPLC analysis of EM66-like immunoreactivity (LI) in the rat pituitary and adrenal glands. (A) Elution profile of recombinant EM66 chromatographed onto a Vydac C18 column. Fractions of 1 ml were collected, dried, and radioimmunoassayed for EM66. Protein extracts prepared from 50 pituitary anterior lobes (B) and from 30 adrenal glands (C) were pre-purified on Sep-Pak cartridges, analyzed as described above, and radioimmunoassayed for EM66. The bars above the peaks indicate the elution position of recombinant EM66, as indicated in A.
Radioimmunoassay
The concentrations of EM66-like material in the rat tissues were measured by RIA. Purified recombinant EM66 was iodinated by the chloramine-T method and separated from free iodine on Sep-Pak C18 cartridges using a gradient of acetonitrile (0–100%) in 0.1% TFA. Radioiodinated EM66 eluted at 32% acetonitrile. The RIA was performed in 20 mM veronal buffer, pH 9.1, containing 3% BSA. The EM66 antiserum, used at a final dilution of 1:60,000, was incubated with 7000 cpm of tracer/tube in the presence of recombinant EM66 (standard peptide) or HPLC fractions. After a 2-day incubation at 4C, the antibody-bound fraction was immunoprecipitated by addition of 200 μl goat anti-rabbit γ-globulins (1:30), 200 μl normal rabbit serum (1:150), and 500 μl of 5% polyethyleneglycol 8000. After a 2-hr incubation at RT, the mixture was centrifuged (5000 × g, 4C, 30 min), and the pellet containing the bound fraction was counted on a γ-counter (LKB; Wallack, Rockville, MD).
Results
Characterization of EM66-IR in Pituitary and Adrenal Gland Extracts
Reversed-phase (RP)-HPLC analysis combined with RIA detection was conducted to characterize EM66 in rat pituitary and adrenal extracts (Figure 2). Recombinant EM66 eluted as a single peak with a retention time of 17.5 min (Figure 2A). A major EM66-IR peak exhibiting the same retention time as the recombinant peptide was detected in the adenohypophysial (Figure 2B) and adrenal (Figure 2C) gland extracts. A more hydrophobic EM66-immunoreactive compound eluting at 22.5 min was also resolved in the adenohypophysial gland extract (Figure 2B).
Distribution of EM66-IR in the Pituitary
The EM66 antiserum strongly labeled many cells in the anterior lobe (Figure 3A and 3C) and moderately stained most melanotrophs in the intermediate lobe of the rat pituitary (Figure 3C and 3D). At a higher magnification, EM66-IR was also observed in nerve endings in the neural lobe (Figure 3D). No staining was seen when the EM66 antiserum was preabsorbed with 10−6 M of the recombinant peptide and when the primary antibody was substituted with non-immune rabbit serum or PB, thus confirming the specificity of the immunoreaction (Figure 3B).
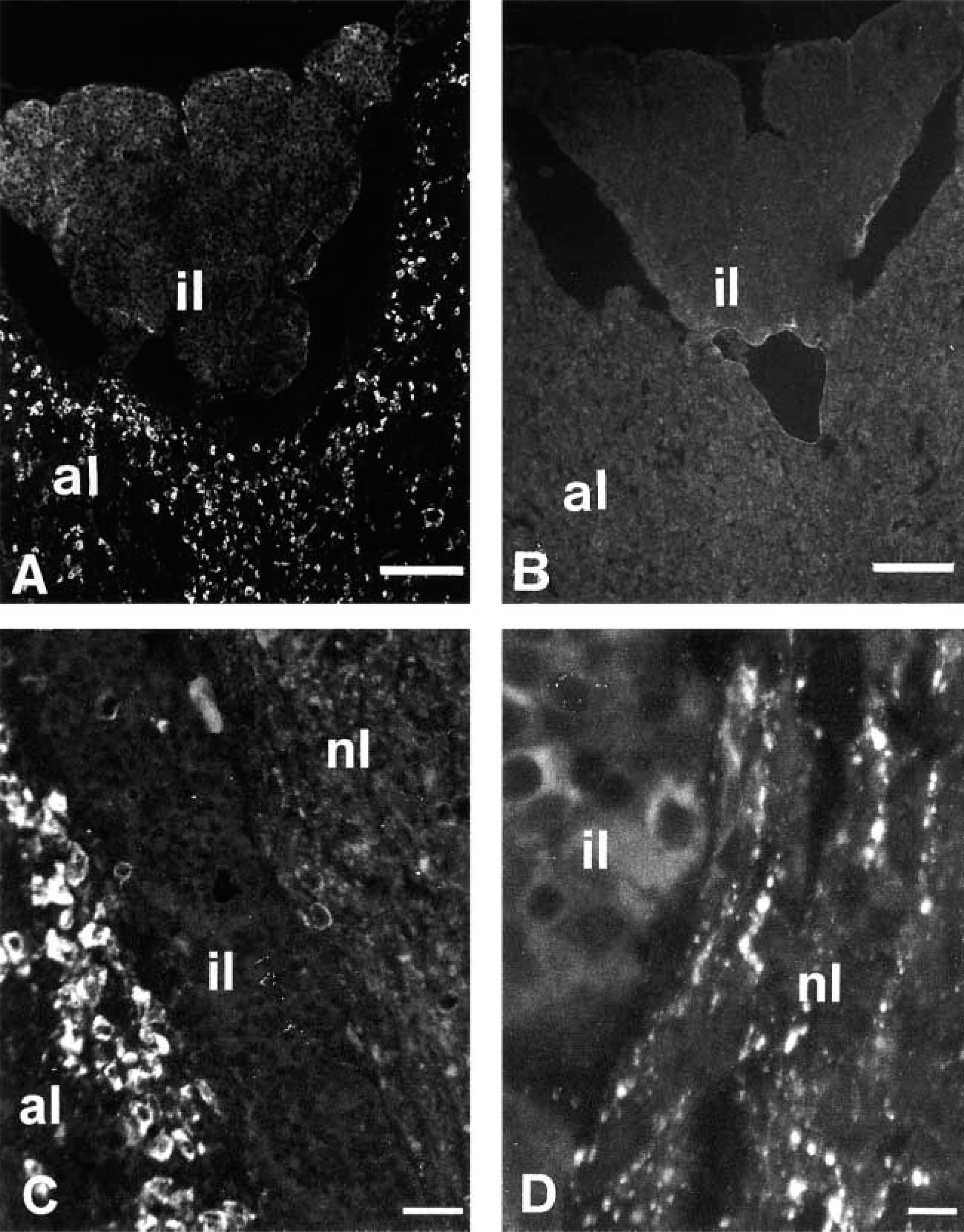
Photomicrographs showing the distribution of EM66-like immunoreactivity in the rat pituitary. Consecutive sections of a rat pituitary gland were incubated with the EM66 antiserum (A) or with the same antiserum preabsorbed overnight with 10−6 M purified EM66 (B). (C) Intense immunofluorescence labeling was observed in many cells of the anterior lobe (al) and moderate labeling of melanotrophs in the intermediate lobe (il) as well as nerve fibers in the neural lobe (nl). (D) Higher magnification showing the distribution of EM66-like immunoreactivity in the intermediate and neural lobes. Bars:
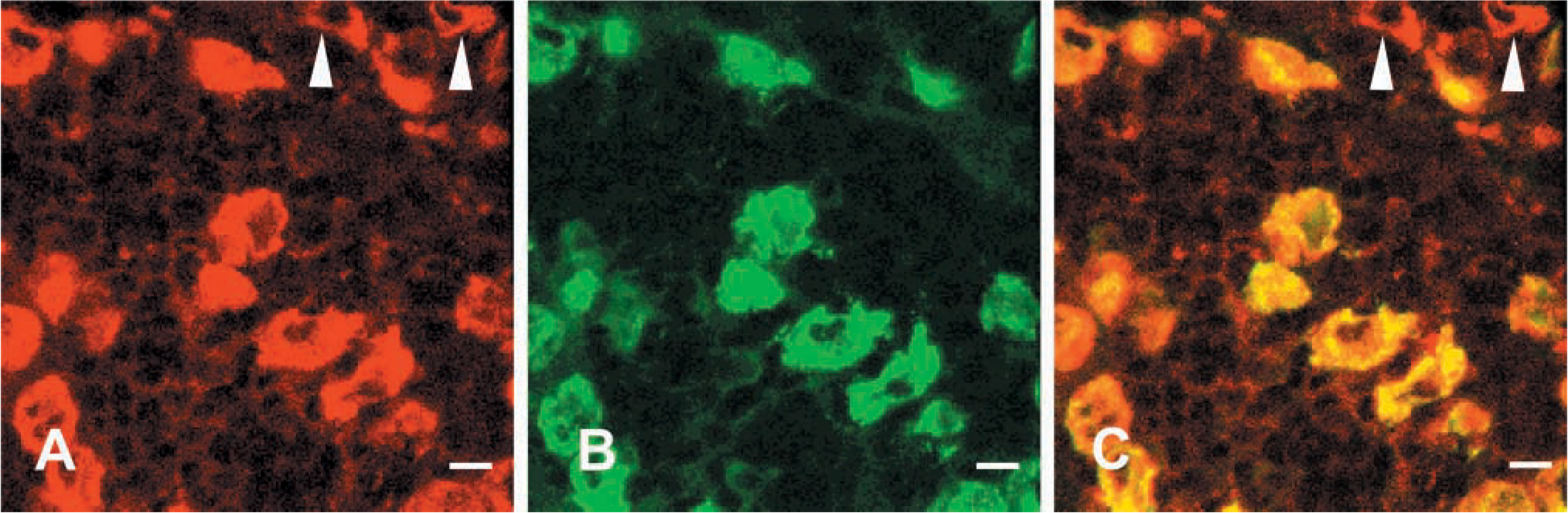
Confocal laser scanning microscope photomicrographs comparing the distribution of EM66 and LHβ immunoreactivity in the rat pituitary. Sections through the anterior lobe were labeled with a rabbit antiserum against EM66 (A) revealed with DAR/TXR and with a monoclonal antibody against the LHβ subunit (B) revealed with GAM/FITC. Combination of the two images acquired in A and B showed the presence of EM66-like immunoreactivity in all gonadotrophs (C). A few cells that were stained by the EM66 antibody were not immunoreactive to LHβ (arrowheads). Bars = 10 μm.
Co-incubation of pituitary sections with the anti-EM66 antiserum and the anti-LHβ antibody revealed that virtually all gonadotrophs contain EM66-IR (Figures 4A-4C). However, some EM66-IR cells did not exhibit any LHβ-IR (Figures 4A-4C), indicating that the peptide occurs in cells other than gonadotrophs. Incubation of consecutive sections of the adenohypophysis with the anti-EM66 (Figure 5A) and anti-PRL (Figure 5B) antisera showed that some EM66-IR cells were lactotrophs (Figure 5A and 5B). Similarly, incubation of consecutive sections with the anti-EM66 (Figure 6A) and the anti-TSHβ (Figure 6B) antibodies revealed that EM66-IR is present in thyrotrophs (Figure 6A and 6B). On the contrary, corticotrophs that were immunostained with the ACTH antibodies (Figure 7B) were totally devoid of EM66-IR (Figures 7A-7C).
The different types of pituitary cells can readily be identified on the basis of their ultrastructural characteristics (Kurosumi 1968; Pelletier 1984). Somatotrophs are characterized by the presence of a large number of round or slightly ovoid secretory granules with a diameter of about 300–400 nm. Lactotrophs contain the largest granules of any pituitary cell. The mature secretory granules are often ovoid or elliptical, with a diameter ranging from 600 to 900 nm. Gonadotrophs are characterized by the presence of two types of granules: the small (200 nm in diameter) and the large ones (350–700 nm in diameter). Corticotrophs can be identified by the presence of a single row of secretory granules (200 nm in diameter) exclusively located along the plasma membrane. Thyrotrophs have the smallest secretory granules (about 140 nm in diameter) of any cell type. EM66-IR material was only observed in secretory granules in gonadotrophs and lactotrophs. In gonadotrophs, both large and small secretory granules were decorated with gold particles (Figure 8A). The labeling of secretory granules of lactotrophs was generally weaker than that observed in gonadotrophs (Figure 8B). No labeling was observed in secretory granules of somatotrophs (Figure 8A). When the antiserum was absorbed with the antigen, no association of gold particles with any cell types in the anterior pituitary could be observed (data not shown).
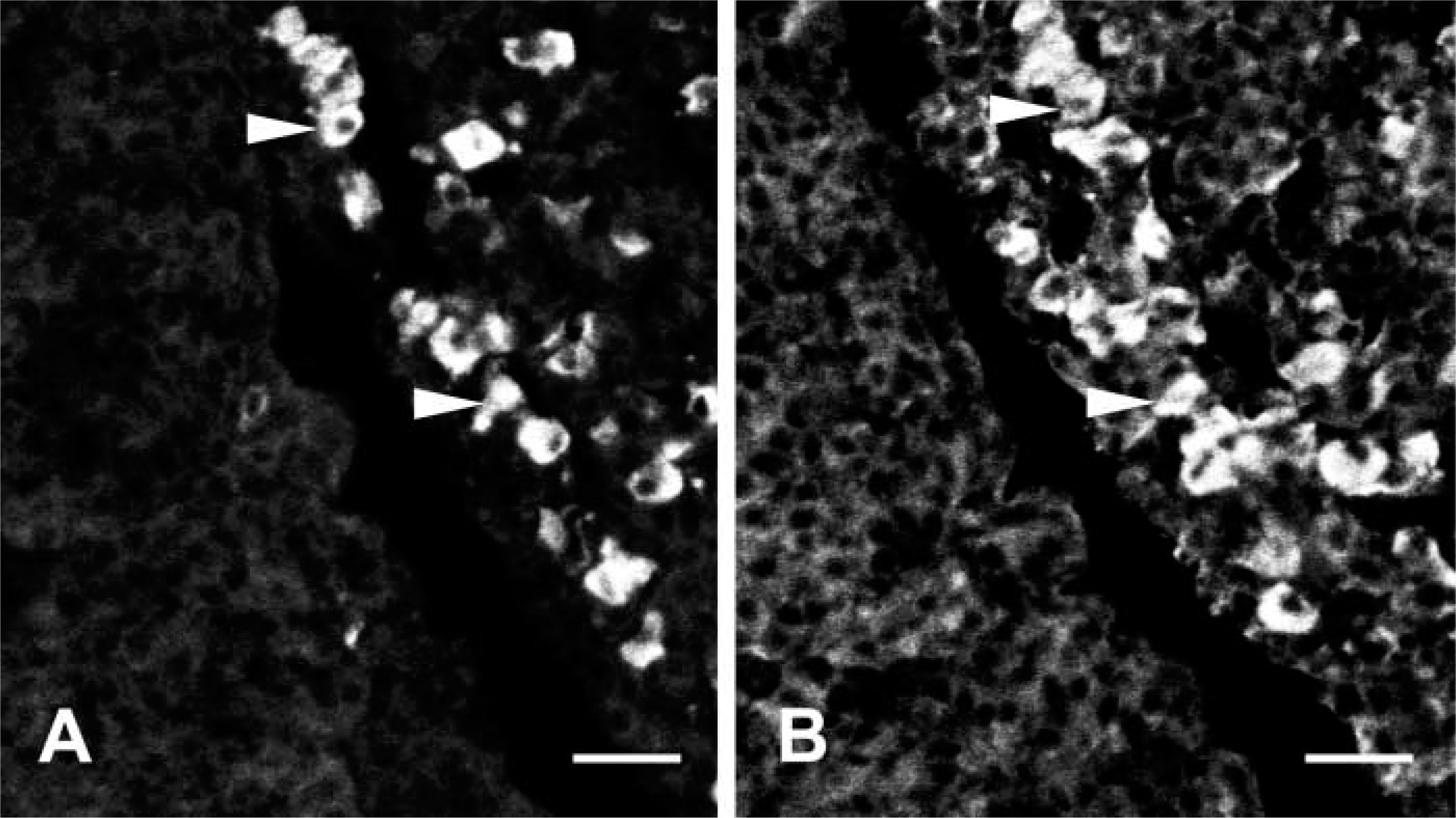
Photomicrographs showing the distribution of EM66- and PRL-like immunoreactivity in the rat pituitary. Consecutive sections through the anterior lobe of the pituitary were immunostained with antisera against EM66 (A) and PRL (B). Several lactotrophs were immunoreactive to EM66 (arrowheads). Bars = 20 μm.
Immunofluorescence labeling of EM66 and TSH in the rat pituitary. Consecutive sections through the anterior lobe of the pituitary were immunostained with antisera against EM66 (A) and TSHβ (B). A thyrotroph that was stained by the EM66 antibody is indicated by an arrowhead. Bars = 20 μm.
Distribution of EM66-IR in the Adrenal Gland
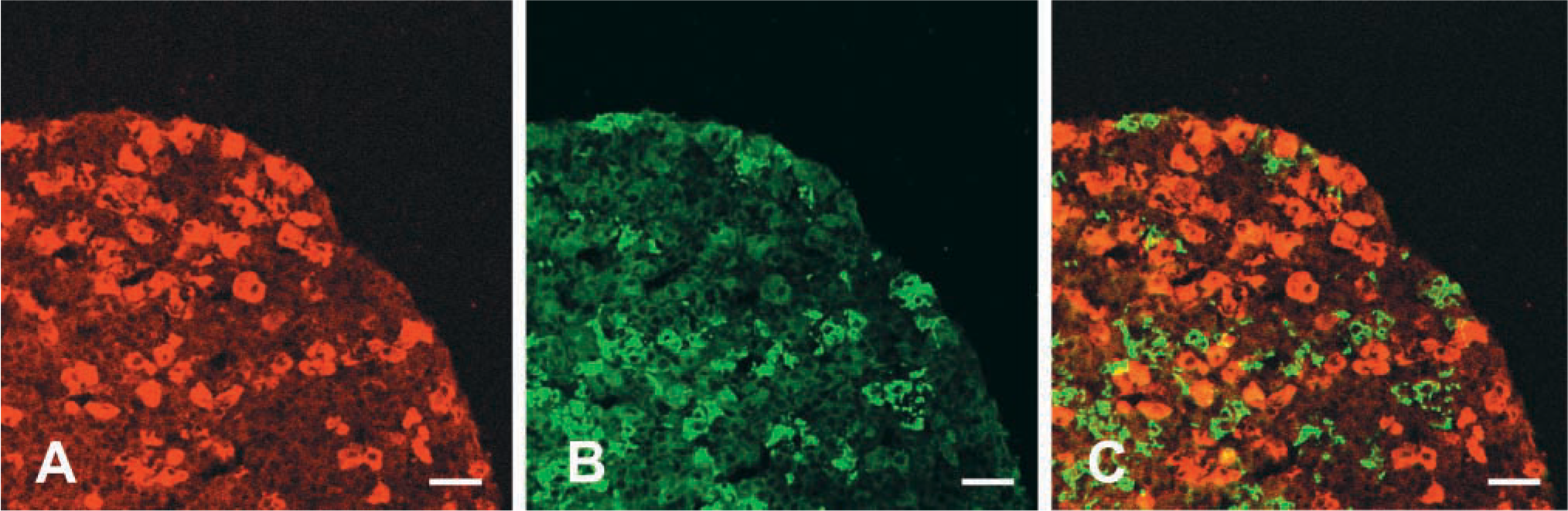
The EM66 antibody strongly labeled the whole medulla (Figure 9A) and groups of cells located in the cortex (Figure 9A and 9B) of the rat adrenal gland. Preabsorption of the primary antiserum with 10−6 M EM66 totally abolished the labeling (data not shown). Co-incubation of adrenal sections with the anti-EM66 and anti-TH antibodies revealed that virtually all adrenomedullary cells that were immunolabeled with the EM66 antiserum were also stained with the TH antibodies (Figures 10A-10C), indicating that EM66 is present in chromaffin cells. However, a few TH-positive cells were devoid of EM66-IR (Figure 10C). Double immunolabeling of adrenal slices with the anti-PNMT and anti-TH antibodies (Figures 10D-10F) revealed that the EM66-negative chromaffin cells were also PNMT-negative (Figure 10C and 10F), indicating that EM66 is present only in adrenergic cells and not in noradrenergic cells of the rat adrenal gland.
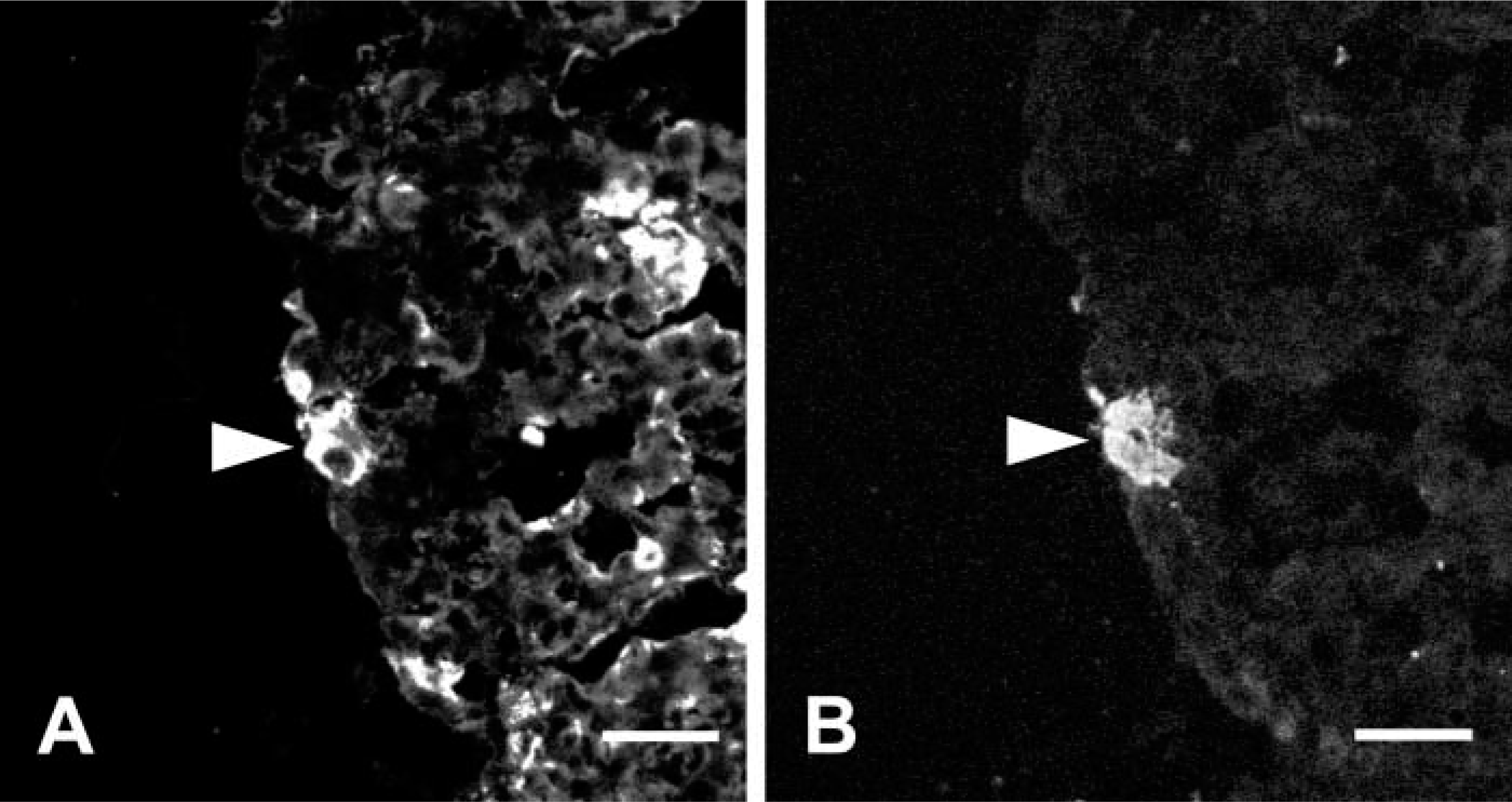
Confocal laser scanning microscope photomicrographs comparing the distribution of EM66 and ACTH-IR in the rat pituitary. Sections through the anterior lobe were labeled with a rabbit antiserum against EM66 (A) revealed with DAR/TXR and with a sheep antiserum against ACTH (B) revealed with DAS/FITC. Combination of the two images acquired in A and B showed the absence of EM66-like immunoreactivity in corticotrophs (C). Bars = 40 μm.
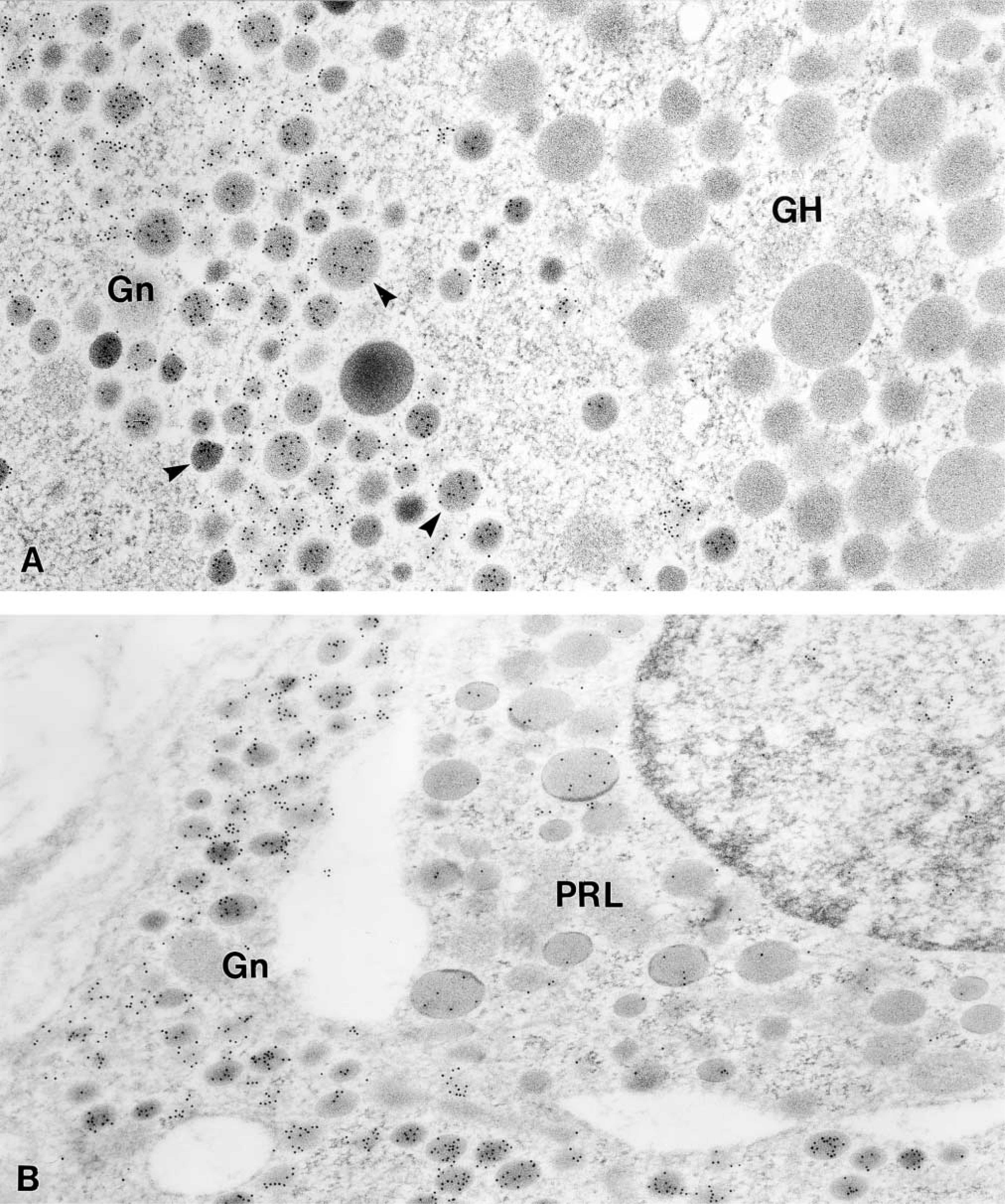
Immunoelectron microscopic localization of EM66 in the anterior pituitary gland. (A) Gold particles are located over the small and large secretory granules (arrowheads) of a gonadotroph (Gn). An adjacent somatotroph (GH) is unlabeled. (B) A few gold particles is also observed over the secretory granules of a prolactin cell (PRL). An adjacent gonadotroph (Gn) is more strongly labeled. Original magnification x 50,000.
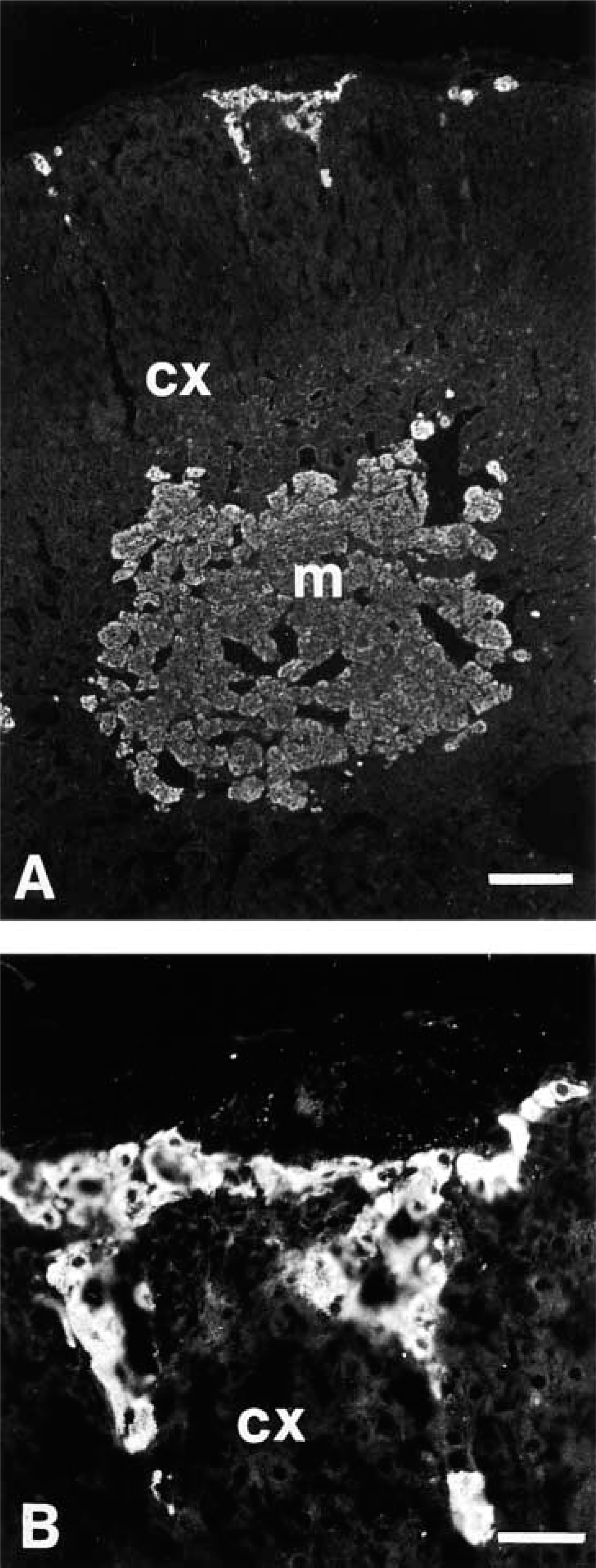
Photomicrographs showing the distribution of EM66-like immunoreactivity in the rat adrenal gland. (
Immunocytochemical labeling of chromaffin cells showed that EM66-IR was restricted to secretory granules (Figure 11A and 11B). In contrast, adrenocortical cells did not exhibit any staining (Figure 11A). When the antiserum was absorbed with the antigen, no association of gold particles with any cell types in the adrenal gland could be observed (data not shown).
Discussion
The present study has demonstrated the occurrence of a novel SgII-derived peptide, EM66, in the rat pituitary and adrenal, and has described its cellular and subcellular distribution in these endocrine glands.
Several lines of evidence indicate that SgII is a precursor protein that gives rise to biologically active peptides. (a) SgII contains pairs of basic amino acids that are known to be privileged targets of PCs in endocrine and neuroendocrine cells (Gerdes et al. 1988; Fischer-Colbrie et al. 1990; Anouar et al. 1996). (b) SgII is cleaved by the convertases PC1 and PC2 (Tooze et al. 1994; Laslop et al. 1998). (c) A post-translational processing product of SgII, the peptide SN, has been identified in the brain of frog and mammals (Vaudry and Conlon 1991; Marksteiner et al. 1993). (d) SN is released from endocrine and nerve cells upon stimulation (Kirchmair et al. 1994; Turquier et al. 2001). We have previously shown that SgII, in addition to SN, can give rise to the peptide EM66 in human adrenal gland (Anouar et al. 1998). Because the sequence of EM66 is highly conserved among species (Anouar et al. 1996), we postulated that the antisera directed against the human peptide may allow more precise characterization of the occurrence and distribution of EM66 in different endocrine tissues of rat. Analysis of tissue extracts by reversed-phase HPLC combined with RIA has revealed that EM66 is formed in the rat pituitary and adrenal, indicating that this SgII-derived peptide can be generated in various tissues and in different species. In the anterior pituitary, a second peak of EM66-IR material was also resolved. Owing to its higher retention time, this peak may correspond to an intermediate processing product of SgII, i.e., a C- and/or N-extended form of EM66.
In the anterior pituitary, intense EM66-IR was observed in all gonadotrophs as revealed by double labeling using a monoclonal antibody against LHβ. This finding is consistent with previous reports showing that SgII is a major product of gonadotrophs, in which its biosynthesis and release are regulated by gonadotropin-releasing hormone and sex steroids (Chanat et al. 1988; Anouar et al. 1991; Anouar and Duval 1992; Lloyd et al. 1992; Fischer-Colbrie et al. 1995; Watanabe et al. 1998). The intense EM66-IR observed in all gonadotrophs suggests that this peptide may represent a sensitive circulating marker for the diagnosis of human gonadotrope adenomas. In particular, EM66 may represent a valuable tool to characterize non-functioning pituitary tumors whose secretory granules have been shown to store SgII in the absence of the resident hormones (Rosa et al. 1992; Vallet et al. 1997).
Immunofluorescence labeling and co-localization experiments revealed that EM66-IR could also be detected in few lactotrophs and thyrotrophs, but not in corticotrophs. Given that corticotrophs have been shown to express SgII mRNA (Lloyd et al. 1992), the lack of EM66 labeling suggests that either the SgII protein is not expressed or the mature EM66 peptide is not produced in these cells, at least at detectable levels using our antibodies. In addition, electron microscopic analysis showed that somatotrophs are also devoid of EM66-IR. The differential occurrence of EM66-IR may reflect cell-specific processing of SgII that could be ascribed to the efficiency with which PCs can act on the cleavage sites of EM66 in different pituitary cells. Thus, it is now well established that both PC1 and PC2 are expressed in several cell types of the anterior pituitary, including gonadotrophs (Marcinkiewicz et al. 1993), corticotrophs (Day et al. 1992), and lactotrophs (Muller et al. 1998), and it has been shown that these processing enzymes are colocalized with chromogranins/secretogranins in pituitary and adrenal secretory granules (Scopsi et al. 1995). However, the expression level and the activity of each PC may vary depending on the cell type (Day et al. 1992; Marcinkiewicz et al. 1993), probably leading to cell-specific processing of SgII. Further studies using different cell lines transfected with SgII and PCs will be necessary to address this issue and to determine the efficiency of cleavage of EM66 from SgII. However, it should be noted that the HPLC analysis and the differential IHC localization of EM66 in the pituitary cells strongly suggest that this peptide is cleaved with a high efficiency and is the major immunoreactive SgII-derived form recognized by our antibody.
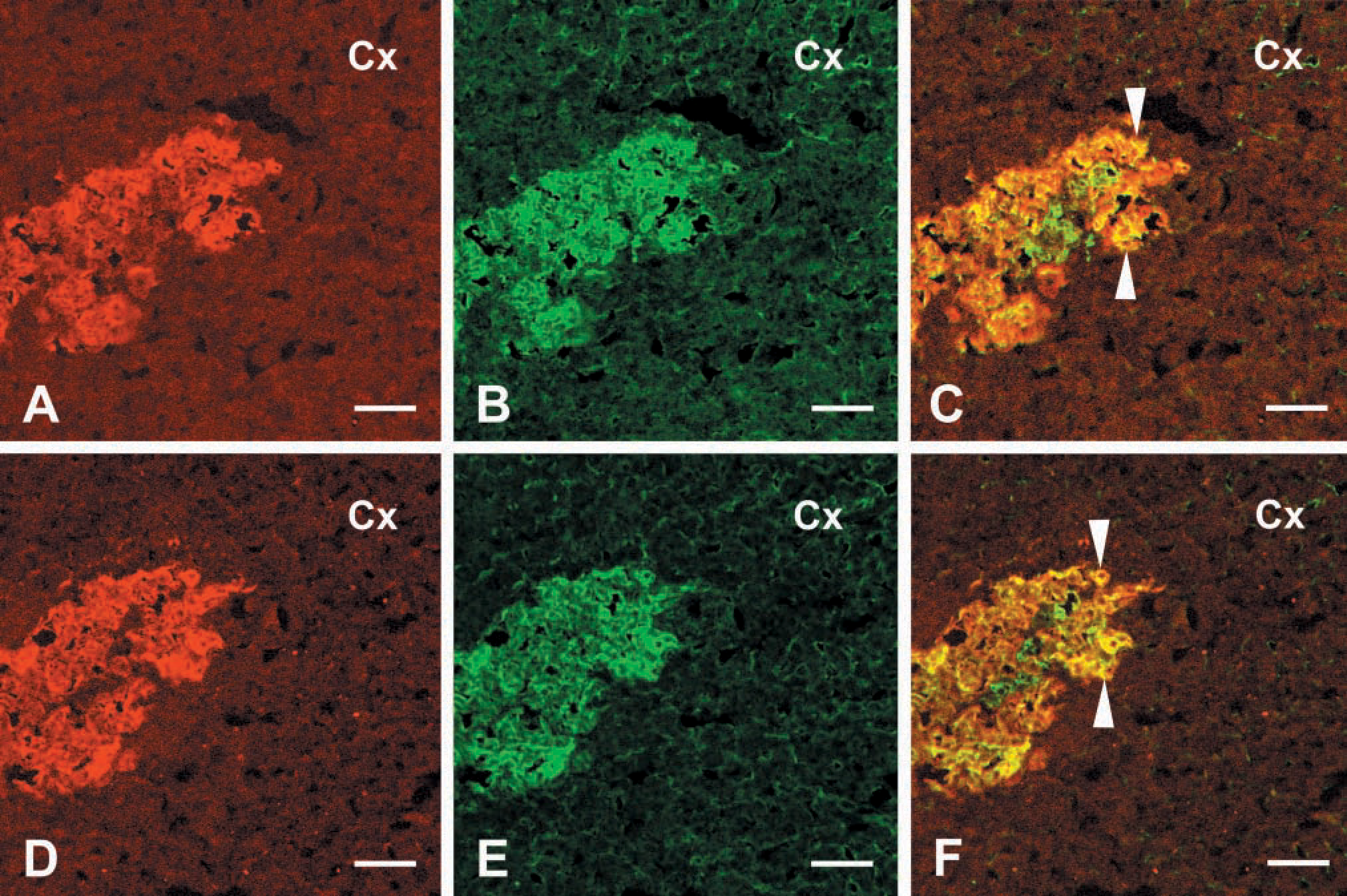
Confocal laser scanning microscope photomicrographs comparing the distribution of EM66, PNMT, and TH immunoreactivity in the rat adrenal gland. Sections through the adrenal were labeled with rabbit antiserum against EM66 (A) or PNMT (D) revealed with DAR/TXR and with a monoclonal antibody against TH (B,E) revealed with GAM/FITC. Combination of the two images acquired in A and B, and in D and E, revealed the presence of EM66 and PNMT in the same adrenochromaffin cells, i.e., adrenergic cells (arrowheads, C and F). Bars = 100 μm.
The present data also provide novel information regarding the cellular localization of a SgII-derived peptide in the rat adrenal medulla. Double labeling using antibodies against EM66, TH, and PNMT revealed that EM66-IR is restricted to a subset of TH-immunopositive cells, i.e., PNMT-expressing adrenergic cells. This finding highlights again the cell-specific occurrence of EM66 within endocrine tissues and implies either that SgII is expressed only in adrenergic cells, or that different mechanisms of SgII processing occur in adrenergic and noradrenergic chromaffin cells in vivo leading to production of EM66 in the former but not in the latter cells. Several studies using different experimental systems have shown that SgII can be cleaved by PC1 and PC2 in rat pheochromocytoma PC12 cells to give rise to several fragments including SN (Dittie and Tooze 1995; Hoflehner et al. 1995; Laslop et al. 1998). In addition, the presence of PC1 and PC2 has been described in chromaffin granules of the adrenal medulla (Kirchmair et al. 1993). However, it remains to be shown whether these enzymes are able to generate EM66 in chromaffin cells and whether the absence or presence of EM66-IR in chromaffin cell subpopulations is related to differential expression of PC1 and PC2 in these cells. Several reports have previously shown the existence of rays and islets of chromaffin cells in the adrenal cortex (Palacios and Lafarga 1975; Gallo-Payet et al. 1987; Bornstein et al. 1991; Perraudin et al. 1993; Berka et al. 1996). The present data indicate that these extramedullary adrenochromaffin cells also contain EM66-IR, suggesting that EM66 could play a role in the paracrine communication that occurs between chromaffin and steroidogenic cells in the adrenal gland (Bornstein and Ehrhart-Bornstein 1992; Ehrhart-Bornstein et al. 1998; Bornstein et al. 2000; Delarue et al. 2001).
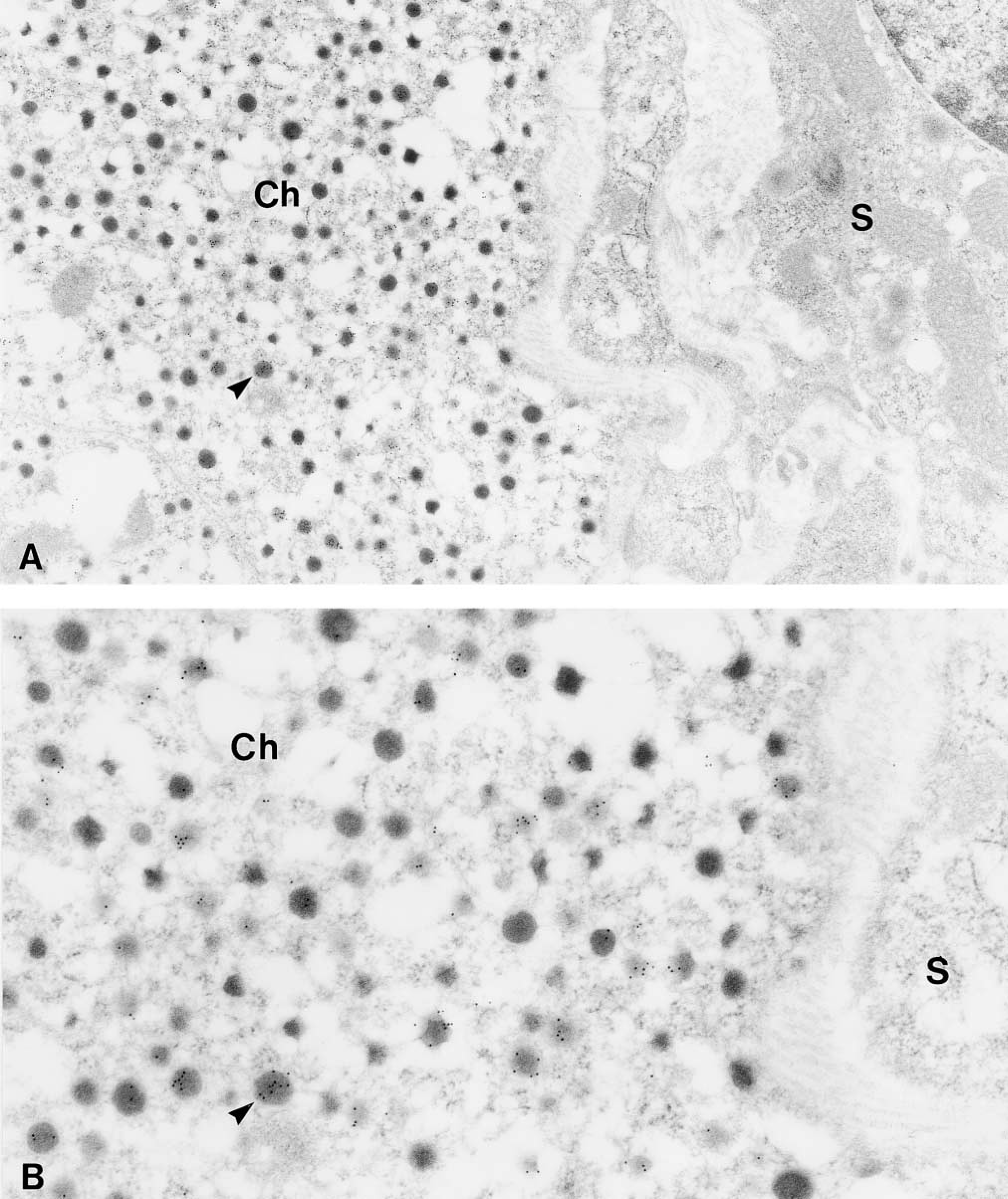
Immunoelectron microscopic localization of EM66 in the adrenal gland. (A) Gold particles are located over secretory granules (arrowhead) of a chromaffin cell (Ch) whereas an adjacent steroid-secreting cell (S) is unlabeled. Original magnification X25,000. (B) Higher magnification of a portion of the chromaffin cell. Original magnification X50,000.
Electron microscopic immunocytochemistry revealed that EM66-IR is confined to secretory vesicles of anterior pituitary and adrenomedullary chromaffin cells. Together with the HPLC data showing that mature EM66 peptide is present in the pituitary and the adrenal gland, these results imply that EM66 is formed into secretory granules. Consequently, it is conceivable that EM66 can be released into the external milieu by anterior pituitary cells and adrenomedullary cells on stimulation. This hypothesis raises the possibility that EM66 may exert hormonal activities. In fact, several observations suggest that EM66 may play endocrine functions. (a) The sequence of this peptide has been highly conserved during evolution and we have found that EM66 is actually processed in human (Anouar et al. 1998), rat (this study), and frog (Yon et al. unpublished work) endocrine glands. (b) EM66 is generated early during the ontogenesis of the fetal human adrenal gland (Anouar et al. 1998), suggesting that its formation is necessary throughout life. (c) EM66 exhibits a cell-specific occurrence in endocrine tissues, where it is confined to secretory vesicles. Therefore, EM66 may be released on stimulation of endocrine cells to act as an autocrine, paracrine, and/or endocrine factor. Alternatively, EM66 may represent a conserved structural motif allowing SgII to play an intracellular role in the morphogenesis and the function of secretory vesicles because the chromogranin/secretogranin family members are postulated to have a crucial role in these processes (Yoo et al. 2000; Kim et al. 2001). Further studies are required to test these hypotheses.
In conclusion, this study has demonstrated the occurrence of EM66, a novel peptide derived from the processing of SgII, in different cell types of the rat anterior pituitary and in adrenal chromaffin cells. In addition, the present data suggest that EM66 could be stored and released from the secretory granules of these cells and thus support the notion that EM66 may play a role in the endocrine system.
Footnotes
Acknowledgements
Supported by grants from the Institut National de la Santé et de la Recherche Médicale (INSERM U413) and the Conseil Régional de Haute-Normandie. We are very grateful to Dr JL Do Régo for CLSM data analysis and Ms H Lemonnier for skillful technical assistance. We thank Drs JF Roser (Davis, CA), and J Cote (Laval University, Quebec, Canada) for generously supplying the LHβ antibodies and the ACTH antiserum, respectively.
