Abstract
The soft palate is the only structure that reversibly separates the respiratory and gastrointestinal systems. Most species can eat and breathe at the same time. Humans cannot do this and malfunction of the soft palate may allow food to enter the lungs and cause fatal aspiration pneumonia. Speech is the most defining characteristic of humans and the soft palate, along with the larynx and tongue, plays the key roles. In addition, palatal muscles are involved in snoring and obstructive sleep apnea. Considering the significance of the soft palate, its function is insufficiently understood. The objectives of this study were to document morphometric and immunohistochemical characteristics of adult human soft palate muscles, including fiber size, the fiber type, and myosin heavy chain (MyHC) composition for better understanding muscle functions. In this study, 15 soft palates were obtained from human autopsies. The palatal muscles were separated, cryosectioned, and stained using histological and immunohistochemical techniques. The results showed that there was a fast type II predominance in the musculus uvulae and palatopharyngeus and a slow type I predominance in the levator veli palatine. Approximately equal proportions of type I and type II fibers existed in both the palatoglossus and tensor veli palatine. Soft palate muscles also contained hybrid fibers and some specialized myofibers expressing slow-tonic and embryonic MyHC isoforms. These findings would help better understand muscle functions.
Keywords
Introduction
The soft palate is the only structure that reversibly separates the respiratory and gastrointestinal systems. The human soft palate is a particularly important structure in the oropharyngeal region. Functionally, movements of the soft palate during upper airway motor tasks are involved with speech, swallowing, and breathing. Speech is the most defining characteristic of humans and the soft palate, along with the larynx and tongue, plays the key roles. Anatomically, soft palate is characterized by containing five pairs of palatal muscles, including the levator veli palatini (LVP), musculus uvulae (MU), palatopharyngeus (PP), palatoglossus (PG), and tensor veli palatini (TVP) 1 that intertwine in a complex fashion. Our recent studies 2 have demonstrated that the human soft palate muscles are innervated by multiple cranial nerves, including the pharyngeal branches of the glossopharyngeal (IX) and vagus (X) nerves as well as the lesser palatine nerve (LPN). Some palatal muscles receive dual or triple innervation. The specialized motor innervation suggests that the soft palate muscles may have distinct contractile properties with appropriate speed and strength for performing their distinct functions.
The wide range of physiological behaviors of the soft palate in speech, swallowing, and breathing would be associated with the content of myosin heavy chain (MyHC) isoforms expressed by the muscle fibers. We hypothesized that the soft palate muscles innervated by the same cranial nerve would have similar muscle fiber types and that the muscles involved in respiration would have slower muscle fibers than those involved in swallowing. Different fiber types within a muscle have different maximum force, contraction velocity, and fatigability. Muscles predominantly containing type I fibers are relatively slow contracting and fatigue resistant, whereas those containing more type II fibers are faster contracting and more sensitive to fatigue. 3 However, limited data are available regarding MyHC expression and fiber type distribution in the human soft palate muscles. 4
This study focused on determining the internal specializations of adult human soft palate muscles such as fiber size, the fiber type, and MyHC composition for better understanding muscle functions.
Materials and Methods
Tissue Source and Demographic Features
Fifteen whole-mount specimens (tongue-soft palate-pharynx-larynx-upper esophagus) were obtained from deceased adult human subjects without known systemic neuromuscular disorders. The specimens were provided by the Department of Pathology at Mount Sinai Medical Center in New York.
Demographic features of the subjects are summarized in Table 1. Specifically, the 15 autopsied specimens used in this study were obtained from subjects of both sexes (nine men and six women), with a mean age of 59 years (range, 34–73 years). The cause of death for each subject is given in Table 1. The mean postmortem interval between death and tissue preparation was 32 hr (range, 24–40 hr); this postmortem interval does not hamper reliable histochemical analysis of autopsied tissues.5,6
Demographic Data of Subjects.
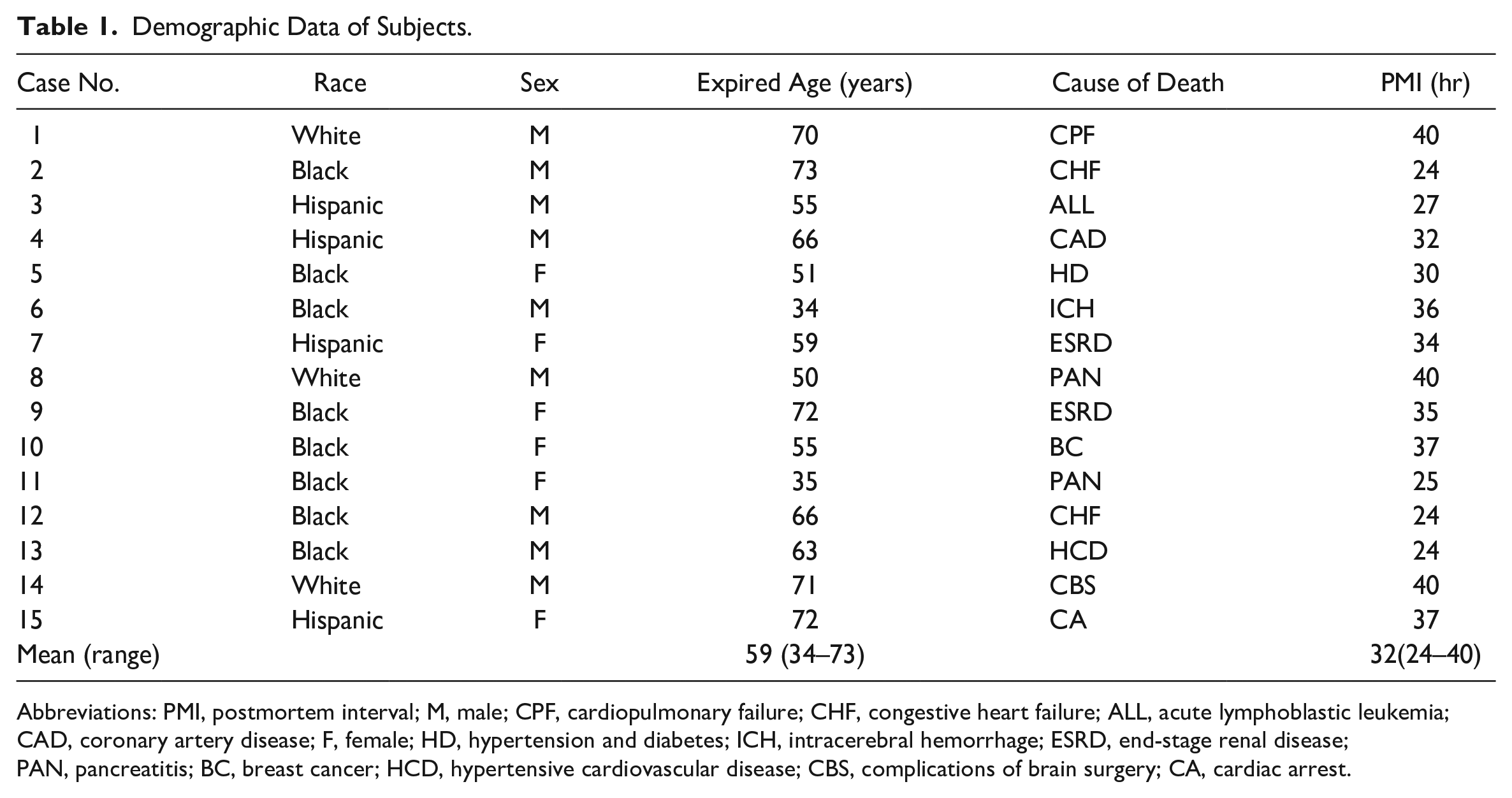
Abbreviations: PMI, postmortem interval; M, male; CPF, cardiopulmonary failure; CHF, congestive heart failure; ALL, acute lymphoblastic leukemia; CAD, coronary artery disease; F, female; HD, hypertension and diabetes; ICH, intracerebral hemorrhage; ESRD, end-stage renal disease; PAN, pancreatitis; BC, breast cancer; HCD, hypertensive cardiovascular disease; CBS, complications of brain surgery; CA, cardiac arrest.
Muscle Sample Preparation and Staining Methods
Each of the removed entire soft palates was bisected in the midline to divide the specimen into equal and symmetrical halves. Fifteen hemi-soft palates were used in this study, whereas the remaining 15 hemi-specimens were used for other purposes. In each hemi-soft palate, five palatal muscles including the LVP, MU, PP, PG, and TVP were sampled as illustrated by Fig. 1. Each of the muscle samples (~10 × 5 × 5 mm) was frozen by immersion in melting isopentane cooled by dry ice, cut transversely (10-µm thick) on a cryostat (Reichert-Jung 1800; Mannheim, Germany) at −25C, and stored at −80C until staining was performed.
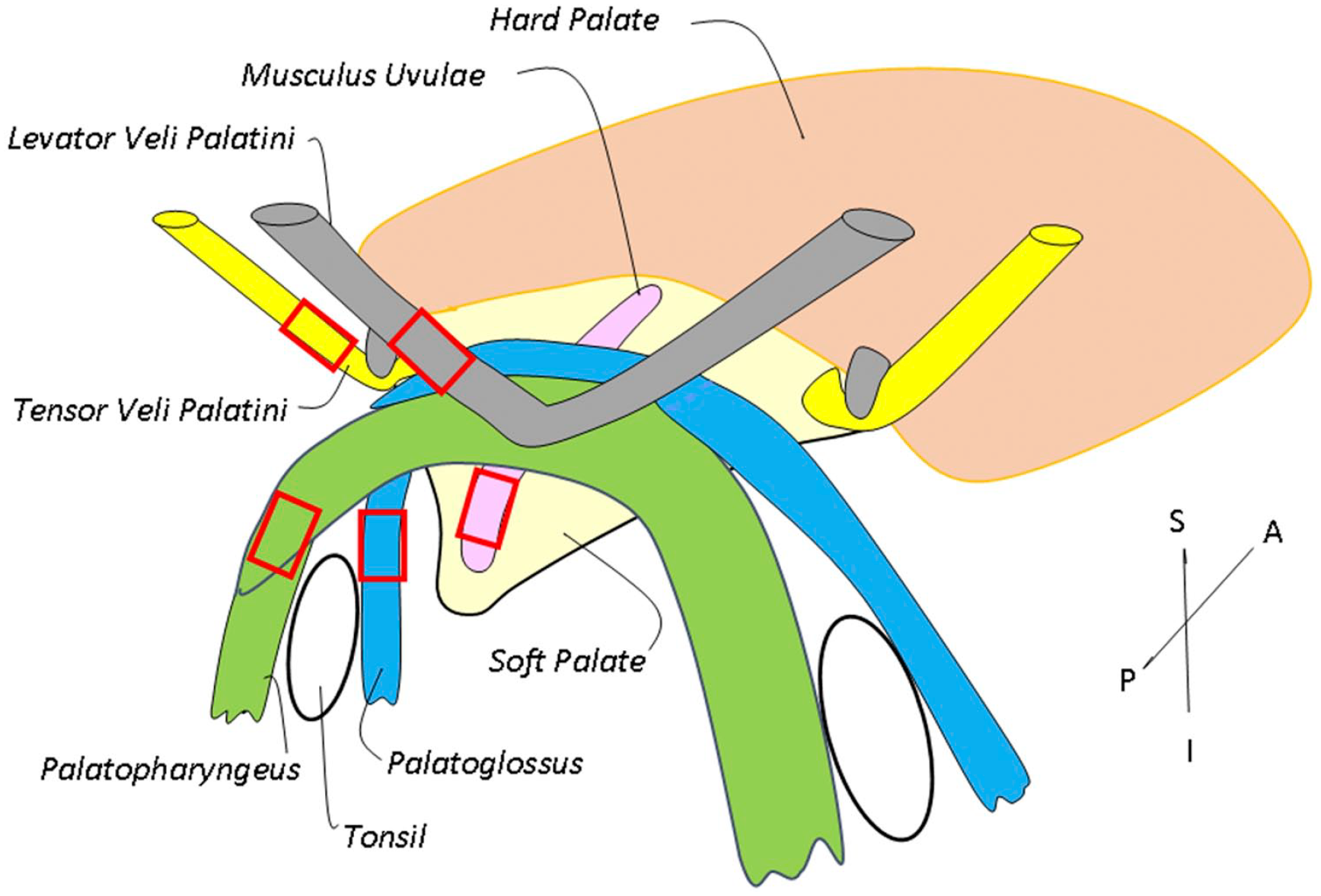
Schematic diagram of the human soft palate muscles, showing tissue sampling sites (enclosed).
Serial cross-sections from each muscle sample were mounted on sets of seven slides. For each set, section 1 was stained with hematoxylin and eosin staining (H&E) to examine muscle structure. Section 2 was stained for succinic dehydrogenase (SDH), an oxidative enzyme, to examine the oxidative capacity of the muscle fibers. 7 Sections 3–7 were immunostained with type-specific anti-MyHC antibodies to examine fiber type and MyHC composition.
Immunohistochemistry
Monoclonal Antibodies (mAbs)
The muscle sections 3–7 were incubated with type-specific anti-MyHC mAbs against three major (MyHCI, MyHCIIa, and MyHCIIx) and two unusual [slow-tonic (MyHC-ton) and embryonic (MyHC-emb)] isoforms (Table 2). The specificity and characterization of these mAbs against MyHC isoforms have been previously established. Specifically, the mAb NOQ7-5-4D is specific to type I MyHC,8–10 MY-32 to all fast type MyHCs,11,12 SC-71 to type IIa MyHC,12–14 ALD-58 to MyHC-ton,15,16 and F1.652 to MyHC-emb. 17
mAbs Used for Immunohistochemistry.
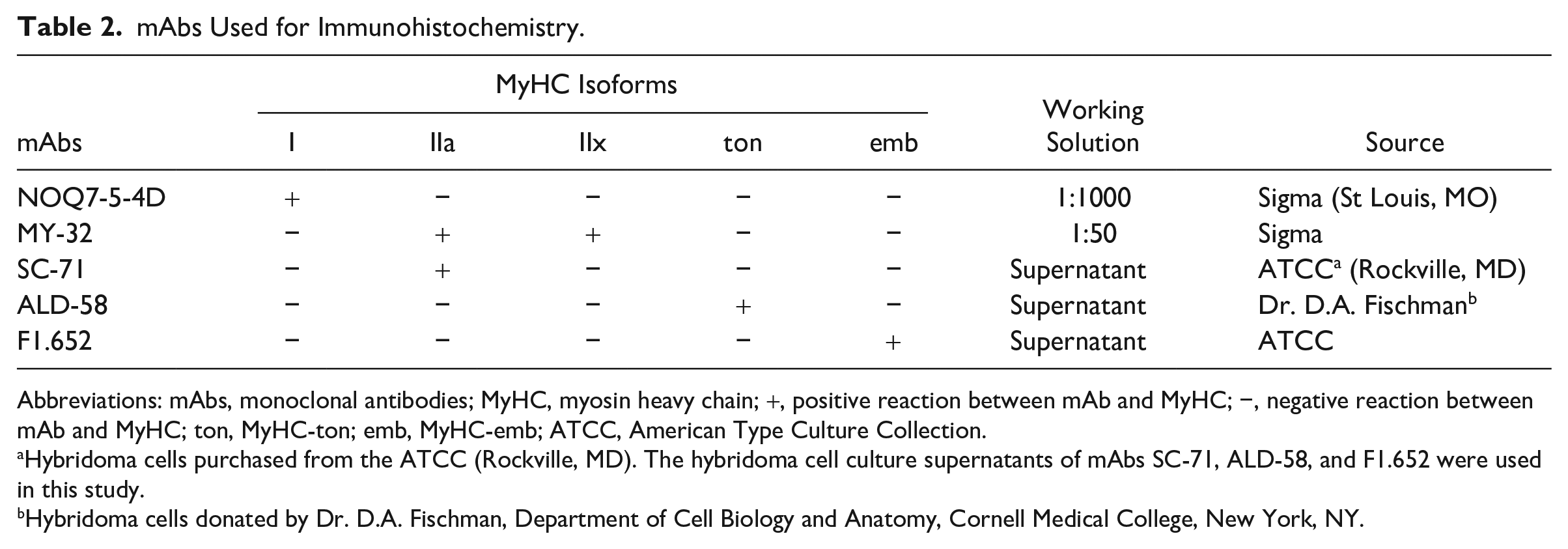
Abbreviations: mAbs, monoclonal antibodies; MyHC, myosin heavy chain; +, positive reaction between mAb and MyHC; −, negative reaction between mAb and MyHC; ton, MyHC-ton; emb, MyHC-emb; ATCC, American Type Culture Collection.
Hybridoma cells purchased from the ATCC (Rockville, MD). The hybridoma cell culture supernatants of mAbs SC-71, ALD-58, and F1.652 were used in this study.
Hybridoma cells donated by Dr. D.A. Fischman, Department of Cell Biology and Anatomy, Cornell Medical College, New York, NY.
Avidin-Biotin-Complex (ABC) Method
The major MyHC isoforms were detected with ABC method according to instructions for Vectastain PK-6102 kit (Vector Labs; Burlingame, CA) as described in our previous publications.18–21 Briefly, tissue sections were (1) fixed in 4% paraformaldehyde for 10 min; (2) incubated in a blocking solution composed of 2% bovine serum albumin (BSA) and 0.1% Triton X-100 at room temperature (RT) for 20 min to prevent nonspecific binding; (3) incubated with anti-MyHC mAbs for 1 hr at RT (mAb NOQ7-5-4D) or overnight in a humid chamber at 4C (mAbs MY-32 and SC-71); (4) processed with a Vectastain anti-mouse IgG (ATCC; Rockville, MD) for 1 hr at RT; (5) reacted for 1 hr at RT in ABC reagent; and (6) reacted for ~10 min at RT with a solution containing 3,3′-diaminobenzidine (DAB) as chromogen to localize peroxidase for primary antibodies according to DAB substrate kit (cat. no SK-4100; Vector Labs). The slides were then washed with water for 5 min 3×, dehydrated, and coverslipped in Permount Mounting Medium (Fisher Scientific). Two additional control sections were treated using the aforementioned procedures but without incubation with a primary antibody.
Immunofluorescence
Unusual MyHC-ton and MyHC-emb containing fibers in the palatal muscles were detected using indirect immunofluorescent staining as described in our previous publications.18–21 Briefly, serial cross-sections adjacent to those stained with ABC method were (1) fixed in 4% paraformaldehyde for 10 min; (2) blocked in 2% BSA with 0.1% Triton X-100 for 20 min; (3) incubated with two anti-MyHC mAbs at 4C for either 12 hr (mAb F1.652) or 24 hr (mAb ALD-58); (4) blocked again with 1% BSA at RT for 5 min; and (5) incubated in a fluorescein isothiocyanate-labeled goat anti-mouse IgG (FITC-GAM; 1:50 dilution; Sigma, St Louis, MO) at RT for 60 min. The stained sections were mounted with Vectashield mounting medium (Vector Labs) and kept in dark at 4C. Control sections were stained as described above except that the incubation with the primary antibody was omitted.
Image Acquisition and MyHC-Based Muscle Fiber Typing
The stained sections were viewed on a Zeiss photomicroscope (Axiophot-1; Carl Zeiss, Goettingen, Germany) equipped with epifluorescence optics. Sections were photographed using a USB 3.0 digital microscope camera (Infinity 3-3URC; Lumenera Corp., Ottawa, Ontario, Canada).
Images at a magnification of 200× were used to classify fiber types and determine the relative frequency of fibers containing different MyHC isoforms. On each stained cross-section, approximately 200 fibers were randomly selected in each muscle for single-fiber MyHC composition analysis. The photographed images were imported into an image-processing program (ImageJ v.1.45s; National Institutes of Health, Bethesda, MD) to identify and mark positive and negative fibers that reacted to a specific mAb.
Three major muscle fiber types were categorized by reaction profiles to MyHC antibodies: type I (NOQ7-5-4D+, MY-32−, SC-71−), type IIA (MY-32+, SC-71+, NOQ7-5-4D−), and type IIX (MY32+, SC71−, NOQ7-5-4D−). Each was expressed as a percentage of the total number of fibers examined. The fibers were classified into three major pure fibers (types I, IIA, and IIX) expressing MyHCI, IIa, or IIx, and hybrid fibers (types I/IIA and IIA/IIX) coexpressing two or more different adult MyHC isoforms.
Two unusual MyHC (MyHC-ton and MyHC-emb) containing fibers were also identified using ImageJ, and their expression patterns and frequencies were determined.
Myofiber Size Measurement
The muscle sections stained with NOQ7-5-4D were used for measuring the least fiber diameter using a previously established method. 7 In each muscle, at least 100 fibers were randomly selected to measure the least fiber diameter by using ImageJ software.
Statistical Analyses
Fiber diameters and relative proportions of the major and unusual MyHC-containing fibers in each of the palatal muscles were computed. All data were reported as means ± standard deviation. Data on myofiber size in diameter, percentages of major and unusual MyHC-containing fibers were analyzed statistically using mixed model for repeated measures with the five palatal muscles as repeated variable. The procedure Proc MIXED/SAS 9.4 was utilized to conduct the evaluation of the effects and subsequent pairwise comparison. All data analysis was performed in SAS 9.4 (SAS Institute Inc.; Cary, NC). Any p<0.05 was considered an indication of statistical significance.
Results
Myofiber Size
Table 3 summarizes the mean fiber diameter for each palatal muscle. Specifically, the mean diameters of the type I and type II fibers were measured to be 39 and 41 µm for MU, 48 and 53 µm for PP, 46 and 50 µm for LVP, 49 and 50 µm for PG, and 42 and 49 µm for TVP, respectively. The mean diameter of the type I fibers (45 µm) in all palatal muscles was relatively smaller as compared with that of the type II fibers (49 µm) (p=0.0007). Overall, the mean fiber diameter of all the MU fibers (40.3 µm) was relatively smaller than that of other four palatal muscles (average 49 µm, range: 45.6–50.4 µm) [p≤0.0001 to 0.0008; except for the comparison between MU and TV (p=0.0008), all other pairwise comparisons with MU yielded p<0.0001].
Percent Composition of Fiber Diameters.
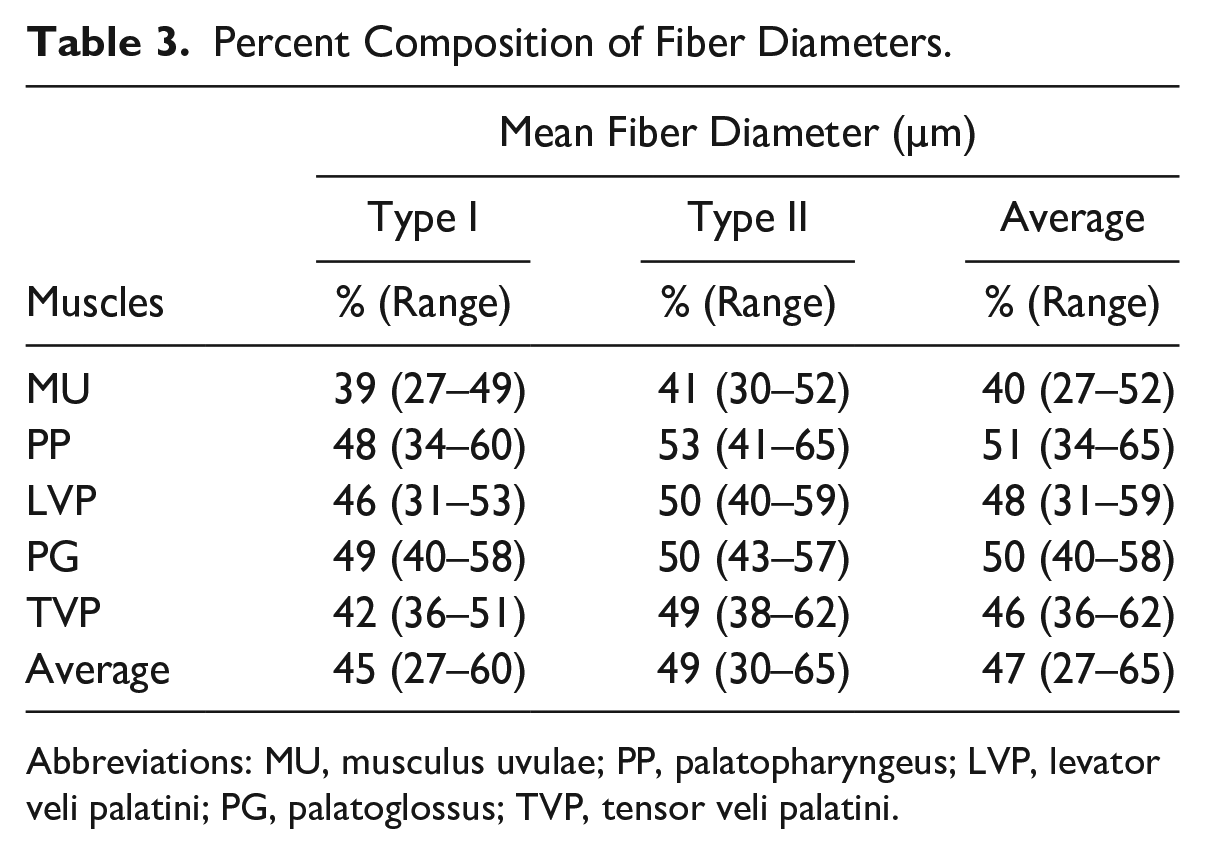
Abbreviations: MU, musculus uvulae; PP, palatopharyngeus; LVP, levator veli palatini; PG, palatoglossus; TVP, tensor veli palatini.
SDH Activity
The sections stained for SDH exhibited small spherical diformazan granules that were preferentially distributed toward the fiber periphery (Fig. 2). The muscle fibers were stained darkly, moderately, or weakly. In general, slow type I fibers in limb muscles have higher level of SDH than fast type II fibers. However, type II fibers in all soft palate muscles also had stronger SDH activity as compared with the biceps brachii (Fig. 2). These findings suggest that the human soft palate muscles are relatively fatigue resistant.
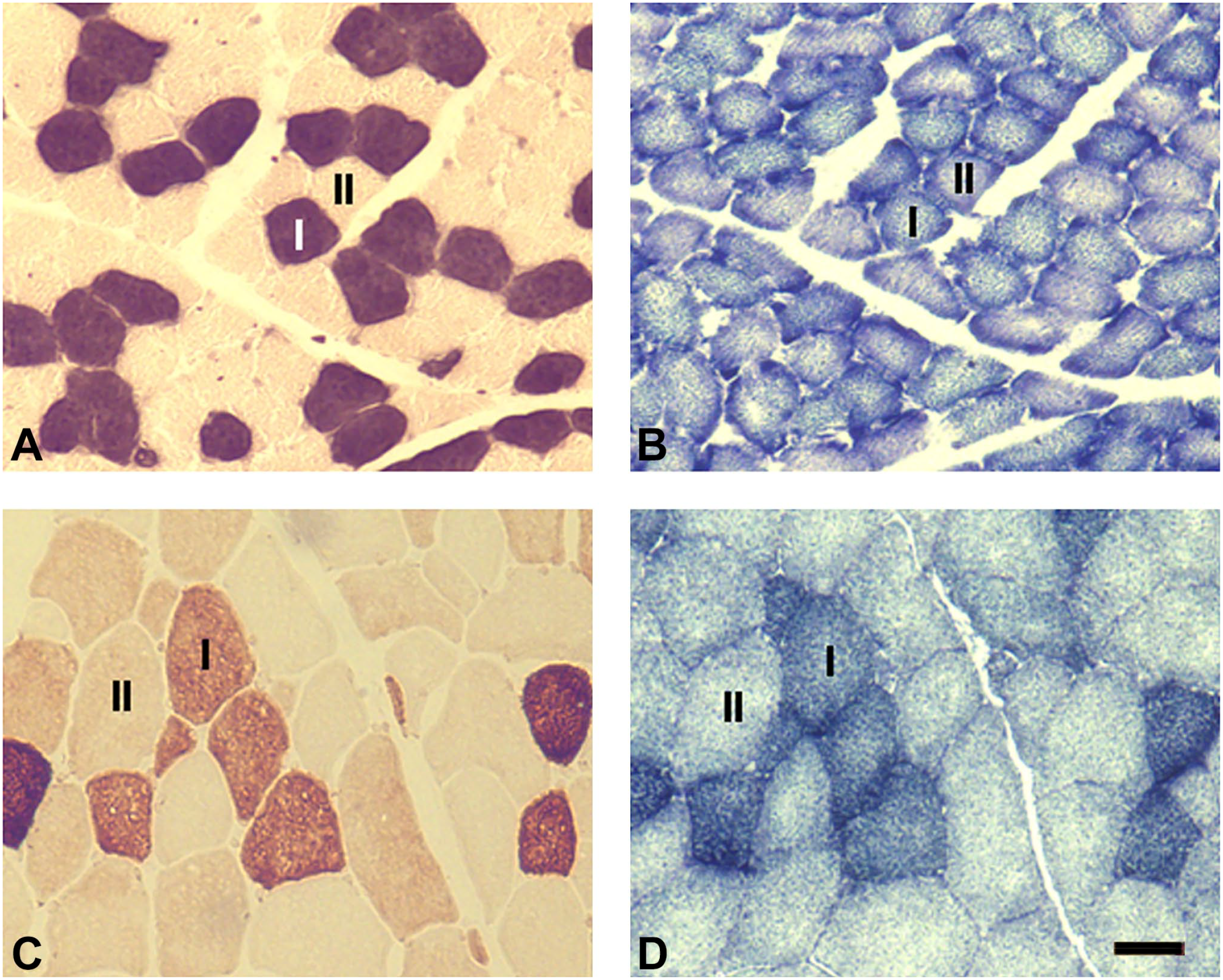
Cross-sections from human tensor veli palatini (TVP) (A-B) and from human biceps brachii (C-D) stained for NOQ7-5-4D (A, C) and succinic dehydrogenase (SDH) (B, D). Note that the fast type II fibers in the TVP had a higher level of SDH activity than those in the biceps brachii. Bar = 50 µm.
Distribution of the Major MyHC-Based Fiber Types
Figure 3 is an example showing fiber-to-fiber comparison to identify major fiber types. The mean proportions of the major MyHC-based fiber types in each of the human palatal muscles are shown in Fig. 4. Specifically, MU was demonstrated to be a fast muscle that contained 12% type I fibers, 76% type II fibers (74% IIA and 2% IIX), and 12% hybrid fibers (4% I/IIA and 8% IIA/IIX). PP was also a fast muscle containing 15% type I, 42% type II (39% IIA and 3% IIX), and 43% hybrid fibers (11% I/IIA and 32% IIA/IIX). In contrast, LVP was demonstrated to be a slow muscle that was composed predominantly of type I fibers (75%), with a balance of 16% type II (15% IIA and 1% IIX) and 9% hybrid fibers (4% I/IIA and 5% IIA/IIX). Approximately equal percentages of slow type I and fast type II fibers were observed in both PG (48% type I, 19% IIA, 5% IIX, 11% I/IIA, and 17% IIA/IIX) and TVP (50% type I, 24% IIA, 2% IIX, 8% I/IIA, and 16% IIA/IIX). The results showed that the fast type II population was larger in the MU and PP as compared with the LVP (p<0.0001), PG (p<0.0001), and TVP (p<0.0001), respectively. In contrast, slow type I population was larger in LVP (p=0.0437), PG (p=0.0006), and PP (p=0.0020) as compared with MU, respectively. Distribution of the hybrid I/IIA and IIA/IIX fibers varied among the palatal muscles. Specifically, a relatively higher percentage of I/IIA fibers was found in the PP (11%) and PG (11%) as compared with MU (4%; p<0.0001) and LVP (4%; p<0.0001), whereas a higher percentage of IIA/IIX was observed in the PP (32%), PG (17%), and TVP (16%) as compared with the MU (8%) (p<0.0001, <0.0001, and 0.0002, respectively) and LVP (5%) (p<0.0001, <0.0001, and <0.0001, respectively).
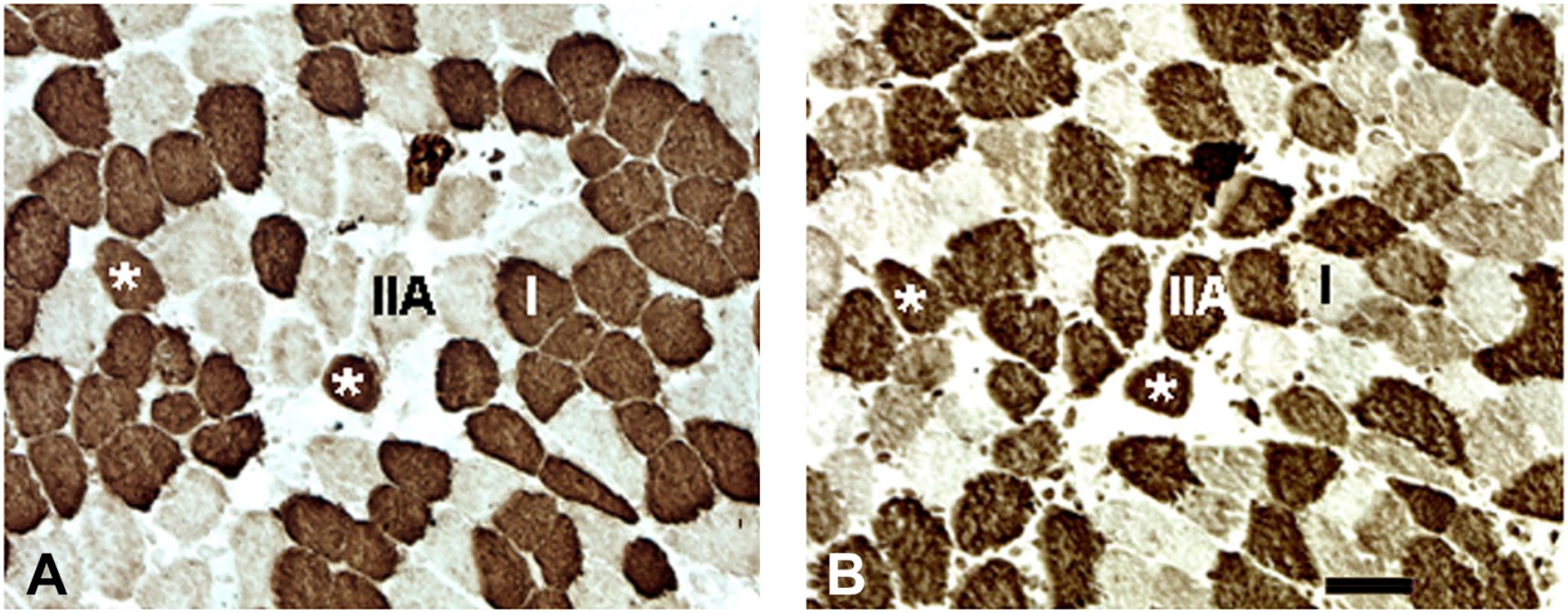
Cross-sections of human levator veli palatini stained with monoclonal antibodies NOQ7-5-4D specific to type I myosin heavy chain (MyHC) (A) and SC-71 to type IIa MyHC (B), showing the fiber types I, IIA, and I/IIA hybrid fibers (asterisks). Bar = 50 µm.
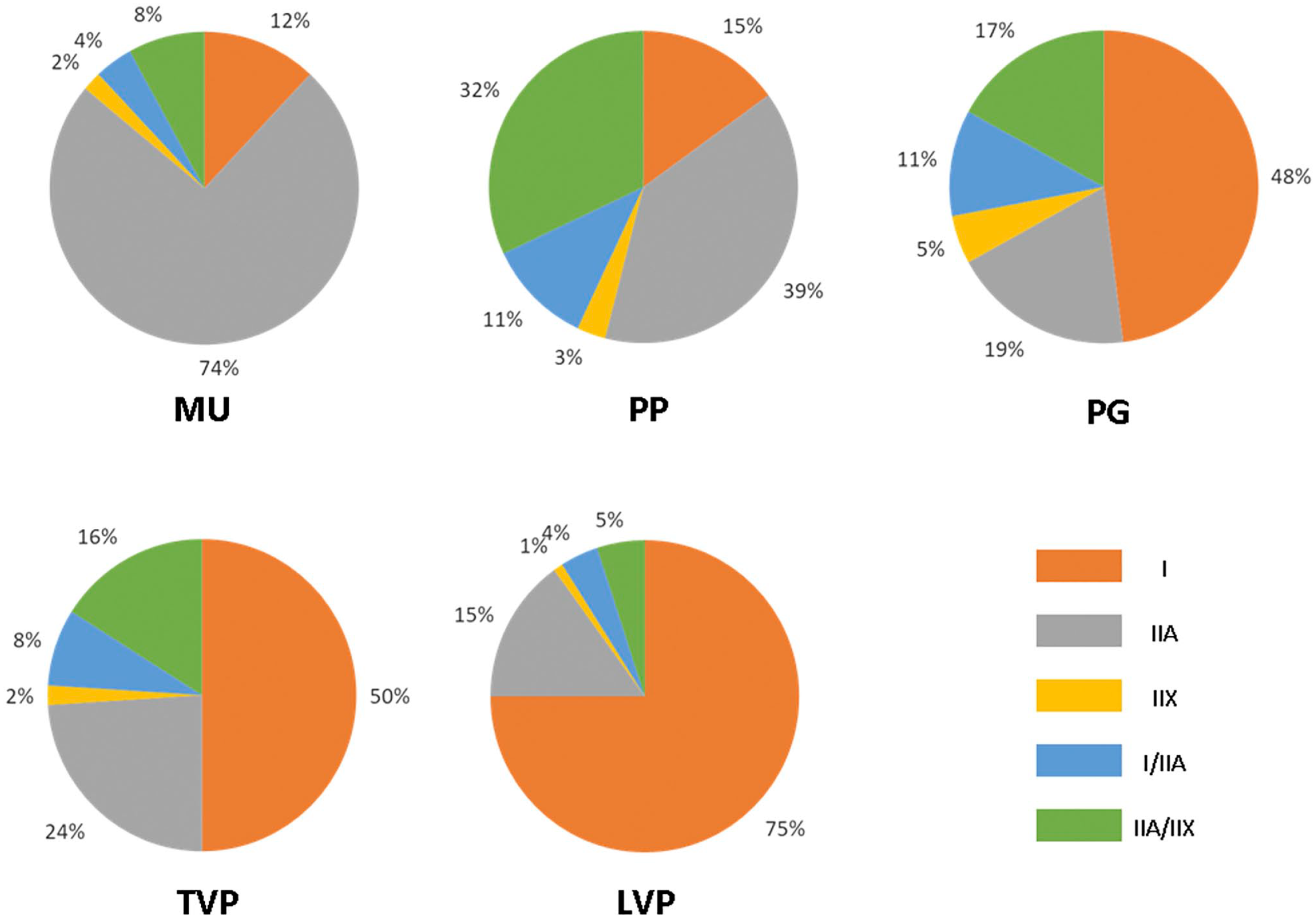
Relative percentages of the myosin heavy chain-based fiber types in adult human MU, PP, PG, TVP, and LVP muscles. Abbreviations: MU, musculus uvulae; PP, palatopharyngeus; PG, palatoglossus; TVP, tensor veli palatini; LVP, levator veli palatini.
Expression of the Unusual MyHC Isoforms
Some fibers of the adult human palatal muscles expressed unusual MyHC isoforms (i.e., MyHC-ton and MyHC-emb). The unusual MyHC isoforms were found to coexist with the major MyHCs. Specifically, MyHC-ton was coexpressed with slow type I MyHC, whereas MyHC-emb coexisted with fast type II MyHCs (Fig. 5). MyHC-ton was expressed by 2%, 3%, 12%, 18%, and 26% of the type I fiber population in the MU, PP, PG, TVP, and LVP, respectively. In contract, MyHC-emb were expressed by 29%, 17%, 12%, 11%, and 7% of the type II fiber population in the MU, PP, PG, TVP, and LVP, respectively (Fig. 6). Clearly, slow LVP contained more MyHC-ton fibers than fast MU (p<0.0001) and PP (p<0.0001) as well as PG (p<0.0001) and TVP (p=0.0021), whereas fast MU contained significantly more MyHC-emb fibers than the slow LVP (p<0.0001) and other palatal muscles (all comparisons with MU reported p<0.0001).
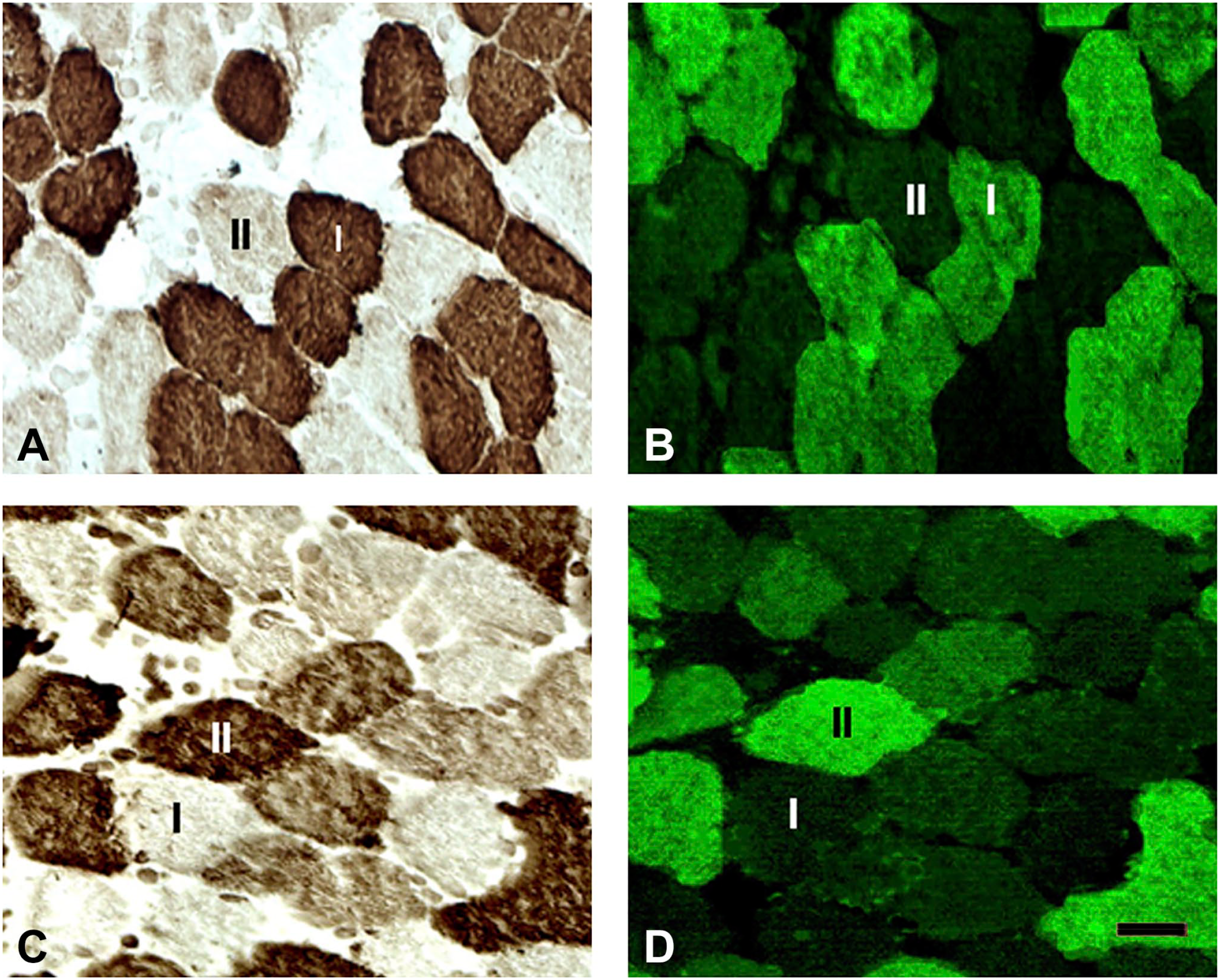
Cross-sections of adult human tensor veli palatini (TVP) stained with monoclonal antibodies NOQ7-5-4D (A), ALD-58 (B), SC-71 (C), and F1.652 (D). Note that the TVP contains muscle fibers expressing myosin heavy chain (MyHC)-ton (B) and MyHC-emb (D) isoforms. MyHC-ton was coexpressed with MyHCI, whereas MyHC-emb coexisted with MyHCII. Bar = 25 µm.
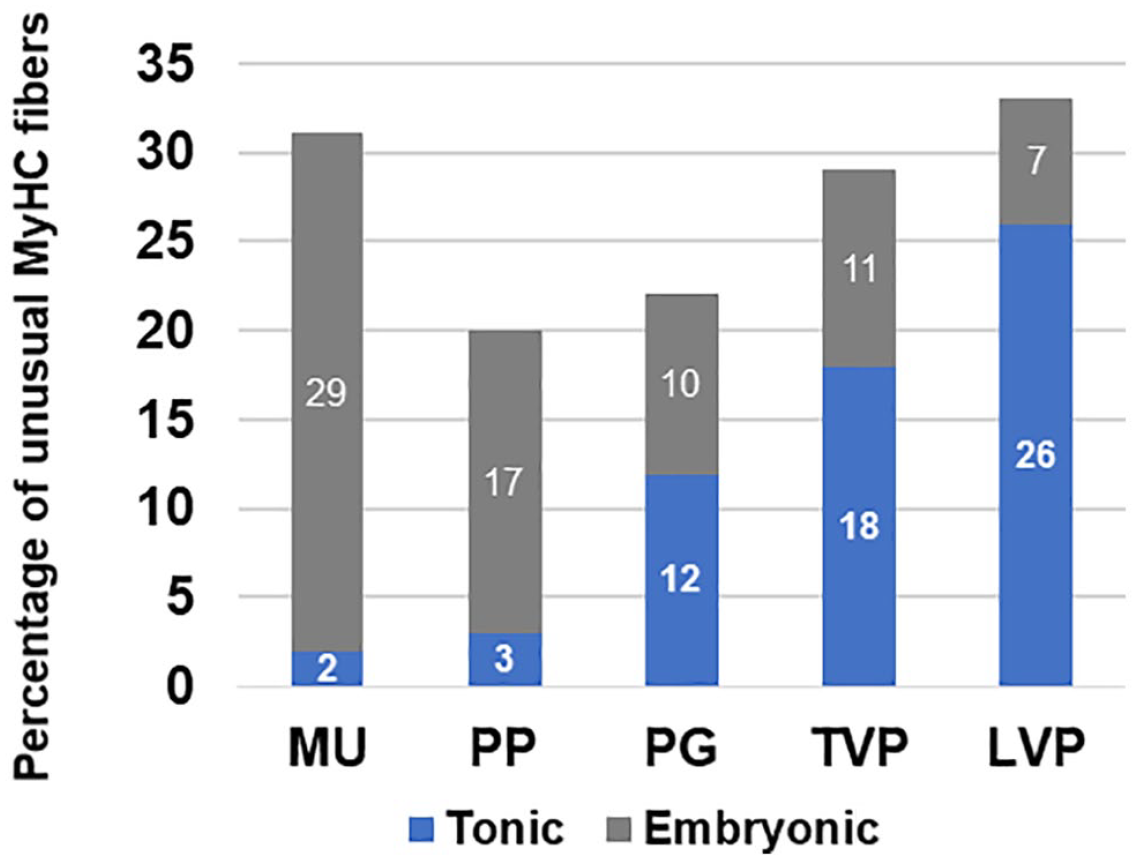
Relative percentages of the MyHC-ton and MyHC-emb containing fibers in adult human MU, PP, PG, TVP, and LVP muscles. Abbreviations: MyHC, myosin heavy chain; MU, musculus uvulae; PP, palatopharyngeus; PG, palatoglossus; TVP, tensor veli palatini; LVP, levator veli palatini.
Discussion
This study investigated MyHC composition and fiber type distribution in adult human soft palate muscles. Several key findings are obtained from this study. First of all, soft palate muscles differ in the relative prevalence of fiber types. Specifically, a type I predominance in the LVP and a type II predominance in the MU and PP muscles were demonstrated. Approximately equal percentages of slow type I and fast type II fibers were observed in the PG and TVP muscles. Second, different percentages of major hybrid fibers (i.e., I/IIA and IIA/IIX) existed in the soft palate muscles. In the PP muscle, the IIA/IIX hybrid fibers accounted for one third of the total fiber population. Third, adult human soft palate muscles were more similar to human pharyngeal and laryngeal muscles that contained myofibers expressing unusual MyHC-ton and MyHC-emb. MyHC-ton isoform coexpressed with slow type I MyHC, whereas MyHC-emb coexisted with fast type II MyHC. These specialized MyHC fibers may play an important role in muscle contractile properties needed for speech, respiration, and swallowing. Finally, fast type II fibers in all soft palate muscles also had stronger SDH activity as compared with those in the biceps brachii. These findings suggest that the human palatal muscles are relatively fatigue resistant.
The human soft palate is an anatomically complex structure which is composed of five pairs of muscles. These muscles are typically divided functionally into elevators (i.e., LVP and MU), depressors (i.e., PP and PG), and tensor (i.e., TVP), according to their mechanical actions. These muscles produce a variety of intricate but well-controlled movements, including elevation and depression of the soft palate, as well as alterations in palate shape and stiffness.
LVP is a thick, rounded muscle which originates at the apex of the petrous portion of the temporal bone and the medial wall of the Eustachian tube. Its fibers run anteroinferiorly and reach the midline in the middle 40% of the velum and insert at the palatine aponeurosis. 22 Our study showed that the human LVP receives dural innervation from both the pharyngeal branch of the X nerve and LPN. 2 LVP is the primary muscle for velar elevation and velopharyngeal closure for speech production23,24 and swallowing. 25 During swallowing and speech, velopharyngeal closure is achieved by the velum, lateral and posterior pharyngeal walls. During swallowing, the velopharyngeal closure prevents food regurgitation into the nasal cavity, and during phonation, it regulates nasal resonance. Contraction of the LVP elevates and retracts the velum and extends posteriorly to close against the posterior pharyngeal wall for non-nasal articulations. Dysfunction of the LVP results in hypernasal speech.26,27 LVP electromyographic (EMG) activities have been recorded during swallowing 25 and respiration. 28 This study showed that LVP contained a high proportion of slow type I fibers and about one third of the type I fibers contained MyHC-ton. The content of fiber type and MyHC composition in the LVP is suited for its slower contractions with longer durations in elevating and stretching the soft palate as demonstrated.23,29
MU is a paired muscle adjacent to each other and lies close to the nasal surface of the velum. Its fibers originate from the posterior margin of the palatine aponeurosis and run along the midline coursing the entire length of the soft palate.30,31 Our recent study showed that human MU muscle is innervated by the LPN only. 2 This novel finding challenges the current and prevailing view that the LPN is a sensory nerve that innervates the palatal mucosa and palatine glandular tissue. 1 MU muscle is regarded as a velar elevator, stiffness modifier, and extensor. 32 Contraction of the MU lifts the soft palate and straightens the curved velum to contact with the posterior pharyngeal wall (velar extension) during speech and swallowing.31–36 This study showed that the MU is a fast muscle with a higher proportion of type II fibers that facilitate rapid muscle contraction during speech and swallowing.
PP, a palate depressor, extends throughout the entire length of the pharynx. Its fibers course inferiorly and laterally behind the tonsil forming the posterior tonsillar pillar and insert into the posterior border of the thyroid cartilage. Our study demonstrated that PP receives triple innervation from the LPN and pharyngeal branches of the IX and X nerves. 2 The type II predominance in the PP indicates that it is a fast muscle for rapid phasic movements of the soft palate. Contraction of the PP depresses soft palate, elevates pharynx, and narrows fauces as seen during swallowing37,38 and speech.39–42
PG, a palate depressor, arises from the palatine aponeurosis of the soft palate. It extends from the lateral part of the soft palate to the side of the posterior tongue and forms the anterior tonsillar pillar. 43 Our study demonstrated that the human PG is innervated by the pharyngeal branches of the IX and X nerves. 2 Contraction of the PG lowers soft palate, elevates posterior tongue, and constricts the anterior tonsillar pillars, as seen during swallowing 44 and speech. 45 During speech, PG contraction elevates the posterior tongue to produce oral constrictions, and lowers the velum to open the velopharyngeal port for the production of nasal phonemes.39,43–47 Application of negative upper airway pressure resulted in increased inspiratory EMG activities of the PG in normal subjects. 28 Approximately equal percentages of slow type I and fast type II fibers in the PG as demonstrated by this study could be more suited to its functional needs.
TVP, a palate tensor, is a thin, broad, flat muscle that originates from the cranial base and the lateral wall of the Eustachian tube. 48 The TVP is innervated by the mandibular division of the trigeminal nerve. 1 Contraction of the TVP tenses and spreads the soft palate, helping to close off the nasal cavity and opens the Eustachian tube during swallowing and yawning to regulate the middle ear pressure. 49 Although it is also active in swallowing and phonation, 25 the TVP plays a critical role in respiration. It exhibits inspiratory phasic activity50–53 or constant activity throughout the respiratory cycle. 54 The TVP is thought to stiffen the soft palate and serve as a pharyngeal dilator muscle for maintenance of airway patency.54–56 Electrical stimulation of the TVP reduces airway collapsibility. 56 This study showed that the TVP had about equal proportions of slow type I and fast type II fibers. This muscle also contained more specialized fibers expressing MyHC-ton isoform as compared with the palatal fast muscles such as MU and PP. The specializations of the TVP muscle fibers could allow the muscle to be well suited for generating the tonic muscle tone, steady and sustained contraction, fine movement, and delicate postural adjustments needed in humans to maintain stiffness and patency of the potentially collapsible velopharyngeal airway during respiration and to shape the velum for speech articulation.
The contractile properties of a given muscle can be indicated by its fiber type and MyHC composition. The maximal shortening velocity of the major fiber types in mammalian skeletal muscles is ranked IIX > IIA > I.3,57–60 The hybrid fibers have intermediate contractile properties.57,58,61,62 Muscles with a high proportion of fast type II fibers facilitate rapid, phasic, and powerful movements, whereas those with a high percentage of slow type I fibers are associated with fine movements and tonic activities.63–66 Hybrid fibers may permit the muscle to generate the desired form of contraction and relaxation, which allows highly specific fine movement. MyHC composition and fiber-type distribution suggest that MU and PP could be mainly involved in rapid movements, whereas the LVP, PG, and TVP would be suited to continuous and tonic contractions that are needed for different upper airway motor tasks such as swallowing, speech, and respiration.
Human cranial muscles with multiple functions have complex patterns of MyHC expression.67,68 Soft palate muscles exhibited unique characteristics similar to the laryngeal 16 and pharyngeal18–21 muscles which contain specialized muscle fibers expressing some unusual MyHC isoforms such as MyHC-ton and MyHC-emb that are not found in limb muscles. MyHC-ton-containing fibers exist in some cranial muscles that perform fine movements as required by specific physiological tasks. MyHC-ton fibers differ from most other muscle fibers in that they do not exhibit a twitch contraction but rather have contractions that are prolonged, stable, precisely controlled, and fatigue resistant. 69 Thus, the MyHC-ton containing fibers could play an important role in the regulation of the muscle tone. In the soft palate, the LVP and TVP with predominance of slow type I and MyHC-ton containing fibers which would be well adapted for tonic contraction, highly specific fine movement, and postural adjustments needed to stabilize the soft palate during respiration for maintaining upper airway patency, thus preventing soft palate and lateral pharyngeal walls from collapse with inspiratory negative pressure. MyHC-emb isoform normally expressed in developing muscle fibers and completely substituted by the adult isoforms in limb muscles still persists in some adult specialized cranial muscles including those in the larynx, 70 pharynx,18,19 and soft palate (this study). The muscle fibers expressing developmental MyHC isoforms have a lower shortening velocity compared with adult fast MyHC fibers. 58 As MyHC-emb isoform coexisted with type II MyHCs in the palatal muscles, its presence may expend the contractile velocity range in a fast fiber population for enhancing a more precise control of palatal muscle activity.
In conclusion, documenting the morphometric and histochemical characteristics of the human soft palate muscles is important for better understanding their functions. In addition, the data from this study are useful for further research to determine the alterations in the muscle morphology and fiber type distribution of the palatal muscles induced by either normal aging or some pathological conditions, which may lead to novel therapies for individuals with soft palate-related speech, swallowing, and breathing disorders.
Footnotes
Acknowledgements
We thank the Department of Pathology of Mount Sinai Medical Center for provision of human specimens used in this study. We also thank two anonymous reviewers for their thoughtful and constructive comments on this manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LM contributed to conception and design of research and manuscript writing; JC and JL performed sample sectioning, immunostaining, photographing, and data collection; TN conducted statistical analyses; SS prepared graphs and performed literature search; LM and IS interpreted results; IS, BB, and MC edited and revised manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health Grant 5R01 DC014679 from the National Institute on Deafness and Other Communication Disorders (to L.M.).
