Abstract
The purpose of this study was to compare the immunohistochemical expression of tenascin-C (Tn-C) regarding clinicopathological variables and its association with the clinical behavior of central giant cell lesions (CGCLs). Forty-eight paraffin-embedded samples of CGCLs were selected. Based on clinical and radiographic features, the lesions were classified as aggressive (A-CGCLs) and non-aggressive (NA-CGCLs) subtypes. Histological assessment included the microvessel count (MVC), multinucleated giant cell (MGC) count, and the proportion of tissue area involved by mononuclear stromal cells/interstitial fibrosis. Immunoreactivity, immunolocalization, and distribution patterns of Tn-C were studied immunohistochemically. The association between Tn-C expression and clinicopathological characteristics was analyzed separately and adjusted for confounders using logistic regression models. A significantly greater proportion of cases with moderate-to-intense, intracellular, and diffuse staining of Tn-C was observed in A-CGCLs. CGCLs with a size ≥3.3 cm, fast growth, cortical disruption, high MVC/MGC counts, and low interstitial fibrosis showed a significantly greater frequency of moderate-to-intense, intracellular, and diffuse staining. Logistic regression analysis indicated a strong/independent association of these three immunohistochemical parameters with the aggressiveness of lesions. These data appear to suggest a possible role for Tn-C in the etiopathogenesis of CGCLs of the jaws, where its upregulation might favor the destructive behavior of A-CGCLs.
Introduction
Central giant cell lesions (CGCLs) of the jaws are non-neoplastic proliferative and osteolytic intraosseous disorders of unknown etiology that show a varied and unpredictable biological behavior.1–3 They affect mainly females and patients aged <20 years,4,5 and occur most frequently in the mandible. 6 The radiographic appearance of the lesions is diverse, alternating among small unilocular lesions and large multilocular well-defined radiolucencies with different grades of cortical plate involvement.6,7 Based on clinical and radiographic features, CGCLs have been categorized as aggressive lesions (A-CGCLs), characterized by pain, rapid growth, cortical disruption, and recurrence, and non-aggressive lesions (NA-CGCLs), usually present as asymptomatic, slow-growing, non-perforating, and non-recurring cases.6,8
Histologically, both variants of CGCLs are recognized by the occurrence of multinucleated giant cells (MGCs) intermingled with polygonal and spindle-shaped mononuclear stromal cells (MSCs) distributed in a fibrous and vascular stroma, along with hemosiderin deposits and foci of hemorrhage.6,9 Notwithstanding, it appears that not only the cellularity but also the proportion of fibrous tissue is highly variable across the lesions 10 so that they may range from entirely cellular to a myxoid or to a basically fibrous tissue with scant foci of MGCs. 11 Based on these histological characteristics, many studies have attempted to establish immunohistochemical biomarkers that might predict the clinical behavior of the lesions, but the results have been inconsistent and controversial.
Given that the microenvironment of CGCLs is constituted by a complex extracellular matrix (ECM) and various cell populations, the focus of interest in these osteolytic lesions should be directed toward the evaluation of the role played by the ECM proteins in dissolving the calcified matrix at sites with increased resorptive activity. Nevertheless, although compelling data support that both MGCs and stromal cell populations express different ECM proteins,1,12,13 currently available data on this issue remain limited. Among these proteins, tenascin-C (Tn-C), a hexameric multimodular glycoprotein, has been considered as an ECM modulator that acts as an acute-phase protein in response to mechanical stress. 14 Although it is not expressed in the majority of normal adult tissues, it may be specifically and transiently upregulated during acute inflammation and permanently expressed during chronic inflammation and tumorigenesis.15,16 Although relatively little has been published with regard to the expression of this glycoprotein in CGCLs,12,17 it has been hypothesized that the dysregulation of Tn-C may promote the migration, differentiation, and cellular proliferation in some odontogenic lesions.18,19
Considering that ECM proteins may have a probable role in the initiation, evolution, and destructive behavior of CGCLs, the purpose of this study was to compare the immunohistochemical expression of Tn-C regarding clinicopathological variables, as well as elucidate its association with the clinical behavior of CGCLs of the jaws.
Materials and Methods
Study Cases and Tissue Collection
This laboratory-based study was carried out using the clinical registers and buffered formalin-fixed/paraffin-embedded tissue samples of 48 cases of CGCLs of the jaws collected from the files of the Laboratory of Oral Pathology and Bioanalysis of the Faculty of Dentistry, University of Antioquia. The study was approved by the Institutional Research Ethics Board and was performed in accordance with the Helsinki Declaration. Only cases with complete demographic, clinical, and radiographic registers, as well as blocks with adequate amount of tissue were considered eligible for the analysis. All demographic variables that could have an effect on the immunomarker expression pattern, including gender, age, smoking, and alcohol consumption, were gathered. The cases were characterized by single experienced stomatologist and researcher (S.I.T.-A.) as A-CGCLs if they exhibited at least one of the following criteria: pain, fast growth, cortical disruption, tooth displacement, root resorption, or recurrence. On the contrary, asymptomatic, slow-growing, non-perforating, and non-recurring lesions were considered as NA-CGCLs. 7 Informed consent for laboratory tests, sampling of biomaterial, and use of registers for investigation purposes was obtained from each patient/custodian at the time of the biopsy procedure.
Histological Analysis
Four-µm-thick sections stained with hematoxylin and eosin were assessed by another qualified researcher (G.A.F.-M.) using an image analysis software (Zen Pro; Carl Zeiss, Oberkochen, Germany) fitted to a Zeiss Axiolab microscope (Carl Zeiss). In each section, 10 high-power fields (HPFs; 400× magnification) were examined. After acquisition and digitalization of the images, histological evaluation included the mean of microvessel count (MVC) and MGCs per HPF, as well as the percentage of tissue area involved by MSCs or interstitial fibrosis per HPF. A graticule superimposed on digitized images, dividing the HPFs into 100 equal squares, was used to facilitate the counting. The average count of the 10 HPFs was taken to represent the score for each sample.
Immunohistochemical Technique and Scoring
Immunohistochemical staining was performed on 4-µm-thick sections mounted onto positively charged glass slides. Endogenous peroxidase activity was blocked with 6% solution of hydrogen peroxide for 5 min. Antigen retrieval was carried out by heating slides immersed in citrate buffer (Target Retrieval Solution; DakoCytomation, Carpinteria, CA), pH 6.1, in a steamer for 30 min, and endogenous avidin–biotin activity was quenched with Biotin Blocking System (DakoCytomation) for 20 min. Slides were incubated for 1 hr with 100 µl of rat antihuman Tn-C (MAB3358) monoclonal antibody (R&D Systems; Minneapolis, MN) diluted 1:50, as primary antibody. The standard streptavidin–biotin–peroxidase complex method was performed to bind the primary antibody with the use of a Histostain-Plus Bulk Kit (LAB-SA Detection System; Invitrogen, CA) as broad-spectrum biotinylated secondary antibody. Diaminobenzidine (DakoCytomation) and Harris hematoxylin (Merck; Darmstadt, Germany) were used as chromogen and counterstain materials, respectively. Furthermore, sections of oral pyogenic granuloma served as appropriate positive control samples, 16 whereas negative controls were prepared replacing the primary antibody with antibody diluent (DakoCytomation).
To ascertain objectivity and consistency, Tn-C tissue expression was qualitatively assessed by another researcher (D.M.I.-G.) blinded to the clinical and histological data using the same microscope and image analyzer system at 400× magnification in relation to the whole viewed surface under the following parameters: immunoreactivity (i.e., weak vs moderate-to-intense), immunolocalization (i.e., intracellular vs interstitial), and distribution pattern (i.e., focal vs diffuse).
Statistical Analysis
Statistical analysis was performed using a standard statistical software (IBM SPSS 26.0; Chicago, IL). To facilitate the analysis, continuous variables were dichotomized based on the mean values estimated from all cases regardless of their clinical behavior as the cut-off point. Reproducibility analysis for both histological and immunohistochemical data was assessed by double assessments for each particular examination achieved by each observer with 10 cases randomly selected using random number tables. The interval among the test–retest assessments was 2 months. The Cohen’s kappa statistic (κ) was used to evaluate the reliability between the two sets of estimates. Between-group comparisons were achieved using the Fisher’s exact or the Pearson’s Chi-square tests to detect differences in relation to demographic, clinicopathological, and immunohistochemical findings. Furthermore, immunohistochemical parameters significantly associated with an aggressive behavior identified in the between-group comparisons were considered to be included in univariate and multivariate analyses adjusting for demographic variables with a level of significance of p<0.20 to establish the strength/independence of the associations. Positive associations were regarded as valid when the odds ratio (OR) was >2 and the confidence interval (CI) was >1.0. All tests were two-sided, and p<0.05 indicated statistical significance.
Results
Reproducibility of Histological/Immunohistochemical Analyses
Intraobserver agreement in each series of data recorded per case by the same examiner was excellent for MGC/MSC counts, as well as for the immunoreactivity, immunolocalization, and distribution pattern of Tn-C (κ = 1.000). Also, intraobserver reproducibility was high for MVC and interstitial fibrosis (κ = 0.889 and 0.862, respectively).
Summary of Demographic, Clinical, Histological, and Immunohistochemical Findings
Complete clinical/radiographic registers and biopsy material from 33 cases of A-CGCLs (11 males and 22 females), ranging from 4 to 58 years of age (mean, 18.4 years), and 15 cases of NA-CGCLs (8 males and 7 females), ranging from 7 to 52 years of age (mean, 25.0 years), were included. The mean follow-up time was 3.4 years (ranging from 1 to 8 years). Overall, the lesions varied in size from 1 to 10 cm (mean, 3.3 cm). Fifteen lesions (31.2%) were located in the maxilla and 33 (68.7%) in the mandible. The evolution time of the disease from the onset of symptoms to the moment of clinical evaluation ranged from 2 to 36 months (mean, 13 months). A-CGCLs were recognized by the presence of one or more of the following findings: moderate-to-severe pain/numbness (14 cases), fast growth rate (29 cases), dental root resorption (9 cases), cortical bone disruption (25 cases), tooth displacement with loosening of variable severity (15 cases), and/or recurrence after surgical resection (10 cases). These recurrences, involving eight mandibular and two maxillary sites, were observed after a mean follow-up of 4.2 years (range, 1–8 years). By contrast, although all NA-CGCLs comprised slow-growing, non-perforating, and non-recurring cases, seven long-standing lesions were associated with mild horizontal tooth displacement, and one patient complained of mild tenderness related to traumatic injury of soft tissues.
Between-group comparisons of demographic, histological, and immunohistochemical parameters assessed from the cases are summarized in Table 1. As is evident, although no statistically significant differences between the study groups related to gender, age stratum, smoking habits, or alcohol consumption (all p>0.05, Chi-square) were observed, both gender and age stratum fulfill the condition to be deemed as confounders of the association among immunohistochemical staining results and the disease behavior, as they attained p values <0.20 in bivariate tests. As for as histological assessment, a significantly greater percentage of specimens with high MVC (≥3.1/HPF), high MGC count (≥6.3/HPF), and high MSC density (≥23%/HPF) was observed in A-CGCLs in comparison with those of NA-CGCLs (all p<0.05, Fisher’s exact test). Conversely, NA-CGCLs showed a significantly higher frequency of cases (p<0.001) with high interstitial fibrosis (≥33.7%/HPF).
Bivariate Comparisons of Demographic, Histological, and Immunohistochemical Parameters According to Diagnosis Category.
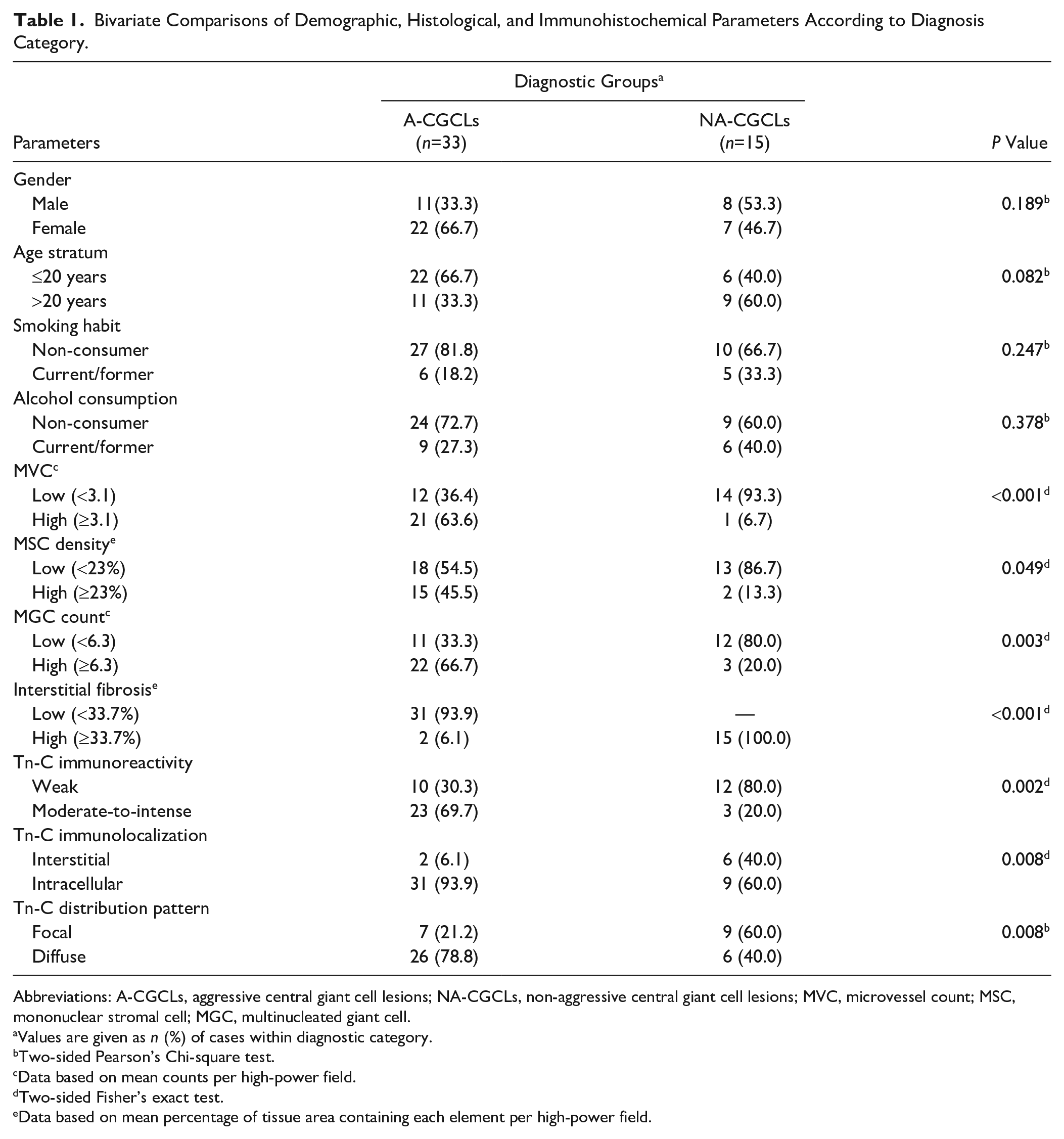
Abbreviations: A-CGCLs, aggressive central giant cell lesions; NA-CGCLs, non-aggressive central giant cell lesions; MVC, microvessel count; MSC, mononuclear stromal cell; MGC, multinucleated giant cell.
Values are given as n (%) of cases within diagnostic category.
Two-sided Pearson’s Chi-square test.
Data based on mean counts per high-power field.
Two-sided Fisher’s exact test.
Data based on mean percentage of tissue area containing each element per high-power field.
Immunohistochemical analysis revealed heterogeneous sample-to-sample variations in the immunoexpression profile of Tn-C. Whereas negative controls showed no labeling expression in the studied samples (Fig. 1A), positive controls disclosed intense reactivity for the biomarker, mainly present in connective tissue adjacent to the epithelium, but also extending into stromal connective tissue, with a diffuse, reticular, and interstitial pattern (Fig. 1B). With regard to CGCLs, immunohistochemical staining confirmed the presence of Tn-C within the lesional micromilieu in all the samples. Irrespective of the clinical behavior, its immunoreactivity was weak in 22 samples (45.83%) and moderate-to-intense in 26 samples (54.17%). Although 8 (16.7%) out of 48 cases showed interstitial positivity, usually expressed as extracellular linear and reticular fibers of variable thickness and intensity (Fig. 1C), 40 cases (83.3%) showed heterogeneous intracytoplasmic immunoreactivity in MGCs and scant polygonal MSCs. Endothelial and spindle-shaped cells were unreactive (Fig. 1D). As to distribution of the reaction, 16 (33.3%) out of 48 cases demonstrated variable labeling with focal stained stromal or cellular areas intermingled within unstained zones, whereas in 32 cases (66.7%) a diffuse staining was seen throughout lesional tissue. It was noteworthy that the cases with moderate-to-intense, intracellular, and diffuse staining were significantly more frequent in A-CGCLs (all p<0.01, Fisher’s exact or Chi-square comparison tests) than those in non-aggressive subtypes of the lesions.
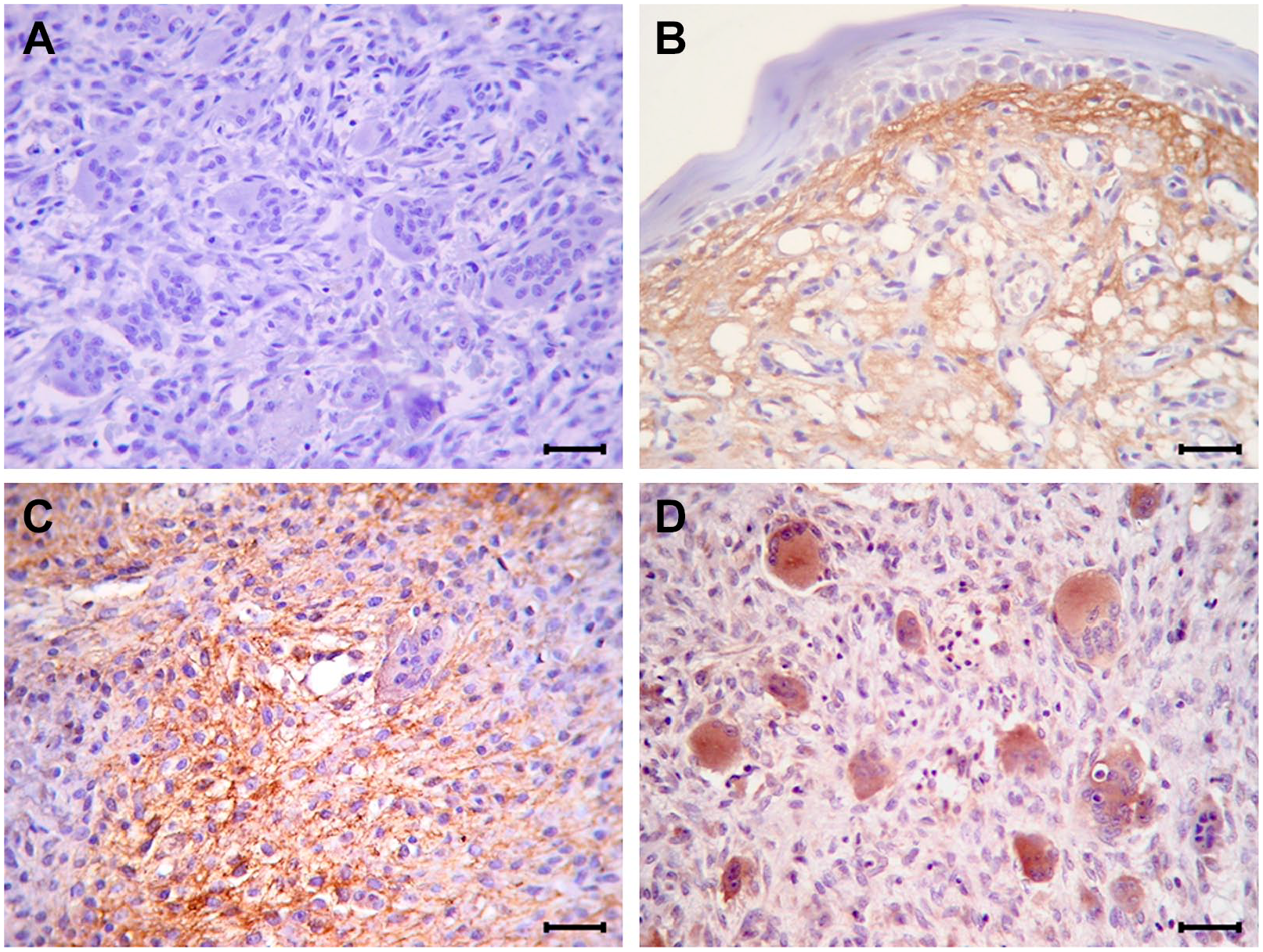
Representative photomicrographs of the immunohistochemical reactivity for Tn-C in tissue sections (streptavidin–biotin–peroxidase complex, hematoxylin counterstain). (A) Negative control showing no staining. (B) Positive control that shows a band of intense Tn-C immunoreactivity in the subepithelial connective tissue and Tn-C positive fibers arranged in a loose reticular pattern projecting into stromal connective tissue of oral pyogenic granuloma. (C) NA-CGCL showing a focal intense interstitial expression of Tn-C distributed in a reticular/fibrillar pattern. (D) A-CGCL with diffuse and moderate-to-intense cytoplasmic staining in MGCs and in some polygonal MSCs. Original magnification: 400×; bars: 20 µm. Abbreviations: Tn-C, tenascin-C; NA-CGCL, non-aggressive central giant cell lesion; A-CGCL, aggressive central giant cell lesion; MGC, multinucleated giant cell.
Comparison Between Tn-C Expression and Clinicopathological Characteristics
Bivariate comparisons for the immunohistochemical expression of Tn-C according to dichotomized clinicopathological characteristics of CGCLs irrespective of diagnostic categories are outlined in Table 2. Although no significant differences (p>0.05, Fisher’s exact and Chi-square tests) were detected in the immunoreactivity, immunolocalization, and distribution pattern of Tn-C in regard to location of the lesions, presence of symptoms, evolution time of disease, root resorption, tooth displacement, recurrence, or MSC density, those CGCLs with a size ≥3.3 cm, fast growth rate, presence of cortical disruption, high MVC, high MGC count, and low interstitial fibrosis presented a significantly greater proportion (p<0.05) of moderate-to-intense, intracellular, and diffuse staining.
Comparison of Immunohistochemical Findings According to Dichotomized Clinicopathological Characteristics of CGCLs of the Jaws Regardless of the Biological Behavior.
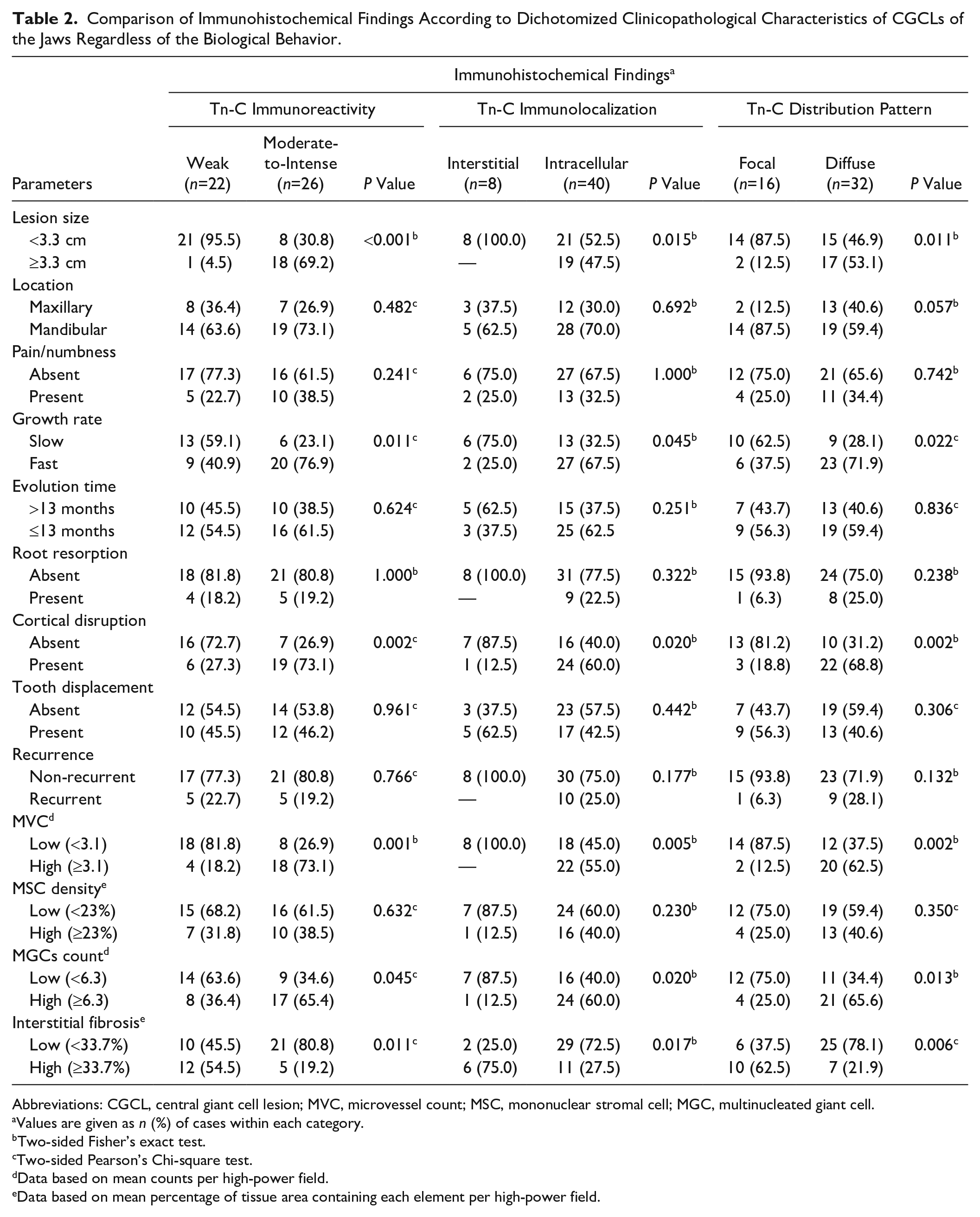
Abbreviations: CGCL, central giant cell lesion; MVC, microvessel count; MSC, mononuclear stromal cell; MGC, multinucleated giant cell.
Values are given as n (%) of cases within each category.
Two-sided Fisher’s exact test.
Two-sided Pearson’s Chi-square test.
Data based on mean counts per high-power field.
Data based on mean percentage of tissue area containing each element per high-power field.
Outcomes From Multivariate Binary Logistic Regression Analysis
The outcomes of univariate and multivariate binary logistic regression analyses for association of immunohistochemical variables with A-CGCLs are outlined in Table 3. It can be noted that the OR for A-CGCLs was significantly increased (p<0.05, Wald’s test) for those cases with moderate-to-intense, intracellular, and diffuse staining. After adjusting for gender and age stratum, all these three parameters continued strongly and independently associated with the aggressiveness of the lesions (p<0.05). In addition, the Hosmer–Lemeshow goodness-of-fit test probability values varied from 0.208 to 0.809, thus indicating adequate calibration and fit of the multivariable models. Similarly, the c-statistic estimation, closer to 1.0 for all the three scores in the adjusted models, indicated a great discriminative power.
Summary of Univariate and Multivariate Binary Logistic Regression Analyses for the Association of TN-C Expression With A-CGCLs Adjusted for Gender and Age Stratum.

Abbreviations: TN-C, tenascin-C; A-CGCLs, aggressive central giant cell lesions; OR, odds ratio; CI, confidence interval.
Hosmer–Lemeshow goodness-of-fit test.
c-statistic value.
Odds ratio (95% confidence interval).
Wald test.
Discussion
Although a great number of histomorphometrical, histochemical, immunohistochemical, genetic, and ultrastructural studies have focused on the analysis of different variables that might be associated with the development of CGCLs of the jaws and have led to hypotheses as the causes of these osteolytic processes, its pathogenesis and pathophysiology remain poorly understood. Moreover, although there is a continuous search for specific biomarkers that allow differentiating among aggressive and non-aggressive lesions, the prediction of its complex clinical behavior remains challenging using the currently available histopathological and molecular markers. Assuming that the relevant role played by the ECM components in tissue injury and tumorigenesis may be not only of mechanical but also of signaling nature, 16 this study proposed to explore whether Tn-C overexpression possesses some predictive value concerning ongoing disease behavior. Accordingly, the current report included a representative number of cases with different clinical presentations throughout the range of aggressiveness of CGCLs. It could be noted that whereas aggressive and non-aggressive subtypes were statistically similar in regard to gender, age stratum, smoking habit, and alcohol consumption, parameters such as gender and participant’s age met the preset conditions to be considered as confounders for the association among immunohistochemical outcomes and A-CGCLs in the final analysis models. In addition, consistent with the findings of earlier studies regarding the relationship of histological changes associated with CGCLs,20–22 the data presented herein confirmed that an increased MVC, a higher MSC density, and a greater MGC count, as well as lower interstitial fibrosis, are also important variables linked to the aggressiveness of lesions that might reflect the cumulative effect of immunological and/or non-immunological stressors affecting tissue homeostasis.
It has been acknowledged that Tn-C prevents cell adhesion,16,23 promotes cell migration, 24 and increases cell proliferation, 23 through the interaction with several ECM molecules and cell-surface receptors, therefore affecting tissue architecture, tissue resilience, and cell responses by means of induction of proinflammatory cytokines and oncogenic signaling molecules. 16 In agreeance with the former, this study showed that the proportion of samples with moderate-to-intense, intracellular, and diffuse staining were significantly higher in A-CGCLs than those in NA-CGCLs. Taking into account that all specimens were treated in the same way, differences in staining intensity and in its spatial distribution may be a reflection, on the one hand, of alterations in the absolute concentrations of the protein present within the lesional microenvironment but, on the other hand, due to differences in the sample composition, as although the usual frequency reported for A-CGCLs has been lower than that of NA-CGCLs,3,7,22 in this study a greater number of cases were typified as having an aggressive behavior, which could have had a profound influence on the immunohistochemical findings. In addition, a possible explanation for dissimilarities in the marker’s immunolocalization might be linked to the existence of several isoforms of Tn-C and posttranscription glycosylation.25,26 In this line, it has been stated that Tn-C splicing patterns appear to be related to cell adhesion modulation so that whereas some isoforms can play a structural and adhesive role, others can promote cell dissemination.26,27 Unlike this study, where both intracytoplasmic and interstitial Tn-C were reported for the samples, other studies found that the immunostaining was completely stromal in CGCLs, with no evidence of intracellular positive reaction.12,17 The reasons for these conflicting results could be due in part to the sample size and composition, or technical factors, including different antibodies and/or immunohistochemical processing techniques used. Even so, the intracytoplasmic location of Tn-C has been described in pleomorphic adenomas and carcinoma ex pleomorphic adenomas, 26 glioblastomas and oligodendrogliomas, 28 idiopathic interstitial lung disease, 29 and recurrent intracranial ependymomas, 30 as well as in medullary thyroid microcarcinomas and C-cell hyperplasia. 31 It has been further proposed that, as Tn-C is mainly synthetized in mesenchymal cells, a dysregulated production and/or secretion mechanism may be a probable reason for its intracytoplasmic accumulation. 26
Whereas nothing is known about the association of Tn-C immunoexpression with clinicopathological features in CGCLs of the jaws, some investigations conducted on malignant lesions have demonstrated that the Tn-C content might be linked to characteristics of local invasiveness and disease progression.26,27,32,33 In this study, although no significant differences were observed between the differentially expressed Tn-C protein and clinicopathological parameters such as location, symptoms, evolution time of disease, root resorption, tooth displacement, recurrence, and MSC density, it was striking that those lesions with a size ≥3.3 cm, fast growth rate, cortical disruption, high MVC, high MGC count, and low interstitial fibrosis showed a significantly greater proportion of moderate-to-intense, intracellular, and diffuse staining. Altogether, these findings suggest that the upregulation of Tn-C might be a key factor in the behavior of CGCLs acting in multiple processes such as the proliferation of MGCs, early stromal remodeling, lesion growth, angiogenesis, potentiation of proinflammatory cytokines, and the inhibition of immunosurveillance.16,31
It is also necessary to underline that this study established a strong/independent association of Tn-C expression in terms of intensity, intracytoplasmic location, and diffuse distribution of immunostaining, as robust indicators for CGCLs aggressiveness. Considering not only that Tn-C functions as an adhesion modulatory ECM protein through the antagonization of the adhesive effects of fibronectin and competition with syndecan-4, the blocking of the ability of fibroblasts to deposit and contract the matrix, and the alteration of the morphology and signaling pathways of adherent cells, 23 but also that the association was essentially based on the higher count of immunoreactive MGCs, the present results indicate that Tn-C might promote the proliferation and stimulation of MGCs within the lesional tissue mediated by cell-to-cell and cell–ECM interactions, thereby leading to various grades of aggressiveness of CGCLs of the jaws.
As a final point, based on different immunohistochemical findings, several therapeutic alternatives, including the use of subcutaneous/nasal application of calcitonin, subcutaneous injection of interferon-2α, intralesional corticosteroid injections, bisphosphonates, and denosumab, have been suggested with variable outcomes to reduce the size of the lesions and minimize the functional and physical sequelae resulting from the surgical resection of CGCLs. 34 Considering that some Tn-C isoforms might be expressed in CGCLs of the jaws, functional studies will be of importance in predicting prognosis and in tailoring specific therapies based on radioactively labeled antibodies against Tn-C as it has been previously proposed for specific solid tumors.27,35,36
In conclusion, these data appear to suggest a possible role for Tn-C in the etiopathogenesis of CGCLs of the jaws, where its upregulation might favor the destructive behavior of A-CGCLs.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SIT-A is the main researcher who contributed to this work, including the conceptualization and design of the study, acquisition of clinical and radiographic data, the analysis and interpretation of data, and writing of original draft. DMI-G participated in its design, carried out the immunohistochemical procedures, and contributed to acquisition and analysis of data and to manuscript preparation. GAF-M contributed to the histological processing/assessment of the samples and critically evaluated and supplemented the manuscript. All the above authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Technical Research Council of the Faculty of Dentistry, University of Antioquia (Code 2020-27931).
