Abstract
Rat limbal niche cells (LNCs) have been proven to induce transdifferentiation of oral mucosal epithelial cells (OMECs) into corneal epithelial-like cells termed transdifferentiated oral mucosal epithelial cells (T-OMECs). This investigation aimed to evaluate the effect of subconjunctival T-OMEC injections on alkali-induced limbal stem cell deficiency (LSCD) in rats. LNCs were cocultured with OMECs in the Transwell system to obtain T-OMECs, with NIH-3T3 cells serving as a control. Subconjunctival injection of single T-OMEC or OMEC suspension was performed immediately after corneal alkali injury. T-OMECs were prelabeled with the fluorescent dye CM-DiI in vitro and tracked in vivo. Corneal epithelial defect, opacity, and neovascularization were quantitatively analyzed. The degree of corneal epithelial defect (from day 1 onward), opacity (from day 5 onward), and neovascularization (from day 2 onward) was significantly less in the T-OMEC group than in the OMEC group. Cytokeratin 12 (CK12), pigment epithelium–derived factor, and soluble fms-like tyrosine kinase-1 were expressed at a higher rate following T-OMEC injection. Some CM-DiI-labeled cells were found to be coexpressed with CK12, Pax6, and ΔNp63α in the corneal epithelium after subconjunctival injection. Subconjunctival injection of T-OMECs prevents conjunctival invasion and maintains a normal corneal phenotype, which might be a novel strategy in the treatment of LSCD:
Keywords
Introduction
Limbal stem cell deficiency (LSCD), a severe blinding keratopathy, is a significant threat to the visual acuity of affected patients. 1 Some commonly implicated etiopathogenic factors for LSCD include chemical/thermal burns, Stevens–Johnson syndrome, ocular cicatricial pemphigoid, Sjogren’s syndrome, and other chronic ocular surface inflammatory conditions. 2 Limbal epithelial stem/progenitor cells (LESCs) are essential for the replacement of corneal cellular loss when under traumatic wounding. 3 However, LESCs and their limbal niche are irrevocably damaged in LSCD, 4 resulting in recurrent epithelial defects, corneal neovascularization, and invasion of conjunctival epithelium. 5 The current therapeutic strategies for LSCD involve cultivated limbal epithelial transplantation (CLET) and transplantation of small pieces of limbal tissue, termed simple limbal epithelial transplantation (SLET).6,7 Both CLET and SLET have shown promising results in unilateral LSCD patients after transplantation of limbal epithelium obtained from the contralateral healthy eye.8,9 However, in bilateral LSCD patients, the outcomes were less impressive on account of the severe immune rejection after allogeneic limbal transplantation. 10
Oral mucosal epithelial cells (OMECs) are a potential resource to restore LESCs because OMECs and corneal epithelial cells are derived from the same ectoderm. 11 For decades, cultivated OMECs have shown feasibility in ocular surface reconstruction.12–14 However, cultivated autologous oral mucosal transplantation (COMET) is restricted by peripheral corneal neovascularization after transplantation.15,16 In our previous studies, OMECs were transdifferentiated into corneal epithelial-like cells by coculturing with limbal niche cells (LNCs) in the Transwell system. 17 Compared with OMECs, transdifferentiated oral mucosal epithelial cells (T-OMECs) showed higher expression of antiangiogenesis factors and were less likely to stimulate neovascularization after transplantation.18,19
Clinically, the transplantation of cultivated oral mucosal graft faces extreme challenges in terms of requirements of the techniques used in cell cultivation, surgical manipulations of an ophthalmologist, and its overall expenses.20,21 Moreover, the ideal culture substrate for cultivated oral mucosal graft has remained uncertain. 22 Compared with graft transplantation, subconjunctival injection of cells is a convenient route and is shown to have some effect in treating LSCD. 23 Therefore, we speculate that subconjunctival injection of T-OMECs might be a feasible strategy in the treatment of LSCD. This investigation was conducted to evaluate the therapeutic effect of subconjunctival T-OMEC injections on rat LSCD models.
Materials and Methods
Animals
Eight-week-old male Sprague Dawley rats (weighing 150–180 g) were provided by the Animal Research Committee of the Huazhong University of Science and Technology (Wuhan, China). This study was approved by the Institutional Animal Care and Use Committee at Tongji Medical College, Huazhong University of Science and Technology (IACUC number S2281, 2019).
Cell Isolation and Culturing
The isolation and culturing of LNCs were reported previously. 24 Rats were anesthetized by intraperitoneal injection of 1% pentobarbital sodium (40 mg/kg) and then sacrificed by cervical dislocation. After enucleation, a 3-mm-diameter trephine was used to separate the central cornea and limbus. The limbus was cut into six small sections, digested with 1 mg/mL collagenase A at 37C for 4 hr, and incubated in 0.25% trypsin and 1 mM ethylenediaminetetraacetic acid (T/E) at 37C for 15 min to yield single cells. The cells were then seeded at a density of 1 × 104/cm2 into a six-well plate coated with 5% Matrigel and cultured in modified embryonic stem cell medium composed of Dulbecco’s modified Eagle medium/nutrient mixture F-12 (DMEM/F12) supplemented with 10% knockout serum, 5 μg/mL insulin, 5 μg/mL transferrin, 5 ng/mL sodium selenite, 4 ng/mL basic fibroblast growth factor (bFGF), 10 ng/mL human leukemia inhibitory factor (hLIF), 50 μg/mL gentamicin, and 1.25 μg/mL amphotericin B. These cells were passaged at a ratio of 1:4 until passage 4 (P4), and the medium was changed every 2 days.
The isolation of OMECs was the same as described previously. 25 The epithelial layer of rat buccal oral mucosa was separated from the connective tissue by submucosal saline injection, dissected using ophthalmic scissors, digested with 10 mg/mL dispase II at 37C for 40 min to remove excess submucosal connective tissue, and incubated in T/E at 37C for 15 min to obtain single cells. OMECs were then cocultured with NIH-3T3 cells (ATCC; Manassas, VA) in the Transwell system. NIH-3T3 cells were first treated with 4 μg/mL mitomycin C at 37C for 2 hr to restrain growth as soon as they reached confluence in a six-well plate. After that, single OMECs were seeded into the Transwell insert coated with 5% Matrigel at a density of 5 × 104/cm2, with NIH-3T3 cells serving as feeder layers. Supplemented hormonal epithelial medium (SHEM) consisting of DMEM/F12 (1:1) supplemented with 10% fetal bovine serum, 5 μg/mL insulin, 5 μg/mL transferrin, 5 ng/mL sodium selenite, 0.45 μg/mL hydrocortisone, 20 ng/mL human epidermal growth factor (hEGF), 10 ng/mL hLIF, 50 μg/mL gentamicin, and 1.25 μg/mL amphotericin B was added to the Transwell insert (1.5 mL) and the bottom plate (2.6 mL), respectively. Stratified epithelial sheets were obtained by lowering SHEM (1.5 mL) to the bottom of the insert and culturing at the air–fluid interface, termed air-lifting. The culture medium was changed every 2 days.
Our group reported the culture of T-OMECs. 17 T-OMECs were obtained by coculturing primary OMECs with LNCs in the Transwell system. Similarly, P4 LNCs were treated with 4 μg/mL mitomycin C for 2 hr before OMECs were inoculated at a density of 5 × 104/cm2 on the insert and cultured in SHEM. Stratified epithelium of T-OMECs was formed by air-lifting culture. Immunofluorescence staining of cytokeratin 12 (CK12; specific corneal epithelial marker 26 ), Pax6 (master regulator of the eye and corneal epithelium 27 ), and ΔNp63α (epithelial stem cell marker 28 ) was conducted to verify the corneal-like transdifferentiation in T-OMECs. All materials used in cell isolation and culturing are included in Supplementary Table S1.
Induction of Rat LSCD Model
Rats were first anesthetized by intraperitoneal injection of pentobarbital sodium, and then their right eyes were topically anesthetized with 4 mg/mL of oxybuprocaine (Santen Pharmaceutical Co., Ltd; Osaka, Japan). Annular filter papers (outer diameter of 8 mm, inner diameter of 3 mm) were saturated in 1 M sodium hydroxide for 20 sec and applied to the peripheral cornea and limbus for 40 sec. Then, the alkali-burned cornea was immediately rinsed with 60 mL saline for 2 min. Levofloxacin (Santen Pharmaceutical Co., Ltd) was applied to the eyes 3× a day to prevent potential bacterial infections after alkali burn.
Assessment of Ocular Surface
The rat corneas with alkali burn were observed on days 0–7, 14, 21, and 28, respectively. Tropicamide (Shenyang Sinqi; Liaoning, PRC) was used to dilate the pupils to prevent the interference of iris blood vessels. Corneal fluorescein staining, opacity, and neovascularization were recorded.
After 28 days, the rats were sacrificed, and the right eyeballs were removed. The corneas were separated from the outer 2 mm limbal zone, embedded in paraffin, and then stained with hematoxylin and eosin (H&E), periodic acid–Schiff (PAS), mucin 5AC (MUC5AC; a marker for goblet cells in conjunctival epithelium 29 ), α-smooth muscle actin (α-SMA; a marker for vascular smooth muscle cells 30 ), CD34 (a marker for vascular endothelial cells 31 ), CK12, and cytokeratin 13 (CK13; a mucosal marker for conjunctival epithelium and oral mucosal epithelium 32 ) antibodies for immunofluorescence. The corneas from the left eyes were used as controls.
Subconjunctival Injection of T-OMECs/OMECs
Eight rats were randomly divided into T-OMEC group and OMEC group, with four rats in each group. The stratified OMECs cocultured with LNCs or NIH-3T3 cells in the Transwell system were digested in dispase II at 37C for 4 hr to dissolve the Matrigel and incubated in T/E at 37C for 15 min to yield single cells. After regulating the cell concentration to 1 × 106/mL, 50 μl of suspended T-OMECs or equivalent OMECs was injected subconjunctivally at the 12 o’clock and 6 o’clock positions about 2 mm away from the limbus immediately after corneal alkali injury. The epithelial defect area, corneal opacity score, length and area of new blood vessels, immunofluorescence, and Western blot were compared between the T-OMEC (treated) group and OMEC (control) group. The formula for calculating the area of corneal new blood vessels 33 and the criterion of corneal opacity score 34 are listed in Supplementary Tables S2 and S3.
In Vivo Tracking of T-OMECs
T-OMECs cultured in Transwell insert were labeled with CellTracker CM-DiI (cell membrane red fluorescent probe) by incubating in the dark at 37C for 5 min and then at 4C for 15 min. CM-DiI solution of 5 and 10 μM were compared in terms of the capacity of fluorescent labeling. The positive labeling rate in 10 μM was significantly higher than that in 5μM (17.7 ± 5.9% for 5 μM, 85.9 ± 4.7% for 10 μM, p<0.01, n=4; Supplementary Fig. S1). Therefore, T-OMECs labeled with 10 μM CM-DiI were injected under the conjunctiva and tracked in vivo. The corneas were ultimately harvested for immunofluorescence of CK12, CK13, Pax6, and ΔNp63α.
Western Blot
Protein concentration was determined by the bicinchoninic acid assay (Beyotime; Shanghai, China). The samples were lysed by radioimmunoprecipitation assay buffer. The proteins were then extracted, separated by SDS-PAGE electrophoresis, transferred to polyvinylidene difluoride membranes, blocked with 5% (w/v) skim milk, and incubated with specific antibodies and their respective secondary antibodies. β-actin was used as an internal reference. Immunoreactive band detection was performed by an enhanced chemiluminescence kit (ECL; PerkinElmer, Waltham, MA), and the film was scanned by BandScan to analyze the gray values.
H&E Staining
The samples were stained with hematoxylin for 8 min, washed thrice, soaked in 1% acid and 1% ammonia, rinsed with distilled water, and dehydrated with 70% and 90% alcohol for 10 min. The sections were then stained in acidic eosin solution for 2–3 min, dried, and observed under a microscope.
PAS Staining
The sections were first treated with the periodic acid solution for 10 min, rinsed with distilled water, soaked in Schiff solution for 10 min, counterstained with hematoxylin for 3 min, dehydrated, sealed, and observed under a microscope. The goblet cells were stained red.
Immunofluorescence Staining
The rat corneas were first fixed with 4% paraformaldehyde for 60 min at room temperature and then embedded in paraffin for sectioning. The paraffin sections were deparaffinized with xylene, rehydrated, and performed for antigen retrieval by incubation in 0.01 M citrate (pH = 6.0) at 95C for 20 min and blocking with 10% goat serum for 60 min at room temperature. The specimens were incubated with primary and corresponding secondary antibodies and finally stained with 4´,6-diamidino-2-phenylindole (DAPI). Fluorescence images were obtained by a laser confocal fluorescence microscope (LSM 700; Carl Zeiss, Thornwood, NY). All antibodies used for Western blot and immunofluorescent staining are included in Supplementary Table S4.
Statistics
All data are shown as mean ± SD. Unpaired Student’s t-test was used to compare the differences between the treated and control groups. The test results used two-tailed p values, and the differences were considered statistically significant at p<0.05.
Results
Transdifferentiation of OMECs Into Corneal Epithelial-like Cells
To verify the transdifferentiation of OMECs into corneal epithelial-like cells, we identified the molecular phenotypes of the central cornea, limbus, oral mucosa, and stratified T-OMECs cultured in Transwell insert before transplantation. Anatomically, Pax6 and CK12 were expressed in the central corneal and limbal epithelium. CK12 and CK13 were coexpressed in the transitional zone of cornea and conjunctiva, and CK13 was expressed in the epithelial layer outside the limbus but not in the central cornea. Vimentin was stained in the stroma of central cornea and limbus. Furthermore, ΔNp63α was expressed in the basal epithelial layer of limbus rather than in the central cornea (Fig. 1A). In contrast, the oral mucosal epithelium did not express Pax6 and CK12 but showed strong immunoreactivity for CK13. And ΔNp63α was expressed in the basal layer of oral mucosal epithelium. After coculturing with LNCs, T-OMECs showed expression of Pax6 and CK12. Some T-OMECs were CK12-positive (yellow arrow), whereas some were still negative (pink arrow). The expression of CK13 was downregulated in T-OMECs. Besides, ΔNp63α was expressed in the basal layer of stratified T-OMECs (Fig. 1B). Western blotting confirmed the upregulation of CK12. Meanwhile, antiangiogenic factors pigment epithelium–derived factor (PEDF) and soluble fms-like tyrosine kinase-1 (sFlt-1) were expressed significantly more in T-OMECs compared with OMECs (Fig. 1C). Based on the above results, the successful transdifferentiation of OMECs into corneal epithelial-like cells with enhanced expression of antiangiogenic factors was confirmed.
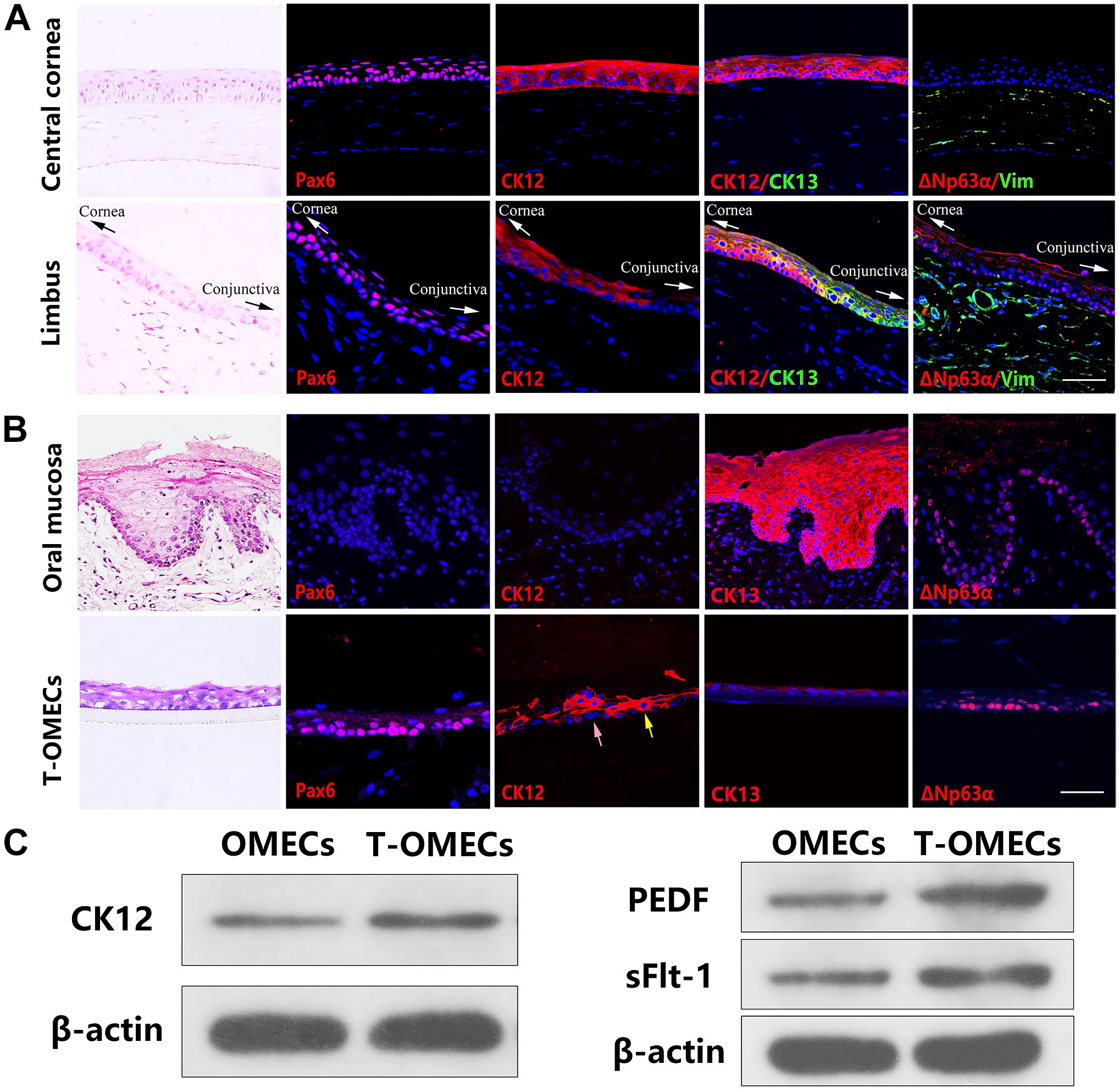
Transdifferentiation of oral mucosal epithelial cells into corneal epithelial-like cells. H&E and immunofluorescent staining of Pax6, CK12, CK13, ΔNp63α, and vimentin (Vim) in central cornea and limbus (A). H&E and immunofluorescent staining of Pax6, CK12, CK13, and ΔNp63α in oral mucosa and stratified T-OMECs cultured in the Transwell system before transplantation. After coculturing, some T-OMECs expressed CK12 (yellow arrow), and some were still negative (pink arrow, B). Western blot of CK12, PEDF, and sFlt-1 in OMECs and T-OMECs; β-actin was used as a loading control (C). Scale bars: 50 μm. Abbreviations: CK12, cytokeratin 12; CK13, cytokeratin 13; H&E, hematoxylin and eosin; OMECs, oral mucosal epithelial cells; PEDF, pigment epithelium-derived factor; sFlt-1, soluble fms-like tyrosine kinase-1; T-OMECs, transdifferentiated oral mucosal epithelial cells.
Expression of MUC5AC, α-SMA, CD34, and CK13 in the LSCD Model
To identify the LSCD model, we observed the rat corneas for 28 days after alkali burn. General observation showed evident neovascularization, conjunctivalization, corneal opacity, and persistent epithelial defects in the rat corneas. On day 0, the fluorescein-stained area covered the limbus. On day 7, peripheral corneal neovascularization, corneal opacity, and a huge area of fluorescein staining were observed. By day 28, the corneas were covered by conjunctiva and neovascular membrane (Fig. 2A).
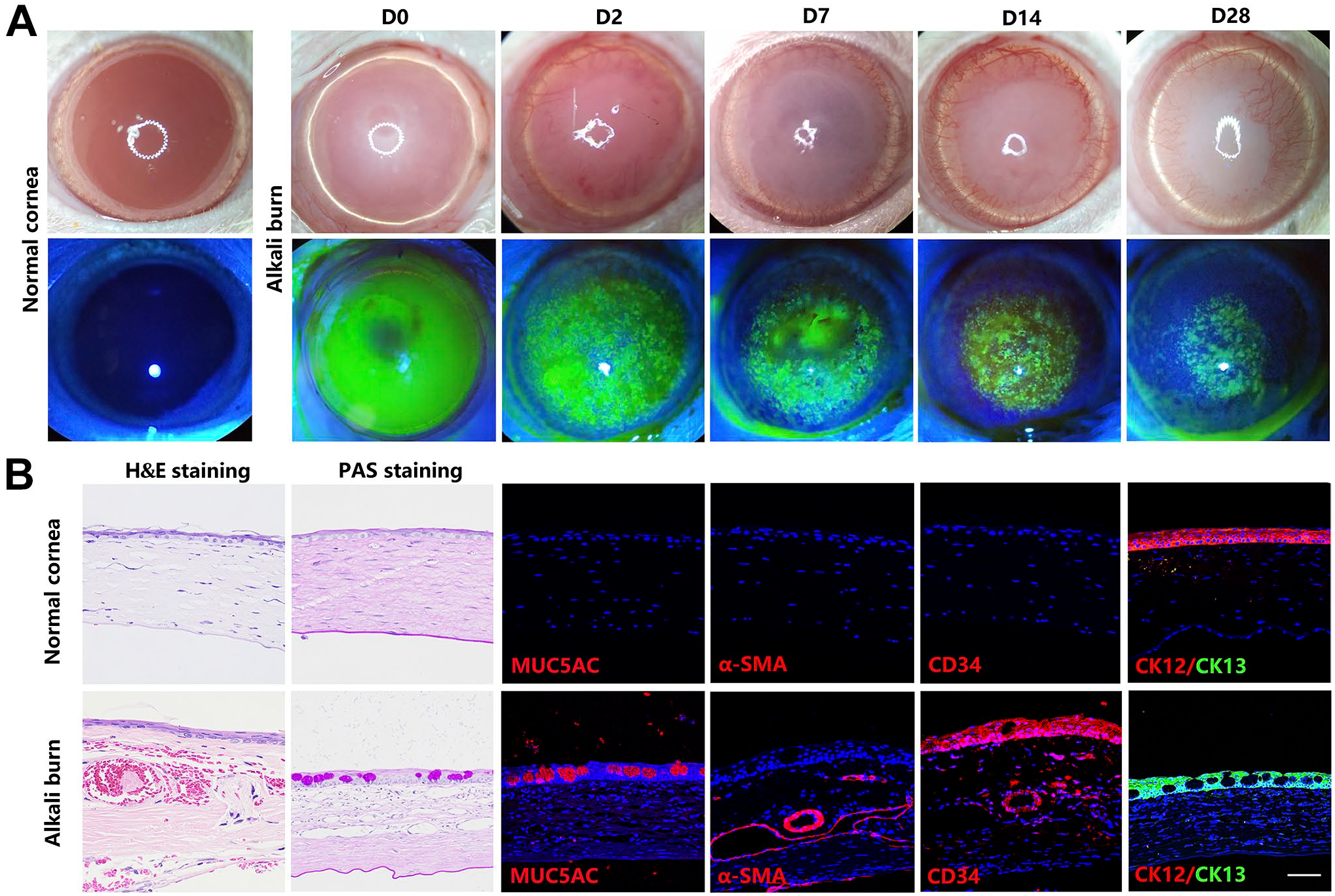
Identification of the rat LSCD model. Light and fluorescein staining of normal and alkali-burned corneas (A). H&E staining, PAS staining, and immunofluorescence staining of MUC5AC, α-SMA, CD34, CK12, and CK13 of normal and alkali-burned corneas (B). Scale bar: 50 μm. Abbreviations: α-SMA, α-smooth muscle actin; CK12, cytokeratin 12; CK13, cytokeratin 13; H&E, hematoxylin and eosin; LSCD, limbal stem cell deficiency; MUC5AC, mucin 5AC; PAS, periodic acid–Schiff.
The staining of the alkali-burned cornea showed neovascular tissues in the corneal stroma by H&E staining, goblet cells in the original corneal epithelium by PAS staining, and strong immunoreactivity for MUC5AC, α-SMA, and CD34 by immunofluorescence. The normal corneal epithelium expressed CK12 but not CK13, whereas the alkali-burned cornea showed negative CK12 and positive CK13 expression (Fig. 2B). Collectively, the expression of MUC5AC, α-SMA, CD34, and CK13 in the alkali-induced LSCD model indicated corneal conjunctivalization and neovascularization.
Subconjunctival Injection of T-OMECs Accelerated Corneal Epithelial Wound Healing
T-OMECs or OMECs were injected under the conjunctiva to compare their therapeutic effect on rat LSCD models. On day 0, both T-OMEC and OMEC groups showed typical limbal epithelial defects after alkali burn. On day 5, the T-OMEC group was stained with a significantly smaller area by fluorescein than the OMEC group. On day 7, only a small area of corneal defects was observed in the T-OMEC group, whereas large flaky defects were present in the OMEC group. By day 28, the corneal epithelium had recovered entirely in the T-OMEC group, compared with the huge defective area in the OMEC group (Fig. 3A and B). The difference in corneal epithelial defect between the two groups appeared from day 1 onward (p=0.04173 on day 1, p=0.04994 on day 2, p=0.02408 on day 3, p=0.01648 on day 4, p=0.01921 on day 5, p=0.00568 on day 6, p=0.00113 on day 7, p=0.00801 on day 14, p=0.00209 on day 21, and p=0.00147 on day 28, n=4), with 0.00 ± 0.00% in the T-OMEC group vs 25.74 ± 4.54% in the OMEC group on day 28 (Fig. 3C). The difference in corneal opacity score between the groups was evident from day 5 to day 28 (p=0.01628 on day 5, p=0.03278 on day 6, p=0.03278 on day 7, p=0.01038 on day 14, p=0.00331 on day 21, and p=0.00032 on day 28, n=4; Fig. 3D). Based on these data, we concluded that subconjunctival injection of T-OMECs accelerated corneal epithelial wound healing.
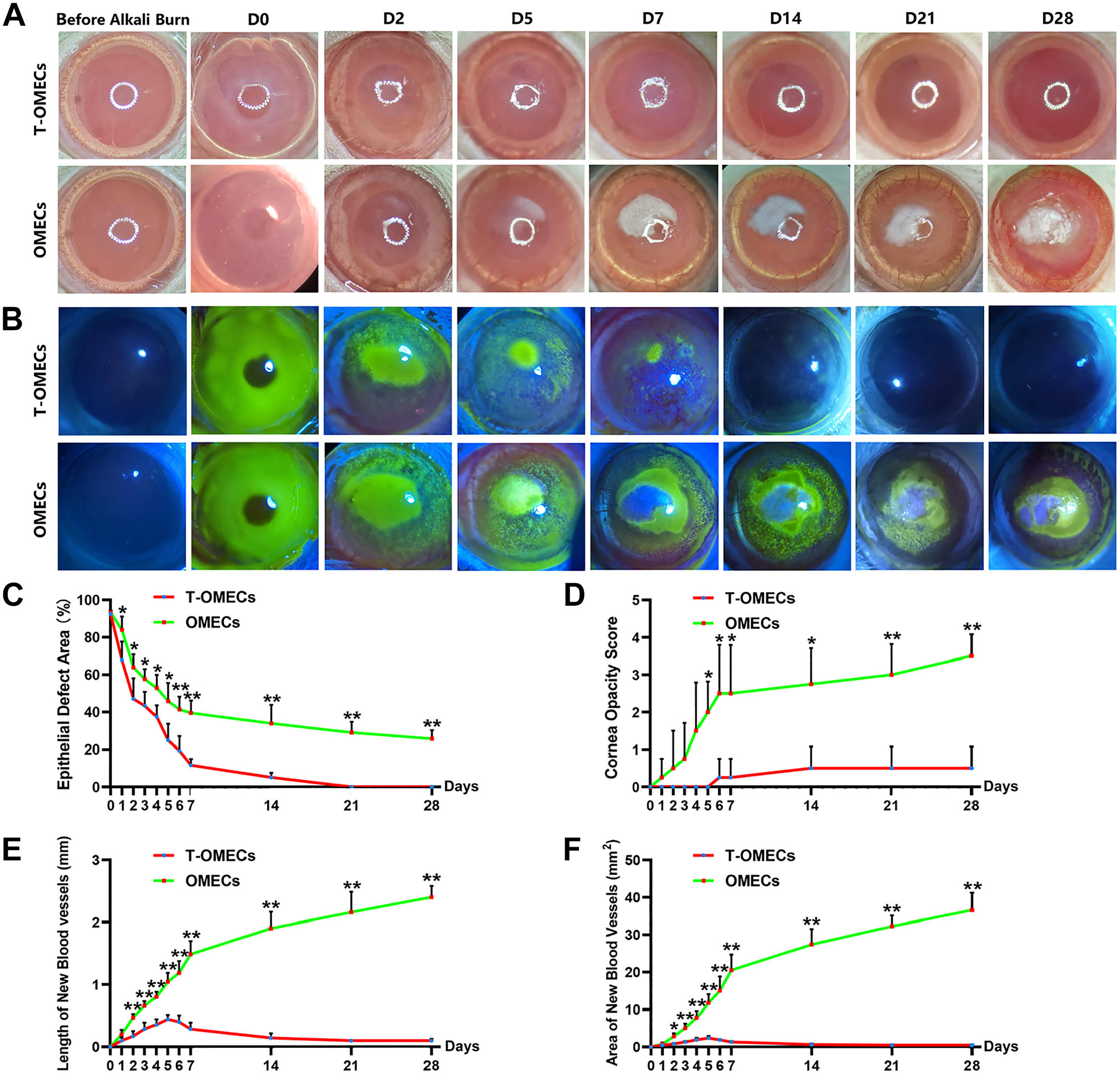
General observation of rat corneas following subconjunctival injection of T-OMECs/OMECs. Light and fluorescein staining of rat corneas in the T-OMEC and OMEC groups (pictures showed the same animal in each group, A and B). The variation tendency of epithelial defect area (%) (C), cornea opacity score (D), length of new blood vessels (E), and area of new blood vessels (F) in the T-OMEC and OMEC groups. *p<0.05, **p<0.01, n=4. Abbreviations: OMECs, oral mucosal epithelial cells; T-OMECs, transdifferentiated oral mucosal epithelial cells.
Subconjunctival Injection of T-OMECs Prevented Corneal Conjunctivalization and Neovascularization
To determine the role T-OMECs played in preventing conjunctivalization and neovascularization, we quantitatively calculated the length and area of corneal new blood vessels. The difference in the length of new blood vessels between the two groups started from day 2 after alkali burn (p=0.00137 on day 2, p=0.00164 on day 3, p=0.00029 on day 4, p=0.00112 on day 5, p=0.00101 on day 6, p=0.00030 on day 7, p=0.00050 on day 14, p=0.00102 on day 21, and p=0.00010 on day 28, n=4; Fig. 3E). In addition, the difference in the area of new blood vessels appeared from day 2 onward (p= 0.01566 on day 2, p=0.00743 on day 3, p=0.00469 on day 4, p=0.00293 on day 5, p=0.00624 on day 6, p=0.00275 on day 7, p=0.00098 on day 14, p=0.00020 on day 21, and p=0.00058 on day 28, n=4), with 0.50 ± 0.15 mm2 in the T-OMEC group vs 36.56 ± 4.67mm2 in the OMEC group on day 28 (Fig. 3F).
According to the results of H&E and PAS staining, the T-OMEC group maintained a normal corneal phenotype compared with neovascularization and conjunctivalization in the OMEC group. The immunofluorescence pattern in the T-OMEC group was MUC5AC−/α-SMA−/CD34−, and that in the OMEC group was MUC5AC+/α-SMA+/CD34+, which demonstrated the lack of conjunctival and vascular invasion with T-OMEC injection (Fig. 4A). Western blot analyses showed significantly higher expression of CK12, PEDF, and sFlt-1 in the T-OMEC group than in the OMEC group (p=0.00203 for CK12, p=0.00188 for PEDF, and p=0.00384 for sFlt-1, n=3; Fig. 4B). These results suggested that subconjunctival injection of T-OMECs prevented corneal conjunctivalization and neovascularization.
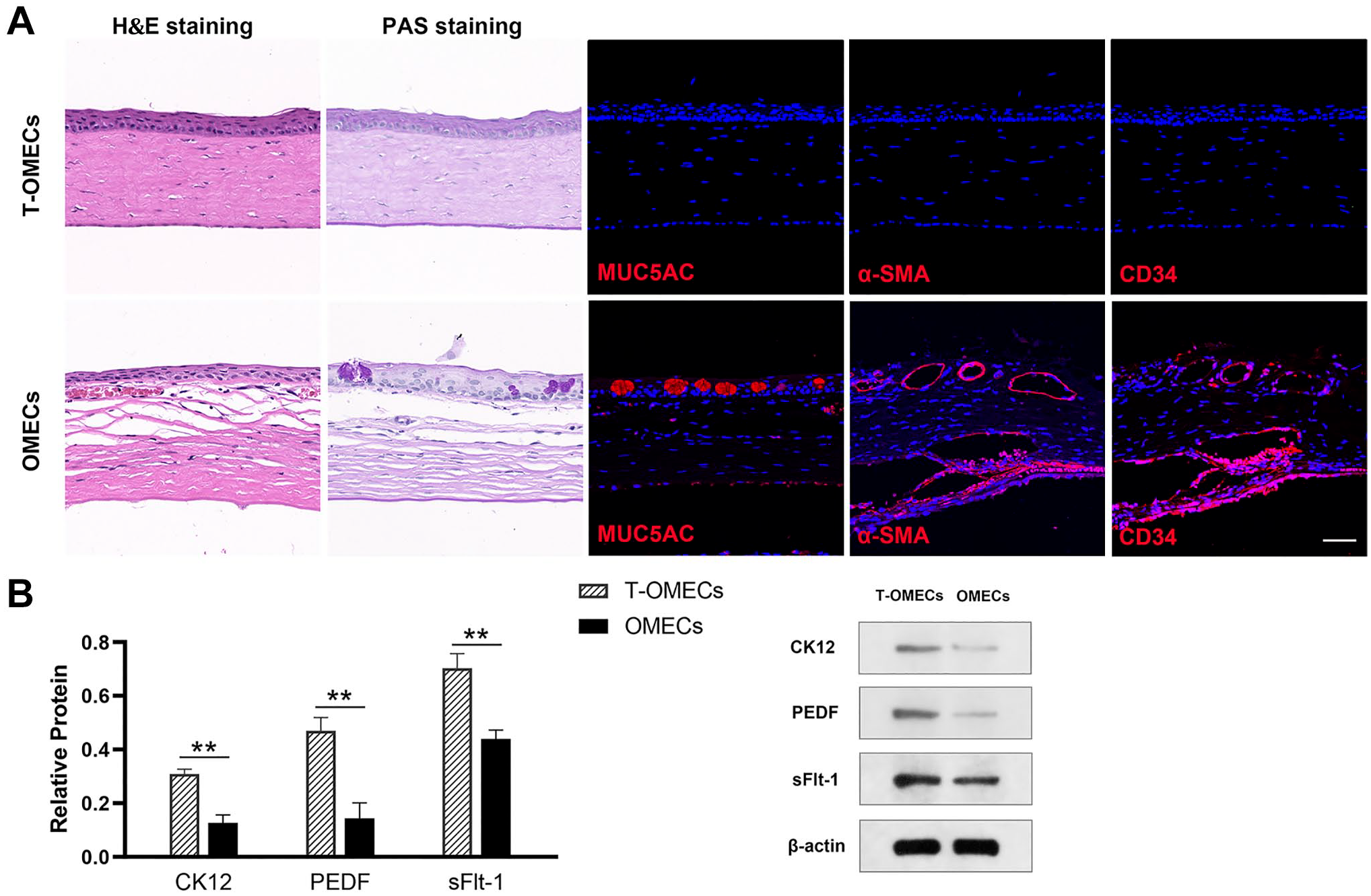
Identification of rat corneal phenotypes following subconjunctival injection of T-OMECs/OMECs. Immunofluorescence staining of MUC5AC, α-SMA, and CD34 of rat corneas in the T-OMEC and OMEC groups (A). Western blot analysis of CK12, PEDF, and sFlt-1 in the two groups (**p<0.01, n=3); β-actin was used as a loading control (B). Scale bar: 50 μm. Abbreviations: α-SMA, α-smooth muscle actin; H&E, hematoxylin and eosin; MUC5AC, mucin 5AC; OMECs, oral mucosal epithelial cells; PAS, periodic acid–Schiff; PEDF, pigment epithelium–derived factor; sFlt-1, soluble fms-like tyrosine kinase-1; T-OMECs, transdifferentiated oral mucosal epithelial cells.
In Vivo Tracking of T-OMECs
To investigate whether T-OMECs directly contribute to ocular surface reconstruction, we labeled T-OMECs with CM-DiI (red fluorescence) before subconjunctival injection. After injection, these labeled cells were monitored for survival and migration under the conjunctiva in the LSCD model. The red fluorescence was found in the 12 o’clock and 6 o’clock (injection sites) regions of rat eyeballs. The distance between the red fluorescence cluster and the limbus reduced gradually: the red fluorescence was observed at about 1 mm outside the limbus on day 7, reached the limbus on day 14, and was found within the cornea on day 28. Therefore, we confirmed that T-OMECs survived in the rat eyeballs for at least 28 days and gradually migrated into the cornea (Fig. 5).
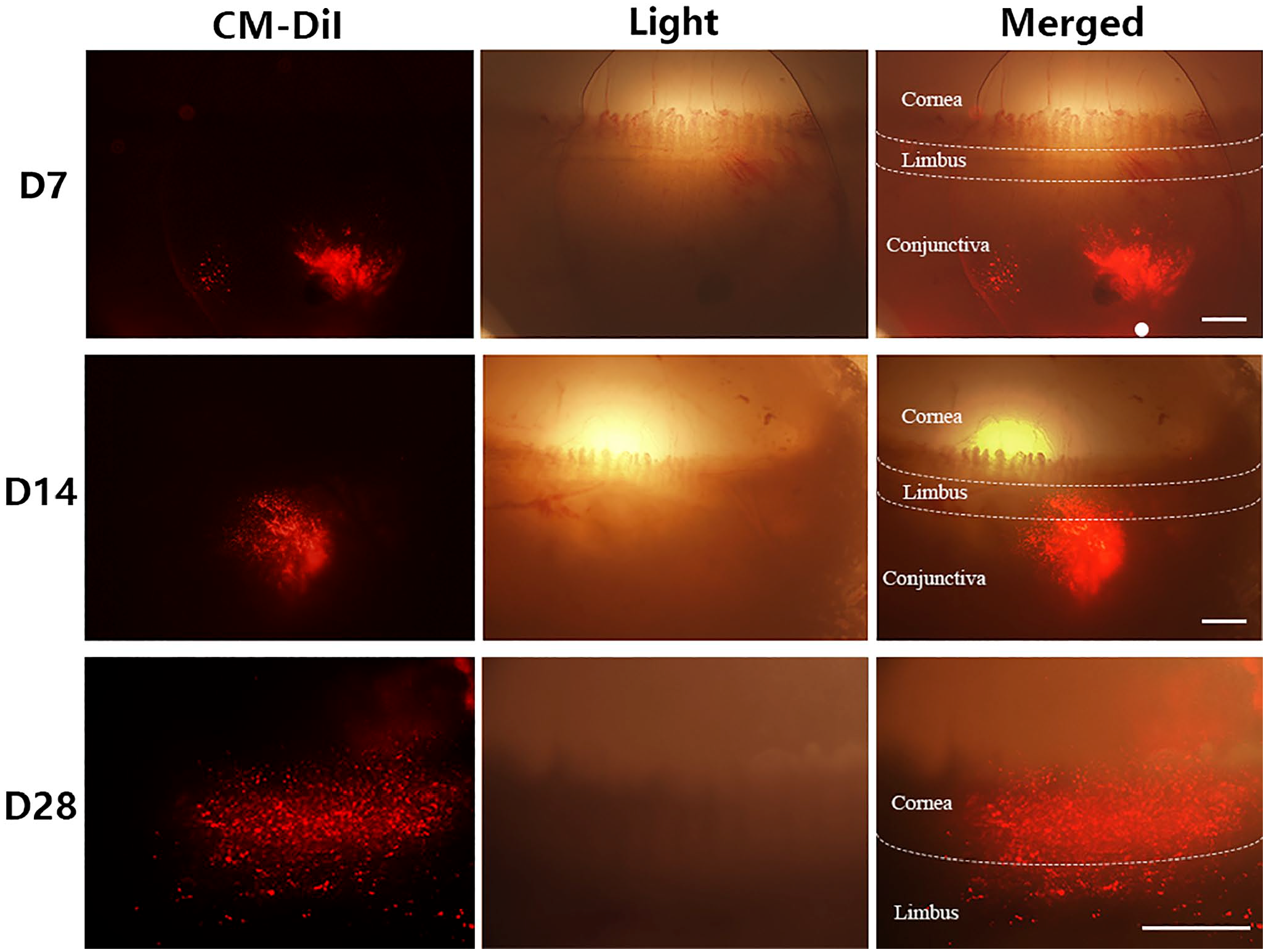
In vivo tracking of T-OMECs. Fluorescent and light photographs of T-OMECs in vivo on days 7, 14, and 28 after subconjunctival injection. Cornea, limbus, and conjunctiva were distinguished by white dotted lines. The white dot showed the initial injection site of T-OMECs. Scale bars: 500 μm. Abbreviations: T-OMECs, transdifferentiated oral mucosal epithelial cells.
Migration of T-OMECs After Subconjunctival Injection
Immunostaining of rat corneas at 28 days after injection further confirmed the migration of T-OMECs into the corneal epithelium. Double immunofluorescence staining for CK12/CK13 demonstrated the disappearance of the transition zone between the cornea and the conjunctiva in the OMEC group as the CK13+ conjunctival epithelium invaded the CK12+ corneal area. Conversely, the T-OMEC group maintained the normal CK12/CK13 transition zone (Fig. 6A). CM-DiI+ cells were detected in the basal layer of corneal epithelium. Interestingly, some of these CM-DiI+ cells still expressed CK12 (yellow arrows) and others were negative (pink arrow, Fig. 6B), which was consistent with the characteristics of T-OMECs we cultured in the Transwell system. Besides, some CM-DiI+ cells were also found to be coexpressed with Pax6 (yellow arrows) in the basal membrane of corneal epithelium (Fig. 6C). Furthermore, several CM-Dil+/ΔNp63α+ cells were detected in the epithelium (yellow arrows) and subjacent stroma of rat cornea (Fig. 6D). These results collectively proved that T-OMECs directly contributed to ocular surface reconstruction in the LSCD model.
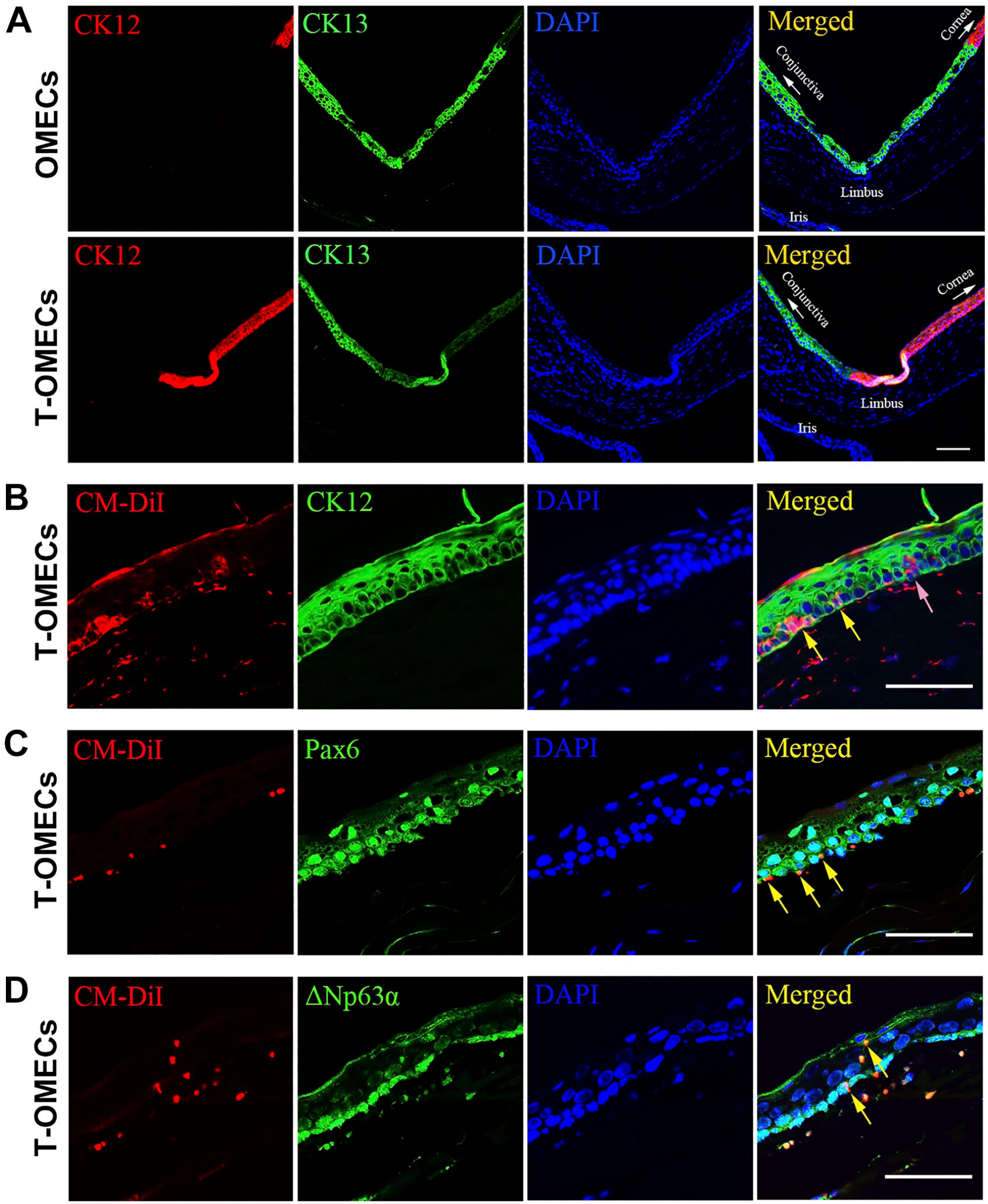
Migration of T-OMECs after subconjunctival injection. Double immunofluorescence staining for CK12 and CK13 of limbus in the OMEC and T-OMEC groups (A). Double immunofluorescence staining for CM-DiI and CK12, Pax6, or ΔNp63α of T-OMECs in the rat cornea (B–D). CM-DiI+ cells were detected in the basal layer of corneal epithelium. Some of these CM-DiI+ cells expressed CK12 (yellow arrows) and some were negative (pink arrow, B). Some CM-DiI+ cells were also found to be coexpressed with Pax6 (yellow arrows) in the basal membrane of corneal epithelium (C). Furthermore, several CM-DiI+/ΔNp63α+ cells were stained in the epithelium (yellow arrows) and subjacent stroma of rat cornea (D). Scale bars: 50 μm. Abbreviations: CK12, cytokeratin 12; CK13, cytokeratin 13; OMECs, oral mucosal epithelial cells; T-OMECs, transdifferentiated oral mucosal epithelial cells.
Discussion
For patients suffering from LSCD, the scarcity of corneal donors, 35 the limitations of CLET and SLET, and the postoperative problems of COMET15,36 create a need for an alternative strategy. In our previous studies of T-OMECs, we successfully induced their transdifferentiation into corneal epithelial-like cells with strong antiangiogenesis ability.17–19 In this investigation, we performed subconjunctival injection of T-OMECs in rat LSCD models. The results of this study—prevention of conjunctival invasion and maintenance of corneal-like phenotype by subconjunctival T-OMECs—demonstrate its beneficial role in LSCD.
Clinically, patients with LSCD often present with severe epithelial defects, corneal opacity, neovascularization, and conjunctivalization, which results in irreversible loss of vision.2,5,29 Both CLET and SLET have limitations on the damage to the contralateral eye in unilateral LSCD patients and immunological rejection after allotransplantation in bilateral LSCD patients.10,37 COMET has remained a significant strategy as it saves the risk of inflicting damage to the alternate healthy eye to harvest limbal tissues for CLET or SLET procedures and has a lower risk of rejection. 38 However, it faces extreme challenges in terms of potential murine infectious diseases in using 3T3 feeder cells and various degrees of postoperative corneal neovascularization.16,39 Fortunately, in this study, subconjunctival injection of T-OMECs accelerated corneal epithelium wound healing of the LSCD model with encouraging results of preventing corneal conjunctivalization and neovascularization. T-OMECs were obtained using homogeneous LNCs as feeder layers and shown to express more antiangiogenic factors compared with OMECs. These transdifferentiated cells showed great potential in the treatment of LSCD with less postoperative problems.
In contrast to the traditional COMET surgery,12,14,40 the injected T-OMECs were transdifferentiated into the corneal epithelial-like phenotype and could stably survive in the cornea. Hiroaki et al. showed that one rabbit cornea attained cornea-specific phenotypes of CK12 and Pax6 after OMEC transplantation, but whether these phenotypes were derived from the transdifferentiation of OMECs or the proliferation of residual LESCs was difficult to ascertain. 41 Similarly, both Gaddipati et al. 42 and Nakamura et al. 43 lacked substantial evidence to suggest that the CK12 phenotype was derived from the transdifferentiation of transplanted OMECs. In addition, the problem of neovascularization could not be neglected in the studies of Gaddipati et al. This study provides conclusive evidence of the maintenance of corneal-like phenotype in T-OMECs after transplantation, which contributes to explain its positive effect on ocular surface reconstruction in rat LSCD models.
Prior subconjunctival injection for LSCD involved mesenchymal stem cells.44–46 In this study, the injected cells were epithelial-derived and morphologically more similar to corneal epithelial cells. These characteristics enabled them to exist stably in the cornea and help support the recovering of corneal epithelium. Limbal mesenchymal stem cells, 44 bone marrow mesenchymal stem cells, 47 or adipose-derived mesenchymal stem cells 48 may provide therapeutic effects in LSCD via improving the microenvironment of LESCs. The current investigation, however, proved that the cells injected under the conjunctiva migrated into the basal layer of corneal epithelium and maintained their corneal-like phenotype, thus contributing to the regeneration of the damaged LESCs. According to our observation, T-OMECs migrated focally in the 12 o’clock and 6 o’clock regions, not spreading to all directions inside the conjunctival stroma, and were guided to the corneal epithelium ultimately. The results of tracking T-OMECs in vivo confirmed that these cells could survive in the LSCD model for an extended period. Moreover, some of the injected T-OMECs continued to express CK12, Pax6, and ΔNp63α while stably existing in the corneal epithelium. Therefore, we hypothesize that these transdifferentiated cells might contribute toward the restoration of the damaged LESCs from preexisting pools of stem cells.
For the emergency treatment of patients with acute alkali burns, autologous serum, hormones, and antiangiogenic drugs are often injected under the conjunctiva following washing with a large amount of saline,49,50 indicating that subconjunctival injection is feasible in the clinical setting. We choose subconjunctival injection as the route of delivery for its convenience compared with the strict COMET procedure for LSCD treatment.20,21 Considering the potent proliferative potential of LNCs in vitro, 24 we can obtain abundant LNCs in vitro in a short time using only a small amount of donated corneal material or redundant limbal material from keratoplasty. Similarly, OMECs are more accessible and only require a few scrapings of buccal mucosa from the patients. 51
Although T-OMECs were successfully transplanted into the rat corneas, the outcomes in humans remain unknown and will be investigated by our team in the future. In the follow-up investigations, we intend to improve the experimental protocols and conditions, explore the underlying mechanism of transdifferentiation, enhance the transdifferentiation efficiency and migration ability, and observe the long-term homeostasis of transplanted T-OMECs in the cornea. Such models will effectively improve the functionality of LESCs and create opportunities for the patients to reconstruct ocular surface and improve vision. We believe that with further studies and breakthroughs in vitro and in vivo, this novel strategy will present an exciting prospect for ocular surface reconstruction in patients with LSCD.
In conclusion, subconjunctival injection of T-OMECs plays a beneficial role in treating LSCD in a rodent model. T-OMECs survive in the ocular surface for as long as 28 days, migrate into the corneal epithelium, prevent conjunctival invasion, and stably maintain their corneal-like phenotype. The proposed mechanism for the above-seen effects is stem cell restoration from preexisting pools. Overall, this study provides an alternative strategy for the treatment of LSCD.
Supplemental Material
sj-zip-1-jhc-10.1369_0022155420980071 – Supplemental material for Subconjunctival Injection of Transdifferentiated Oral Mucosal Epithelial Cells for Limbal Stem Cell Deficiency in Rats
Supplemental material, sj-zip-1-jhc-10.1369_0022155420980071 for Subconjunctival Injection of Transdifferentiated Oral Mucosal Epithelial Cells for Limbal Stem Cell Deficiency in Rats by Yu-Ting Xiao, Hua-Tao Xie, Xin Liu, Chao-Ye Duan, Jing-Yu Qu, Ming-Chang Zhang and Xin-Yue Zhao in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We would like to thank Editage for English language editing.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors Contributions
YTX contributed to the collection and assembly of data, interpretation, and manuscript writing. HTX was responsible for the conception and design of the study and manuscript writing. XL analyzed the data. CYD and JYQ contributed to the revision of the manuscript. MCZ and XYZ were responsible for the conception, design and interpretation, and final approval of the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Program of China (2017YFE0103500), National Natural Science Foundation of China (grant number: 81600708), and the Fundamental Research Funds for the Central Universities (HUST: 2019kfyXMBZ065). These fundings provided sufficient funds for the purchase of materials and antibodies.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
