Abstract
Here, we describe an ethylenediaminetetraacetic acid (EDTA)-based bone demineralization procedure that uses cation-exchange resin and dialysis tubing. This method does not require solution changes or special equipment, is faster than EDTA alone, is cost-effective, and is environmentally friendly. Like other EDTA-based methods, this procedure yields superior tissue preservation than formic acid demineralization. Greater protein antigenicity using EDTA as opposed to formic acid has been described, but we also find significant improvements in carbohydrate-based histological staining. Histological staining using this method reveals cartilage layers that are not distinguishable with formic acid demineralization. Carbohydrate preservation is relevant to many applications of bone demineralization, including the assessment of osteoarthritis from bone biopsies and the use of demineralized bone powder for tissue culture and surgical implants. The improvements in time, expense, and tissue quality indicate this method is a practical and often superior alternative to formic acid demineralization:
Introduction
Bone demineralization is a common practice in bone and cartilage research and in medical diagnostics. Bone samples for biopsy and cytological study usually must be demineralized before histological analysis; demineralization is also required to generate demineralized bone powder, used for scaffolding in bone tissue engineering. 1 Despite various options, no single decalcification method is ideal for optimizing time, tissue preservation, cost, and waste disposal.
A number of different demineralization methods have been devised, including electrolytic, strong acid, weak acid, and calcium chelator extraction. Electrolytic methods of decalcification are fast, but the heat produced by electrolysis compromises tissue quality and the resulting tissue is not useful for histology. 2 Acid extraction and calcium chelator methods, while slower, are better for preserving cytological detail. The use of ethylenediaminetetraacetic acid (EDTA) as a calcium chelator yields the highest degree of tissue preservation, but requires significantly more time. 3 Two common methods use either formic acid or EDTA. Because formic acid is rapid, it is widely preferred over EDTA for most applications. In contrast, EDTA is used in cases requiring better tissue preservation, usually for immunostaining. However, as most applications are impacted by tissue preservation, it is questionable whether the speed of formic acid is worth the risk of compromised tissue analysis.
Several studies have proposed adding ion-exchange resin to enhance bone demineralization. Most of these studies combined acids (nitric, hydrochloric, or formic) with cation-exchange resin, and whereas some found improvements,2,4 others found either no improvement over acid alone 5 or degradation of the resin by the acid. 6 Surprisingly, most studies do not use EDTA combined with resin. EDTA on its own takes a relatively long time to decalcify tissue, but has advantages over acid extraction methods for enzyme and DNA preservation and for immunostaining.4,7,8 Using EDTA instead of formic acid also reduces the cost of waste disposal and the need for lab safety precautions, as EDTA waste requires neither a fume hood nor hazardous waste removal. The major drawback of EDTA, that is, the time required for demineralization, should be theoretically reduced using a cation-exchange resin with a higher affinity for calcium than does EDTA. Also, adding a cation-exchange resin to EDTA obviates the need for daily solution changes, thus reducing cost. Comparisons of speed and cost were among the goals of this study.
An obstacle to overcome with the ion-exchange resin is contamination of the tissue with the resin. When uncontained resin is placed directly with the bone during decalcification, the resin can contaminate the surface of the tissue. This is particularly problematic for procedures using pulverized bone, where the resin cannot be separated from the bone powder. One solution to this problem is the use of an in-line pump to circulate the decalcification solution through an ion-exchange column. Reports of this method have suggested either a mild acidic fixative pumped through an ion-exchange resin column to simultaneously fix and decalcify bone and bone marrow biopsies 9 or pumping decalcifying solution over a cation-exchange column to demineralize bone while maintaining its osteoinductivity for use in allografts. 10 Although this method is certainly effective, not all labs (especially histology labs) possess pumps and columns, making this a less practical option.
Our solution is to demineralize bone using EDTA with cation-exchange resin enclosed in dialysis tubing. This method results in improvements in decalcification time and expense over EDTA alone, and retains the superior tissue preservation inherent in EDTA-based demineralization. One particular benefit from our method (or likely any method using EDTA) is enhanced preservation of carbohydrate in the tissue. The implications of this preservation are far-reaching and combined with the other advantages show that this proposed demineralization procedure is preferable to other existing methods for most applications.
Methods
Chicken Tissue
Whole roaster chickens were purchased from a local market. Segments of vertebrae weighing between 2 and 10 g were dissected out, with some surrounding soft tissue intact (similar to a bone biopsy tissue sample). The tissue pieces were fixed in 4% paraformaldehyde (in 0.1 M sodium phosphate buffer at pH 6.8) for 4 days and then washed 2 days in PBS (Dulbecco’s phosphate-buffered saline; Hyclone; Cytiva, Marlborough, MA), all at room temperature with orbital shaking.
Demineralization Treatments
The chicken vertebral bone pieces were divided into four decalcification treatment groups and one control group (below). Because the vertebrae varied in dimension and were not expected to decalcify at the same rate, the segments were assigned to groups non-randomly such that each group contained a range of sizes and shapes. The demineralization solution for treatment groups 1 to 3 was 0.5 M EDTA (ACS grade; Sigma-Aldrich, St. Louis, MO) at pH 7.5 with 5% sucrose to preserve tissue quality.
Treatment Group 1: EDTA With Cation-exchange Resin
Cellulose phosphate cation-exchange resin (1.5 g; Sigma-Aldrich; cat. no. C2258) was first activated (stripped of any residual contaminating ions) by a 5-min treatment with NaOH (1 g in 50 ml of dH2O) and successive washes with dH2O until the pH fell below 10, followed by a 5-min treatment with HCl (2 ml of 12 N HCl with 48 ml of dH2O) and washes with dH2O until the pH rose above 3.0. The resin was then briefly equilibrated in the EDTA solution and subsequently transferred into a length of dialysis tubing (Spectra/Por, MWCO 6-8kD; Spectrum Chemical, New Brunswick, NJ), which was secured on both ends with plastic dialysis tubing clamps (Fig. 1A). Three bone segments were placed in a lidded container with the resin/dialysis tubing and 150 ml of EDTA/sucrose and placed on an orbital shaker (80 rpm) at room temperature for 5 days. Alternatively, our laboratory has successfully demineralized bone with this method using a magnetic stir bar to provide continuous circulation while the bone was suspended by string in mesh specimen biopsy bags (ThermoFisher Scientific; Waltham, MA).

The EDTA/resin method. (A) Image showing cellulose phosphate cation-exchange resin encased in dialysis tubing, in a beaker filled with EDTA/sucrose solution. (B) Schematic of the EDTA/resin method, in which the cation-exchange resin acts as a calcium sync for EDTA, as EDTA chelates calcium from bone. The constant binding of calcium by the resin allows recycling of EDTA, which increases the speed of demineralization. Abbreviation: EDTA, ethylenediaminetetraacetic acid.
Treatment Group 2: EDTA, No Solution Change
Three bone segments were placed in a lidded container with 150 ml of EDTA/sucrose and placed on an orbital shaker at room temperature for 5 days.
Treatment Group 3: EDTA, Solution Changed Daily
Three bone segments were placed in a lidded container with 150 ml of EDTA/sucrose and placed on an orbital shaker at room temperature for 5 days. The solution was replaced every 24 hr. This is the standard EDTA method.
Treatment Group 4: Formic Acid
Three bone segments were placed in mesh bags and suspended in a covered glass container with 40× weight:volume of tissue of 15% formic acid (250 ml for three bone pieces), which was continuously agitated by a magnetic stir bar for 5 days in a chemical fume hood. The solution was replaced every 24 hr.
Treatment Group 5: Control
Two bone segments were placed in 150 ml of PBS on an orbital shaker for 5 days.
Quantification of Demineralization
For all treatment groups, X-rays were taken every 24 hr for 5 days using a Toshiba Rotanode X-ray tube (unit model: E7242GX). The parameters for X-ray imaging were the same each day, such that the tissue was imaged at a distance of 40 inches from the X-ray source using 60 kVp at 2.5 mA and scanned with a 25.2 × 30.3 cm Fujifilm FCR IP cassette (type CC), and the tissue pieces were arranged in the same configurations and imaged together. To assess the degree of demineralization, digital rectangles were fit to minimize normalized black space around the bones, and the same rectangles were used each day. The mean grayscale values of rectangles in each treatment group were calculated using ImageJ software (National Institutes of Health, Bethesda, MD) and averaged over each day. Although X-ray is one of the best methods to determine demineralization, it is not perfect by any means; slight changes in background grayscale level and the potential variations that can occur if the bone pieces are not placed exactly in the same orientation every day introduce some variation.
Immunostaining and Histology
After demineralization, tissues from the resin and formic acid groups were either cryoembedded for immunostaining or paraffin embedded for histological staining. For cryoembedding, demineralized tissue was cryoprotected overnight in 30% sucrose in 0.1 M sodium phosphate buffer (pH 6.3) and then infiltrated in OCT medium (Tissue-Tek; Sakura Finetek, Torrance, CA) at room temperature, first for 3 hr in 1:1 30% sucrose to OCT and then for 3 hr in 100% OCT. Samples were then cryoembedded in plastic molds using dry ice and ethanol. Cryosections of 14 to 20 µm were collected and applied to either Superfrost Plus (Fisherbrand) or agar-coated glass slides. Sections for immunostaining were blocked for 1 hr in normal goat serum blocking buffer (0.03 M Tris [pH 7.5], 0.15 M NaCl, 1% glycine, 0.4% Triton X100, 10% goat serum) before antibody incubation. Two primary antibodies (cell supernatants used at 1:100 dilution; Developmental Studies Hybridoma Bank, Iowa City, IA) were used for this study: RT97, which recognizes the neurofilament, and II-II6B3, a collagen 2–specific antibody whose antigen is prevalent in cartilage. The specificity of both antibodies was verified by secondary antibody–only controls, as well as imaging in both FITC (not shown) and Texas Red channels. Secondary antibody staining was achieved using a 1:500 dilution of goat anti-mouse (593 nm) MAb (Molecular Probes; Eugene, OR).
For wax embedding and histological staining, demineralized tissue was processed in a Tissue-Tek VIP 6 processor starting with an ethanol series until dehydrated. The tissue was then cleared in Clear-Rite followed by Tissue Prep paraffin. Samples were then embedded in Tissue-Tek paraffin using the Tissue-Tek TEC embedding station. Sections of 5 to 7 µm were cut using a Jung RM2035 microtome and transferred to glass slides.
Histological analysis was achieved using hematoxylin and eosin (H&E), alcian blue/picrosirius red staining, or SYTOX green fluorescent nuclear staining. H&E is a general stain specific for cell nuclei and cytoplasm. Picrosirius red, which primarily stains collagen fibers, was used as a stain for fibrocartilage in intervertebral discs, and Alcian blue, which stains acidic polysaccharides, was used for general cartilage staining. For these stains, paraffin sections were dewaxed with xylenes and rehydrated through a series of EtOH washes to water. H&E staining was achieved with a 1-min immersion in Gill’s Hematoxylin #2 (Polysciences; Warrington, PA), followed by a 5-min continuous wash in dH2O bath and the first dehydration steps with 50% EtOH and 70% EtOH. The slides were subsequently immersed in 0.25% eosin Y (Sigma-Aldrich)/80% EtOH/0.5% glacial acetic acid for 5 sec and then washed twice with 70% EtOH, 2 min each. Dehydration continued with 2-min incubations with 95% EtOH, then 100% EtOH, and finally with xylenes before mounting in DPX media (Electron Microscopy Sciences; Hatfield, PA). Alcian blue and picrosirius red staining was performed on rehydrated sections on slides by incubation in Alcian blue (0.048% Alcian Blue 8GX dye [Sigma-Aldrich] in 70% EtOH, 20% glacial acetic acid) for 15 min, followed by a water wash and then 1 hr in picrosirius red (0.1% Direct Red 80 [Sigma-Aldrich], in 1.3% saturated picric acid). The slides were subsequently washed in acidified water (0.5% glacial acetic acid), dehydrated through a series of EtOH washes to xylenes, and mounted in DPX mounting media. SYTOX green staining on dewaxed paraffin sections was achieved by a 30-min incubation at room temperature with dilute SYTOX green (ThermoFisher Scientific; 0.2 µl of 5 mM stock per 1 ml Dulbecco’s PBS), followed by washing in PBST (Dulbecco’s PBS with 0.1% Tween), dehydration in an ethanol series to xylenes, and mounting in DPX media.
Histology and immunofluorescence imaging were performed on a Zeiss Axioscope A1 microscope in conjunction with a Jenoptik ProgRes C14 Plus digital camera and accompanying software. Experiments comparing formic acid and EDTA/resin-treated sectioned tissue were performed simultaneously (i.e., antibody dilutions for the two treatments were prepared in the same tube and applied at the same time, and for histology, both treatments were stained at the same time using the same batches of stains and washes), and images for each experiment were captured with the same parameters (contrast, gain, exposure, etc.) to allow direct comparison. All experiments conformed to guidelines specified by the Montana State University Institutional Animal Care & Use Committee and biosafety committees.
Results
Our EDTA/resin method was assessed for its ability to decalcify bone, for its suitability to histology and immunohistochemistry (IHC), and for its cost, and these aspects were compared with other decalcification methods. Our method is shown in Fig. 1, in which the cation-exchange resin is encased in dialysis tubing (Fig. 1A) and calcium is removed from bone by chelation with EDTA, with the resin serving as a calcium reservoir (Fig. 1B; see the “Discussion” section for further information on the chemistry behind the method). Decalcification rates were compared between our EDTA/resin method and formic acid, standard EDTA (replacing EDTA solution daily), EDTA without solution replacement, and PBS-only control. Because formic acid is a predominant method, we further compared our EDTA/resin procedure with formic acid for IHC and histology. Relative costs of the methods were assessed for EDTA/resin, formic acid, and standard EDTA procedures.
We chose to analyze the demineralization of tissue pieces with variable bone and soft tissue content, to better approximate bone biopsies and to investigate the effects of the methods on soft tissue. This decision to not use standardized bone content replicates led to variable rates of decalcification for each condition tested (due to variable bone content in each tissue specimen), but several trends were nonetheless observed.
Speed of Decalcification
Decalcification speed was estimated using the mean grayscale values of bone samples on daily X-rays (Fig. 2A). Grayscale values between 0 (black) and 255 (white) decreased in all non-control groups over the course of the treatments as calcium was removed from the tissue. Our data indicate that formic acid is the most rapid demineralization method, but addition of cation-exchange resin to EDTA is faster than EDTA alone (Fig. 2B). The fastest rates of demineralization across the different treatments occurred in the first 24 hr; we therefore determined the slopes of the demineralization curves from 0 to 24 hr (averaged over the number of bone pieces for each condition) for a comparison of rates. The slopes were calculated (in order of fastest to slowest) as −0.21 grayscale/hr for formic acid, −0.13 for EDTA/resin, −0.08 for EDTA (changed daily), −0.08 for EDTA (no daily solution changes), and +0.002 for the PBS-only control. Our data also indicate that the bone pieces were fully decalcified by day 2 in formic acid (as evidenced by the plateau of the curve) and by day 3 in EDTA/resin, but had not fully decalcified in the other treatments by the end of the 5-day time course (our ability to X-ray the bone pieces was limited to five consecutive days, which prevented demineralization end point determinations for the remaining groups).
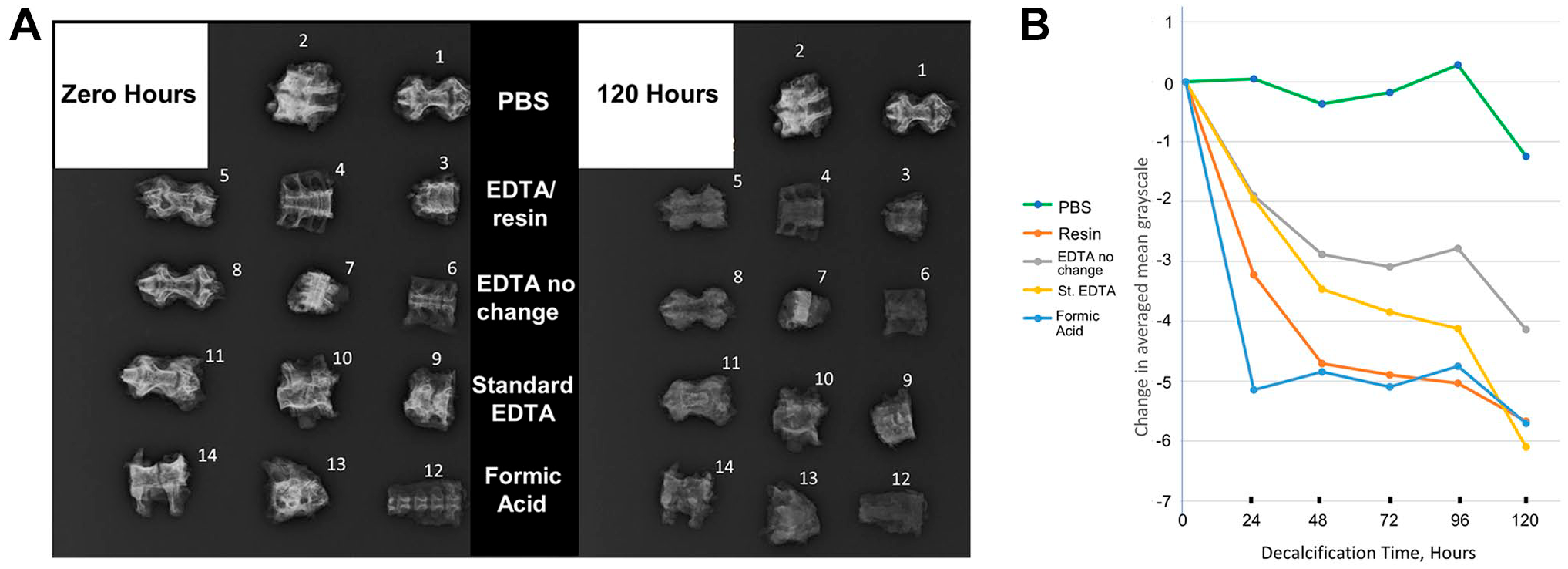
Comparison of demineralization rates by different demineralization methods. (A) Raw X-ray data from the zero time point and the final 120-hr time point of demineralization. The middle panel denotes the methods used, which includes the PBS control, our EDTA/resin method, EDTA no change (EDTA/sucrose solution only, with no daily solution changes), standard EDTA (EDTA/sucrose solution changed daily), and formic acid (15% formic acid, changed daily). (B) Graph of changing pixel grayscale values for each method over time. Pixel values for the three bone pieces in each treatment were averaged and adjusted to set 0 time points at 0 pixel value. Error bars are not shown because the tissue specimens within and between the different groups vary in bone and soft tissue content and are therefore not replicates; this graph is intended to demonstrate overall demineralization trends. Formic acid was found to be the fastest method of demineralization, followed by EDTA/resin, standard EDTA, EDTA no change, and, finally, the PBS control. Only the formic acid and EDTA/resin treatments resulted in complete demineralization before the end of the 5-day time course. Abbreviations: EDTA, ethylenediaminetetraacetic acid; PBS, phosphate-buffered saline.
Tissue Preservation
Considering that formic acid is a predominant demineralization method, we compared formic acid with our EDTA/resin procedure for immuno-antigen preservation and for the quality of histological stains. For outward tissue appearance, it was apparent that formic acid treatment results in tissue discoloration (Fig. 3A), including lightening at the outer edges and darkening of the bone. EDTA/resin demineralization resulted in tissue that looked essentially the same as before treatment (Fig. 3B). Upon sectioning and histological staining, it was also observed that the formic acid–treated tissue exhibited more compacted hyaline cartilage in the bone endplate (Fig. 3C) than with EDTA/resin demineralization (Fig. 3D; see Fig. 3E for schematic), representing a loss in tissue structure. The pieces of bone chosen for this analysis were consecutive cervical vertebrae from the same chicken, so we would not expect significant differences in tissue architecture unless caused by the demineralization method.
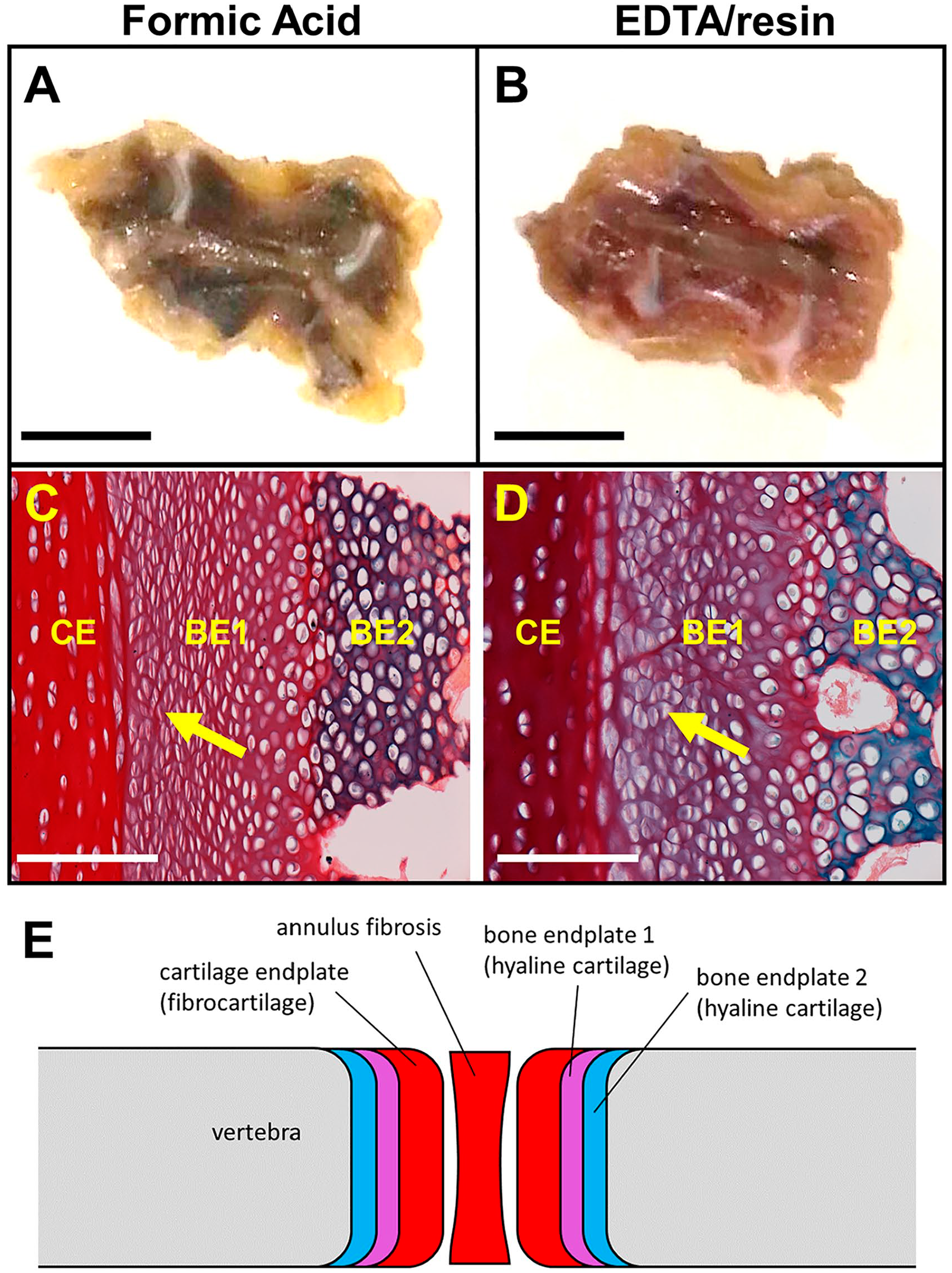
Tissue integrity comparison between formic acid and EDTA/resin methods. (A, C) Formic acid treatment; (B, D) EDTA/resin treatment. (A) Image of formic acid–treated chicken cervical vertebra, showing tissue discoloration. Scale bar, 1 cm. (B) Image of EDTA/resin-treated chicken cervical vertebra, with no discoloration. Scale bar, 1 cm. (C) Same vertebra as in (A), paraffin embedded and stained with alcian blue and picrosirius red. Yellow arrow denotes a bone endplate cartilage layer with compressed cells (layer coincides with bone endplate 1 hyaline cartilage layer; see Fig. 3E). White scale bar, 100 µm. (D) Same EDTA/resin-treated vertebra as in (B), paraffin embedded and stained with alcian blue and picrosirius red. Yellow arrow denotes the same tissue layer as noted in (C), showing improved cell morphology. White scale bar, 100 µm. E, Schematic of vertebral and intervertebral tissues. Chicken cervical vertebrae at 7 to 8 weeks of age lack a nucleus pulposus, but the annulus fibrosus of the disc is evident. Abbreviation: EDTA, ethylenediaminetetraacetic acid.
Immuno-antigen Preservation
Our EDTA/resin method and the formic acid method were compared for immunofluorescence staining using antibodies specific to soft tissue (bone tends to be highly autofluorescent, making soft tissue more appropriate for fluorescence staining comparison). Cryoembedded and sectioned chicken cervical vertebral tissue was stained with either RT97 MAb to detect neurofilament or α-Collagen 2 MAb (Col2) to detect cartilage. True to the variable nature of antibodies, RT97 staining was unaffected by the demineralization method (Fig. 4A and B), but Col2 staining was greatly reduced in the formic acid relative to EDTA/resin treatment (Fig. 4C and D). Interestingly, Col2 immunoreactivity was most obviously affected in the bone endplate hyaline cartilage juxtaposed to the intervertebral disc endplate (for bone endplate cartilage 1 layer, see Fig. 3E), and the same tissue layer was found to be compacted and lacking tissue architecture with formic acid demineralization (Fig. 3C). As has also been shown by other studies, 3 therefore, EDTA-based decalcification is overall superior to formic acid for tissue integrity and the preservation of protein antigens.
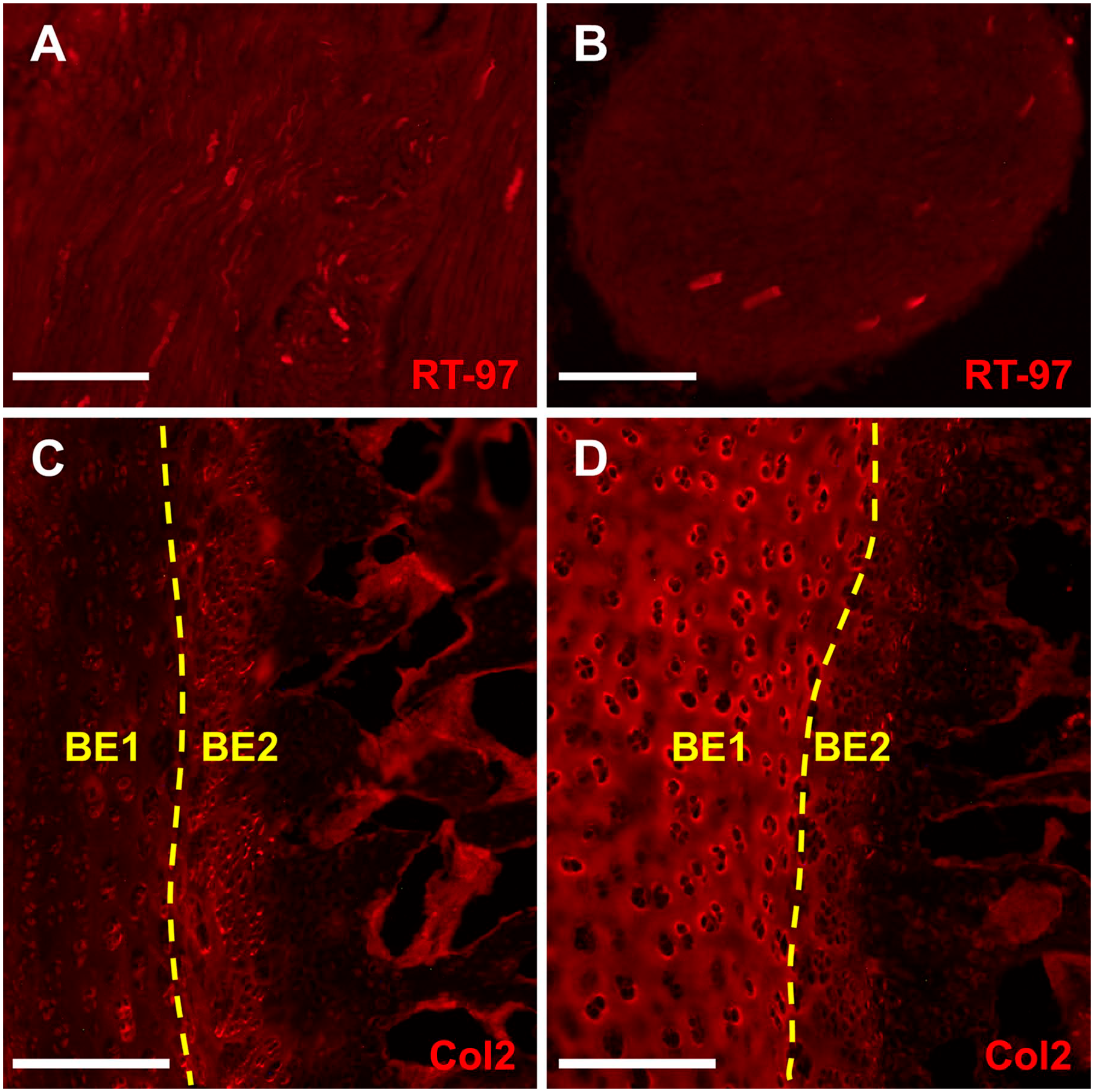
Immunofluorescent staining comparison between formic acid and EDTA/resin methods. (A, C) Formic acid treatment; (B, D) EDTA/resin treatment; all panels show cryosections of chicken cervical vertebrae. (A) Formic acid–treated cryosection immunostained for neurofilament using RT97 MAb. White scale bar, 100 µm. (B) EDTA/resin-treated cryosection immunostained using RT97 MAb. White scale bar, 100 µm. (C) Formic acid–treated cryosection immunostained for collagen 2 using the Col2A1 antibody. White scale bar, 200 µm. (D) EDTA/resin-treated cryosection immunostained for collagen 2. White scale bar, 200 µm. No significant differences between demineralization methods were observed for RT97 MAb staining, but collagen 2 staining was substantially inhibited with formic acid treatment compared with EDTA/resin. Abbreviation: EDTA, ethylenediaminetetraacetic acid.
Histological Preservation
Two histological stains, H&E and alcian blue/picrosirius red, were applied to assess potential staining quality differences between formic acid and EDTA/resin demineralization methods. Overall tissue appearance and nuclear integrity were examined with H&E, and nuclear staining was further investigated by SYTOX green staining. Unlike other reports, 7 we found no significant difference in cell nuclei stain between formic acid and EDTA/resin treatments, either by H&E or by SYTOX green (Fig. 5A-D). However, EDTA/resin demineralization showed delineation of a cartilage layer not apparent with formic acid (Fig. 5E and F). In this case, the tissue layer most compromised by formic acid was the intervertebral disc endplate cartilage, which displayed greater stain intensity in the EDTA/resin treatment. The combined histology and immunofluorescence results indicate that the hyaline cartilage and neighboring fibrocartilage at the bone to disc junction are negatively impacted by formic acid demineralization.
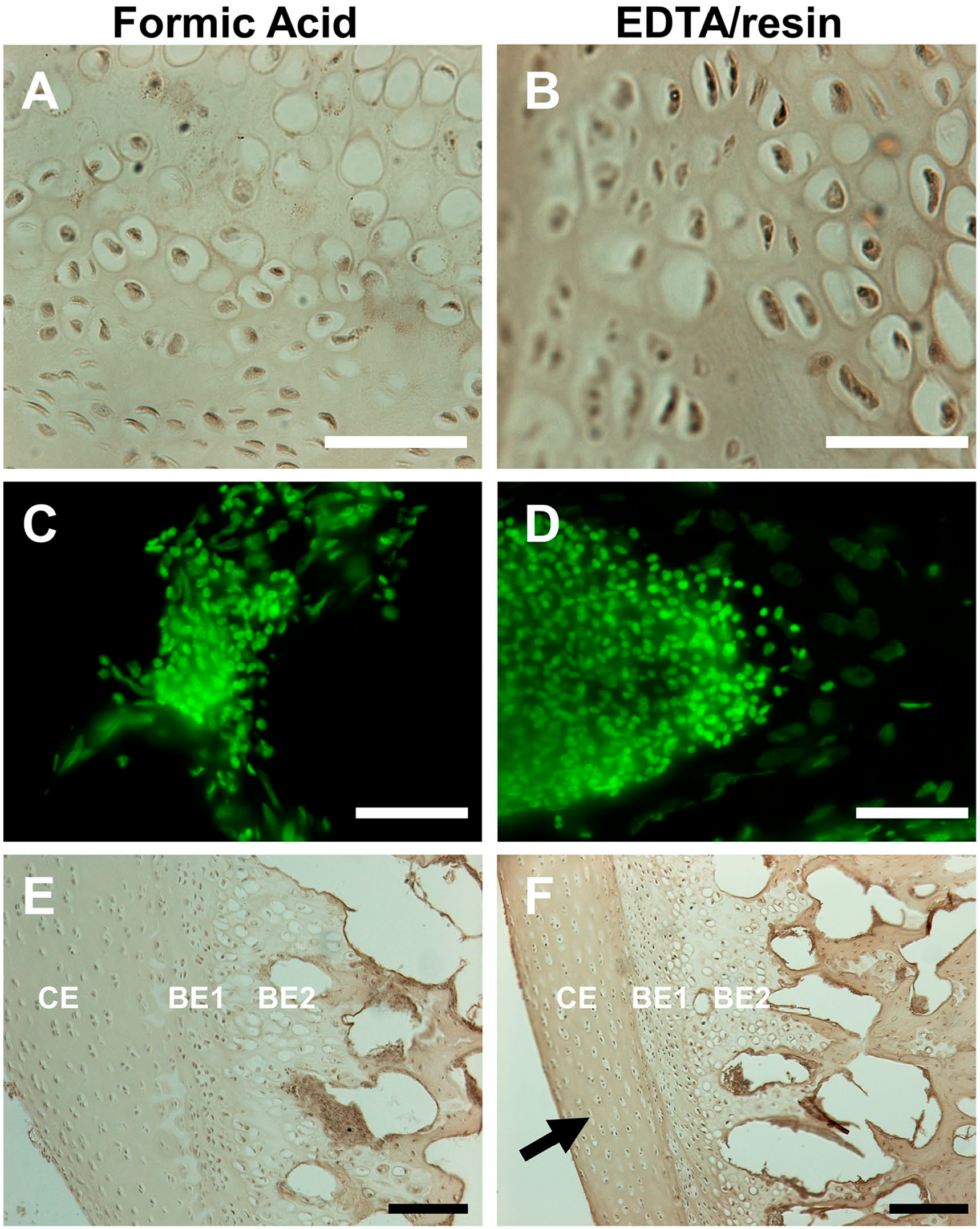
H&E and SYTOX green staining comparison between formic acid and EDTA/resin methods. (A, C, F) Formic acid treatment; (B, D, G) EDTA/resin treatment; all panels show images of chicken cervical vertebrae. (A) H&E-stained paraffin section, formic acid treatment, showing hyaline cartilage cells. White scale bar, 50 µm. (B) H&E-stained paraffin section, EDTA/resin treatment, showing hyaline cartilage cells. White scale bar, 50 µm. (C) SYTOX green–stained nuclei of bone marrow cells, formic acid treatment. White scale bar, 100 µm. (D) SYTOX green–stained nuclei of bone marrow cells, EDTA/resin treatment. White scale bar, 100 µm. (E) H&E staining, formic acid treatment, view of intervertebral disc and bone tissue. Black scale bar, 250 µm. (F) H&E staining, EDTA/resin treatment, view of intervertebral disc and bone tissue. Arrow shows specific staining of the cartilage endplate of the intervertebral disc, not seen with formic acid treatment. Black scale bar, 250 µm. Abbreviations: H&E, hematoxylin and eosin; EDTA, ethylenediaminetetraacetic acid; CE, cartilage endplate; BE1, bone endplate 1 layer; BE2, bone endplate 2 layer; these tissue layers are detailed in the schematic in Fig. 3E.
Alcian blue/picrosirius red stain is used to differentiate different types of cartilage, with picrosirius red specific to collagen fibrils in fibrocartilage and alcian blue specific to acidic polysaccharides abundant in the extracellular matrix of hyaline cartilage. When comparing formic acid and EDTA/resin-demineralized tissue, it is evident that formic acid treatment results in reduced alcian blue staining in a cell layer already identified to be compromised, the segment of hyaline cartilage directly apposed to the fibrocartilage at the meeting of bone endplate to intervertebral disc endplate (Fig. 6A-D). Also, the staining of fibrocartilage of the central annulus fibrosus cartilage of the disc is negatively affected (Fig. 6B). With formic acid treatment, these cartilaginous tissues appear red from the picrosirius red stain, but in the EDTA/resin-treated tissue, the structures are purple, an additive effect from picrosirius red and alcian blue staining of the same structures. Alcian blue staining of the hyaline cartilage facing trabecular bone at the bone endplate appears to be unaffected by formic acid, which suggests that the loss of alcian blue staining may be cell layer–specific. The purple staining of bone endplate hyaline cartilage apposed to disc fibrocartilage indicates this is a separate tissue layer that expresses more abundant acidic polysaccharides than the neighboring bone-associated hyaline cartilage. This same region (what we refer to as bone endplate layer 1) was seen to be more compacted in the formic acid–demineralized tissue (Fig. 3C) and more prone to loss of Col2 staining (Fig. 4C), indicating that the morphology, Col2 preservation, and extracellular matrix of this particular cell layer are vulnerable to formic acid–induced degradation. Because H&E staining highlights fibrocartilage using EDTA/resin but not formic acid treatment, and alcian blue/picrosirius red reveals a pattern of acidic polysaccharides with EDTA/resin not seen with formic acid, we conclude that EDTA/resin is superior to formic acid for the identification of distinct cell layers in disc and bone-associated cartilage.
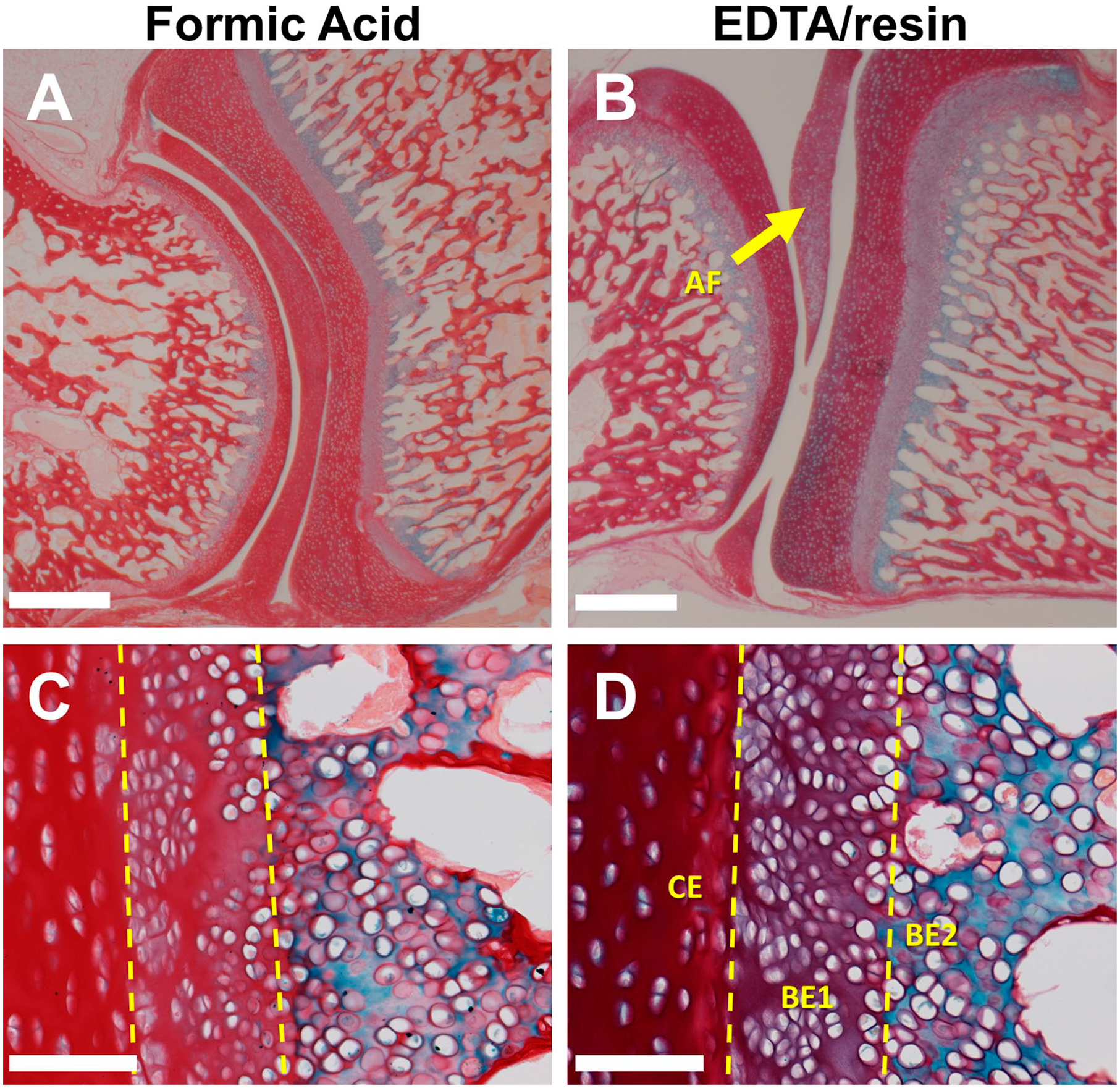
Alcian blue and picrosirius red histological staining comparison between formic acid and EDTA/resin methods. (A, C) Formic acid treatment; (B, D) EDTA/resin treatment; these panels show paraffin sections of chicken cervical vertebrae; the enlarged images in (C) and (D) are from the same tissue specimen but not from the same sections as in (A) and (B). (A) Formic acid–treated bone and intervertebral disc tissue. White scale bar, 1 mm. (B) EDTA/resin-treated bone and intervertebral disc tissue; arrow points to annulus fibrosus (AF). White scale bar, 1mm. (C) Higher magnified view of cartilage endplate of the intervertebral disc and bone endplate layers of formic acid–treated tissue. White scale bar, 100 µm. (D) View of cartilage endplate of the intervertebral disc and bone endplate layers of EDTA/resin-treated tissue. White scale bar, 100 µm. Note that the BE1 (bone endplate 1) layer in the EDTA/resin-treated tissue is purple, and this method better distinguishes this layer from the red disc cartilage endplate layer (CE) compared with formic acid–treated tissue. Alcian blue staining overall is more prominent with EDTA/resin compared with formic acid, indicating enhanced preservation of carbohydrate. Abbreviation: EDTA, ethylenediaminetetraacetic acid.
Cost Analysis
The experimental conditions for the above analyses were engineered for the decalcification of several bone pieces simultaneously. To assess the relative costs of the different methods, costs were compared for the decalcification of one 5 g tissue piece each, according to our usual protocol. Prices for reagents were determined from the Sigma-Aldrich website (www.sigmaaldrich.com, January 2020). The reagents were ACS grade, unless otherwise indicated. In our hands, a 5-g tissue specimen requires approximately 8 days in EDTA for demineralization using the standard method, 3 days using formic acid, and 4 days using our EDTA/resin protocol. Costs are detailed in Table 1, showing that the EDTA/resin method delivers an approximately 600% cost reduction for a 5-g tissue specimen compared with the standard EDTA procedure and is similar in expenditure to formic acid demineralization.
Cost Comparison by Method.
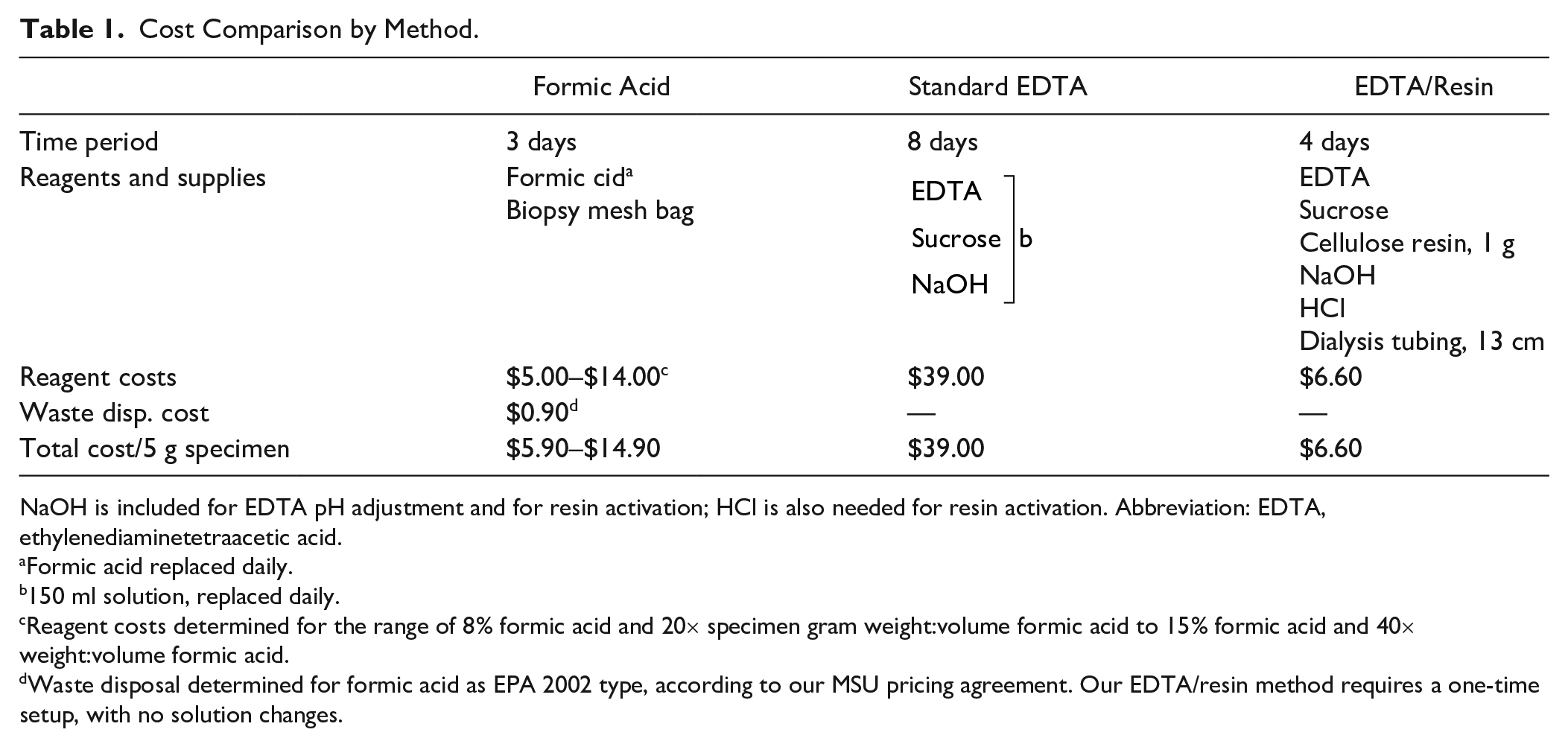
NaOH is included for EDTA pH adjustment and for resin activation; HCl is also needed for resin activation. Abbreviation: EDTA, ethylenediaminetetraacetic acid.
Formic acid replaced daily.
150 ml solution, replaced daily.
Reagent costs determined for the range of 8% formic acid and 20× specimen gram weight:volume formic acid to 15% formic acid and 40× weight:volume formic acid.
Waste disposal determined for formic acid as EPA 2002 type, according to our MSU pricing agreement. Our EDTA/resin method requires a one-time setup, with no solution changes.
There are additional aspects of the methods that impact cost, such as personnel time. It takes approximately 1 hr to activate the resin. For each solution change for the other methods, it takes about 5 min each. The time required to prepare the EDTA solution for both the standard EDTA and EDTA/resin methods also adds additional personnel time that is greater than that required to prepare diluted formic acid. The cost of resin is small (less than $1), but the resin can be retrieved and reactivated, which could further reduce the overall cost of our EDTA/resin method. Also, although we used ACS and reagent grade reagents, lower grade chemicals may also be effective, which would lower overall costs for all methods. Purchase of bulk chemicals would also lower the overall costs for all methods.
We used a 40× weight:volume ratio for the volume of formic acid solution to ensure that our bone pieces were adequately submersed in solution while suspended above the bottom of the glass container and stir bar. Formic acid demineralization, however, has been reported to be effective at half that volume, or 20× the weight:volume of the tissue, using formic acid at 8% dilution. 11 Both scenarios are reflected in Table 1.
Waste disposal cost for formic acid was estimated by the MSU Office of Research Compliance, considering the contracts in place for bulk waste disposal ($1.50/liter for EPA 2002 type). However, as was also estimated by the MSU ORC, if the formic acid must be disposed of and containerized as a “non-reactive lab pack” or as an “acidic waste stream organic,” the cost would be approximately $60 ($100/liter).
Discussion
Tissue mineralization in vertebrates occurs in bone, cartilage, teeth, dermal structures, antlers, and so on and in pathological conditions such as kidney stones and bone cancers. 12 The degree of mineralization and the organic components vary, but the mineral common to all situations is hydroxyapatite. Hydroxyapatite is the primary calcium mineral, but in bone is accompanied by approximately 10% of calcium carbonate and carbonate apatite, combined. 13 Mineralized material constitutes 70% to 80% of total mineralized tissue weight, 13 depending on the tissue type. Although mineralization provides tissue hardness for the formation of internal and external vertebrate scaffolding, it hinders the analysis of those same tissues. Paraffin-based histology and cryoembedding techniques, for example, require that the tissue has a density/hardness near to that of the embedding medium to allow manageable sectioning of the tissue. Analysis, therefore, usually requires demineralization, which breaks the calcium binding to phosphate and hydroxyl groups in hydroxyapatite to yield pliable material. Formic acid does this by protonating the hydroxyapatite phosphate and hydroxyl groups, thus liberating calcium ions. 14 EDTA, in contrast, chelates calcium ions from the bone mineral and draws calcium away from the tissue. 3 We strove to optimize tissue preservation and hence elected to focus on EDTA as the demineralizing agent.
In designing our method, we reasoned that a faster and less expensive improvement over EDTA alone could promote an EDTA-based method over formic acid for bone demineralization. Theoretically, as EDTA pulls calcium from the bone, a cation-exchange resin with a higher affinity for calcium than does EDTA would draw calcium from the solution, thus continuously recycling EDTA, with the resin serving as a calcium sync. A schematic representing this phenomenon is illustrated in Fig. 1B.
Evidence of a higher affinity for calcium by the resin compared with EDTA was observed by the faster rates of decalcification by the addition of cellulose phosphate resin to EDTA compared with EDTA alone (Fig. 2B). This was true whether or not the EDTA solution was replaced daily. While we only tested cellulose phosphate, we suspect that other cation-exchange resins (i.e., Dowex 50 [Sigma], AG 50 [Bio-Rad, Hercules, CA], etc.) may perform at least as efficiently. Cellulose phosphate is a weak cation exchanger, and as such operates over a smaller pH range. It binds cations by its phosphonic groups; strong cation exchangers usually bind by sulfonic groups. It has a unique thread-like structure compared with other ion-exchange beads that may or may not affect its performance relative to other resins. Cellulose phosphate is relatively inexpensive (which is why it was chosen for this study), but has a lower exchange capacity than Dowex 50 or AG 50 (according to their respective product specifications), meaning it has a lower overall binding capacity for cations. The extremely small pore size of dialysis tubing would prevent any of these resins from leaching into the surrounding solution. A further investigation comparing different cation resins for bone demineralization would be an interesting correlate to this study.
In terms of speed alone, formic acid was the fastest demineralization method that we tested. Our EDTA/resin method was the next best procedure for speed, followed by EDTA with daily replacement, EDTA with no solution replacement, and, finally, the PBS-only control. The EDTA/resin method required an additional day for demineralization compared with formic acid, and the other methods did not reach plateau end points at the conclusion of the experiment at 5 days. Although formic acid was superior in speed, however, our histology and immunostaining comparisons call into question whether the sacrifice of tissue integrity is worth the somewhat faster decalcification time frame.
By the histology and immunostaining parameters presented in this study, the EDTA/resin method was superior to formic acid demineralization. These analyses collectively demonstrate the higher degree of tissue preservation and retention of extracellular matrix components when EDTA (with cation-exchange resin to enhance its action) as opposed to formic acid is the demineralizing agent. Overall tissue morphology, as well as the ability to distinguish distinct cartilage tissue layers, is demonstrably better using our method compared with formic acid. A recent study evaluated the effects of hydrochloric acid, formic acid, and EDTA on tissue, DNA, and RNA preservation. 15 Although we did not observe significant differences between EDTA/resin and formic acid with SYTOX green staining of DNA, these researchers, using higher resolution methods, found significant deleterious effects of acid compared with EDTA for nucleic acid preservation. Analogous to our findings, they also observed diminished histology and immunostaining with acid decalcification, especially with hydrochloric acid and with prolonged exposure times. EDTA and formic acid are both very effective at bone demineralization, but EDTA treatment is clearly superior for the preservation of soft tissues and nucleic acid.
As a calcium chelator, EDTA has only minor effects on protein conformation and carbohydrate retention in tissue. It has been observed to cause a loss of glycosaminoglycans (GAGs) in tissue after 12 days of demineralization treatment, but only by approximately 10%. 16 The time reduction inherent in our method likely reduces that loss even more. In contrast, acids can denature proteins by breaking weak hydrogen bonds. Protein unfolding by denaturation can affect antigenicity, as some antibodies recognize three-dimensional epitopes. This could explain our immunofluorescence data where the α-neurofilament antibody was unaffected, but the α-Col2 antibody staining was greatly reduced. Carbohydrates are also an important consideration. They are not crosslinked by formaldehyde fixation 17 and are therefore vulnerable to formic acid extraction. Heating dilute solutions of formic acid is known to hydrolyze carbohydrate chains, resulting in the release of monosaccharides from the chains. 18 It is unknown whether hydrolysis is occurring here, with long exposure time but no heat, but there is ample evidence that formic acid is proficient at carbohydrate extraction/solubilization from both plant 18 and animal 16 tissue, and notably is used in this capacity as a pretreatment for the production of fuel alcohols from plant material. Formic acid can also esterify carbohydrates. 19 It is possible, therefore, that carbohydrate esterification could result in reduced alcian blue staining. Thus, the inferior histological staining with formic acid demineralization could be from carbohydrate solubilization, modification, or both; answering this question would require further study.
Beyond the destructive effects of formic acid on tissue preservation, the resulting impacts on tissue analysis must be considered. The potential protein denaturation and resulting loss of antigenicity are obvious pitfalls to using formic acid, negatively impacting protein analysis after formic acid treatment. Protein analysis, in general, is greatly hindered if demineralization degrades the proteins. This limitation holds true whether the tissue is analyzed by IHC or by other methods that require tissue solubilization such as mass spectrometry. The negative impacts on carbohydrate are at least as significant. Considering that the vast majority of acidic polysaccharide content in cartilage are GAGs, 20 it is safe to assume that the reduced alcian blue staining we observe indicates that GAGs are compromised with formic acid treatment. One of the major applications of bone demineralization is in joint tissue biopsies, and the reduction of GAGs in articular cartilage has long been used as a diagnostic feature of early osteoarthritis. 21 A histological index, the Osteoarthritis Research Society International (OARSI) score, is used to assess the degree of osteoarthritis. 22 This index rates the stage of disease by morphology and by histological identification of GAGs. The OARSI score also stresses that consistent methods must be used to adequately gauge disease severity, but rates (inaccurately, in our view) formic acid–based and EDTA-based decalcification equally. Considering formic acid’s ability to extract carbohydrates from fixed tissue, 16 and our results that show a formic acid–specific loss of alcian blue staining in articular cartilage, we posit that EDTA-based decalcification methods are superior to formic acid for histological osteoarthritis assessment.
Apart from histological staining, retention of carbohydrates in pulverized bone powder is relevant to surgical and cell culture applications. Demineralized bone powder in tissue culture and surgical implants acts as scaffolding material, providing an ad hoc extracellular matrix. Cells function according to their surroundings, and proteins and carbohydrates are major influencing factors in that matrix. It stands to reason that demineralization methods that affect both of those components need to be carefully assessed, and the effects on cells should be tested to ensure that the system performs appropriately. We therefore suggest that our EDTA/resin method be evaluated for demineralized bone powder applications and hypothesize that it would yield improved scaffolding material.
Cost is often an overriding consideration in laboratories. Our EDTA/resin procedure and the lowest range estimate for formic acid demineralization are similar in expense. EDTA/resin, however, constitutes a cost reduction of approximately 6× the cost of the standard EDTA method and less than half of the cost incurred with the formic acid conditions used in this study. Waste disposal costs for formic acid vary considerably between institutions and can greatly increase overall expenditure. Formic acid is a hazardous chemical to lab personnel and to the environment. For this reason, and for the additional practices that must be undertaken in laboratories for formic acid use, EDTA/resin is a cost-effective and more easily manageable demineralization method.
Our laboratory has successfully performed EDTA/resin demineralization on tissue from a number of different species, including chicken, emu, quail, dove, alligator, frog, pig, cow, and human. The method is effective for both trabecular and compact bone, although compact bone can require extra time, usually an extra day (for a 5-g specimen), for demineralization. We have not attempted to demineralize enamel or dentin, but their hydroxyapatite-based mineral suggests they could also be decalcified by this method. In conclusion, the improved tissue morphology, protein and carbohydrate preservation, reduced time compared with EDTA alone, and low cost and environmental impacts (Table 2) show that the EDTA/resin method is a practical and often superior alternative to EDTA alone and formic acid bone demineralization.
Cumulative Comparison of Methods.
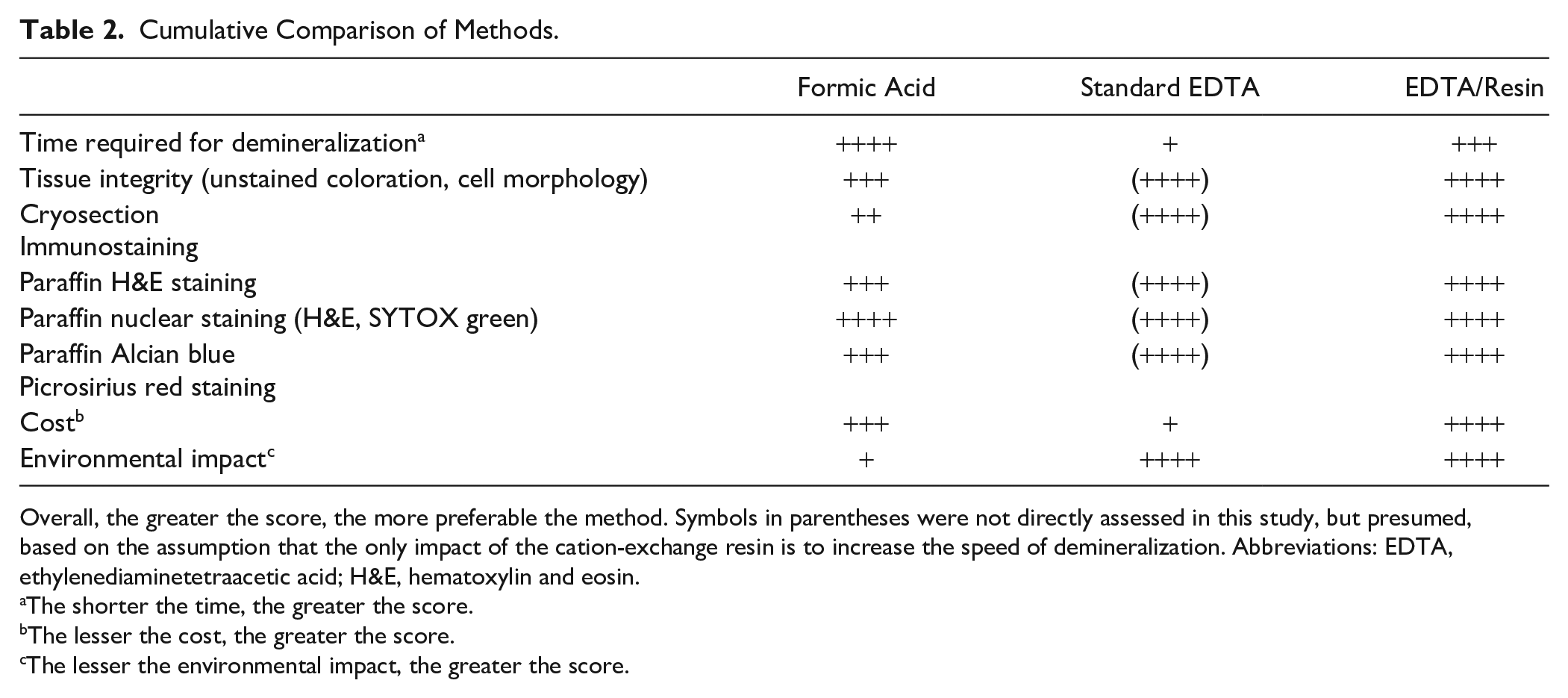
Overall, the greater the score, the more preferable the method. Symbols in parentheses were not directly assessed in this study, but presumed, based on the assumption that the only impact of the cation-exchange resin is to increase the speed of demineralization. Abbreviations: EDTA, ethylenediaminetetraacetic acid; H&E, hematoxylin and eosin.
The shorter the time, the greater the score.
The lesser the cost, the greater the score.
The lesser the environmental impact, the greater the score.
Footnotes
Acknowledgements
We are indebted to Codi Frances at the MSU Student Health Center for providing X-ray images. Additional thanks go to Kevin Surya and Dr. Roger Bradley for discussion of the manuscript and to Patrick Ryan for providing MSU waste disposal costs. We also sincerely thank Gerry Ohrstrom and George Lucas for their kind donations to the project.
Competing Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A provisional patent application has been submitted by D.J.R. and J.R.H. for the commercial production of resin/dialysis tubing bone demineralization packets. The remaining authors claim no financial or non-financial competing interests.
Author Contributions
EN performed the decalcification rate experiments and analysis, and the histology stains and cowrote the manuscript. MEJ performed the formic acid decalcifications and tissue embedding and sectioning. JRH contributed to experimental planning and funding acquisition. DJR performed the immunofluorescence experiments, cowrote the manuscript, and supervised the project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by an MSU Faculty of Excellence grant to D.J.R. and by private contributions from Gerry Ohrstrom and George Lucas. The MSU Histology Core Facility was funded by National Institutes of Health IDeA Program COBRE Grant GM110732.
