Abstract
Introduction
Meningococcal disease is associated with high mortality. When acute kidney injury (AKI) occurs in patients with severe meningococcal disease, it is typically attributable to sepsis, although meningococcal disease and lipopolysaccharide release are rarely investigated. Therefore, we evaluated renal tissue in a mouse model of meningococcal disease.
Methods
Female BALB/c mice were induced to AKI by meningococcal challenge. Markers of renal function were evaluated in infected and control mice.
Results
In the infected mice, serum concentrations of tumor necrosis factor alpha, interferon gamma, interleukins (IL-1β, IL-2, IL-4, IL-5, IL-6, IL-10, and IL-12), and granulocyte–macrophage colony-stimulating factor were elevated, as was renal interstitial infiltration with lymphocytes and neutrophils (p < 0.01 for the latter). Histological analysis showed meningococcal microcolonies in the renal interstitium, without acute tubular necrosis. Infected mice also showed elevated renal expression of toll-like receptor 2, toll-like receptor 4, and Tamm–Horsfall protein. The expression of factors in the intrinsic pathway of apoptosis was equal to or lower than that observed in the control mice. Urinary sodium and potassium were also lower in infected mice, probably due to a tubular defect.
Conclusion
Our findings corroborate those of other studies of AKI in sepsis. To our knowledge, this is the first time that meningococci have been identified in renal interstitium and that the resulting apoptosis and inflammation have been evaluated. However, additional studies are needed in order to elucidate the mechanisms involved.
Introduction
Meningococcal disease continues to be a public health concern. The clinical spectrum of such disease ranges from meningitis to meningococcemia, and the associated mortality is as high as 20%. 1 In Brazil, more than 23,000 cases of meningococcal disease were reported to the National Health Ministry between 2008 and 2018, as confirmed in the database of the Information Technology Department of the Brazilian Unified Health Care System (http://tabnet.datasus.gov.br/cgi/tabcgi.exe?sih/cnv/niuf.def, last accessed 23 April 2018). After meningococci reach the nasopharynx and are internalized, they are recognized by pathogen-associated molecular patterns and damage-associated molecular patterns, which bind to pattern-recognition receptors to induce innate immunity.2,3 It is known that the members of one group of pattern-recognition receptors, the toll-like receptors (TLRs), especially TLR2, TLR4, and TLR9, are involved in meningococcus recognition.4 After the meningococci have been recognized, there is a cascade of reactions, including cytokine release and leukocyte adhesion to endothelial cells, which typically culminates in apoptosis. However, some studies have shown that meningococci, like other bacteria, can block the apoptotic cascade in the intrinsic pathway.5–7
Many patients with meningococcemia evolve to septic shock and, as a consequence, acute kidney injury (AKI). Some patients with AKI develop tubular lesions, leading to electrolyte excretion disorders.8,9 It remains unclear whether tubular disorders occur because of sepsis per se or because of a local effect of the meningococci. Although meningococcal DNA has been found in kidney specimens obtained from animals and in postmortem tissue samples,10,11 the presence of meningococci has not been correlated with tubular defects. Therefore, the aim of this study was to analyze renal inflammation and apoptosis in a mouse model of meningococcal disease, as well as to characterize meningococci in renal tissue, as described in previous studies.12–14
Material and methods
Animals and experimental protocol
Thirty female BALB/c mice were divided into three groups of 10 mice each—control, comprising mice submitted to a sham procedure; low-dose (LD), comprising mice challenged with 0.5 mL containing 1 × 107 serogroup B meningococci and 10 mg of human holotransferrin administered intraperitoneally, and high-dose (HD), comprising mice challenged with 0.5 mL containing 5 × 107 serogroup B meningococci and 10 mg of human holotransferrin—as described elsewhere.15–17 The sample size was determined with the objective of using the minimum number of animals, thus complying with the best practices in animal use. Because they were syngeneic mice, the results obtained are expected to be similar among them. There were no statistically significant differences among the animals in terms of age and weight. The meningococcal strain was NZ98/254. After a 24-h observation period, the mice were euthanized by exsanguination. Blood samples, urine samples (obtained by bladder puncture), and tissue samples were then collected. Kidney tissue samples were submitted to histological analysis, flow cytometry, and Western blotting. Tissue samples were stored in RPMI medium at −80°C.
All experiments were performed in accordance with the requirements of the United Kingdom Home Office (Scientific Procedures) Acts, 1986. The study was approved by the Research Ethics Committees of the University of São Paulo School of Medicine (number 109/14), in the city of São Paulo, Brazil, and Public Health England (number PPL 30/2906), Porton Down, in the city of Salisbury, United Kingdom. Animals were inoculated and some of the experiments were performed at the Public Health Laboratory Service Centre for Applied Microbiology and Research (Porton Down, Salisbury, UK).
Kidney sections
Half of each kidney was fixed in 10% formalin, after which it was blocked in paraffin and cut into sections. Some sections were stained with hematoxylin–eosin (H&E) and analyzed by two pathologists, working independently. For immunohistochemistry, sections were deparaffinized, endogenous peroxidase was blocked using a commercial kit (spring Bioscience, Pleasanton, CA), and the sections were incubated overnight, with an anti-myeloperoxidase antibody (ab9535; Abcam, Cambridge, MA), an anti-CD3 antibody (1:100, DAKO, California, USA), and an anti-galectin-3 (anti-MAC-2) antibody (1:12,500, Cedarlane Laboratories, Burlington, Ontario, Canada). For visualization, we used anti-rabbit, anti-goat, and anti-rat horseradish peroxidase-conjugated polymers, respectively (DHRR; spring Bioscience) and commercial 3,3′ diaminobenzidine.
Meningococcal antigens were visualized by immunofluorescence. After deparaffinization, sections were blocked with bovine serum albumin 6% (to avoid nonspecific antigen binding) and incubated overnight with anti-α-actin antibody (1:800; Sigma-Aldrich, St Louis, MO). The sections were then incubated with anti-mouse antibody (Alexa Fluor 546; Life Technologies, Carlsbad, CA) and finally with fluorescein isothiocyanate-conjugated anti-Neisseria meningitidis antibody (1:50; Abcam).
Apoptosis
One half of one kidney from each animal was homogenized in a cell dissociator (GentleMACS; Miltenyi Biotec, Bergisch Gladbach, Germany). The suspension was filtered, the supernatant was treated with a red cell lysis solution, and cells were counted after staining with trypan blue. The cells were diluted in PBS and fixed with methanol. After three washes, the primary antibodies were added, the samples were washed again, and the secondary antibody was added. Flow cytometry was performed with antibodies against p53 (ab26; Abcam), caspase 3 (31A1067; Abcam), cytochrome c (37BA11; Abcam), BAX (ab32503; Abcam), and caspase 9 (ab32539; Abcam). The cells were marked with K-cadherin, which is expressed exclusively in epithelial cells of the adult kidney. The results obtained by flow cytometry (Beckman Coulter, Fullerton, CA) were analyzed with Summit software, version 4.3.
We also assessed renal apoptosis by terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) assay, using a commercial in situ cell death detection kit (Roche Diagnostics, Mannheim, Germany). Positive cells were quantified by an independent investigator, who analyzed all fields.
Cytokine analysis
Blood samples were pooled and cytokine concentrations determined with a commercial kit (Mouse Cytokine Magnetic 10-Plex Panel, LMC0001M; Invitrogen/Life Technologies, Carlsbad, CA). The following cytokines were analyzed: granulocyte–macrophage colony-stimulating factor, interleukin (IL)-1β, IL-2, IL-4, IL-5, IL-6, IL-10, and IL-12
TLR2, TLR4, and Tamm–Horsfall Analysis
Kidney samples were analyzed on 10% polyacrylamide minigels, with 50 μg of protein per lane, and transferred to PVDF membranes. Blots were blocked with 5% nonfat milk in TBS for 1 h and incubated overnight with the following primary antibodies: anti-TLR2 (goat, 1:500; R&D Systems, Minneapolis, MN); anti-TLR4 (1:500; Santa Cruz Biotechnology, Santa Cruz, CA); and anti-Tamm–Horsfall (1:500, Millipore, Billerica, MA). After three washes, the membrane was incubated with horseradish peroxidase-conjugated secondary antibodies: anti-mouse (1:4000; Abcam) for TLR4; anti-goat (1:10,000; Sigma) for TLR2; and anti-sheep (1:2000, R&D Systems, Minneapolis, MN) for Tamm–Horsfall protein (THP). Chemiluminescence was detected with a gel documentation system (Alliance; Uvitec, Cambridge, UK), and proteins were quantified with Scion Image software, version 4.0.3. (Scion Corporation, Frederick, MD). The results were normalized to β-actin and are presented as percentages of the control values.
Biochemical analysis
Urine samples were centrifuged for 30 min at 4000 g. Urinary levels of sodium and potassium were measured with an electrolyte analyzer (EasyLyte Na/K; Medica Corporation, Bedford, MA).
Statistical analysis
Differences among the means of multiple parameters were analyzed by one-way analysis of variance, followed by the Newman–Keuls test. Quantitative data are expressed as mean ± standard deviation. The statistical analysis was performed with GraphPad Prism software, version 8.2.0 (GraphPad Software, Inc, San Diego, CA). Values of p < 0.05 were considered statistically significant.
Results
Study period
Among the 30 mice studied, there were five (two in the LD group and three in the HD group) that died within the first 20 h post challenge. Because the remaining mice were extremely lethargic, we decided to terminate the experiment at hour 22.
Histological analysis
Except for small foci of basophilic tubules, no abnormalities were detected in the kidneys of the control group mice. However, bacterial microcolonies were detected in the kidneys of mice in the two infected groups. Although most of the microcolonies were located in the cortex, some were detected in the medulla. In most cases, the bacterial aggregates were in interstitial tissue, although some were within tubular cells and there were signs suggestive of bacteria within the lumina of tubules. Their presence did not provoke a detectable inflammatory response or acute tubular necrosis. In one case, there was a focus of tubular degeneration (Figure 1). [insert Figure 1.] Kidney sections and distribution of microcolonies of meningococci in mice in the low-dose (LD) group (challenged with 1 × 107 serogroup B meningococci and transferrin) and high-dose (HD) group (challenged with 5 × 107 serogroup B meningococci and transferrin). (A) Subcapsular focus of basophilic tubules; (B) interstitial bacterial microcolony (arrow) and intracellular bacteria in the renal cortex (double arrow); (C) focus of degenerated cells in the cortex; (D) bacteria in the interstitial tissue appear to have invaded the adjacent proximal convoluted tubule cells (H&E staining, magnification, × 100); and (E) bar graph showing the number of colonies per section.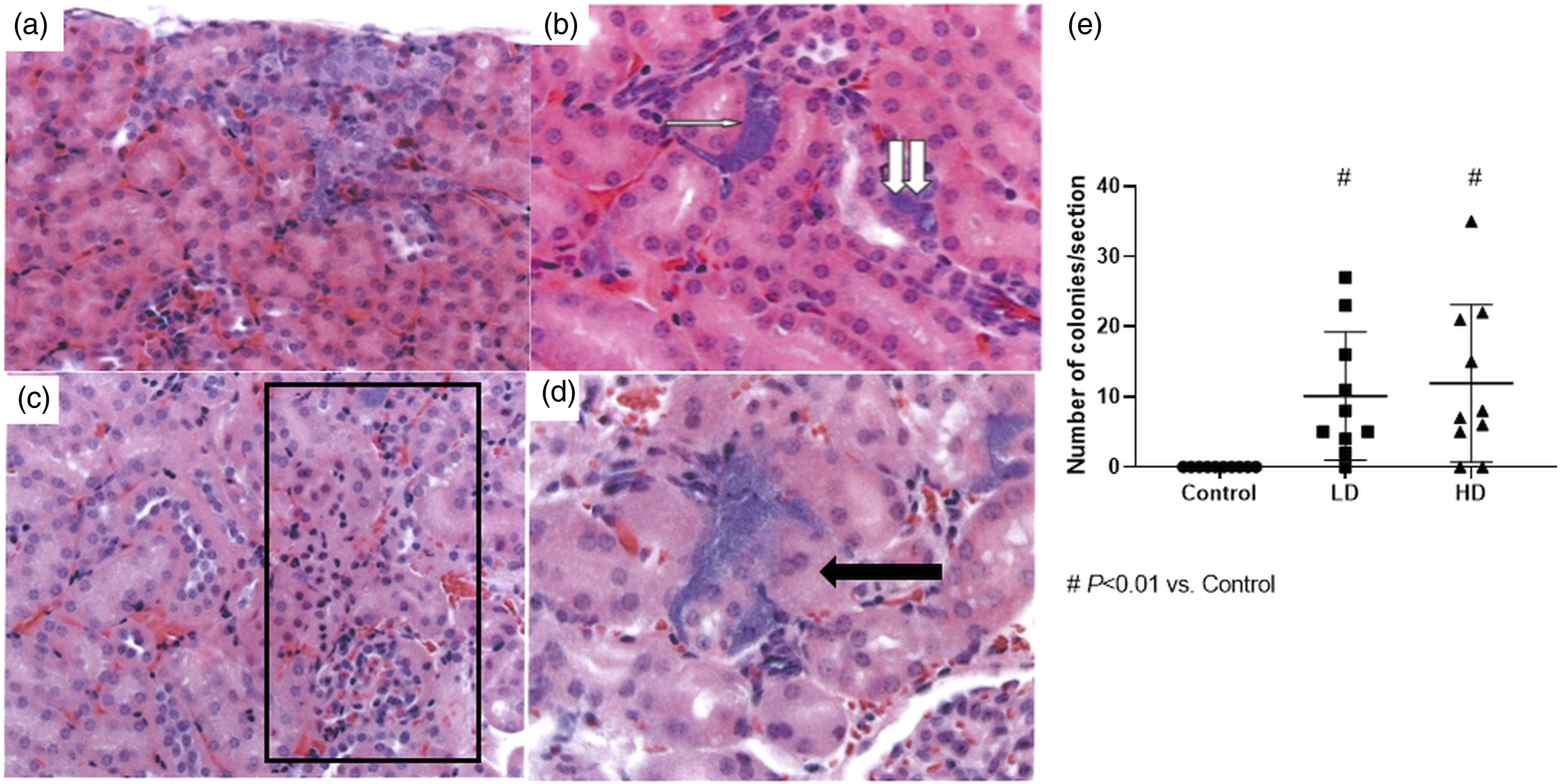
Immunofluorescence
Meningococci were detected in the renal interstitium. Colocalization with α-actin showed that there were no bacteria within the blood vessels (Figure 2). [insert Figure 2.] Colocalization of Neisseria meningitides. (A) Control group; (B) low-dose group; and (C) high-dose group. Note the blood vessel (double white arrow). Note also the meningococcal colony (single arrow) in the renal interstitium. Magnification, × 400.
Leukocyte infiltration
Infiltration of the renal tubulointerstitium by neutrophils, lymphocytes, and macrophages, as quantified by determining the expression of myeloperoxidase, CD3, and galectin-3 (MAC-2), respectively (Figure 3), was significantly greater in the LD and HD group mice than in the control group mice. Although lymphocyte infiltration was more pronounced in the HD group than in the LD and control groups, the difference was not statistically significant (Figure 3). [insert Figure 3.] Evaluation of leukocytes in renal tissue: (A) Anti-myeloperoxidase (anti-MPO) staining (black arrow) in the control and high-dose (HD) groups; (B) anti-MPO+ cell counts in the control, low-dose (LD), and HD groups; (C) anti-CD3 staining (black arrow) in the control and high-dose (HD) groups; (D) anti-CD3 + cell counts in the control, LD, and HD groups; (E) anti-MAC-2 staining (black arrow) in the control and high-dose (HD) groups; and (F) anti-MAC-2 cell counts in the control, LD, and HD groups. Magnification, × 400.
TLR2, TLR4, and Tamm–Horsfall expression
Figure 4 shows the protein expression of TLR2, TLR4, and Tamm–Horsfall in kidney tissue. TLR2 and TLR4 were more strongly expressed in the LD and HD groups than in the control group, although the difference was statistically significant only for TLR2 (p < 0.001). [insert Figure 4]. In addition, the THP expression was significantly higher in the LD and HD groups than in the control group (Figure 4). Bar graphs and Western blots, showing expression of toll-like receptor 4 (TLR4), toll-like receptor 2 (TLR2), and Tamm–Horsfall protein (THP) in kidney tissue samples from mice in the control, low-dose (LD), and high-dose (HD) groups.
Cytokines
Figure 5 shows the concentrations of the cytokines analyzed in sera. Because it was not possible to evaluate each animal and the samples were pooled, we were unable to perform a statistical analysis. However, it was clear that the concentrations of all of the cytokines tested were greater in the mice challenged with N. meningitidis. [insert Figure 5.] Serum concentrations of cytokines. IL, interleukin; GM-CSF, granulocyte–macrophage colony-stimulating factor.
Evaluation of apoptosis
Figures 6(a) and (b) show the TUNEL-stained (apoptotic) cells and their counts, respectively. The number of apoptotic) cells was significantly greater in the HD group than in the LD and control groups (11.7 ± 6.0 cells/mm2 vs. 3.7 ± 1.1 and 2.7 ± 1.0 cells/mm
2
; p < 0.01, respectively). Figure 6(c) shows the flow cytometry counts of proteins within the intrinsic pathway. [insert Figure 6.] (A) Apoptosis analysis through terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) assay and marked cell (arrow) in the low-dose (LD) and high-dose (HD) groups (magnification, × 40); (B) graph bar showing the number of TUNEL-stained cells in kidney tissue; and (C) flow cytometry of stained cells.
Urine analysis
We determined the concentrations of sodium and potassium in pooled urine samples. The results are presented in Figure 7. [insert Figure 7.] As can be seen in the figure, the concentrations of sodium and potassium were lower in the LD and HD groups than in the control group, indicating that there was renal tubular dysfunction in the former. Sodium and potassium concentrations in urine samples obtained from mice in the control, low-dose (LD), and high-dose (HD) groups.
Discussion
To our knowledge, this is the first time that intact meningococci have been identified within the renal cortical and medullary interstitial spaces, as well as (potentially) within the tubular lumina. This finding could explain the tubular dysfunction, characterized by lower concentrations of sodium and potassium in the urine, observed in the animals induced to AKI in our study. However, the differences in sodium and potassium concentrations could also be attributable to the fact that the animals were sick. Other studies of meningococcal disease have shown deficiencies in electrolyte concentrations, although the mechanisms involved have not been elucidated.8,9,18 Acute tubular necrosis was not seen in our analysis, which corroborates the findings of studies employing a cecal ligation and puncture model of sepsis.19,20
When there is an inflammatory process in renal tissue (or elsewhere), neutrophils and macrophages are the first immune cells recruited. In the present study, neutrophil and macrophage infiltration was significantly more pronounced in the infected mice than in the control mice. This finding was expected, given that the concentrations of cytokines, most of which participate in neutrophil adhesion and macrophage infiltration were higher among the mice challenged with N. meningitidis. Nevertheless, there were no differences between the groups in terms of infiltration by lymphocytes. That can be explained by the fact that lymphocytes are recruited in the later stages of inflammation and that our study was focused on the acute phase.
The expression of TLR2 and TLR4, which are recognized as receptors for pathogen-associated molecular patterns, was significantly higher in our infected mice. It is known that TLR4 is the major lipopolysaccharide receptor, triggering a cascade that culminates in apoptosis. However, some genera of bacteria, including Neisseria, can inhibit apoptosis via the intrinsic pathway. It has been demonstrated that PorB, a surface protein of meningococci, can alter the polarization of mitochondrial membrane, preventing Ca2+ efflux.5,6 In contrast, Deo et al. 21 demonstrated that PorB could trigger the apoptotic cascade in Neisseria gonorrhoeae. Therefore, we cannot rule out the possibility that other proteins at different trigger points impede the pathway. If so, that could explain our finding of lower BAX activity in infected mice. The low activity of cytochrome c and caspase nine could be due to inhibition by PorB in the mitochondrial membrane. Finally, the TUNEL assay is a nonspecific method of investigating apoptosis, 22 because it stains for DNA fragmentation only. Therefore, the higher numbers of TUNEL-stained cells in the infected mice could be attributed to intense gene transcription caused by the intense inflammation process. Also known as uromodulin, THP is a glycoprotein expressed exclusively by renal tubular cells lining the thick ascending limb of the loop of Henle. 23 It is a frequently used marker for cortical and medullary thick ascending limb renal segments, all of which strongly express TLR4. These segments also showed a significant increase in THP in a cecal ligation and puncture model of sepsis. Therefore, it is possible that THP is a ligand of TLR4. 24 El-Achkar et al. demonstrated that THP plays a role in TLR4-mediated Cox-2 signaling in a sepsis model. 25 Further studies are needed to establish firm, direct links among THP, TLR4, and meningococcal disease.
Our study has some limitations. Because of the small quantity of urine and blood samples, we were unable to assess all markers of renal function. In addition, the extrinsic pathway was not examined and we therefore do not know the importance of its role in AKI. However, to our knowledge, this is the first time that meningococci and the consequent apoptosis have been evaluated in renal tissue.
Conclusion
Our findings corroborate those of other studies of AKI in sepsis. Nevertheless, further studies are needed in order to elucidate the mechanisms involved in the apoptotic cascade resulting from meningococcal disease.
ORCID iD
Karin R Kolbe https://orcid.org/0000-0002-9681-8151
Stephen Thomas https://orcid.org/0000-0003-2368-8058
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, São Paulo Research Foundation; Grant no. 2010/19012–0); and by the Laboratórios de Investigação Médica (Medical Investigation Laboratories) of the Faculdade de Medicina da Universidade de São Paulo (University of São Paulo School of Medicine). L. Andrade is the recipient of a grant from the Brazilian Conselho Nacional de Desenvolvimento Científico e Tecnológico (National Council for Scientific and Technological Development; Grant no. 301193/2016–2019).
Ethics approval
Ethical approval for this study was obtained from Research Ethics Committees of the University of São Paulo School of Medicine (number 109/14), in the city of São Paulo, Brazil, and Public Health England (number PPL 30/2906), Porton Down, in the city of Salisbury, United Kingdom.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation. All experiments were performed in accordance with the requirements of the United Kingdom Home Office (Scientific Procedures) Acts, 1986.
