Abstract
Grading of pancreatic neuroendocrine tumors (pNETs) is currently based on mitotic rate and Ki67 proliferation index. Phosphohistone-H3 (PHH3) is an effective marker for mitosis that has been proposed to use in grading various NETs. It remains unclear which method more accurately predicts grade and clinical outcome. Cases of pNET were evaluated using immunohistochemical stains for Ki67 and PHH3. In addition, each case was evaluated for necrosis, lymphovascular invasion, and perineural invasion and compared with stage. R project statistical analysis was used for comparisons. Sixty-three cases were included in the study including 29 males and 34 females (M:F 0.9) with a median age of 59 years (ranging 34–84). There was not a significant discrepancy in the stratification of tumor grades for Ki67 and PHH3. PHH3 significantly predicted lymph node metastasis (p=0.041). Necrosis correlated with overall survival (p=0.017). The results suggest that PHH3 is an effective marker for determining mitotic activity and can be used alternative to Ki67. In addition, necrosis may be included in the reporting of pNET as it may play a prognostic role. Larger scale studies are warranted to understand the biology and behavior of these tumors.
Introduction
Neuroendocrine tumors (NETs) are distinct entities of epithelial origin that arise in many organ systems. The nomenclature, staging, and grading of NETs throughout all organ systems varies and has frequently evolved in the past decade. 1 Pancreatic NETs (pNETs) are a particularly rare entity within this group of malignancies with about 1,000 new cases per year in the United States. 2 Data on the epidemiological course of pNET is limited, but the incidence appears to be rising.3,4 In 2010, the World Health Organization (WHO) released the latest classification guidelines for these tumors 5 that divided NETs into three grades. These grades are further stratified into well-differentiated NETs [low grade (G1) and intermediate grade (G2)] or poorly differentiated NETs [high grade (G3)]. According to these guidelines, the grading of such tumors requires reporting of mitotic index. The determination of mitotic index is obtained through mitotic figure count on hematoxylin and eosin (H&E)–stained specimens as well as immunohistochemical staining with Ki67. The percentage of cells that stain positively correlates to tumor proliferation, and therefore determines the grade of the tumor. The combination of mitotic figure count and Ki67 proliferation rate has been shown to have a fairly accurate prognostication of NETs.6 –9 However, the WHO grading scheme (Table 1) creates a narrow cutoff particularly between the well-differentiated entities (G1 and G2). Ki67 protein is present in cell nuclei in G1, S, G2, and M phase cell cycles. The major limitation of Ki67 immunohistochemistry (IHC) staining is the subjectivity in interpretation in addition to possible nonspecific staining in apoptotic cells. Like Ki67, IHC marker for phosphohistone-H3 (PHH3) stains cells undergoing mitosis and can specifically aid in determining mitotic activity. PHH3 may have advantages over Ki67 as it does not highlight cells undergoing cell death or apoptosis.10,11 This can give PHH3 a possible advantage over Ki67 index and routine H&E mitotic count as it will lessen the amount of apoptotic figures misidentified as mitotic figures and possibly make it a better tool to assess grading in NETs.
Pancreatic Neuroendocrine Tumors Grading Scheme According to World Health Organization 2010 Classification of the Digestive System.
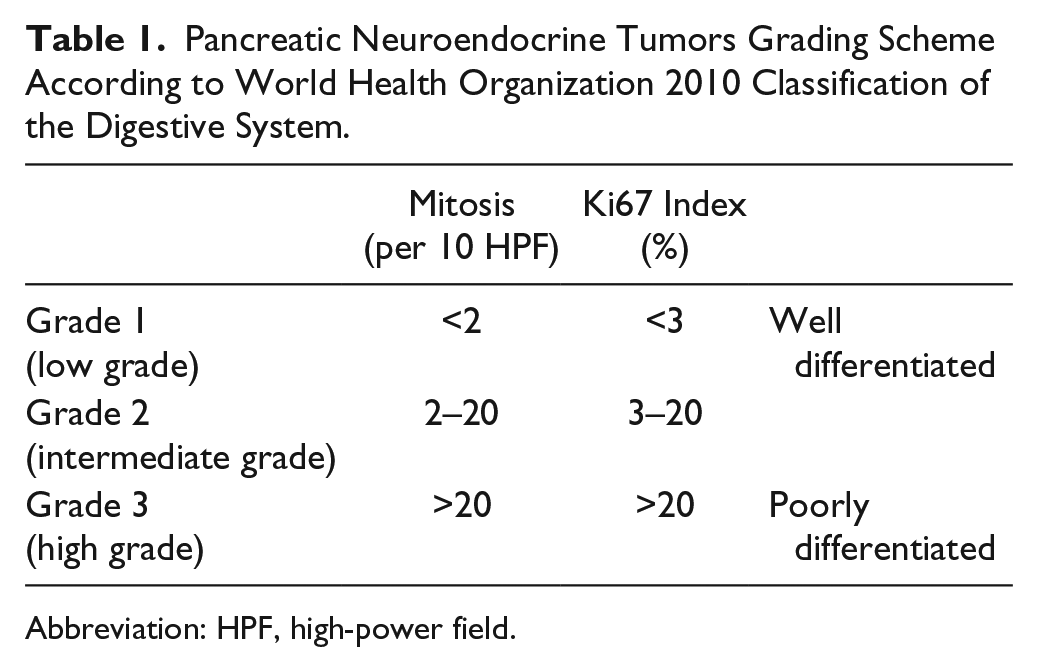
Abbreviation: HPF, high-power field.
PHH3 has been studied in other tumors where mitotic count is important for classification, such as meningioma, melanoma, and astrocytoma.12–14 The investigation of PHH3 in the setting of pNETs has been limited. In these limited studies, the usefulness of PHH3 as a tool for assessing mitotic count has been examined.15,16
In the current study, we aim to compare mitotic count determined by PHH3 with the Ki67 index and H&E-based mitotic count for the grading and prediction of clinical outcome for pNETs. In addition, because the presence of necrosis, lymphovascular invasion (LVI), or perineural invasion (PNI) is not included in the current staging scheme, determining those parameters in conjunction with mitotic proliferation may additionally add to more accurate staging and prognostication. Therefore, we also studied the role of tumor necrosis, PNI, and LVI in prognostication.
Materials and Methods
Tissue Samples
The study was approved by the University of Alabama at Birmingham (UAB) Institutional Review Board in accordance with the Declaration of Helsinki. Patients who were diagnosed and treated with non-functional pNET between January 2006 and December 2013 were included in this study. Pathology results were obtained in UAB Cerner systems and clinical chart review using electronic medical records was performed to retrieve patient demographics; treatment modalities and clinical outcome including recurrence were assessed from clinical chart review. Patients with excisional biopsy or resection were included and biopsy only cases were excluded. Also excluded were cases in which pancreatic adenocarcinoma was concurrently present. Formalin-fixed, paraffin-embedded (FFPE) blocks were retrieved from the UAB Pathology Archives.
Immunohistochemistry
Five-mm sections were obtained from FFPE block preparations. Heat-induced epitope retrieval was applied in a sodium citrate buffer (Ventana Medical Systems; Tucson, AZ), at 97C, for 20 min. Staining was performed using an antibody raised against PHH3 (rabbit polyclonal, cat. no 369A, 1:100; Cell Marque, Rocklin, CA) using a semi-automated immunostainer (Dako Autostainer Link 48; Dako, Carpinteria, CA) and an Envision FLEX HRP system (Dako, Carpinteria, CA). The antibody for Ki67 (rabbit monoclonal, predilute; Ventana Medical Systems) was performed using an automated immunostainer (Benchmark XT, Benchmark Ultra; Ventana Medical Systems; and Ultravision LP polymer system; Labvision, Fremont, CA). Diaminobenzidine tetrachloride was used to visualize the antibody–antigen complex. The tissue was counterstained with hematoxylin. Appropriate positive and negative controls were included. Strong nuclear immunoreactivity of PHH3 or Ki67 was regarded as positive staining. PHH3 antibody was validated by using 10 positive (tissues with identifiable mitotic activity by H&E stains) and negative (tissues without identifiable mitotic activity by H&E stains) controls, respectively, before the study.
Mitotic Index, Proliferation Index, and Histologic Grading
Routine H&E slides were reviewed for confirming the diagnosis and grading by two surgical pathologists. These cases were graded based on the current WHO guidelines 5 using mitotic count on (H&E) sections and Ki67 proliferation index using IHC. Cases of pNET were assessed for the presence of necrosis, PNI, and LVI.
On H&E sections, the mitotic index was scored as the total number of mitotic figures in non-overlapping 10 consecutive high power fields (HPFs; original magnification ×400) where they were most prevalent. Similarly, the PHH3 mitotic index was scored as the total number of tumor cells in 10 consecutive HPFs where the immunolabeling was most prevalent. 17 The scoring for mitotic counts was performed based on the field diameter and is as follows: ≤9 (score of 1), 10 to 19 (score of 2), and ≥20 (score of 3). The Ki67 proliferation index was scored as the mean percentage of Ki67-labeled tumor cells from 10 successive HPFs in most prevalent fields.
Three grades (I–III) based on mitotic count and Ki67 were assessed and compared with PHH3 results. These grades were compared with disease recurrence and the presence of metastases to lymph nodes (LNs) at the time of diagnosis to determine which method better predicts clinical outcome. Disease recurrence was defined as the presence of new local recurrence or new lesions elsewhere documented in the electronic charts. The presence of LN metastases was determined through histomorphologic review. In addition, the presence of necrosis, LVI, or PNI was compared with disease recurrence, LN metastases as well as the stage of disease.
Statistical Analysis
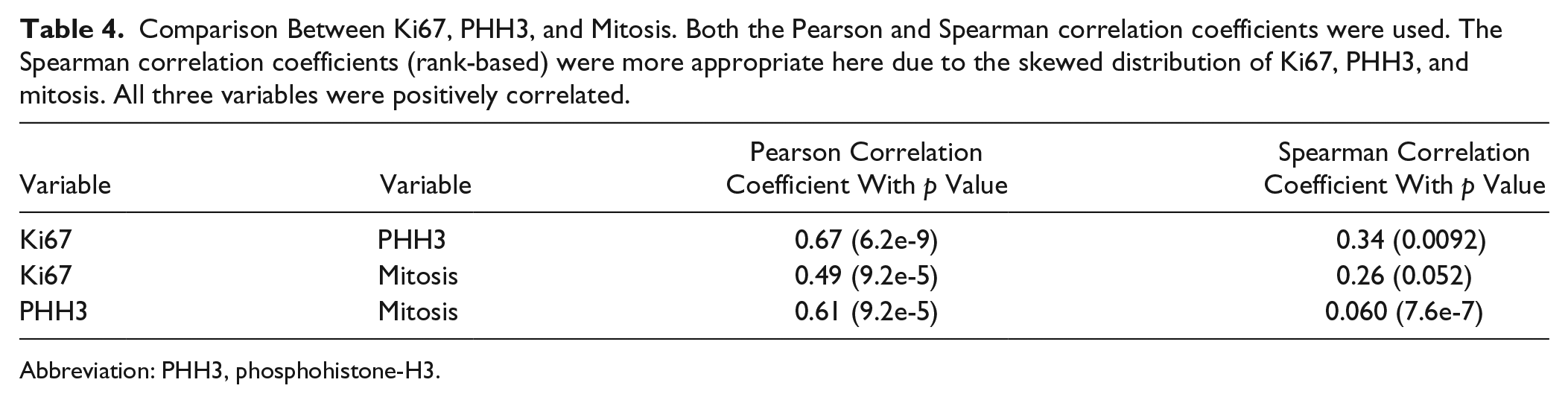
Comparison between Ki67, PHH3, and mitosis was performed using the Pearson and Spearman correlation coefficients. Also, the comparison of Ki67, PHH3, and mitosis to the stage, LN metastasis, recurrence, disease-free survival (DFS), and overall survival (OS) was performed. For the analysis of stage, LN metastasis, and recurrence, the Kruskal–Wallis Test [a non-parametric version of analysis of variance (ANOVA)] was used. The Fisher exact test was used to compare PNI, LVI, necrosis, and stage to the recurrence. For the survival analysis, the Cox proportional hazards regression model was used. We performed the statistical analysis using R project (http://www.r-project.org/). A p value of 0.05 was considered statistically significant.
Results
A total of 63 cases with pNET excisions were included in the study. The clinicopathological findings and tumor characteristics are summarized in Table 2. There were 29 males and 34 females (M:F 0.9) with a median age of 59 years (ranging 34–84). The DSF rate was 87% with 7 (11%) patients exhibiting documented metastatic disease/recurrence. Of these seven cases, 6 (86%) developed metastatic disease to the liver. The remaining case involved metastasis to the celiac trunk. One case had both pancreatic recurrence and liver metastases. One disease-related death due to extensive metastatic disease leading to multisystem organ failure was identified. Tumor size varied from 0.9 to 9.5 cm. Multiple primary tumor nodules were present in four cases (6%). The pancreatic tail was the most common site of presentation (n=40, 51%) followed by the pancreatic head (n=20, 27%), and the uncinate (n=3, 5%). LN metastases were present in nine cases, ranging from one to eight positive LNs identified. Staging based on American Joint Committee on Cancer 18 guidelines detected 22 T1 lesions (35%), 26 T2 lesions (41%), 14 T3 lesions (22%), and one T4 lesion (2%), and the staging and grading results are summarized in Table 3. Necrosis was identified in six cases (10%), LVI was present in 40 (64%), and PNI was present in 39 (62%) of the cases.
Summary of Clinicopathological Data and Tumor Characteristics.
The Grading of 63 Cases Based on Mitotic Count, Ki67, and PHH3 Compared With Disease-Free Survival.

Abbreviation: PHH3, phosphohistone-H3.
Grading based on H&E mitotic count, Ki67, and PHH3 was determined (Fig. 1). Grading using mitotic count on H&E-stained slides yielded 39 grade 1 (62%), 20 grade 2 (32%), and four grade 3 (6%) diagnoses. Grading with Ki67 yielded 33 grade 1 (52%), 26 grade 2 (41%), and four grade 3 (6%) diagnoses. Grading using PHH3 yielded 36 grade 1 (57%), 24 grade 2 (38%), and three grade 3 (5%) diagnoses. There was no statistical significance in grading using any of the methods (p>0.05; Table 2). Comparisons of Ki67 index, PHH3 index, and mitotic index were positively correlated (Table 4 and Fig. 2).
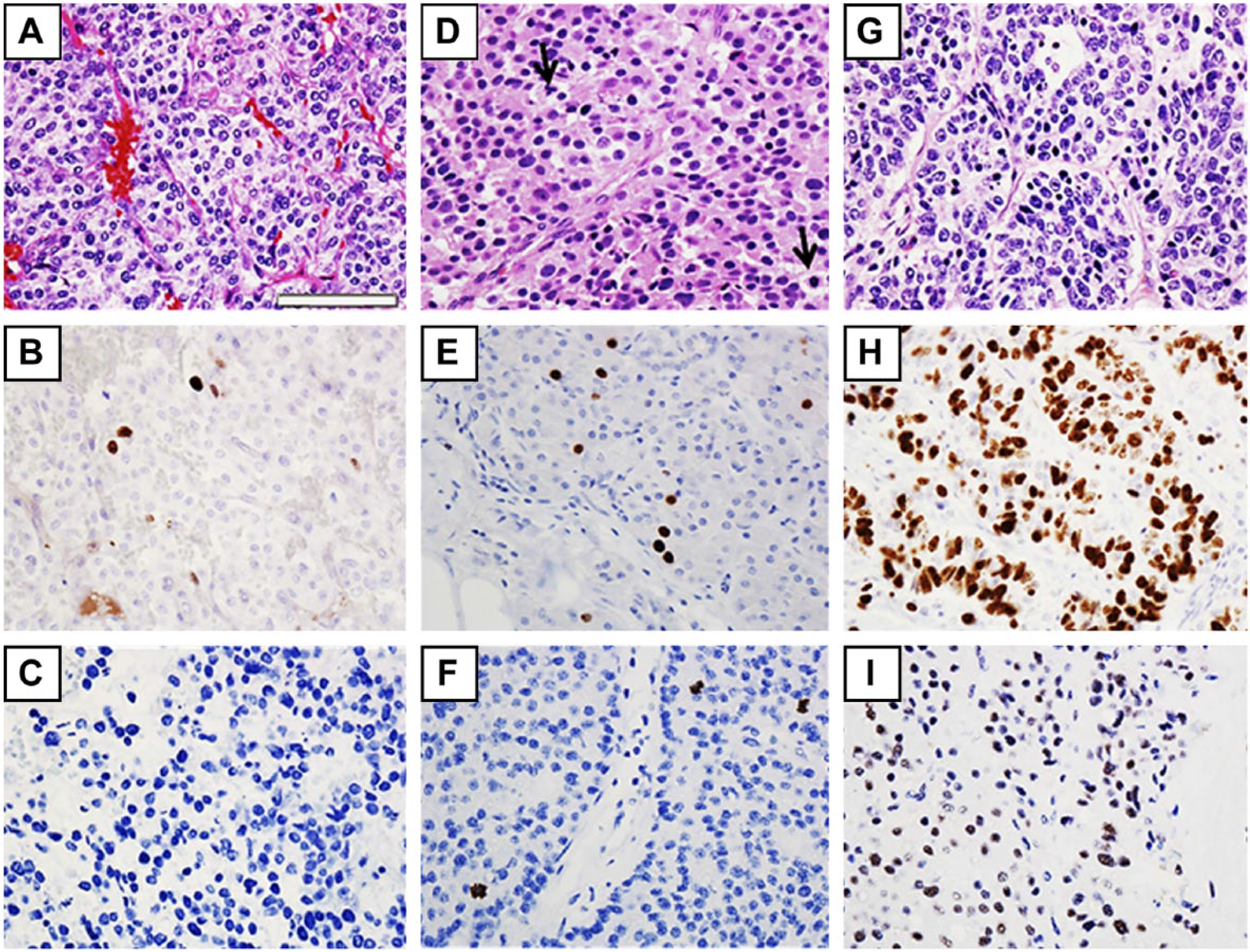
Grade 1 pancreatic neuroendocrine tumor (A) with Ki67 with a proliferation index of <2% (B) and phosphohistone-H3 (PHH3) showing no mitotic activity (C). The H&E stain of grade 2 pancreatic neuroendocrine tumor with two mitotic figures (arrows; D) showing increased Ki67 with a proliferation index of 3–20% (E) and increased mitotic activity by PHH3 (three mitoses per high power field; F). High-grade (grade 3) tumor (G) with >20% proliferation rate via Ki67 (H) and increased mitotic figures highlighted by the PHH3 stain (I). The scale size is the same for all the figures. Scale, 90 µm.
Comparison Between Ki67, PHH3, and Mitosis. Both the Pearson and Spearman correlation coefficients were used. The Spearman correlation coefficients (rank-based) were more appropriate here due to the skewed distribution of Ki67, PHH3, and mitosis. All three variables were positively correlated.
Abbreviation: PHH3, phosphohistone-H3.
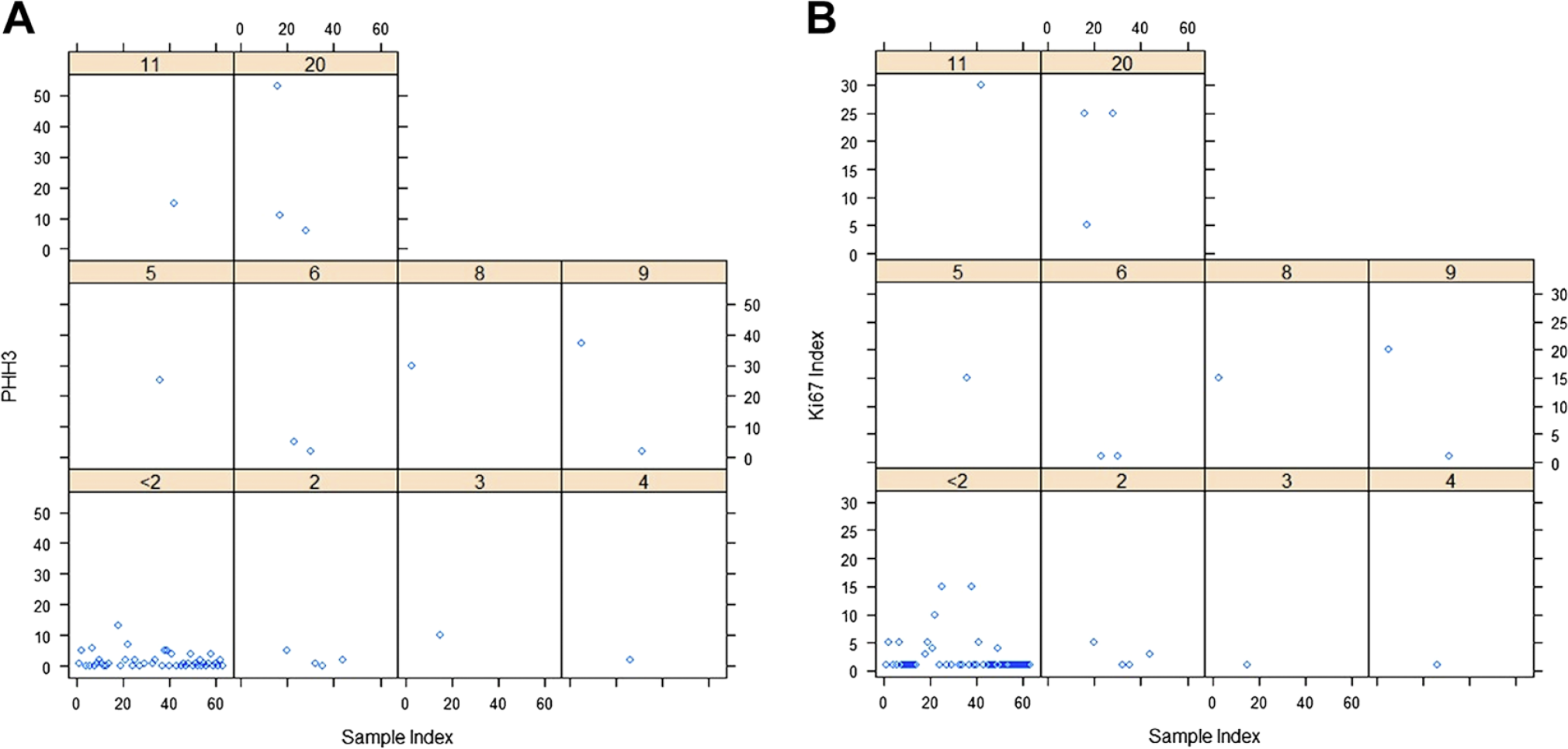
Scatter plots for phosphohistone-H3 (PHH3) (A) and Ki67 (B) stratified by the mitotic counts; within each subfigure, the x-axis is the sample index, the y-axis is PHH3/Ki67, and the highlighted is the corresponding mitotic counts (n=63).
The p values for the comparison between DFS curves of different grades were 0.069, 0.20, and 0.19 for Ki67, PHH3, and mitotic count, respectively. The p values from a Cox proportional hazards regression model with the gender and age as covariates were also not significant.
To asses which grading technique better predicts the presence of LN metastases, a contingency table for the grades and the presence of LN metastases was made. The p values from the Fisher exact test of contingency tables were 0.92, 0.041, and 0.15 for Ki67, PHH3, and mitoses, respectively. This indicates that PHH3 significantly correlates with the presence of LN metastases (p=0.041). There was no significant correlation between the disease stage and Ki67, PHH3, or mitotic index.
PNI, LVI, and necrosis were compared with the recurrence, DFS, and OS. The only statistically significant correlation was found between necrosis and OS (p=0.017). PNI, LVI, or necrosis was not associated with recurrence or DFS.
Ki67, PHH3, or mitotic index did not significantly predict the recurrence. The PHH3 index was the only method that correlated with the LN metastasis.
Discussion
pNETs are uncommon tumors arising from endocrine cells of pancreas and the incidence has risen remarkably in the United States over the last two decades, possibly as a result of better detection on new imaging techniques and endoscopy.1,19 pNET is often associated with a prolonged survival; however, there is a substantial variability in patient outcome due to the poor understanding of natural history and lack of predictor factors and prognostication. 20 In pathology practice, the widely used grading system 21 is mainly based on the mitotic index and quantitative Ki67 proliferation index. However, recognition of mitotic figures may be challenging and has been shown to be greatly subjective. PHH3 is a histone protein located in eukaryotic chromatin, and has been demonstrated to coincide with mitosis temporally and spatially. It is present when chromosomes are fully condensed and is negligible in the interphase phase.22,23
Several recent studies have documented the utility of PHH3 immunolabeling in accurately identifying mitotic figures in various tumor types with significant results.17,24,25 We investigated the utility of this antibody in pNET grading. We also sought the significance of other pathological parameters, including tumor necrosis, PNI, and LVI, and their effect on clinical outcomes.
Our findings suggest that PHH3 is an effective marker for determining mitotic activity when compared with Ki67- and H&E-based mitotic count and can be used alternatively to Ki67. There was a negligible discrepancy in the stratification of tumor grades for Ki67 and PHH3. In determining the correlation between these grading methods and disease recurrence, Ki67, PHH3, and mitotic figure counts all were not statistically significant. This indicates grading alone does not directly predict recurrence for any grading method, and that PHH3 is similar to Ki67 and mitotic index in correlating with recurrence. PHH3 had a statistically significant correlation with the metastatic disease in the LN when compared with Ki67.
According to our results, the presence of PNI, LVI, and necrosis was not predictive of recurrence. However, the presence of necrosis was very strongly associated with decreased OS. The presence of necrosis is known to be associated with aggressive disease in NETs, and was extensively present in our one case of fulminate disease. However, PNI or LVI did not correlate with the clinical outcome likely due to the small sample size.
The utility of PHH3 in pNETs has not been extensively studied. Most recently, Voss et al. 16 examined the utilization of PHH3 in comparison with the current WHO grading scheme of mitotic figure count and Ki67 in 63 well-differentiated pNET resected surgical specimens. Similar to our results, their study showed that PHH3 correlated well with Ki67 indices. In addition to using PHH3 for mitotic rate similar to Ki67, they examined, as we did in our study, the utilization of PHH3 as a tool for mitotic count analysis. They found PHH3 to be less time consuming and had improved inter-observer variability when compared with H&E. An earlier study by Dragonova-Tacheva et al. 15 compared PHH3- and Ki67-based grading in 23 pNET cytological specimens. As with our study, they found a correlation between Ki67 and PHH3 grading, with the range of PHH3 positivity being narrower than Ki67 requiring more cells to be examined. They also suggested the utility of PHH3 as a tool for mitotic count, rather than determining a proliferation index. To date, the only other published study of PHH3 in NETs has been conducted by Fung et al. 26 in which 19 well-differentiated gastrointestinal cytological specimens were not limited to the pancreas. This study concluded that Ki67 and PHH3 have significantly correlated labeling indices. It also found that Ki67 had a stronger staining than PHH3 allowing for the easier detection of staining hot spots, making Ki67 a more ideal stain for NET grading. Both studies examining cytological specimens did not examine clinical outcome and were conducted using digital image analysis systems.
Ki67 has been shown to correlate with the presence of necrosis and angio-invasion in the presence of NETs. 26 However, the presence of angio-invasion is not part of the WHO criteria and its correlation with clinical outcome has been minimally studied since the new WHO classification. Demir et al. 27 retrospectively examined 82 resected pNET cases for the presence of angio-invasion or necrosis. The authors reported that both angio-invasion and necrosis predicted worse OS. Although our study did not find a strong correlation between angio-invasion and stage, we found similar results for the presence of necrosis. The study however did not examine the presence of PNI in pNETs, and no studies since the new WHO criteria have been published examining the relation between PNI and clinical outcome.
In conclusion, PHH3 produces similar grading results to the Ki67 proliferative rate in regard to predicting disease recurrence or the presence of metastatic disease on presentation. This study therefore suggests that PHH3 mitotic count could be used in grading pNETs as a possibly better alternative tool precluding the subjective interpretation between the observers. Similar studies with a larger sample size to develop more accurate quantification methods and PHH3 grading cut-offs to determine mitotic count are warranted for further grading modifications in pNETs.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
DP organized the study, DP and JT collected and analyzed the data and wrote the manuscript, and KZ performed statistical analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study supported by the Adams Resident Research Fund from the Department of Pathology at the University of Alabama at Birmingham (to JT).
