Abstract
Congo red (CR) is a histological stain used for the detection of extracellular amyloids mediating various neurodegenerative diseases. Given that damaged photoreceptors appear to degenerate similarly to other nerve cells, CR staining was evaluated in experimentally injured porcine retina. CR staining appeared mostly as discrete cytosolic deposits with no obvious plaque formation during the investigated time period. Increases of CR labeling coincided temporally with the known accumulation of mislocalized opsins and increases of cell death. Coculture, either with human retinal pigment epithelium (ARPE) or human neural progenitor (ReN) cells, was accompanied by a significant reduction of CR labeling. Of particular interest was the reduction of CR labeling in cone photoreceptors, which are important for the perception of color and fine details and afflicted in age-related macular degeneration (AMD). Electron microscopy revealed inclusions in the inner segment, cell body, and occasionally synaptic terminals of photoreceptor cells in cultured specimens. Closer examinations indicated the presence of different types of inclusions resembling protein aggregates as well as inclusion bodies. The current results indicate that injury-related response resulted in accumulation of CR deposits in photoreceptor cells, and that trophic and/or structural support attenuated this response.
Introduction
Microscopic deposits of aggregated proteins constitute a hallmark of neurodegenerative disorders that progressively replace the parenchyma of the affected organ, resulting in progressive loss of functionality and eventually organ failure. Amyloid deposition results from a variety of precursor proteins, 1 which are misfolded and mainly aggregated into extracellular deposits. Neurodegenerative and neurological disorders result from depositions of extracellular plaques and/or intracellular inclusions consisting of aggregated proteins.
For instance, extracellular deposits of amyloid β and intracellular accumulation of phosphorylated tau in Alzheimer’s disease cause damage to synapse composition and dendritic spines, leading to cognitive decline. 2 Neuronal loss in Parkinson’s disease is correlated with intracellular Lewy bodies consisting of α-synuclein, parkin, and phosphorylated neurofilament. 3 The dye Congo red (CR) has been used for histological demonstrations of deposited amyloidogenic protein in various degenerative diseases. 4 CR binds mainly to extracellular fibrils, but some intracellular protein tangles and inclusions also bind CR and may be considered as intracellular amyloids. 1 Accumulating immunoelectron microscopy data suggest that an accumulation of amyloid β precedes the detection of extracellular material.5,6
Accumulation and aggregation of defective proteins leading to pathology may occur in ocular tissues as a result of age-related cellular stress and inherited alterations of protein structure.7–10 Initiation of photoreceptor degeneration may be a result of aberrant aggregation of misfolded opsins that accumulate in subsets of photoreceptor cells and disturb protein homeostasis. Degeneration of rod photoreceptors in retinitis pigmentosa is mostly associated with rhodopsin mutations, resulting in accumulation of mislocalized protein aggregates. 11 Although such data from cone photoreceptors are scarce, aggregation of the different cone photoreceptor opsins has been shown to occur before cone degeneration. 12 It is not known if opsins can form intracellular protein aggregates that are toxic for the afflicted photoreceptor. However, amyloid β and its precursor protein have been observed in different rodent models of retinal degeneration and retinas from patients suffering from Alzheimer’s,11–16 which substantiates that amyloids may contribute to photoreceptor degeneration in various retinal disorders. 17
As the name age-related macular degeneration (AMD) implies, visual impairment affects central vision by degeneration of cone photoreceptors located in the macular region. AMD is a complex neurodegenerative disorder causing visual impairment or blindness in the elderly population. The complexity of AMD involves not only loss of retinal pigment epithelium (RPE) and photoreceptor cells, but also secondary injuries including gliosis, neural remodeling, and loss of synaptic integrity.18,19 Malfunction of the RPE, initiated by an accumulation of intracellular deposits (so-called drusen), results eventually in photoreceptor degeneration. In addition, recent data indicate that AMD may arise in cone photoreceptors via oxidative damage resulting in protein aggregation. 20 Defective trafficking and accumulation of mislocalized cone opsin may also result in endoplasmic reticulum stress, a cellular response that eventually results in apoptosis and photoreceptor degeneration. 21 Further investigations of protein aggregation mechanisms and putative involvement in the degeneration of cone photoreceptors are, thus, of considerable importance for the understanding of AMD.
In the current study, we evaluated putative amyloid-like protein deposits in cultured porcine retina exposed to cyclic light by CR staining under fluorescence microscopy. In the experimentally injured photoreceptors, small intracellular deposits were intensely positive for CR, and electron microscopy revealed the presence of dense inclusions. In cocultures of retinal explants together either with human RPE cells or human neural progenitor cells to provide structural and/or trophic support, respectively, significant reductions of CR labeled deposits were observed.
Materials and Methods
Tissue
Experimental procedures were approved by the Swedish Board of Agriculture (Jordbruksverket; Jönköping, Sweden). Porcine eyes were collected from a local slaughterhouse and transported in cold CO2-independent media (Gibco; Life Technologies, Carlsbad, CA). Dissection was carried out as described previously. 22 Briefly, the anterior segment was removed as well as the lens and vitreous body. The neural retina was gently detached from the underlying pigment epithelium, and retinal explants of about 10 mm2 were punched out. Start of explant cultures was achieved within 30–40 min after collection.
Feeder Layers Using ARPE or ReNcell CX Progenitor Cell Line
The human adult RPE-19 (ATCC; cat. no CRL-2302, Manassas, VA) cell line (here referred to as ARPE) was cultured to confluence onto Millicell-PCF 0.4 μm (Millipore; Bedford, MA) culture plate inserts. ARPE feeder layers consisted of 1.6 × 105 cells/insert between passages 6–12. Culture media was composed of DMEM/F12 (Gibco) supplemented with 2% B27-supplement (Gibco), 1% N2-supplement (Gibco), 1% penicillin/streptomycin, and 2 mM glutamine (Sigma-Aldrich; St. Louis, MI). Cell culture media was exchanged every third day.
The ReNcell CX progenitor (Millipore) cell line (ReN) was used as a feeder layer for production of trophic factors. 22 Passages between 9–13 were used. ReN cells proliferated in laminin-coated (20 μg/mL; Sigma-Aldrich) culture flasks, and culture media was DMEM/F12 (Gibco) supplemented with 2% B27-supplement (Gibco), 1% N2-supplement (Gibco), 0.2% Heparin 5000 IU/mL (Leo Pharma; Malmö, Sweden), 1% penicillin/streptomycin, and 2 mM glutamine (Sigma-Aldrich). Culture media supplemented with human epidermal growth factor (EGF; Millipore), 20 ng/mL, and human basic fibroblast growth factor (bFGF; Millipore), 20 ng/mL, was exchanged every third day. Before coculture, 2 × 105 ReN cells/well were seeded in 6-well culture dishes.
Retinal Explants and Coculture With or Without Feeder Layers
Retinal explants of about 10 mm2 were divided into 4 equal sections and explanted on Millicell-PCF 0.4 μm culture plate inserts (Millipore) with the vitreal side oriented upward. One or two retinal sections were explanted onto each insert, which was placed in 6-multiwell culture dishes containing 1.2 ml of culture medium per well. The culture medium consisted of DMEM/F12 (Gibco) supplemented with 2% B27-supplement (Gibco), 1% N2-supplement (Gibco), 0.2% Heparin 5000 IU/mL (Leo Pharma), 1% penicillin/streptomycin, and 2 mM glutamine (Sigma-Aldrich).
The ARPE cells were allowed to grow to confluence on the insert before the coculture started. Explants were placed on inserts with or without ARPE cells, and placed in 6-well culture dishes.
By growth factor withdrawal, the ReN cells were allowed to differentiate for 3 days, whereupon the coculture started. Inserts with explanted retina were placed in 6-well culture dishes with or without ReN cells.
Retinas were cultured at a light/dark cycle (8 hr illumination of warm white light) for 24 hr, 48 hr, 3 and 5 days at 37C with 95% humidity and 5% CO2. The specimens were immersion-fixed using 4% paraformaldehyde (Alfa Aesar; Karlsruhe, Germany) in Sorensen’s phosphate buffer (Sigma-Aldrich) overnight at 4C. Several rinses with Sorensen’s followed, after which the specimens were cryoprotected in increased concentrations of sucrose (10–25%; Sigma-Aldrich) in Sorensen’s. The tissue samples were embedded in cryomount (HistoLab; Gothenburg, Sweden), cryosectioned at 10–12 μm, and stored at −20C. Normal retinas, directly fixed after dissection and cryosectioned, were used as uncultured controls. Further controls included retinas cultured for 3 days in darkness.
Congo Red Staining
Congo red dye is considered as a pan-stain for amyloidal proteins and can be visualized by fluorescent microscopy using Texas red filter.23,24 Frozen slides were thawed for at least 10 min and stained for Congo red (Sigma-Aldrich) according to the manufacturer’s instructions. Immersed slides were mounted using Vectashield with 4′,6-diamidino-2-phenylindole (DAPI; Vector Laboratories, Burlingame, CA). Rehydrated control sections (cardiac amyloidosis), obtained from Histo-Center AB (Gothenburg, Sweden), were stained in parallel with retinal sections and were used as positive controls.
Congo Red Staining of Photoreceptor Cells
To investigate the cellular distribution of presumptive amyloidosis in the outer nuclear layer (ONL), CR-stained sections were also assessed for the expression of cone opsin and rod rhodopsin. The CR-stained sections were incubated overnight at 4C in a moist chamber either with rabbit anti-red/green cone opsin (long/middle wave [L/M]-opsin) antibody (Millipore) at 1:1000, or a mouse anti-rhodopsin antibody (Millipore) at 1:2000.
After rinsing with PBS (Medicago; Uppsala, Sweden) of pH 7.2, the sections were incubated with Alexa 488 conjugated donkey anti-rabbit IgG Fab (1:500; Jackson Laboratories, West Grove, PA) or FITC-conjugated goat anti-mouse IgG Fab (1:500; Jackson Laboratories) fluorescent secondary antibodies at room temperature for 30–45 min in darkness. All antibodies were diluted in 1% BSA in PBS.
Transmission Electron Microscopy
Specimens grown on tissue inserts were fixed using 2% paraformaldehyde (Alfa Aesar) and 2% glutaraldehyde (Agar Scientific; Stansted, UK) in 0.1 M PBS (Medicago) for 2 hr at 4C. Postfixation in 1% osmiumtetroxid (Agar Scientific) for 1 hr followed, whereupon the specimens were dehydrated for embedding in Epon resin (Sigma-Aldrich). Ultra-thin sections from selected areas were counterstained with uranyl acetate (Ted Pella Inc.; Reading, CA) and lead nitrate (BDH; Dubai, UAE), and examined using a JEOL 1400 Plus Transmission electron microscope (JEOL; Tokyo, Japan). Images were captured using a Matakataki CMOS digital camera (JEOL).
Microscopy and Histological Quantitation
Labeled sections were viewed using an Olympus BX60 with appropriate filter settings and imaged with an Olympus DP71 camera (Olympus–Europe; Hamburg, Germany). Images were adjusted for brightness and contrast (Adobe CC 2015; Adobe Systems, San Francisco, CA). The density of CR deposits were quantified with ImageJ software (National Institutes of Health; Bethesda, MD).
Presentation of Data and Statistical Analyses
Qualitative or quantitative comparisons were made between treated and untreated explants (the latter serving as controls), and 4–7 eyes were used. Non-cultured retina are used to show normal histology. All quantitative data are presented as mean ± SEM. Statistical calculations were performed with GraphPad Prism 7 (La Jolla, CA) and Student’s t-test was used for unpaired samples. Results were considered statistically significant when p<0.05.
To quantify the density of CR deposits, images were acquired from the midportion of each section. CR densities were measured in 2–5 areas in 2–3 retinal sections/explant using ImageJ (https://imagej.nih.gov/ij), which were normalized to 1000 um2 of the ONL.
To quantify the beneficial effect of ARPE or ReN cells on cone photoreceptor survival, the ratio of surviving cones was calculated using the following formula: L/M-labeled cones without CR / total number of L/M-labeled cones in a given section × 100.
Results
Retinal Morphology
As demonstrated previously, 22 cultured porcine retinas display similar cellular layered architecture with nuclear and plexiform layers as normal retina (Figs. 1A and B). Distortions resembling outer retinal tubulation,25,26 and tissue cavities were evident in the ONL after 3 and 5 days in vitro (DIV; Figs. 1G and H).22,27 Cone photoreceptor outer segments degenerate in vitro and cone opsin immunoreactivity redistributes and accumulates in the inner segments and/or rudimentary outer segments as well as in cone cell somata (compare Figs. 1A and B).
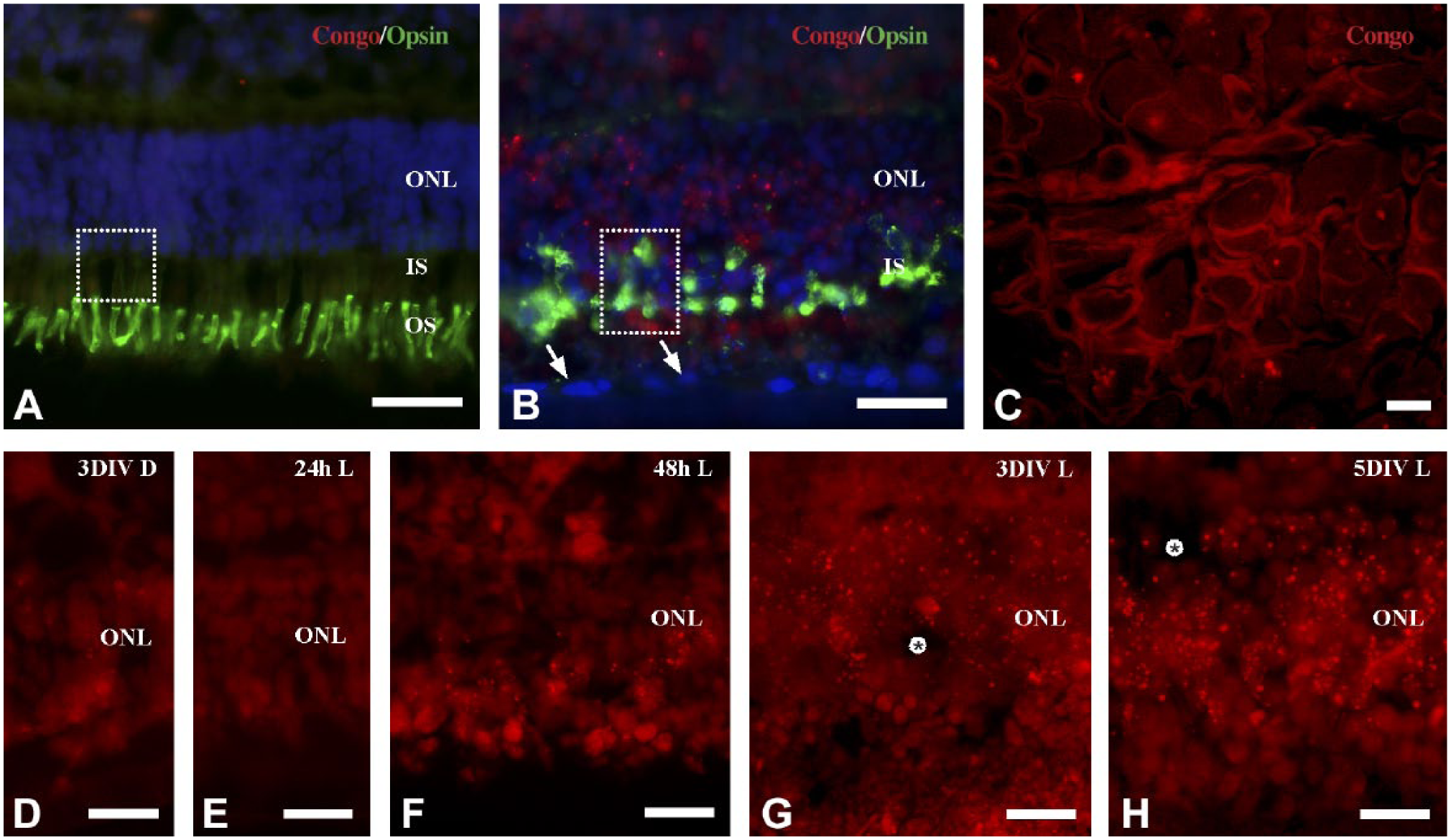
Micrographs of CR labeling in experimental and control tissues. CR+ labeling in the ONL of normal (A) and cultured (B, D–H) retina. No CR+ labeling in the ONL of normal retina (A) compared with 5 DIV retina. Note also expression of L/M-opsin in the OS of normal retina and in the rudimentary IS of cultured retinas (B). Arrows in (B) indicate the ARPE feeder layer. Boxed area in A and B approximately indicates the regions that were analyzed by electron microscopy. (C) CR+ labeling of cardiac amyloidosis. Muscle parenchyme is surrounded by fluorescent extracellular amyloid deposits. No or single CR+ labeled deposits were observed in retina cultured for 3 DIV in darkness (D) or 24 hr in light-exposed cultures (E). The density of CR+ labeled deposits increases in light-exposed cultures from 48 hr (F) to 3 (G) and 5 (H) days of culture. Tissue cavities were observed in the ONL of 3 and 5 DIV retina (asterisks in G and H). A, B are DAPI counterstained. Abbreviations: CR, Congo red; ONL, outer nuclear layer; OS, outer segments; IS, inner segments; DIV, days in vitro; DAPI, 4,6-diamidino-2-phenylindole; D, dark-reared; L, light-reared. Scale bars A–H = 20 µm.
Congo Red Fluorescence
CR-positive (CR+) labeling in cultured retinas showed a clear red color in the ONL with Texas red filter using ultraviolet light.23,24 No CR+ labeling was observed in the ONL of normal, uncultured retina (Fig. 1A). Examination of positive control tissue (heart amyloidosis) showed strong CR fluorescence confined to extracellular amyloid deposits (Fig. 1C).
CR+ labeling in photoreceptors was evident in the form of compact and homogeneous globular deposits without radiating fibrils (Fig. 1B). The deposits varied in size and were associated with photoreceptor cell somata, seemingly with a perimembranous and/or cytosolic distribution. Although single extracellular deposits were observed (Fig. 3B), large extracellular plaques were not observed. The density of CR+ deposits increased with time in vitro and light exposure. Only single CR+ deposits were observed in the ONL after 24 hr of culture and in control cultures dark-reared for 3 DIV (Fig. 1D and E). CR+ deposits were readily evident at the outer inner aspect of the ONL after 48 hr (Fig. 1F). The density of CR+ deposits increased further and was consequently distinguished throughout the ONL in 3 and 5 DIV cultures (Figs. 1G and H).
Quantitation of CR Fluorescence in ARPE and ReN Cell Cocultures
A general feature of cultured retinas is the aberrant localization of the opsins in rod and cone photoreceptors.27,28 Mislocalized L/M-opsin was detected in cone somata and in cone synaptic pedicles (Figs. 2A and D). Because coculture with ReN cells significantly reduced mislocalized L/M-opsin and increased photoreceptor survival, 22 the impact of cocultured ARPE and ReN cells on CR labeling in the ONL was analyzed. Double labeling for CR and L/M-opsin showed the presence of CR+ both in the rod and the cone photoreceptor population (Figs. 2A–F).
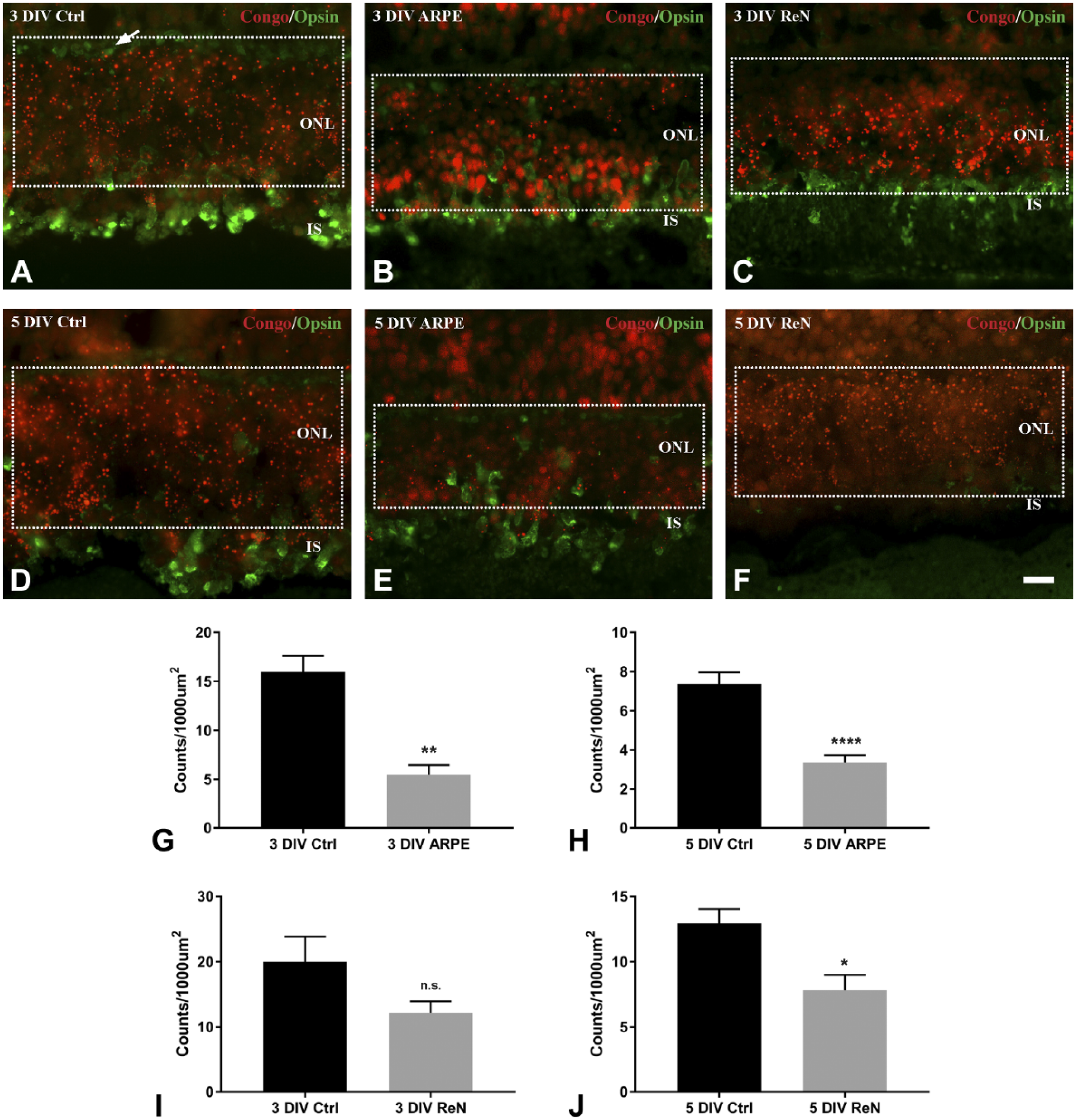
Photoreceptor responses in cultured retinas, controls as well as ARPE and ReN cocultures, in terms of CR+ (red) and L/M-opsin (green) staining. (A–F) In 3 and 5 DIV cocultures with ARPE (B, E) or ReN (C, F), the density of CR+ deposits declined within the ONL. Arrow in A indicates L/M-opsin labeled cone synaptic pedicles. Boxed area in A to F approximately represents the region of the ONL in which the CR+ were estimated. (G–J) Quantitative measures of CR+ deposits in the different culture paradigms. The density of CR+ deposits was not significantly different between 3 DIV control and 3 DIV ReN (I). Measures of CR+ deposits in control and coculture revealed significant decreases in the other culture paradigms (G, H, J). Abbreviations: ARPE, human retinal pigment epithelium; ReN, human neural progenitor; CR, Congo red; DIV, days in vitro; ONL, outer nuclear layer; IS, inner segments. Scale bar in F = 20 µm. *p<0.05, **p<0.01, ****p<0.0001, Student’s t-test.
Quantitative measurements of CR+ deposits in the entire ONL revealed a general reduction of CR. Comparisons of CR+ between controls and ARPE cocultures showed significant decreases both at 3 and 5 DIV (p=0.0019 and p<0.0001, respectively; Figs. 2G and H). While coculture with ReN for 3 DIV did not significantly reduce the density of CR+ (p=0.103; Fig. 2I), there was a significant reduction after 5 DIV (p=0.018; Fig. 2J).
Aberrant opsin trafficking with accumulation/aggregation of visual pigments to photoreceptor cell somata, inner segment, and synaptic terminal is a general feature of injured in situ retinas, 29 as well as in cultured retinas.22,27 We hypothesized that the accumulation of mislocalized opsin may be associated with the CR+ deposits, and investigated whether CR colabeled either for rhodopsin or cone L/M-opsin immunoreactivity. In general, there was a variable abundance of colabeled photoreceptors particularly in the inner part of the ONL. Within single colabeled rod and cone photoreceptors, CR+ deposits and immunolabeling for rhodopsin (Fig. 3A, insert) and L/M-opsin appeared to be localized to separate subcellular compartments (Figs. 3A and B). CR+ deposits exhibiting a yellowish hue, indicating colabeling with the L/M-opsin, were not clearly evident (see Fig. 3B).
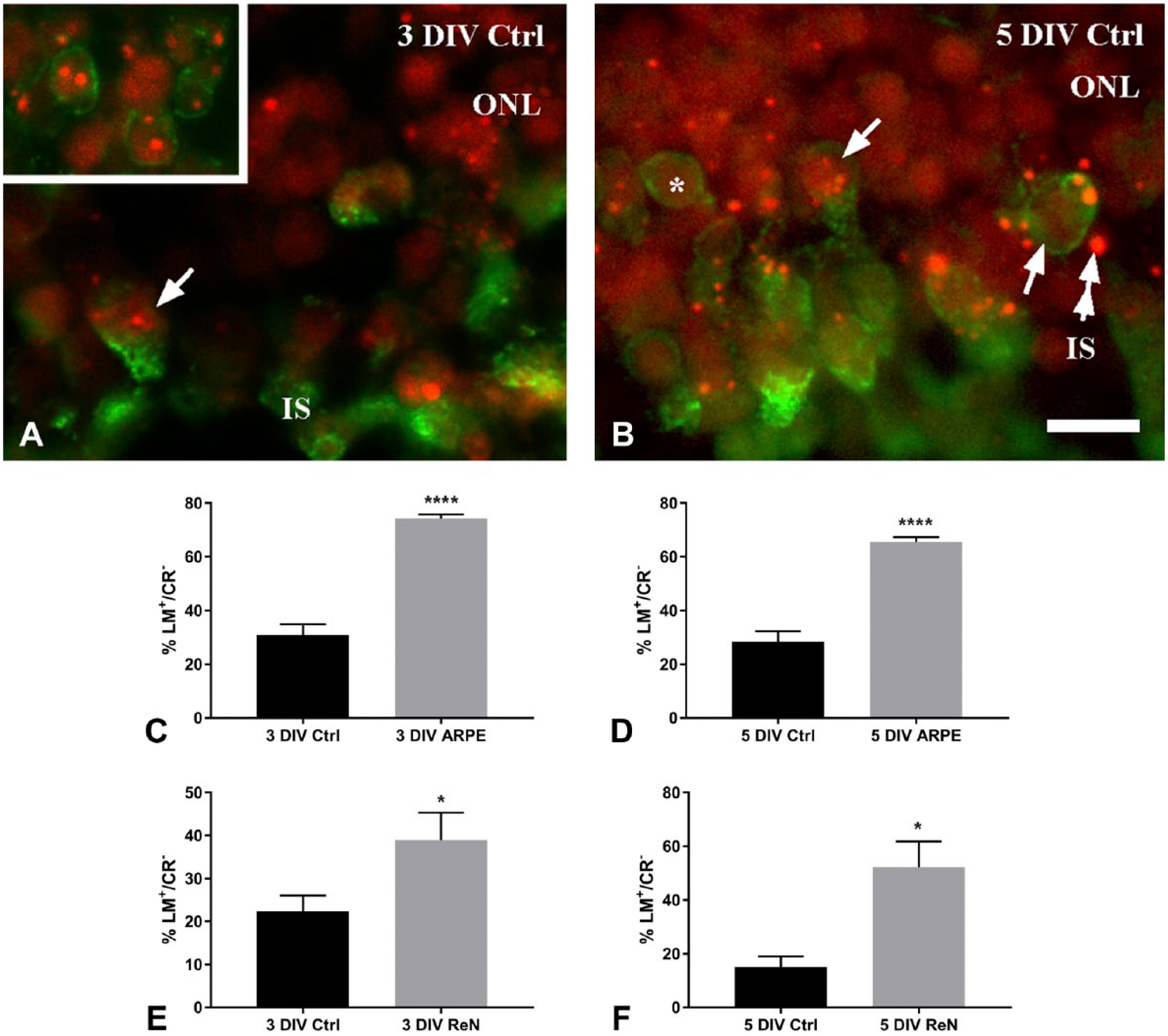
Immunofluorescence images of CR+ (red) and L/M-opsin (green) staining and quantitative estimates of cone survival as measured in terms of the ratio of L/M-opsin positive cells that lack CR labeling (LM+/CR−). (A, B) CR+ deposits of variable size in cone photoreceptors (arrows) at 3 and 5 DIV, respectively. (A insert) Immunofluorescence showing CR+ in rhodopsin (green) immunostained rods at 3 DIV. Note also the presence of single extracellular CR+ deposits (double arrows in B). Quantitative measurements revealed significant increases of LM+/CR− cones in all cocultures (C–D). Abbreviations: CR, Congo red; DIV, days in vitro; ONL, outer nuclear layer; IS, inner segments; ARPE, human retinal pigment epithelium; ReN, human neural progenitor. Scale bar in B = 20 µm. *p<0.05, ****p<0.0001, Student’s t-test.
Coculture with both ARPE and ReN indicated a reduction of CR+ deposits in the outer part of the ONL where cone cell bodies reside. To further analyze if ARPE and ReN cells provide beneficial structural and/or trophic effects on cone photoreceptors, the number of L/M-opsin labeled (LM+) cones was estimated. A ratio of cells positive for L/M-opsin (LM+) and negative for CR (CR−) divided by the total number LM+ was established and compared between controls and cocultures (Table 1). The ratio of LM+/CR− cones in the outer ONL at 3 and 5 DIV differed significantly, with higher ratios of cones without CR+ deposits in specimens cocultured either with ARPE or ReN cells (Figs. 3C–F).
Ratio of LM-Positive Cones Without Congo Red (LM+/CR−) at 3 and 5 DIV.

A total of 4710 LM+ cones with/without CR were counted (2010 at 3 DIV and 2700 at 5 DIV). Abbreviations: LM, long/middle wave; CR, Congo red; DIV, days in vitro; ARPE, human retinal pigment epithelium; ReN, human neural progenitor.
Transmission Electron Microscopy
Electron microscopy was focused at the outer part of the retina (box areas in Figs. 1A and B) on specimens cocultured for 5 days with ARPE cells. Low-power electron microscopy revealed the presence of cytosolic electron-dense spherical inclusions in different photoreceptor cell compartments, particularly in the inner segment but also close to cone nuclei and occasionally in cone synaptic pedicles (Figs. 4A and C). The inclusions were not observed in normal retina (Fig. 4B). Photoreceptor inner segments were rudimentary with a broad and blunt morphology, and contained spherical to ovoid mitochondria (Fig. 4C).
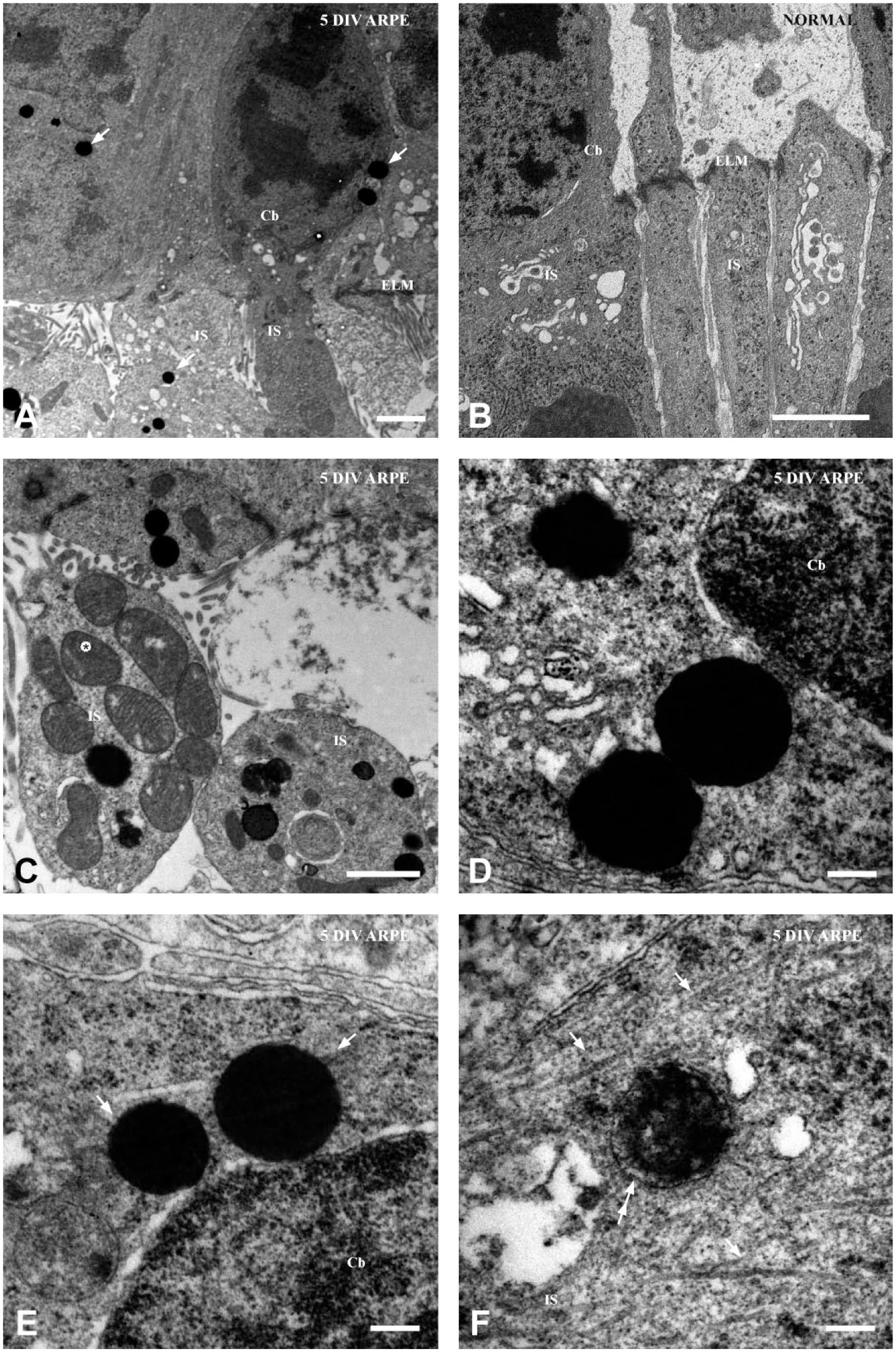
Electron micrographs of photoreceptors with inclusions. Because of good tissue preservation, focus is on retinas cocultured for 5 days with ARPE cells. Low-power micrographs showing electron-dense spherical inclusions (arrows) close to the nucleus and in the inner segments (A). (B) Normal retina showing photoreceptor cell bodies and inner segments without inclusions. (C) Inclusions vary in number and were also found close to mitochondria (asterisk) in the inner segment. (D–F) Representative inclusions observed in photoreceptors. Inclusions were mostly dense and spherical but occasionally lobulated (D). High magnification revealed the presence of filamentous material (arrows in E) radiating out from single inclusions. Inclusion in the inner segments close to filamentous material (arrows in F). Note also the less dense matrix and surrounding membrane-like structure (double arrows in F). Abbreviations: ARPE, human retinal pigment epithelium; DIV, days in vitro; Cb, Cell body; IS, inner segments; ELM, external limiting membrane. Scale bars A–B = 2 µm, C = 1 µm, D–F = 0.2 µm.
Most of the inclusions observed showed variations in size and were composed of an electron-dense and non-lamellar even matrix. Inclusions with electron-lucent material in their matrix were not observed. High magnification showed the presence of different inclusions that appeared to be non-membrane-bound in general (Figs. 4D and E). A minor fraction of the inclusions were lobular with a flocculent matrix enclosed by a membrane (Fig. 4F), while other inclusions had a more lobulated appearance but even matrix (Fig. 4D). Filamentous material, free ribosomes, and the endoplasmic reticulum could be noted in the vicinity of some inclusions (Figs. 4D and E). The filamentous material appeared as a thin corona radiating from the inclusion (Fig. 4E) or as long filaments close to the inclusion (Fig. 4F). The latter could be observed between the inner segment and cell body.
Discussion
The findings presented in this study describe an injury-related occurrence of CR+ deposits in photoreceptors in cultured porcine retina, and demonstrate that different experimental culture conditions modulate the density of CR+ staining. First, CR+ staining was upregulated during the culture period and was readily evident after 3 DIV among photoreceptor cell bodies in the ONL. Upregulation of CR+ deposits was also associated with exposure to cyclic light. This suggests that the formation of CR+ deposits appears in concert with previously described photoreceptor damage and the erratic accumulation of the opsins.22,27 Second, coculture either with ARPE cells or ReN cells resulted in reduced CR+ staining, particularly among cone photoreceptors localized to the outer aspect of the retina. Finally, electron microscopy of rod and cone photoreceptors showed the presence of various intracellular electron-dense inclusions that appeared to coincide with the localization and form of the CR+ deposits. The data from the current study indicate that an amyloidosis-like process as revealed by CR may occur among photoreceptors in light-injured retina. The coculture data show a significant beneficial effect by trophic, and perhaps also structural, support on a detrimental cascade that leads to photoreceptor degeneration.
Given the accumulation of mislocalized rhodopsin and L/M-opsin in cultured porcine retina,22,27 we hypothesized that the opsins could be amyloidogenic particularly following light exposure. CR+ staining appeared as intracellular deposits of variable size in photoreceptors indicating that aggregated protein(s) are stained by the dye. The general distribution of CR+ deposits within cone cell bodies and inner segments coincided with immunoreactivities for rhodopsin and L/M-opsin, however, single CR+ deposits showed no obvious colocalization with the opsins. The reason for this may be that the opsins do not contribute to the formation of CR+ deposits, or that the opsins are denatured during aggregation and not recognized by the antibodies. Studies on photoreceptors with P23H-mutant rhodopsin show formation of cytotoxic protein aggregates and inclusion bodies, which may be involved in photoreceptor degeneration.30,31 A recent study demonstrates that mouse S-opsin and human blue-sensitive cone photoreceptors, in particular, can form aggregates and determine the degeneration rate of photoreceptors. 12 It is possible that an accumulation of the opsins may result in high protein levels that facilitate self-association into compact protein aggregates that are labeled by CR. Opsin aggregation may be also enhanced when the amounts of 11-cis retinal are limited 12 : 11-cis retinal was neither supplemented to the cultures nor is it produced by the ARPE cells. 32
The presence of different protein inclusions in ageing cones is known from previous electron microscopy studies.25,33,34 A general observation as reported in previous studies is the presence of inclusions close to mitochondria and having an association with filaments in the inner segment myoid. Subsets of inclusions in the current study were spherical and displayed an electron-dense matrix resembling lysosomal-like inclusions and/or refractile inclusion bodies. Such bodies were also found in cones, 34 and their ultrastructural appearance differs from lipofuscin granules and multivesicular bodies. 35 We also observed inclusions with or without a membrane as well as granular inclusion close to the nucleus, all of which also lacked the electron-lucent material observed in lipofuscin granules. 35 Differences in electron microscopic features suggest the presence of subsets of inclusions that may differ in pathophysiological significance in the degenerating cones. Compared with inclusion bodies in Alzheimer’s, the inclusions in the present study may be true inclusion bodies or aggresomes recruited to the inclusion body.
By trophic and structural support, there was a significant decrease of CR+ staining in general among photoreceptors as well as in the cone subpopulation. Trophic support to photoreceptors in vitro by cocultured progenitor cells is most likely accomplished by the presence of various growth factors.22,36 Beneficial effect(s) provided by the ARPE to the photoreceptors may involve cell-derived growth factors in combination with structural support. ARPE is known to secrete vascular endothelial growth factor (VEGF) and pigment-epithelium-derived factor (PEDF), 32 and gene profiling shows the expression of other growth factors as well. 37 A variety of growth factors including VEGF and PEDF as well as brain-derived neurotrophic factor (BDNF) and bFGF have neuroprotective effects and promote photoreceptor survival.38,39 Also, the presence of shed immunolabeled outer segments between the explant and ARPE feeder layer indicates an improved trafficking of opsins into the outer segments and subsequent reduced burden of aggregated opsins and higher survival rate of photoreceptors.
The current study is focused on cone photoreceptor degeneration and injury-related morphological alterations at the subcellular level that may be related to AMD. In comparison with human AMD retinas,26,28 some morphological features were also observed in the current study and include opsin redistribution and rudimentary inner segment. The presence of inclusions in the cone population is another common feature found in light-exposed cultured retinas 27 as well as AMD retinas.33,34 Another similarity is the movement of spherical and ovoid mitochondria into the rudimentary cone inner segment and toward nuclei. 26 The anomalous location of the mitochondria and their oxidative phosphorylation and generation of reactive oxygen species may create an environment that damages proteins and organelles in the inner segment. Misfolded and damaged proteins, including amyloid beta, have been reported to accumulate in photoreceptor 40 and are present in drusen from AMD retinas.9,41 Dysfunction of mitochondria and oxidative stress, resulting in protein damages, appears to be a common feature of age-related neurodegenerative disorders, 42 including AMD.20,26
In summary, although accumulating circumstantial data indicate that amyloid formation contributes to photoreceptor degeneration, direct evidence is lacking. From the current in vitro study, it is suggested that light-exposed photoreceptors in general and cones in particular exhibit an amyloidogenic-like process, as demonstrated by CR staining. Coculture with ARPE and ReN cells provided trophic and/or structural support that resulted in diminished CR+ labeling. Because ReN cells coculture is known to support proper trafficking of cone opsin, it may be suggested that L/M-opsin may have amyloidogenic properties. However, this hypothesis could not be confirmed with conventional immunohistochemical techniques. By electron microscopy, it was evident that different subsets of inclusions were formed in the injured photoreceptors. Based on morphological criteria, some of the inclusion may be regarded as inclusion bodies that have been described in other neurodegenerative diseases (e.g., Alzheimer’s). Some inclusions in the photoreceptors may be classified as protein aggregates.
Footnotes
Acknowledgements
The Microscopy Facility at the Department of Biology, Lund University, is acknowledged for technical assistance with electron microscopy.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: CM and KJ designed and executed experiments; CM, DD, AK, and KJ wrote the article, and all authors have read and approved the manuscript as submitted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support was provided by the faculty of Medical Sciences at Örebro University, Olle Engkvist Foundation, Ögonfonden, the Crown Princess Margaretas Committee for the Blind, Edwin Jordan and Sven and Dagmar Salen Foundations.
