Abstract
Fragile X mental retardation-related protein 1 (FXR1) is a cytoplasmic RNA-binding protein highly conserved among vertebrates. It has been studied for its role in muscle development, inflammation, and tumorigenesis, being related, for example, to metastasizing behavior in human and canine uveal melanoma. Anti-FXR1 antibodies have never been validated in the canine species. To investigate FXR1 expression in canine melanocytic tumors, the present study tested two commercially available polyclonal anti-human FXR1 antibodies, raised in goat and rabbit, respectively. The cross-reactivity of the anti-FXR1 antibodies was assessed by Western blot analysis, and the protein was localized by IHC in a set of normal canine tissues and in canine melanocytic tumors (10 uveal and 10 oral). Western blot results demonstrated that the antibody raised in rabbit specifically recognized the canine FXR1, while the antibody raised in goat did not cross-react with this canine protein. FXR1 protein was immunodetected using rabbit anti-FXR1 antibody, in canine normal tissues with different levels of intensity and distribution. It was also detected in 10/10 uveal and 9/10 oral melanocytic tumors. The present study validated for the first time the use of anti-FXR1 antibody in dogs and highlighted different FXR1 protein expression in canine melanocytic tumors, the significance of which is undergoing further investigations.
Introduction
FXR1 is an autosomal gene encoding for the cytoplasmic RNA-binding protein Fragile X mental retardation-related protein 1 (FXR1). FXR1 is a homologue of the Fragile X mental retardation syndrome protein FMR1, and both belong to the family of Fragile X mental retardation-related RNA-binding proteins together with Fragile X mental retardation-related protein 2 (FXR2).1–3 These proteins are involved in nuclear export, cytoplasmic transport, and translation control of target mRNAs. 4 FMR1, FXR1, and FXR2 are more than 70% homologous in their N-terminal half and have the same functional domains. 4 The human FXR1 and FXR2 genes are autosomally encoded and are located, respectively, in 3q28 and 17p13.2. 4
FMR1 spans about 40 kb encoding a mRNA of 3.9 kb. The N-terminal region has a functional nuclear localization signal (NLS), followed by two K homology (KH) domains, a nuclear export signal (NES), and an RGG box in the C-terminal region. 4 Its homologues FXR1 and FXR2 have two KH domains, like FMR1, and an RGG box. FXR1, FMR1, and FXR2 share 63% of amino acid identity in the first half of the protein. 5 Expression of the protein is similar but not overlapping; for example, FXR1 is highly expressed in muscle and heart, whereas FMR1 is almost absent, 4 while FXR2 is more expressed than FXR1 in the brain. 5 FXR1 and FXR2 are predominantly cytoplasmic, even if FXR2 and one FXR1 isoform have also a nucleolar targeting signal.4,5
FXR1 is highly conserved among vertebrates. 4 The FXR1 gene is conserved in chimpanzee, Rhesus monkey, dog, cow, mouse, rat, chicken, zebrafish, and frog. 6 FXR1 is a protein of 621 amino acids with a molecular mass of 69,721 Da. Posttranslational modifications include dimethylation of Arg-445. 7 Basic Local Alignment Search Tool (BLAST) analyses between human and dog FXR1 proteins reveal 99% identity (619/621 amino acids). 8 In human, mouse, and laboratory models (e.g., frog Xenopus laevis), FXR1 is normally expressed in a wide variety of tissues, with the highest level in muscle, heart, and testis,2,4,5 and it is considered to play a role in muscular cell development, 9 and in the inflammatory process by controlling the expression of tumor necrosis factor-α (TNF-α)10,11 and regulating the migration of monocytes. 12 Moreover, FXR1 has been investigated in oncology for its potential role as a key regulator of tumor progression, resulting in an overexpression at the genetic level, for example, in human lung squamous cell carcinoma, non-small cell lung cancer,13,14 and colorectal cancer. 15 It has been supposed that FXR1 affects DNA stability, 16 either regulating target mRNA expression by miRNA pathways 17 or regulating posttranscription by direct interaction with mRNA. 9 FXR1 was also demonstrated to be overexpressed in human and canine uveal malignant melanoma with a positive correlation with the metastatic potential of the tumor.18,19
Currently, the expression of FXR1 protein in normal canine tissues has not been investigated and, despite a few molecular studies on gene regulation, there are no data concerning the expression of FXR1 protein in canine melanocytic tumors. To the best of the authors’ knowledge, anti-FXR1 antibodies were not previously validated in the canine species. Therefore, in the present study, the authors tested the immunoreactivity of FXR1 protein in normal canine tissues and melanocytic tumors.
Materials and Methods
Western Blot
Canine fibroblasts, canine serum, and human serum were used as samples during Western blot experiments.
Primary cultures of fibroblasts were established from abdominal full-thickness skin excised from the margins of a specimen obtained during therapeutic surgical procedure, as previously described with slight modifications. 20 Immediately after collection, skin fragments were placed in PBS on ice; all the fat tissue was removed from the skin with scissors, skin tissues were trimmed to 0.5 cm × 0.5 cm fragments, and finely minced into small pieces evenly distributed onto the bottom of a tissue culture 25-cm2 flask in DMEM Medium with GlutaMax (Gibco Thermo-Fisher; cat. no 10569-010) supplemented with 10% of FBS. After 24 hr of culture, when the pieces have adhered, the medium volume was gradually increased to 5 ml. After a week, when a substantial outgrowth of cells was observed, the explants were removed from the center of the outgrowth with a scalpel, and the medium was replaced. Fibroblasts were cultured until the outgrowth has spread to cover at least 70% of the growth surface.
All the chemicals and reagents used in this part of study were purchased from Sigma-Aldrich-Merck KGaA; Darmstadt (Germany), except for those specifically mentioned.
Cells at passage 0 were harvested by incubation for 5 min on ice with 0.25% trypsin in 0.01% EDTA and collected by centrifugation. Protein extraction was carried out on ice or at 4C. Fibroblasts were homogenized in 5 μl/mg TES buffer (10 mM Tris–HCl, pH 7.6; 1 mM EDTA, 0.25 M sucrose) and centrifuged at 10,000 × g, for 30 min, at 4C. Protein concentration values were determined by the Pierce BCA Protein Kit (VWR), using BSA as a protein standard, according to the manufacturer’s manual.
A goat anti-FXR1 polyclonal antibody (cat. no ab51970; Abcam, Cambridge, UK), produced against the peptide corresponding to aa 317–330 of human FXR1, and a rabbit anti-FXR1 polyclonal antibody (ab50841; Abcam), produced against a synthetic human peptide non-conserved between FXR1 and FXR2, were used throughout the experiments. Because the cross-reactivity of the antibodies had never been validated before in dogs, a preliminary step to assess their possible utilization for canine FXR1 detection was carried out. Serum from human and dog, and protein extracted from canine fibroblasts, were separated on a 12% SDS-PAGE and blotted onto nitrocellulose membrane. Before gel separation, 1 µl ß-mercaptoethanol (Sigma-Aldrich; St. Louis, MO) was added to each sample.
Immunolabeling was performed using primary antibodies at 0.25 µg/ml for 1 hr at room temperature, while an anti-goat IgG labeled with peroxidase (Sigma-Aldrich) and an anti-rabbit IgG labeled with peroxidase (Vector Laboratories; Burlingame, CA) were used as secondary antibodies (1:5000 dilution for 45 min at room temperature), respectively. Antibodies were diluted using Roti-Block 1× (Carl Roth; Karlsruhe, Germany). Immunoreactive bands were visualized by enhanced chemiluminescence (ECL) using Immobilon Western Chemiluminescent HRP Substrate (Millipore; Billerica, MA). To further assess the specificity of the secondary antibodies, Western blotting experiments were repeated replacing the primary antibodies with PBS and using only the respective secondary antibodies labeled with peroxidase (1:5000 dilution for 45 min at room temperature), to perform a negative control and detect any possible nonspecific bands. Human serum was used as positive control. Fibroblasts were used as negative control, as these cells do not express FXR1 protein. 21
IHC
Immunohistochemical expression of FXR1 protein was tested in canine normal tissues and melanocytic tumors. A pool of canine tissues was examined, using microarrays of normal tissue created by assembling multiple formalin-fixed samples (5 × 5 × 3 mm each) in the same paraffin block. Tissues examined included stomach, intestine, pancreas, liver, thyroid, adipose tissue, lung, kidney, testis, brain, heart, adult, and fetal skeletal muscle. IHC was also performed on conventional tissue sections of canine oral (n=10) and uveal (n=10) melanocytic tumors.
Serial sections were cut 4 μm thick and mounted on polylysine-coated slides (Menzel-Gläser; Braunschweig, Germany). Immunohistochemical staining with the standard avidin-biotin-peroxidase complex (ABC) method was performed.
Preliminary setup of the immunohistochemical protocol compared different heat-induced antigen retrieval methods (microwave oven, pressure cooker, water bath) and serial concentration of the primary antibody. The chosen protocol applied to the sections was the following: sections were deparaffinized in xylene and rehydrated through a descending series of ethanol concentrations. The endogenous peroxidase activity was blocked with 0.3% H2O2 in methanol for 30 min. Antigen retrieval was performed by heating the slides in citrate buffer solution (pH 6.5) in a water bath at 95C for 30 min, followed by cooldown in buffer at room temperature for 30 min. Sections were, therefore, incubated for 20 min at room temperature with normal goat serum (1:70) to block any nonspecific protein binding. Serial sections were incubated at 4C overnight in a humidified chamber with the primary antibody: rabbit polyclonal anti-FXR1 (ab50841; Abcam, Cambridge, UK), 1:100 dilution.
Sections were then rinsed in Tris buffer solution, and incubated with the secondary anti-rabbit biotinylated antibody (1:200, 30 min at room temperature; Vector Laboratories), followed by rinsing in Tris buffer and, therefore, incubation with the ABC reagent (30 min at room temperature; Vector Laboratories). The chromogen 3-amino-9-ethylcarbazole (AEC; Vector Laboratories) was applied for 15 min and, after rinsing in tap water, slides were counterstained with Mayer’s hematoxylin (Diapath srl; Martinengo, Italy) for 2 min. Slides were, therefore, dried and mounted in aqueous mounting agent (Aquatex, Merck; Darmstadt, Germany).
Negative controls for the technique were carried out by replacing the primary antibodies with rabbit IgG (Santa Cruz; Dallas, TX).
Fibroblasts were used as negative controls. Canine fetal muscle was used as positive control.
Immunolabeling was semi-quantitatively scored with postexamination masking method 22 by two pathologists (L.N., C.G.) simultaneously. At the time of the samples’ evaluation, a prior extensive revision of human literature was intentionally not performed to avoid bias in the scoring of tissues. In normal canine tissue and melanomas, the percentage of positive cells was recorded (<10%, 10–30%, 30–50%, 50–80%, >80%), and the intensity of single cell staining was scored from 1+ (mild) to 3+ (marked). A 3+ signal equated to an intensity easily seen on low magnification, whereas a 1+ signal was seen on high magnification, and 2+ bordered between a strong and a weak staining signal. 23 The immunoreactive score of Remmele and Stegner (IRS score) was calculated combining the intensity and ratio of positivity as follows 24 : IRS score (0–12) = percentage of positive cells (with no positive cells = 0, <10% positive cells = 1, 10–50% of positive cells = 2, 50–80% of positive cells = 3, >80% of positive cells = 4) × intensity of staining (with no positive cells = 0, 1+/mild = 1, 2+/moderate = 2, 3+/marked = 3).
Results
Western Blot
Western blotting experiments were carried out to determine whether the antibodies cross-reacted with the respective canine protein. The rabbit anti-FXR1 antibody cross-reacted with a band of 60–70 kDa, corresponding to the predicted molecular mass (predicted molecular mass = 68 kDa, band detected by the producer = 65 kDa; Fig. 1). Canine fibroblasts did not show any cross-reactivity. In the negative control for the secondary antibody, the incubation with the lone anti-rabbit secondary antibody did not show reactive bands (see Supplementary Material). Conversely, goat anti-FXR1 antibody showed bands lower than expected molecular mass (predicted molecular mass = 68 kDa, band detected by the producer = 80 kDa; see Supplementary Material). These results indicated that rabbit anti-FXR1 polyclonal antibody specifically recognized the canine FXR1 protein and did not react with other canine serum proteins, while goat anti-FXR1 appeared nonspecific.
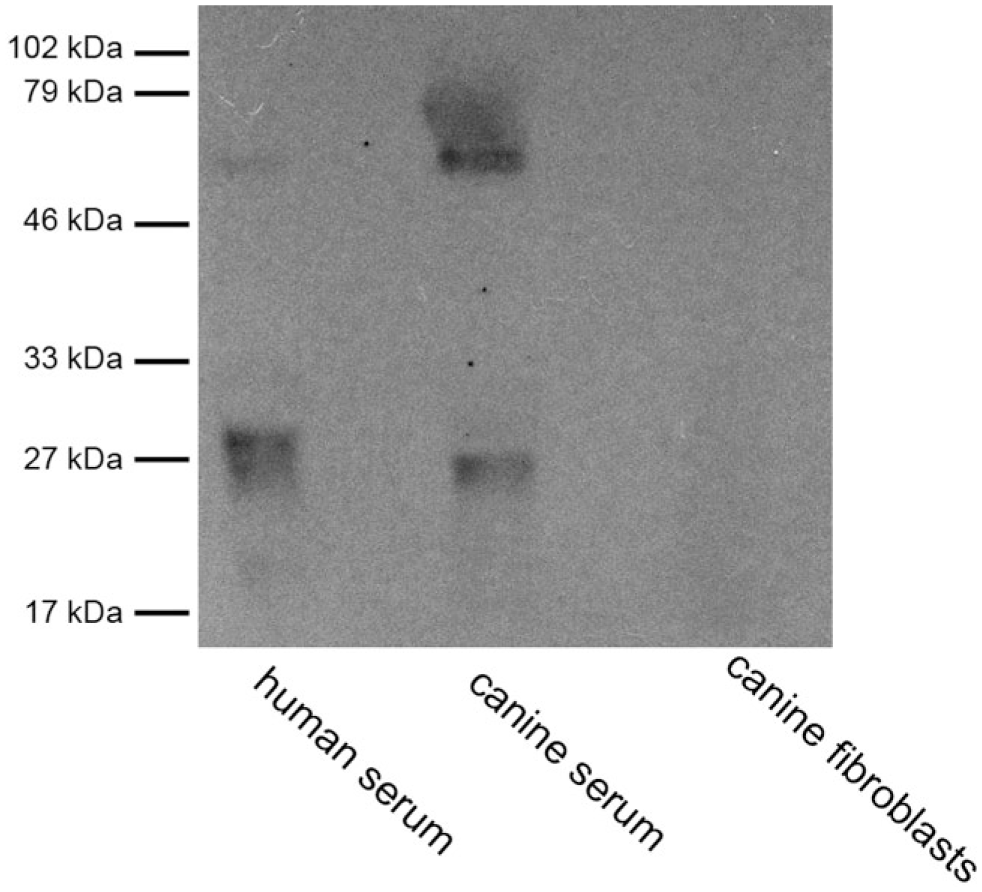
Human and canine sera and canine fibroblasts, detection of FXR1 protein, Western blot analysis. Rabbit polyclonal anti-FXR1 antibody ab50841. Abbreviation: FXR1, Fragile X mental retardation-related protein 1.
IHC
Based on Western blot results, rabbit anti-FXR1 antibody was used to perform immunohistochemical tests.
Different normal canine tissues stained positive, as detailed in Table 1. Staining was always intracytoplasmic, and its intensity varied from mild (1+) to marked (3+; Fig. 2).
Immunohistochemical Expression of FXR1 in a Series of Normal Canine Tissues.
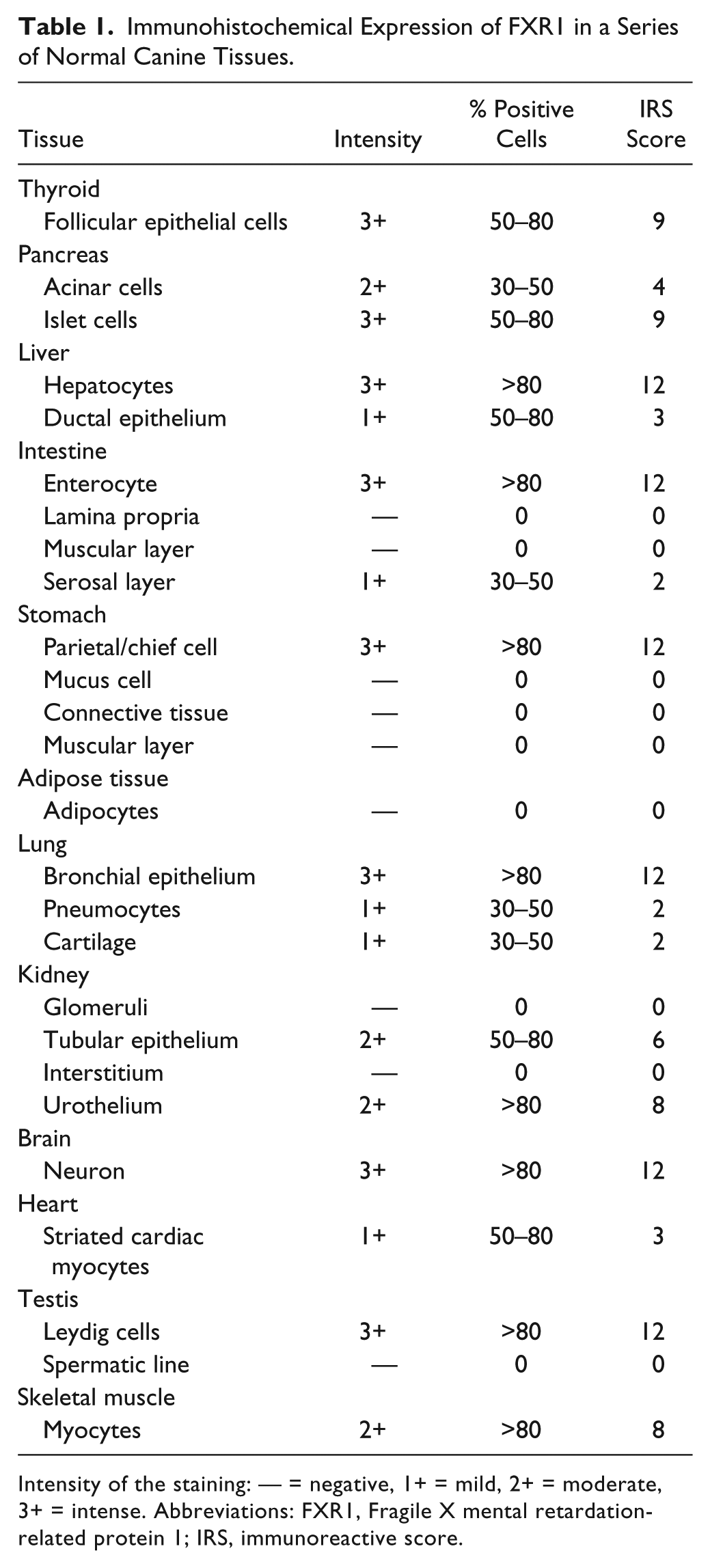
Intensity of the staining: — = negative, 1+ = mild, 2+ = moderate, 3+ = intense. Abbreviations: FXR1, Fragile X mental retardation-related protein 1; IRS, immunoreactive score.
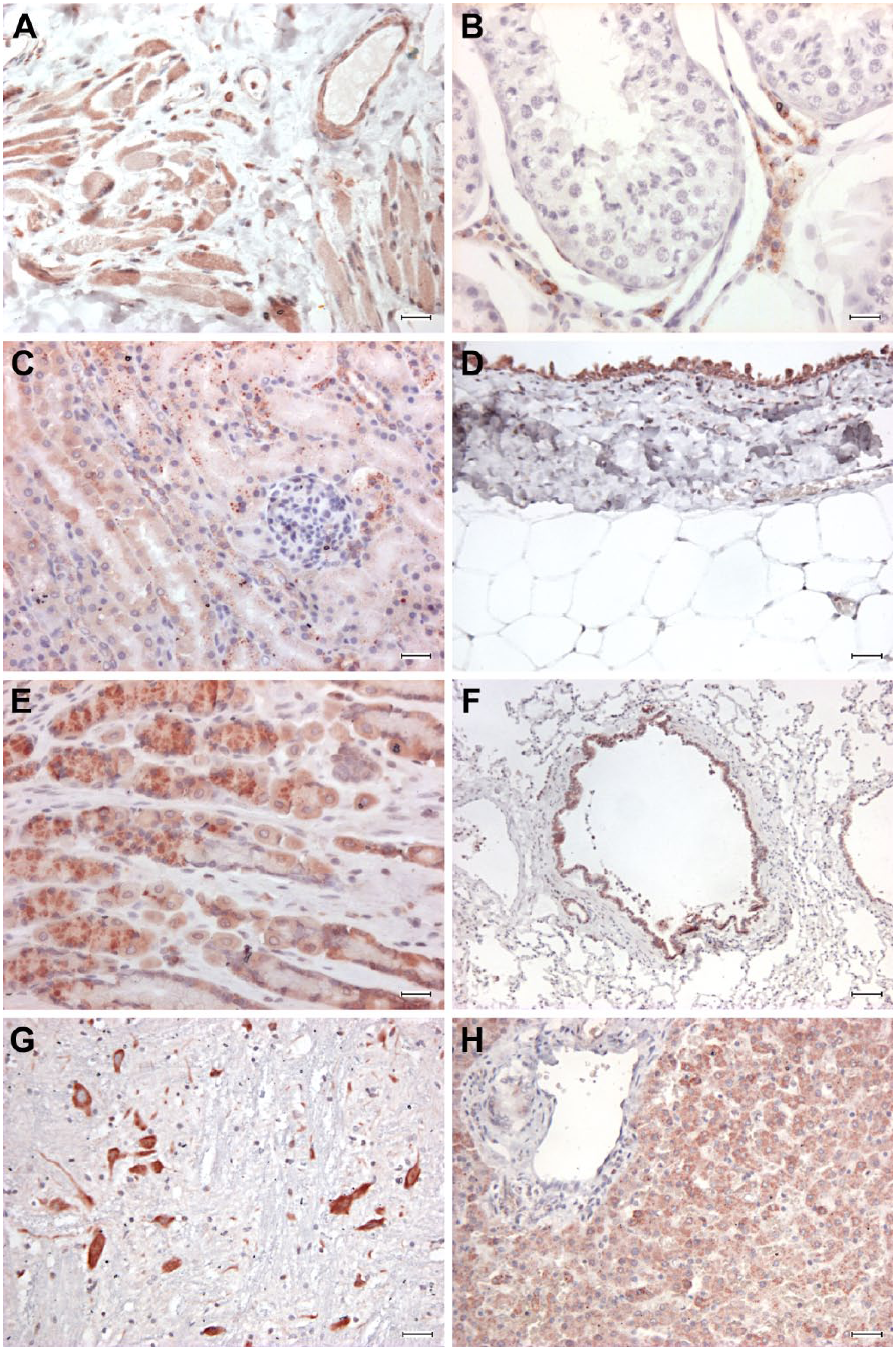
Normal tissues, dog. Immunohistochemical staining anti-FXR1, rabbit polyclonal anti-FXR1 antibody ab50841, ABC method, AEC chromogen. (A) Skeletal muscle, diffuse moderate to intense immunostaining of skeletal fibers. (B) Testis, intense immunostaining of interstitial cells. (C) Kidney, moderate reactivity of tubular epithelium and no reactivity of renal glomeruli. (D) Kidney, intense immunostaining of the urothelium, negative staining of connective tissue. (E) Stomach, intense immunoreactivity of parietal/chief cells. (F) Lung, intense staining of bronchial epithelium and mild scattered staining of pneumocytes. (G) Brain, intense reactivity of neurons. (H) Liver, diffuse moderate immunoreactivity of hepatocytes and mild reactivity of ductal epithelium. Abbreviations: FXR1, Fragile X mental retardation-related protein 1; ABC, avidin-biotin-peroxidase complex; AEC, 3-amino-9-ethylcarbazole. A–C, E scale bars = 25 μm; D scale bar = 50 μm; F scale bar = 100 μm; G–H scale bars = 50 μm.
In detail, in the gastrointestinal tract, the mucosal epithelium exhibited strong immunoreactivity in the stomach (parietal/chief cells) and intestine (enterocytes), while the connective tissue of the lamina propria and the muscular layers were not stained. In the liver, hepatocytes were strongly positive and ductal epithelium stained mildly for FXR1 expression. Pancreatic acini stained moderately. In the lung, pneumocytes stained mildly and bronchial epithelium intensely; in the kidney, tubular epithelial cells and the pelvis urothelium were diffusely and moderately positive, while glomeruli did not stain. Thyroid follicular epithelial cells were diffusely and strongly positive. Neurons in the brain stained intensely for FXR1. Adult striated skeletal muscles was diffusely, moderately FXR1 positive, while striated cardiac muscles were mildly positive. Canine fetal skeletal muscle was intensely immunoreactive. In blood vessels, the endothelium multifocally stained moderately positive for FXR1, while the smooth muscle of the tunica media was mostly negative, and only occasionally mildly positive. An intense signal was detected in the Leydig cell of the testis, whereas Sertoli and seminal cells were always negative. Stromal connective tissue was consistently and diffusely negative for FXR1.
Concerning melanocytic tumors, 9 (9/10) oral melanomas were FXR1 positive (Table 2, Fig. 3A–B). The immunostaining was always intracytoplasmic. Four cases were above 80% of positive neoplastic cells, one ranged 50–80%, two 30–50%, one 10–30%, and one case was negative. Immunohistochemical staining intensity ranged, in all cases but one, from intense to moderate. Ten (10/10) uveal melanocytic tumors were positive to FXR1 immunostaining. The cases were homogeneously and variably distributed among the semiquantitative classes of evaluation (Table 2; Fig. 3C–D). Three cases were above 80% of positive neoplastic cells, four varied between 30–50%, and three had less than 10% of positive neoplastic cells. Three cases were intensely positive, six moderately, and one case was mildly positive. In tissue-circumscribing tumors, scattered melanocytes with dendritic appearance were recognizable, and they did not stain. No immunolabeling was observed in negative controls.
Immunohistochemical Expression of FXR1 in 10 Canine Oral and Uveal Melanocytic Neoplasms.
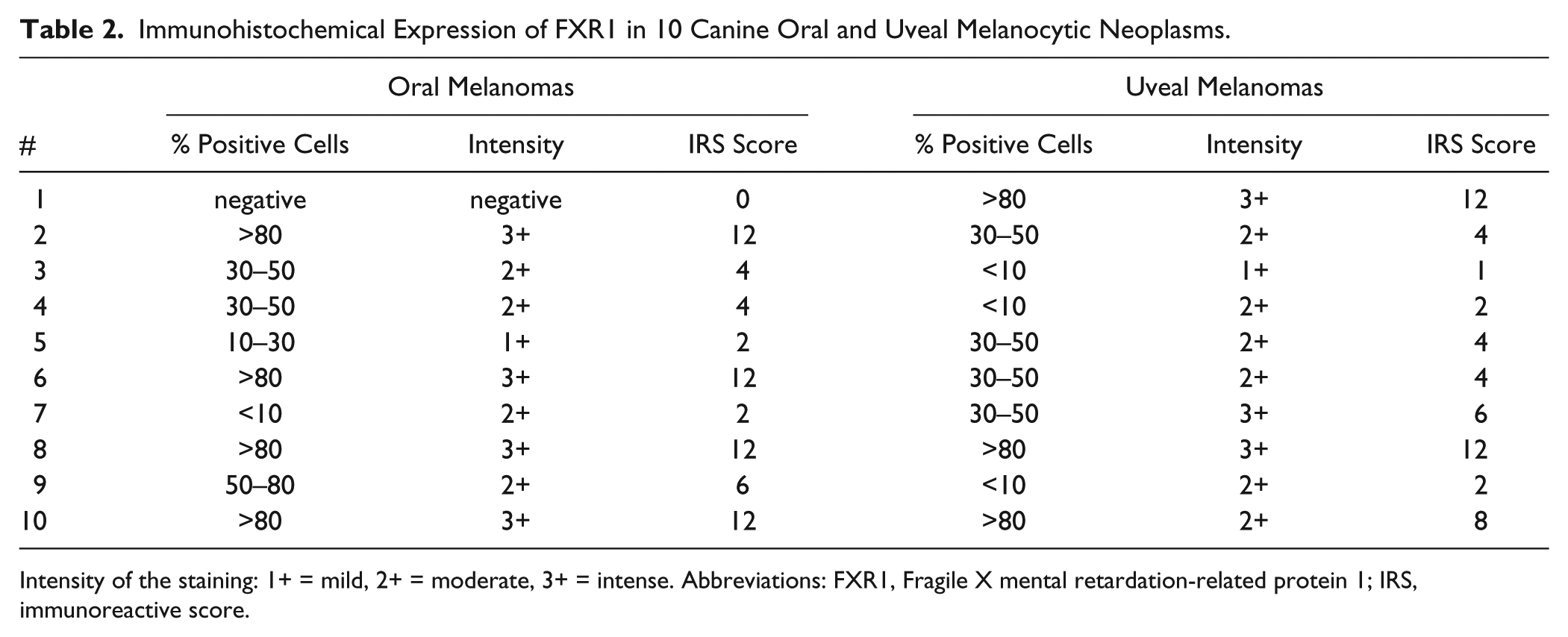
Intensity of the staining: 1+ = mild, 2+ = moderate, 3+ = intense. Abbreviations: FXR1, Fragile X mental retardation-related protein 1; IRS, immunoreactive score.
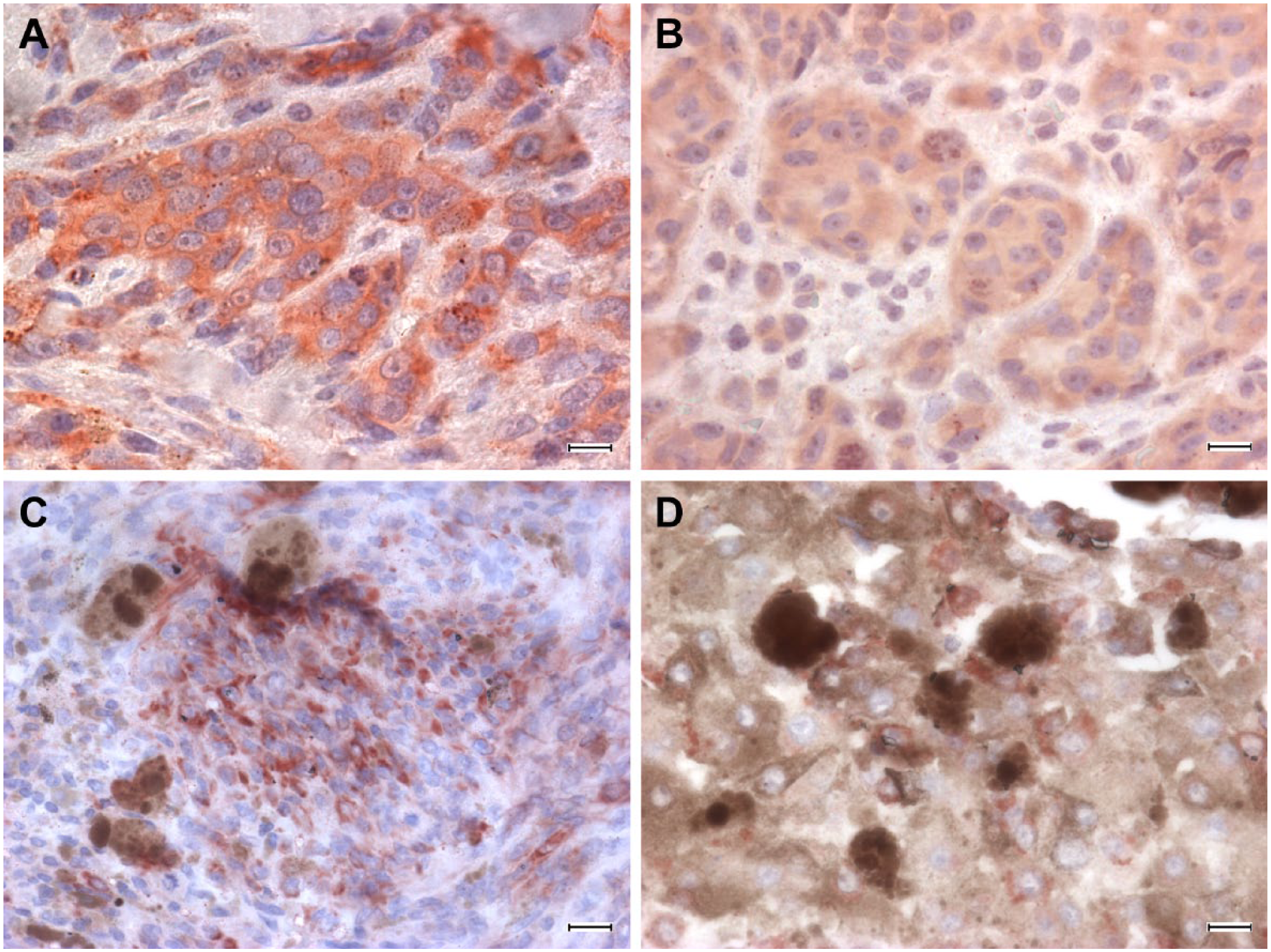
Melanoma, dog. Immunohistochemical staining anti-FXR1, rabbit polyclonal anti-FXR1 antibody ab50841, ABC method, AEC chromogen. (A–B): oral melanoma, diffuse intense to moderate staining of neoplastic cells. (C–D): uveal melanoma, intense staining of neoplastic cells, multifocal to scattered. Abbreviations: FXR1, Fragile X mental retardation-related protein 1; ABC, avidin-biotin-peroxidase complex; AEC, 3-amino-9-ethylcarbazole. Scale bars = 12.5 μm.
Discussion
FXR1 is a cytoplasmic RNA-binding protein whose codifying gene’s overexpression has been related to tumor progression in different human cancers,13–15 and more specifically to metastasizing behavior in human and canine uveal melanoma.18,19
In the current literature, few studies investigated FXR1 protein localization in normal human tissue14,25 and in different tumors.13–15,19 Detailed tissue expression of FXR1 has been described in the Human Protein Atlas. 21 To the best of the authors’ knowledge, no data are available concerning the tissue immunolocalization of FXR1 protein in the canine species. Moreover, commercially available anti-human antibodies were not previously validated in dogs.
The commercially available anti-human polyclonal anti-FXR1 antibodies tested in the present study were predicted to react with the canine species. On Western blotting, a rabbit anti-FXR1 antibody showed a band with a molecular mass consistent with what is expected as per manufacturer’s indications (predicted molecular mass: 68 kDa, band detected by the producer: 65 kDa), and, therefore, it was considered to react specifically with human and canine serum. Moreover, the polyclonal antibody rabbit anti-FXR1 is raised against an immunogen sequence within aa 338–513, a sequence that is not present in the FXR2 sequence, which is characterized by a homology of 70% with FXR1, 4 further endorsing the specific binding of this antibody to FXR1 protein. The goat anti-FXR1 antibody recognized a band with a molecular mass lower than expected, and it was, therefore, considered not specific and excluded from further analysis.
In the present study, the specific tissue immunolocalization of FXR1 protein was investigated for the first time in the canine species. On immunohistochemical examination, FXR1 protein was detected in normal canine tissues with different degrees of intensity, and the result was, it was present in at least one tissue or cell population in all organs tested. These results in canine tissues were consistent with the immunohistochemical expression of FXR1 in human tissues as described in the Protein Atlas. 21 Connective tissue in the dog was not immunoreactive: this finding was consistent with the predicted results according to Western blot results and human data. 21 An intense FXR1 expression was particularly observed in canine fetal skeletal muscle, testicular interstitial cells of Leydig, and neurons. These results were consistent with the human literature, where FXR1 is reported to be expressed in different tissues,6,25 even if studies on FXR1 expression in normal tissue mostly relied on investigations of gene or mRNA expression.2,6 Anyway, canine and human tissues exhibited also slight differences in FXR1 expression, for example, in the testis, where the spermatic line resulted as negative, whereas it is reported to be strongly positive in humans. It may be reasonable to hypothesize that tissue-specific isoforms of FXR1 exist and that they probably have a different distribution and expression in different species. Since 1995, reverse transcription (RT)-PCR analyses on human tissues revealed that different isoforms of FXR1 exist in brain and testis, presumably suggesting a tissue-specific alternative splicing. 2 Isoforms share extensive regions of identity and vary only in specific domains. 2 This fact probably accounts for tissue-specific functioning of the protein,2,5 thus, explaining subtle differences in the expression between humans and dogs.
The FXR1 gene has been previously shown to be overexpressed in a subset of human and canine uveal malignant melanomas with higher tendency toward a metastasizing behavior.18,19 However, to the best of the authors’ knowledge, there is no knowledge about the expression of the FXR1 protein in canine melanomas, and no data indicating if FXR1 is peculiar to uveal melanomas or if it is also expressed in melanomas arising in different body sites. In the present study, melanoma of the oral cavity was selected because it can be an aggressive tumor, but its progression and metastasizing behavior are not always predictable based only on histomorphological findings. Interestingly, in the cohort of cases investigated in the present study, FXR1 immunostaining was observed in all but one of the tested oral melanomas, with different levels of staining intensity and percentage of positive cells (results ranged from a negative case up to more than 80%). These data indicated that the FXR1 protein is actually expressed also in canine oral melanomas, and the results may suggest a further investigation of the possible relation between the immunohistochemical expression and the biological behavior.
In the cohort of uveal melanocytic tumors examined, all cases were positive for FXR1 even though with different levels of protein expression in terms of staining intensity and number of stained neoplastic cells. The immunohistochemical expression of FXR1 in melanoma should be compared, in the future, with the follow-up data, to determine if it may provide a prognostic meaning, as already demonstrated for the FXR1 gene.18,19
In the present study, two anti-FXR1 protein antibodies were tested, and the rabbit polyclonal anti-FRX1 showed a more specific binding to the canine FXR1 protein. The distribution of a positive immunohistochemical signal obtained with rabbit anti-FXR1 antibody may indicate its potential ability to discriminate among different classes of protein expression possibly related to the tumor behavior. Further studies are ongoing to correlate FXR1 immunostaining with a prognostic significance in canine melanomas.
Supplemental Material
JHC766292_Supplemental_Figure_1_online_supp – Supplemental material for Immunohistochemical Expression of FXR1 in Canine Normal Tissues and Melanomas
Supplemental material, JHC766292_Supplemental_Figure_1_online_supp for Immunohistochemical Expression of FXR1 in Canine Normal Tissues and Melanomas by Laura Nordio, Andreia T. Marques, Cristina Lecchi, Alberto M. Luciano, Damiano Stefanello and Chiara Giudice in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
The authors are grateful to Prof. Fabrizio Ceciliani for the technical support in the Western blot experiments.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: all authors (LN, ATM, CL, AML, DS, CG) contributed to the experimental design, writing, and editing of the manuscript; AML cultured fibroblast cells; LN, ATM, and CL performed the Western blot; LN and CG performed the histology and IHC; and all authors have read and approved the manuscript as submitted.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
