Abstract
The standard method for detecting triple-stranded DNA over the last 1.5 decades has been immune detection using antibodies raised against non-canonical nucleic acid structures. Many fluorescent dyes bind differentially to nucleic acids and often exhibit distinctive staining patterns along metaphase chromosomes dependent upon features, including binding to the major and minor DNA grooves, level of chromatin compaction, nucleotide specificity, and level of dye stacking. Relatively recently, the fluorochrome Thiazole Orange (TO) was shown to preferentially bind to triplex DNA in gels. Here, we demonstrate that TO also detects triplex DNA in salivary gland chromosomes of Drosophila melanogaster and Rhynchosciara americana identical in location and specificity to observations using antibodies. This finding may enable triple-stranded DNA investigations to be carried out on a much broader and reproducible scale than hitherto possible using antibodies, where a frequently encountered problem is the difference in detection specificity and sensitivity between one antibody and another.
Introduction
Use of fluorescent dyes for chromosome staining started in the 1960s when acridine orange was found to label chromosomes, 1 although banding patterns were not recognized at this time. Later, Caspersson and coworkers were able to detect reproducible banding patterns in human chromosomes using quinacrine mustard and assumed that the staining specificity was determined by preferential binding of the dye’s mustard group with the N-7 nucleophilic center of guanine and presumably followed the distribution of G/C (guanine-cytosine) enriched DNA. 2 However, quinacrine dihydrochloride (Atebrin—used in huge quantities during World War II as an antimalarial agent) was shown to stain the human Y chromosome with the same specificity as quinacrine mustard 3 and cast doubts on the G/C specificity of quinacrine mustard. Indeed, later work confirmed that quinacrine bright regions (bands) in human chromosomes were A/T (adenine-thymine) rich, although contradictory interpretations were drawn on whether the fluorescence patterns were entirely due to differential base binding, differential quenching, or a combination of both. 4 However, A/T enriched fluorescent banding patterns in chromosomes were obtained with Hoechst 33258,5,6 4′,6-diamidino-2-phenylindole (DAPI),7,8 and G/C enriched bands with chromomycin. 8 Despite extensive studies on the interaction between organic dyes and nucleic acids, little was determined on the ability of most fluorochromes to bind differentially to conformations other than double- or single-stranded DNA.
Assessment of non B-DNA structures in chromosomes has been mainly achieved through development of antibodies to specific nucleic acid conformations. Antihybrid DNA.RNA antibodies9,10 have been employed to detect corresponding structures after in situ hybridization in chromosomes using either RNA probes11–14 or to map transcriptionally active regions because transcribed RNA tends to form duplex molecules with its template DNA.15–21 Triplex DNA, of particular interest for this study, is created when poly-purine or poly-pyrimidine single-stranded DNA binds in the major groove of the double helix, forming Hoogsteen or reverse Hoogsteen hydrogen bonds. Although Watson-Crick base pairing predominates over the bulk of genomic DNA, the possibility that naturally occurring triple-helical nucleic acid structures play functional roles in eukaryotes has received increasing attention, and many types of cancer exhibit enhanced levels of triplex DNA binding proteins. 22 Antibodies to triplex conformations were found to react with mammalian and insect chromosomes.23–31 Evidence for triple-helices in the control of gene expression has also been shown by combining immunocytochemical detection with molecular biology methods.32,33
The fluorophore known as Thiazole Orange {TO, 4-[3-methyl-2,3-dihydro(benzo-1,3-thiazole)-2-methylidene]quinolinium iodide} binds to both RNA and DNA 34 and has been used extensively in flow cytometry. More recent in vitro studies demonstrated that the affinity of TO to triplex and quadruplex DNA is orders of magnitude higher than for double-stranded DNA. 35 In addition, TO was found to selectively stain nucleoli of immortalized cells, suggesting the presence of non-canonical nucleic acid configurations in the nucleolar domain characterized by a high abundance of RNA molecules. Extensive cytoplasmic labeling was presumed to result from TO interaction with tRNAs known for their potential to adopt unusual helical shapes. While the Lubitz et al. study demonstrates that TO may preferentially bind to triplex and quadruplex DNA in gels, the observed nucleolar and cytoplasmic localization in cells might also reflect RNA-dye binding 34 independent of strand complexity. We consider that an in situ validation of the specificity of TO staining to triple-stranded DNA necessitates doubly stained preparations in which the specificity of TO staining to triplex DNA in chromosomes can be directly compared with immunological detection, as demonstrated previously in the pericentric heterochromatin of polytene chromosomes from the salivary glands of Rhynchosciara americana (Diptera: Sciaridae) and Drosophila melanogaster. 31 Our observations confirm the correspondence of labeling between TO and triplex immunodetection showing that the dye TO can be used as an in situ marker for some triple-helical DNA structures.
Materials and Methods
Animals
Rhynchosciara americana larvae were collected in the coastal region of Mongaguá in the state of São Paulo, Brazil. Drosophila melanogaster larvae (Canton S) came from laboratory stocks.
Preparation of Chromosome Spreads
Salivary glands and brain ganglia were fixed in ethanol-acetic acid (3:1), squashed in 40% acetic acid, frozen in liquid nitrogen, and stored in ethanol at −10C after coverslip removal. For enzymatic treatments, the slides were rehydrated in 1× TBS (25 mM Tris, 0.14 M NaCl, 3 mM KCl) followed by incubation at room temperature with RNase A (Calbiochem; San Diego, CA) diluted (0.2 mg/ml) in 2× SSC (1× SSC: 0.15 M NaCl, 0,015 M sodium citrate) for 2 hr. The slides were washed in 1× TBS prior to staining procedures.
TO Staining
TO (Sigma-Aldrich Chemical Co.; St. Louis, MO) was dissolved in methanol and its concentration determined with a Cary 50 Bio UV Spectrophotometer (Varian Instruments; Walnut Creek, CA) using the mean of 3 wavelength values according to the TO data sheet. Distinct concentrations of TO were tested for chromosome staining: dissolving the stock solution (0.3 mM) in 1× PBS following incubation at room temperature for 5 min and subsequent washing in 1× PBS for 20 min. The slides were mounted in antifading medium (VectaShield, Vector Labs.; Burlingame, CA) and inspected with epifluorescence optics (Nikon Instruments Inc.; Melville, NY), using excitation wavelengths of 488 and 532 nm.
Immunocytochemical Detection of Triple-helical Nucleic Acids
The slides were rehydrated in 1× TBS followed by incubation at room temperature in 1× TBS, 0.1% Triton X-100 (TBST), 2% low-fat powdered milk for 20–30 min. Slide incubations with purified anti-poly(dA).poly(rU).poly(rU)31,36 diluted 1:50 in TBST solution were performed in a moistened chamber at room temperature for 2 hr. After washes in TBST, the slides were incubated for 1 h at room temperature with sheep IgG anti-rabbit IgG conjugated with tetramethylrhodamine (TRITC; Sigma Chemical Co.; St. Louis, MO) diluted 1:100 in TBST solution. The slides were washed twice in TBST for 30 min and finally in 1× TBS for 5 min. Preparations were counterstained with DAPI. The slides were mounted in antifading medium (VectaShield, Vector Labs.) and inspected with epifluorescence optics (Nikon Instruments Inc.).
Simultaneous Visualization of Anti-triplex and TO Staining
The slides were first processed for anti-triplex detection, as described above. Images were captured with an Axiophot 2 Photomicroscope (Carl Zeiss; Oberkochen, Germany) equipped with a Zeiss charge-coupled device (CCD) camera (AxioFan MRm) and coupled to an image analysis software package (ISIS, Zeiss). Chromosomal sites that were labeled by antibodies (TRITC channel) displayed no fluorescent signal in the FITC channel. Precise coordinates of each image were registered for further inspection after TO staining. The coverslips were pried off by washing the slides in TBST for 1 hr at room temperature and once in 1× TBS for 10 min. The chromosome spreads were then stained by TO, as described previously, and fluorescent signals captured in the FITC channel.
“Endogenous” Hybridization
The slides kept frozen in ethanol were briefly immersed in 1× TBS and subsequently incubated in 50% acetic acid at 45C for 5 min. For detection of endogenous RNA.DNA hybrid, the slides were washed in TBST for 5 min and incubated with goat IgG anti-poly-(rA).poly-(dT)9,10 diluted 1:50 in TBST. After 1 hr at room temperature in a moistened chamber, the slides were washed twice in TBST for 30 min and incubated with rabbit IgG anti-goat labeled with TRITC (Sigma Chemical Co.; St. Louis, MO) diluted 1:200, as described above. The slides were washed twice in TBST for 30 min and finally once in 1× TBS for 5 min. Mounting and inspection of the slides were done as described for immunostaining.
Results
Millimolar TO Concentrations Do Not Bind Differentially to Chromosomal DNA of Drosophila and Rhynchosciara
Initially, we tested TO staining in the millimolar range (0.1–0.3 mM) on polytene chromosomes of D. melanogaster and R. americana untreated with RNase. The same results were seen when 20–60 µM concentrations of TO were used. The TO labeling at millimolar concentrations corresponded exactly with the DAPI staining. Fluorescence from TO excitation could be visualized using either FITC or TRITC filters (Fig. 1A and B). Chromosome spreads treated with RNase A and subsequently stained by TO did not display significant changes in fluorescence levels relative to untreated slides, suggesting that RNA does not contribute significantly to labeling intensity. This was confirmed by the observation that the well-formed nucleoli of Drosophila, a nuclear region that has long been characterized by intense rRNA synthesis and accumulation, always appeared very weakly labeled by TO (Fig. 1D). “DNA puffs” are typical of sciarid polytene chromosomes of the salivary gland and also examples of regions in which high rates of DNA and RNA synthesis take place. 37 Also in this case, TO staining, if any, is weak (Fig. 2) even though uridine incorporation and RNA in the form of DNA.RNA hybrids are readily detected in these structures, as seen in the B2 “DNA puff” (Fig. 3).
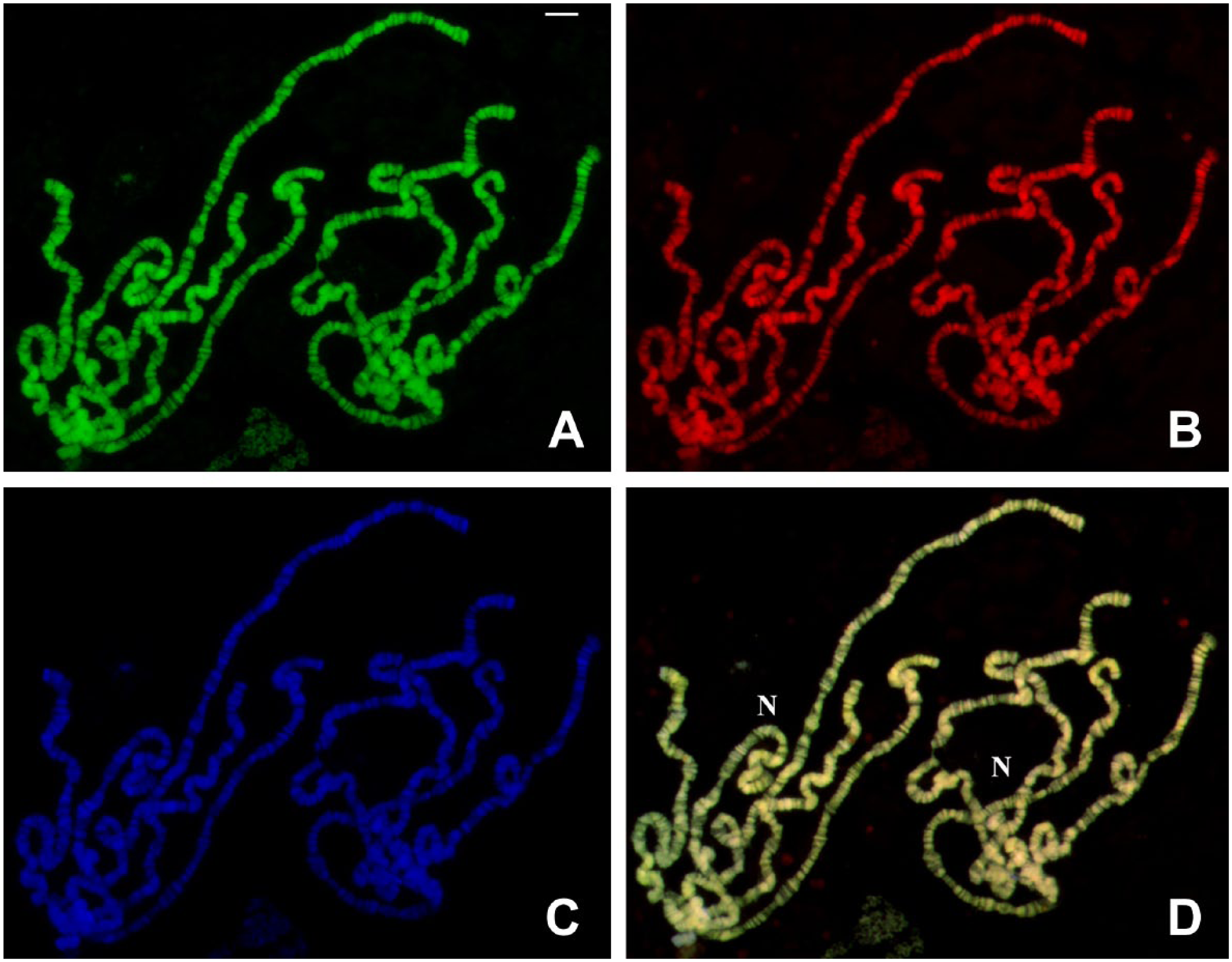
TO and DAPI staining in polytene chromosomes of D. melanogaster untreated with RNases. (A) TO staining captured in the FITC channel. (B) TO staining visualized in the TRITC channel. (C) DAPI staining of the same chromosomes and (D) the superimposed (A–B) images. Areas around which nucleoli are formed appear indicated (N). TO in millimolar concentration was employed in the experiment. Bar = 10 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole; FITC, fluorescein isothiocyanate; TRITC, tetramethylrhodamine.
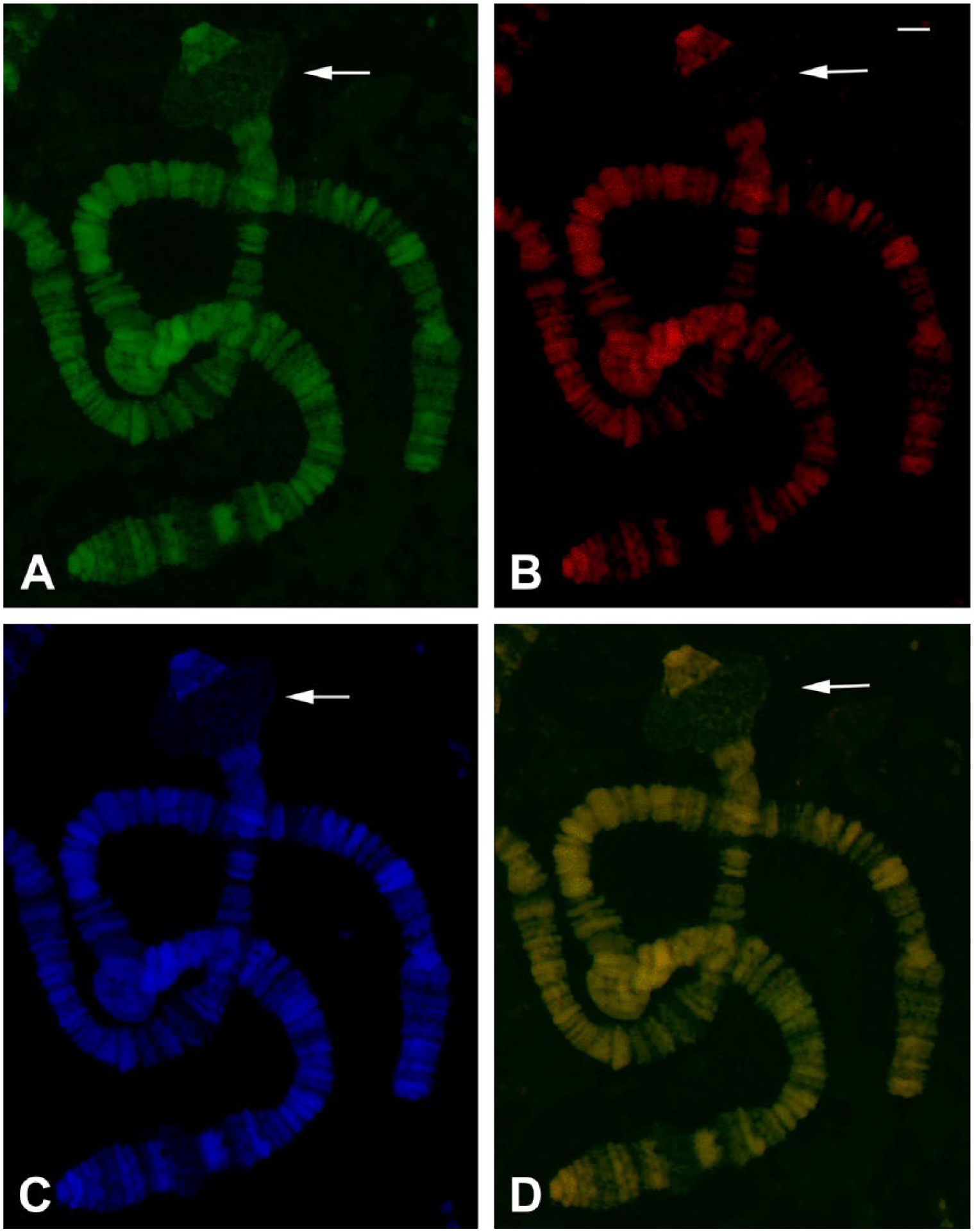
TO and DAPI staining in polytene chromosomes of R. americana untreated with RNases. (A) TO staining captured in the FITC channel. (B) The corresponding TO staining visualized in the TRITC channel. (C) DAPI staining of the same chromosomes. (D) FITC and TRITC channels superimposed. The arrows indicate the B2 DNA puff site. TO concentration in the millimolar range was employed in the experiment. Bar = 20 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole; FITC, fluorescein isothiocyanate; TRITC, tetramethylrhodamine.
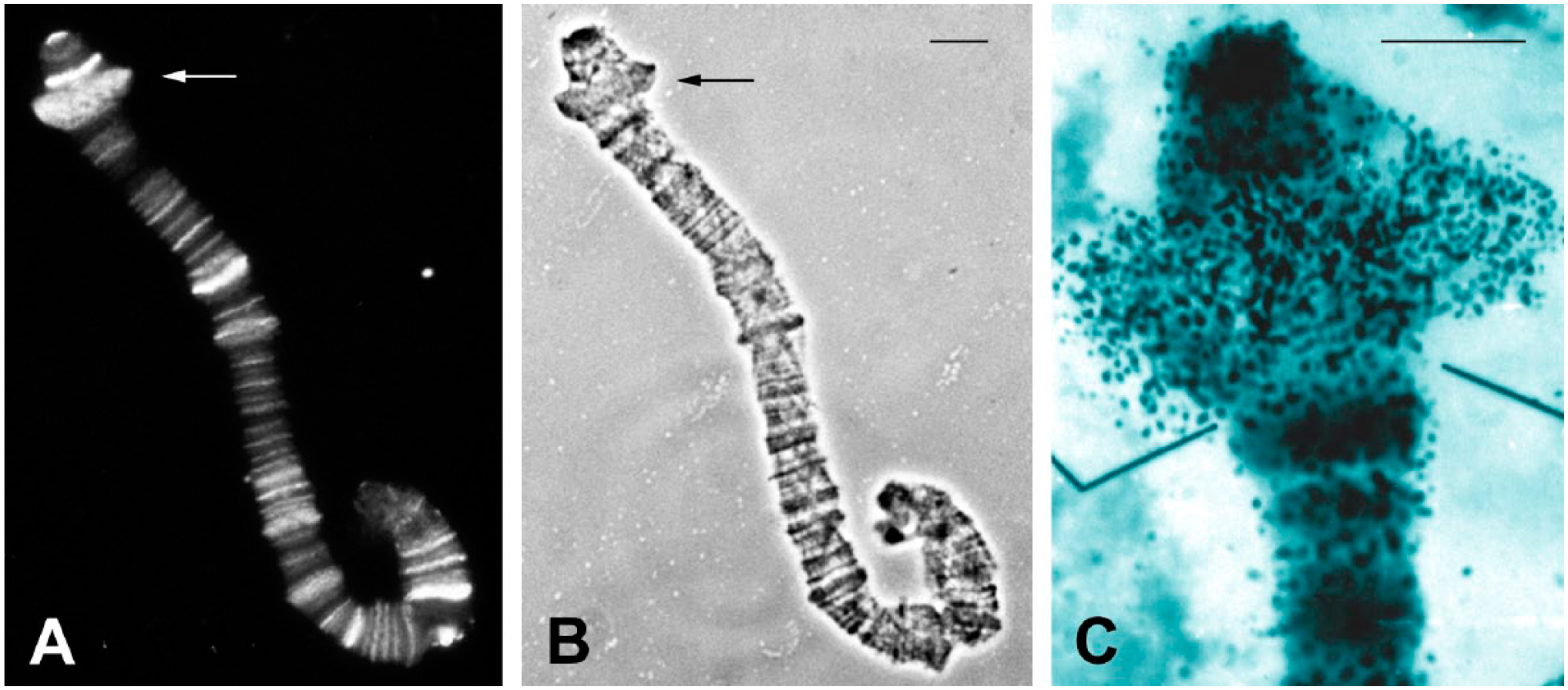
RNA detection into DNA puff B2 of R. americana. (A) Fluorescent detection of endogenous RNA.DNA hybrid in salivary gland chromosome B of R. americana. The arrows point to the DNA puff B2 under immunofluorescense and in its corresponding (B) phase contrast image. (C) Autoradiogram modified from 38 showing RNA synthesis in the form of granular labeling from uridine incorporation into DNA puff B2. Bars at top = 20 μm.
Nanomolar Concentrations of TO Allow Visual Differentiation of Specific Regions of Drosophila and Rhynchosciara Polytene Chromosomes
In distinction with the homogeneously stained chromatin obtained by mM TO solutions, use of 20–60 nM concentrations exhibited small areas of more intense staining using either FITC or TRITC filters. In polytene chromosomes of D. melanogaster, TO invariably bound to a small part of the whole pericentric heterochromatin (Fig. 4). These results are very similar to those obtained previously using immunodetection of triple-stranded DNA (Fig. 5). In R. americana, more intense regions of TO staining were restricted to pericentric sites of the 4 polytene chromosomes (Fig. 6), and these results also agree with those obtained with antibodies to triplex nucleic acids. 31 Because the TO and immunological labeling were first applied individually to separate slides, double labeling for antitriplex and TO was performed to allow alternate detection of putative triplex DNA by either TO or immunofluorescence on the same chromosome spreads. Despite a strong reduction of TO signal that was consistently seen in the FITC channel, a precise overlap of the TO and triplex immunodetection was always observed (Figs. 7 and 8).
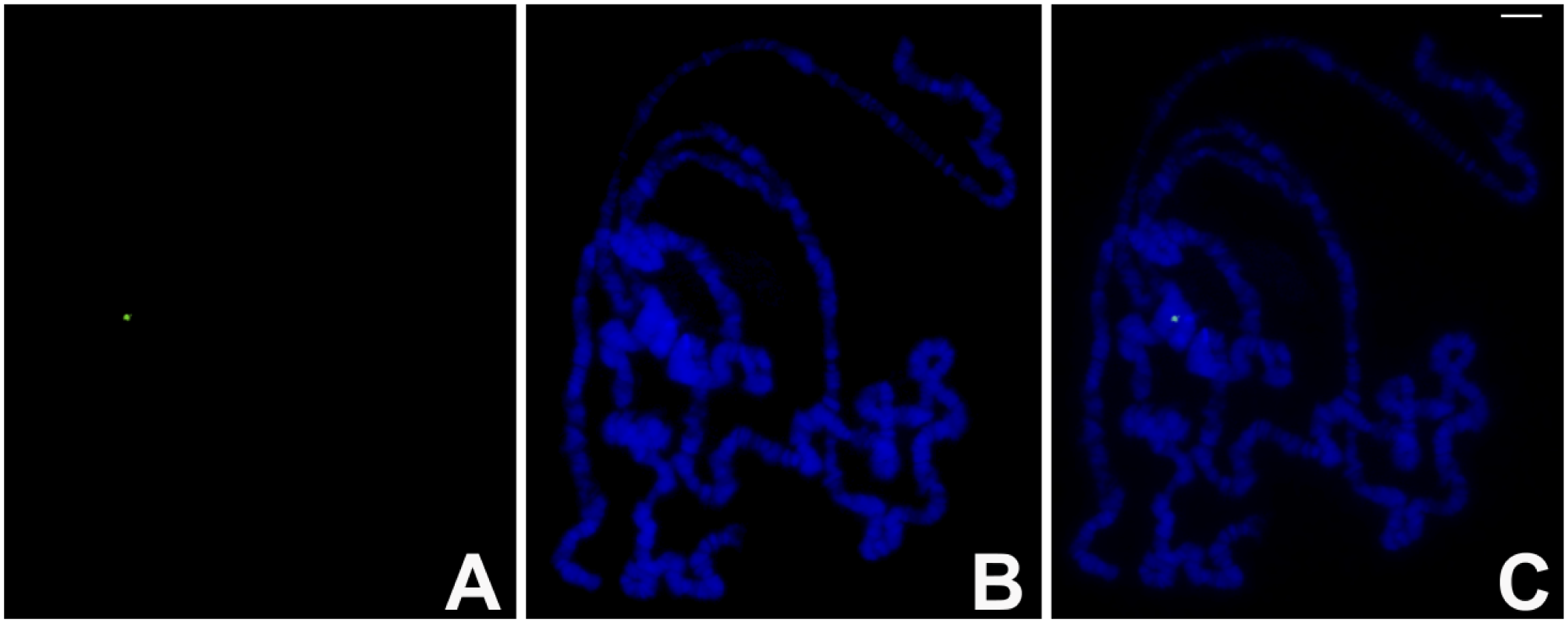
TO and DAPI staining in polytene chromosomes of D. melanogaster. (A) TO staining captured in the FITC channel. (B) The corresponding DAPI staining and (C) the two superimposed images. TO concentration in the nanomolar range was employed in the experiment. Bar = 10 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole; FITC, fluorescein isothiocyanate.
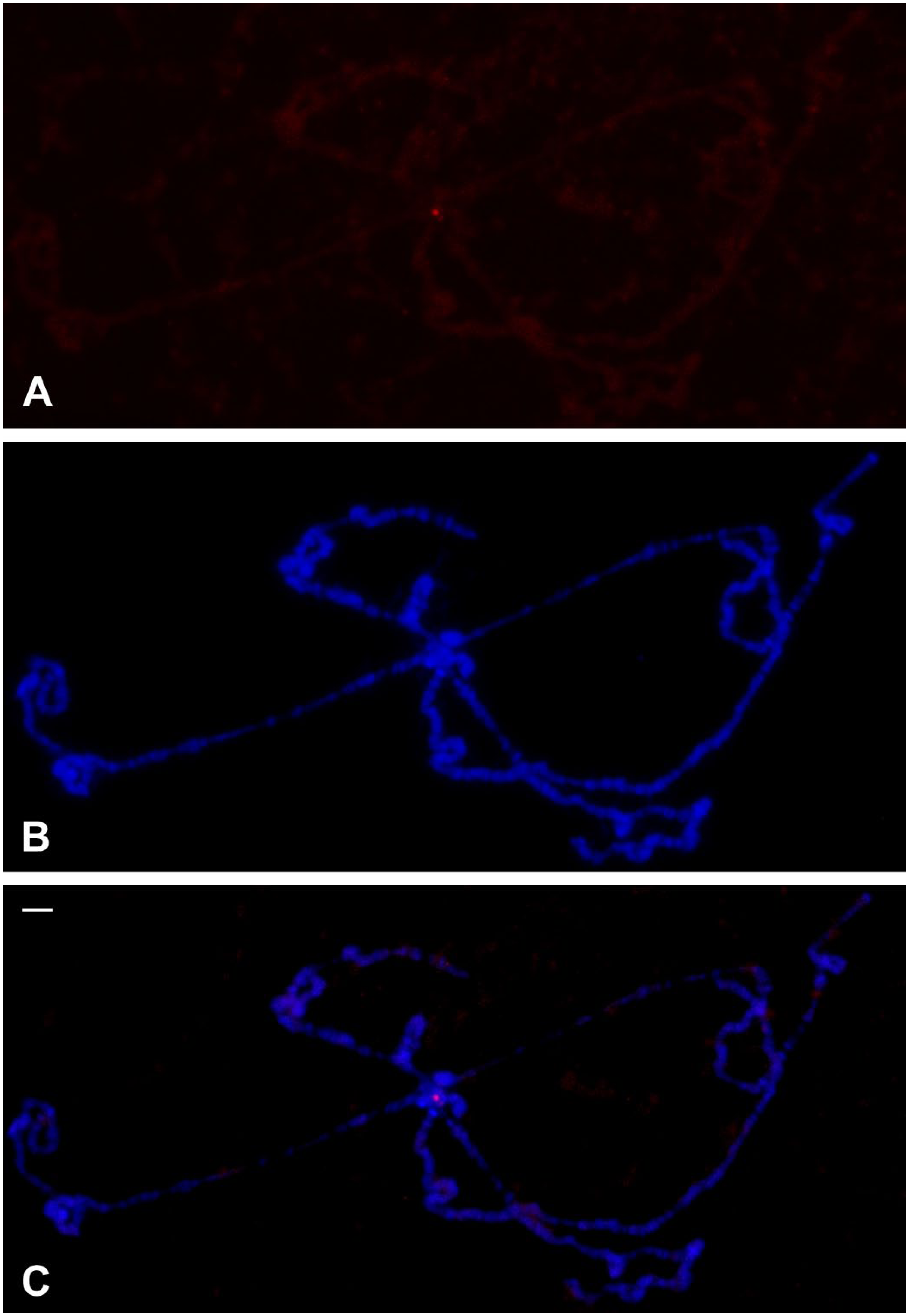
Immunological detection of triplex DNA in polytene chromosomes of D. melanogaster. (A) Localization of anti-poly(dA).poly(rU).poly(rU) antibodies in polytene chromosomes of wild type D. melanogaster, previously treated with RNases. (B) DAPI staining and (C) merged signals. Bar = 10 μm. Abbreviation: DAPI, 4′,6-diamidino-2-phenylindole.
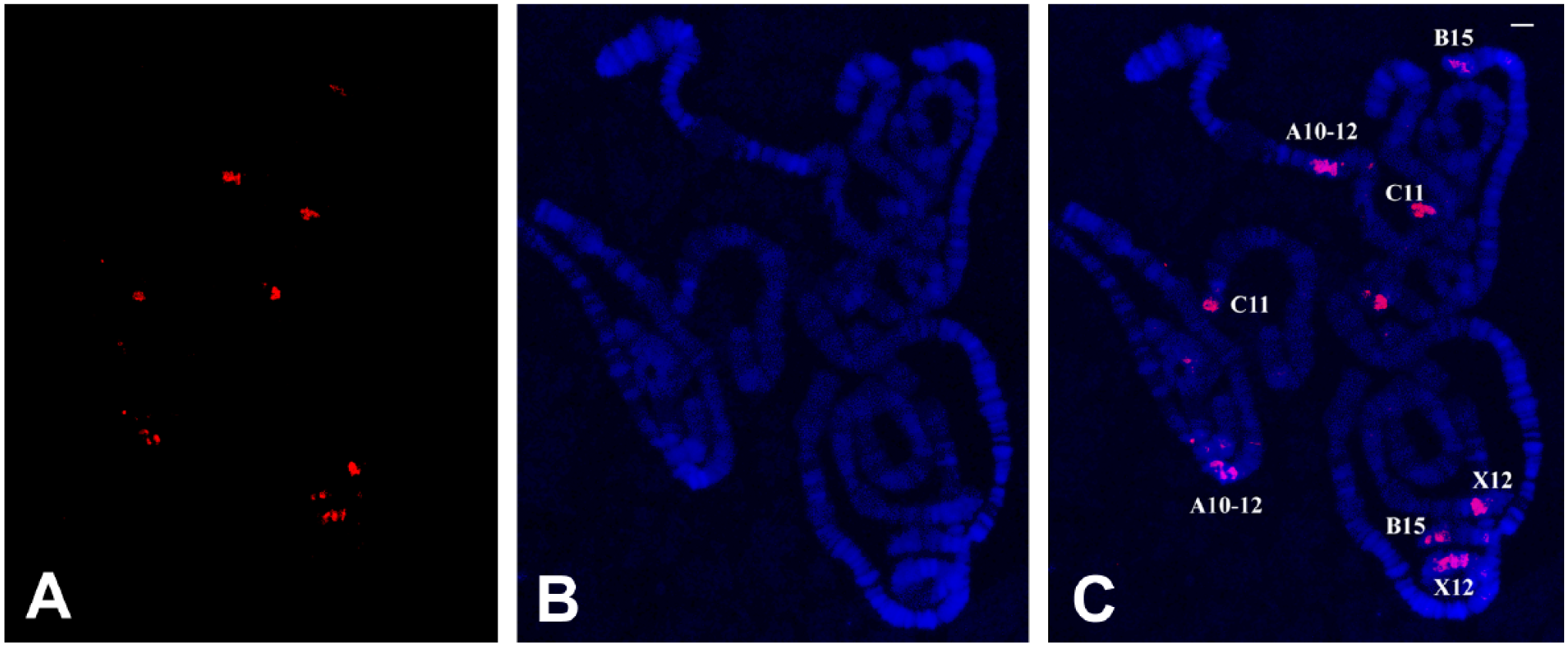
TO and DAPI staining in polytene chromosomes of R. americana. (A) TO signals captured in the TRITC channel. (B) The corresponding DAPI staining and (C) the 2 superimposed images. The identification of chromosome regions was performed previously. 14 TO concentration in the nanomolar range was employed. Bar = 20 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole.
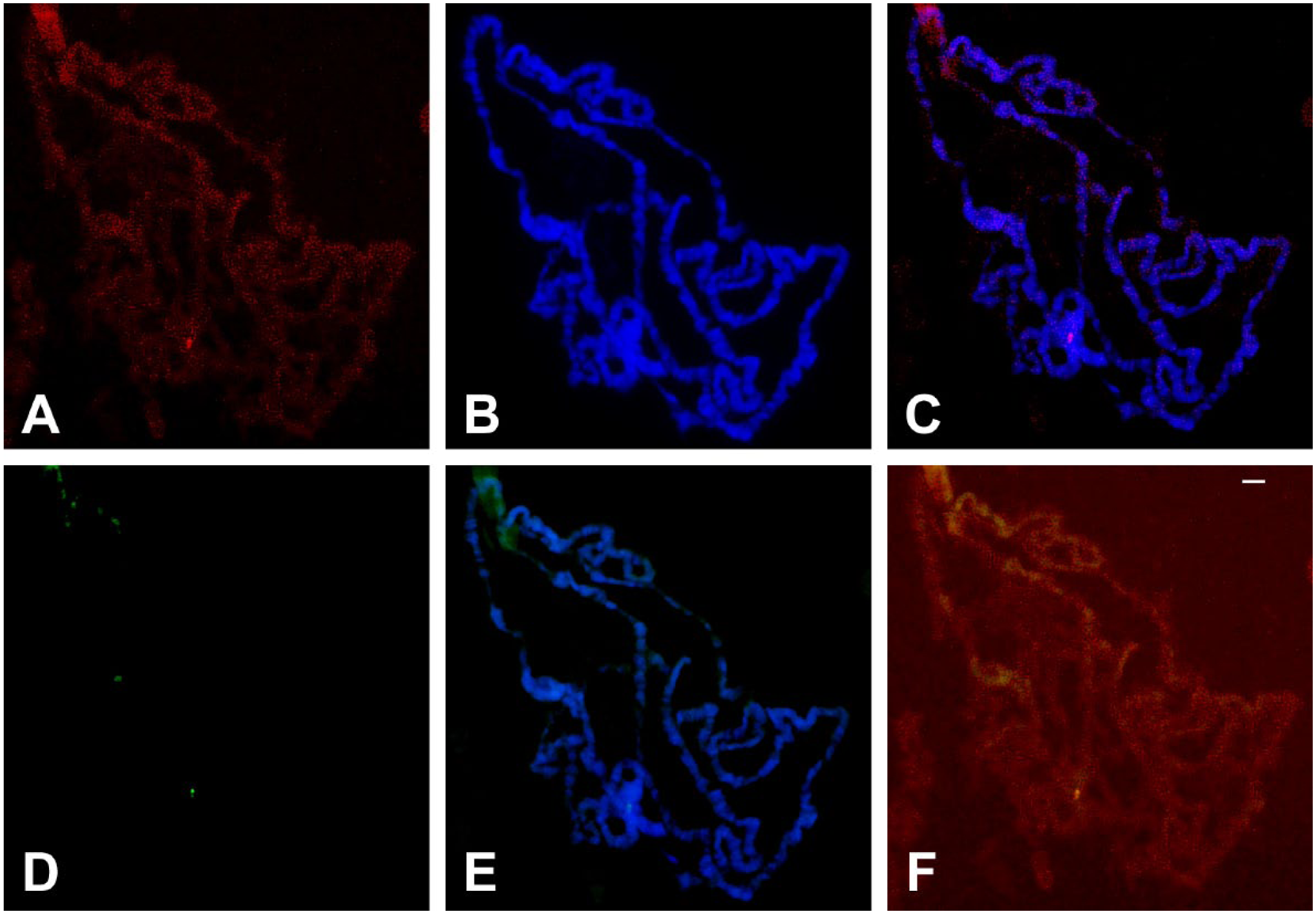
Simultaneous detection of antitriplex and TO staining in polytene chromosomes of D. melanogaster. (A) Localization of anti-poly(dA).poly(rU).poly(rU) antibodies in polytene chromosomes of wild type D. melanogaster. (B) DAPI staining and (C) the merged signals. (D) TO staining captured in the FITC channel. (E) Merged signals of TO and DAPI staining. (F) Signals from TRITC and FITC channels are superimposed. Bar = 10 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole; FITC, fluorescein isothiocyanate; TRITC, tetramethylrhodamine.
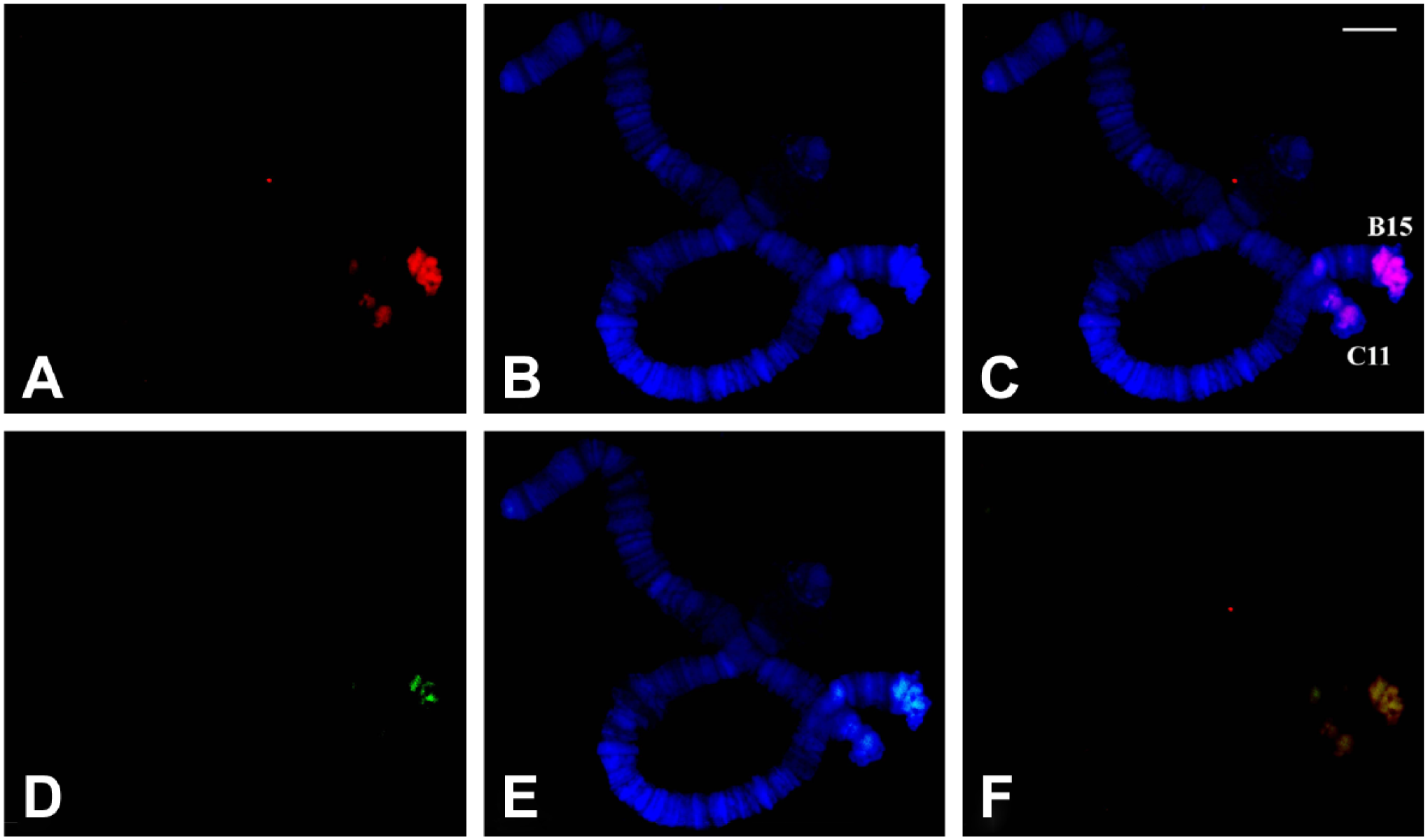
Simultaneous detection of antitriplex and TO staining in polytene chromosomes of R. americana. (A) Localization of anti-poly(dA).poly(rU).poly(rU) antibodies in polytene chromosomes of R. americana. (B) DAPI staining and (C) the merged signals. (D) TO staining captured in the FITC channel. (E) Merged signals of TO and DAPI staining. (F) Fluorescence from TRITC and FITC channels is superimposed. Bar = 20 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole; FITC, fluorescein isothiocyanate; TRITC, tetramethylrhodamine.
TO Staining in Diploid Cells of Drosophila
Compared with the extent of pericentric heterochromatin as defined by Heterochromatin Protein 1 (HP1) in salivary gland nuclei of Drosophila, 39 the TO binding area is very small. The question arises of whether fluorescence from TO can be detected in the heterochromatin of cell types in which nuclear domains are not enlarged. With the aim of assessing the limits of TO detection of triplex DNA, we tested the staining capability of the dye in neuroblast nuclei of Drosophila that are significantly smaller than those from salivary glands. Considering the huge difference in the nuclear volume of the 2 cell types, neuroblast nuclei appear to have a much larger proportion of the heterochromatin labeled by TO than do polytene nuclei of the salivary gland (Fig. 9).
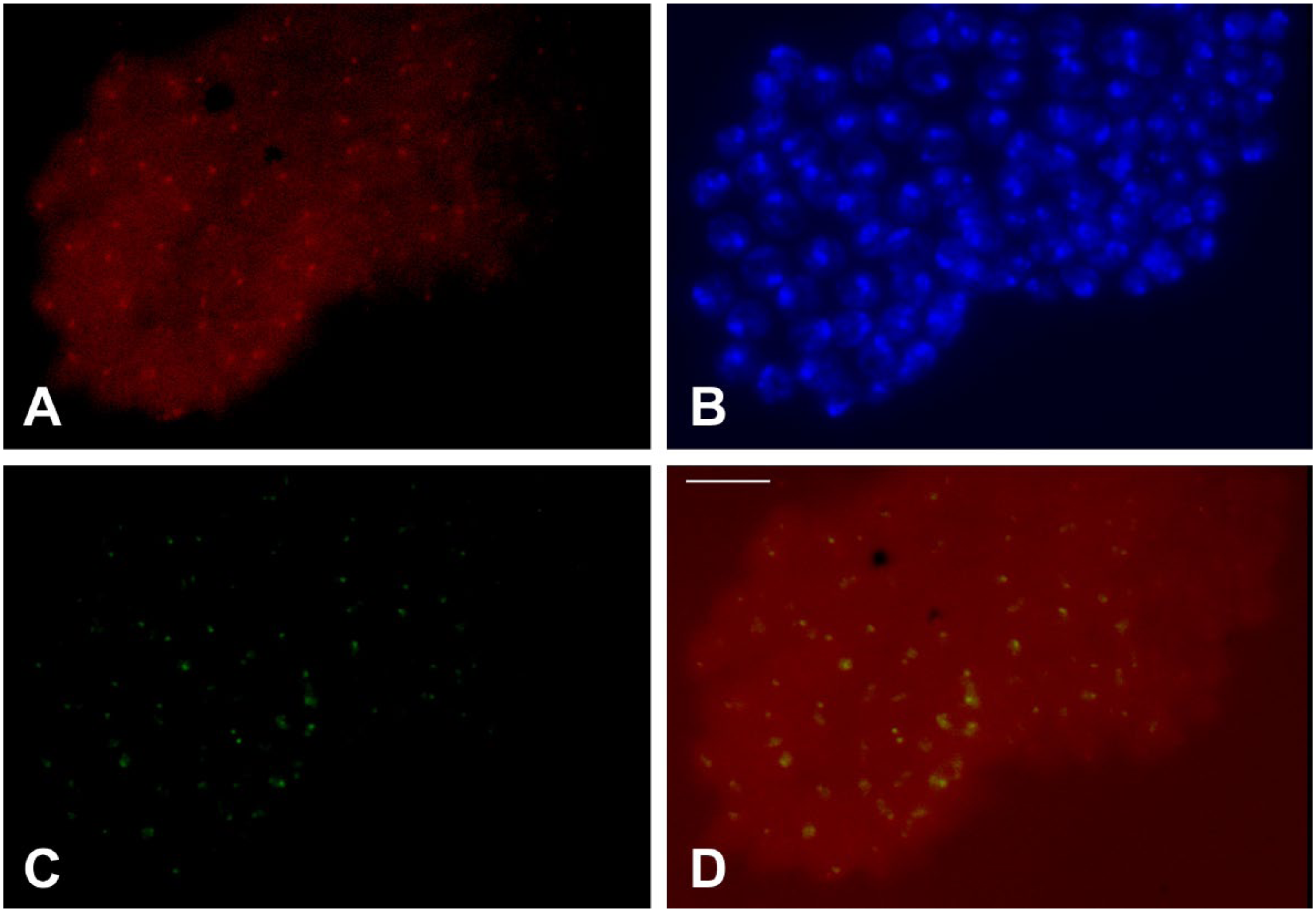
Simultaneous detection of antitriplex and TO staining in neuroblast nuclei of D. melanogaster. (A) Antibody signals captured in the TRITC channel. (B) The corresponding DAPI staining. (C) TO staining captured in the FITC channel. (D) Fluorescence from TRITC and FITC channels is superimposed. Bar = 10 μm. Abbreviations: TO, Thiazole Orange; DAPI, 4′,6-diamidino-2-phenylindole; TRITC, tetramethylrhodamine; FITC, fluorescein isothiocyanate.
Discussion
To our knowledge, this is the first article to validate the potential of a fluorochrome to detect in situ non-canonical DNA structures, such as triple-helices, by comparison with detection by antibodies raised against three-stranded nucleic acid configurations. Although dye concentration and incubation time appear to be the main determinants for detection of triplex DNA by TO in this study, it is likely that excitation light intensity and differential fading during illumination may also play a role in detection, as observed for many other fluorochromes widely used in chromosome studies, such as acridine orange. Furthermore, similar to acridine orange that fails to reveal the difference between double- and single-stranded DNA at high dye concentrations, TO also fails to distinguish between double- and triple-stranded DNA unless low dye concentrations are used.
The differential staining patterns produced by low concentrations of TO in polytene chromosomes were similar to those observed with polyclonal antibodies to triplex DNA described in the literature,31,36 in which labeling is restricted to the pericentric heterochromatin in Drosophila polytene chromosomes. However, monoclonal antibodies to triple-stranded DNA have been shown to react with many Drosophila and human chromosome sites.24,25 The precise antigenic determinants of the polyclonal and monoclonal antibodies have not been determined in detail, but they are probably not the same, and this may explain their different in situ binding behavior. Although the polyclonal antibodies employed in this work were raised to the homopolymeric poly-(dA).poly-(rU).poly-(rU) complex, 36 they can also bind DNA triplexes comprising G/C as components of deoxy-nucleotides. 33 Therefore, the antibodies interact with a specific nucleic acid conformation irrespective of its base composition. Triplex DNA synthesized for the assays with TO in gels contained poly-deoxy-guanine and poly-deoxy-cytosine. 35 Whether the TO preference for triple-stranded DNA is preserved in the case of triplexes composed of poly-(dA) and poly-(dT) remains to be elucidated.
A large number of short poly-(puric).poly-(pyrimidic) tracts is extremely frequent in genomes and have the potential to form triple-helices.40,41 As polyclonal antibodies bind few regions in R. americana and D. melanogaster genomes, 31 this may be due to the existence of constraints imposed by triple-stranded tract length. The same restrictions may operate also in the case of TO staining so that labeled sites in Drosophila and Rhynchosciara probably correspond to sites in which only long triplex tracts are present. The low RNA binding by TO was inferred by low fluorescence levels in nucleoli, polytene chromosome puffs, and cytoplasm on slides that did not undergo RNase treatments. The data obtained in this work argue that both double- and triple-stranded DNA interact with the dye with little, if any, demonstrable RNA binding.
TO and antitriplex antibody labeling in polytene chromosomes of D. melanogaster is not spread over the entire heterochromatic region occupied by HP1, 39 suggesting that sequences were selectively targeted in pericentric heterochromatin. This observation can be extended to binding to neuroblast nuclei, although staining is comparatively more extensive in these cells. As DNA underreplication is not known to occur in the heterochromatin of diploid cells, the data argue that three-stranded DNA structures may be underreplicated in the salivary gland and probably in other polytene tissues of Drosophila. If such a statement is correct, clues on sequence identification of TO and antibody targets might be expected by exploiting probes for triplex nucleic acid conformations together with Drosophila mitotic chromosomes.
Human mitotic spreads prepared for in situ hybridization were also used in this work and did not show brighter fluorescence in localized chromosome regions after TO staining. Given the ability of TO to bind preferentially not only to triplex but also to quadruplex DNA structures, 35 one might expect labeling at telomeres because canonical telomere repeats may form G-quartets 42 detectable by antibodies to quadruplex DNA. 43 Whether the negative results are related to limits inherent in our current observational system or to quadruplex tract length or slide preparation requires further investigation.
In conclusion, this work demonstrates the differential affinity of TO with triple-stranded DNA in chromatin and opens up the possibility of rapid direct detection of triple-stranded DNA in the nuclei of various organisms and tissues. It seems likely that TO will provide uniform results but with much less variability in detecting triplex DNA targets. However, that still remains to be demonstrated.
Footnotes
Acknowledgements
The authors thank Francisco Flausino and Carlos Lopes for maintenance of Drosophila stocks, Dr. Carla Rosenberg for help with figures, and Dr. AngelaVianna Morgante for the gift of human mitotic spreads.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Both authors have contributed to this article as follows: EG performed experiments; PLP drafted the work and wrote the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo), grants 2013/03840-0 and 2017/02588-6.
