Abstract
The intranuclear distribution of HMGI/Y proteins was analyzed by immunofluorescent staining in several cell lines using a polyclonal antibody that stained a fibrogranular network. In actively growing 3T3 fibroblasts, HMGI/Y proteins were mainly localized to heterochromatin masses, whereas in quiescent cells they were more diffusely distributed. Double labeling experiments showed a co-localization of HMGI/Y with DNA topoisomerase IIα. These results are in agreement with previously published biochemical data and indicate a possible involvement of HMGI/Y proteins in several nuclear functions, including chromatin organization and gene expression.
H
Moreover, it has been shown that transforming viruses are no longer able to transform rat thyroid cells if the cells also transcribe an anti-sense message for HMGI-C (Berlingieri et al. 1995). HMGI/Y proteins bind to scaffold-associated regions (SARs), i.e., to highly A-T-rich DNA sequences that are involved in attaching chromatin loops to a nucleoskeletal structure and are also associated with active genes (Zhao et al. 1993). Because no information is available concerning the distribution of HMGI/Y during interphase, we have performed an immunocytochemical analysis of the spatial distribution in the nucleus of HMGI/Y by means of a specific polyclonal antibody (Giancotti et al. 1996). To this end, cells growing on coverslips were fixed with 4% paraformaldehyde, permeabilized with 0.5% Triton X-100, and processed as reported by Neri et al. (1997). In rat thyroid PC E1A + v-raf cells (which produce high levels of these polypeptides), the antibody stained several large dots with a finer, punctate immunoreactivity dispersed in between (Figure 1a). In contrast, in rat PC Cl 3 cells (which do not express these proteins; Berlingieri et al. 1995) no immunoreactivity was detected (Figure 1b). These results showed specificity of the antibody for immunocytochemical analysis. Using confocal laser scanning microscope (CLSM), it was possible to distinguish in HeLa cells a nuclear fibrogranular network stained by the antibody, with some faintly positive granules in the nucleoli (Figure 1c), consistent with the results of Yang-Yen and Rothblum (1988) showing involvement of HMGI/Y in rDNA synthesis. Because HMGI/Y proteins are preferentially localized to G/Q- and C-bands of chromosomes (Disney et al. 1989), we analyzed the distribution of HMGI/Y relative to DNA in interphase nuclei. As shown in Figure 1d, in quiescent Swiss 3T3 cells the antibody to HMGI/Y gave a fine punctate pattern. In contrast, in actively growing cells the immunofluorescent pattern was characterized by large dots and and interspersed punctate staining (Figure 1f). We stress that DAPI staining for DNA revealed no differences between the two types of cells, because the large heterochromatic regions were detected both in quiescent and in serum-stimulated cultures (compare Figure 1e with Figure 1g). In these regions, centromeric heterochromatin is replicated during the late S-phase (Nakayasu and Berezney 1989). Because Saitoh and Laemmli (1994) showed that HMGI/Y and topoisomerase IIα co-localize along the scaffold of mitotic chromosomes, we analyzed by double immunostaining and CLSM whether this type of association was present in the interphase nucleus of HeLa cells. As shown in Figure 1j, almost all of the immunofluorescent signal due to HMGI/Y (Figure 1h, red) co-localized with that due to topoisomerase IIα (Figure 1i, green). Topoisomerase IIα was immunostained by monoclonal antibody Ki-S1 (Boehringer-Mannheim; Mannheim, Germany).
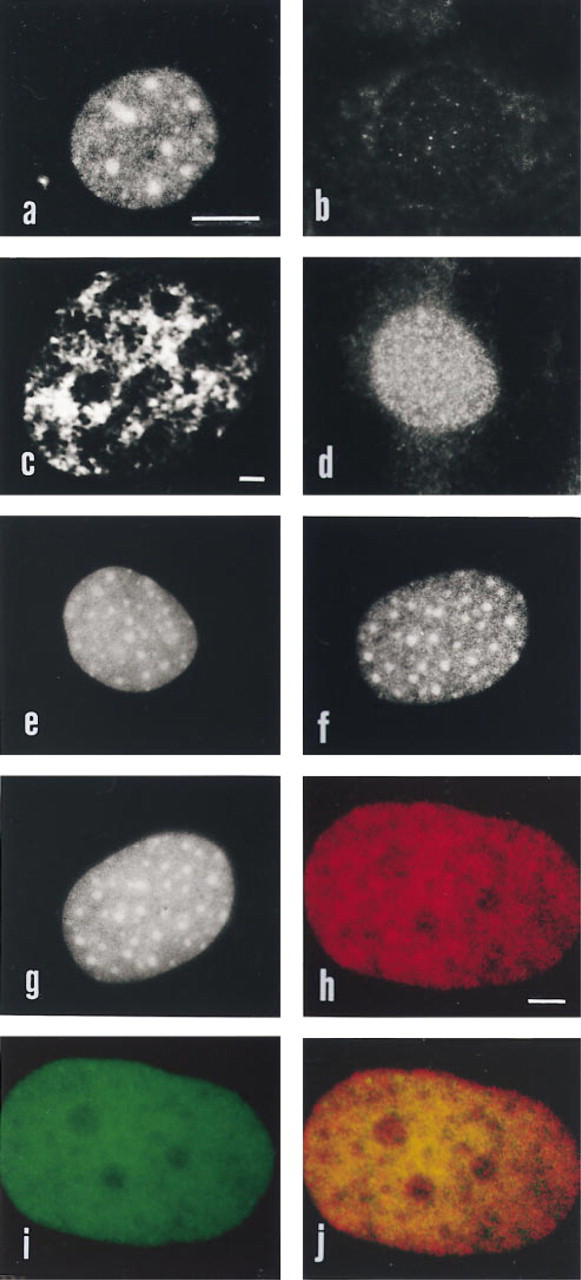
See text for details. Bars: a,b,d-g = 5 μm; c = 1 μm; h-j = 1 μm.
In conclusion, we believe that our data are in agreement with previous findings (obtained by biochemical and molecular biology techniques) indicating an involvement of HMGI/Y in several key nuclear functions (Bustin and Reeves 1996). They also show that some of the architectural features found in mitotic chromosomes are maintained in the interphase nucleus. Future investigations with the antibody used for the present study may provide additional information on the roles played by HMGI/Y proteins.
Footnotes
Acknowledgements
Supported by Associazione Italiana per la Ricerca sul Cancro (A.I.R.C.) 1996 grants to AMM and VG, and from Italian MURST 40% and 60% grants to Università di Trieste.
