Abstract
The membrane receptor megalin is crucial for normal fetal development. Besides its expression in the developing fetus, megalin is also expressed in the human placenta. Similar to its established function in the kidney proximal tubules, placental megalin has been proposed to mediate uptake of vital nutrients. However, details of megalin expression, subcellular localization, and function in the human placenta remain to be established. By immunohistochemical analyses of first trimester and term human placenta, we showed that megalin is predominantly expressed in cytotrophoblasts, the highly proliferative cells in placenta. Only limited amounts of megalin could be detected in syncytiotrophoblasts and least in term placenta syncytiotrophoblasts. Immunocytochemical analyses furthermore showed that placental megalin associates with structures of the endolysosomal apparatus. Combined, our results clearly place placental megalin in the context of endocytosis and trafficking of ligands. However, due to the limited expression of megalin in syncytiotrophoblasts, especially in term placenta, it appears that the main role for placental megalin is not to mediate uptake of nutrients from the maternal bloodstream, as previously proposed. In contrast, our results point toward novel and complex functions for megalin in the cytotrophoblasts. Thus, we propose that the perception of placental megalin localization and function should be revised.
Keywords
Introduction
The human placenta ensures fetal growth via its continuous supply of vital nutrients (e.g., carbohydrates, lipids, amino acids, vitamins, minerals) and a panel of signaling molecules from the maternal bloodstream to the fetus. Also, the placenta serves as a protective barrier and removes fetal waste products. Maternal–fetal exchange of nutrients, signaling molecules, and waste products occurs across the membranes of the placental chorionic villi which mainly consist of cytotrophoblasts and syncytiotrophoblasts.1–4
Cytotrophoblasts are mononuclear cells that can either undergo mitotic cell division or differentiate into non-proliferative syncytiotrophoblasts. Syncytiotro-phoblasts fuse to form a layer of multinuclear cells called the syncytium, which lines the chorionic villi.1–4 During the second trimester and throughout the pregnancy, the cytotrophoblast layer below the syncytium gradually gets thinner and discontinuous.1–4
The syncytiotrophoblasts have no transcriptional activity, and the syncytial layer is maintained by continuous proliferation, differentiation, and fusion of differentiating cytotrophoblasts. 5 Accordingly, placental growth is strictly dependent on cytotrophoblast proliferation and differentiation, especially during the first trimester when the placenta grows rapidly. Importantly, a number of growth factors have been shown to have a dramatic influence on placental and fetal development and growth. For example, clinical studies have shown that levels of insulin-like growth factors (IGFs) in the maternal circulation correlate with fetal growth6,7 and, furthermore, that birth weight is positively correlated with cord blood levels of IGF-1.8,9 Likewise, epidermal growth factor (EGF) has been suggested to influence villous trophoblast function and placental development. 5 In addition, transforming growth factor beta 1 has been shown to play a major role in controlling trophoblast proliferation, differentiation, and invasion.10,11
Cytotrophoblasts and syncytiotrophoblasts are clearly different, fundamentally because the cytotrophoblasts are proliferative, whereas the syncytiotrophoblasts are not. But they also share characteristics. Both cell types, like most cell types, require advanced vesicular transport machineries such as the endolysosomal apparatus to conduct their many and diverse functions.12,13 A shared feature of vesicular transport machineries is the participation of transmembrane receptor proteins and their cytosolic adaptor protein partners.5,12,13
Megalin, also known as low-density lipoprotein receptor–related protein 2 (LRP2), is a transmembrane receptor with ability to mediate uptake and trafficking of various ligands. Megalin has predominantly been studied in the kidney proximal tubular epithelium.14–16 Here, it localizes to the apical membrane as well as the endolysosomal apparatus of proximal tubular cells and mediates reabsorption of many different ligands from the glomerular ultrafiltrate.14–16 Megalin has furthermore been shown to be expressed in both cytotrophoblasts and syncytiotrophoblasts of human placenta and, here, suggested to function as a nutrient receptor and mediate uptake of nutrients from the maternal bloodstream (similar to its functions in the kidney).17–21 However, the function of placental megalin has not been systematically investigated in suitable models, and the suggestions regarding the role of megalin in human placenta therefore remain purely speculative.
The megalin protein contains an enormous extracellular ligand–binding region, and the cytoplasmic tail of megalin holds multiple recognition sites for cytosolic adaptor proteins known to mediate intracellular trafficking of vesicles in the endolysosomal system.15,16,22 Furthermore, megalin is also able to colocalize with established markers of endosomes (e.g., EEA1 and Rab5).15,16,22,23
The previous studies reporting megalin expression in first and third trimester cytotrophoblasts and syncytiotrophoblasts are not unanimous and reflect various and inconsistent observations and conclusions. A thorough investigation and comparison of the expression and subcellular localization of megalin in first and third trimester placenta is required to further elucidate megalin function in the human placenta.
In the present study, we have conducted detailed histological and cytological investigations of the expression and subcellular localization of megalin in first trimester and term placenta as well as a biochemical characterization of placental megalin.
Materials and Methods
Tissues
Two first trimester placentas of gestational age 9.5 weeks (exactly 9 weeks + 4 days) were obtained from induced abortions. Three term placentas were obtained from uncomplicated pregnancies, delivery at full term (weeks 38–41). All material was collected with informed patient consent and was approved beforehand by the Danish National Committee on Health Research Ethics (Approval No. KF (01) 258206 and 20060063, respectively). Tissue for immunohistochemical analyses was fixed in 0.1-M cacodylate buffer including 4% formaldehyde (pH 7.4) by immersion for 2 hr followed by further processing. Tissue for subsequent membrane preparations was snap-frozen in liquid nitrogen and stored at −80C.
Antibodies
The polyclonal rabbit anti-human megalin antibody used in this study is well characterized24–26 and was a kind gift from Dr. Soren K. Moestrup, Aarhus University, Denmark. Protein G–purified rabbit anti-human megalin antibody was used (immunofluorescence: 10 µg/ml; IHC: 6.60–8.25 µg/ml; immunocytochemistry: 1.65 µg/ml). Mouse monoclonal anti-human EEA1 antibody (ab15846; Abcam, Cambridge, UK; immunofluorescence: 1:50) was used for colocalization studies. For immunohistochemical analyses, visualization was performed using horse radish peroxidase (HRP)–conjugated secondary goat anti-rabbit antibody (1:200; P044801-2; Dako, Glostrup, Denmark) for light microscopy or 10-nm gold-conjugated secondary goat anti-rabbit antibody (1:50; EM.GAR10; BioCell, Cardiff, UK) for electron microscopy. Secondary antibodies (Life Technologies, Naerum, Denmark) were used for immunofluorescence analyses (Invitrogen Alexa Fluor 488 goat anti-rabbit [A11008] and Alexa Fluor 546 goat anti-mouse [A11030]; 1:200). Visualization of megalin in immunoblotting was performed using protein G–purified rabbit anti-human megalin antibody together with secondary goat anti-rabbit IgG (alkaline phosphatase–conjugated; 1:8000; Sigma-Aldrich, Brøndby, Denmark).
IHC and Immunocytochemistry
Placental tissue for light microscopic investigations was dehydrated in graded alcohols and embedded in paraffin. Paraffin sections of 2 µm were cut on a Leica RM 2165 microtome (Leica, Ballerup, Denmark) and processed as previously described. 27 Briefly, sections were heated to 60C for 1 hr and placed in xylene overnight, before rehydration in graded alcohols and blocking for endogenous peroxidase activity. Rehydrated sections were heated in Tris-EGTA buffer to 100C for antigen retrieval in a microwave for approximately 10 min, cooled and permeabilized with 0.05% saponin (1% BSA, 0.2% gelatin, and 0.05% saponin in 0.01-M PBS), and blocked for endogenous peroxidase activity before incubation with primary antibodies. Sections were incubated with a primary antibody in 0.01-M PBS, 0.1% BSA, and 0.02-M NaN3, followed by incubation with HRP-conjugated secondary antibody. Peroxidase labeling was visualized by incubation with diaminobenzidine and 0.03% H2O2 for 10 min. Sections were counterstained with Mayer’s hematoxylin stain and examined in a Leica DMR (Leica, Wetzlar, Germany) microscope equipped with a Leica DFC320 camera (Leica, Wetzlar, Germany) and processed using Adobe Photoshop 8.0 software.
Placental tissue for electron microscopic investigation was transferred to 12% gelatin, infiltrated for 2 hr with PBS buffer including 2.3-M sucrose (pH 7.4), and ultimately frozen in liquid nitrogen. Cryosections of approximately 60 nm were sliced using a Leica EM FC6 cryo-ultramicrotome (Leica, Ballerup, Denmark). For immunolabeling, cryosections were processed as previously described. 28 Briefly, cryosections were incubated overnight, at 4C, with primary antibody in buffer containing 2% human serum followed by incubation, at 4C, for 2 hr, with 10-nm gold-conjugated secondary antibody. Cryosections were then embedded in methylcellulose containing 0.4% uranyl acetate and examined using a JEOL JEM-1400+ transmission electron microscope (JEOL, Freising, Germany).
Cell Culture
The human choriocarcinoma cell lines BeWo and JEG-3 were purchased at ATCC (#ATCC CCL-98 and #ATCC HTB-36). BeWo and JEG-3 cells were cultured in F-12K Medium (Kaighn’s Modification of Ham’s F-12 Medium; ATCC 30-2004) and Eagle’s Minimum Essential Medium (ATCC 30-2003) (LGC Standards, Wesel, Germany), respectively, supplemented with 10% heat-inactivated FBS from PAA Laboratories (Fisher Scientific, Roskilde, Denmark), 100 U/ml penicillin, and 100 µg/ml streptomycin (Life Technologies). The human choriocarcinoma cell line JAR was a kind gift from Associate Professor Trine Fink (Laboratory for Stem Cell Research, Aalborg University, Denmark). JAR cells were cultured in RPMI-1640 medium (Life Technologies) supplemented with 10% heat-inactivated FBS, 100 U/ml penicillin and 100 µg/ml streptomycin, and 2-mM
Immunofluorescence Staining of JAR Cells and Confocal Microscopy
Membrane proteins in living, non-permeabilized JAR cells were labeled at 4C in the absence of detergents, and fixed, permeabilized JAR cells were stained at room temperature in the presence of Triton X-100, both essentially as described by Pedersen et al. 29 Nuclei were visualized using Hoechst 33342 (1 µg/ml; Sigma-Aldrich, Copenhagen, Denmark). Images were captured using a Zeiss LSM-780 confocal microscope system (Carl Zeiss MicroImaging GmbH, Jena, Germany) using a 63×/1.20 water objective.
RNA Extraction
Total RNA from BeWo, JEG-3, and JAR cells was extracted and purified using RNeasy Mini Kit (Qiagen, Copenhagen, Denmark), according to the manufacturer’s protocol. Genomic DNA was removed by an additional on-column DNase digestion step.
cDNA Synthesis and qRT-PCR
Total RNA was reverse transcribed using Applied Biosystems High Capacity RNA-to-cDNA Kit (Life Technologies) according to manufacturer’s protocol. Quantitative real-time PCR (qRT-PCR) reactions were performed using Applied Biosystems TaqMan Fast Advanced Master Mix (Life Technologies) and the following TaqMan assays: LRP2 Hs00189742_m1 and ACTB Hs01060665_g1. Reactions were performed using a standard fast TaqMan program of the Applied Biosystems 7500 Fast Real-Time PCR system (Life Technologies). Each sample was run in triplicates for each TaqMan assay. Relative expression levels were calculated using 7500 Software (version 2.0.6; Applied Biosystems) and Microsoft Office Excel 2007. ΔCt values were calculated as Ct (target) minus Ct (normalizing gene: ACTB); ΔΔCt values were calculated as ΔCt (target) minus ΔCt (calibrator). Relative quantification values were calculated as 2−(ΔΔCt). All qRT-PCR experiments and calculations were performed as far as possible in accordance with the MIQE guidelines. 30
Preparation of Solubilized Term Placenta Membrane Proteins
Solubilized placental membrane proteins were prepared essentially as described previously.31,32
Ligand Affinity Chromatographic Purification and MS Identification of Placental Megalin
Megalin was purified from solubilized human placental or kidney cortex membranes essentially as described by Moestrup et al. 32 using sepharose beads coated with receptor-associated protein (RAP; a ligand for megalin 33 ). Eluted fractions were analyzed on non-reducing 4% to 12% Invitrogen Novex Bis-Tris Mini Gels (Life Technologies) and subsequent silver staining. A presumed megalin-containing ~500- to 600-kDa protein band was excised from the gel (one of the top fractions) and subjected to in-gel tryptic digestion before identification by liquid chromatography–tandem mass spectrometry (MS) with a Q-Tof Premier mass spectrometer (Waters, Milford, MA) essentially as previously described. 34 Proteins were identified in the SwissProt protein database (Homo sapiens) using the online version of the Mascot MS/MS Ions Search facility (Matrix Science, Ltd., London, UK). 35 The parameters are as follows: doubly and triply charged ions with up to two missed cleavages, peptide tolerance of 20 ppm, one variable modification, Carbamidomethyl-C, and an MS/MS tolerance of 0.05. Contaminating peptides such as keratins and trypsin were disregarded. Individual ion scores above 24 indicated identity or extensive homology giving a less than 5% probability that the observed match was a random event. Thus, if the score of a given peptide in the search is greater than 24, it means that the probability that the match was a random event is below 5%. At least one “bold red” significant peptide was required in the search.
Immunoblotting
Total homogenate of human term placenta and human kidney cortex, proteins purified by ligand (RAP) affinity chromatography from human placenta or human kidney, and cell lysates were analyzed using non-reducing 4% to 12% Invitrogen Novex Bis-Tris Mini Gels (Life Technologies) and transferred to polyvinylidene difluoride (PVDF) membranes (Bio-Rad, Copenhagen, Denmark). Blots were developed using the NBT/BCIP developing system (Roche) or ECL Western Blotting Substrate (from Pierce, Thermo Fisher Scientific, Naerum, Denmark), according to manufacturer’s protocol. Images were captured using a Fujifilm LAS-3000 and Image Reader LAS-3000 software, version 2.2 (both GE Healthcare, Brøndby, Denmark).
Results
Immunohistochemical and Immunocytochemical Detection of Megalin in Human Placenta
Immunohistochemical detection of megalin in human placenta showed an intense cytoplasmic labeling of cytotrophoblast cells in human first trimester (9.5 weeks; Fig. 1A and C) and term placenta (Fig. 1B and D). In accordance with previous studies of megalin in the human placenta,17,20 we detected an apical expression of megalin in the syncytiotrophoblast layer in first trimester placenta. However, in contrast to previous studies, we were not able to detect a distinct labeling for megalin in the syncytiotrophoblast layer of term placenta (Fig. 1B and D; see Table 1 for a summary of megalin expression and localization).
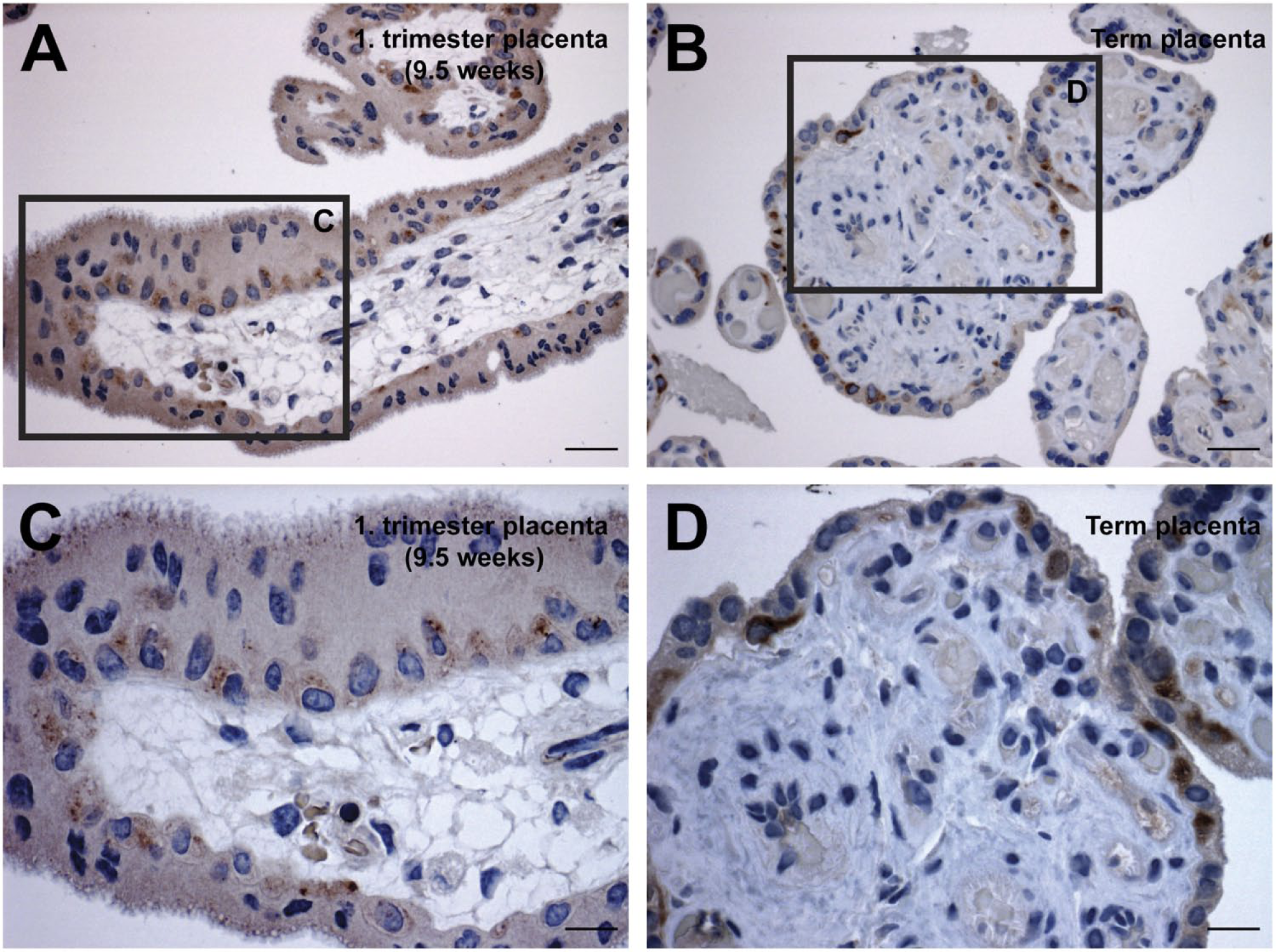
Immunohistochemical detection of megalin in human first trimester and term placenta. Immunoperoxidase staining for megalin in first trimester (A and C) showed that megalin is predominantly expressed in cytotrophoblasts but can also be identified apically in syncytiotrophoblasts. In human term placenta (B and D), megalin was only observed in cytotrophoblast cells. Scale bars: A, C = 20 µm; B, D = 10 µm.
Overview of Megalin Expression and Localization in Human Placenta.

Table summarizing the expression pattern of megalin in human first trimester and term placenta. xxx indicates strong expression and (x) very weak/non-detectable expression.
To determine the subcellular localization of megalin in cytotrophoblast cells and in the syncytiotrophoblast layer, we performed immunocytochemical detection of megalin on ultrathin cryosections from first trimester and term placenta tissue. In first trimester placenta cytotrophoblasts, megalin was predominantly observed in vesicular structures in the juxtanuclear region (Figs. 2 and 3A) near the Golgi apparatus. We furthermore observed megalin on the plasma membrane facing the basement membrane, neighboring cytotrophoblasts, and neighboring syncytiotrophoblasts as well as in endocytic invaginations, vesicular structures near the plasma membrane, and on the outer limiting membrane and luminal vesicles of multivesicular bodies (MVBs; Figs. 2 and 3A–C). Immunocytochemical detection of megalin in term placenta showed a similar subcellular localization in cytotrophoblasts (Fig. 4). However, in contrast to the first trimester cytotrophoblast cells, megalin was predominantly observed on and near the plasma membrane in term placenta cytotrophoblast cells (Fig. 4).
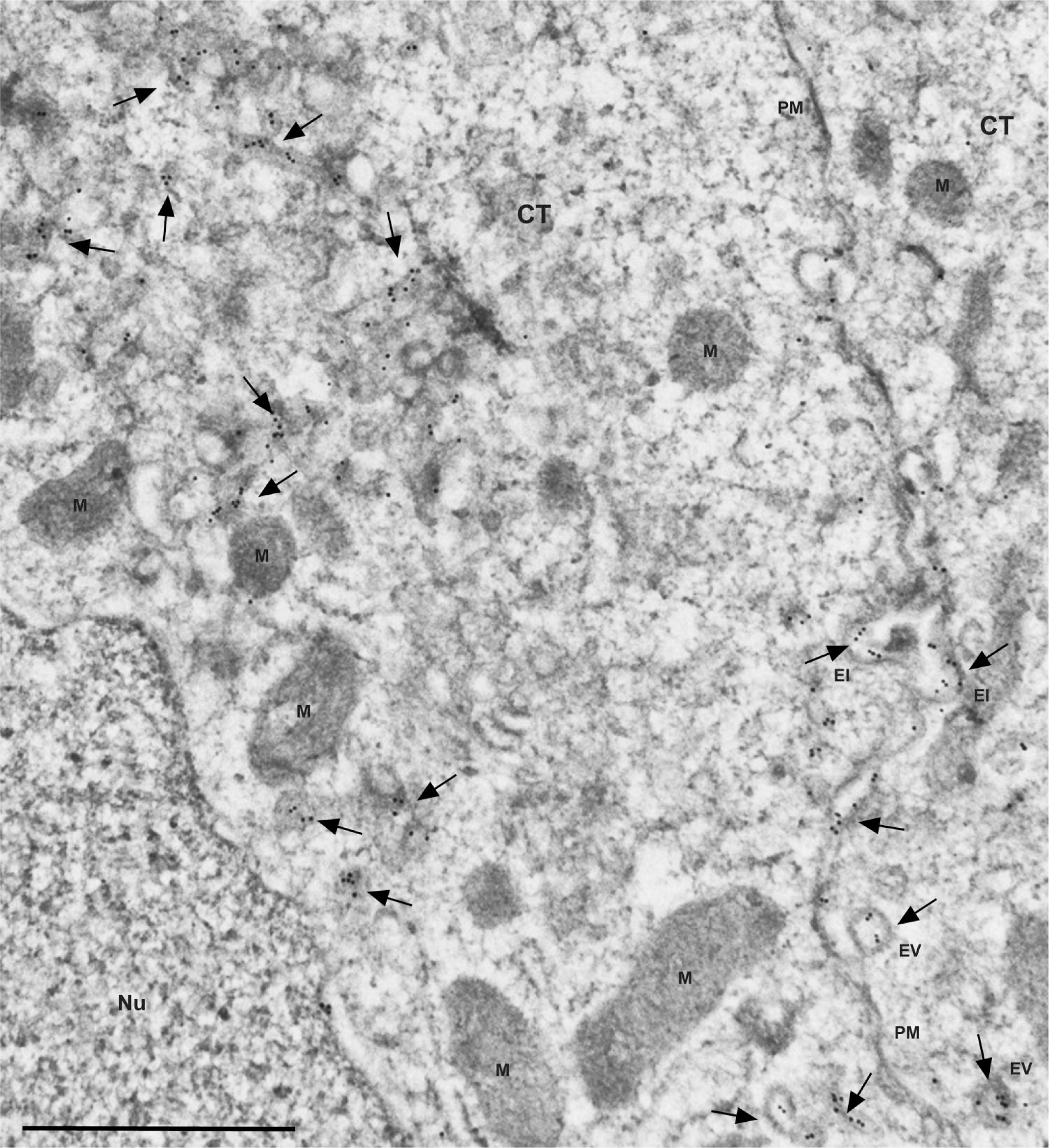
Immunocytochemical detection of megalin in ultrathin cryosections from first trimester human placenta. Clusters of gold particles (arrows) show a massive vesicular juxtanuclear expression of megalin in first trimester cytotrophoblasts. Megalin was furthermore detected on the plasma membrane facing the neighboring cytotrophoblasts as well as in endocytic invaginations and vesicles near the plasma membrane. Scale bar = 1 µm. Abbreviations: PM, plasma membrane; CT, cytotrophoblast; M, mitochondrion; EI, endocytic invagination; EV, endocytic vesicle; Nu, nucleus.
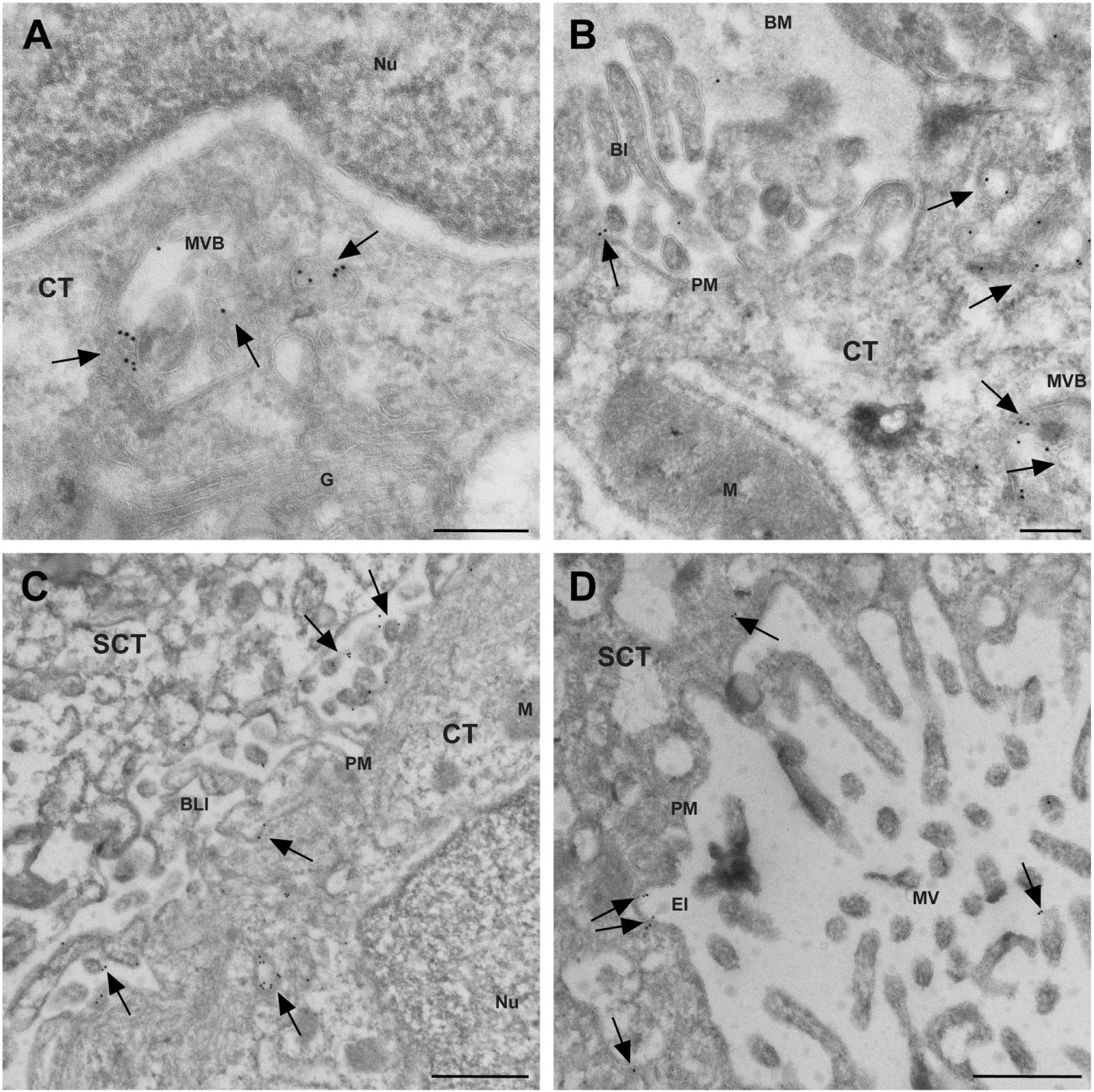
Immunocytochemical detection of megalin in ultrathin cryosections from first trimester human placenta. Gold particles (arrows) show that megalin localizes to vesicular structures near the nucleus and Golgi apparatus as well on the intraluminal vesicles and outer limiting membrane of MVBs (A). Megalin (arrows) was also observed on the basolateral plasma membrane facing the basement membrane and neighboring syncytiotrophoblast cells as well as in MVBs and smaller vesicular structures near the basolateral plasma membrane (B and C). In syncytiotrophoblast cells, megalin (arrows) was predominantly observed on the apical plasma membrane and in vesicular structures near the apical plasma membrane (D). Scale bars: A, B = 200 nm; C, D = 500 nm. Abbreviations: Nu, nucleus; MVB, multivesicular bodies; CT, cytotrophoblast; G, Golgi apparatus; BM, basement membrane; PM, plasma membrane; M, mitochondrion; SCT, syncytiotrophoblast; BLI, basolateral interdigitations; Nu, nucleus; EI, endocytic invagination; MV, micro villi; BI, basal infolding.
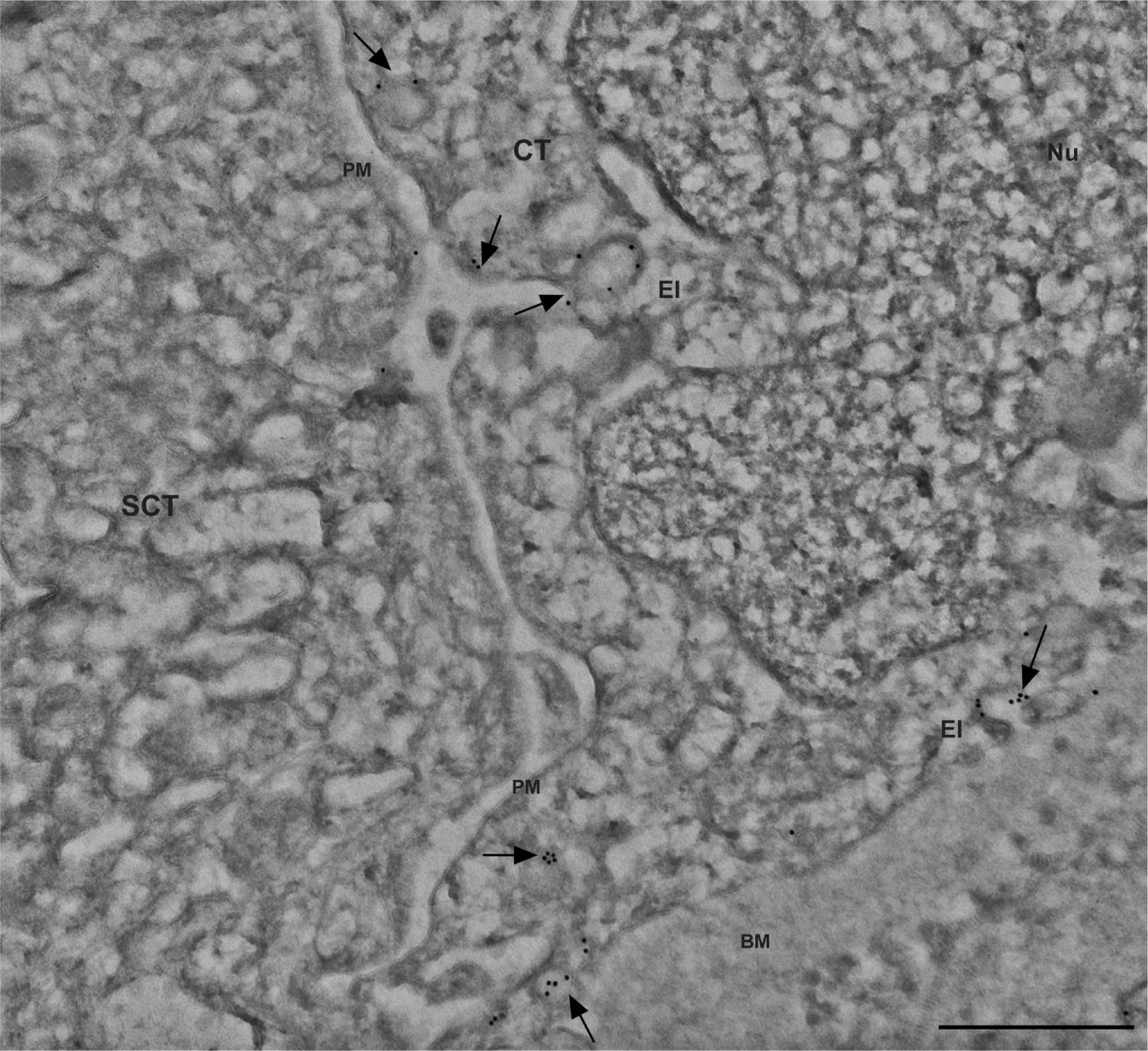
Immunocytochemical localization of megalin in cytotrophoblasts of human term placenta. Gold particles (arrows) show that megalin localizes near or on the plasma membrane of cytotrophoblast cells of human term placenta. More specifically, megalin was observed in endocytic invaginations and vesicular structures in close proximity to the plasma membrane. Scale bar = 500 nm. Abbreviations: CT, cytotrophoblast; Nu, nucleus; PM, plasma membrane; EI, endocytic invagination; SCT, syncytiotrophoblast; BM, basement membrane.
Interestingly, only a weak expression of megalin was observed in first trimester syncytiotrophoblasts and expression of megalin appeared to be extremely weak or absent in term placenta syncytiotrophoblasts (Table 1). In the first trimester syncytiotrophoblasts, megalin was primarily observed in vesicular structures in the apical region of the cells as well as on the apical plasma membrane (Fig. 3D).
Megalin Expression and Localization in Trophoblast-Like Cell Lines
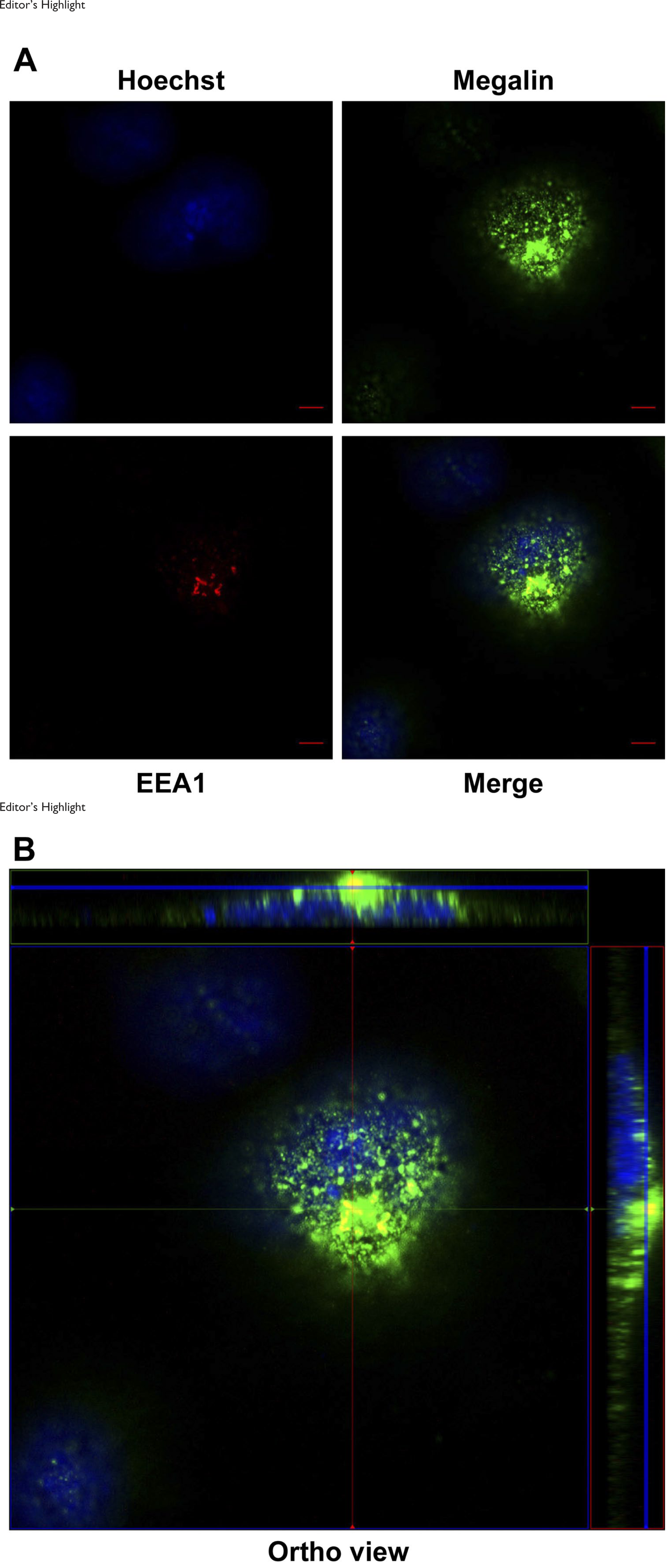
We also investigated the LRP2 mRNA and megalin protein expression in the first trimester trophoblast-like choriocarcinoma-derived cell lines BeWo, JEG-3, and JAR.36–38 Interestingly, as shown in Fig. 5A and B, we measured that JEG-3 and JAR cells express 707- and 997-fold more LRP2 mRNA than BeWo cells, respectively. We made a similar observation at protein level, where no megalin could be detected in BeWo cells, although four times more cell lysate was loaded of these cells compared with JEG-3 and JAR (Fig. 5C). Furthermore, we investigated the subcellular localization of megalin in JAR cells by immunofluorescence analyses. Megalin labeling of living JAR cells at 4C in the absence of detergent showed that megalin was present at the plasma membrane (Fig. 5D and E). More specifically, megalin appeared to be localized in clusters perhaps similar to the endocytic invaginations, we observed by immunocytochemistry analyses of sections of first trimester placenta. In permeabilized JAR cells, megalin appeared to localize extensively to vesicle-like structures of many different sizes (Fig. 6A; green image), which is in agreement with the data of our immunocytochemistry analyses of sections of first trimester placenta. In addition, megalin was identified in EEA1-positive compartments (EEA1 alone in the red image and potential colocalization with megalin in merged image; Fig. 6A and B; B representing an ortho display of A).
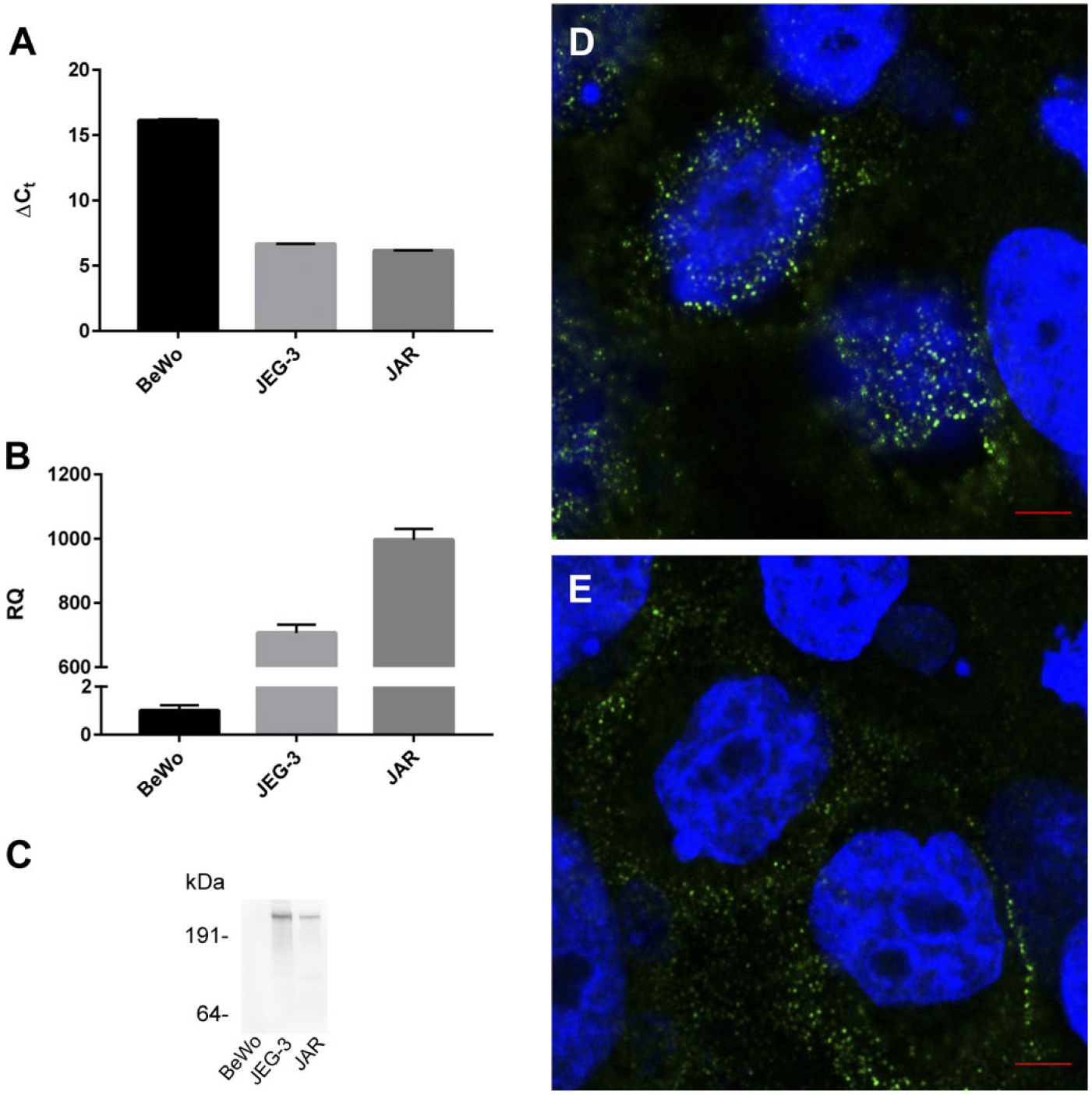
(A) Quantitative real-time PCR (qRT-PCR) analyses of the LRP2 expression in BeWo, JEG-3, and JAR cells. ΔCt values are displayed. ACTB was used as normalizer. (B) qRT-PCR analyses of the LRP2 mRNA expression in BeWo, JEG-3, and JAR cells. For calculation of relative quantification values, BeWo cells were used as calibrator. (C) Immunoblotting of total lysate of BeWo, JEG-3, and JAR cells using protein G–purified anti-human megalin antibody (10 µg/ml); 200-, 50-, and 50-µg total lysates of BeWo, JEG-3, and JAR cells, respectively, were loaded. (D and E) Immunofluorescent labeling of megalin at the surface of JAR cells. JAR cells were labeled with protein G–purified anti-human megalin antibody (10 µg/ml) at 4C in the absence of detergent before fixation and labeling with Alexa Fluor 488 fluorescence–labeled secondary antibody. Z-stacking was performed, stacks of 0.315 µm. Top (D) and middle (E) slices shown. Nuclear staining in blue (Hoechst). Scale bars: D, E = 5 µm. Abbreviation: LRP2, lipoprotein receptor–related protein 2.
Editor’s Highlight
Biochemical Characterization of Placental Megalin
Finally, we performed a biochemical characterization of placental megalin. We investigated the molecular weight and electrophoretic mobility of megalin expressed in term placenta and first trimester–derived JAR cells and compared it with renal megalin. As seen in Fig. 7A, term placenta megalin and JAR cell–derived megalin were both recognized by the purified anti-megalin antibody previously used for studies of human renal and melanoma megalin.24–26 Also, the molecular weight of term placenta megalin and JAR cell–derived megalin was similar to that of renal megalin (Fig. 7A). Furthermore, the purified anti-megalin antibody only detected full-length megalin in total term placenta homogenate (as well as a smaller protein of ~200 kDa, which was also recognized in kidney homogenate) and only full-length megalin in total cell lysate of JAR cells (Fig. 7A). The smaller protein in placenta and kidney homogenate is most likely a megalin degradation product, formed during postmortem tissue handling. Finally, placental megalin was purified by ligand (RAP) affinity chromatography from total term placenta homogenate. Elution fractions were analyzed by SDS-PAGE and silver staining (Fig. 7B) as well as immunoblotting with anti-megalin antibody (not shown). A high-molecular-weight band of ~500 to 600 kDa corresponding to the band we observed in immunoblotting was excised from the silver-stained gel and subjected to in-gel digestion and subsequent MS analysis. This identified LRP1 (data not shown), which was previously identified in and purified from human term placenta,39–42 as well as megalin/LRP2 protein (one significant peptide identified with a score of 35, Fig. 7C).
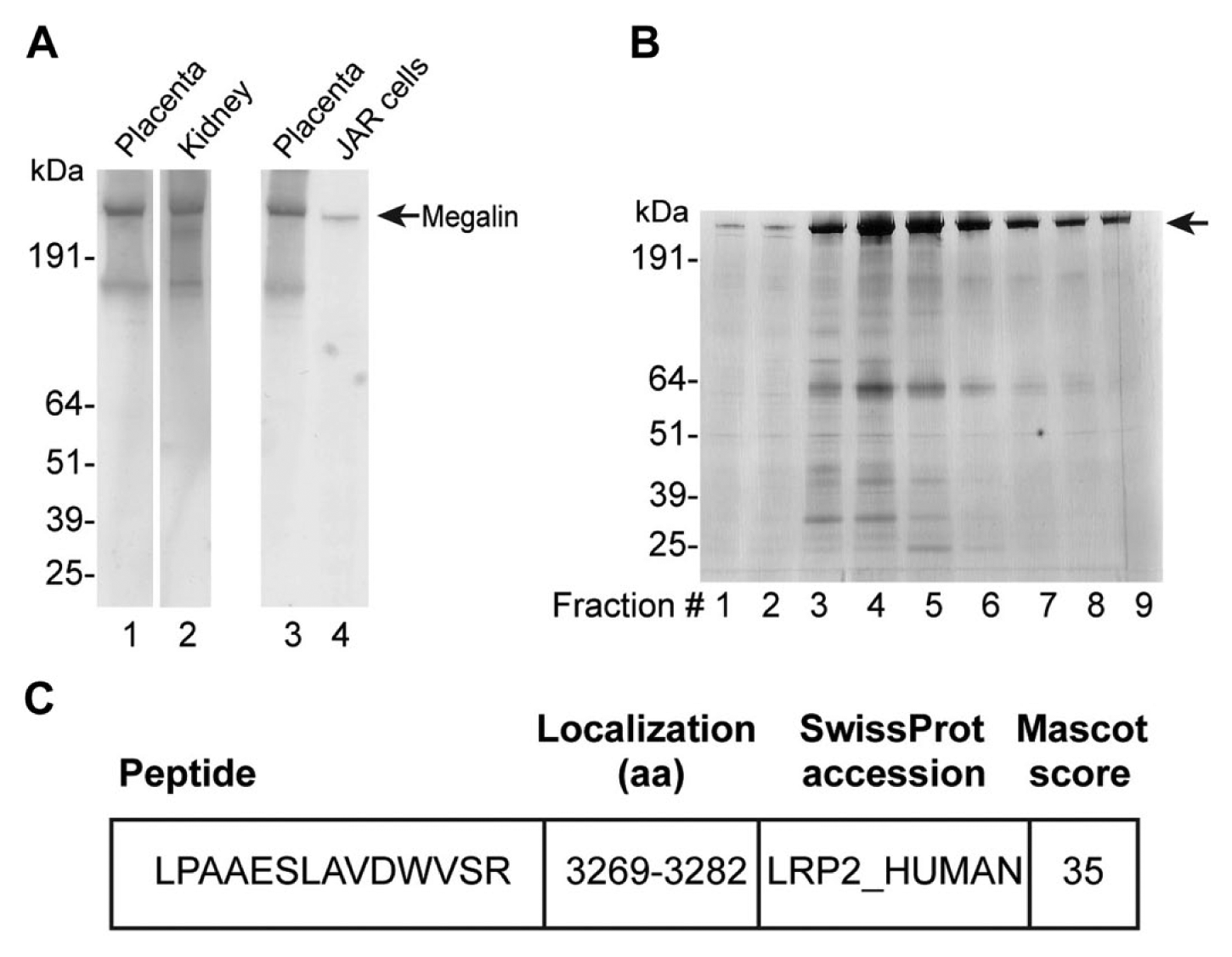
(A) Non-reducing SDS-PAGE (4%–12% polyacrylamide gels) and immunoblotting of 200-µg total homogenate of human term placenta (lane 1), 20-µg human kidney cortex (lane 2), 200-µg total homogenate of human term placenta (lane 3), and 50-µg total JAR cell lysate (lane 4). Detection of megalin was performed using protein G–purified anti-human megalin antibody (10 µg/ml). (B) Proteins were purified from homogenate of human term placenta by ligand affinity chromatography, and elution fractions were analyzed by non-reducing SDS-PAGE (4%–12% polyacrylamide gel) and silver staining; 25 µl of each elution fraction (1–9) loaded. Strong high-molecular-weight band presumed to contain megalin. (C) A high-molecular-weight band of ~500 to 600 kDa presumed to contain megalin was excised, and the presence of megalin/LRP2, as well as LRP1; (data not shown) in the excised gel piece was confirmed by liquid chromatography–tandem mass spectrometry and identification of the peptide indicated here. Abbreviation: LRP, lipoprotein receptor–related protein.
Discussion
In the present study, we investigated the expression and subcellular localization of megalin in human first trimester and term placenta. Immunohistochemical detection of megalin in first trimester placenta revealed a strong expression of megalin in cytotrophoblasts as well as a weaker expression in the syncytial layer. Analyses of human term placenta revealed that megalin is predominantly expressed in the cytotrophoblasts with only an imperceptible megalin expression in term placenta syncytiotrophoblasts.
Immunohistochemical detection of megalin across gestational ages, ranging from 6 weeks to term, was recently reported by Burke and coworkers. 20 In agreement with our observations of megalin expression in first trimester placenta, they identified megalin in both cytotrophoblasts and syncytiotrophoblasts. 20 However, in contrast to our observations, they reported megalin expression in both cell types in term placenta and did not specify differences in expression levels between cytotrophoblasts and syncytiotrophoblasts. Other studies of megalin expression in human term placenta have reported expression either in the cytotrophoblasts 17 or in the syncytial layer only 21 but not simultaneous expression in both cell types. The differences between our study and the previous studies of megalin in human placenta are very intriguing. We think they may be a result of differences in antibody specificity, tissue preparation, section thickness, and/or the immunohistochemical method applied. For the present study of megalin in human placenta, we used a well-characterized,24–26 protein G–purified rabbit polyclonal antibody raised against ligand affinity–purified human megalin. Most importantly, the specificity of the antibody has been verified on renal tissue from patients lacking the megalin protein. 25 Furthermore, as shown in the present study, this anti-megalin antibody only recognizes full-length megalin and a potential megalin degradation product (also observed in total kidney homogenate) in immunoblotting of total placenta homogenate. Our study illustrates that what has previously been reported as a heterogeneous labeling of the syncytial layer in term placenta might instead be strong labeling of cytotrophoblasts and only weak or absent labeling of the syncytiotrophoblasts in term placenta.
Our observation is further supported by the results from our mRNA and protein analyses of the three trophoblast-like choriocarcinoma-derived cell lines; BeWo, JEG-3, and JAR. These cell lines provide an alternative to isolating primary trophoblasts, as they share many biochemical and morphological properties with placental villous trophoblasts.36–38 In addition, by treatment with forskolin, BeWo cells can be stimulated to form a syncytial monolayer by differentiation and fusion of cells.38,43,44 This has not been shown for JEG-3 and JAR cells. Accordingly, BeWo cells have been suggested to be more “syncytiotrophoblast-like” than JEG-3 and JAR cells and a better model for studying transplacental transport mechanisms.36–38,45 We show here that JEG-3 and JAR cells express substantially more LRP2 mRNA and megalin protein than BeWo cells in line with our immunohistochemical observations regarding low megalin expression in syncytiotrophoblasts versus cytotrophoblasts. We therefore speculate that perhaps the megalin expression is downregulated during cytotrophoblast differentiation to syncytiotrophoblasts and/or during establishment of the multinuclear syncytial layer, especially in term placenta.
Our immunocytochemical investigations of placental megalin localization showed that megalin localized to the apical plasma membrane facing the maternal bloodstream as well as to intracellular vesicles close to the apical membrane in first trimester syncytiotrophoblasts. In line with our immunohistochemical analyses, we did not observe any distinct labeling for megalin in term placenta syncytiotrophoblasts.
In the cytotrophoblasts of human first trimester and term placenta, we identified megalin on the plasma membrane facing the apical as well as the basolateral sides, the plasma membrane invaginations, and in numerous intracellular vesicular structures. The subcellular localization of placental megalin was also investigated in JAR cells, where megalin was observed to localize to the plasma membrane as well as intracellular vesicular structures. Within intracellular vesicular structures of JAR cells, we observed that megalin was able to colocalize with a marker of early endosomes, EEA1. Combined, these observations are consistent with compartments associated with the endolysosomal system as previously reported for megalin in cells of the kidney proximal tubular epithelium, retina pigment epithelium, and non-pigmented ciliary epithelium22,28,46–48 as well as the endocytic receptor function ascribed to megalin in these tissues. It furthermore points toward a similar role for placental megalin in first trimester and term placenta cytotrophoblasts as well as in first trimester syncytiotrophoblasts.
Our results thus indicate that in first trimester placenta, megalin might serve the previously suggested role and mediate uptake of nutrients from the maternal bloodstream via the syncytial layer. In term placenta, however, megalin-mediated uptake of nutrients from the maternal bloodstream via the syncytial layer seems rather unlikely due to very low megalin expression. Accordingly, we suggest that the general perception of megalin as a nutrient receptor in human placenta throughout pregnancy should be revised.
Intriguingly, and different from what we observed for megalin in first trimester syncytiotrophoblasts, we detected cytotrophoblast-derived megalin at both the apical and basolateral membranes of the cytotrophoblasts as well as in vesicular structures close to these membranes. This indicates that megalin could be involved in uptake and transport of biomolecules delivered to the cytotrophoblasts by the syncytial layer, by other cytotrophoblasts, or by the fetus and not only from the maternal bloodstream as previously suggested.
Our immunocytochemical detection of megalin in first trimester and term placenta cytotrophoblast cells furthermore identified megalin in vesicular structures in the juxtanuclear region, near the Golgi apparatus. To our knowledge, this has only been reported for megalin once previously, in rat yolk sac cells (Brown Norway/Moloney sarcoma virus [BN/MSV]) 49 which too are proliferative cells like the cytotrophoblasts. This may suggest a strong and ongoing de novo synthesis of megalin in these cells. Notably, cytotrophoblasts and BN/MSV cells are significantly different from the highly differentiated, non-proliferative, and specialized epithelial cells typically expressing megalin, for example, retina pigment epithelium and proximal tubular epithelium. Interestingly, none of the studies of megalin expression, localization, and function in non-proliferative epithelial cells have reported megalin presence in the juxtanuclear area.22,28,46–48 Our detection of megalin in these compartments of human cytotrophoblasts thus points toward potential novel and yet to be discovered functions of megalin in these cells.
The highly proliferative cytotrophoblasts are considered to be the placental stem cells, 5 feeding cells to the syncytial layer. In line with a prominent expression of megalin in stem cell–like cytotrophoblasts, megalin expression was likewise recently described in the ocular ciliary marginal zone of mice 50 which holds undifferentiated retinal stem and progenitor cells in vertebrates. 51 Here, megalin has been proposed to modulate sonic hedgehog signaling during embryonic development. 50 Megalin has furthermore been identified in the subependymal zone of the lateral ventricles in adult brain and to modulate bone morphogenetic protein 4 signaling in these cells. 52 Again, the subependymal zone is considered a neurogenic stem cell niche crucial for generation of new neurons and glial cells. 52 The IGF-1 and EGF growth factors known to promote placental development and growth as well as fetal growth 5 are both established ligands for megalin,15,16 and a recently published study of megalin function in melanoma cells revealed that megalin may be involved in modulation of IGF-1 signaling in these cells. 26 In the context of growth factor signaling, MVBs were previously shown to play key roles in the control of growth factor signaling (e.g., EGF signaling), 53 and interestingly, we observed megalin on the luminal vesicles of MVBs as well as their outer limiting membrane in cytotrophoblasts of first trimester and term placenta. To our knowledge, megalin has not previously been identified in MVBs. Based on our novel data and these previous studies, we speculate that megalin might contribute to modulation of signaling pathways in placental trophoblasts and thus play a role in development and growth of the human placenta. However, this remains purely speculative at this point.
Summarized, we have contributed significantly to an improved understanding of megalin expression and localization in the human placenta by studying the intracellular localization of megalin in the cytotrophoblasts and syncytiotrophoblasts in early gestation as well as in term placenta. To our knowledge, this is the first time a detailed study of the subcellular localization of megalin in human placenta has been performed. Our results question some of the previous assumptions and conclusions regarding megalin function in human placenta and point toward novel and complex functions. We believe that future studies designed from a revised perspective are warranted to further elucidate the function of placental megalin.
Footnotes
Acknowledgements
We thank Gitte Fynbo Biller, Inge Kjaergaard, Mona Britt Hansen, and Hanne Sidelmann (all Aarhus University) for technical assistance; Soren K. Moestrup (University of Southern Denmark) for anti-megalin antibody; and Soren K. Moestrup and Rikke Nielsen (Aarhus University) for fruitful discussions.
Authors’ Note
The author Mette Madsen has previously published under the name Mette Kristiansen.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
TS: Participated in the design of the study and analyses of data, performed the IHC and immunocytochemistry analyses, and partially drafted the manuscript. EIC: Participated in the design of the study and analyses of data. JNC: Performed quantitative real-time PCR and immunoblotting and participated in manuscript preparation. TK: Performed purification of megalin from placenta and kidney tissue. NU: Provided placental tissue for IHC, immunocytochemistry, and biochemical characterization of megalin. AL: Provided placental tissue for IHC and immunocytochemistry. BH: Performed mass spectrometry experiments and data analyses. MM: Responsible for the design of the study, performed immunofluorescence analyses, involved in data analyses, and partially drafted the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Danish Council for Independent Research, Novo Nordisk Foundation, A.P. Møller Foundation, and Aarhus University.
