Abstract
Glycyrrhizic acid (GA), an active ingredient in licorice, has multiple pharmacological activities. However, the effects of GA on sepsis-induced acute lung injury (ALI) have not been determined. Tthe aim of this study was to investigate the molecular mechanism involved in the effects of GA against sepsis-induced ALI in rats. We found that GA alleviated sepsis-induced ALI through improvements in various pathological changes, as well as decreases in the lung wet/dry weight ratio and total protein content in bronchoalveolar lavage fluid, and a significant increase in the survival rate of treated rats. Additionally, GA markedly inhibited sepsis-induced pulmonary inflammatory responses. Moreover, we found that treatment with GA inhibited oxidative stress damage and apoptosis in lung tissue induced by ALI. Finally, GA treatment significantly inhibited NF-κ B, JNK and P38 MAPK activation. Our data indicate that GA has a protective effect against sepsis-induced ALI by inhibiting the inflammatory response, damage from oxidative stress, and apoptosis via inactivation of NF-κB and MAPK signaling pathways, providing a molecular basis for a new medical treatment for sepsis-induced ALI.
Introduction
Sepsis, a common complication of postoperative critical patients, is a systemic inflammatory response syndrome caused by infection and is also one of the leading causes of death. The lung is the most vulnerable organ during sepsis and about 25% to 50% of patients with sepsis may experience acute lung injury (ALI) (Cohen 2002). With advances in supportive care, the mortality rate for patients with ALI has decreased over time but is still as high as 70%. Undoubtedly, the identification of therapeutic and preventive approaches that are innovative, safe and effective is crucial for successful sepsis treatment.
During ALI, the permeability of the alveolar capillary membrane is increased and inflammatory cells from the blood are recruited by inflammatory cytokines (Matthay and Zemans 2011). The remarkable characteristic of ALI is acute pulmonary edema and interstitial fibrosis caused by oxidative stress and overwhelming inflammation in the lung (Ruffmann 1998; Hotchkiss and Karl 2003; Puneet et al. 2005). Research has also shown that pulmonary microvascular endothelial cells in apoptosis is increased in patients with ALI, whereas the pulmonary microvascular permeability is significantly strengthened; this demonstrated some correlation between apoptosis and pulmonary microvascular permeability (Damarla et al. 2014). Thus, there is reason to believe that apoptosis of pulmonary endothelial cells may cause the increase in pulmonary microvascular permeability and pulmonary edema seen in this condition.
Glycyrrhizic acid (GA), obtained from the root of the Glycyrrhiza glabra shrub, is the primary bioactive constituent of licorice and possesses a wide range of pharmacological and biological benefits, such as anti-inflammatory, anti-viral, antioxidant, anti-tumoral and hepatoprotective actions (Abe et al. 1982; Baba and Shigeta 1987; Sato et al. 1996; Wang and Nixon 2001; Ming and Yin 2013). This compound is commonly used in Asia as a therapeutic agent for chronic viral hepatitis (Arase et al. 1997; van Rossum et al. 2001; Yoshida et al. 2007). Recent studies also have shown that GA also offers various protective effects in the lung. A study by Menegazzi et al. (2008) showed that GA can attenuate the development of carrageenan-induced lung injury in mice. GA has also been shown to alleviate lung injury in rats exposed to benzo(a)pyrene (Qamar et al. 2012), and another study showed that GA treatment is associated with the attenuation of lipopolysaccharide-induced acute lung injury by inhibiting cyclooxygenase-2 and inducible nitric oxide synthase expression (Ni et al. 2011). However, the effect of GA on sepsis-induced ALI and its related molecular mechanism(s) are poorly understood. In the present study, we established a model of sepsis-induced ALI using cecal ligation and puncture in rats to investigate the therapeutic potential and detailed molecular mechanisms of GA.
Materials & Methods
Drugs and Antibodies
GA was purchased from Meilun Biotic Co., Ltd (Dalian, China). GA was dissolved in 10% DMSO to a concentration of 5 mg/ml before use. The chemical structural of GA is shown in Fig. 1. The antibodies used for western blotting were as follows: anti-Bcl-2 (Boster; Wuhan, China), anti-Bax (Boster), anti-cleaved caspase-3 (Abcam, Cambridge, MA), anti-iNOS (Bioss; Beijing, China), anti- p-IκB-α (Bioss), anti-IκB-α (Bioss), anti-p-JNK (Santa Cruz; Dallas, TX), anti-JNK (Santa Cruz), anti-p-P38 (Santa Cruz), anti-P38 (Santa Cruz), and anti-β-actin (Boster).
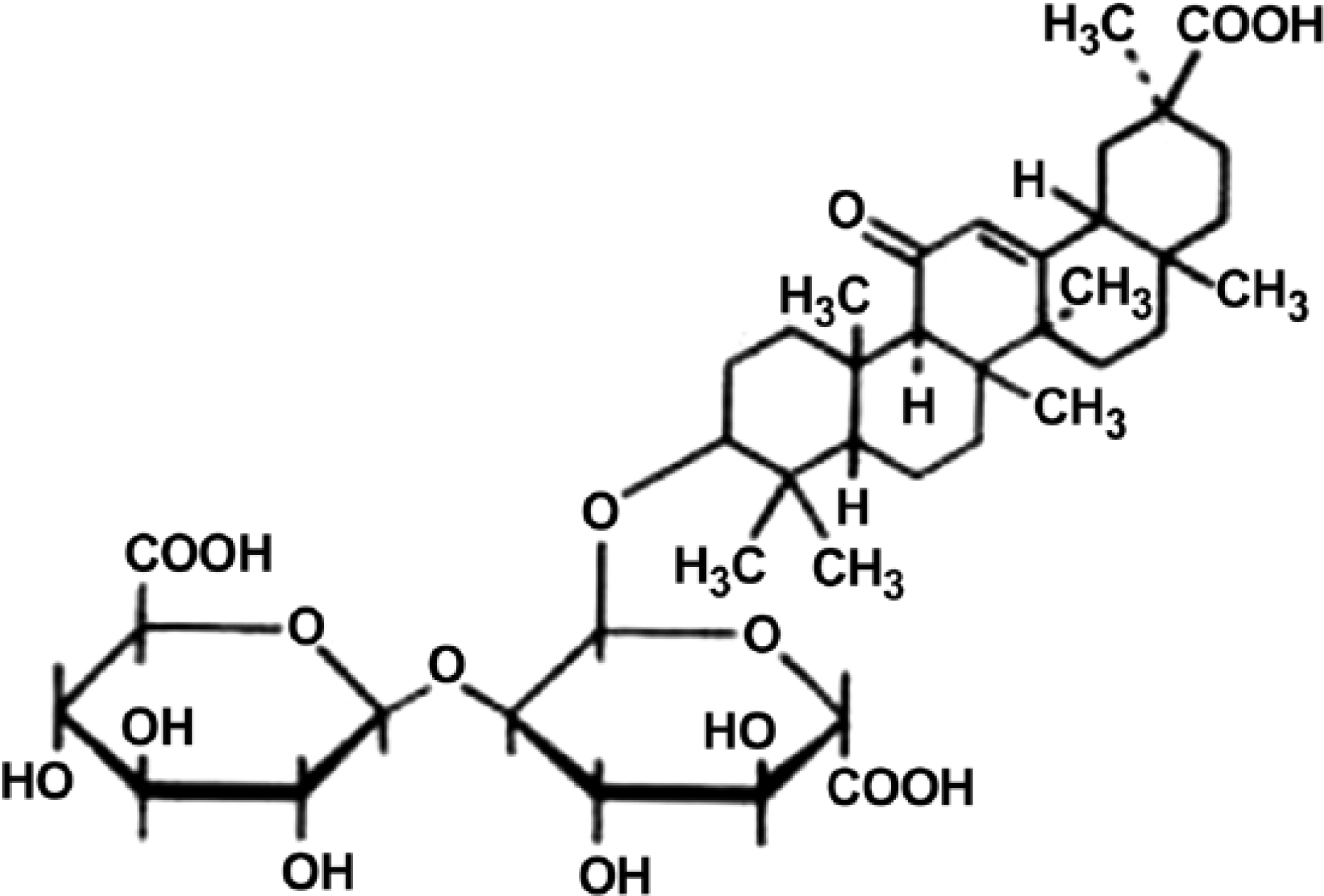
Chemical structure of GA. The molecular formula of GA is C42 H62 O16 and its molecular weight is 822.93.
Animals and Experimental Protocol
Pathogen-free male Sprague-Dawley rats (weight, 200 ± 20 g) were purchased from Beijing Vital River Laboratory Animal Co., Ltd. (Beijing, China). The animals were maintained at 23°C under a 12 hr light-dark cycle with free access to food and water. The night before the experiments, food was restricted but the animals had free access to water. Thirty rats were randomly divided into five experimental groups (n=6 per group): sham operation group, GA (50 mg/kg) group, sepsis group, sepsis plus GA (25 mg/kg) and sepsis plus GA (50 mg/kg). We induced sepsis by cecal ligation and puncture (CLP). Briefly, the rats were anaesthetized by injection with sodium pentobarbital (50 mg/kg) and a laparotomy was performed through a 2-cm ventral midline abdominal incision. The cecum was punctured twice at different sites with an 18-gauge needle and gently compressed until feces had extruded; the cecum was then repositioned. The incision was closed in two layers. The sham operation group underwent laparotomy through a midline incision, but the cecum remained unpunctured. Animals in the GA (50 mg/kg), sepsis plus GA (25 mg/kg) and sepsis plus GA (50 mg/kg) groups were intraperitoneal injected with 25 mg/kg or 50 mg/kg GA immediately after CLP, whereas the sham operation group and sepsis group received an intraperitoneal injected of isovolumetric 10% DMSO. Twenty-four hours after surgery, all of the animals were euthanized and peripheral blood and lung tissues were collected for further tests. All animal experiments were carried out strictly in accordance with international ethical guidelines and the National Institutes of Health Guide concerning the Care and Use of Laboratory Animals. The experiments were approved by the Institutional Animal Care and Use Committee of Shengjing Hospital of China Medical University.
Survival Curves
To observe the effect of GA on survival, another 40 rats were randomly divided into four experimental groups (n=10 per group): sham operation group, sepsis group, sepsis plus GA (25 mg/kg) and sepsis plus GA (50 mg/kg). Observations of survival were performed every 12 hr until the endpoint at 96 hr.
Hematoxylin-Eosin (H&E) Staining
Lung tissue samples were fixed in 10% buffered formalin for 48 hr and were then dehydrated by washing in ascending grades of ethanol. Samples were then sectioned and embedded in paraffin wax. Five-μm lung sections were prepared for routine H&E stains. H&E stain was used to observe the morphological changes to the lung tissue sections among the various groups. Under 200× magnification, each section was randomly selected and five fields of view were photographed.
Lung Wet/Dry (W/D) Weight Ratio
Lung W/D weight ratio was determined to assess the lung edema. The fresh right middle lobe was measured using an electronic scale and then dried in an oven at 85°C for 48 hr until the weight remained constant. Dry weights were measured, and the W/D ratios were calculated.
Inflammatory Cell Counts in Bronchoalveolar Lavage Fluid (BALF)
BALF was concentrated by centrifugation (1000 rpm, 10 min at 4°C), and the cell pellet was resuspended in 0.5 ml PBS. Total cell count in the BALF was determined with a hemocytometer. A differential leukocyte cell count was performed using 10 μl of the cell suspension, which was transferred to a glass slide and stained with Giemsa stain.
Protein Concentration in Lung BALF
The concentration of protein in the BALF was determined by the BCA protein estimation kit (Beyotime; Haimen, China).
ELISA
The levels of tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, nitrotyrosine (NT), and 8-Oxo-2’-deoxyguanosine (8-OhdG) were measured by commercially available ELISA kits according to the manufacturer’s instructions (Uscn Life Science; Wuhan China). Concentrations were calculated by generating a standard curve using standard proteins and analyzed using curve expert 1.3 software.
Determination of Nitric Oxide (NO)
The amount of nitrite (a stable metabolite of NO) in the lung tissues was detected by the Griess Reagent System (Beyotime) according to the manufacturer’s recom-mendation. Absorbance was measured at 540 nm and the nitrite concentration was determined using various kalium nitrite concentrations (1, 2, 5, 10, 20 and 50 μM) as a standard.
Detection of MPO and SOD Activities and MDA Concentration
Lung samples were homogenized with 0.9% normal saline and centrifuged. Superoxide dismutase (SOD) and myeloperoxidase (MPO) activities, as well as the malondialdehyde (MDA) concentration were determined using commercial kits produced by Jiancheng Bioengineering Institute (Nanjing, China, according to the manufacturer’s instructions. MPO and SOD activities were measured by colorimetry. Results are expressed as MPO units/g of protein and SOD units/mg of protein. The MDA concentration is expressed as mM/mg of protein (mM/mgprot).
Terminal Deoxynucleotidyl Transferase-mediated dUTP Nick End Labeling (TUNEL) Staining
Apoptotic cells in lung tissues were detected by TUNEL on rat lung tissue sections using the DeadEnd™ Colorimetric TUNEL System (Promega; Madison, WI) following the manufacturer’s protocols. The numbers of TUNEL-positive cells were counted from six sections under a microscope (400×).
Western Blot Analysis
Total protein in the lung tissue samples was lysed in RIPA buffer and then denatured. The protein concentration was determined by the BCA protein estimation kit (Beyotime). Equal quantities of protein samples were separated by SDS-PAGE and then transferred to polyvinylidene fluoride membranes. The membranes were blocked with 5% non-fat dry milk in PBS, and incubated with anti-Bcl-2 (1:1000), anti-Bax (1:1000), anti-cleaved caspase-3 (1:1000), anti-iNOS (1:1000), anti-p-I κ B α (1:500), IκB-α (1:1000), anti-p-P38 (1:200), anti-P38 (1:200), anti-p-JNK (1:200), anti-JNK (1:200), anti-β-actin (1:1000) antibodies respectively, at 4°C overnight. Membranes were then washed and incubated with goat anti-rabbit/mouse secondary antibodies conjugated with horseradish peroxidase (1:5000), followed by ECL detection reagent (Beyotime). Photographs were taken and the optical densities of the bands were scanned and quantified with the Gel Doc 2000 (Bio-Rad; Hercules, CA).
Immunohistochemistry
The expression of iNOS in lung tissue was detected by immunohistochemistry. Lung tissue was fixed in 4% paraformaldehyde, then embedded in paraffin. Lung sections of 5 μm were prepared and placed on poly-L-lysine-coated slides, then kept in an oven at 60°C for 24 hr to increase section adherence to the slide. The slides were deparaffinized using xylene and dehydrated by graded concentrations of alcohol, then placed in a retrieval solution and incubated for 15 min in a microwave at high power. The slides were then cooled and washed with wash buffer and incubated sequentially with primary antibody and biotin-labeled secondary antibody. Finally, the sections were stained with DAB, counterstained with hematoxylin, dehydrated, cleared in xylene, and fixed. Under 400× magnification, five different microscopic fields were randomly chosen.
Electrophoretic Mobility Shift Assay (EMSA)
EMSA was performed using a commercial kit (Viagene Biotech Inc.; Tampa, Florida) according to the manufacturer’s instructions. The nuclear proteins were isolated as stated above. Briefly, the binding reaction comprised 15 µg of nuclear extract and labeled DNA probe in a total volume of 15 µl, and this reaction mix was incubated for 20 min at room temperature. Electrophoresis was performed with a 4% TBE gel at 180 V for 80 min. Following electrophoresis, the DNA–protein complexes were then blotted onto nylon membranes and detected using chemiluminescence.
Statistical Analysis
All data are expressed as the mean ± SD. One-way ANOVA followed by Bonferroni’s multiple comparison test were used for comparisons among experiment groups using GraphPad Prism 5 software (GraphPad Inc., La Jolla, CA). Survival data were analyzed utilizing log-rank or χ2. A p-value of less than 0.05 was considered statistically significant.
Results
Effect of GA on Survival Rate
As shown in Fig. 2, the survival rate was significantly decreased in the sepsis group as compared with the sham operation group; however, the survival rate was more than doubled following GA treatment as compared with the sepsis group. This result confirmed the protective effect of GA on mortality in rats with sepsis.
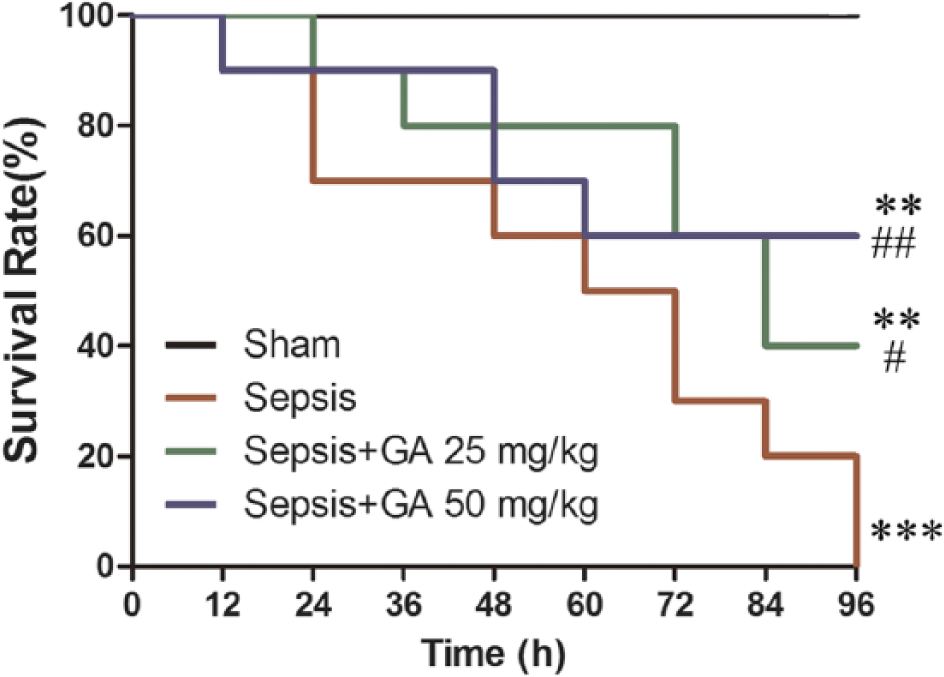
The survival rate of rats in different treatment groups. The survival curves of rats (n=10) with indicated treatment were monitored. **p<0.01, ***p<0.001 versus the sham operation group;
Protective Effect of GA against Sepsis-induced ALI
A large number of infiltrating inflammatory cells were identified in lung sections from the sepsis group by H&E staining assay, and the lung injury score was significantly higher as compared with the sham operation group, as shown in Fig. 3A. However, GA could attenuate the lung injury, with a significantly lower number of inflammatory cell infiltration measured. The lung W/D ratio was lower in GA treatment group, as shown in Fig. 3B. To further confirm the effect of GA on ALI, we detected the total protein content in BALF. As shown in Fig. 3C, the protein content was significantly higher in the sepsis group, with GA resulting in lower protein content measuring almost half that seen in the sepsis group. These results may lead us to conclude that GA has a protective effect against sepsis-induced ALI.
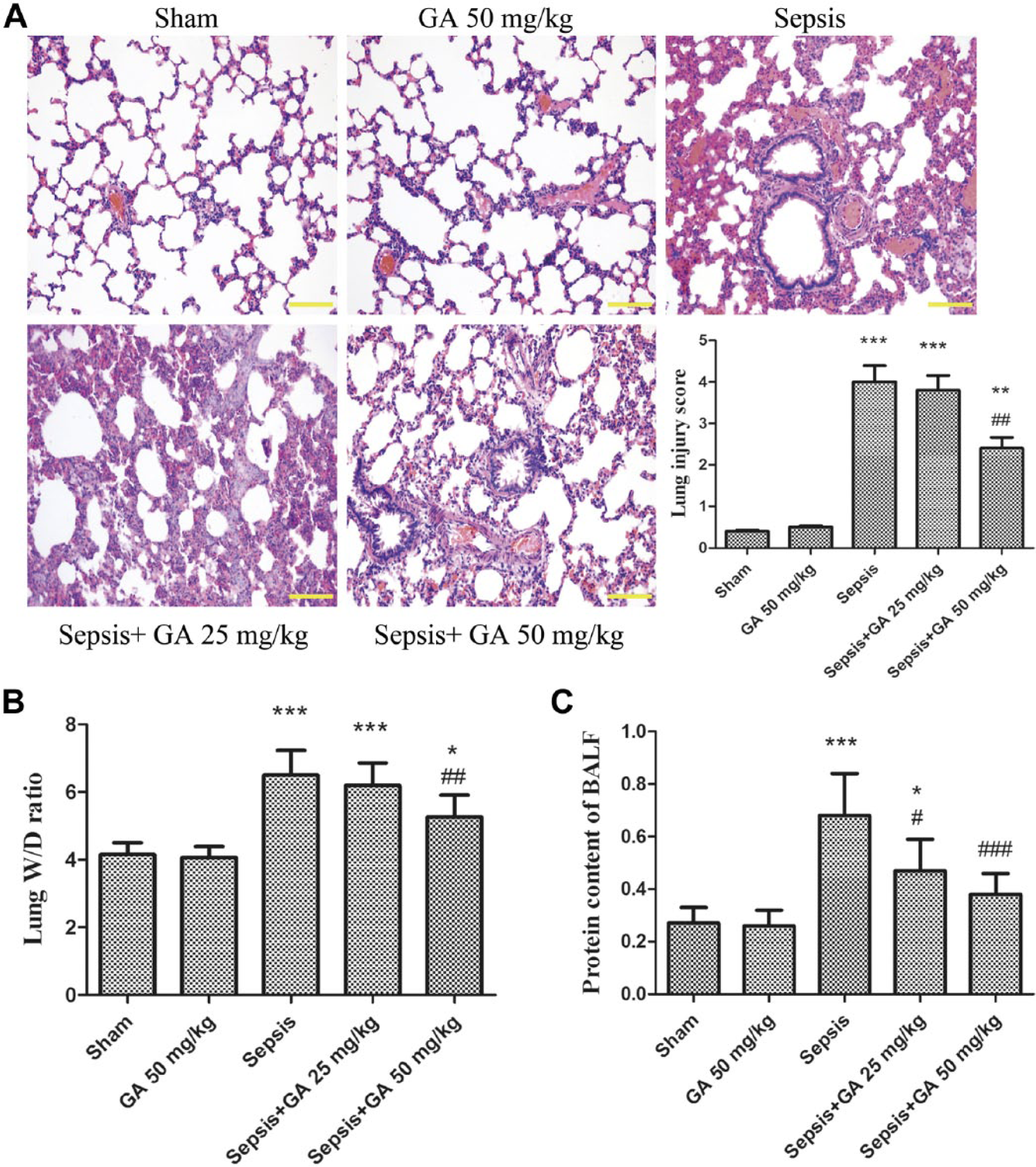
Protective effect of GA against sepsis-induced ALI. (A) Pathological changes to lung tissue detected by H&E staining (magnification 200×) and lung injury score. (B) The W/D ratio of lung tissue from different groups. (C) The total protein content of BALF from different groups. The results shown are representative of at least three independent experiments. Each value represents the mean ± SD (n=6). *p<0.05, **p<0.01, ***p<0.001 versus the sham operation group;
Effect of GA on Pulmonary Inflammatory Responses Induced by ALI
Since the essence of ALI is an uncontrolled inflammation reaction in the lung, we detected the white cells counts, MPO activity, and production of several inflammatory cytokines, such as TNF-α, IL-1β and IL-6. As shown in Fig. 4, the number of white cells in the BALF, the MPO activity in the lung tissue, and the levels of TNF-α, IL-1β and IL-6 in the peripheral blood were markedly higher in the sepsis group as compared with the sham operation group, which could be reduced by GA treatment at different concentrations. Next, we determined the white cell counts in the peripheral blood and the ascitic fluid. As shown in Supplemental Fig. 1, GA significantly prevented the sepsis-induced the increase in multinuclear leukocyte number in the peripheral blood; however, there was no obvious effect of GA on white cell counts in the ascitic fluid. Moreover, we investigated the role of GA on peripheral blood leukocyte apoptosis in vitro. The result suggested that GA significantly induced leukocyte apoptosis in vitro (Supplementary Fig. 2). To further elucidate the molecular mechanism of GA-induced leukocyte apoptosis, we detected the mitochondrial DNA expression level in peripheral blood leukocytes. As shown in Supplemental Fig. 3, the levels of NADH dehydrogenase, cytochrome B, and cytochrome C oxidase were obviously higher in the sepsis group as compared with the sham group, and treatment with GA could prevent the increased levels of this mitochondrial DNA. These data suggest the beneficial effect of GA on inflammatory responses against CLP-induced lung injury in rats.
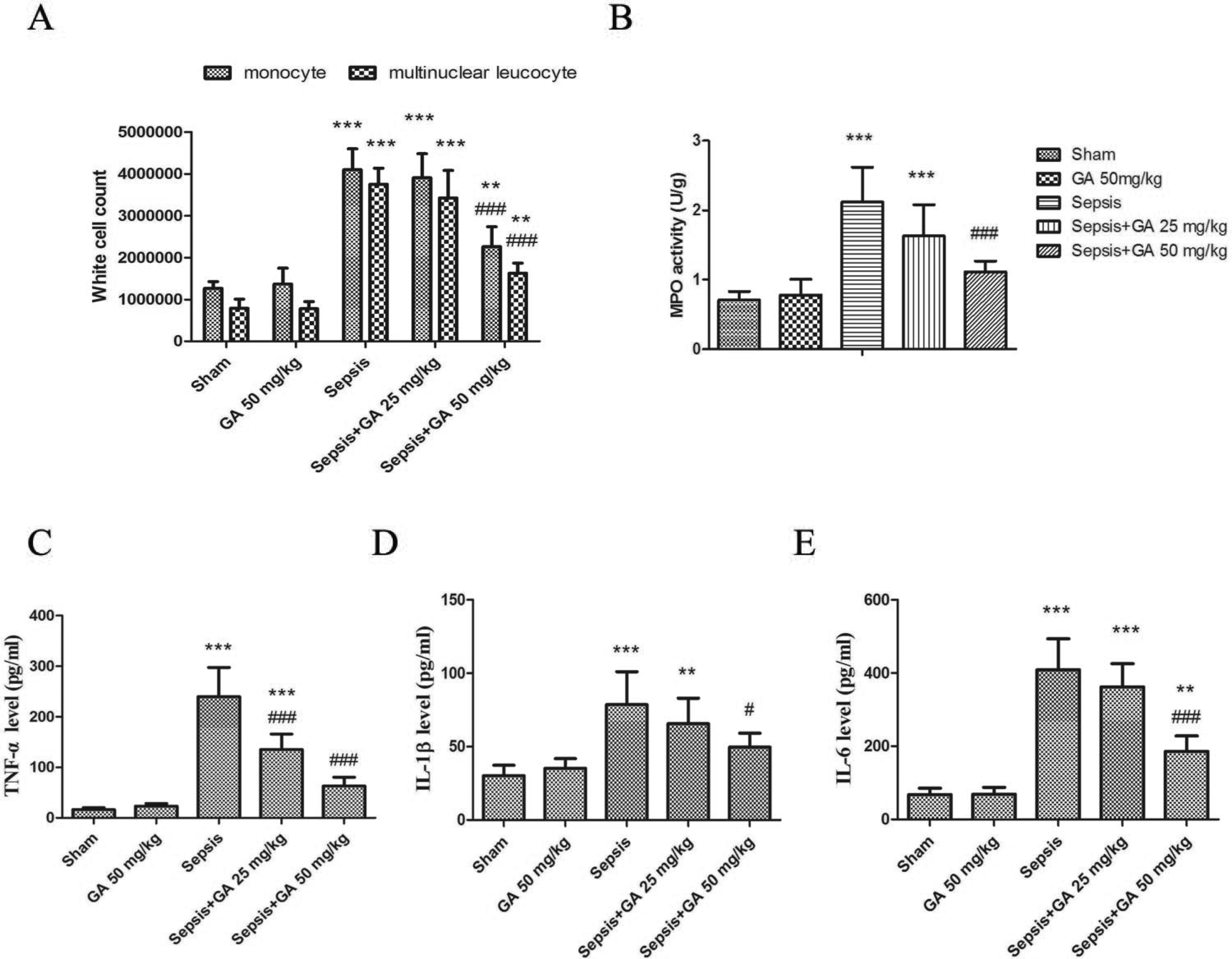
GA reduced the inflammation in sepsis-induced ALI in rats. (A) White blood cell count of BALF from different groups. (B) Activity of MPO of lung tissue from different groups. (C-E) TNF-α, IL-1β, IL-6 levels in the peripheral blood, as determined by ELISA. Results shown are the representative of at least three independent experiments. Each value represents the mean ± SD (n=6). **p<0.01, ***p<0.001, versus the sham operation group.
Effect of GA on NO Production and iNOS Expression
To evaluate the effect of GA on sepsis-induced inflammation-related mediators and proteins, the production of NO and expression of iNOS were detected. As shown in Fig. 5A, sepsis induced by CLP resulted in a two-fold increase in NO production compared with sham group, whereas GA significantly inhibited sepsis-induced the production of NO. Moreover western blot was used to evaluate the expression of iNOS. As shown in Fig. 5B, GA markedly inhibited sepsis-induced the expression of iNOS in the same manner as it inhibited the production of NO. To further illuminate the expression of iNOS, immunohistochemical staining was adopted. Our results showed that sepsis-induced the expression of iNOS was suppressed by GA treatment and was chiefly accorded with those of western blot (Fig. 5C).
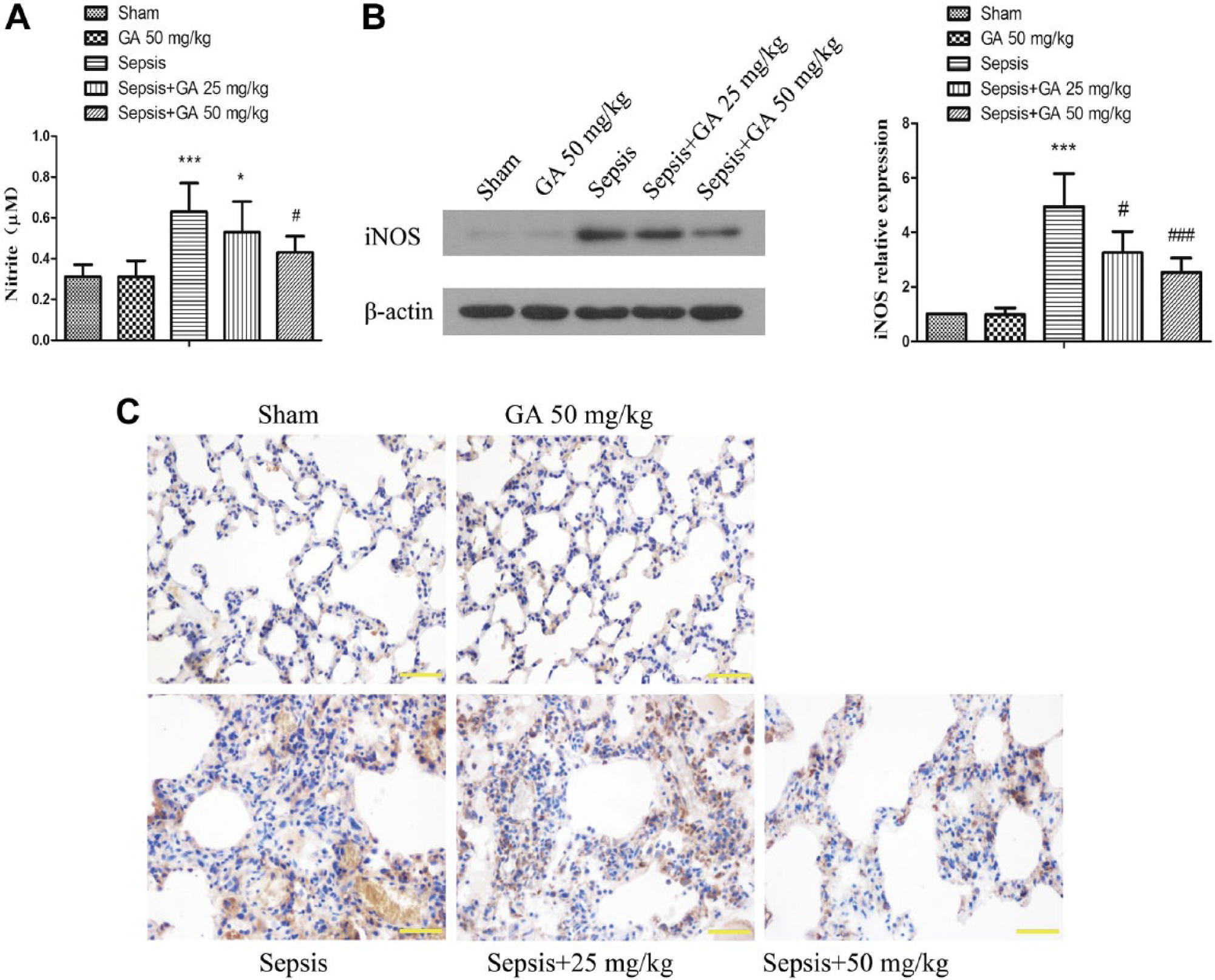
GA inhibited the production of NO and expression of iNOS in lung tissue induced by ALI. (A) The amount of nitrite in the lung tissue was detected by the Griess Reagent System. (B) The expression of iNOS in lung tissue was measured by western blot. (C) The expression of iNOS in lung tissue was confirmed by immunohistochemical staining (magnification 400×). The results shown are representative of at least three independent experiments. Each value represents the mean ± SD (n=6). *p<0.05; ***p<0.001, versus the sham operation group.
Effect of GA on Oxidative Stress Damage Induced by ALI
To evaluate the effect of GA on sepsis-induced oxidative stress damage, the level of MDA was measured. As shown in Fig. 6A, sepsis induced by CLP resulted in a significant increase in MDA production as compared with that seen in the sham group, whereas GA significantly attenuated the sepsis-induced production of MDA by more than 50%. Furthermore, the activity of SOD, a group of endogenous antioxidants, was evaluated in the lung tissue of rats. As shown in Fig. 6B, SOD activity was significantly lower in the sepsis group as compared with that in the sham group, whereas SOD activity was maintained with GA treatment. 8-OHdG, a biomarker of DNA peroxidation, was also significantly elevated in the sepsis group and attenuated with GA treatment (Fig. 6C). Finally, we measured NT, an indirect index of toxic reactive nitrogen species production. The results showed that GA treatment significantly lowered NT formation by nearly 50% as compared with that in the sham group (Fig. 6D).
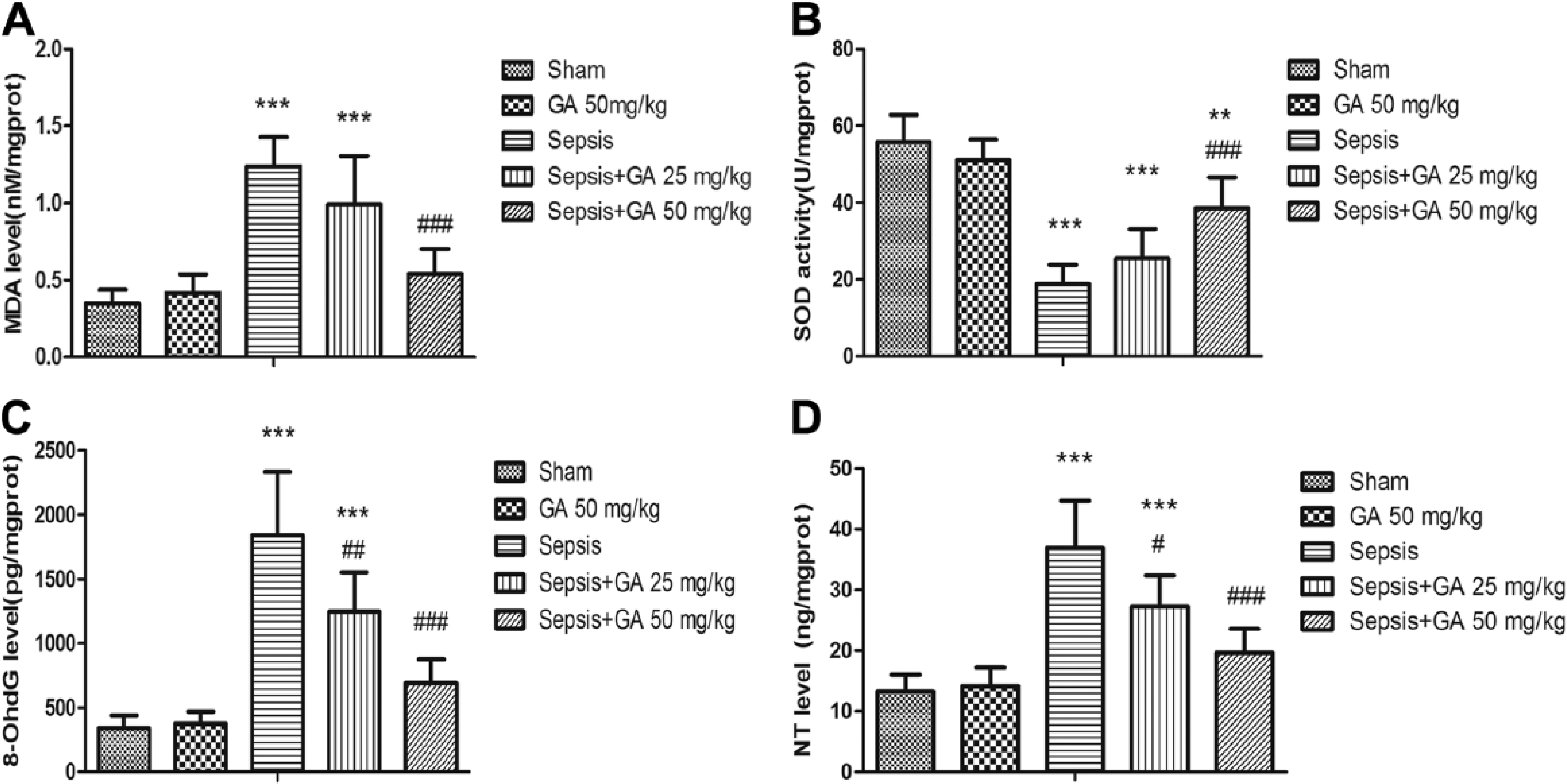
GA attenuated sepsis-induced oxidative stress damage. Malondialdehyde (MDA) production (A), superoxide dismutase (SOD) activity (B), 8-Oxo-2’-deoxyguanosine (8-OhdG) concentration (C), and nitrotyrosine (NT) concentration (D) in lung tissues from the different experimental groups. The results shown are representative of at least three independent experiments. Each value represents the mean ± SD (n=6). ***p<0.001 versus the sham operation group;
Effect of GA on Sepsis-induced Cell Apoptosis
To investigate the effect of GA on ALI-induced parenchymal cell apoptosis in lung tissue, TUNEL staining was performed. The nuclei of TUNEL-positive cells appears as brown. As shown in Fig. 7A and 7B, compared with the sham group, the number of TUNEL-positive cells was significantly higher in the sepsis group, with GA leading to an approximately 30% reduction in the number TUNEL-positive cells induced by ALI.
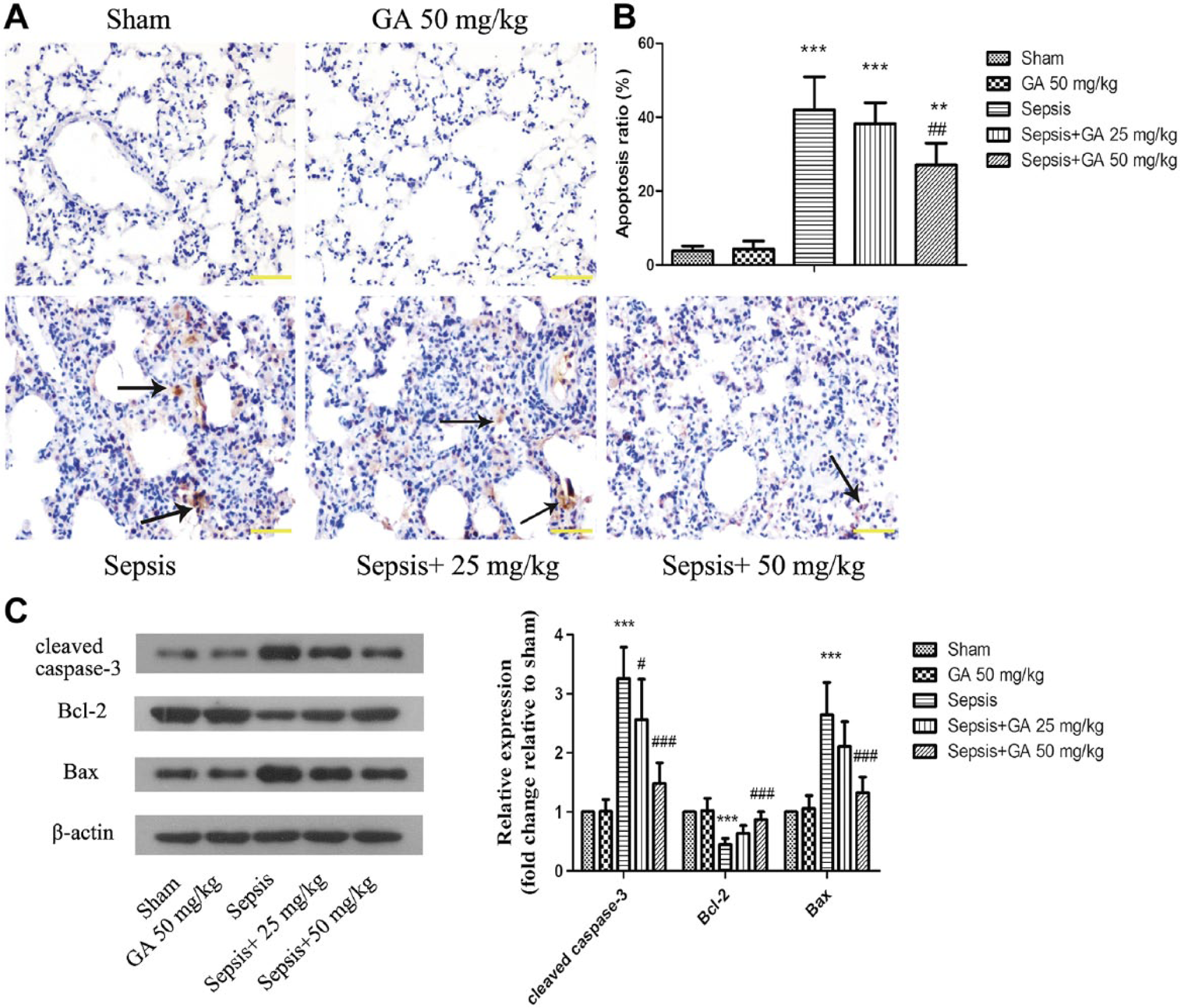
GA inhibited ALI-induced apoptosis in lung tissue. (A) The apoptosis of parenchymal cells in lung tissue was detected by TUNEL staining (magnification 400×). The arrows indicate TUNEL-positive cells. (B) The degree of apoptosis was calculated from TUNEL staining. (C) Apoptosis-related proteins were detected by western blot. β-actin was used as a loading control. The results shown are representative of at least three independent experiments. Each value represents the mean ± SD (n=6). **p<0.01, ***p<0.001 versus the sham operation group;
Moreover, we assessed the expression of a number of apoptosis-related proteins using western blotting. As shown in Fig. 7C, the expression of cleaved caspase-3 protein, a downstream effector during apoptosis, was higher in the sepsis group, as was the expression of Bcl-2. In contrast, the expression of Bax was significantly lower in the sepsis group. Thus, the ratio of Bcl-2/Bax was significantly lower in sepsis rats. Treatment with GA could attenuate the expression of cleaved caspase-3 and lead to a higher ratio of Bcl-2/Bax.
Effect of GA on NF-κB, JNK, and p38 MAPK Activation
To further explore the potential mechanism through which GA relieves sepsis-induced ALI, we determined the effects of GA on NF-κB, JNK, and p38 MAPK signaling proteins. As shown in Fig. 8A, the expression level of p-IκB-α was higher whereas that of IκB-α lower in the sepsis group as compared with the sham group, and these changes were mitigated by GA treatment. Moreover, the DNA binding activity of NF-κB was detected by EMSA assay. The result showed that GA markedly inhibited the elevation of DNA binding activity induced by ALI (Fig. 8B). As shown in Fig. 9, the levels of p-JNK and p-P38 were elevated in the sepsis group as compared with the sham group, and these activations were significantly affected by GA treatment.
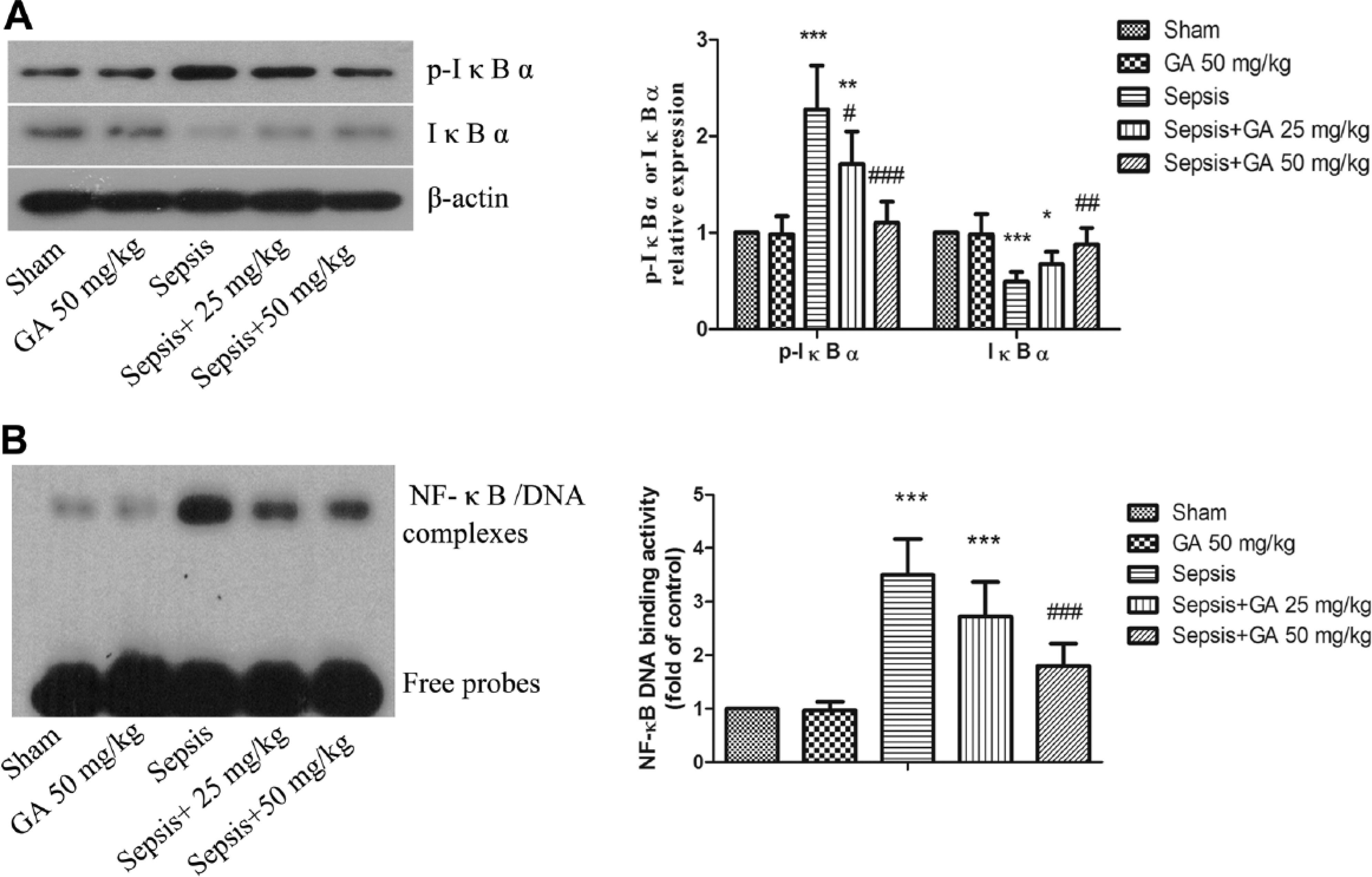
GA inhibited NF-κB activation induced by ALI. (A) The expression of p-IκB-α and IκB-α were detected by western blotting and the result of the densitometric analysis is shown. β-actin was used as a loading control. (B) DNA binding activity of NF-κB, as assessed by EMSA assay. The results shown are representative of at least three independent experiments. Each value represents the mean ± SD (n=6). *p<0.05, **p<0.01; ***p<0.001, versus the sham operation group.
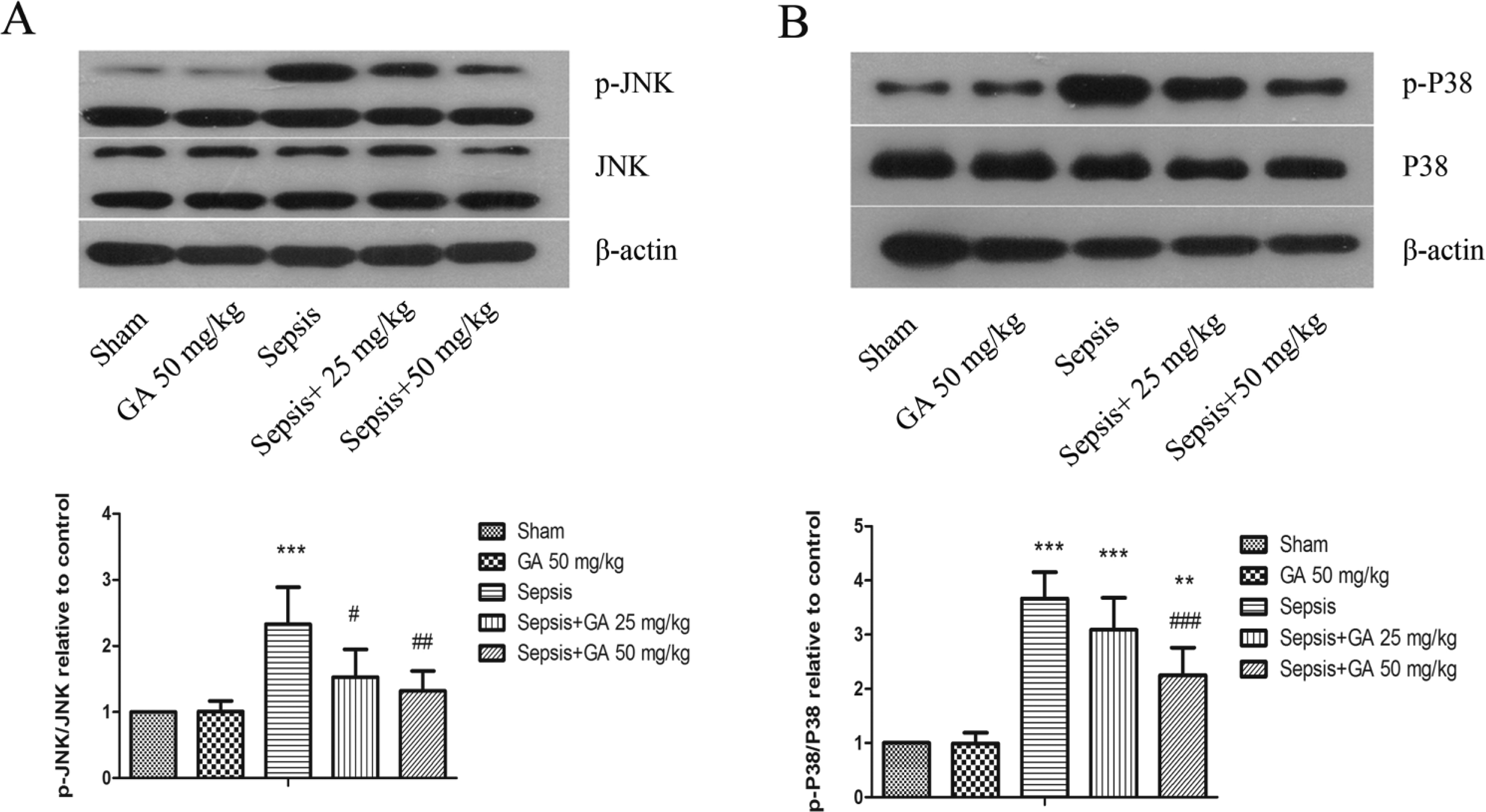
GA inhibited JNK and p38 MAPK activation induced by ALI. The expression of p-JNK and JNK (A), p-p38 and p38 (B) in lung tissues, as detected by western blotting and analyzed by densitometry. β-actin was used as a loading control. The results shown are representative of at least three independent experiments. Each value represents the mean ± SD (n=6). **p<0.01, ***p<0.001, versus the sham operation group.
Discussion
GA is the main active ingredient in licorice, which is a traditional Chinese medicine that is safe and non-toxic. In this study, we observed the effect of GA on sepsis-induced ALI in rats and investigated its potential mechanisms of action.
We established a sepsis-induced ALI model in rats by CLP. The CLP sepsis model is currently regarded as the gold standard for sepsis-related studies because of its high stability, good repeatability, and wide applicability. The tissue necrosis and gut-derived severe peritoneal cavity infection in rats subjected to CLP will result in a systemic inflammatory response syndrome (SIRS) (Ritter et al. 2003). The obvious lung damage is likely induced by endotoxins and other bacterial toxins residing in the intestines in this CLP model. Previous studies have shown that rats exhibit lung injury, low-oxygen, neutrophil cell infiltration, and alveoli and interstitial edema approximately 18–72 hr after CLP (Matute-Bello et al. 2008). During sepsis, the overwhelming inflammation increases the permeability of pulmonary epithelial cells and causes the quick accumulation of liquid in the lungs, which results in acute pulmonary edema accompanied by pulmonary interstitial fibrosis (Perl et al. 2011). Our sepsis model caused significant increases in the lung tissue histological score and W/D weight ratio, demonstrating the effectiveness and feasibility of this method. After treatment with GA, the lung tissue histological score was dose dependently decreased. Likewise, the W/D weight ratio and total protein content in BALF—both reflective of the severity of ALI—were obviously decreased, suggesting that GA offers a protective effect against lung damage. Moreover, the survival rate of ALI model rats was higher in rats in the GA treatment group. Collectively, we can conclude that GA is a novel and effective drug that could be used to attenuate sepsis-induced ALI in rats.
A large number of clinical and experimental research has indicated that ALI is typified by an uncontrolled inflammation reaction in the lung. Leukocytes are important inflammatory effector cells and play key roles in the occurrence, development and outcome of inflammation (Ge et al. 2005). A delay or defect in leukocyte apoptosis could result in an intensified inflammatory response. As such, the timely and appropriate removal of excessive leukocytes through induction of apoptotic pathways is closely related to controlling the inflammatory response and alleviating tissue injury (Filep and El Kebir 2009). According to our results, GA can effectively reduce the proportion of sepsis-induced leukocytes in vivo, presumably through apoptosis, as noted in in vitro conditions.
Neutrophil mitochondria, although deficient in respiration, still play a role in restricting the apoptosis of neutrophils (Maianski et al. 2004). Thus, we further sought to quantify the blood levels of some representative mitochondrial DNAs. Our results indicate that GA relieves lung injury by decreasing the levels of mitochondrial DNAs. In sepsis, numerous inflammatory mediators and lipid metabolites are produced in the body, which promote the production of cytokines, reactive oxygen species (ROS), and proteases to amplify the inflammatory reaction. In sepsis-induced ALI, TNF-α, IL-1β, IL-6, and NO are the important pro-inflammatory cytokines and mediators, and their interactions and subsequent cascade activation promotes the progression of ALI. TNF-α is the initiation factor of the inflammatory cascade reaction in lung injury (Standiford et al. 1990; Martin et al. 1992). IL-1β enhances the recruitment of inflammatory cells into airspaces and alters vascular permeability, leading to fluid transport and subsequent lung edema formation (Pugin et al. 1996; Olman et al. 2002). Because IL-6 is mainly produced in the lung, the level of IL-6 is a good index to assess the early injury of the lung (Spooner et al. 1992). A study by Gultekin et al. (2007) showed that leptin treatment ameliorated acute lung injury in rats by decreasing the production of pro-inflammatory cytokines. In the present study, the numbers of leukocytes and the activity of MPO were lower in the GA treatment group, which illustrates the significant anti-inflammatory effect of GA. Furthermore, we found that GA caused a remarkable decline in the expression of inflammatory cytokines TNF-α, IL-1β, IL-6. NO is an inflammatory mediator produced by iNOS in pulmonary vessel endothelial cells. iNOS can be induced by various cytokines in pathological states (Gaston et al. 1994; Bloomfield et al. 1997). In our study, we found that GA could dose-dependently decrease the levels of NO and iNOS in rats with ALI. Iuvone et al. found that iNOS gene knockout rats showed reduced severity in lung injury as compared with control littermates, proving that the excessive NO produced by iNOS was an important mechanism in the pathogenesis of lung injury (Iuvone et al. 1996).
Next, we investigated the influence of GA on oxidative stress to further explore the mechanisms of GA alleviating ALI. Lipid peroxidation plays an important role in ALI (Tariq et al. 2003). During the course of ALI, neutrophil granulocytes release high levels of ROS through phagocytosis and discharging particles. The excessive ROS produces lipid peroxidation, of which the end-product is MDA. In our studies, MDA, SOD, 8-OhdG, and NT were measured to show the degree of oxidative stress damage. We found that GA could markedly inhibit the production and activity of these parameters. Others have also shown that GA can ameliorate oxidative stress in fructose-induced metabolic syndrome-X in rats (Sil et al. 2013). Furthermore, a study by Li et al. (2011) showed that GA has antioxidant and immune activity in mice with allergic rhinitis. Therefore, the antioxidant function of GA could alleviate the oxidative stress-mediated injury in lung.
Some recent studies have found that wound, infection, inflammatory mediators, and ROS cam induce apoptosis in pulmonary vascular endothelial cells and alveolar epithelial cells (Wang et al. 2013). Cell apoptosis in pulmonary tissues is involved in the development and progression of lung injury during sepsis (Chang et al. 2012). Research shows that the activation of caspase-3 plays an important role in the apoptosis of lung tissue cells. Caspase-3 is a key enzyme and regulatory protein in downstream apoptotic signaling pathways and is also critical in the cascade reaction of caspases. Most of the apoptosis is induced following the activation of caspase-3-mediated signaling pathways. Thus, caspase-3 acts as the final executor of apoptosis at the core of the apoptosis cascade. Bcl-2 and Bax are important regulatory factors of apoptosis among Bcl-2 family members (Dewson and Kluck 2009; Low et al. 2011). The ratio of Bcl-2/Bax determines the incidence of apoptosis. Bcl-2 can prevent activation of the pro-apoptotic protein Bax (Postolow et al. 2011), and studies have shown that excessive ROS can cause mitochondrial permeability transport pore opening and mitochondrial dysfunction, which leads to caspase-3 activity (Li et al. 1997; Schon and Manfredi 2003; Rigoulet et al. 2011). Our present findings show that GA can inhibit pulmonary vascular endothelial cell and alveolar epithelial cell apoptosis and changes in the expression of related proteins.
The process of inflammatory cell activation and inflammatory mediator release is regulated by nuclear transcription factors (Imanifooladi et al. 2010), proteins that bind specifically to promoters and enhancer nucleotide sequences to promote gene transcription. NF-κB is an important nuclear transcription factor widely distributed in the body, with a key role in cell proliferation, differentiation, adhesion, apoptosis, inflammation, and immunity (Ghosh et al. 1998). Several groups have shown that NF-κB is activated in response to sepsis, and that inhibiting NF-κB activity can alleviate the inflammatory reaction and improve the survival rate of experimental animals with sepsis (McCubrey et al. 2006; Imanifooladi et al. 2010; Xie et al. 2010). Mitogen-activated protein kinases (MAPKs)—a family of serine/threonine protein kinases—extensively participate in the regulation of signal transduction involved inn cell division, proliferation, motility, differentiation and apoptosis, among other pathways (Torii et al. 2006; Dhillon et al. 2007). p38 MAPK and JNK are two important family members that regulate the release of inflammatory mediators (Silvers et al. 2003). Lipopolysaccharide promotes the production and release of a large number of inflammatory mediators through the activation of MAPK, and this has been shown to be important in the onset and progression of ALI (Thobe et al. 2007; Asaduzzaman et al. 2008). In our studies, NF-κB, JNK, and p38 MAPK were all elevated in ALI, and GA inhibited their expressions, offering a protective effect against sepsis. We consider that NF-κB, JNK, and p38 MAPK might constitute an important molecular mechanism of GA activity in response to ALI; however, further research is needed to ascertain how the exact mechanism.
In conclusion, our results demonstrate that GA can alleviate sepsis-induced ALI, increase the survival rate of septic rats, and inhibit inflammatory responses, oxidative stress damage, and apoptosis in lung tissue induced by ALI via the inactivation of NF-κB, JNK, and p38 MAPK. Accordingly, we conclude that GA has the potential to be developed as a novel treatment for ameliorating the inflammation and pain associated with in sepsis-induced ALI.
Footnotes
Author Contributions
ZHY was responsible for the experimental design. ZHY and ZM contributed to the execution of experiments, data statistics, and manuscript composition. WY, LFC and ZZG participated in performing the experiment and in the manuscript preparation and submission. ZHY was responsible for the funding application and the supervision and management of the project. All authors have contributed to and approved the final manuscript.
Competing Interests
The authors declared no potential competing interests with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the National Key Clinical Specialties of Ministry of Health (No.: 2012-649).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
