Abstract
Objective
Carotid atherosclerosis, a major cause of ischemic cerebrovascular events, is characterized by a pro-inflammatory and pro-oxidant vascular microenvironment. The current risk score models based on traditional risk factors for cardiovascular risk assessment have some limitations. The identification of novel blood biomarkers could be useful to improve patient management. The aim of the study was to evaluate the association of selected inflammation- and oxidative stress-related markers with the presence of severe stenosis and/or vulnerable plaques.
Methods
Circulating levels of soluble CD40 ligand, interleukin-10, macrophage inflammatory protein (MIP)-1α, endoglin, CD163, CD14, E-selectin, tumor necrosis factor-α, monocyte chemoattractant protein-1, C-Reactive protein, CD40 L + T lymphocytes, total antioxidant capacity, glutathione reductase activity, and protein carbonyl content were determined in patients with carotid atherosclerosis.
Results
Multiparametric analysis showed significantly higher levels of MIP-1α in patients with stenosis ≥70% than in patients with stenosis <70%, and significantly higher levels of CD14 in patients with hypoechoic (vulnerable) lesions compared to those with hyperechoic (stable) ones. The area under the curve obtained by the receiver operating characteristic curve analysis was 0.7253 for MIP-1α and 0.6908 for CD14.
Conclusions
Our data suggest that circulating MIP-1α and CD14 levels are associated with the presence of advanced stenosis and of vulnerable carotid plaques.
Introduction
Carotid atherosclerosis is a leading cause of ischemic strokes. 1 Research studies have shown that traditional cardiovascular disease risk factors play a key role in the insurgence of this pathology. 2
Atherosclerosis is widely considered an inflammatory disease initiated by endothelial damage, 3 and once it started, different factors participate to atherosclerotic plaque development. The retention of low-density lipoproteins (LDL) in subendothelial space of the vascular wall is the first step that favors the formation of the atherosclerotic lesion. 4 Subsequently, the subendothelial LDLs are oxidized by the vascular cells, promoting a pro-inflammatory response characterized by the expression of chemokines and adhesion molecules, as well as the recruitment of inflammatory cells, in particular monocytes and T lymphocytes, within the intima of the vascular endothelium. Activated by the pro-inflammatory vascular microenvironment, these cells release cytokines and chemokines, which support the inflammatory process, and also produce pro-oxidant mediators such as reactive oxygen species (ROS), which cause endothelial cell damage and accelerate the atherosclerotic process. 5 Thus, a chronic vicious circle between inflammation and oxidative stress occurs, leading to the destabilization of atherosclerotic plaques. 6
Current risk score models rely on traditional risk factors, such as age, sex, smoking, blood pressure, diabetes, total cholesterol, high-density lipoproteins cholesterol, and family history. These models are valuable tools for the management of patients with cardiovascular disease; nevertheless, they have important limitations, particularly for patients with intermediate cardiovascular risk. 7 Measuring circulating levels of oxidative stress- and inflammation-related markers is of particular interest for improving cardiovascular risk assessment, especially in asymptomatic subjects. 8
Several molecules related to inflammation and endothelial dysfunction are currently under study as biomarkers for risk stratification 7 and different studies have shown that inflammation-related biomarkers are predictive of clinical events in atherosclerosis.9-11 The analysis of these types of markers is a field of great interest in order to identify new tools to improve the detection of vulnerable plaques.
The aim of the present study is to evaluate the possible association between a panel of selected inflammation- and oxidative stress-related mediators and the presence of severe stenosis and vulnerable plaques. For this purpose, we determined the concentrations of circulating factors in the peripheral blood of patients affected by carotid atherosclerosis. The following mediators were analyzed: soluble CD40 Ligand (sCD40 L), interleukin (IL)-10, macrophage inflammatory protein (MIP)-1α, endoglin, CD163, CD14, E-selectin, tumor necrosis factor (TNF)-α, monocyte chemoattractant protein (MCP)-1, C-Reactive Protein (C-RP), total antioxidant capacity of plasma, glutathione reductase activity, protein carbonyl content, and the percentages of CD40 L + T lymphocytes. We divided patients according to the degree of stenosis and plaque echogenicity. The possible association between these mediators and the baseline and clinical characteristics of patients was also considered in the analysis.
Methods
Subjects
Sixty-nine consecutive patients with carotid atherosclerosis (44 men and 25 women, median age 70, range 53–85) were recruited from 2011 to 2015 at the Department of Surgical Sciences, University of Rome “La Sapienza” to carry out a cross-sectional study with a multi-marker strategy. Exclusion criteria were recent infections (< 1 month), autoimmune diseases, malignancy, and inflammatory diseases before enrollment. Blood samples were obtained by venipuncture and processed within 1 hr from collection. Samples collected in non-heparinized tubes were centrifuged and sera were stored at −80°C until use. Samples collected in heparinized tubes were used to determine the percentage of surface CD40 L positive T cells.
Echo-color Doppler
Echo-color Doppler images to diagnose carotid atherosclerotic disease were acquired on an ultrasound system equipped with a 7.5 MHz frequency linear-array transducer. Fifty patients had severe carotid artery stenosis (≥70%) and 19 had asymptomatic moderate carotid stenosis (< 70%). In 61 patients, carotid plaques were classified according to their echogenicity: uniformly or predominantly (>50%) hypoechoic (vulnerable) plaques, and uniformly or predominantly (>50%) hyperechoic (stable) plaques. 12 Forty-three patients had hyperechoic plaque, and 18 patients had hypoechoic plaque.
Determination of circulating inflammation- and oxidative stress-related mediators
Serum concentrations of sCD40 L, IL-10, MIP-1α, endoglin, CD163, CD14, E-selectin, adiponectin, and C-RP were determined by commercially available enzyme linked immunosorbent assay (ELISA) kits (R&D Systems, Inc. Minneapolis, MN, USA; Cayman Chemical, Ann Arbor, MI, USA for C-RP only). Serum concentrations of TNF-α and MCP-1 were determined by commercially available ELISA sets (OptEIA set, BD Biosciences, San Jose, CA, USA). All ELISA assays were performed according to manufacturer’s instructions. Minimum levels of detection were 4.2 pg/ml for sCD40 L; <3.9 pg/ml for IL-10; <10 pg/ml for MIP-1α; 0.007 ng/ml for endoglin; 0.177 ng/ml for CD163; <125 pg/ml for CD14; 0.009 ng/ml for E-selectin; 0.246 ng/ml for adiponectin; 46.9 pg/ml for C-RP; and 3.9 pg/ml for TNF-α and MCP-1. Total antioxidant capacity of plasma, glutathione reductase activity, and protein carbonyl content were determined by commercially available assay kits (Cayman Chemical, Ann Arbor, MI, USA). Assay ranges were 0.044–0.330 mM for total antioxidant capacity, 20–255 nmol/min/ml for glutathione reductase activity, and 1–10 mg/ml for protein carbonyl content.
Determination of circulating CD40 L + T lymphocyte percentages
Surface CD40 L expression in circulating T lymphocytes was evaluated by flow cytometry. Whole blood samples were stained with peridinin chlorophyll protein (PerCP)-conjugated CD3 specific monoclonal antibody (MoAb) (BD Biosciences, San Jose, CA, USA) and phycoerythrin (PE)-conjugated CD40 L specific MoAb (BD Biosciences) for 30 minutes on ice in the dark and then treated with fluorescence-activated cell sorted (FACS) lysing solution (BD Biosciences) for 10 minutes. After washing, cells were fixed in 1% paraformaldehyde and analyzed by flow cytometry. PE-conjugated mouse IgG1 (BD Biosciences) was used as a control. The typical forward and side scatter gates for lymphocytes together with a CD3+ gate (logical gate) were set to exclude contaminating cells. Data were obtained on a FACScan flow cytometer and percentages of CD40 L + T cells were determined with CellQuest software (BD Biosciences).
Statistical analysis
We used non-parametric Mann–Whitney U test for non-Gaussian distributions of continuous data and Pearson chi square test or Fisher’s exact test for categorical data to evaluate the statistical significance between groups. We applied the receiver-operator characteristic (ROC) curves to evaluate the ability of mediators to discriminate between groups of patients. To assess the independent role on atherosclerosis risk of the considered molecules, a multivariate analysis was conducted by multiple logistic regression, all the factors significantly associated in the univariate analysis were included in the model, clinical characteristics were also included when confounders. Statistical analyses were performed using Stata 15; p values <0.05 were considered statistically significant.
Ethical committee approval
The study was approved by Sapienza University Ethical Committee N. 687/11, (approved in July 21, 2011), and conducted in accordance with the declaration of Helsinki. Informed written consent was obtained from all participants.
Results
Clinical characteristics and inflammation- and oxidative stress-related markers according to degree of stenosis
Univariate analysis of clinical characteristic distribution in patients divided according to the degree of stenosis or to plaque echogenicity.
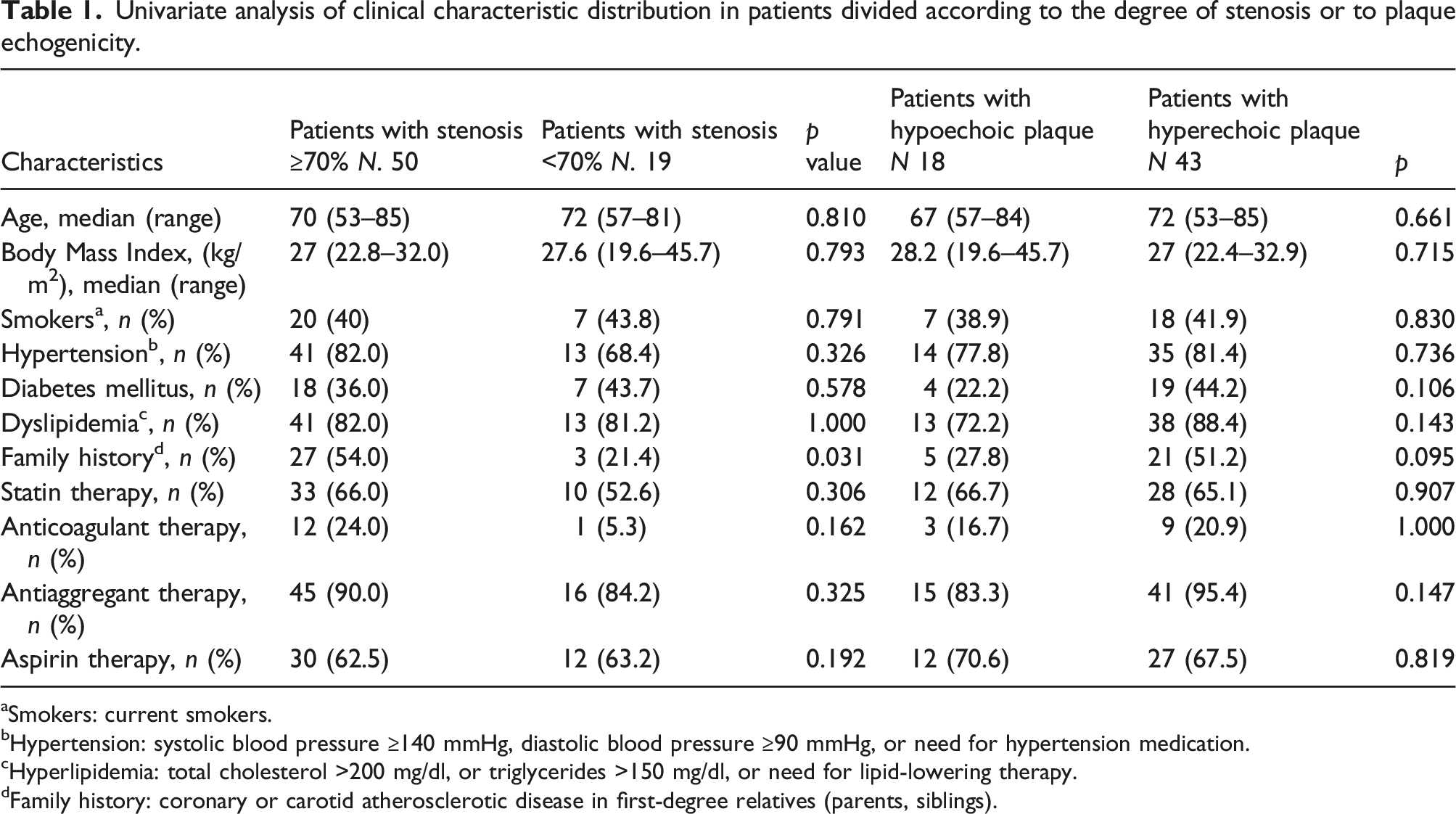
aSmokers: current smokers.
bHypertension: systolic blood pressure ≥140 mmHg, diastolic blood pressure ≥90 mmHg, or need for hypertension medication.
cHyperlipidemia: total cholesterol >200 mg/dl, or triglycerides >150 mg/dl, or need for lipid-lowering therapy.
dFamily history: coronary or carotid atherosclerotic disease in first-degree relatives (parents, siblings).
Univariate analysis of the relation between circulating marker levels and the degree of stenosis. Medians, interquartile ranges, and minimum and maximum values in patients divided according to the occurrence of severe (≥ 70%) or moderate (< 70%) carotid stenosis are reported. Statistical analysis by Mann–Whitney U test.

Multivariate analysis of the risk of stenosis ≥70% by circulating marker levels. Statistical analysis by multiple logistic regression.

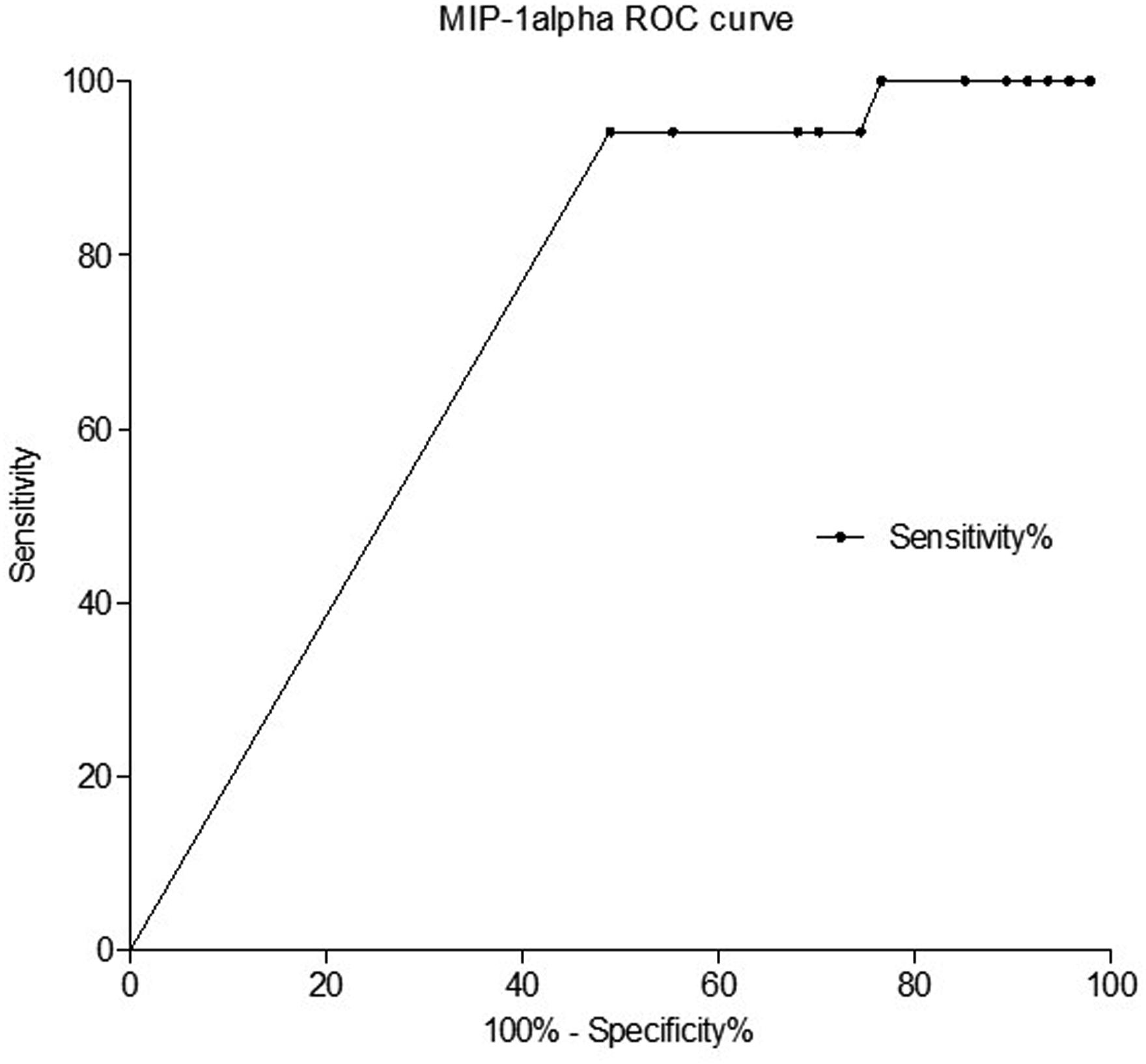
Receiver-operator characteristic (ROC) curve of MIP-1α. Analysis of MIP-1α in patients with stenosis
Clinical characteristics and inflammation- and oxidative stress-related markers according to plaque echogenicity
Univariate analysis of CD14 circulating levels according to plaque morphology. Medians, interquartile ranges, and minimum and maximum values in patients divided according to the presence of hypoechoic or hyperechoic plaques. Statistical analysis by Mann–Whitney U test.

Analysis of data did not reveal any significant difference for the other markers under study (data not shown). As the morphology of the plaque did not result association with other biological or clinical parameters by univariate analysis, no possible confounding factors are present and multivariate analysis was not performed. ROC curve analysis for CD14 circulating levels in the two groups of patients showed that the area under the ROC curve was 0.6908 (95% confidence interval: 0.5471 to 0.8345; p = 0.02804) (Figure 2). Receiver-operator characteristic (ROC) curve of CD14. Analysis of CD14 levels in patients with hypoechoic vs hyperechoic plaques. Area under the ROC curve: 0.6908; 95% confidence interval: 0.5471 to 0.8345; p = .02804.
Discussion
The results of the present study show significantly different levels of MIP-1α and CD14 in patients with carotid atherosclerosis in relation to the degree of stenosis and to the echogenicity of plaques, respectively, suggesting a possible association of these mediators with the presence of severe stenosis and of vulnerable plaques. In particular, our data show increased levels of the pro-inflammatory chemokine MIP-1α in patients with severe carotid stenosis, confirming the association between the pro-inflammatory status and the progression of carotid stenosis growth.
Chemokines play a key role in cardiovascular events as they coordinate inflammation and leukocyte recruitment. 13 In the past, several chemokines including MIP-1α have been detected within atherosclerotic lesions. 14
MIP-1α belongs to the CC chemokine family and is produced during infection or inflammation 15 ; it is released by activated platelets and promotes the recruitment of leukocytes in atherothrombosis. 16
Clinical studies have reported elevated serum concentrations of MIP-1α in patients with congestive heart failure. 17 Furthermore, patients with acute myocardial infarction and severe left ventricular dysfunction have increased concentrations of this chemokine when compared to healthy subjects. 18
Our data are in accordance with previous studies, demonstrating a positive correlation between the occurrence of myocardial infarction and MIP-1α levels and its independent association with the risk of short-term mortality in patients with acute coronary syndrome. These findings suggest the strong prognostic potential of this chemokine to identify high-risk patients.19,20
Our results on CD14 levels suggest the association between this biomarker and the composition of carotid lesions. Decisions on therapeutic intervention in patients with carotid atherosclerosis are mainly based on the evaluation of the severity of stenosis; however, this clinical management is not satisfactory. 21 Ultrasound analysis of atherosclerotic plaques provides useful information on their composition. Hypoechoic lesions are characterized by neovascularization and surface irregularities. They are more prone to clinical complications compared to hyperechoic plaques, characterized by smooth surface and extensively calcified areas. 22 Nevertheless, the imaging analysis of atherosclerotic lesions has some limitations.
The identification of high-risk plaques before they become symptomatic is a major milestone, although not yet definitively achieved. 22 In particular, it has been observed that some plaques undergo rupture without showing ultrasound characteristics of vulnerability. Moreover, some plaques with imaging features of vulnerability do not undergo rupture. 23
Standardization of ultrasound tissue characterization is difficult to achieve, as it is complex, operator-dependent, and affected by artifacts due to the different image settings. 22
Immune-regulated inflammatory pathways and oxidative processes have a pivotal role in the pathogenesis and progression of atherosclerosis. 24 The detection of inflammatory and oxidative stress-related markers in patients affected by atherosclerotic disease could be useful to ameliorate the identification of plaques at high risk of progression.
Previous clinical studies have evaluated the relationships between markers of inflammation and the characteristics of carotid plaques in terms of morphology and intima-media thickness (IMT) with important, even if not definitive, results.9,25
Our results demonstrate the association of CD14 serum levels with the presence of hypoechoic plaques and are therefore in accordance with previous studies reporting an association of this molecule with the severity and instability of atherosclerotic disease. 26
This marker is a molecule expressed on the surface of immune cells, particularly monocytes that play a pivotal role in the mechanisms responsible for the initiation, progression, and complication of atherosclerotic disease. In a recent study, Cocker et al. demonstrated that [18F]-fluorodeoxyglucose (18FDG) uptake analyzed with positron emission tomography was associated to symptomatic plaques. This marker is related to the presence of macrophage inflammatory cell activation and inflammatory leukocyte burden. 27 Our data on CD14 are in accordance with the observations by Cocker and colleagues.
The CD14 receptor is a monocyte activation marker and an increased density of membrane-bound CD14 has been reported in patients with acute coronary syndrome. 28
In another study, Lee et al. observed that urinary CD14 levels were higher in patients with coronary artery disease (CAD) compared to healthy controls, and were associated to the different extents of CAD. 26 Furthermore, a previous study performed in human carotid plaques demonstrated that intra-plaque CD14 expression was significantly increased in complicated plaques with signs of thrombosis. 29
Nevertheless, data on the role of CD14 as marker of cardiovascular risk are conflicting. In a case-control study, no differences were observed for plasma CD14 levels in patients with stable angina and controls, 30 and no association was determined between circulating levels of CD14 and CAD or IMT.30,31 A prospective study performed in a cohort of healthy men did not demonstrate any ability of CD14 to predict future coronary events. 32
The results of the present study, which show increased levels of CD14 in patients with hypoechoic plaques, sustain the association of circulating CD14 increased levels with atherosclerotic lesion instability, and support the possible usefulness of this monocyte antigen as biomarker for diagnostic screening.
Our results showing that different inflammation-related molecules are associated to degree of stenosis and echogenicity of the lesion are in accordance with previous studies, which demonstrate that IMT and plaque composition are associated to different atherosclerotic risk factors, suggesting that echogenicity and degree of stenosis contain different information.25,33
On this topic, a key aspect is the fact that clinical and experimental observations indicate IMT as a phenotype different from atherosclerosis. Studies based on meta-analysis showed that evaluation of IMT in the absence of plaque, and its progression or regression, are not reliable predictors of cardiovascular risk.34-37 Instead, information on plaque area proved a stronger predictor of risk than IMT. 38 Furthermore, although age and hypertension are main contributors to IMT, they are not necessarily signs of atherosclerosis development. 39
Intimal thickening is characterized by lipid deposition without the formation of a necrotic core, which forms due to macrophage infiltration, foam cell formation, and defective clearance of debris, and is considered an indicator of plaque development. Therefore, IMT is not able to distinguish between intimal thickening and the presence of a necrotic core. 39 Interestingly, IMT and plaque area are associated to different genetic determinants. 40 These observations indicate that IMT is biologically, genetically, and pathologically different from atherosclerosis and it has been proposed to refer to IMT as “arterial injury” or “arteriopathy”, instead of atherosclerosis. 41
Various ultrasound phenotypes reflect different mechanisms involved in the progression of atherosclerotic lesions. In particular, endothelial dysfunction, lipid accumulation, oxidative stress, and smooth muscle cell proliferation have been proposed as the main factors involved in determining total plaque area. 42 Instead, inflammation, reduction of matrix, and thrombosis are considered the factors most involved in the occurrence of stenosis and occlusion of the vessel lumen. 42
These observations suggest that different plaque phenotypes can reveal different biomarkers and this is a relevant aspect in the assessment of biomarkers for atherosclerosis progression.
This study has some important limitations that are worth taking into consideration when interpreting results. First, the low number of patients were recruited, in particular those with moderate carotid stenosis. Recruitment of these patients is usually more difficult, since this condition is asymptomatic and patients may carry out fewer medical checks. Second, the lack of significance observed for many markers could be related to the lack of power analysis due to sample size, that is, fewer observations than those needed. If determined in a larger population, it is plausible to assume that the distribution of some inflammation- or oxidative stress-related markers may be significantly different in patients divided according to the degree of stenosis or to plaque echolucency. Finally, another important limitation is the cross-sectional design of the study. This type of study does not allow to assess the causality of the associations of MIP-1α and CD14 with degree and morphology of the plaque and, more importantly, it does not allow to evaluate the predictive value of these molecules, which would be essential for determining the usefulness of these biomarkers in identifying patients at risk of clinical events.
Conclusions
In conclusion, the present study suggests that CD14 and MIP-1α circulating levels are possible markers associated with the occurrence of vulnerable plaques and severe stenosis in carotid atherosclerosis. Further studies conducted on larger populations are needed to confirm these results and should be considered longitudinal studies, in order to verify the usefulness in clinical practice of these biomarkers in combination with imaging techniques for patient monitoring.
Footnotes
Acknowledgments
We thank Dr. Cecilia Damiano for the careful linguistic revision of the manuscript.
Author contributions
EP: acquisition and interpretation of data, draft of the article; BB: acquisition of data and revision of the article; MET: statistical analysis of data; BS and RC: recruitment of subjects and sample collection; RR: design of the work and revision of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research of this article: this work was supported by the Italian Ministry of University and Research [Ateneo grants 2015, 2016, 2017 of “Sapienza” University of Rome].
Ethical approval
Ethical approval for this study was obtained from the Sapienza University Ethical Committee (N. 687/11).
Informed consent
Written informed consent was obtained from all subjects before the study.
