Abstract
Lung cancer in humans is majorly represented by non-small cell lung cancer cells, and there is a constant search for an efficient therapeutic approach. This study aims to find the anti-proliferative and apoptotic effects of matricin on H1299 cells via activation of MAPK pathway. Non-small cell H1299 cells were subjected to viability analysis with MTT assay and anti-proliferation analysis with different concentrations of matricin. Apoptosis was determined with annexin V/propidium iodide (PI) and flow cytometric analysis. Analysis of oxidative stress markers, reduced glutathione, lipid peroxidation (LPO), superoxide dismutase (SOD), and catalase (CAT) activities were done using standard assay kits. Apoptosis enzymes caspase-3, caspase-8, and caspase-9 levels were measured using colorimetric kit analysis. Western blot analysis on apoptotic proteins was performed to determine the involvement of MAPK pathway activation in apoptosis. Matricin significantly (P < 0.01) exerted anti-proliferative activities on H1299 cells in a dose-dependent manner. Flow cytometric apoptosis analysis showed increasing concentrations of matricin had increased apoptosis (P < 0.01) in the H1299 cells. Levels of oxidative stress markers were altered significantly (P < 0.01) by matricin. Caspase-3, caspase-8, and caspase-9 levels were significantly increased (P < 0.01) in matricin-treated H1299 cells. Western blot analysis showed decreased expression of anti-apoptotic Bcl-2, increased expressions of Bax and phosphorylated JNK, ERK 1/2, and p-38 MAPK proteins in matricin-treated H1299 cells. Matricin has significant anti-proliferative and apoptosis-inducing effects via activation of MAPK pathway in non-small cell lung cancer cells.
Introduction
Lung cancer is considered as one of the most deleterious malignancies worldwide. High percentage of death cases due to lung cancer is being reported every year throughout the world. 1 The percentage is increasing from the past few decades with no signs of recovery. Non-small cell lung cancer cells in individuals account for major lung cancer including large-cell carcinomas, adenocarcinomas, and squamous cell carcinomas. 2 Chemotherapy and radiotherapy treatments are still in practice for lung cancer treatment and patients do live a little longer. Unfortunately, the uncontrollable levels of side effects due to chemotherapy, vigorous progression of disease, intrinsic resistance to chemotherapy, and acquired resistance lead to death of patients. 3 Hence, there is need for a better therapeutic approach to be incorporated with the conventional treatment for lung cancer. Till date, there is a constant search for an efficient treatment for human lung cancer. Therapeutic agents from natural products are commonly approached in target of their effectiveness in promoting anti-proliferative effects in cancer cells. 4 Previously, it was confirmed that phytochemicals from plants are able to promote apoptosis in non-small cell lung cancer cells.5,6
Considering the effects of phytochemicals on cancer cells, matricin, a sesquiterpene compound that is majorly found in the essential oil extracts of chamomile plant (Matricaria recutita L.), was chosen to be studied for its anti-proliferative and apoptosis-inducing effects on non-small cell H1299 cancer cells. 7 Matricin is a promising phytocompound with anti-inflammatory, antioxidant, and anti-microbial properties and is described to possess abilities as a pro-drug in similarity with synthetic drugs.8–10 Moreover, sesquiterpenes have been reported to possess apoptosis-inducing properties along with anti-cancer effects on non-small cell lung cancer cells and are also known to exert only negligible amounts of toxicity.11,12 Recently, the anti-proliferative effects of matricin on human pancreatic carcinoma cells have been reported by Fang et al. 13 In this study, the potential of matricin to exert anti-proliferative and apoptosis-inducing effects on non-small cell H1299 lung cancer cells is reported.
Materials and methods
Cell culture and chemicals
Matricin and all analytical chemicals were obtained from Sigma Aldrich Inc., USA. Colorimetric assay kits for apoptosis detection, antibodies, immunoglobulins, and all western immunoblot assay kits were purchased from Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA. Non-small cell lung cancer cells H1299 and human bronchial epithelial cells Beas-2b were purchased from ATCC (Manasas, VA, USA). The cell lines were cultured in DMEM media containing 100 µg/mL streptomycin, 100 U/mL penicillin, 10% fetal bovine serum, and supplied with 5% CO2 at 37°C. The cultured cell lines were pre-treated with matricin (0–80 µM) in culture media to test the anti-proliferative and apoptosis-inducing effects. This study was conducted in the cell culture laboratory of Qingdao Hiser Medical Center from September 2018 for a period of 7 months.
Cell viability assay
Cytotoxicity of matricin was evaluated using MTT assay on the viability of H1299 and Beas-2b cells. The method was done following Zhang et al. 14 where cells were seeded in 96-well plates with 1 × 104 cells/well and treated with matricin for 24 h. Then, the medium was replaced with fresh MTT (0.5 mg/mL) in each well and incubated for 4 h. The solid formazan formed was dissolved in dimethyl sulfoxide (DMSO) and the absorbance readings were recorded at 570 nm using a microplate reader.
Annexin V/propidium iodide (PI) staining for apoptotic analysis
The H1299 cells were added at 2 × 105 cells/well overnight in 6-well plates, similar to the method of Lee et al. 6 The cells were treated with matricin (0–80 µM) for 24 h, harvested, and thoroughly rinsed with cold phosphate-buffered saline (PBS). Then, the cells were stained with annexin V and PI using FITC-annexin V apoptosis detection kit (BD Biosciences, CA, USA) following the manufacturer’s protocols and analyzed with flow cytometry (Calibur, BD Biosciences, USA).
Determination of GSH, LPO, SOD, and CAT levels
The levels of oxidative stress markers were determined using commercially available biochemical assay kits following the method of Santhanam et al. 15 All the results were recorded using UV-vis spectrophotometer. Briefly, glutathione (GSH) was determined by measuring the formation of 5-thiol-2-nitrobenzoic acid at 412 nm. Lipid peroxidation (LPO) was measured at 535 nm by thiobarbituric acid (TBA) conjugate formation with malondialdehyde (MDA). Activity of catalase (CAT) was determined by measuring decomposition rate of H2O2 at 270 nm, whereas activity of superoxide dismutase (SOD) was determined by measuring the inhibition rate of pyrogallol auto-oxidation at 470 nm.
Colorimetric assay to determine apoptosis enzyme levels
Colorimetric assay to determine caspase-3, caspase-8, and caspase-9 levels were performed with commercial kit for apoptosis detection (Roche Diagnostics, CA, USA) following the manufacturer’s protocols. The H1299 cells were pre-treated with matricin (0–80 µM) for 24 h, harvested, and thoroughly rinsed with cold PBS, subjected to colorimetric assay and measured using a UV-vis spectrophotometer following the method of Zhang et al. 16 The experiments were performed in at least three replicates.
Western blot analysis for MAPK pathway determination in apoptosis
The western blot analysis was performed according to the protocols of Min et al. 17 The H1299 cells were pre-treated with matricin (0–80 µM) for 24 h, harvested, and thoroughly rinsed with cold PBS, and centrifuged to obtain cell lysate proteins. Proteins were extracted in radioimmunoprecipitation assay (RIPA) buffer, and immunoblotting was performed using sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gel electrophoresis. The gels were transferred to polyvinylidene fluoride membranes, which were blocked with 5% bovine serum albumin for 2 h in Tris-buffered saline with Tween 20 (TBST) at room temperature. The membrane was incubated with primary antibodies overnight at 4°C. The immunoblots tested were antibodies for apoptosis marker proteins Bcl-2, Bax, and phosphorylation of MAPK pathway–related JNK, ERK 1/2, and p-38 proteins. The membrane was then incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies and TBST for another 1 h. The membrane was washed and positive immunoblots were developed using electrochemiluminescence (ECL) detection system and detected with enhanced chemiluminescence kit.
Statistical analysis
The results are represented as mean ± standard deviation for minimum of three replicates for each experiment. The statistical significance was determined using SPSS windows statistical package (SPSS 21.0, Chicago, USA) by performing one-way analysis of variance coupled with Tukey’s honestly significant difference (HSD) test. P values less than 0.05 (P < 0.05) and (P < 0.01) were statistically significant.
Results
Cytotoxic effect of matricin on H1299 and Beas-2b cells
Matricin (0–80 µM) was treated on non-small lung cancer H1299 cells and normal bronchiol epithelial Beas-2b cells for a period of 24 h. The results depict that matricin has significant cytotoxicity (P < 0.01) toward H1299 cells as the cell viability of H1299 were decreased at each time interval of 6, 12, 18, and 24 h (Figure 1). The IC50 of matricin was valued at 54 µM on H1299 cell viability at 24 h. Interestingly, matricin did not cause significant cytotoxicity on Beas-2b cells at the same time interval and concentration.

Cell viability and cytotoxicity induced by matricin on non-small cell lung cancer H1299 cells and normal bronchial Beas-2b cells at 6-, 12-, 18-, and 24-h time intervals. Data presented as mean and standard deviation of three replicates. Significance level *P < 0.05, **P < 0.01 compared with untreated control cells.
Apoptosis induced by matricin on H1299 cells
Annexin V/PI staining test was performed to detect apoptosis in matricin-treated H1299 cells. The flow cytometric results are shown in Figure 2. The results indicate matricin has induced significant apoptosis in H1299 cells, specifically at the highest dose 80 µM. The percentage of apoptotic cells were increased linearly with increasing dose of matricin. Highest percentage of apoptotic cells were 59.3% at 80 µM matricin treatment, compared with the untreated H1299 cells which showed 4.6% apoptosis.
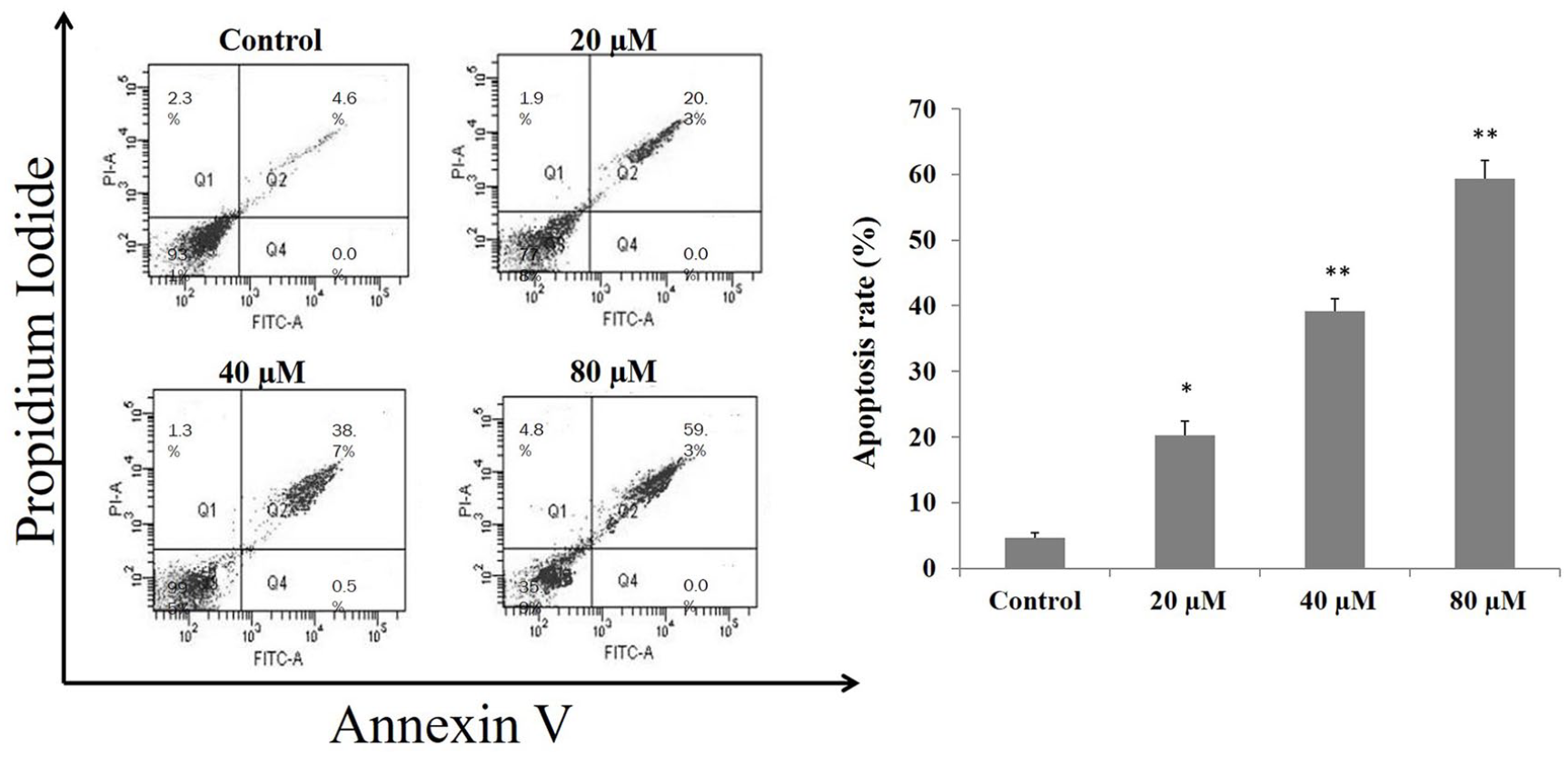
Annexin V/PI double staining flow cytometry results for apoptosis induced by matricin in H1299 cells in a dose-dependent manner. Data presented from at least three independent replicates. Significance level *P < 0.05, **P < 0.01 compared with untreated control cells.
Effect of matricin on oxidative stress markers in H1299 cells
Matricin-treated H1299 cells showed very mild difference in the oxidative stress profile compared with untreated control H1299 cells. Results given in Table 1 show there was a significant decrease (P < 0.01) in the levels of LPO and elevation in the levels of GSH, SOD, and CAT for highest dose of matricin as compared with untreated H1299 cells. The levels of oxidative stress markers in H1299 cells were significantly (P < 0.01) manipulated by matricin.
Effects of matricin on oxidative stress markers in H1299 cells.
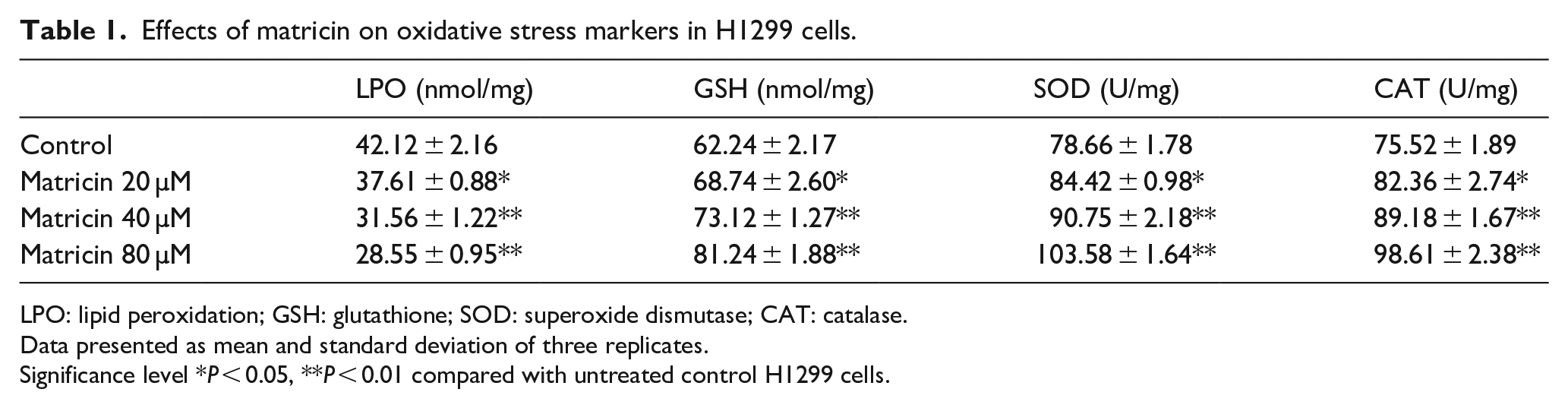
LPO: lipid peroxidation; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase.
Data presented as mean and standard deviation of three replicates.
Significance level *P < 0.05, **P < 0.01 compared with untreated control H1299 cells.
Matricin-induced apoptosis enzymes in H1299 cells
The effects of matricin treatment on apoptosis marker enzymes caspase-3, caspase-8, and caspase-9 in H1299 cells were observed and results are shown in Figure 3. Matricin significantly caused increase in the activities (P < 0.01) of apoptosis enzymes caspase-3, caspase-8, and caspase-9 in H1299 cells compared with untreated control H1299 cells. The activities of caspase-3, caspase-8, and caspase-9 were highest at the matricin treatment of 80 µM in H1299 cells compared with untreated control H1299 cells.

Matricin triggers the activities of apoptosis enzymes caspase-3, caspase-8, and caspase-9 with increasing dose concentration in H1299 cells. Data presented as mean and standard deviation of three replicates. Significance level *P < 0.05, **P < 0.01 compared with untreated control cells.
Expressions of apoptosis and MAPK pathway–related proteins
Western blot analysis results of Bcl-2, Bax, JNK, ERK 1/2, and p-38 proteins in matricin-treated H1299 cells are shown in Figure 4. From the results, it was found that the expression of anti-apoptosis protein Bcl-2 was decreased with increasing concentration of matricin treated in H1299 cells compared with untreated cells. In contrast, the pro-apoptosis protein Bax was highly expressed with increasing concentration of matricin as compared with untreated control H1299 cells. Protein expressions of MAPK pathway–related JNK, ERK 1/2, and p-38 in phosphorylated form were significantly increased in matricin-treated H1299 cells in a dose-dependent pattern compared with the untreated H1299 cells.

Western blot analysis results of pro-apoptotic Bax, anti-apoptotic Bcl-2, and phosphorylation of MAPK pathway–related JNK, ERK 1/2, and p-38 with relevance to matricin treatment on H1299 cells. β-actin was used as the internal loading standard. Significance level *P < 0.05, **P < 0.01 compared with untreated control cells.
Discussion
Phytochemical compounds have widely shown positive results for anti-cancer and anti-proliferative properties against malignancies and in vitro model of cancer cells.1,4,12 From the results of this study, it was found that matricin was able to cause cytotoxicity to non-small cell lung cancer H1299 cells in an increasing dose with time but viability of normal bronchial epithelial Beas-2b cells were not harmed by matricin. The effect of matricin on cell viability of non-neoplastic Beas-2b cells and the cytotoxicity caused in proliferation of H1299 cells can be related to the results of Singh et al. 18 The IC50 value of matricin in this study was to be found higher on H1299 cells as compared with the study pancreatic cancer cells, where matricin inhibited 50% of capan-2 cell viability at 20 µM. 13 Comparatively, other sesquiterpenes like parthenolide and costunolide exhibited IC50 values at 54.5 µM on A549 cell viability and 23.9 µM on H1299 cell viability, respectively.11,19 Apoptosis-inducing mechanism is one of the fundamental criteria related to anti-cancer properties of a compound, and matricin has significantly induced apoptosis in H1299 cells as proven in the flow cytometric analysis. The results show that matricin was able to induce apoptosis and cease proliferation of almost 60% of the H1299 cells at 80 µM concentration. The ability of matricin to induce apoptosis in H1299 cells is associated to the elevation of apoptosis marker enzymes, caspase-3, caspase-8, and caspase-9. These enzymes are well known as apoptosis inducers in cancer cells and they have interrelated functions. Colorimetric assay results on the activities of caspase-3, caspase-8, and caspase-9 enzymes exhibited that matricin significantly triggered the activation of these enzymes which can be related back to the flow cytometric apoptosis results. Moreover, these findings are in agreement with the results of Lee et al. 6 and Min et al. 17
Anti-apoptotic Bcl-2 and pro-apoptotic Bax proteins are regulators of apoptosis through activation of a cascade of events. Western blot analysis results showed increased expression of Bax in matricin-treated H1299 cells, whereas the Bcl-2 expression was reduced with increasing dose of matricin. These results indicate the similarity of apoptosis-inducing effects of natural compounds in lung cancer cells as reported by Czerwonka et al. 20 Defensive mechanism in cancer cells is associated with the inhibition of the apoptotic factors, hence blocking the antioxidant defense in cancer cells and triggering the apoptotic factors are possible using phytochemical compounds. 4 The levels of oxidative stress markers in H1299 cells were significantly altered by matricin, where antioxidant enzyme activities and GSH levels were elevated whereas LPO levels were reduced. Past results have proven the effectiveness of antioxidant compounds in anti-cancer mechanism.6,21 MAPK pathway activation is an important progenitor in anti-proliferation and advancement of apoptotic mechanism in cancer cells. 16 Oxidative stress helps to break the antioxidant defense of cancer cells and promote the apoptosis-inducing mechanism within the cell. 21 Increased expressions of phosphorylated JNK, ERK 1/2, and p-38 proteins with increasing concentration of matricin in the western blot analysis proved that matricin triggered apoptosis in H1299 cells through activation of MAPK pathway. The results of MAPK pathway activation are similarly described in the findings of Min et al. 17 Overall, matricin has shown significant anti-proliferative and apoptosis-inducing effects in non-small cell lung cancer H1299 cells. Limitations in this study are the detailed mechanism of apoptosis inducing and the specific small molecule markers responsible for apoptosis were not determined. Hence, a detailed in vivo study involving animal lung cancer model should be performed to delineate the mechanism of matricin on specific apoptosis molecular markers, compared with a commercially available apoptosis-inducing compound.
Conclusion
To conclude, this study shows matricin has significant cytotoxic effects on non-small cell lung cancer H1299 cells, and the apoptosis inducing might be partially induced by MAPK pathway and related apoptosis enzymes. These results support the use of matricin in a new therapeutic approach for human lung cancer treatment. Validation of matricin using an in vivo lung cancer model is needed to ascertain the bioavailability and pharmacokinetics of the compound for a successful clinical venture.
Footnotes
Author contributions
All authors share equal contribution for manuscript writing and editing. X.B. and W.W. collected and analyzed data; Y.W. analyzed data and performed statistical analysis; J.L. provided the experimental design and validated the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
