Abstract
Evaluation of the number of type II alveolar epithelial cells (AECs) is an important measure of the lung’s ability to produce surfactant. Immunohistochemical staining of these cells in lung tissue commonly uses antibodies directed against mature surfactant protein (SP)-C, which is regarded as a reliable SP marker of type II AECs in rodents. There has been no study demonstrating reliable markers for surfactant system maturation by immunohistochemistry in the fetal sheep lung despite being widely used as a model to study lung development. Here we examine staining of a panel of surfactant pro-proteins (pro–SP-B and pro–SP-C) and mature proteins (SP-B and SP-C) in the fetal sheep lung during late gestation in the saccular/alveolar phase of development (120, 130, and 140 days), with term being 150 ± 3 days, to identify the most reliable marker of surfactant producing cells in this species. Results from this study indicate that during late gestation, use of anti-SP-B antibodies in the sheep lung yields significantly higher cell counts in the alveolar epithelium than SP-C antibodies. Furthermore, this study highlights that mature SP-B antibodies are more reliable markers than SP-C antibodies to evaluate surfactant maturation in the fetal sheep lung by immunohistochemistry.
Fetal lung development begins in early gestation and undergoes a process of organogenesis and differentiation occurring across five phases: embryonic, pseudoglandular, canalicular, saccular, and alveolar (Harding and Bocking 2001). Expansion of peripheral airways occurs in the embryonic and pseudoglandular phases during early gestation. Thinning of the pulmonary mesenchyme and alveolar epithelial cell (AEC) differentiation occur in the canalicular phase. Maturation of the surfactant system occurs in the saccular phase over the latter stages of gestation and is an important step toward air breathing in extrauterine life (Flecknoe et al. 2003). Although alveolar development begins at ~30 weeks gestation and the lung is functionally mature at birth, it remains in the alveolar phase of development, and in humans, more than 90% of alveoli are formed during the first 3 years of life (Burri 1984).
The sheep fetus has been widely used as an animal model of human lung development to study both structural and molecular mechanisms regulating lung development due to its large body size, long gestation, and relative ease of surgical manipulation (Pringle 1986; Hooper et al. 1993; Maritz et al. 2001; Moss et al. 2002; Orgeig et al. 2010). The development of the sheep lung resembles that of the human lung much more closely than that of rodents because in sheep and humans, the lung spends similar total proportions of gestation in each phase of lung development (Pringle 1986; Lock et al. 2013). For example, in the case of the rat, there is an extended embryonic phase with relatively short periods of gestation spent in the remaining phases of lung development. Furthermore, alveolar development is significantly delayed, as true alveoli are not found in the rat lung until after birth (Burri 1984; Pringle 1986).
Pulmonary surfactant is a complex mixture of lipids and proteins, which primarily functions to reduce surface tension at the air-liquid interface and prevent alveolar collapse after birth (Pérez-Gil and Weaver 2010). This complex mixture of 90% lipids and 10% proteins is synthesized, stored, and secreted from type II AECs lining the alveoli. Surfactant protein (SP)-B and SP-C are synthesized as immature precursor proteins (pro–SP-B and pro–SP-C) that undergo a series of posttranslational modifications, including proteolytic cleavage in the intracellular machinery, including the endoplasmic reticulum, Golgi, and multivesicular body (Guttentag et al. 1998; Gobran and Rooney 2001; Pérez-Gil 2001; ten Brinke et al. 2002). Following this, mature SPs are packaged into lamellar bodies within type II AECs, where they are stored until secretion at the air-liquid interface (Ballard et al. 2003). The small hydrophobic SP-B and SP-C function cooperatively to promote insertion and spreading of surfactant phospholipids at the air-liquid interface to adequately permit exchange of respiratory gases and prevent alveolar collapse throughout the respiratory cycle (Pérez-Gil 2008). In addition, the large hydrophilic glycoproteins SP-A and SP-D play important roles in innate immunity within the lung (Kabha et al. 1997; Linke et al. 2001; Ofek et al. 2001; Kishore et al. 2006).
Maturation of the surfactant system occurs in late gestation and is vitally important for the transition to extrauterine life. In addition to molecular examination of surfactant components in the lung, electron microscopy and histological and, more specifically, immunohistochemical techniques provide important cellular and structural details about not only development of this vitally important system but also lung parenchymal development across gestation. Evaluation of the number of type II AECs in the alveolar epithelium is an important measure of the lungs’ ability to produce surfactant and is therefore commonly measured via immunohistochemistry targeting the surfactant proteins (Nogee et al. 1993a; Lin et al. 1996; Lin et al. 1999; Nogee et al. 2000; Fehrenbach 2001; Nogee et al. 2001; Sisson et al. 2010). The current gold standard for evaluation of type II AECs in lung tissue is electron microscopy, but this technique is expensive and laborious, and thus immunohistochemistry has typically used SP-C antibodies. For example, in mice and rats, SP-C has been regarded as the only specific SP marker of type II AECs as SP-B is expressed in its immature form in both type II cells in the alveolar epithelium and Club cells in the airways (Weaver 1998; Lin et al. 1999; Fehrenbach 2001). Moreover, as SP-A and SP-D have important immune functions, they are expressed more widely within the lung, for example, in type II AECs, Club cells, macrophages, and serous cells, as well as in many non-pulmonary tissues (Crouch 1998; McCormack 1998). Hence, their expression is not a useful indicator of type II AEC number. Despite the well-defined characterization of staining patterns for rodents, there has been no study demonstrating reliable markers for measuring maturation of the surfactant system via immunohistochemistry in the sheep. Thus far, the optimal procedure available for identifying type II AECs in sheep lungs is based on ultrastructural criteria with the presence of lamellar bodies, apical microvilli, cell-cell junctions, and cuboidal shape, which allows for the clearest distinction of the type II and type I AEC phenotypes (Flecknoe et al. 2000; Fehrenbach 2001; Flecknoe et al. 2002; Flecknoe et al. 2003). However, electron microscopy only allows the ability to assess the maturity rather than functionality of these cells. It is for this reason that immunohistochemistry is a more appropriate method to evaluate surfactant system development by identifying the functionality of cells within the lung capable of producing surfactant.
In this study, we examine the staining of a panel of surfactant pro-proteins (pro–SP-B and pro–SP-C) and mature proteins (SP-B and SP-C) in the fetal sheep lung at three time points during late gestation (120, 130, and 140 days; term, 150 ± 3 days), which corresponds to the overlapping saccular/alveolar phases (Pringle 1986), to identify the most reliable marker of mature surfactant producing cells in the lung of the sheep fetus.
Materials and Methods
Ethics and Animal Care
Experimental protocols for animal work were approved by the University of Adelaide and the University of South Australia/IMVS Animal Ethics Committees. Thirteen pregnant Merino ewes and their fetuses were used in this study. Ewes were provided with water ad libitum and fed once daily with a diet that was calculated to provide 100% of the energy requirements for the maintenance of a pregnant ewe bearing a singleton fetus, as specified by the Ministry of Agriculture, Fisheries and Food UK (Alderman et al. 1975).
Postmortem and Collection of Lung Tissue
Pregnant ewes were humanely killed at 120 (n=4), 130 (n=4), and 140 (n=5) days’ gestation (term, 150 ± 3 days) with an overdose of sodium pentobarbitone (Virbac Pty Ltd, Milperra, NSW, Australia). Fetal sheep were delivered by hysterotomy and body weights recorded. Fetal lungs were dissected and weighed; samples were immersion fixed in 4% paraformaldehyde and, following processing, were embedded in paraffin for subsequent immunohistochemical analysis.
Antibody Panel Selection
A panel of primary antibodies (Table 1) directed against surfactant pro-proteins (rabbit α-recombinant mouse pro–SP-C and rabbit α-human pro–SP-B [Seven Hills Bioreagents; Cincinnati Children’s Research Foundation, OH]) and mature surfactant proteins (rabbit α-human recombinant SP-C [Seven Hills Bioreagents], rabbit α-human mature SP-B [Seven Hills Bioreagents], and rabbit α-bovine mature SP-B [generously donated by Professor J. Whitsett, Cincinnati Children’s Research Foundation, Ohio]) were used in this study.
Primary Antibody Panel Selection for Use in Lung Tissue from Sheep Fetuses.

SP, surfactant protein.
Optimization of Final Diluted Antibody Concentrations for Use in Immunohistochemistry
Lung tissue sections of 5 µm thickness were cut from one block per animal, and serial sections were used for staining with each of the five aforementioned antibodies. Antibody staining patterns observed with all five antibodies were restricted to similar regions of the alveolar epithelium in all serial tissue sections. Lung tissue slides were baked at 60°C for 1 hr followed by deparaffinization and rehydration. After rehydration, endogenous peroxide solution activity was blocked with 3% hydrogen peroxide (Sigma-Aldrich; St. Louis, MO), followed by heat-induced antigen retrieval in citrate buffer (pH 6.0). Slides were incubated overnight with a primary antibody of interest at 4°C following incubation with non-immune serum (serum blocking solution; Histostain-Plus Kit; Invitrogen, Carlsbad, CA) to prevent nonspecific binding. Negative control slides with the primary antibody omitted were used to demonstrate no nonspecific binding of the secondary antibody or reagent contamination (Fig. 1C). In addition, negative control slides where incubation with primary antibody was substituted for rabbit serum (Sigma-Aldrich) at the same protein concentration as the diluted primary antibodies (1:500 and 1:1000) were carried out at each gestational age (Figs. 2, 3, 4A,B). This negative control is regarded as the proper negative control to demonstrate no nonspecific binding of immunoglobulins interacting with cell and tissue components or due to nonspecific mechanisms (Hewitt et al. 2014), thus highlighting primary antibody specificity in positive control slides in the alveolar epithelium observed in this study. Negative controls were incubated overnight at 4°C in parallel with test slides under the same experimental conditions. A Histostain-Plus kit (Invitrogen) was used with horseradish peroxidase and Histostain-Plus broad spectrum 3,3′-diaminobenzidine chromagen for visualization of the SP-positive cells. All sections were counterstained with Mayer’s hematoxylin (Sigma-Aldrich).
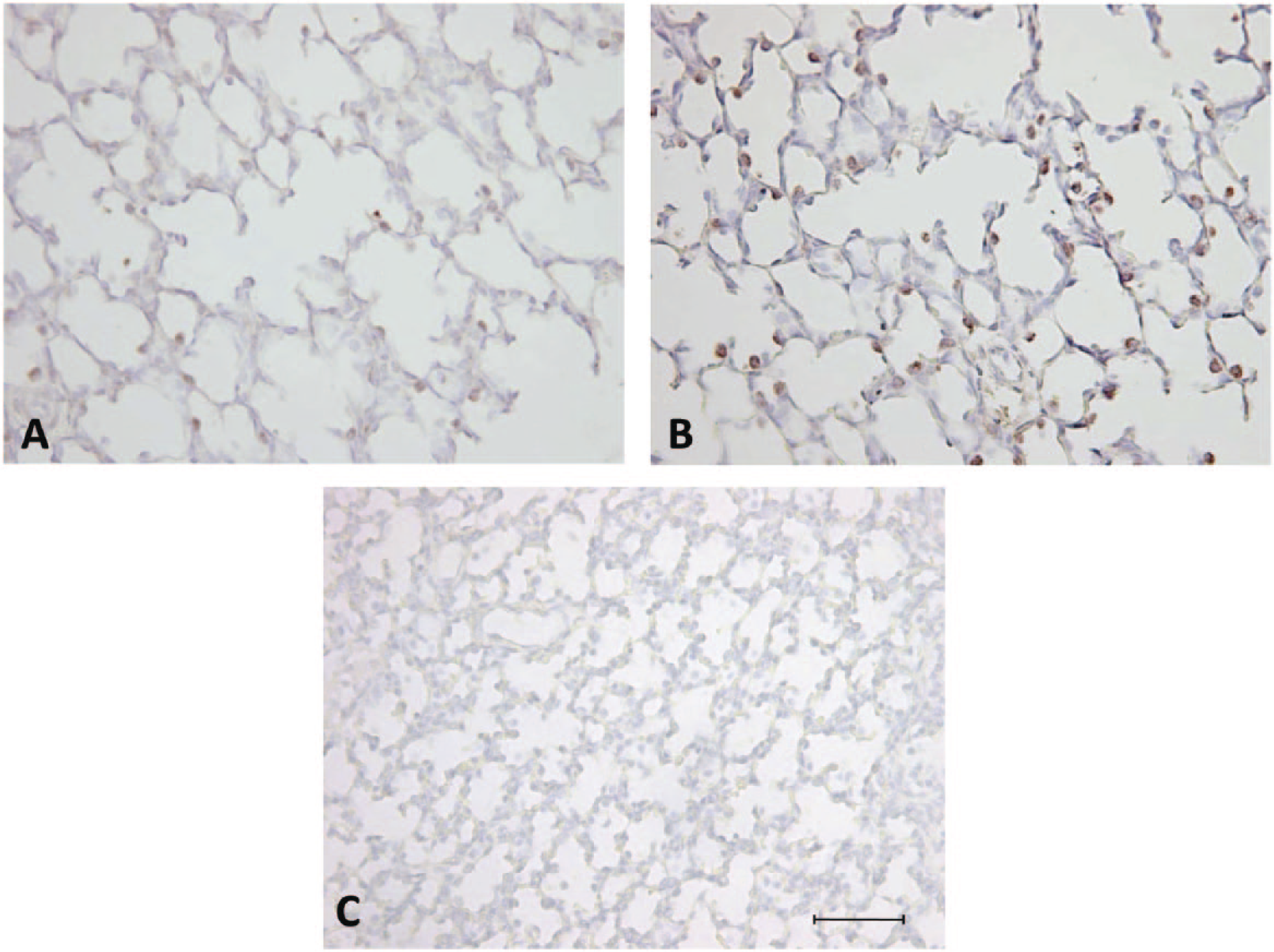
Antigen retrieval enhances primary antibody reactivity and increases staining of surfactant protein (SP)–positive cells identified in the alveolar epithelium in the fetal sheep lung. Micrographs of lung tissue from sheep fetuses stained at 140 days’ gestation using an antibody directed against anti-human mature SP-B without antigen retrieval (A) and anti-human mature SP-B with heat-induced antigen retrieval (B). No primary antibody negative control fetal lung tissue counterstained with Mayer’s hematoxylin (C). Scale bar = 50 µm.
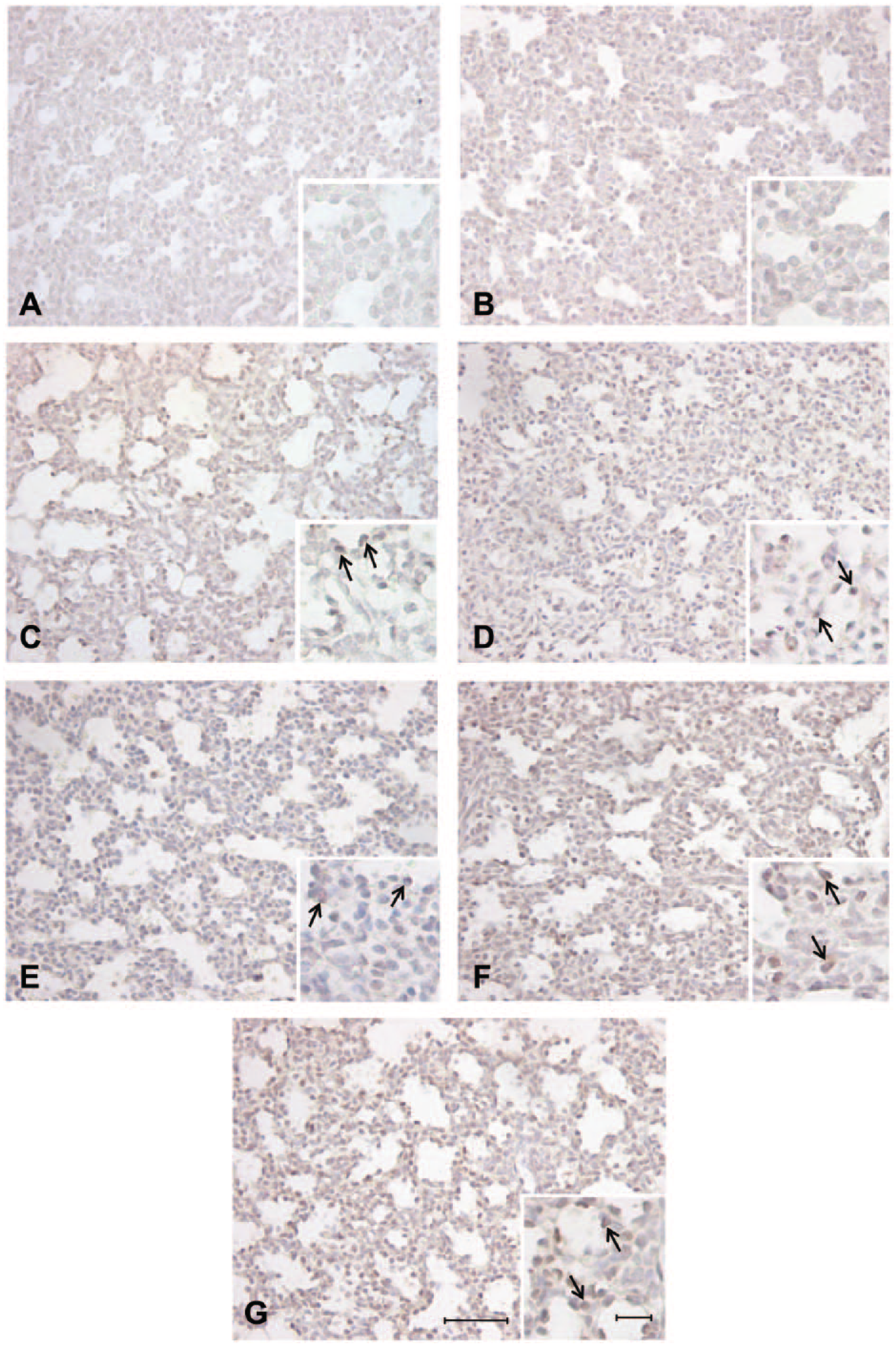
Positive staining in the alveolar epithelium observed in the fetal sheep lung at 120 days’ gestation identified with antibodies directed against anti-mouse pro–surfactant protein (SP)-C (C), anti-human pro–SP-B (D), anti-human mature SP-C (E), anti-human mature SP-B (F), and anti-bovine mature SP-B (G). Negative controls incubated with rabbit serum at 1:1000 (A) and 1:500 (B) demonstrating no nonspecific staining in the alveolar epithelium. Arrows = positively stained cells present in the alveolar epithelium. Scale bar = 50 µm and high-magnification insert scale bar = 10 µm.
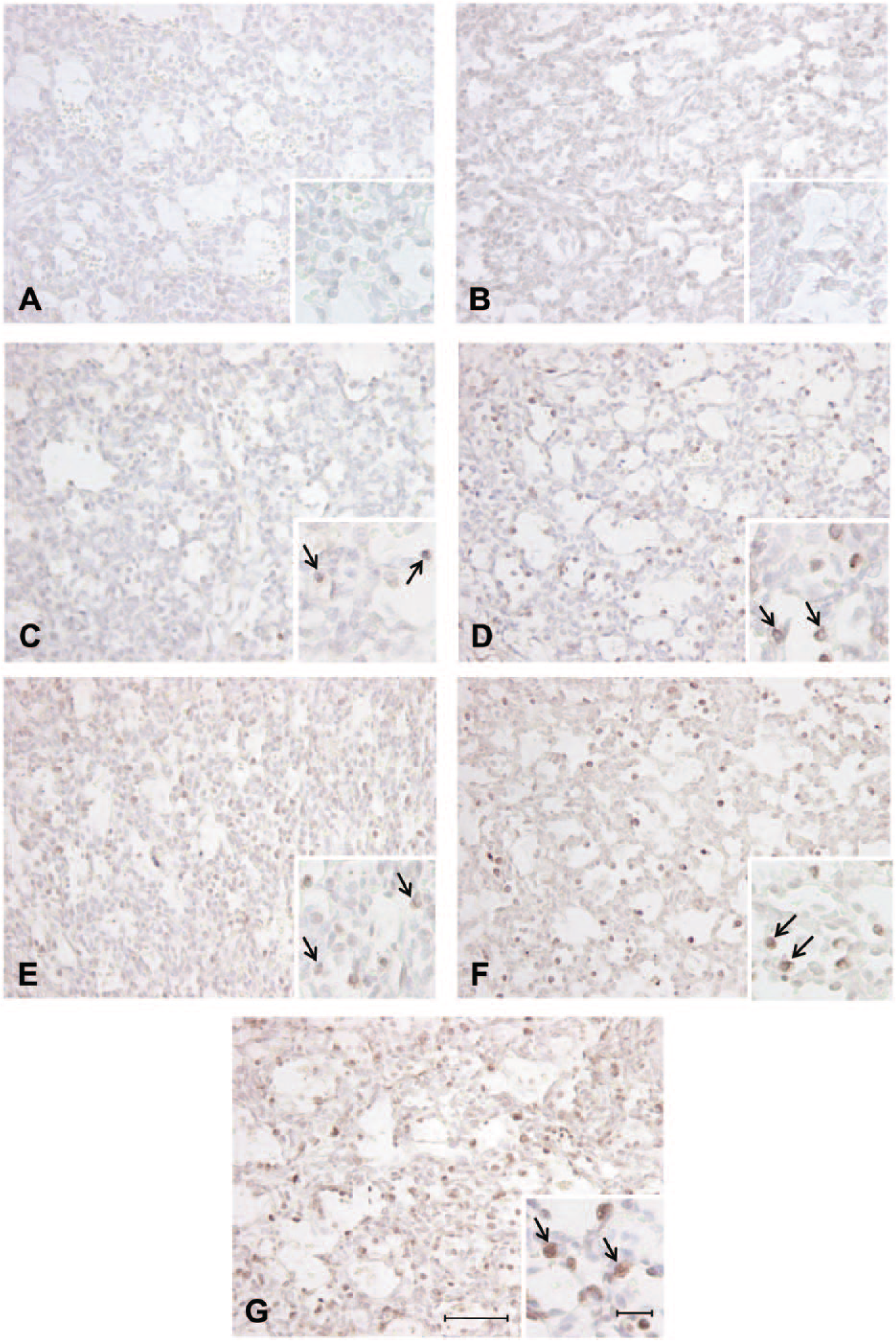
Positive staining in the alveolar epithelium observed in the fetal sheep lung at 130 days’ gestation identified with antibodies directed against anti-mouse pro–surfactant protein (SP)-C (C), anti-human pro–SP-B (D), anti-human mature SP-C (E), anti-human mature SP-B (F), and anti-bovine mature SP-B (G). Negative controls incubated with rabbit serum at 1:1000 (A) and 1:500 (B) demonstrating no nonspecific staining in the alveolar epithelium. Arrows = positively stained cells present in the alveolar epithelium. Scale bar = 50 µm and high-magnification insert scale bar = 10 µm.
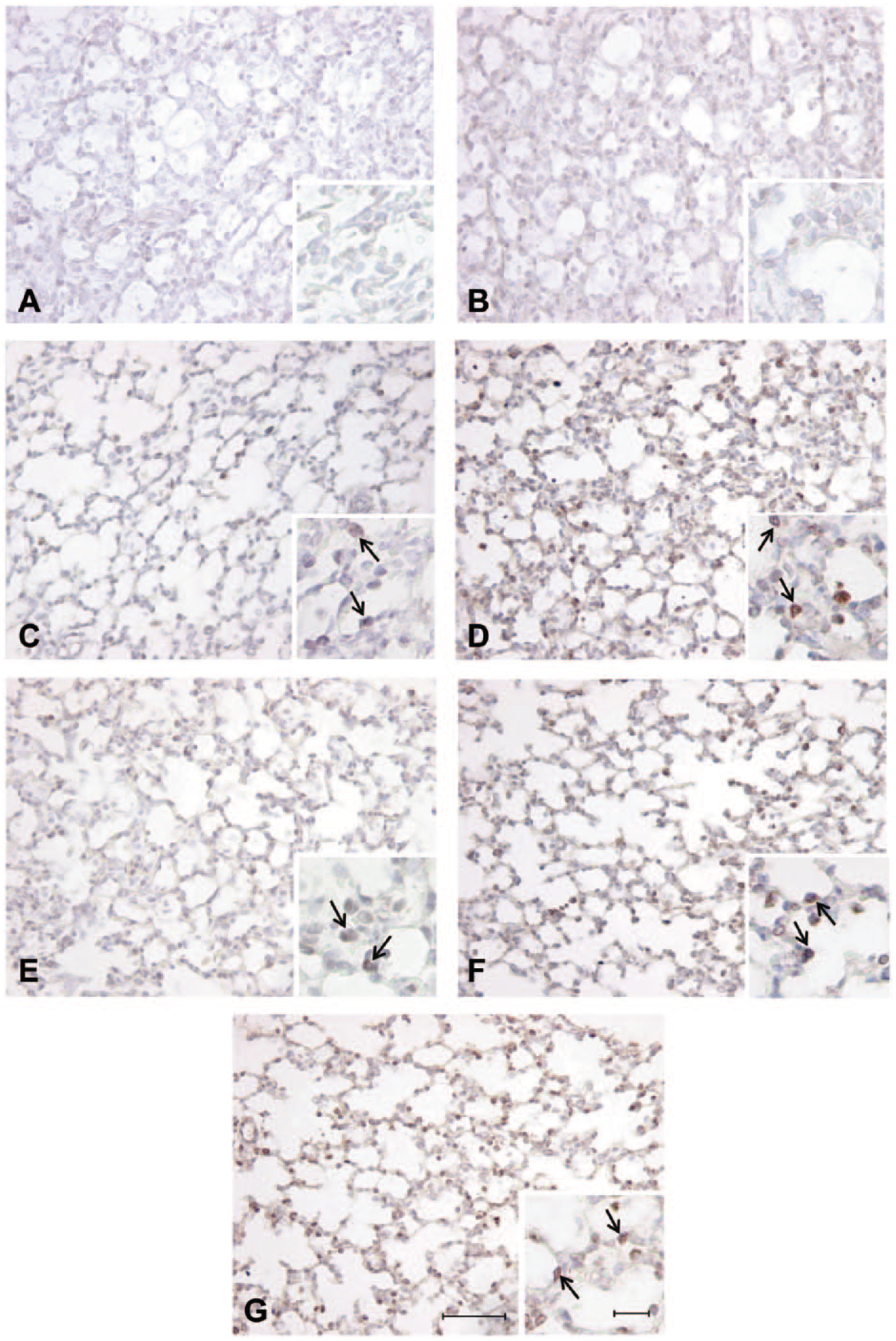
Positive staining in the alveolar epithelium observed in the fetal sheep lung at 140 days’ gestation identified with antibodies directed against anti-mouse pro–surfactant protein (SP)-C (C), anti-human pro–SP-B (D), anti-human mature SP-C (E), anti-human mature SP-B (F), and anti-bovine mature SP-B (G). Negative controls incubated with rabbit serum at 1:1000 (A) and 1:500 (B) demonstrating no nonspecific staining in the alveolar epithelium. Arrows = positively stained cells present in the alveolar epithelium. Scale bar = 50 µm and high-magnification insert scale bar = 10 µm.
Before use in immunohistochemistry on test samples, each final antibody concentration was optimized within the immunohistochemistry protocol (as above) by manipulating a range of test conditions. First, slides were subjected to either heat-induced antigen retrieval for 10 min in citrate buffer (pH 6.0) or did not undergo retrieval. This process was used to restore the reactivity of the epitope of interest to enhance binding with the primary antibody of interest. This method was found to enhance primary antibody reactivity with the tissue for all of the tested antibodies (Fig. 1). Furthermore, a range of antibody concentrations (1:500 to 1:3000) taking into account antibody supplier specification suggestions, in addition to substrate-chromagen reaction staining durations (4–10 min), were tested to give a desired primary antibody reactivity to produce a discernible brown precipitate that was easily distinguished from surrounding lung tissue when counterstained with Mayer’s hematoxylin.
Following final antibody optimization, immunohistochemistry for each of the five antibodies was performed on serial sections for each animal at each gestational age (final antibody concentrations listed in Table 1). Antigen retrieval was carried out for all slides for each antibody. Following optimization, the substrate-chromagen reaction was allowed to occur for the same time for all slides incubated with each individual antibody. Images of stained slides at each of the gestational ages were taken using an Olympus CX40RF200 microscope attached to an Olympus U-TV1X-2 camera (Olympus; Tokyo, Japan) to allow for visual localization comparison between antibodies on serial sections of lung tissue.
Quantification of SP Positively Staining Cells within the Fetal Lung Using Antibodies against Pro–SP and Mature SP
To evaluate the ability of the panel of antibodies to identify surfactant-producing cells within the fetal lung (during maturation of the surfactant system from late canalicular through saccular and alveolar phases of development; 120–140 days’ gestation), the numerical density of positively staining cells in the alveolar epithelium was determined by point counting as previously described (McGillick et al. 2013; McGillick et al. 2014). Characteristic positively staining cells were present within the alveolar epithelium of tissue sections and exhibited cytoplasmic staining (Fig. 5). In addition, the numerical density of positive cells using anti-human and anti-bovine mature SP-B was determined to test for specificity differences in staining of cells with each primary antibody. Briefly, sections were examined using Visiopharm Computer Assisted Stereological Toolbox (NewCAST) software (Visiopharm; Hoersholm, Denmark). Analysis was carried out by a trained individual who was blinded to treatment groups. Sixty counting frames (×600 magnification) of the alveolar epithelium were randomly selected per section. Point counting using an unbiased counting frame with an area of 20,000 µm2 was used to estimate the numerical density of SP-positive cells within the fetal lung sections. Using the four corners of the test frame, the reference space was estimated from the number of points falling on lung tissue in each field of view. Positively staining cells were identified by their cuboidal shape and location in the alveolar epithelium (Fig. 5). The numerical density of SP-positive cells per mm2 of lung tissue was obtained using the following equation (Bruel et al. 2005; McGillick et al. 2013; McGillick et al. 2014):

where ΣQ¯ (Surfactant positive) represents the total number of SP-positive cells counted in all counting frames of one fetal lung tissue section, and ΣP (Lung tissue) represents the total number of points falling on lung tissue in each field of view. P is the number of points that were used to count the points included within the reference space (four corners per counting frame), and
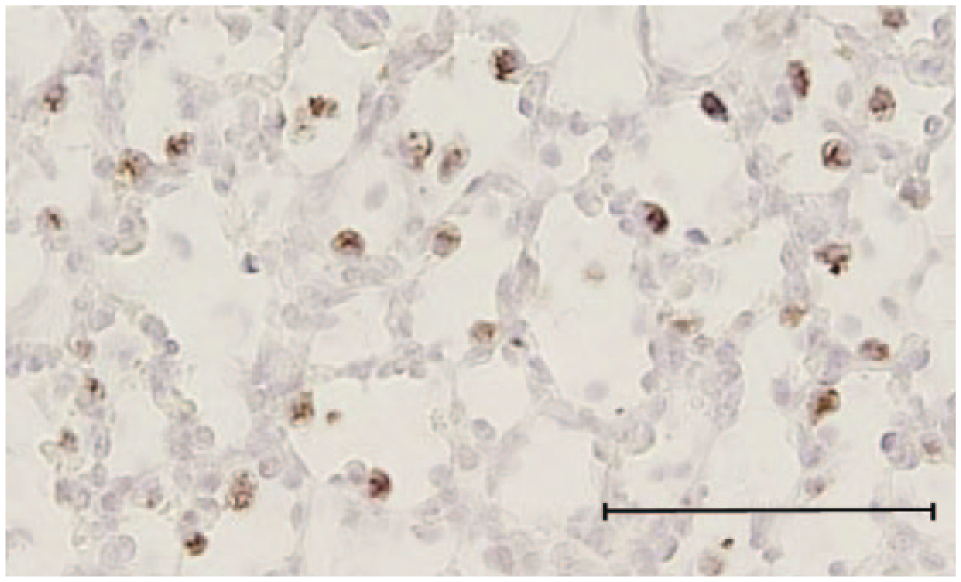
Intracellular staining with anti-human mature surfactant protein (SP)-B antibody in a lung sample from a 140-day sheep fetus. Micrograph showing typical positively stained cells present in the alveolar epithelium, demonstrating the ability to identify specific staining of epithelial cells. Scale bar = 70 µm.
Statistical Analyses
All data are presented as mean ± SEM. A probability level of 5% (p<0.05) was used as the significance level in all statistical tests. As serial sections for each animal were used for staining with anti-recombinant mouse pro–SP-C, anti-human mature SP-C, pro–SP-B, and mature SP-B antibodies, numerical density of positively stained cells present in the alveolar epithelium was analyzed using a two-way analysis of variance (ANOVA) with repeated measures by STATA 11 (StataCorp LP; College Station, TX), where the factors were gestational age and antibody (antibody was a repeated measure). Similarly, numerical density of positively stained cells identified by anti-human and bovine mature SP-B antibodies was analyzed using a two-way ANOVA with repeated measures where the factors were gestational age and antibody. When a significant interaction between major factors was identified, the data were split on the basis of the interacting factors and reanalyzed. Duncan’s new multiple-range post hoc test was used to identify significant differences between mean values.
Results
Comparison of Positive Staining of Cells in Fetal Sheep Lung from 120–140 Days’ Gestation Using a Panel of Pro- and Mature SP-B and SP-C Antibodies
At 120 days’ gestation, there was positive staining observed in the alveolar tissue where epithelial cell differentiation had occurred in the canalicular phase of development and surfactant maturation had begun at this gestational age, and thus staining of some SP-positive cells was observed at this gestational age (Fig. 2). At both 130 and 140 days’ gestation, there was also positive staining identified in the alveolar epithelium with all antibodies (Figs. 3 and 4). However, there was visually less positive staining observed within the alveolar epithelium using the pro- and mature SP-C directed antibodies (Figs. 2, 3, and 4C,E) compared with that of the pro–SP-B and mature SP-B antibodies (Figs. 2, 3, and 4D,F,G) observed from 120 to 140 days’ gestation.
Comparison of the Numerical Density of Positively Stained Cells in the Alveolar Epithelium Identified with Anti-Pro and Mature SP-B and SP-C Antibodies in the Fetal Sheep Lung from 120–140 Days’ Gestation
When comparing the positive staining of cells in the alveolar epithelium from 120 to 140 days’ gestation, there was a significant increase (p<0.05) with gestational age in the numerical density of cells identified with SP-B (both pro- and mature antibody) but not with SP-C (both pro- and mature antibody) directed antibodies. There was no difference in the numerical density of positively stained cells present in the alveolar epithelium identified with both the pro- and mature SP-C directed antibodies (Fig. 6). When comparing the numerical density of positive cells identified in the alveolar epithelium with SP-B directed antibodies, despite no difference at 120 and 140 days’ gestation, there was a significantly higher density of positive cells observed at 130 days’ gestation with the use of the mature SP-B antibody compared with the pro–SP-B antibody (p<0.05; Fig. 6). Thus, regardless of the age examined, mature SP-B identified the most positively staining cells in the alveolar epithelium in the fetal sheep lung from 120 to 140 days’ gestation.
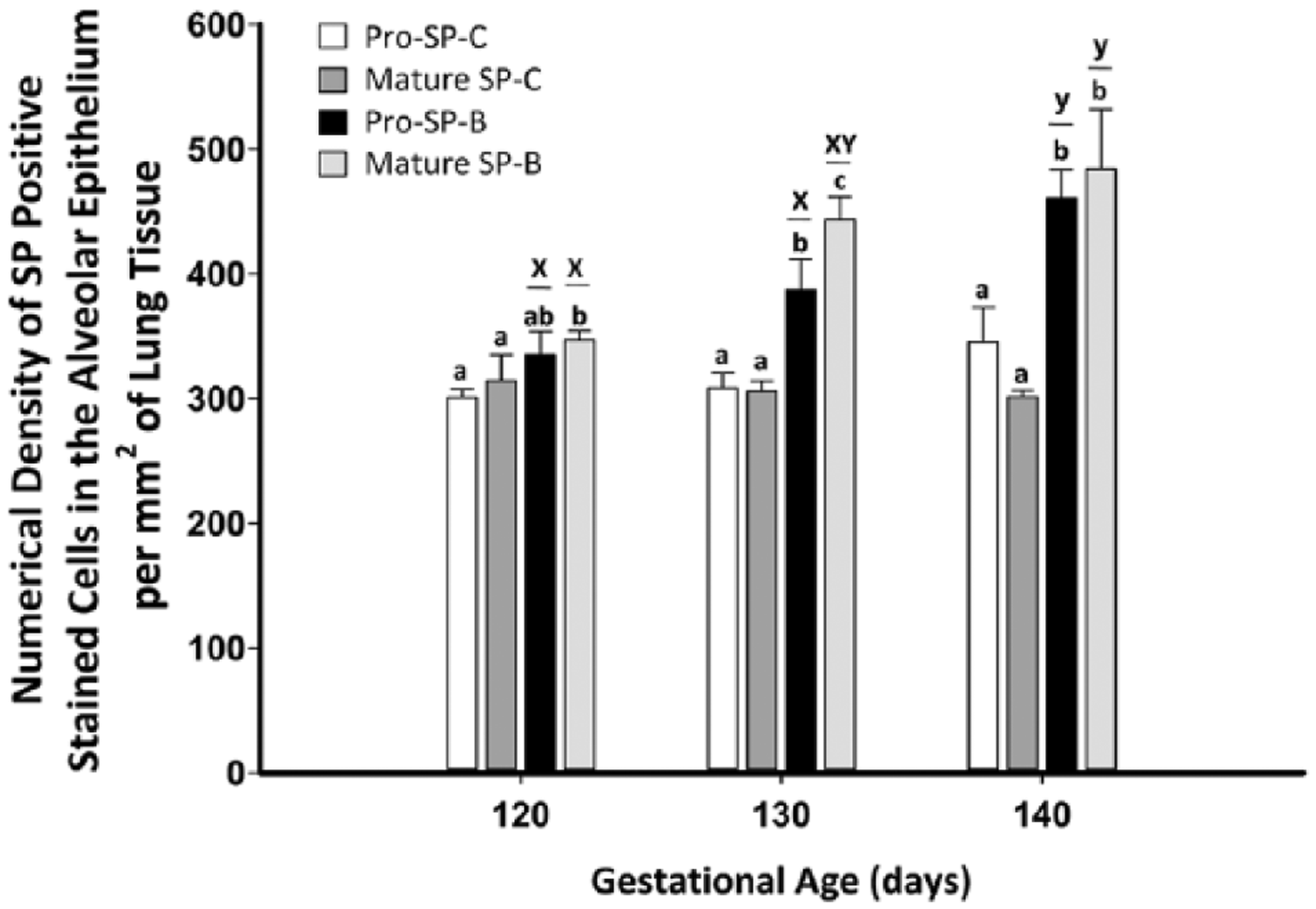
The numerical density of positively stained cells in the alveolar epithelium increased from 120 to 140 days’ gestation as expected when using anti-pro and mature surfactant protein (SP)-B directed antibodies (as indicated by the letters x and y), but the density of positive cells was unchanged between 120 and 140 days when using anti-pro- and mature SP-C directed antibodies. At 120 to 140 days’ gestation, pro- and mature SP-B directed antibodies detected the increase in the numerical density of SP-positive cells in comparison to SP-C directed antibodies (as indicated by the letters a, b, and c). However, the use of a mature SP-B antibody reliably detected the most positively stained cells present in the alveolar epithelium from 120 to 140 days’ gestation. Data are expressed as mean ± SEM. Data were analyzed by two-way analysis of variance (ANOVA) with repeated measures (p<0.05); different letters between bars indicate significant differences; x and y, effect of gestational age for each antibody (one-way ANOVA); a and b, effect of antibody at each age (one-way ANOVA).
Comparison of Staining with Anti-Bovine and Anti-Human Mature SP-B directed Antibodies to Identify Surfactant Positive Cells Present within the Alveolar Epithelium from 120–140 Days’ Gestation in the Fetal Sheep Lung
There was a significant increase in the numerical density of positively staining cells identified with anti-mature SP-B antibodies between 120 and 140 days’ gestation, independent of the specificity of the antibody used to detect mature SP-B expression, that is, anti-human versus anti-bovine SP-B (p<0.05; Fig. 7). However, there was no difference in the numerical density of SP-B–positive cells identified in the alveolar epithelium detected by either mature SP-B directed antibody at each individual gestational age.
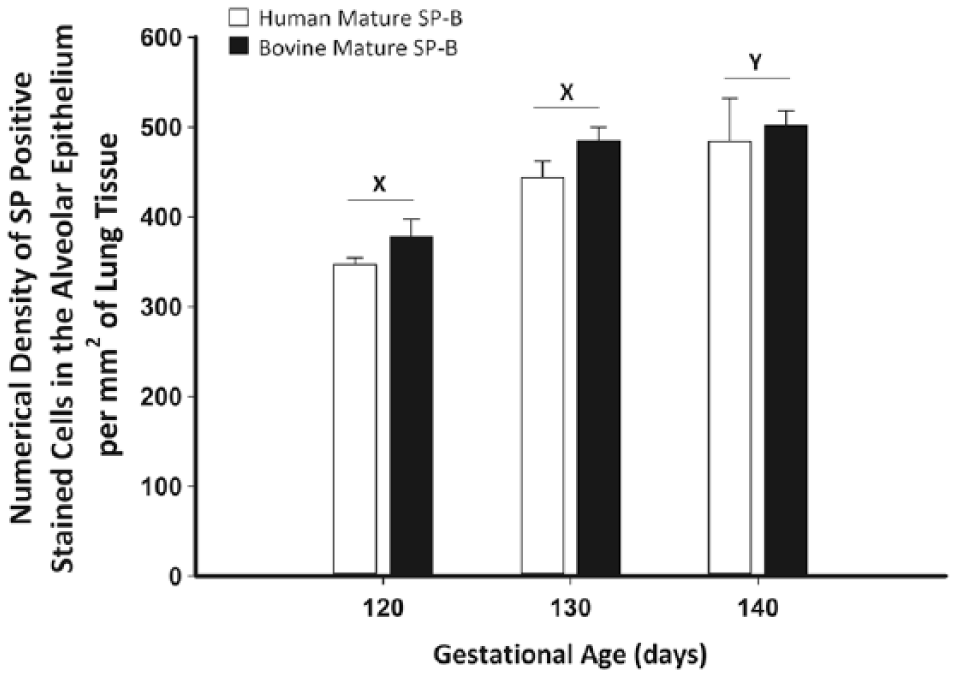
There was no difference in the numerical density of positively stained cells present in the alveolar epithelium at each gestational age between the anti-human mature surfactant protein (SP)-B antibody (open bars) and rabbit anti-bovine mature SP-B antibody (closed bars). There was, however, an increase in the number of positively stained cells identified in the alveolar epithelium from 120 to 140 days’ gestation (as indicated by the letters x and y). Data are expressed as mean ± SEM. Data were analyzed by analysis of variance (ANOVA) with repeated measures (p<0.05). Different letters indicate significant differences; x and y represent the effect of gestational age.
Discussion
With the goal of identifying functional type II AECs in the sheep fetus, we have demonstrated staining with pro- and mature SP-B and SP-C antibodies in the alveolar epithelium of the lung in late gestation. Furthermore, we have quantified positive staining of cells in the alveolar epithelium from 120 to 140 days’ gestation, during the saccular/alveolar phase of lung development, and have identified mature SP-B directed antibodies as the most reliable marker of surfactant system maturation in the fetal sheep lung.
SP expression begins in undifferentiated epithelial cells in the bronchiolar epithelium, before migration and differentiation of type II AECs to the distal parts of the lung (Khoor et al. 1997). In previous studies, SP-C has been used widely as a reliable marker for identifying SP-positive cells as an indicator of maturation on the surfactant system in rodent lungs (Fehrenbach 2001; Nogee et al. 2001; Sisson et al. 2010) as, unlike SP-B, SP-C is specifically expressed in type II AECs (Kalina et al. 1992; Kasper and Singh 1995; Weaver 1998; Lin et al. 1999). Although the SP-C precursor has previously been detected in cuboidal cells lining the bronchioloalveolar portals in newborn infants (Khoor et al. 1994), these cells are thought to be precursors of cells lining the terminal airways and differentiate into mature type II AECs or into type I AECs at the alveolar-capillary interface (Iwatsuki et al. 1993; Khoor et al. 1994). It has also been previously demonstrated that murine Club cells are not capable of processing pro–SP-B to its mature peptide (Lin et al. 1999) and that staining of mature SP-B was rarely seen in the bronchiolar epithelium in human fetal lung tissue (Khoor et al. 1994).
Evaluation of surfactant-producing cells in the alveolar epithelium is an important measure of the lung’s ability to produce surfactant. In this study, we were able to evaluate for the first time the staining of positive cells from 120 to 140 days’ gestation using a panel of both pro- and mature SPs to identify the most reliable marker in the late-gestation fetal sheep lung by immunohistochemistry. Despite the limitation that the lung tissue was immersion not instillation fixed, this series of experiments was standardized across late gestation. Another consideration is the natural decrease in tissue density with advancing gestational age as we examined lung tissue from 120 to 140 days. We normalized for this as the cell-counting data are expressed as numerical density of SP-positive cells per mm2 of lung tissue using an equation that takes into account the number of points falling on lung tissue for each counting frame placed over the fetal tissue sample. This therefore provides an internal correction for any change in tissue density between age groups evaluated. Pro-SP antibody staining was examined because it was suggested that the principally intracellular nature of these proteins would be a more reliable marker to identify surfactant-producing cells compared with mature SPs. Mature SP-B and SP-C are secreted from type II AECs during late gestation, and thus it is possible that some airway lumen and parenchymal staining observed with the anti-mature SP-B and-C antibodies could be due to extracellular staining following secretion to the air-liquid interface. However, this was rarely identified and could be distinguished readily from intracellular specific staining of epithelial cells, thereby yielding an accurate numerical density of positively staining cells in the alveolar epithelium from 120 to 140 days’ gestation. Between 120 and 140 days’ gestation in the fetal sheep lung, a similar numerical density of positively stained cells was identified between the pro- and mature SP directed antibodies for both SP-B and SP-C. However, the SP-B directed antibodies were more reliably able to identify positively staining cells and their natural increase with gestational age in the alveolar epithelium from 120 to 140 days’ gestation than SP-C directed antibodies. This was a surprising result as both SP-B and SP-C are highly conserved among mammals (Foot et al. 2007; Potter et al. 2007), and in mice, SP-C is regarded as a reliable marker for type II AECs, where it is used extensively in studies of embryological development to identify early lung buds (Wert et al. 1993; Hyatt et al. 2007).
Furthermore, the presence of intermediate AECs, characterized by distinct features of both type I and type II AECs, has been identified within the fetal sheep lung throughout development (Flecknoe et al. 2002; Flecknoe et al. 2003). Although little information exists on surfactant protein expression of this intermediate AEC, it has been suggested that the presence of these cells may complicate the ability to quantify the number of surfactant-producing cells because they contain lamellar bodies, which may be capable of secreting surfactant proteins. However, the proportion of AECs of this intermediate phenotype remains low throughout gestation (0.1%–6%) in the fetal sheep (Flecknoe et al. 2003). Moreover, there is no significant difference in the number of these cells during maturation of the surfactant system from 120 to 140 days’ gestation, the period that is relevant to the current study (Flecknoe et al. 2002; Flecknoe et al. 2003). Thus, any change in the numerical density of cells identified in late gestation is unlikely to be influenced by this intermediate cell population. Furthermore, our aim was to determine the numerical density of cells capable of producing surfactant, and thus any contribution by these intermediate cells does not detract from our findings. If further characterization of type II AEC differentiation and phenotype was to be of particular interest, an additional marker such as ATP-binding cassette subfamily A member 3 (ABCA3) may be targeted. This has an important role in normal surfactant metabolism and packaging of surfactant lipids within lamellar bodies, the cytoplasmic inclusion bodies within type II AECs responsible for surfactant storage and secretion (Ban et al. 2007). ABCA3 expression has been identified in both the mouse and human lung and is restricted to the cuboidal epithelium in the late canalicular phase of development and is a robust marker of differentiated type II AECs in the adult lung (Stahlman et al. 2007). However, unlike staining observed with the use of some surfactant proteins, it is not expressed within the bronchiolar epithelium (Weaver 1998; Stahlman et al. 2007). It has been demonstrated that dual immunolocalization staining with ABCA3 and SP-B is associated with staining within adult mouse type II cells (Stahlman et al. 2007); thus, in the context of the current study, it would be reasonable to expect similar staining and quantification of positive cells between the use of this marker and an SP-B antibody.
As a mature SP-B directed antibody allowed detection of the most positively staining cells in the alveolar epithelium regardless of the age examined between 120 and 140 days’ gestation, we proceeded to determine if the specificity of the antibody used influenced the ability to detect these positive staining cells. We used both an anti-human and anti-bovine directed mature SP-B antibody because evolutionarily, the sheep (Ovis aries) is more closely related to the cow (Bos taurus) than to humans (Foot et al. 2006). Despite the different origin of the antibody target, the species specificity of the mature SP-B antibody used did not affect the ability to assess maturation of the surfactant system by quantifying the numerical density of positively stained cells in the alveolar epithelium from 120 to 140 days’ gestation in the fetal sheep lung.
The numerical density results indicate that despite the same criteria used to identify positive cells, the use of either the anti–pro-SP-C or anti-mature SP-C antibody underestimates the numerical density of measurable SP-positive stained cells present in the alveolar epithelium in the fetal sheep lung from 120 to 140 days’ gestation. The increase in numerical density of positively stained cells observed in this study using SP-B antibodies and no change observed with SP-C antibodies in late gestation is consistent with a previous study of SP mRNAs in the fetal sheep lung, where expression of SP-C plateaued from 138 to 142 days’ gestation and 2 weeks after birth, whereas SP-B mRNA expression increased between 111 and 138 days, peaking at 142 days’ gestation (Flecknoe et al. 2003). This is consistent with the importance of SP-B for surfactant packaging and function in the lung at birth, because loss of SP-B is lethal at birth (Nogee et al. 1993b; Clark et al. 1995), being required for surfactant function, as well as processing of pro–SP-C (Vorbroker et al. 1995).
Despite no significant difference in the estimates obtained for numerical density of positively staining cells when using the anti-human mature SP-B and the anti-bovine mature SP-B directed antibodies, there was a reduced numerical density of positively stained cells identified with the anti–pro-SP-B antibody at 130 days’ gestation relative to the anti-mature SP-B antibody (Fig. 6). For this reason, the similarities in the data obtained with use of the mature SP-B antibodies suggest that they are acceptable and preferable for use to determine the density of type II AECs in the alveolar epithelium of fetal sheep lungs during the saccular/alveolar phase of development from 120 to 140 days’ gestation relative to pro- and mature SP-C directed antibodies.
In the literature, both SP-B and SP-C have been widely used as a marker of type II AECs in rodent lungs (Lin et al. 1999; Fehrenbach 2001; Sisson et al. 2010), but in this study, in comparison to antibodies directed against SP-C, the anti-mature SP-B antibodies stained the cells of the alveolar epithelium of fetal lung tissue more reliably, resulting in significantly higher positive cell counts. Furthermore, the specificity of the mature SP-B antibody used in this study (anti-human and anti-bovine mature SP-B) demonstrated no significant difference between the numerical densities obtained from positive cells present in the alveolar epithelium, thus indicating that either anti-human or anti-bovine mature SP-B antibodies are reliable markers to be used to evaluate surfactant system development in the fetal sheep lung in late gestation.
Footnotes
Acknowledgements
We acknowledge the assistance of Anne Jurisevic, Laura O’Carroll, and Stacey Holman for participating in the collection of lung tissue.
Author Contributions
MCL, EVM, SO, and JLM were responsible for the conception and design of the experiments. MCL, EVM, SO, ICM, and JLM were each involved in data acquisition. MCL, EVM, SO, SZ, and JLM were involved in analysis and interpretation of the data. MCL, EVM, SO, SZ, ICM, and JLM drafted the article and all authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The animal component of the work was funded by NHMRC Project Grant 456418 (ICM/JLM). The molecular component of the work was funded by NHMRC Project Grant APP1030853 (JLM/SO). JLM was funded by a South Australian Cardiovascular Research Network Fellowship (CR10A4988) and a NHMRC Career Development Fellowship (APP1066916).
