Abstract
Surfactant proteins in different glandular structures of the oral cavity display antimicrobial activity for protection of invading microorganisms. Moreover, they are involved in lowering liquid tension in fluids and facilitate secretion flows. Numerous investigations for studying the occurrence of surfactant proteins in glandular tissues were performed using different methods. In the oral cavity, minor salivary glands secrete saliva continuously for the maintenance of a healthy oral environment. For the first time, we could show that infantile labial glands show expression of the surfactant proteins (SP) SP-A, SP-B, SP-C, and SP-D in acinar cells and the duct system in different intensities. The stratified squamous epithelium of the oral mucosa revealed positive staining for SPs in various cell layers.
Introduction
Surfactant proteins are mainly synthesized in the lung where they were described and analyzed first.1–3 They account for 10% of the pulmonary surfactant, a complex mixture of lipids and proteins. Surfactant-specific proteins such as surfactant protein (SP) A (SP-A) and SP D (SP-D) belong to the collectins, a protein family that is characterized by a collagen-like region and a C-type lectin domain. These large hydrophilic proteins play a role in the initial phases of host defense and function as antibody independent opsonins. 4 Stimulation of macrophages by SP-A and SP-D leads to enhanced phagocytosis of various bacteria such as Pseudomonas aeruginosa.5,6 On the contrary, studies revealed that P. aeruginosa degrades SP-A by producing an enzyme, called Pseudomonas elastase. 7 Moreover, SP-A and SP-D are involved in surfactant metabolism8,9 and regulation of inflammation. 10 The characteristic surfactant function, that is, the adsorption and spreading of surfactant lipids at the alveolar air-liquid phase to lower surface tension, is taken over by the SPs B (SP-B) and C (SP-C). 11 These hydrophobic SPs perturb packing of phospholipids leading to a relaxation of the membrane. 12 SP-B deficiency is associated with lethal, neonatal respiratory distress syndrome (RDS) and chronic obstructive pulmonary disease (COPD).13,14 Also, the palate, lung, and nasal epithelial clone (PLUNC) proteins belong to the family of SPs. These are related proteins in humans that are important for surface tension and innate immunity in epithelial secretions of the respiratory tract and oral cavity. 15 PLUNC1 is also present in the tear film. 16 Up to now, SPs were detected in many locations throughout the human body such as in glandular structures of the sinonasal tract,17,18 airway submucosal gland cells, 19 lacrimal gland, 20 parotid and submandibular gland, 21 furthermore in skin, 22 brain, 23 testis, 24 female reproductive tract, 25 gastrointestinal tract, 26 and urinary tract. 27 Even in bacteria such as Staphylococcus aureus and P. aeruginosa, SPs and genes were proved with antibodies and primers to human SP-A, SP-B, SP-C, and SP-D. 28 In addition, two new SPs with functions in immune regulation, SP-H (surfactant-associated protein [SFTA]3), 29 and with physicochemical properties, SP-G (SFTA2), 30 were described. SPs are produced early in human fetal development not only in lung pneumocytes type II but also in glandular structures and epithelium in the lung.31,32
Labial glands are located in the lamina propria of upper and lower lips mucosa and categorized as minor salivary glands. They are characterized by mucous tubular and serous acinar endpieces, often the distal end of mucous tubules was capped by serous cells forming a half-moon (demilune cells). Intralobular ducts drain into the main excretory duct. True striated and intercalated ducts are rarely seen. Myoepithelial cells are found in secretory endpieces and small ducts. 33 Salivary glands produce saliva necessary for functions such as digestion, taste, and swallowing. Although minor salivary glands provide less than 10% of the total volume of whole saliva, the continuous production of saliva in the absence of local stimuli guarantees lubrication of the oral mucosa and protein secretion. 34 Among others, immunoglobulins and antimicrobial proteins are secreted by minor salivary glands. 35 The expression of lactoferrin, lysozyme, cathelicidin, and defensin-1, -2, -3 was detected in minor salivary glands, for example, in labial, lingual (von Ebner’s gland), and buccal glands.36–38 SP-A and SP-D as components of the innate immune system as well as SP-B and SP-C were identified in saliva and in epithelial structures of the parotid and submandibular gland. 21 SPs in various glandular tissues in humans are described in literature so far, but there are no data available on SP-A, SP-B, SP-C, and SP-D expression in infantile labial glands. This study complements information about SPs in glandular tissues.
Materials and Methods
Samples of lip tissue were obtained during surgical corrections of cleft lip in infantile patients. Excess tissue of the upper lip was collected and fixed immediately in buffered formalin (4%). The study group comprised 28 individuals (16 males and 12 females) aged 2 to 9 months, with 1 patient being 3 years of age. The study was performed according to the guidelines of the Ethics Committee (Nr. 2183/08).
IHC was performed on 5-µm paraffin sections using the avidin-biotin-horseradish peroxidase method with antibodies to SP-A (rabbit polyclonal ab87674; Abcam, Cambridge, UK), SP-B (mouse monoclonal sc-133143; Santa Cruz Biotechnology, Santa Cruz, TX), SP-C (rabbit polyclonal ab90716; Abcam), and SP-D (mouse monoclonal ab17781; Abcam). Antigen retrieval for unmasking epitopes was performed by microwave irradiation (600 W) in citrate buffer at pH 6.0 for 15 min. Endogenous peroxidase activity was inactivated by treatment of the sections with 3% hydrogen peroxide for 10 min. Sections were then pre-incubated with goat serum for blocking nonspecific binding. Primary antibodies were applied for 1 hr at room temperature and overnight at 4C in the following dilutions: SP-A 1:200, SP-B 1:200, SP-C 1:500, and SP-D 1:400. Incubation with the secondary biotinylated antibody recognizing mouse or rabbit IgGs of the primary antibody was performed at a 1:200 dilution for 30 min (Vector Laboratories, Inc.; Burlingame, CA). Afterward, peroxidase-labeled streptavidin was applied to the slides for 45 min (Vector Laboratories, Inc.). Brown color was developed in diaminobenzidine with hydrogen peroxide (Sigma; St. Louis, MO). For counterstaining, hematoxylin was used. For rinsing the sections between the different incubation steps (3 times for 5 min), PBS, pH 7.4, was used. Human lung tissue served as positive control. Isotype-negative controls were performed on all specimens. In addition, negative controls, in which the primary antibody was omitted and replaced by buffer, were treated equally. For histological orientation, sections were stained with H&E. 39 Images were viewed with a Nikon microscope and digital camera (Nikon; Düsseldorf, Germany). Labeling results were categorized in weak, medium, and strong staining.
Results
General Morphology
The labial glands were aligned as globular structures in the oral mucosa (Fig. 1A). They were located in the lamina propria of the labial mucosa and showed the characteristic feature of a mixed salivary gland with serous and mucous endpieces; mucous endpieces predominated and displayed often serous demilunes at the distal end of the mucous tubule. Serous glandular cells are grouped in the form of an acinus at the secretory endpiece (Figs. 1B and C). Intercalated ducts were rarely seen in sections. Intralobular ducts showed mostly a two-layered epithelium leading into the excretory duct that emptied into the oral cavity. A stratified squamous epithelium lined the oral mucosa.
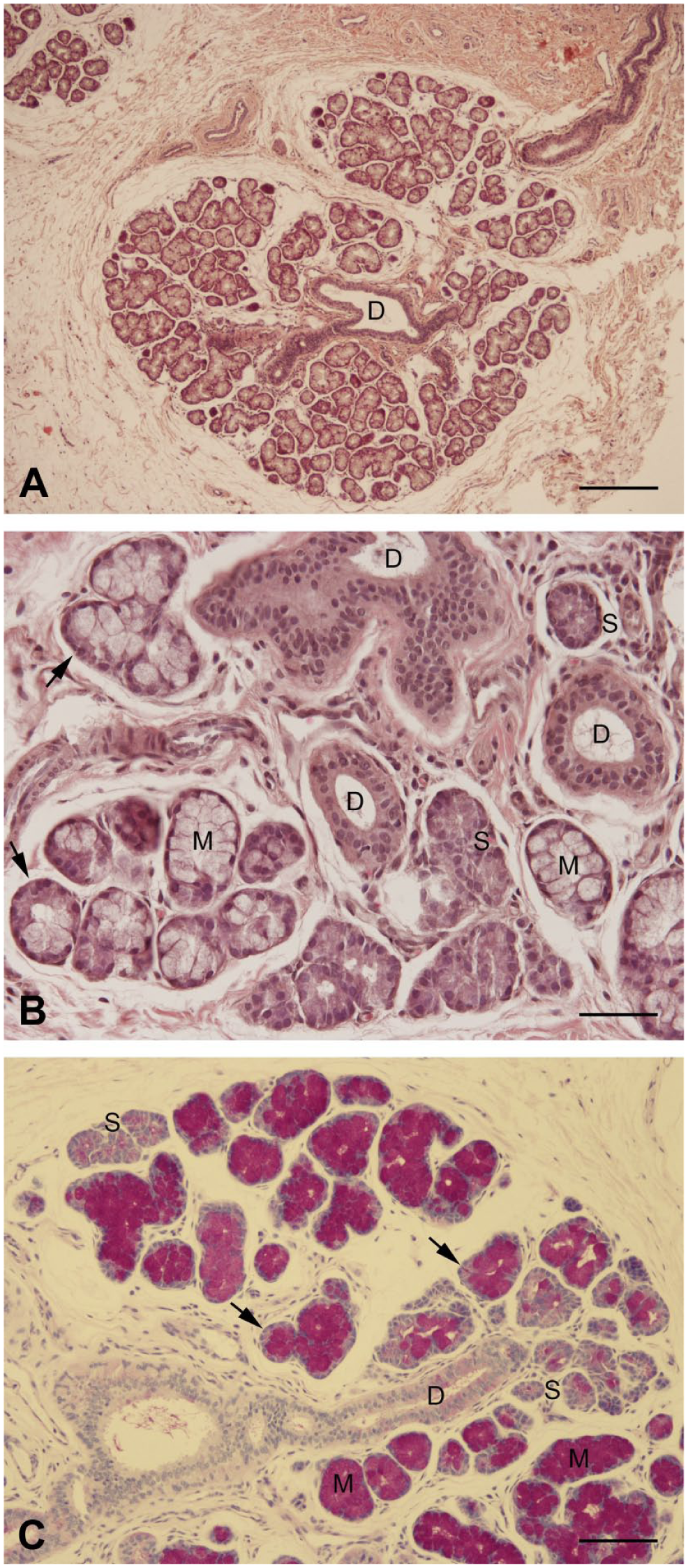
Labial glands at low (A) and high magnification (B, C). H&E stain (A, B). Periodic acid-Schiff stain (C). (D) duct, S (serous cells), M (mucous cells), arrows (demilune cells). Scale bars: A = 250 µm, B = 50 µm, C = 100 µm.
Surfactant Protein A
Ductal cells of seromucous glands produced medium to strong SP-A signals in all cell layers. Partially, the basal cytoplasm of the lowest duct layer was stronger stained. Acinar cells were stained with a weak to medium intense staining. Mucous cells were SP-A negative (Fig. 2A). The basal and intermediate layer of the oral mucosa revealed SP-A antibody reactivity; the superficial cell layer remained unstained (Fig. 2B). Incubation with antibodies to SP-A resulted in a positive immunoperoxidase staining of type II pneumocytes in the human lung (Fig. 2C).
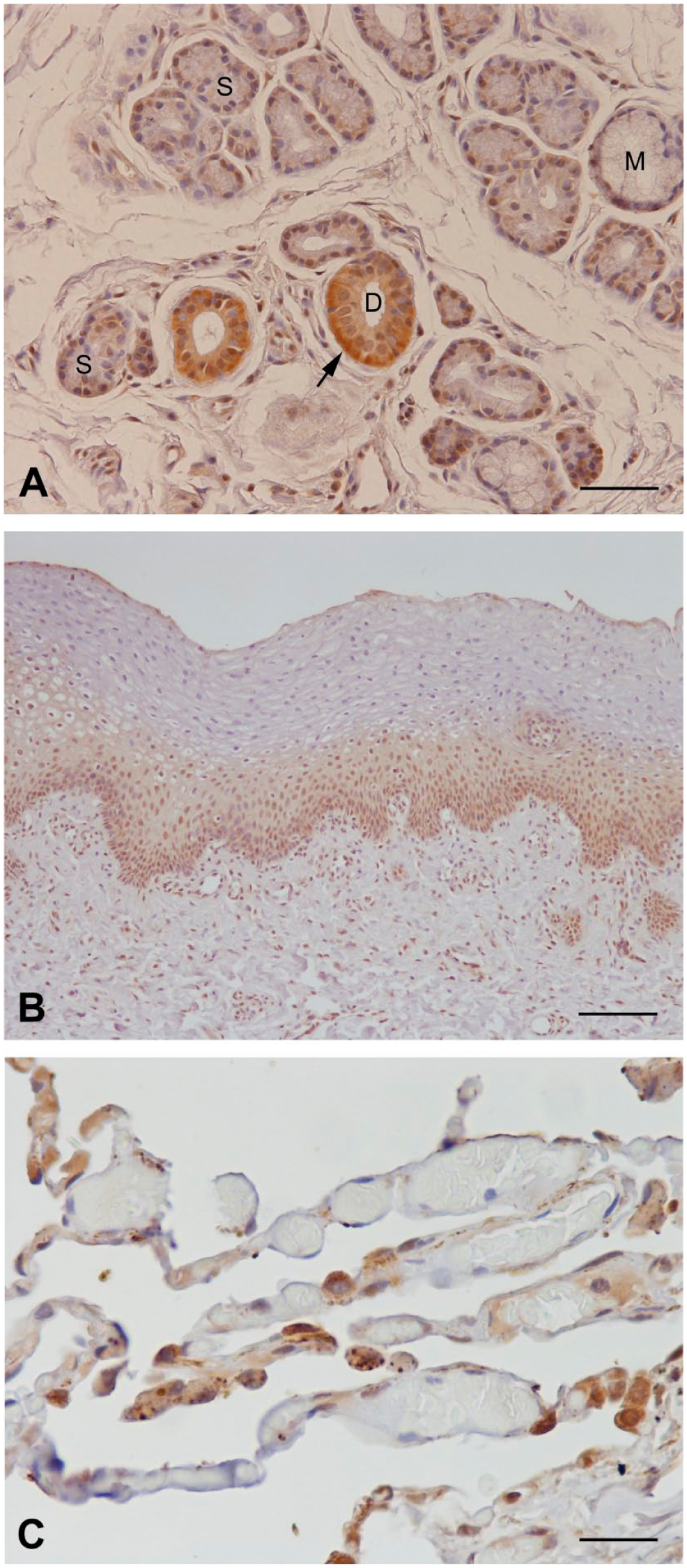
Immunolocalization of SP-A. (A) SP-A-detection, especially in ductal cells of labial glands, with strong expression in the basal cytoplasm (arrow). S (serous cells), M (mucous cells), D (duct). (B) SP-A expression in the basal and intermediate layer of the mucosa. (C) SP-A in lung tissue (positive control). Scale bars: A = 50 µm; B = 100 µm; C = 25 µm. Abbreviation: SP, surfactant protein.
Surfactant Protein B
SP-B antibody-incubation resulted in a medium to strong staining intensity in acinar cells and demilune cells at the distal end of a mucous glandular endpiece. With respect to the apical and basal cell compartment, the staining was evenly distributed (Fig. 3A). The duct system showed the same staining intensity as the secretory units. However, the SP-B protein was more strongly expressed in the apical part of the top cell layer. Occasionally, apical protrusions were detected (Fig. 3A, inset). Mucous cells exhibited no immunoreaction. The basal layer of the stratified squamous epithelium was stained; the intermediate and superficial layers remained unstained (Fig. 3B). Type II pneumocytes as controls reacted positively (Fig. 3C).
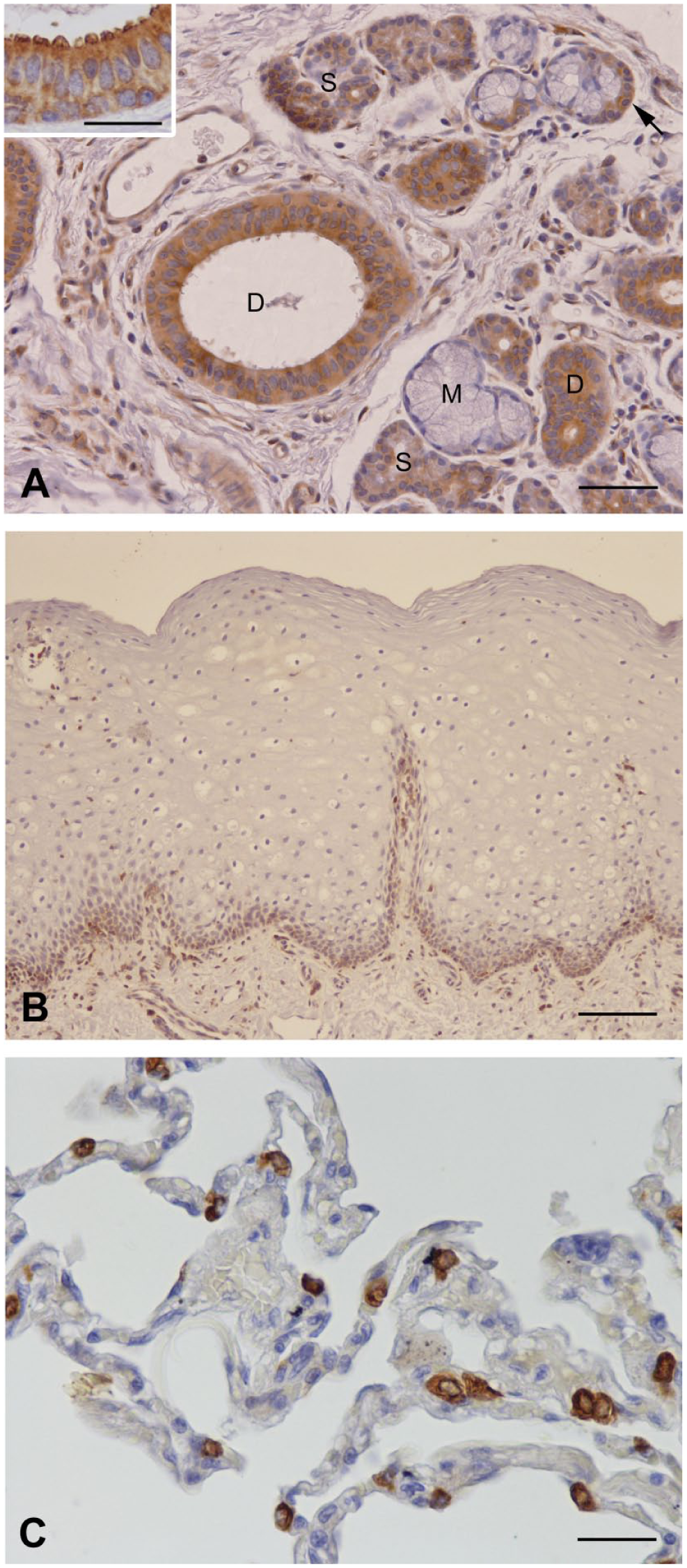
Immunolocalization of SP-B. (A) Positive staining of SP-B in serous and ductal cells. Occasionally, ductal cells exhibited protrusions (inset). S (serous cells), M (mucous cells), D (duct), arrow (demilune cells). (B) SP-B expression in the basal layer of the mucosa. (C) SP-B in lung tissue (positive control). Scale bars: A = 50 µm, inset A = 25 µm; B = 100 µm; C = 25 µm. Abbreviation: SP, surfactant protein.
Surfactant Protein C
SP-C was expressed in serous acinar cells and demilune cells with a medium to strong staining. The same immunoreactivity could be observed for the duct system (Fig. 4A). In contrast to the serous cells, which were evenly stained, intralobular ducts revealed partially an apically stronger staining. The oral mucosa exhibited a positive reaction with the anti-SP-C antibody in the basal and intermediate layers, while the superficial layer remained unstained (Fig. 4B). A positive antibody reaction occurred with type II pneumocytes lining alveoli (Fig. 4C).
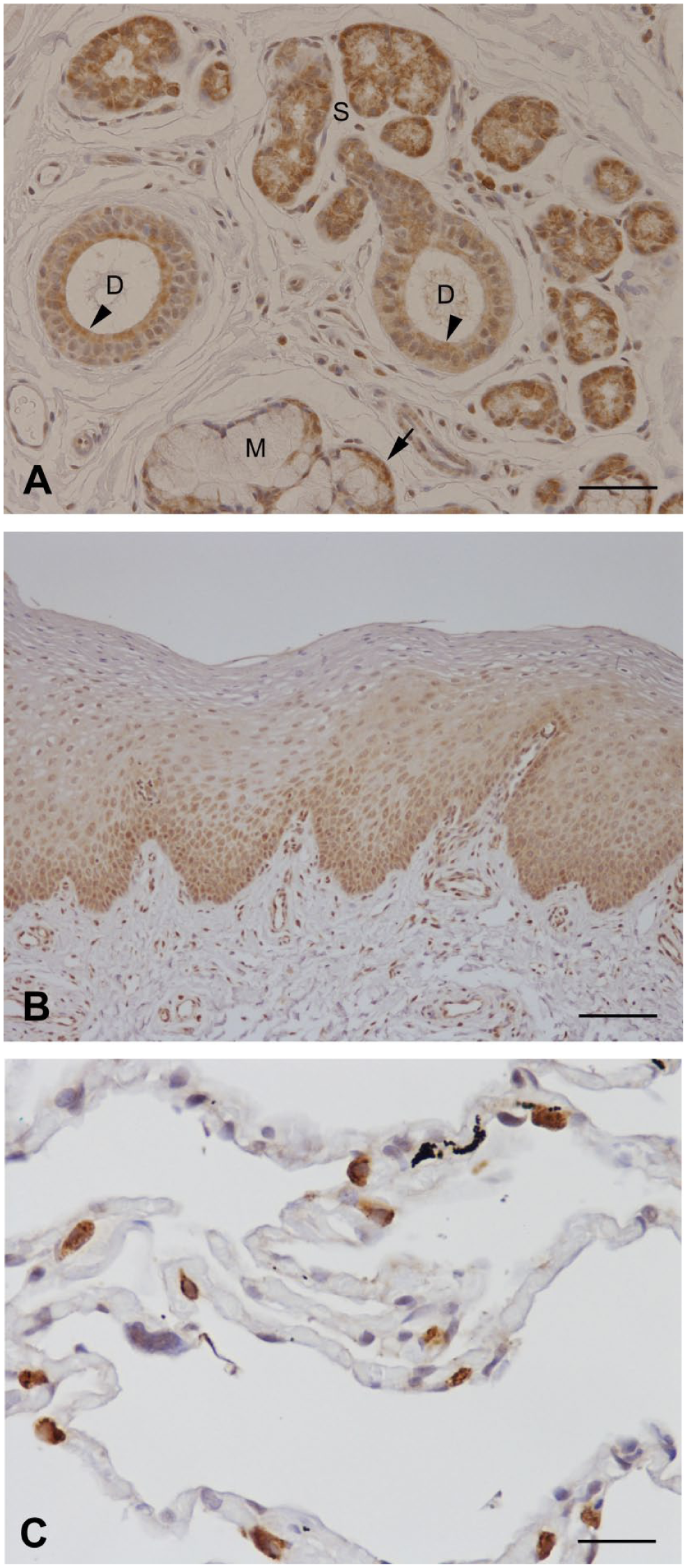
Immunolocalization of SP-C. (A) SP-C expression in serous cells and the duct system; apically strong expression in ductal cells of the top layer (arrowheads). S (serous cells), M (mucous cells), D (duct), arrow (demilune cells). (B) SP-C expression in the basal and intermediate layer of the mucosa. (C) SP-C in lung tissue (positive control). Scale bars: A = 50 µm; B = 100 µm; C = 25 µm. Abbreviation: SP, surfactant protein.
Surfactant Protein D
SP-D is mainly associated with the duct system of the labial glands. Here, the cells revealed a strong reaction to the antibody to SP-D. Especially, the apical part of the cells in the top layer expressed the protein intensively (Fig. 5A). In the upper epithelial layer of the duct, occasionally, granules were visible in the cytoplasm. In serous acinar cells, a supranuclear region was stained in a weak to medium intensity. The basal part of the glandular cells either showed a weak immunoreactivity or remained unstained (Fig. 5A, inset). Mucous glandular cells expressed no SP-D. Basal cells of the stratified squamous epithelium showed a faint antibody reaction, while suprabasal cell layers and interspersed cells in the intermediate layers stained stronger. The superficial layer showed no reaction to the SP-D antibody (Fig. 5B). Positive control sections with human lung tissue exhibited stained type II pneumocytes (Fig. 5C).
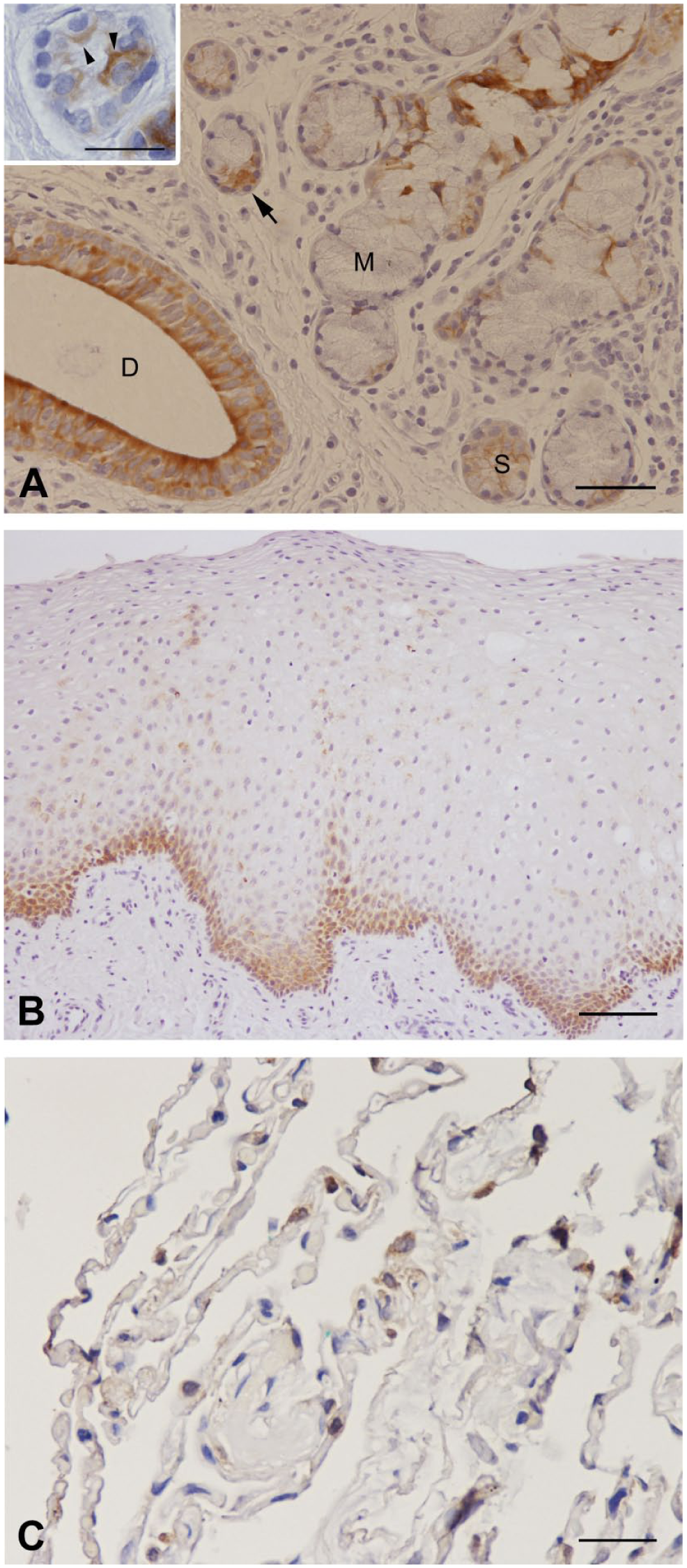
Immunolocalization of SP-D. (A) SP-D antibody reactivity mainly in the duct system; serous acinar cells showed a supranuclear cytoplasmic staining (arrowheads, inset). S (serous cells), M (mucous cells), D (duct), arrow (demilune cells). (B) SP-D expression predominantly in suprabasal layers. (C) SP-D in lung tissue (positive control). Scale bars: A = 50 µm, inset A = 25 µm; B = 100 µm; C = 25 µm. Abbreviation: SP, surfactant protein.
Discussion
SPs such as SP-A, SP-B, SP-C, and SP-D comprise a wide spectrum of activity such as immune defense and physicochemical characteristics in different tissue types throughout the human body.6,9,10 Along with the lung, glandular tissues are one of the most studied in this field.17,20,28 We provide the first investigation on SPs in infantile labial glands and broaden the spectrum of occurrence.
Labial glands exhibited positive SP-A, SP-B, SP-C, and SP-D staining in both the excretory ducts and the acinar cells of the secretory endpieces in different intensities. Mucous cells remained unstained. Salivary glands of the lip drain their secretion product to the stratified squamous epithelium of the oral mucosa. Antibodies to the 4 SPs especially stain the basal cell layer. There are numerous investigations on glandular tissue and the mucosal epithelium dealing with the expression of SPs. Antibody reactivity for SP-A, SP-B, SP-C, and SP-D was demonstrated for the excretory duct system and serous acinus cells in parotid and submandibular glands. 21 In a prior analysis by Bräuer et al., 20 acinar epithelial cells of the lacrimal gland and epithelial cells of the conjunctiva and nasolacrimal ducts revealed SP-A and SP-D expression. Gaunsbaek et al. 40 found SP-A, SP-B, SP-C, and SP-D in acini of the submucosal glands in nasal mucosa. Saitoh et al. 19 investigated submucosal glands of human airway walls and showed a positive SP-A stain over serous cells. The immunolocalization of SP-A and SP-D in seromucinous glands of sinonasal mucosa resulted in an intense expression of the ductal epithelium; no staining, however, was identified in the secretory glandular part. 17 Moreover, the ciliated pseudostratified columnar epithelium was SP-A and SP-D positive. SP-D stained the basal layer of the respiratory epithelium. In our investigation, this applied also for the basal cells of the stratified squamous epithelium in the oral mucosa. Madsen et al. 41 reported a positive SP-D expression in duct epithelial cells within salivary glands in compliance with our findings. Lee et al. 42 localized SP-A to ductal cells of the parotid gland while acinar cells were negative. In our findings, SP-A and SP-D were positive in acinar cells, but were weaker expressed than SP-B and SP-C. The preferred expression of SP-A and SP-D in the duct system seems to be part of the mucosal defense system. Duct systems are exposed to invading microorganisms; secretion of defense proteins in that localization provides direct protection. Human SP-D reactivity was also seen in the intercalated ducts of the pancreas, the small bile ductules of the liver, and in sweat glands. 41 SPs were often related to chronic diseases such as rhinosinusitis 43 or sialadenitis 42 showing then a stronger immunoreactivity. Saliva SPs SP-A, SP-C, and SP-D are increased in periodontal diseases, while SP-B is decreased. 44 SP-A serves as a marker of lung adenocarcinoma, and the transduction of SP-A gene into human lung adenocarcinoma cell lines suppresses the progression of tumor in mouse models. 45 SP-B and SP-C play a major role in lowering the surface tension across the alveolar air-liquid-phase in lung. 11 A similar function was attributed to glandular SPs facilitating mucociliary transport, for example, in the Eustachian tube. 46 In a similar way, SP-B and SP-C may help to improve the flow of secretion material through the duct system of glandular tissues as discussed by Bräuer et al. 20 This quality is also helpful in the flow property in human saliva of the parotid gland, submandibular gland, 21 and tear fluid 20 as all four SPs were detected therein. Interestingly, Gaunsbaek et al. 40 revealed that in nasal lavage and mucus, the levels of SP-A, SP-B, SP-C, and SP-D were not detectable although they could be demonstrated immunohistochemically in acinar cells of nasal submucosal glands and in the surface epithelium. A possible explanation may be that in nasal seromucosal glands, mucous endpieces predominate, consequently, SPs of serous acini were difficult to be detected by conventional methods. In addition, the authors speculate that the large lavage-volume complicates the detection of a low concentration of SPs.
In our investigation, we proved the presence of the SPs SP-A, SP-B, SP-C, and SP-D in infantile labial glands. The presence of all four SPs point, on one hand, to functions such as the reduction of surface tension in saliva, and, on the other hand, to immune defense early in infant development.
Footnotes
Acknowledgements
The authors are grateful to Amela Klaus for skillful technical assistance.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: study design (MS, MRK), acquisition of material (MRK, FF), experiment support (CS), data analysis and manuscript editing (MS), manuscript review (MS, FF, CS, K-DW, MRK), and all authors have read and approved the manuscript as submitted.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
