Abstract
Wnt signaling comprises a group of pathways emanating from the extracellular environment through cell-surface receptors into the intracellular milieu. Wnt signaling cascades can be divided into two main branches, the canonical/β-catenin pathway and the non-canonical pathways containing the Wnt/planar cell polarity and Wnt/calcium signaling. Syndecans are type I transmembrane proteoglycans with a long evolutionary history, being expressed in all Bilateria and in almost all cell types. Both Wnt pathways have been extensively studied over the past 30 years and shown to have roles during development and in a multitude of diseases. Although the first evidence for interactions between syndecans and Wnts dates back to 1997, the number of studies connecting these pathways is low, and many open questions remained unanswered. In this review, syndecan’s involvement in Wnt signaling pathways as well as some of the pathologies resulting from dysregulation of the components of these pathways are summarized.
Wnt Signaling
Wnt signaling is one of the most evolutionarily conserved pathways in metazoans, described in organisms from Hydra to man. Wnt signaling cascades control many aspects of developmental processes, but also have roles in adult tissue homeostasis by regulating cell proliferation, differentiation, polarized migration, genetic stability and apoptosis. Therefore, their dysregulation has been connected to many types of disease.
Members of the Wnt family are secreted, hydrophobic glycoproteins that can act as long-range morphogens or specific short-range ligands for various receptors in order to initiate distinct signaling cascades, depending on receptor–co-receptor combinations and cellular context (Gross and Boutros 2013; van Amerongen 2012). The Wnt-mediated pathways are initiated by the binding of Wnt ligands to Frizzled (Fz), seven-pass transmembrane receptors that resemble G-protein-coupled receptors (GPCR). Ten different Fz receptors and 19 Wnts have been identified in humans and multiple Wnts can bind different Fz receptors, making the exact description of Wnt pathway initiation complicated and still not completely resolved. We will summarize here the two main Wnt pathway branches activated upon ligand–receptor coupling.
Based on the involvement of the β-catenin downstream effector, the Wnt pathways have been divided into two main branches, the canonical Wnt pathway, or Wnt/β-catenin, and the non-canonical Wnt pathways, which contain the planar cell polarity (PCP) and Wnt/calcium signaling. In addition to these, there are other alternative Wnt pathways and receptors not discussed here in detail but recently summarized by others (van Amerongen 2012; Gao and Chen 2010). Generally, Wnt1/2/3a/8a/8b/10a/10b and Fz1/5/7/9 are classified as the elements of the canonical pathway, whereas Wnt4/5a//6/7a/7b/11 and Fz2/3/4/6 are described as non-canonical elements; the remaining binding combinations are, however, not yet deciphered (Clevers 2006; Huelsken and Behrens 2002). Some of Wnt–Fz combinations can also have both canonical and non-canonical functions; e.g., in the absence of the Ror2 co-receptor, Wnt5a can activate the canonical pathway by binding to Fz4 and Lrp6 (Armstrong et al. 2011).
β-Catenin is a key component of canonical Wnt signaling and is a component of cell–cell junctions (Hirano et al. 1992) (Fig. 1A, 1B). The Wnt/β-catenin pathway is the best characterized Wnt pathway to date. In the absence of Wnt ligand, cytosolic β-catenin is targeted for degradation by a complex composed of the scaffolding protein axin, adenomatous polyposis coli (APC), and the two serine/threonine kinases, glycogen synthase kinase 3β (GSK3β) and casein kinase 1 (CK1) (MacDonald et al. 2009). Axin and APC bind β-catenin and increase its N-terminal phosphorylation by the kinases. The first phosphorylation is mediated by CK1 at Ser45, after which GSK3β can phosphorylate other sites, including Thr41, Ser37 and Ser33. The latter two serine phosphorylations facilitate the binding of an E3 ubiquitin ligase, which leads to β-catenin ubiquitination and proteasomal degradation (Liu et al. 2002; Kimelman and Xu 2006). When Wnt binds to Fz and Lrp5/6 co-receptors, the resulting ternary complex recruits the scaffolding protein Dishevelled (Dvl/Dsh) to the plasma membrane, which, in turn, traps the axin/GSK3β complex, preventing the constitutive destruction of β-catenin. The stabilized β-catenin can then translocate into the nucleus and bind to TCF/LEF (T cell factor/lymphoid enhancer factor) transcription factors, and thereby initiate transcription of different genes that have roles in cell differentiation and proliferation (Daniels and Weis 2005) (Fig. 1A). There are many target genes in the canonical pathway, most of which are cell-type specific, with the exception of axin2, which has been described as a negative feedback regulator (Grigoryan et al. 2008).
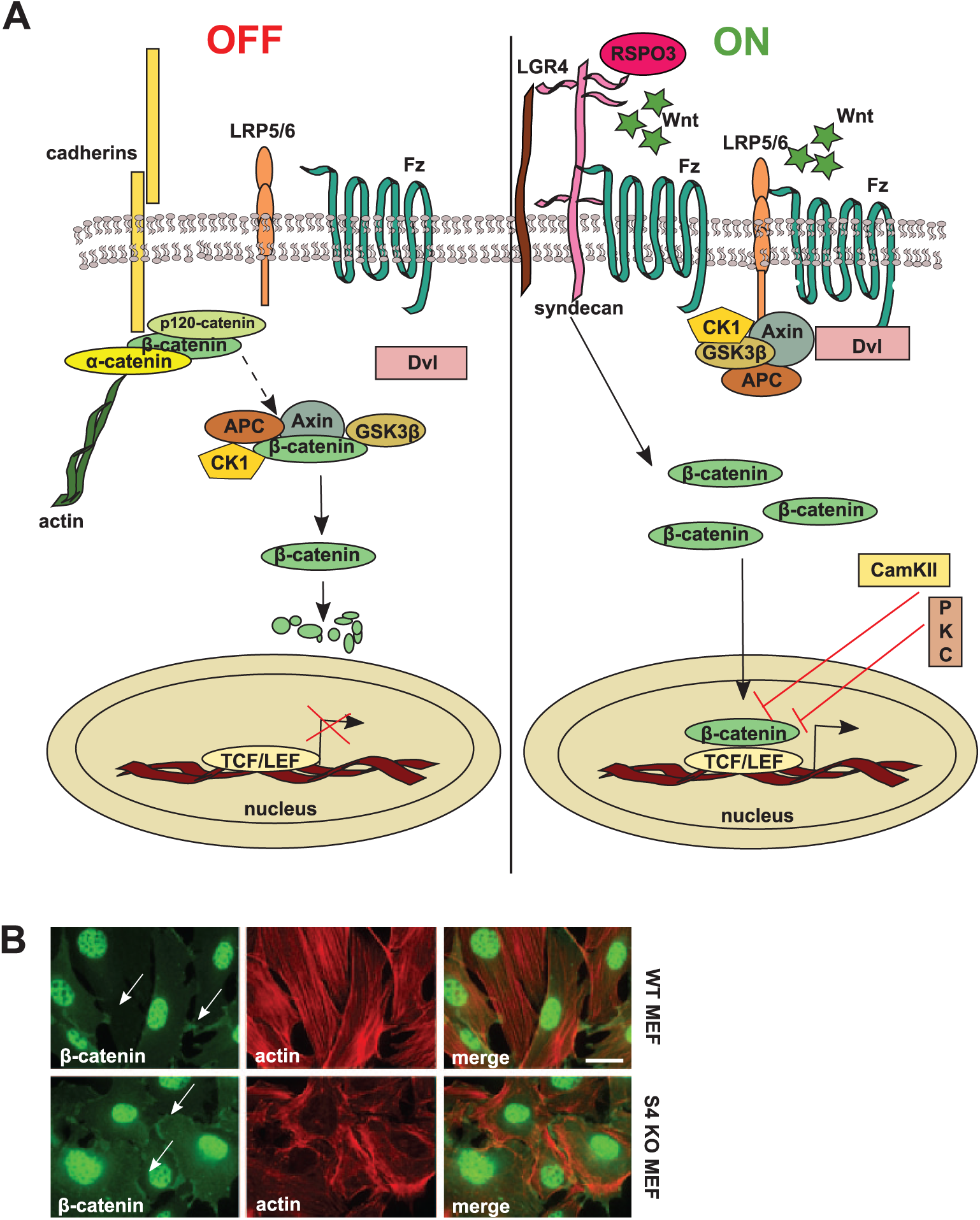
The canonical Wnt/β-catenin pathway. (A) In the absence of Wnt ligands (“OFF” state), β-catenin levels are kept low through their interaction with the destruction complex formed by protein kinases CK1 (casein kinase) and GSK3β (glycogen synthase kinase 3β), the tumor suppressor protein, APC (adenomatous polyposis coli), and the scaffolding protein, axin. After phosphorylation by the kinases, β-catenin is degraded in proteasomes. β-Catenin levels are also regulated by adhesion complexes that contain cadherins and other catenins. During Wnt signaling (“ON” state), Wnt interacts with the frizzled (Fz) receptor and LRP5/6 co-receptors. LRP binds axin and Fz binds dishevelled (Dvl), thereby recruiting the β-catenin destruction complex in the vicinity of plasma membrane. As a consequence, the kinases do not phosphorylate β-catenin, which thus escapes degradation and can instead enter the nucleus to interact with TCF/LEF transcription factors. The Wnt/calcium pathway inhibits the β-catenin pathway, since PKC (protein kinase C) and CaMKII (calcium-calmodulin-dependent kinase) inhibit nuclear β-catenin’s interaction with transcription factors. Interaction of R-spondin 3 (RSPO3) with syndecan-4 activates the Wnt/β-catenin pathway in collaboration with LGR4 (leucine-rich repeat-containing G protein-coupled receptor) receptor. (B) β-catenin localizes to cell–cell contacts (white arrows) and nuclei. β-Catenin was detected in wild type and syndecan-4 null mouse embryonic fibroblasts by indirect immunofluorescence microscopy. Cells were fixed in 4% paraformaldehyde followed by sequential incubations with anti-β-catenin (clone 9587, Cell Signaling Technology, Beverly, MA) and Alexa 488-labelled anti-rabbit IgG (green; Molecular Probes, Carlsbad, CA). Filamentous actin (F-actin) was visualized using Phalloidin-Alexa568 (red; Molecular Probes, Carlsbad, CA). Images were captured on a Zeiss Axioplan epifluorescence microscope, and intensity and contrast were enhanced using Adobe Photoshop CS6 (Adobe Systems Inc., San Jose, CA). Bar, approximately 20 µm.
Cytoplasmic Dvl is considered to be a hub for Wnt signaling cascades, having roles in all branches of this pathway (Gao and Chen 2010). In addition, Dvl has been shown to shuttle between the cytoplasm and nucleus; but, whereas prevention of nuclear localization impairs the canonical pathway, it has no effect on the non-canonical pathway-mediated cell migration during convergent extension in Xenopus (Fanto et al. 2000).
In contrast to the canonical Wnt pathway, non-canonical signaling does not depend on β-catenin but requires the co-receptors Ror2/Ryk/PTK7 instead of Lrp5/6. In the Wnt PCP pathway (Fig. 2), ligand interaction with Fz recruits Dvl, which, in turn, binds to the small GTPase Rho via the formin homology adaptor protein Daam1 (Dvl associated activator of morphogenesis 1). The formin complex further interacts with Rho GEFs leading to Rho nucleotide exchange and activation, which initiates ROCK (Rho-associated protein kinase) signaling and remodels the actin cytoskeleton (Tanegashima et al. 2008). In addition, Dvl can also associate with Rac, which stimulates its downstream effector C-jun-N-terminal kinase (JNK) to regulate polarized cell movements during Xenopus development (Habas et al. 2003) and initiates the MAP kinase cascade with subsequent activation of AP1-mediated (activator protein 1) transcription (Jones and Chen 2007; Oishi et al. 2003; Sebbagh and Borg 2014) (Fig. 2). The PCP pathway is predominantly involved in regulating polarized cell migration during gastrulation, neural tube closure, and cilia orientation in the organ of Corti (Kikuchi et al. 2011; Simons and Mlodzik 2008).
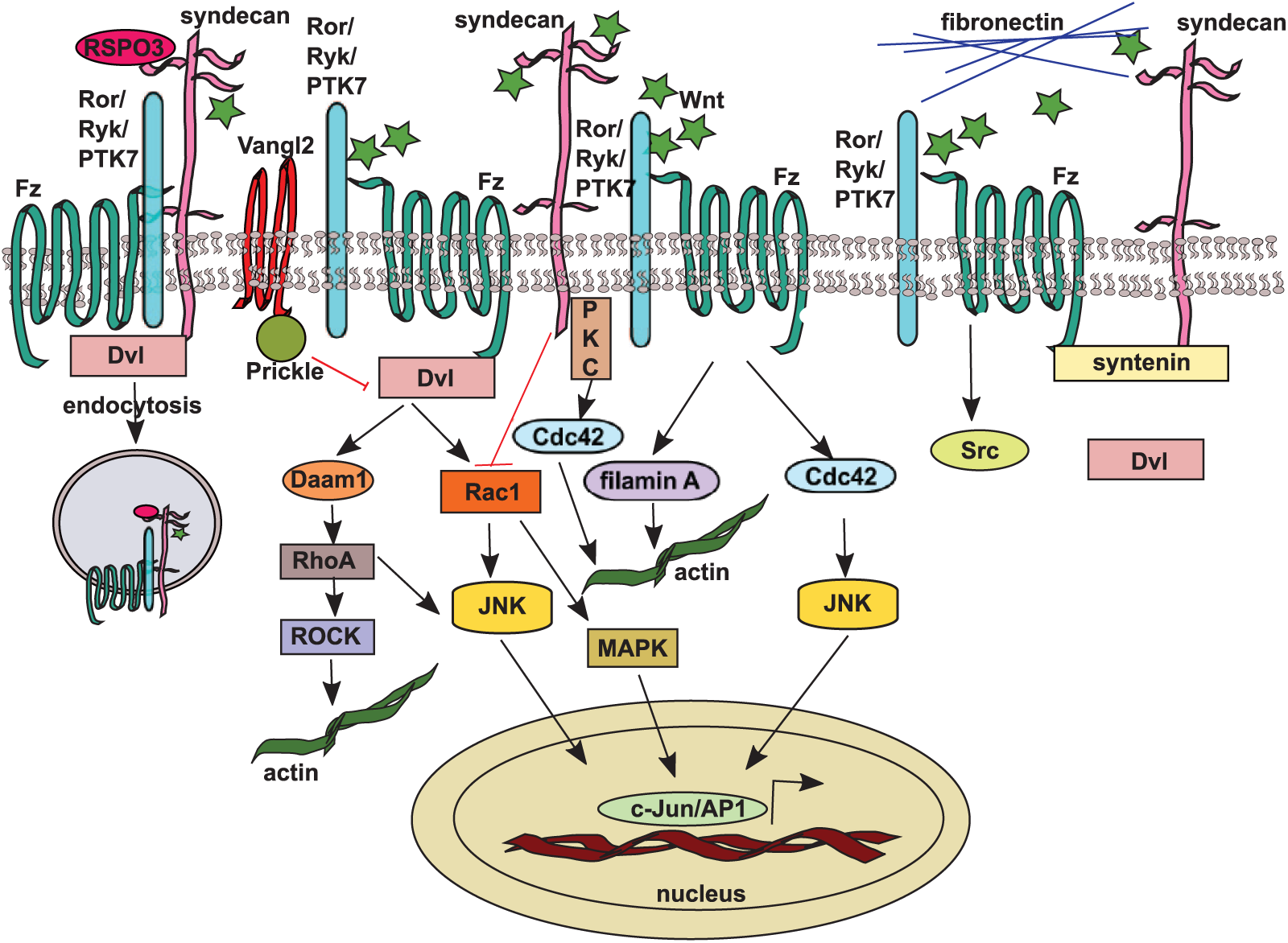
The non-canonical Wnt/planar cell polarity pathway. Wnt stimulates the PCP pathway by activating small GTPases Rho, Rac, and Cdc42 through recruitment of dishevelled (Dvl) by frizzled (Fz) and its co-receptors (Ror/Ryk/PTK7) leading to actin cytoskeleton rearrangements and C-Jun and AP1 (activator protein 1) transcription factor activation via JNK (C-jun-N-terminal kinase) and MAPK (MAP kinase) pathways. The Vangl2 and Prickle complex inhibits the PCP pathway. Syndecans influence the PCP pathway by inhibiting Rac and activating Rho and Cdc42, and they have been shown to interact directly with frizzled 7 and dishevelled during convergent extension in Xenopus. During the same developmental process, syndecan-4 and dishevelled are bound by different domains of syntenin scaffolding protein. Syndecan-4 also interacts genetically with Vangl2 to regulate neural tube closure. RSPO3 exerts its function in PCP signaling by inducing syndecan-4-dependent clathrin-mediated endocytosis of the Wnt complex.
Small changes in intracellular calcium concentrations act as a second messenger. Wnt/Fz signaling via a trimeric G protein activates phospholipase C (PLC), leading to the generation of 1,2 diacylglycerol (DAG) and inositol 1,4,5-trisphosphate (IP3) from the membrane-bound phospholipid phosphatidylinositol 4,5-bisphosphate (PtdIns4,5P2). The formation of IP3 and DAG results in the release of intracellular calcium from endoplasmic reticulum stores and opening of new store-operated calcium channels on the plasma membrane, which increases extracellular calcium influx (Sheldahl et al. 2003; Parekh and Putney 2005). The release of intracellular calcium is sufficient to activate various calcium-sensitive enzymes such as calcium-calmodulin-dependent kinase II (CamKII), protein kinase C (PKC), and the calcium-sensitive phosphatase calcineurin (Slusarski and Pelegri 2007; McNeill and Woodgett 2010; De 2011). Further, calcineurin activates the nuclear factors of activated T cells (NFATs) by dephosphorylation, triggering their nuclear translocation and assembly with partner proteins to form transcriptional complexes (Fig. 3). Re-phosphorylation of NFATs by glycogen synthase kinase (GSK3) leads to their export from the nucleus, ending their cycle of activation; this has been reviewed in detail by Schultz and Yutzey (2004). NFATs can be re-phosphorylated in the cytoplasm as well by factors, such as CK1 (Ghosha et al. 2010) and mTOR (mammalian target of rapamycin) (Yang et al. 2008). Another molecule activated by CamKII is the NLK (Nemo-like kinase), which also enters the nucleus and acts as an inhibitor of the β-catenin pathway through phosphorylation and degradation of TCF/LEF (Ishitani et al. 2003) (Fig. 3). The Wnt/calcium pathway has been mainly implicated in cancer, inflammation and neurodegeneration (Clark et al. 2012); however, many aspects of this pathway remain enigmatic.
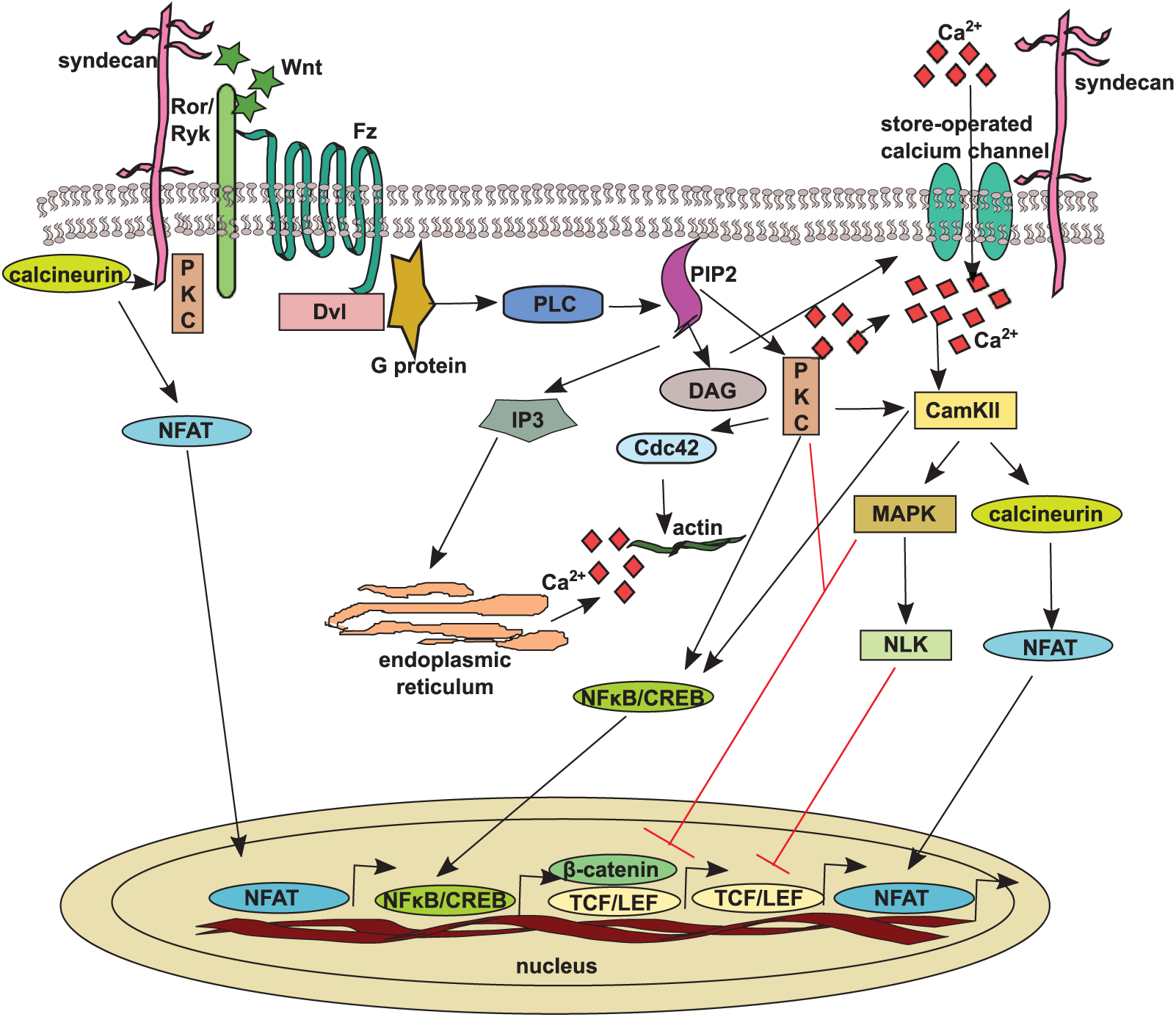
The non-canonical Wnt/calcium pathway. Upon Wnt binding, the frizzled (Fz) receptor and its co-receptors are able to recruit dishevelled and small G-proteins leading to activation of PLC (phospholipase C) and the hydrolysis of phosphatidylinositol 4,5-bisphosphate (PtdIns4,5P2) into IP3 (inositol 1,4,5-triphosphate) and DAG (1,2 diacylglycerol). IP3 binds to its receptor on the endoplasmic reticulum and triggers the release of calcium ions into the cytoplasm. This calcium release and DAG production open the cell-surface store-operated calcium, thereby allowing more calcium to be imported. The increase in cytoplasmic calcium activates CamKII (calcium-calmodulin-dependent kinase II), PKC (protein kinase C), and calcineurin, which leads to the activation and nuclear translocation of NFATs (nuclear factors of activated T cells) to initiate the transcription of different genes. PKC and CamKII also act through NFκB/CREB. CamKII negatively regulates β-catenin nuclear activity directly or via NLK (Nemo0like kinase). Syndecans, by direct interaction with calcineurin and/or PKC, influence NFAT activation.
An interesting aspect of Wnt signaling is the cross-talk between the different branches of the pathway. One example is the interaction between PKC, GSK3β, and β-catenin. GSK3β may be phosphorylated by PKC, leading to the expression of neuroprotective genes via activation and nuclear translocation of β-catenin, whereas PKC phosphorylates β-catenin on Ser33, Ser37 and Ser45, promoting its degradation via Wnt/calcium signaling (De 2011). The Wnt/β-catenin and Wnt/calcium signaling pathways have opposite roles during convergent extension movements, with PKC and CamKII influencing the signaling activity of β-catenin (Kuhl et al. 2001) (Figs. 1, 3). Similarly, distinct Wnt signaling pathways have been shown to have opposing roles in tail fin regeneration (Stoick-Cooper et al. 2007).
Another aspect of antagonistic cross-talk occurs at the ligand–receptor level of Wnt pathways. For example, Wnt5a, which is more specific to the non-canonical pathway, competes for and inhibits binding of Wnt3a (mainly specific for β-catenin pathway) to Fz2, thereby suppressing the β-catenin pathway (Sato et al. 2010). Moreover, the non-canonical Wnt ligands either inhibit (Torres et al. 1996) or activate (He et al. 2004) the β-catenin pathway, raising the possibility that syndecans may also affect both pathways.
Syndecans: Structure and Function
The syndecan family are cell-surface heparan sulfate proteoglycans (HSPGs) and, more precisely, are type I transmembrane glycoproteins. Syndecans have a long evolutionary history, being present in Cnidaria and throughout the Bilateria (Chakravarti and Adams 2006; Couchman 2010; Pataki and Couchman 2012). Invertebrates and primitive chordates have one syndecan gene, whereas four syndecan family members occur in higher vertebrates (Chakravati and Adams 2006). Most cell types, with the exception of erythrocytes, express at least one syndecan. Syndecan-1 (CD138) is expressed at higher levels in epithelial cells, condensing mesenchyme in development, and in plasma cells. Syndecan-2 (fibroglycan) is present in cells of a mesenchymal origin, fibroblasts, and developing neural tissue. Syndecan-3 (N-syndecan) is primarily enriched in the neural tissue and in the developing musculoskeletal system. Syndecan-4 (amphiglycan, ryudocan), on the other hand, is expressed ubiquitously, although at a lower level than the other syndecans in the same cell (Couchman 2010; Xian et al. 2010).
Structurally, syndecans can be divided into three major domains. The N-terminal ectodomain is decorated with several glycosaminoglycan (GAG) chains, mostly heparan sulfate (HS), although syndecan-1 and -3 may also be substituted with chondroitin sulfate (CS) or dermatan sulfate (DS). The ectodomain core shows low sequence homology between syndecan family members. In contrast, the transmembrane and short C-terminal cytoplasmic domains are highly conserved (Zhang et al. 1995; Yoneda and Couchman 2003).
Syndecans are localized to the plasma membrane, having functions in cell adhesion, migration, cytoskeleton organization and differentiation. Some time ago, it was believed that the 20–40 kDa core protein ectodomains of syndecans only functioned to sustain the GAG chains, but now it is clear that the ectodomains of syndecan-1, -2 and -4 have roles in cell adhesion (Whiteford and Couchman 2006; Beauvais et al. 2009). Syndecan-2 and -4 core proteins are smaller and belong to one subfamily, whereas syndecan-1 and -3 belong to another subfamily, based on sequence homology (Choi et al. 2010). The HS chains are close to the N-terminus and attached to serine/glycine sites on the core protein; CS and DS chains of syndecan-1 and -3 are more membrane-proximal (Multhaupt et al. 2009).
The GAG chains are complex polysaccharides that are synthesized in the Golgi through the action of multiple enzymes (Bishop et al. 2007). HS synthesis results in several sulfation modifications that are major determinants of ligand binding, as reviewed in detail by Xu and Esko (2014). A wide range of extracellular ligands are captured by HS chains, including polypeptide growth factors, chemokines, morphogens (including Wnts), extracellular matrix (ECM) proteins, cell–cell adhesion molecules, blood coagulation factors, and lipid-regulating enzymes (Couchman 2010). Based on these interactions, syndecans are often described as co-receptors, since they can collaborate with other receptors like the vertebrate fibroblast growth factor receptors, Wnt signaling Fz receptors (Luyten et al. 2008), Hedgehog signaling members (Shimo et al. 2004), and transforming growth factor β (TGF-β) receptors (Chen et al. 2004). However, some of these interactions are not specific to syndecans, since glypicans may have similar roles (Witt et al. 2013), including a role in Wnt signaling (Capurro et al. 2014). Glypicans are also HSPGs, but with a C-terminus covalently attached to the plasma membrane via a GPI (glycosylphosphatidylinositol) anchor. Again, conserved throughout evolution, mammals express six distinct glypican core proteins. (Filmus and Capurro 2012). Very recent evidence shows that glypicans can inhibit Wnt signaling indirectly because their HS chains can act as scaffolds to sequester both Notum and Wnt on the cell surface in a ternary complex. Notum deacylates Wnt, inhibiting its signaling activity by removing the palmitoleate moiety that is important for receptor binding (Kakugawa et al. 2015).
The short transmembrane domain of syndecans, consisting of 25 hydrophobic amino acids, promotes dimerization or oligomerization of the core proteins (Choi et al. 2005; Dews and Mackenzie 2007). Oligomerization is important for syndecan signaling from the ECM to different cytoplasmic components and for their localization to distinct membrane compartments; therefore, the syndecan homodimer is an essential functional unit (Choi et al. 2005).
All four syndecans can connect with the cytoskeleton via their cytoplasmic domains (Yoneda and Couchman 2003; Okina et al. 2009). These cytoplasmic domains can be divided into three regions: a conserved C1 membrane proximally and a conserved C2 membrane distally flank a variable (V) region that is unique to each syndecan, but nevertheless highly conserved across species (Multhaupt et al. 2009).
In the case of syndecan-2, the C1 region has been shown to interact with ezrin, whereas that of syndecan-3 binds Src kinase and cortactin (Granés et al. 2003; Kinnunen et al. 1998), signaling to the cytoskeleton. These C1 interactions apparently relate to endocytosis. Chen and Williams (2013) have shown that syndecan-1 raft-dependent endocytosis after clustering occurs in two steps. The first step involves the activation of extracellular signal-regulated kinase (ERK), which drives the dissociation of syndecan-1 from α-tubulin and its localization in the lipid rafts of the plasma membrane. In the second phase, the Src family kinases phosphorylate tyrosines in the transmembrane and C regions of syndecan-1, which leads to recruitment of cortactin, an essential mediator of actin-dependent endocytosis (Chen and Williams 2013).
Only in case of syndecan-4 is there substantial information about V region interactions. NMR spectroscopy was used to define a 12-amino acid region within the syndecan-4 V region that forms a twisted clamp dimer. Syndecan-4 binds the phospholipid PtdIns4,5P2 whereas the KKXXXKK motif in this region promotes dimerization (Oh et al. 1998; Whiteford et al. 2008) and provides a platform for the binding and activation of protein kinase Cα (PKCα). This interaction is very important for syndecan-4 signaling. In combination with integrins such as α5β1, syndecan-4 promotes the assembly of stress fibers and focal adhesions (Dovas et al. 2006; Bass et al. 2007). Since the small GTPases Rac and Rho are downstream of PKCα, syndecan-4 affects regulators such as the guanine nucleotide exchange factors (GEF), GTPase activators (GAPs), and guanine dissociation inhibitors (GDIs) (Morgan et al. 2007; Couchman 2010), which influence cell adhesion and migration via RhoA and Rac (Dovas et al. 2010; Bass et al. 2007, 2008). Besides PKCα, the 40-kDa syndesmos protein also binds to syndecan C1 and V regions (Denhez et al. 2002). The function of syndesmos is unclear besides its interactions with the focal adhesion protein, paxillin, and the related Hic5; the latter has been shown to be implicated in nuclear envelope decapping (Multhaupt et al. 2009). The third interacting partner of the V region is the α-actinin cytoskeletal protein, which binds syndecan-4 via its spectrin repeat 4 (Greene et al. 2003; Okina et al. 2009, 2012). Because α-actinin binds other focal adhesion proteins, such as vinculin and zyxin, this interaction provides a link between syndecan-4, focal adhesions, and the cytoskeleton.
The conserved C2 region has a hydrophobic motif terminating in a EFYA (glutamic acid-phenylalanine-tyrosine-alanine) motif or very similar residues. Through this motif, syndecans bind to PDZ domain-containing proteins, notably syntenin (mda-9, melanoma differentiation-associated gene-9) (Beekman and Coffer 2008). Syntenin has been shown to play a scaffolding role in Xenopus non-canonical Wnt signaling by binding the Fz7 receptor through its PDZ1 domain and syndecan-4 or -2 through its PDZ2 domain (Luyten et al. 2008). Syndecan-4 and -2 also interact with GIPC/semcap-1/synectin to regulate adhesion and migration (Tkachenko et al. 2005; Cai and Reed 1999; Gao et al. 2000). Synbindin, also known as trs23, interacts with the syndecan-2 EFYA motif and is involved in postsynaptic vesicle trafficking and dendritic spine maturation in neurons (Ethell et al. 2001). Finally, the calcium/calmodulin-associated serine/threonine kinase (CASK) binds all syndecans and neurexin (Hsueh 2006), and this binding influences its cellular distribution (Multhaupt et al. 2009).
Syndecans and the Wnt Signaling Cascades
The first in vivo evidence for GAG chain involvement in Wnt signaling came from experiments performed in Drosophila (Haerry et al. 1997). The Drosophila ska (suppenkasper) gene encodes UDP-glucose dehydrogenase, which produces glucuronic acid, an important constituent of HS and CS. Therefore, ska mutants failed to produce GAG chains on syndecan and had a cuticle phenotype similar to Wingless (Wg) mutants. The authors also observed genetic interactions between ska, wg and dsh (dishevelled) (Haerry et al. 1997). Supporting these in vivo data, it has been shown that GAG chain removal severely reduces Wg signaling in cultured Drosophila cells, which can be rescued by the exogenous addition of heparin (Reichman et al. 1996).
There are several indications for the involvement of syndecans in the canonical Wnt pathways. In C. elegans, syndecan-1 regulates growth factor signaling in the ventral to dorsal distal tip cell (DTC) migration by affecting Unc5 function through the regulation of EGL20/Wnt and growth factor distributions (Schwabiuk et al. 2009). It has been shown recently that C. elegans syndecan is required for orientation of the mitotic spindle in response to Wnt. Loss of either HS synthesis or syndecan core protein results in a misorientation of spindles of the ABar blastomere where the transient accumulation of syndecan is necessary for Dvl localization (Dejima et al. 2014).
It is well known that syndecan-1 null mice develop almost normally, but are resistant to mammary tumor development upon Wnt1 treatment; although, in this case, there is no direct evidence for the co-receptor activity of HS in Wnt signaling (Liu et al. 2003). By crossing syndecan-1 null mice with mice that overexpress effectors of the β-catenin/T cell factor Wnt pathway, the amplification of mammary progenitor cells as well as tumor development were reduced (Liu et al. 2004). In other experiments, syndecan-1 has been shown to modulate the triple-negative breast cancer stem cell phenotype through regulation of the Wnt and IL6/STAT3 pathways (Ibrahim et al. 2013). In this case, syndecan-1 silencing led to downregulation of LRP6 (Ibrahim et al. 2013). In neuronal progenitor cells (NCPs), which are important for the assembly of the layered mammalian cortex, knockdown of syndecan-1 in vivo reduces NCP proliferation and causes their premature differentiation through a reduction in β-catenin levels and prevention of its activation by Wnt3a, thereby affecting canonical Wnt signaling. This phenotype could be rescued by GSK3β inhibition (Wang et al. 2012). Recent evidence from Astudillo et al. (2014) shows that, in multiple mammalian cell lines and Xenopus embryos, syndecan-4 overexpression and fibronectin inhibit the Wnt/β-catenin pathway. This effect occurs at the LRP6 co-receptor level, as decreased syndecan-4 levels resulted in increased LRP6 activity (shown by its phosphorylation). Furthermore, syndecan-4 restricts the activity of RSPO3 (R-spondin 3) (Astudillo et al. 2014) with which it interacts and induces its clathrin-mediated endocytosis in PCP signaling (Ohkawara et al. 2011). In the canonical Wnt pathway, syndecan-4 restricts the activity of RSPO3, as depletion of HSPGs leads to induction of β-catenin by RSPO3. Results from the same group also suggest that syndecan-4 may decrease the agonist activity of RSPO3 in order to enhance β-catenin signaling through its cognate receptor, LGR4 (Astudillo et al. 2014) (Fig. 1).
Regarding the non-canonical pathway in vertebrates, previous studies have demonstrated that syndecan-4 regulates the Wnt/PCP pathway in Xenopus embryos (Munoz et al. 2006; Matthews et al. 2008; Ohkawara et al. 2011). Munoz et al. (2006) suggested a model where xSyn4 interacts functionally and biochemically with Frizzled7 receptor and its intracellular signal transducer, Dvl during convergent extension. xSyn4 proved to be “necessary and sufficient” for translocation of Dvl to the membrane (Fig. 2). Regarding neural crest (NC) cell migration in Xenopus and zebrafish, Matthews and colleagues demonstrated that syndecan-4, together with the Wnt/PCP pathway, controls the directional migration of NC cells in vivo by regulating membrane protrusions. By FRET (Förster resonance energy transfer) analysis, they have also shown that syndecan-4 inhibits Rac activity, whereas the Dvl/PCP pathway promotes RhoA activity, which inhibits Rac in NC cells (Matthews et al. 2008). These data suggest the existence of two parallel pathways leading to the same effect (Fig. 2). Also in Xenopus, syntenin has been shown to play a scaffolding role for non-canonical Wnt signaling by binding the Frizzled7 receptor through its PDZ1 domain and syndecan-4 or -2 through its PDZ2 domain (Luyten et al. 2008) (Fig. 2).
Syndecan-4 has been connected with Wnt/PCP signaling also during mouse development (Escobedo et al. 2013), as it interacts genetically with Vangl2/Strabismus to regulate neural tube closure, morphogenesis of cochlear stereocilia, and wound healing. In biochemical and cell culture experiments, Escobedo et al. (2013) demonstrated that Vangl2 regulates SDC4 steady-state levels and affects the total levels of expressed HSPGs. Additionally, they demonstrated that the Vangl2 Lp (loop tail) mutant embryos developed craniorachischisis when sulfation of GAG chains was inhibited. Furthermore, syndecan-4 knockout mice reveal defects in stereocilia organization in the inner ear, a hallmark phenotype of disturbed non-canonical Wnt signaling (Escobedo et al. 2013). Lp mutation also disturbs protein trafficking from the endoplasmic reticulum to the plasma membrane (Merte et al. 2010) and alters the localization of other PCP proteins like Vangl1 and Prickle-like2 (Yin et al. 2012); therefore, this mutation can additionally affect the PCP pathway independently of SDC4 (Fig. 2). It has been also suggested that Vangl2 regulates the cell-surface availability of MMP14 matrix metalloproteinase during gastrulation (Williams et al. 2012).
R-spondins (RSPO) are a family of secreted Wnt modulators with important roles in development. In Xenopus embryos, RSPO3 has been shown to bind syndecan-4 and together activate non-canonical Wnt signaling in gastrulation movements and head cartilage morphogenesis. During gastrulation, RSPO3/PCP signaling requires Wnt5a ligand and induces a Fz/Dvl/JNK signaling cascade. RSPO3 exerts its function by inducing syndecan-4-dependent clathrin-mediated endocytosis of the Wnt complex (Ohkawara et al. 2011). RSPO3 also plays a role in canonical Wnt signaling, as discussed earlier (Fig. 2).
Other evidence that SDC4 is involved in non-canonical Wnt signaling in mammals comes from a recent report where Bentzinger et al. (2013) demonstrated that syndecan-4 and Fz7 form a co-receptor complex in satellite cells, and that binding of fibronectin to syndecan-4 stimulated the ability of Wnt7a to induce the symmetric expansion of satellite stem cells in regenerating muscles.
The first evidence that a cell-surface protein can be ubiquitinated and degraded by the non-canonical pathway came from the laboratory of Juan Larraín (Carvallo et al. 2010). They have shown that Wnt5a reduces cell-surface levels and induces mono-ubiquitination of syndecan-4 in cell lines and during Xenopus gastrulation where Dvl plays a key role in regulating syndecan-4 steady-state levels (Carvallo et al. 2010).
Recently published work reveals the importance of syndecan-4 and calcineurin/NFAT pathways in the mammalian heart. Syndecan-4 null mice do not develop concentric myocardial hypertrophy following pressure overload and they have minimal activation of NFAT compared to matching wild type animals. Pull-down experiments have demonstrated a direct syndecan-4/calcineurin interaction initiated by Ser179 phosphorylation in the syndecan-4 cytoplasmic domain. This phosphorylation was increased during pressure overload (Finsen et al. 2011) (Fig. 3). They have also shown that patients with hypertrophic myocardium had higher syndecan-4 phosphorylation levels and NFAT activation, suggesting that syndecan-4 regulates mechanical stress-induced activation of the calcineurin-NFAT pathway in cardiomyocytes (Finsen et al. 2011). The same group also published the importance of syndecan-4 in the differentiation of cardiac fibroblasts into myofibroblasts after aorta banding since, in syndecan-4 null mice, the RNA levels of extracellular matrix components such as collagen I, collagen III, fibronectin as well as smooth muscle actin (SMA) were reduced in comparison to that in wild type mice. This phenomenon was dependent on syndecan-4 Ser179 dephosphorylation by calcineurin (Herum et al. 2013) (Fig. 3). In contrast, Echtermeyer et al. (2011) observed an enhanced activation of the NFAT pathway in syndecan-4 knockout mice. These data suggest an interference between syndecan-4 and the Wnt/calcium pathway, but more work is required to elucidate the details.
In human osteosarcoma cells, syndecan-2 has been identified as a Wnt target HSPG. The high activity of the Wnt/β-catenin pathway in these cells induces a constitutive repression of syndecan-2 transcription by TCFs, whereas Wnt/RhoA signaling blocks the amplification loop of syndecan-2 expression by regulating its shedding from the cell surface. Moreover, syndecan-2 regulates its own repression by inhibiting RhoA signaling (Dieudonne et al. 2010). Therefore, in this particular case, both Wnt pathways contribute to low syndecan-2 expression.
HS ligand binding capacity depends on the amount and patterns of sulfate groups added during HS synthesis in the Golgi apparatus by sulfotransferases, as well as by extracellular 6-O-endosulfatases called Sulfs (Couchman and Pataki 2012). Ai et al. (2003) have suggested a model to explain how the sulfation status of HS chains influences Wnt signaling, called “catch or present”. According to this model, the embryonic cells that do not express the QSulf1 possess sulfated HS chains on the cell surface that catch Wnt ligands with high affinity, competing with Fz receptors. QSulf expression decreases the 6-O sulfation of HS chains leading to decreased affinity towards Wnts and presentation of the Wnt ligand to its Fz receptor to initiate Wnt signaling (Ai et al. 2003). In Drosophila, however, Sulf1 negatively regulates the Wingless pathway by promoting Wg degradation (Kleinschmit et al. 2013).
In total, and based on our current understanding, syndecans have an impact on both the non-canonical and canonical Wnt pathways.
Implications for Pathological Conditions
Taking into account the ubiquitous cellular expression of syndecans and Wnt pathway components as well as their evolutionary conservation and role during development, it is not hard to imagine that the same pathways contribute to the development of various diseases. Most conventional knockouts targeting Wnt pathway components show very severe abnormalities with early embryonic or perinatal mortality (van Amerongen and Berns 2006). Although several tissue-specific conditional mutants have been made in the past 25 years, it is difficult to attribute their phenotype to a specific branch of the Wnt pathway because a) the activity of pathways is important in many different tissues; b) Wnt ligands show highly localized, temporally restricted expression with some degree of redundancy; c) most of the pathway elements have several homologs and many combinations of ligand–receptor binding can occur; and d) some of the components, like Dvl, are situated at the nexus of the canonical and non-canonical pathways. In addition, Wnt pathways intersect with many other pathways. Moreover, little is known about the redundancy between, for example, β-catenin and other members of the armadillo family, such as γ-catenin (plakoglobin) and p120-catenin. For example, it has been shown that plakoglobin can bind TCF/LEF and acts as a transcription activator in human embryonic kidney cells (Williams et al. 2000; van Amerongen and Berns 2006).
Several mutations in PCP pathway elements have been associated with human developmental disorders like craniorachischisis (Robinson et al. 2012), defects in the palate cleft (Yang et al. 2014), spina bifida (Kibar et al. 2007; Lei et al. 2010), cystic disease (Otto et al. 2003), and Meckel-Gruber and Joubert syndromes that are described as ciliopathies showing polydactyly, status inversus and cystic kidney disease (Barker et al. 2014; Leightner et al. 2013).
Altered expressions of PCP proteins have been shown in chronic lymphatic leukemia or acute myeloid leukemia (Kaucká et al. 2013; Prebet et al. 2010), promoting tumor metastasis. β-Catenin nuclear accumulation has been described in schizophrenia patients (Mao et al. 2009) and variant alleles of the LRP6 receptor have been associated with Alzheimer’s disease in a population-based linkage study (De Ferrari et al. 2007). Syndecan-2 was shown to be enriched in the area of the brain affected by Alzheimer’s disease, although this has not so far been coupled to Wnt signaling (van Horssen et al. 2001). Abnormal β-catenin activation has been shown in a range of cancers. One prominent example is colon cancer where mutations to APC activate β-catenin (Morin et al. 1997). In the case of head and neck squamous cell carcinoma (HNSCC), β-catenin nuclear accumulation has been correlated with radiation resistance (Chang et al. 2008). Ma and colleagues published that Wnt3a, β-catenin and LEF1 were consistently upregulated in human osteosarcoma cell lines (Ma et al. 2013). In contrast, APC and β-catenin mutations rarely appear in lung cancer but overexpression of Dvl, Wnt1 and Wnt2 have been correlated with non-small cell lung cancer (He et al. 2004; Uematsu et al. 2003; You et al. 2004). Wnt5a and Fz7 were shown to be upregulated in breast cancer (Lejeune et al. 1995; Yang et al. 2011) but Wnt5a can act as a tumor suppressor in some cancer types, such as neuroblastoma (Blanc et al. 2005), acute myeloid lymphoma (Martín et al. 2010), acute lymphoblastic lymphoma (Roman-Gomez et al. 2007), thyroid carcinoma (Kremenevskaja et al. 2005), colon carcinoma (MacLeod et al. 2007) and esophageal squamous cell carcinoma (Li et al. 2010). Conversely, Wnt5a is a proto-oncogene in prostate cancer (Wang et al. 2010), breast cancer (Pukrop et al. 2006), melanoma (Weeraratna et al. 2002), and pancreatic cancer (Ripka et al. 2007), showing the importance of the Wnt/calcium pathway in cancer. Many other studies involving Fz receptors have been extensively reviewed by Ueno and colleagues (Ueno et al. 2013).
Unlike Wnts, knockouts of syndecans-1, -3 and -4 are mild, with few developmental defects (Bishop et al. 2007; Couchman et al. 2015). Postnatal defects in tissue repair are apparent in the syndecan-1 and -4 null mice but, overall, these data support a hypothesis that there may be redundancy across the syndecans, at least through development. This is reinforced by multiple observations that genetic ablation of enzymes involved in HS synthesis can have severe and often lethal effects (Bülow and Hobert 2006; Bishop et al. 2007). Moreover, there are no syndecan mutations associated with human pathologies (Couchman et al. 2015). However, a recent report suggests that syndecan-1 polymorphism is associated with breast cancer in an Australian cohort (Okolicsanyi et al. 2014). With respect to the co-ordination of Wnt signaling, it is conceivable that there is redundancy not only across the syndecans but this might extend to the glypicans, which although structurally unrelated are nevertheless important, widespread, HSPGs.
As in the case of Wnt signaling, tissue context is very important for syndecan function. For example, syndecan-3 may be pro-inflammatory in rheumatoid arthritis but anti-inflammatory in skin and muscle inflammation (Kehoe et al. 2014). Syndecans have been shown to be deregulated in different cancer types, not surprisingly, given their role in cell–cell and cell–matrix adhesion and migration. It has been also noted that the syndecan ectodomain can be shed from the cell surface and can bind to different receptors competing with growth factors (Manon-Jensen et al. 2010). Wnt signaling in cancer does not always correlate with reduced patient survival; e.g., in melanoma, sarcomas, ovarian, prostate and colorectal cancers, as summarized by Zimmerman et al. (2012). Similarly, the role of syndecans in tumors depends on the tumor stage and type. Syndecan-1 may be downregulated in some types of breast cancers (Beauvais and Rapraeger 2004) but can be upregulated in others, with stromal syndecan-1, in particular, an indicator of poor prognosis (Barbareschi et al. 2003; Lendorf et al. 2011). Syndecan-1 modulates the triple-negative breast cancer stem cell phenotype via LRP6 and IL-6-mediated STAT3 signaling (Ibrahim et al. 2013). Moreover, development of the mammary gland requires syndecan-1 to create a β-catenin/TCF-responsive mammary epithelial cell population; although, this process is independent of Wnt signaling (Liu et al. 2003). The cancer cell environment has a great impact on the behavior of cells, as reviewed in detail by Theocharis et al. (2014, 2015). Syndecan-1, for example, serves as a collagen receptor in squamous cell carcinoma where, together with α2β1 integrin, it promotes cell adhesion. Lowering the levels of syndecan-1 enhanced the cell spreading and motility via the RhoA/Rock pathway, but only on collagen 1 substrates (Ishikawa and Kramer 2010). In orthotopic mouse models of breast cancer, it has been shown that the exosomes secreted by cancer-associated fibroblasts are internalized by breast cancer cells and associate with Wnt11 produced by cancer cells to initiate PCP signaling and increase motility and in vivo metastasis (Luga et al. 2012). The authors show that Wnt/PCP signaling promotes breast cancer metastasis since all the tested core PCP components, such as Fz6, Dvl1, Vangl1 and Pk1 were critical to initiate the protrusive activity and migration of breast cancer cells. This work reveals an intercellular communication whereby exosomes produced by remote fibroblasts mobilize autocrine Wnt/PCP signaling to drive breast cancer metastasis.
Syndecan-1 is also upregulated in pancreatic cancer, whereas the shed form of syndecan-1 is upregulated in myelomas (Seidel et al. 2000) and lung cancer (Joensuu et al. 2002). Syndecan-1 and -4 seem to be important components of the Wnt5a autocrine signaling loop in metastatic melanoma cells (O’Connell et al. 2009). Syndecan-2 also plays a dual function in cancer. It has been reported to be upregulated in colon cancer, Lewis lung carcinoma, ovarian and prostate cancers, melanoma, osteosarcoma and glioma (Munesue et al. 2002, 2007). In breast cancer, syndecan-2 regulates the actin cytoskeleton via p190A Rho GAP and suppresses syndecan-4, as published recently by Lim and Couchman (2014). In osteosarcoma cells, syndecan-2 has been identified as a Wnt pathway target. In these cells, canonical Wnt signaling and also Wnt/RhoA signaling lead to low syndecan-2 transcription and resistance to chemotherapy (Dieudonne et al. 2010). Syndecan-4’s expression is altered in breast cancer, melanoma, malignant mesothelioma and hepatocellular carcinoma (Couchman et al. 2015). In hepatocarcinogenesis, the canonical and non-canonical Wnt pathways have complementary roles. β-Catenin activation initiates the tumor, whereas the non-canonical pathway contributes to tumor progression (Yuzugullu et al. 2009; Pez et al. 2013). Leocata et al. (2007) suggest that syndecan-1 and Wnt1 can be involved in promoting the proliferation and invasion in intraosseous ameloblastomas in odontogenic epithelium. The activated canonical Wnt pathway has been implicated in fibrosis in many different organs, such as the lungs, kidneys and liver, indicating its role in wound healing as well (Chilosi et al. 2003; Surendran et al. 2005; Königshoff et al. 2008, Miao et al. 2013). At early stages after injury, the β-catenin pathway has been shown to be rapidly activated (Petersen and Reddien 2009). During inflammation, however, the Wnt5a/Fz5 pathway has a major role, as described in detail by De (2011). Inhibition of Wnt signaling during mammalian epidermis wound healing prevents the formation of hairs and sweat glands and causes further scarring of the skin; this can be rescued by elevating Wnt signaling (Ito et al. 2007). Syndecan-4 has been shown to be upregulated in a series of inflammatory conditions, such as ischemic myocardial injury (Cizmeci-Smith et al. 1997) and wound closure (Gallo et al. 1996). Wnt signaling has crucial functions during heart development and recovery after injury through the renewal and differentiation of cardiac progenitors via the β-catenin pathway (Qyang et al. 2007; Deb 2014). Moreover, in the heart, a connection between syndcan-4 and calcineurin/NFAT signaling has been published, as discussed earlier. In addition, NFAT has been associated with Down’s syndrome (Arron et al. 2006). Shed syndecan-1 levels are elevated in the dermal wound fluid from patients with acute graft-versus-host disease (Seidel et al. 2003). In chronic inflammation, endothelial syndecan-1 seems to play a negative regulatory role in leucocyte adhesion and migration (Teng et al. 2012; Zhang et al. 2013). Wnt signaling has a role in angiogenesis, as Wnt1 and Wnt5a have been shown to increase survival and proliferation of endothelial cells (Masckauchan et al. 2005, 2006). As can be appreciated, syndecans and Wnt pathways are involved in a multitude of developmental processes and pathologies that arise from wound healing, inflammation, angiogenesis and cancer. There are many indications that the components of these two pathways can serve as important cross-points in different diseases; however, mapping all of the connections between syndecans and the Wnt pathways will require future investigations.
Future Perspectives
Thousands of studies have appeared regarding the Wnt pathways in the past 30 years and the syndecan family in the past 25 years since their discovery. These studies describe the structure of the molecules, their localization and expression from primitive organisms to humans, their mapping into different pathways, and their various functions in development and disease. Despite the intensive study of syndecans and Wnt pathways, the cross-talk between them is less explored. Given their co-receptor and receptor functions, syndecans influence a large variety of signaling pathways, and thus the Wnt signaling pathways may exhibit cross-talk not only between each other but also with other pathways involving hepatocyte growth factor, Notch, mitogen-activated protein kinase 1, the Ras cascade, and bone morphogenetic proteins, among others. Moreover, our understanding of how the cellular and tissue contexts influence these pathways is rudimentary. These problems make it very difficult to specifically target pathways to develop therapeutic strategies for disease. Further experiments are needed to elucidate the more precise function of syndecans in different processes such as inflammation, proliferation, apoptosis and their potential role in the nucleus in specific tissue environments (Stewart et al. 2015). There is also a gap in the literature regarding the phenotypic characterization of syndecan-2 knockout mice. In the case of Wnt pathways, descriptions of conditional knockout mice is missing and the characterization of existing models is incomplete. Furthermore, the exact interactions between Wnt and other pathways also need more work in order to fully understand their roles in development and disease.
Footnotes
Competing Interests
The authors declare no potential competing interests with respect to the research, authorship, and/or publication of this paper.
Author Contributions
All authors contributed to this review as follows: CAP has written the manuscript and prepared the figures; JRC has edited the manuscript and participated in discussions; JB proofread the manuscript and participated in discussions.
Funding
The authors have received the following financial support for the research, authorship, and/or publishing of this article: CAP and JB were supported by project BIOCEV (
