Abstract
Proteoglycans, with their core proteins and attached glycosaminoglycan chains, are recognized as important partners in many biological processes, yet often experimental analysis of their molecular action is considered for only part of these molecules: either the protein or the carbohydrate unit. In this article, we have tried to summarize, with an example of the syndecan family in general and more specifically with syndecan-1, what is known considering the mutual influence of these different components, and we follow whether the nature of the glycosaminoglycan chains matters for these effects.
Keywords
The syndecans are transmembrane proteoglycans (PGs) with the ability to connect the inside of a cell with its surrounding (Couchman 2010). Four isoforms exist in vertebrates with syndecans-1 to -3 expressed in a tissue-specific manner, whereas syndecan-4 is more widely found in various cell types (Kim et al. 1994). The four isoforms are well conserved in their transmembrane and cytoplasmic domains, with the exception of a variable stretch in the intracellular domain. The extracellular domains, containing the attachment sites for glycosaminoglycans (GAGs), are, however, highly variable within the family (Couchman 2010), having different numbers and types of chains attached. Considering cell- and tissue-specific glycosylation (Ledin et al. 2004), the latter fact should not surprise; but what are the implications of such differences for molecular interactions of their carriers? In this perspective, we aim to summarize the current understanding of the effect(s) of glycosylation on the function of the core protein and vice versa.
Syndecan-1 and Its GAG Chains
Syndecan-1, a membrane PG originally isolated from mouse mammary epithelial cells (Rapraeger and Bernfield 1983), is the first hybrid-type PG described bearing both heparan sulfate (HS) and chondroitin sulfate (CS) chains; the latter are shorter at least in mouse mammary epithelial cells (Rapraeger et al. 1985). The notion that GAG chains, particularly the HS chains, would anchor the cells to the extracellular matrix (Rapraeger et al. 1985) was raised in the “early days” of syndecan. A comparison of syndecan-1 from simple and stratified epithelia showed that molecular heterogeneity between the two samples depends largely on the fact that the same core protein is substituted with more and longer GAG chains in the simple epithelium, which may account for differences in how these two tissue types are organized (Sanderson and Bernfield 1988). A reduction in size of the GAGs is also responsible for the polymorphism of syndecan-1 apparent during lung development (Brauker et al. 1991). By 1992, it had been accepted that the GAG chains of syndecans were critical for their function, and it was appreciated that syndecans from different cell types exist as discrete isoforms with variability in type, number, and size of GAG chains attached, resulting in a vast variety of interaction opportunities (Bernfield et al. 1992). The role of HS chains is seen to attract and present different proteins at the cell surface, whereas the role of CS chains is speculated to modify interactions between syndecans and other proteins.
What Defines Where on Syndecan What Type of GAG Is Attached?
Both HS and CS chains are initiated with the same linkage sequence of four sugar units that are O-glycosidically linked to a serine residue in the core protein, -GlcA-Gal-Gal-Xyl-O-Ser (where GlcA, Gal, Xyl, and Ser stand for D-glucuronic acid, D-galactose, D-xylose, and L-serine, respectively). As such, efforts have been undertaken to understand what drives the extension of either of these chain types. Two main aspects may be considered that determine where and which GAGs are attached to the core protein. Features of the core protein itself, that is, the primary sequence surrounding the potential GAG attachment site, the protein’s conformation and its relative abundance, and on the other hand, the capacity of the cellular biosynthetic machinery to produce these chains (Zhang et al. 1995). Thus, the protein has been an early target for studies, whereas the impact of the biosynthetic machinery of the respective cell is still more of an enigma, as less is understood as to how the biosynthetic enzymes are organized and controlled in the Golgi apparatus. Studies with chimeric syndecan-1/Protein A constructs in Chinese hamster ovary cells provide evidence that HS chains are extended on the linker sequence situated in hydrophobic pockets of the core protein (Zhang et al. 1995). As a minimal consensus, serine-glycine repeats will promote the action of xylosyltransferases for attachment of the linkage sequence on murine syndecan-1. Acidic amino acids surrounding these Ser-Gly repeats are necessary but not sufficient for HS attachment, further promoted by additional aromatic amino acids (Zhang and Esko 1994). Increasing the number of Ser-Gly repeats promotes substitution with HS as well, possibly by helping the first GlcNAc-transferase to act on the linkage sequence (Zhang et al. 1995; Romaris et al. 1999). The picture emerges that either HS or CS may substitute the three sites positioned in the N-terminal part of syndecan-1, whereas the two membrane-proximal sites are substituted with CS.
The biosynthetic machinery clearly has an influence on the substitution pattern. Intermediary modifications of the link sequence by sulfation of galactose and phosphorylation of xylose have been suggested as markers for either CS or HS extension (Ueno et al. 2001). Even though the two types of GAGs are synthesized by two different ensembles of enzymes, probably in different compartments of the Golgi, synthesis of sulfated GAGs is interlinked not only by the common linkage sequence as the acceptor site but also through access to common building blocks (sugar units, sulfate donor PAPS); thus, substitution may occur in a competitive state for substrate sites possibly with priority for HS (Uhlin-Hansen and Yanagishita 1993; Presto et al. 2008). Limitation of one or the other building block component may therefore lead to a selective substitution pattern in a given context. Indeed, syndecan-4 (ryudocan), produced in murine L cells or early passages of human endothelial cells, has three attachment sites for GAGs that seem to be occupied with either HS or CS without preference, a promiscuity that derives from the core protein (Shworak et al. 1994). Pointing in the same direction are studies that CS chains purified from syndecan-1 are both structurally and functionally distinct from CS chains attached to syndecan-4 in NMuMG cells, whereas HS chains purified from these two core proteins from the same source are indistinguishable (Deepa et al. 2004). HS chains from sydecan-1 and -4 from the same cell do not differ (Zako et al. 2003), whereas HS chains from the same core protein isolated from different cells do, with implications for the interaction potential of the respective molecules (Kato et al. 1994; Sanderson et al. 1994). Another interesting influence of the position of the GAG attachment site for the HS chains produced has been demonstrated with syndecan-4 constructs that contain variable numbers and sites of GAG attachment. Here, there is no effect of the position within the protein on polymerization of the chains, yet the degree of modification seems to be in relation to the position of the chain on the protein, with higher sulfation identified at positions further from the N-terminus (Gopal et al. 2010).
How Can GAG Chains and Core Protein Affect Each Other?
With a cell- and tissue-specific expression of different isoforms of the core proteins and a dynamically regulated production of their attached GAG chains, the functional possibilities of these molecules not only cover the functions of the individual parts but also provide a mutual impact that may not always be easy to analyze in an unambiguous manner. A GAG chain will have binding capacity for different ligands depending on its structural properties, but, in addition, the scaffold function of the core protein will affect the binding by providing polyvalent arrangements or localizing the chains at defined sites within the plasma membrane (Fig. 1A). This will result in interactions taking place in, for example, lipid rafts or at sites of focal adhesions. Furthermore, depending on the core protein to which the GAG chains are attached, their position will be different for their interaction partners. This relative positioning of the GAG chains closer to or further away from the plasma membrane, in tight or loose association with a receptor or other cellular components, could affect the GAG function more than other structural details of the GAG chain itself. The extracellular part of syndecan can also get released into the surroundings (Fig. 1B). This event removes the ectodomain with the attached GAG chains and any bound ligands from the cell surface, which will not only affect the availability of, for example, growth factors for their cell surface–positioned receptors, but will also create new interaction possibilities for both GAG chains and the core protein due to the relocalization of the shed molecule. The released ectodomains also have the ability to compete with any binding partner of GAG chains on syndecan molecules still in the plasma membrane on both the same and the surrounding cells. The core protein, on the other hand, has properties of its own, whether this is in interaction with itself or different molecules, its synthesis, transport, localization or turnover (Fig. 1C). The presence or absence of a GAG chain, at specific or variable sites, will surely have an impact on such variables, both by steric hindrance and by allowing for direct interaction with both components.
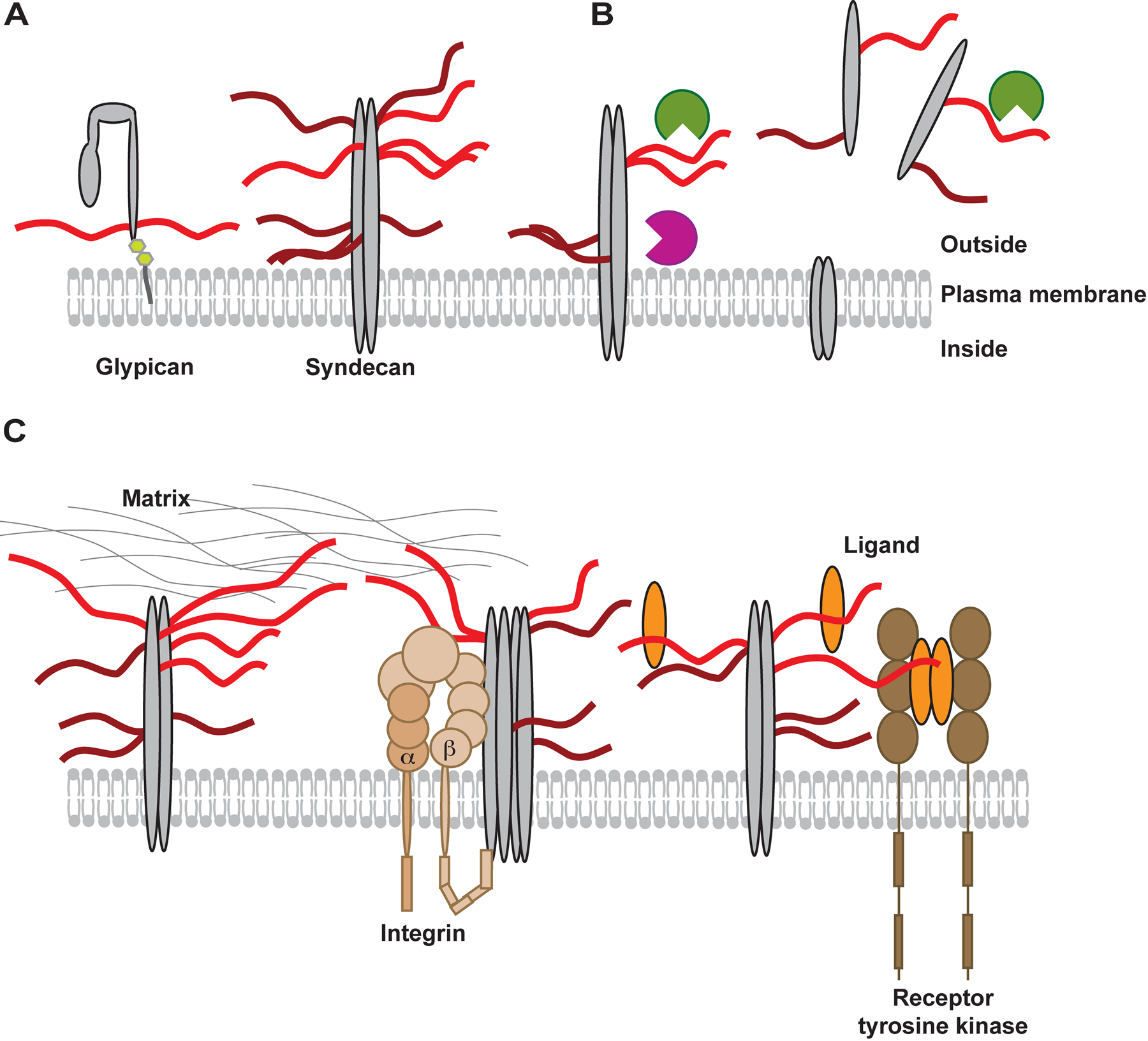
(A) The localization of the glycosaminoglycans (GAGs), heparan sulfate (HS) (light red), and chondroitin sulfate (CS) (dark red) in the glycocalix is dependent on which core protein (in gray shades) the chains are attached to. Due to structural differences of proteoglycans (PGs) (e.g., glypicans [glycosylphosphatidylinositol anchored] and syndecans [transmembrane PG]), the GAGs will be closer to or more distant from the plasma membrane, affecting the range for interaction. (B) In a dynamic cellular environment, the appearance of proteins and GAGs will change over time, affected by both the location of the core protein within the membrane domains and by the actions of sheddases (purple), which act on the core proteins to release intact ectodomains containing attached GAG chains, and endoglycosidase heparanase (green) that cleaves off parts of HS chains outside the cell. (C) Syndecans are known to interact with a vast number of extracellular matrix molecules such as collagen, fibronectin, or laminin; these interactions are stimulated by multivalent binding to more than one HS chain. Syndecans can also interact with membrane-intercalated receptors such as integrins (light brown), controlling adhesive properties of the cells. GAGs could help to enforce such interactions by crosslinking, for example, integrin ligands or by sterically interfering with the close association between PGs and other membrane proteins. The dimers of syndecan are able to further oligomerize, which may be affected by their glycosylation state. Another example of the ternary complexes is formed between fibroblast growth factors (FGFs) (orange), FGF receptors (brown), and HS on, for example, syndecan-1. In this case, it is likely that characteristics of the core protein, the extent of glycosylation, and the fine structure of the attached GAG chains have an impact on cellular response to FGF stimulation.
The Roles of HS and CS for Syndecan Interactions
HS chains have been shown to interact with a variety of matrix molecules (Koda et al. 1985; Saunders and Bernfield 1988) and diverse growth factors (Burgess and Maciag 1989; Robinson and Stringer 2001). Syndecan interaction with, for example, laminin can depend uniquely on the attached GAG chains as demonstrated for syndecan-1, whereas syndecan-4 interactions with laminin require both the core protein and the GAG chains (Okamoto et al. 2003; Carulli et al. 2012). Also, receptors for these matrix molecules (i.e., integrins) can interact with PGs. The syndecan-1 core protein binds via the extracellular domain to, among others, both αvβ3 and αvβ5 integrin during angiogenesis (Beauvais et al. 2009) or with α2β1 integrin during re-epithelialization of lung tissue (Chen et al. 2009), thus potentially forming ternary complexes between an extracellular molecule, a cell surface receptor, and a PG component, similar to the ones well recognized for growth factors, their corresponding receptors, and HS chains (Harmer 2006; Jastrebova et al. 2010). In these latter cases, HS chains in relation to their structural properties have been demonstrated to affect the signals imposed on the cell by the growth factor. In stromal fibroblasts of breast carcinomas, the HS chains on syndecan-1, rather than the ectodomain of the core protein, mediate the paracrine fibroblast growth factor 2 (FGF-2)–dependent signaling to stimulate growth (Su et al. 2007). CS (and the related dermatan sulfate [DS]) chains, on the other hand, also bind to matrix proteins as well as to soluble molecules (Yamada and Sugahara 2008). Although most focus has been put on the role of HS in syndecan, an example from Drosophila melanogaster suggests a selective role for CS in Slit/Robo-driven axon guidance (Chanana et al. 2009). This ligand/GAG interaction is dependent on GAG attachment to a core protein, and the ensuing polyvalent arrangement of the chains has been shown to affect cell invasion for syndecan-1 binding to collagen (Langford et al. 1998). A similar finding has also been observed in the case of syndecan-4, where assembly of focal adhesion sites on fibronectin is strictly dependent on the multivalent appearance of HS chains on the core protein to permit correct organization of the cellular cytoskeleton (Gopal et al. 2010). Not only interaction with extracellular matrix structures but also binding of soluble effector molecules may be potentiated by the cooperative interaction of both HS and CS immobilized on syndecan-1, as demonstrated for midkine and pleiotrophin (Deepa et al. 2004). Here, the value of a polyvalent arrangement, provided by the immobilization of the GAG chains on the core protein, is apparent.
The Scaffold Function of Syndecan-1 Core Protein and Its Shedding
Another potential impact of the core protein is the different types of scaffolds it provides (Liu et al. 1998). Indeed, FGF-2–mediated signaling is initially dependent on syndecan-1, but after removal of syndecan from the cell surface, this function is taken over by glypican (Ding et al. 2005). Thus, two properties of the core protein have an impact. First, the dimension of the core protein and the localization of the GAG attachment sites give HS chains on syndecan a preferential positioning, as compared with those attached to glypican (Fig. 1A). The second relates to the different localization and turnover of these two core proteins, where syndecan is removed from the cell surface in a different manner and timed in another way than glypican.
A common feature of the syndecans is the removal of the extracellular part by proteases, a phenomenon referred to as shedding (Lambaerts et al. 2009). This event releases the ectodomain, including attached GAG chains and any bound ligands, from the cell surface into the extracellular surrounding (Fig. 1B), where it can act as an effector molecule (Wang et al. 2005) influencing the radius and mode of action of its cargo (mostly GAG)–bound ligands, in a similar manner to that performed by heparanase through its endolytic cleavage of HS chains (Sanderson and Yang 2008). The shed ectodomain can thereby compete with the remaining PGs on the cell surface for ligands in the extracellular matrix. Shed ectodomains have been demonstrated to inhibit cell stimulation through reliance on their HS chains and do so more efficiently than free HS chains, suggesting a structural impact of either the protein on the GAG chains or the structural properties of the tested free HS chains needed for ligand-receptor interaction (Mali et al. 1994; Kato et al. 1998). Simultaneously, shedding also detaches the cell from its surrounding matrix, thereby promoting cell migration (Bass et al. 2009; Nikolova et al. 2009). The proteases responsible for shedding belong to the family of matrix metalloproteinases (MMPs), particularly MMP-7, -9, and -14, and ADAMTS. The cleavage site consists of two basic amino acids, arginine and lysine, close to both the plasma membrane and the membrane-proximal GAG attachment sites (in the case of syndecan-1). Different stimuli can increase the shedding, such as growth factors, microbes, chemokines, and cellular stress, but the regulation of this process is not yet clear. A recent study suggests that the HS chains themselves regulate the shedding of syndecans. By treating various cell types with heparin lyase to remove attached HS chains, the amount of syndecan-1 ectodomain in conditioned medium is increased, suggesting that less HS on the core protein will increase the shedding (Ramani et al. 2012), an observation similar to the finding that enhanced heparanase activity also leads to increased sensitivity of the core protein for attack by proteases (Yang et al. 2007). An obvious explanation is the steric hindrance that GAGs can provide, so that the cleavage site is more or less available for these sheddases. Notable in this context is the position of the membrane-proximal GAG attachment sites at positions 205 and 215 relative to the protease target site at positions 243–247 (Wang et al. 2005). Thus, increasing the substitution of these sites, preferentially with CS chains, would be expected to have an impact (so far not tested) on the sensitivity of syndecan for proteolytic attack. Another interesting connection in this triad is the fact that some of the MMPs have been shown to bind to HS and CS themselves, potentially bringing the proteases closer to the substrate site and thus further promoting their mutual influence (Gao et al. 2004).
A third property of the core protein that will crucially affect PG interactions and be affected by the presence of GAGs is the property of self-association. Syndecan-1—as all syndecans—forms homodimers driven by a conserved, cytoplasmic sequence close to the plasma membrane (Tkachenko et al. 2005) and a GXXXG-motif within the transmembrane domain (Dews and Mackenzie 2007). During oligomerization, it is not unlikely that any of the GAG chains on the core protein can help or hinder the process, potentially with different roles for HS and CS and some dependence on their location on the protein (Fig. 1C). As such, it is feasible that oligomerization could be induced by extracellular ligands able to crosslink GAG chains attached to different core proteins. On the other hand, substitution of the membrane-proximal GAG attachment sites could not only impart a steric hindrance for tight association of syndecan core proteins with themselves but also affect interactions with other membrane-intercalated proteins, such as growth factor receptors or integrins. And finally, the tight association of the core protein will endow the GAG chains with an enhanced multivalent appearance, as described above.
What Are the Physiological and Pathological Consequences?
These diverse molecular details will have an influence on the physiological and pathological roles of syndecan-1. Although the functions of syndecan-1 during embryogenesis and development may be shaded by redundancy through other syndecan family members (Xian et al. 2010), the impact of syndecan-1 during wound healing is apparent by increased expression and shedding into the wound, engaging elements of the PG core, GAG-attached growth factors, and sheddases (Elenius et al. 1991; Kato et al. 1998). Lack of syndecan-1 has been associated with enhanced mesenchymal appearance (Kato et al. 1995) and poorly differentiated tumors (Nackaerts et al. 1997), whereas overexpression leads to epithelialization of cells, thus suggesting that syndecan-1 has a role in epithelial-mesenchymal transformation (EMT), important during both embryogenesis and different pathological conditions (Li et al. 2002; Nikolova et al. 2009; Masola et al. 2012). Conversely, increased shedding of syndecan-1 has been associated with poor prognosis for tumor metastases (Purushothaman et al. 2010). The observed different properties of the ectodomains as a stimulator of tumor growth (Yoneda et al. 2012), an inducer of apoptosis (Dhodapkar et al. 1998), or an immune modulator (Li et al. 2002), exploited also by microbes (Park et al. 2001), may hint at a molecular heterogeneity affected by the cellular source of syndecan and its glycosylation.
Perspectives
The impact of syndecan-1 and its GAG chains on the cellular community in a tissue ensemble is multiple and interconnected. As presented in this summary, some attempts have been made to dissect these complex relations. Yet, it may not always be possible to pinpoint a singular effect and, whenever altered, a change may have an influence on various cellular parameters. Thus, an understanding and potential clinical exploitation of the molecular interactions that control cellular activities require that glycosylation is taken into consideration when experimental data are interpreted.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge support by the Swedish Cancer Foundation and the Foundation for Proteoglycan Research at the University of Uppsala.
