Abstract
The removal of excess cytoplasm from elongated spermatids by Sertoli cells is the last essential step in spermatogenesis. It requires cell-to-cell recognition between a Sertoli cell and an elongating spermatid through protein-protein interactions. CEACAM2-L, an adhesion molecule of the immunoglobulin superfamily (IgSF), is present at the plasma membrane covering the excess cytoplasm of elongated spermatids, and is possibly involved in the cell-to-cell recognition. In this study, we investigated the interaction between CEACAM2-L and Poliovirus receptor (PVR), which is also from the IgSF and is expressed by Sertoli cells. Immunohistochemical analysis showed that CEACAM2-L expressed on elongated spermatids was in close contact with PVR-positive cell processes of Sertoli cells. Immunoprecipitation experiments both in vivo and in vitro demonstrated a direct heterophilic interaction between CEACAM2-L and PVR. We show that the N-terminal Ig domain of CEACAM2-L was critical for its interaction with PVR. In addition, we found that CEACAM2-L formed heterophilic trans-tetramers with PVR in transfected COS-7 cells. From these data, we propose that Sertoli cells recognize the excess cytoplasm of elongated spermatids through the PVR-CEACAM2-L interaction in mouse testis.
Introduction
Spermatogenesis is a complex process that occurs within the seminiferous epithelium (SE), and consists of spermatogonial proliferation, meiosis, and spermatid differentiation (spermiogenesis). During spermiogenesis, round spermatids undergo drastic changes in morphology, including the formation of a flagellum and acrosome, nuclear condensation, and cytoplasmic removal (Leblond and Clermont 1952; Clermont 1972).
Spermatogenic cells interact or form direct contacts with Sertoli cells via adhesion molecules—many belonging to the immunoglobulin superfamily (IgSF)—expressed by both cell types, which is required for spermatogenesis (Mruk and Cheng 2004). Desmosome-like junctions connect Sertoli cells to spermatogonia, spermatocytes, or round spermatids, whereas ectoplasmic specializations (ES) localized at the apical regions of Sertoli cells form an attachment with the head of elongated spermatids (Russell 1977; Cheng and Mruk 2002; Mruk and Cheng 2004). Present at ES are the molecules NECTIN-2 and -3 (also known as poliovirus receptor-related [PVRL]-2 and -3, respectively), cell adhesion molecule (CADM) 1, junctional adhesion molecule (JAM)-B and -C (also known as JAM2 and 3, respectively), coxsackievirus and adenovirus receptor (CAR), and carcinoembryonic antigen-related cell adhesion molecule 6-L (CEACAM6-L) (Ozaki-Kuroda et al. 2002; Bouchard et al. 2000; Mueller et al. 2003; Inagaki et al. 2006; Fujita et al. 2006; Surace et al. 2006; van der Weyden et al. 2006; Yamada et al. 2006; Wakayama et al. 2007; Gliki et al. 2004; Wang et al. 2007; Mirza et al. 2006; Kurio et al. 2008). An additional interaction occurs at the apical region of SE, which is essential for the production of slender spermatozoa, where Sertoli cells recognize and phagocytose the excess cytoplasm of elongated spermatids, leaving residual bodies. This is followed by the release of spermatozoa from the SE into the lumen of the seminiferous tubule (i.e., spermiation) (O’Donnell et al. 2011).
We previously reported that CEACAM2-L—a novel member of the IgSF present at the plasma membrane overlying the cytoplasm of elongated spermatids (during steps 13–16)—could be involved in cell-cell adhesion between elongated spermatids and Sertoli cells within the mouse testis (Salah et al. 2012), specifically in the recognition and engulfment of the excess cytoplasm by Sertoli cells. To examine this possibility, in this study we identified molecules present on Sertoli cells that can form a complex with CEACAM2-L. These molecules were PVR, an IgSF member that is highly expressed in immunocompetent cells (Iwasaki et al. 2002) as well as in many cancer cell types and cell lines (Denis 1998) and binds heterophilically to other IgSF molecules such as PVRL-3 (Ikeda et al. 2004) and cluster of differentiation (CD)44 (Freistadt and Eberle 1997). The results in this study demonstrate that PVR binds to CEACAM2-L and potentially mediates the interaction between Sertoli cells and spermatids that is required for cytoplasmic removal and spermatid maturation.
Materials & Methods
Animals
Animal experiments were approved by the animal experimental committee of Kyushu University. Experiments were conducted in accordance with the National Research Council publication, “Guide for Care and Use of Laboratory Animals”. Animals for experiments were obtained from Kyudo Company (Fukuoka, Japan).
Antibodies
Affinity-purified rabbit anti-CEACAM2-L and rat anti-PVR antibodies were characterized previously by Salah et al. (2012) and Wakayama et al. (2007), respectively. Rabbit anti-PVR antibody was purchased from Sigma-Aldrich (St. Louis, MO). Rabbit anti-E-Cadherin antibody was obtained from ECM Biosciences (Versailles, KY). Antibodies were diluted with blocking buffer (PBS containing 5% skim milk and 0.05% Tween-20) before use.
Immunohistochemistry
Adult mouse testes were fixed at 4C for 4 hr in PBS containing 4% paraformaldehyde, washed three times in PBS, incubated in PBS containing 50 mM NH4Cl for 30 min, and then rinsed in PBS. After infiltration with 20% (w/v) sucrose in PBS, the testes were immersed in OCT compound (Tissue-Tek, Miles Inc, Elkhart, IN) and immediately frozen using liquid nitrogen. Frozen sections of 10 µm thickness were cut by a cryostat (CM1850; Leica, Nussloch, Germany). The sections were attached to poly-L-lysine-coated glass slides, washed in PBS, and exposed to 0.1% Triton X-100 in PBS before immunostaining. The sections were double immunostained for 2 hr with anti-CEACAM2-L antibody diluted 1:300 and rat anti-PVR antibody diluted 1:400, followed by incubation for 1 hr with goat anti-rabbit IgG conjugated with fluorescein isothiocyanate (FITC, GE Healthcare BioScience Corp., Piscataway, NJ) and anti-rat IgG conjugated with Cy3 (GE Healthcare BioScience Corp.). For nuclear staining, immunostained samples were incubated for 30 min with PBS containing Hoechst 33342 (Sigma-Aldrich). The samples were washed with PBS before examination by a fluorescence microscope (DMRXA, Leica, Wetzlar, Germany). For control staining, the primary antibody was replaced by pre-immune serum.
Preparation of Mechanically Separated Spermatids for Immunostaining
Fixed mouse testes were decapsulated in PBS and the seminiferous tubule was mass cut into small pieces. The pieces were transferred into a 10-ml tube containing 5 ml of PBS. Tissue pieces were gently passed through 18-gauge then 21-gauge needles 2–5 times. This fragmented material was left on ice to sediment by gravity for 5 min and then the upper layer was transferred to another tube. The cells in the suspension were pelleted by centrifugation, and the pellet was resuspended in a small volume of PBS. Cells in the suspension were attached to poly-L-lysine-coated glass slides. The samples were subjected to double immunostaining for CEACAM2-L and PVR followed by Hoechst nuclei staining, as described above.
Preparation of Membrane Fraction from Seminiferous Tubules for Electrophoresis
Testes of isoflurane-anesthetized adult mice were immersed in ice-cold PBS, and the capsule (tunica albuginea) was torn away by tweezers. Seminiferous tubules released from the testes were untangled and washed several times in PBS at 4C with gentle agitation to remove interstitial cells. A crude membrane fraction of the mouse seminiferous tubules was then obtained as follows. Seminiferous tubules were washed with PBS, homogenized in 0.1 M Tris-HCl (pH 7.4) in the presence of a protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany), and the samples then centrifuged twice for 15 min at 640g. The resultant supernatant was centrifuged at 100,000 ×g for 1 hr to obtain the crude membrane fraction. The samples were extracted in RIPA buffer [50 mM Tris (pH 7.2), 1 mM EDTA, 0.1% SDS, 0.1% Na deoxycholate, 1% Nonidet P-40, protease inhibitor cocktail], and the lysates centrifuged at 10,000 ×g at 4C for 20 min. Clarified supernatants were used as samples for electrophoresis and immunoprecipitation.
Immunoblot Analysis
RIPA-extracted proteins described above were separated by SDS-PAGE on 12% acrylamide gels, and separated proteins were either stained with Coomassie brilliant blue or transferred to Hybond-P membranes (GE Healthcare BioScience Corp.). The membranes were incubated for 2 hr with either the anti-CEACAM2-L antibody diluted 1:20,000 followed by incubation for 1 hr with HRP-conjugated goat anti-rabbit IgG (Bio-Rad, Richmond, CA) diluted 1:2,000, or with the rat anti-PVR antibody diluted 1:5000 followed by incubation for 1 hr with HRP-conjugated goat anti-rat IgG (Sigma Chemical) diluted 1:2000. The membranes were also incubated for 2 hr with the anti-E-Cadherin antibody (ECM Biosciences) diluted 1:10,000 followed by incubation for 1 hr with HRP-conjugated goat anti-rabbit IgG (Bio-Rad, Richmond, CA). Antigen–antibody complexes were visualized using an ECL-Plus detection kit (GE Healthcare Bioscience Corp.).
Immunoprecipitation with NHS Magnetic Sepharose Beads
NHS Magnetic Sepharose beads (GE Healthcare BioScience Corp.) were used for immunoprecipitation following the manufacturer’s instructions. In brief, NHS sepharose beads were activated by ice cold-activation buffer (0.1 M HCl). The anti-CEACAM2-L antibodies or rabbit pre-immune IgG were diluted in a coupling buffer (0.15 M triethanolamine, 0.5 M NaCl, pH 8.3) and rotated with the activated beads for 15 min at room temperature. The beads were then washed serially using blocking buffer A (0.5 M ethanolamine, 0.5 M NaCl, pH 8.3) and buffer B (0.1 M Na-acetate, 0.5 M NaCl, pH 4.0) to block the remaining active groups.
The extracted membrane fraction described above was diluted in a binding buffer (TBS, 50 mM Tris, 150 mM NaCl, pH 7.5) and incubated overnight at 4C with the NHS beads coupled either with the anti-CEACAM2-L antibodies or with rabbit pre-immune IgG. Beads were washed three times with a washing buffer (TBS, 2 M Urea, 0.15 M Tris-HCl, pH 7.5) before the proteins were eluted using an elution buffer (0.1 M glycine-HCl, pH 2.9). Both the eluates and the beads were collected and denatured in SDS-PAGE sample buffer for electrophoresis. To confirm that the IgG fixed to the beads did not release into the eluted samples, proteins from both the eluates and the beads were transferred to membranes and probed only with HRP-conjugated goat anti-rabbit IgG. The eluates were then subjected to immunoblot analysis using the following antibodies: (1) anti-CEACAM2-L antibody diluted 1:20,000, followed by incubation for 1 hr with HRP-conjugated goat anti-rabbit IgG; (2) rabbit anti-PVR antibody diluted 1: 2000, followed by incubation for 1 hr with HRP-conjugated goat anti-rabbit IgG; or (3) rat anti-PVR antibody diluted 1:5000, followed by incubation for 1 hr with HRP-conjugated goat anti-rat IgG. Antigen–antibody complexes were visualized using an ECL-Plus detection kit (GE Healthcare Bioscience Corp.).
Transfection and Immunoprecipitation In Vitro
Full-length mouse Ceacam2-L (accession number AB731450) was PCR-amplified using the following primers: 5′-CTT CGA ATT CTA CCA TGG AGC TGG CCT CAG CAC ATC-3′ (forward) and 5′-TGC AGA ATT CTT CTT TTT TAC TTC TGA ATA AAC TG-3′ (reverse). Full-length mouse Pvr (accession number NM_027514) was PCR-amplified using the following primers: 5′- CTC TCG AAT TCT ACC ATG GCT CAA CTC GCC CGA GC-3′ (forward) and 5′- TGC AGA ATT CCT TGT GCT GTT TGG CTC CA- 3′ (reverse). Amplified PCR products were digested by EcoRI and cloned in-frame into pEGFP-N3 (for Ceacam2-L, Sigma-Aldrich) or pTagRFP-N (for Pvr, Evrogen JSC, Moscow, Russia) expression vectors. COS-7 cells were transfected with either Ceacam2-L-GFP vector or Pvr-RFP vector using Lipofectamine 2000 (Invitrogen, Carlsbad, CA) as per the manufacturer’s instructions. After 48 hr of culture, transfected cells were trypsinized, mixed, and co-cultured in one dish. After 24 hr of culture, the co-cultured cells were lysed in RIPA buffer for 1 hr at 4C, centrifuged, and the resultant supernatant was used either directly for immunoblot analysis or for immunoprecipitation with the anti-GFP antibody (Cell Signaling Technology, Inc. Boston, MA) and protein G-agarose beads (Roche Diagnostics). Non-transfected cells were used as control. Immunoprecipitated proteins were subjected to immunoblot analysis either with the anti-CEACAM2-L antibody followed by incubation with HRP-conjugated goat anti-rabbit IgG or with the rat anti-PVR antibody followed by incubation with HRP-conjugated goat anti-rat IgG. Antigen–antibody complexes were visualized using an ECL-Plus detection kit (GE Healthcare Bioscience Corp.).
For immunofluorescence microscopy, transfected COS-7 cells fixed in PBS containing 4% paraformaldehyde were investigated by the fluorescence microscope after nuclear staining with Hoechst 33342 (Sigma-Aldrich).
Treatment with BS3 Crosslinker in Transfected COS-7 cells
BS3 (Bis [sulfosuccinimidyl] suberate, Pierce Biotechnology, Thermo Scientific, Rockford, IL) was used to investigate the interaction between CEACAM2-L-GFP and PVR-RFP in transfected COS-7 cells. BS3 crosslinker reacts efficiently with primary amino groups (-NH2 ) between pH 7 to 9 to form stable amide bonds.
Transfected COS-7 cells, as described above, were exposed for 1 hr at 4C to PBS in the presence or absence (control) of BS3 crosslinker at a final concentration of 2 mM. The reaction was quenched by the addition of Tris-HCl (pH 7.0) to a final concentration 20 mM and incubated at 4C for 30 min. The cells were then lysed in RIPA buffer for 1 hr at 4C, and the lysates centrifuged at 15,000 rpm for 10 min. The resultant supernatant was used either for immunoblot analysis or for immunoprecipitation with the anti-GFP antibody followed by incubation with protein G-agarose beads, as described above. Non-transfected cells were used as control. Immunoprecipitated proteins were separated by SDS-PAGE and transferred to Hybond-P membranes (GE Healthcare BioScience Corp.). The membranes were incubated for 2 hr either with the anti-CEACAM2-L antibody followed by incubation with HRP-conjugated goat anti-rabbit IgG or with the rat anti-PVR antibody followed by incubation with HRP-conjugated goat anti-rat IgG.
Investigation of Interaction between Mutated CEACAM2-L-GFP and PVR-RFP in COS-7 cells
A mutant form of Ceacam2-L, which lacks the first Ig domain (Ceacam2-LΔIg), was produced as follows: Ceacam2L-GFP-N3 vector was digested with BstEII enzyme, which cuts at (GGTnACC). The cut vector was used as a template for PCR-amplification using 5’ phosphorylated primers: 5′-GCACAAGTGACTGTTATG-3′ (forward) and 5′-GTGACTCAGCCCTCCCTC-3′ (reverse). Amplified PCR products were ligated by T4 ligase (TAKARA Biotech. Co. Japan) and used for the transformation of E. coli competent cells. The coding sequence of Ceacam2-L is 1020 bases and encodes a protein of 340 amino acids, whereas Ceacam2-LΔIg is 714 bases and encodes a 238-amino acid protein.
COS-7 cells were transfected with Ceacam2-LΔIg-GFP vector or Pvr-RFP vector using Lipofectamine 2000. After 24 hr of culture, transfected cells were trypsinized, mixed, and co-cultured in one dish. After 24 hr, the co-cultured cells were subjected either to immunoblot analysis or to immunoprecipitation using the anti-GFP antibody. Immunoprecipitated samples were processed for immunoblot analysis either with the anti-CEACAM2-L antibody or with the rat anti-PVR antibody, as described above. Non-transfected cells were used as control. For immunofluorescence microscopy, fixed COS-7 cells were counterstained with Hoechst 33342 and investigated under a fluorescence microscope.
Glycopeptidase F Treatment
Proteins extracted using RIPA buffer from the membrane fraction of the SE were incubated with anti-CEACAM2-L antibody immobilized on NHS magnetic beads. Immunoprecipitated proteins were incubated at 37C for 15∼20 hr in the reaction buffer in the presence or the absence of glycopeptidase F (2.5 mU/30 µl, TAKARA Biotech. Co.) following the manufacturer’s instructions. Both undigested (control) and digested proteins were subjected to immunoblot analysis with either the anti-CEACAM2-L antibody or rat anti-PVR antibody.
Transfected COS-7 cells overexpressing CEACAM2-L-GFP or PVR-RFP were lysed in RIPA buffer and the lysate centrifuged at 15,000 rpm for 15 min to collect the supernatants. The samples were processed for glycopeptidase F digestion and then subjected to immunoblot analysis with either the anti-CEACAM2-L antibody or rat anti-PVR antibody, as described above.
Results
Immunolocalization of CEACAM2-L and PVR in the Mouse Seminiferous Epithelium
Ceacam2 is composed of 9 exons located on 7A3 in mouse chromosome 7. There are two types of variants expressed in mouse testis. Ceacam2-L has an open reading frame of 1,020 bases and consists of seven exons (1, 2, 5, 6, 7, 8 and 9) encoding a protein of 340 amino acids. It comprises one Ig domain, one IgC2 domain, a trans-membrane region, and a cytoplasmic tail region (Salah et al. 2012). Another short variant of Ceacam2, i.e., Ceacam2-S, has the coding sequence of 816 bases and encodes a protein of 272 amino acids (Han et al. 2001). CEACAM2-L differs from CEACAM2-S in that it has a much longer cytoplasmic tail region. In this study, we used the antibody specific for CEACAM2-L, which does not recognize the shorter variant (Salah et al. 2012).
The expression and cellular localization of CEACAM2-L and PVR in the mouse testis were first examined by immunohistochemistry using the anti-CEACAM2-L antibody and the rat anti-PVR antibody, respectively. Within SE of stage III~VI, heads of elongating spermatids are deeply invaginated into the crypts of Sertoli cells, at which the apical ES is formed. As shown in Fig. 1A, fluorescence microscopy demonstrated that CEACAM2-L was not detectable at the apical ES surrounding the head region of elongating spermatids but was detected at the plasma membranes overlying the excess cytoplasm of elongated spermatids, as reported previously (Salah et al. 2012). On the other hand, PVR immunoreactivity within SE was distributed diffusely from the basal to the apical portions of the epithelium in a radial pattern, which is consistent with the observation of Wakayama et al. (2007) that PVR is present on the plasma membranes as well as in the cytoplasm of Sertoli cells. Extended cytoplasmic processes of Sertoli cells immunostained by the rat anti-PVR antibody were in close contact with the plasma membrane of elongated spermatids and incompletely surrounded the cytoplasm of elongated spermatids (Fig. 1B). Figure 1C shows immunostaining of CEACAM2-L and PVR on elongated spermatids isolated from mechanically fragmented testis. Fluorescence microscopy clearly revealed that CEACAM2-L was detected on the plasma membrane covering the excess cytoplasm of elongated spermatids as well as in their cytoplasm, but not at the sperm head region or on the flagellum (Fig. 1C, top); PVR was not detected on the cells (Fig. 1C, center). Replacement of the anti-CEACAM2-L and rat anti-PVR antibodies with pre-immune serum produced no distinct staining (data not shown), as previously reported (Salah et al. 2012; Wakayama et al. 2007). These observations indicate that CEACAM2-L and PVR are expressed specifically in elongated spermatids and Sertoli cells, respectively.
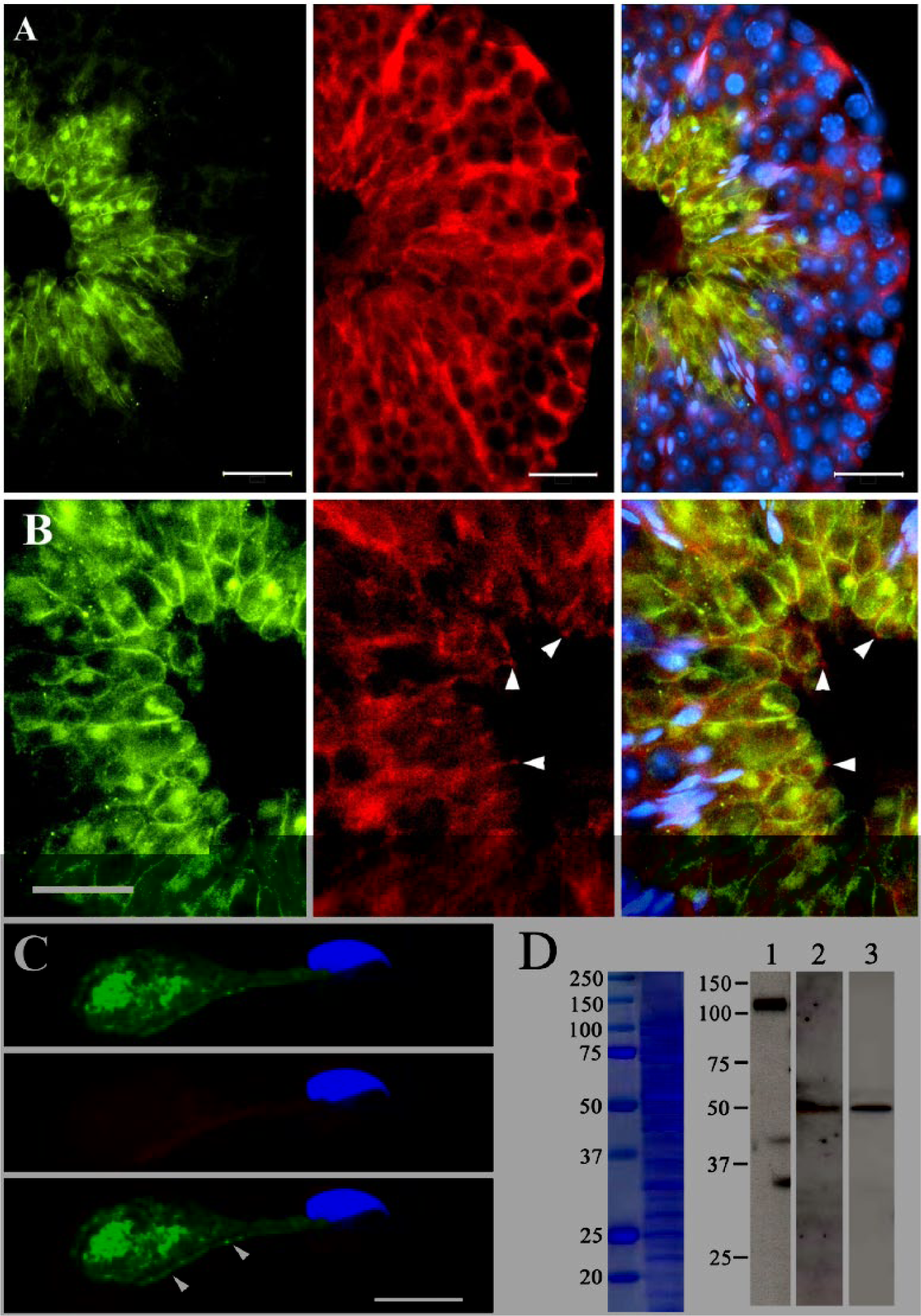
Expression of CEACAM2-L and PVR in the seminiferous epithelium (SE) of adult mouse testis. (A and B) Immunohistochemical localization of CEACAM2-L and PVR in the seminiferous epithelium at stage III~VI of mouse seminiferous tubules. Frozen sections of adult mouse testis were double stained with the anti-CEACAM2-L antibody (green) and the rat anti-PVR antibody (red). Nuclear DNA was stained by Hoechst 33342 (blue). Three-color images are super-imposed in the right panel. CEACAM2-L and PVR were detected on elongated spermatids and Sertoli cells, respectively (A). Extended cytoplasmic processes of Sertoli cells (B, arrowheads, red) were in close contact with the surface of cytoplasm of elongated spermatids labeled by the anti-CEACAM2-L antibody. CEACAM2-L-positive puncta (green), which might represent cell organelles such as Golgi apparatus, were also seen in the cytoplasm of elongating spermatids. Bars: 25 μm. (C) Immunohistochemical localization of CEACAM2-L on mechanically separated spermatids. Spermatids isolated from fixed testis by mechanical fragmentation were double immunostained by the anti-CEACAM2-L antibody (green, top panel) and the rat anti-PVR antibody (red, center panel) followed by nuclear DNA staining by Hoechst 33342 (blue). Three-color images are super-imposed in the bottom panel. CEACAM2-L was detected on the surface covering the excess cytoplasm of elongated spermatids (arrowheads), but not at the head region or flagellum. There was no immunosignal for PVR on the surface. Bar: 10 μm. (D) Immunoblot analysis of E-cadherin, CEACAM2-L, and PVR in the seminiferous tubules of adult mouse testis. Proteins extracted from the membrane fraction of mouse seminiferous tubules were separated by SDS-PAGE. Separated proteins were either stained with Coomassie brilliant blue (left panel) or subjected to immunoblot analysis (right panel) using the anti-E-Cadherin antibody (lane 1), the anti-CEACAM2-L antibody (lane 2) or the rabbit anti-PVR antibody (lane 3). Trans-membrane glycoprotein E-Cadherin was detected at 120 kDa (lane 1). A single band migrating at approximately 50 kDa, which corresponds to CEACAM2-L (lane 2) and PVR (lane 3), respectively, was detected on the blot. Molecular standards are shown on the left in kDa.
To confirm the expression of CEACAM2-L and PVR in SE, the membrane fraction of SE was subjected to immunoblot analysis with antibodies against CEACAM2-L and PVR. We confirmed that the 120 kDa trans-membrane glycoprotein E-Cadherin (a marker for the membrane fraction) was present in the fraction (Fig. 1D). As shown in Figure 1D, both CEACAM2-L and PVR were detected at around 50 kDa on the blot. The sizes of the proteins detected on the blot were larger than the predicted molecular weights of full-length of CEACAM2-L (37.4 kDa) and PVR (44.9 kDa), as calculated from the amino acid sequence deduced from the cDNA sequence.
Interaction between CEACAM2-L and PVR In Vivo
In order to investigate the interaction between CEACAM2-L and PVR in vivo, the membrane fraction of mouse seminiferous tubules was solubilized in RIPA buffer, centrifuged, and the resultant supernatants were used for immunoprecipitation with the anti-CEACAM2-L antibody. Because both CEACAM2-L and PVR appeared around 50 kDa on the blot (see Fig. 1D), they probably overlap with the heavy chain of IgG used for immunoprecipitation. To avoid this overlap on the blot, the anti-CEACAM2-L antibody and pre-immune IgG (for control) were covalently immobilized onto NHS Magnetic Sepharose beads to prevent them from releasing during antigen elution.
RIPA-extracted membrane fraction samples, which are identical to that used for immunoblotting in Figure 1D, were incubated overnight at 4C either with pre-immune IgG or with the anti-CEACAM2-L antibody fixed on the beads, washed extensively, and proteins combined with the beads were released by 0.1 M glycine-Cl (pH 2.9). First, to confirm that IgG were retained on the beads after elution, the released antigens and washed beads were separately subjected to immunoblot analysis with the HRP-conjugated anti-rabbit IgG. As shown in Figure 2A, IgG of both pre-immune and the anti-CEACAM2-L antibody were detected in the beads, but not in the eluates, indicating that IgGs were retained on the beads even after antigen elution. The eluates released from the immunoprecipitated beads were subjected to immunoblot analysis with the anti-CEACAM2-L antibody (Fig. 2B), the rabbit anti-PVR antibody (Fig. 2C), and the rat anti-PVR antibody (Fig. 2D). In addition to CEACAM2-L (Fig. 2B), PVR was detected in the sample immunoprecipitated using the anti-CEACAM2-L antibody (Figs. 2C and D), indicating the direct interaction between CEACAM2-L and PVR in vivo.
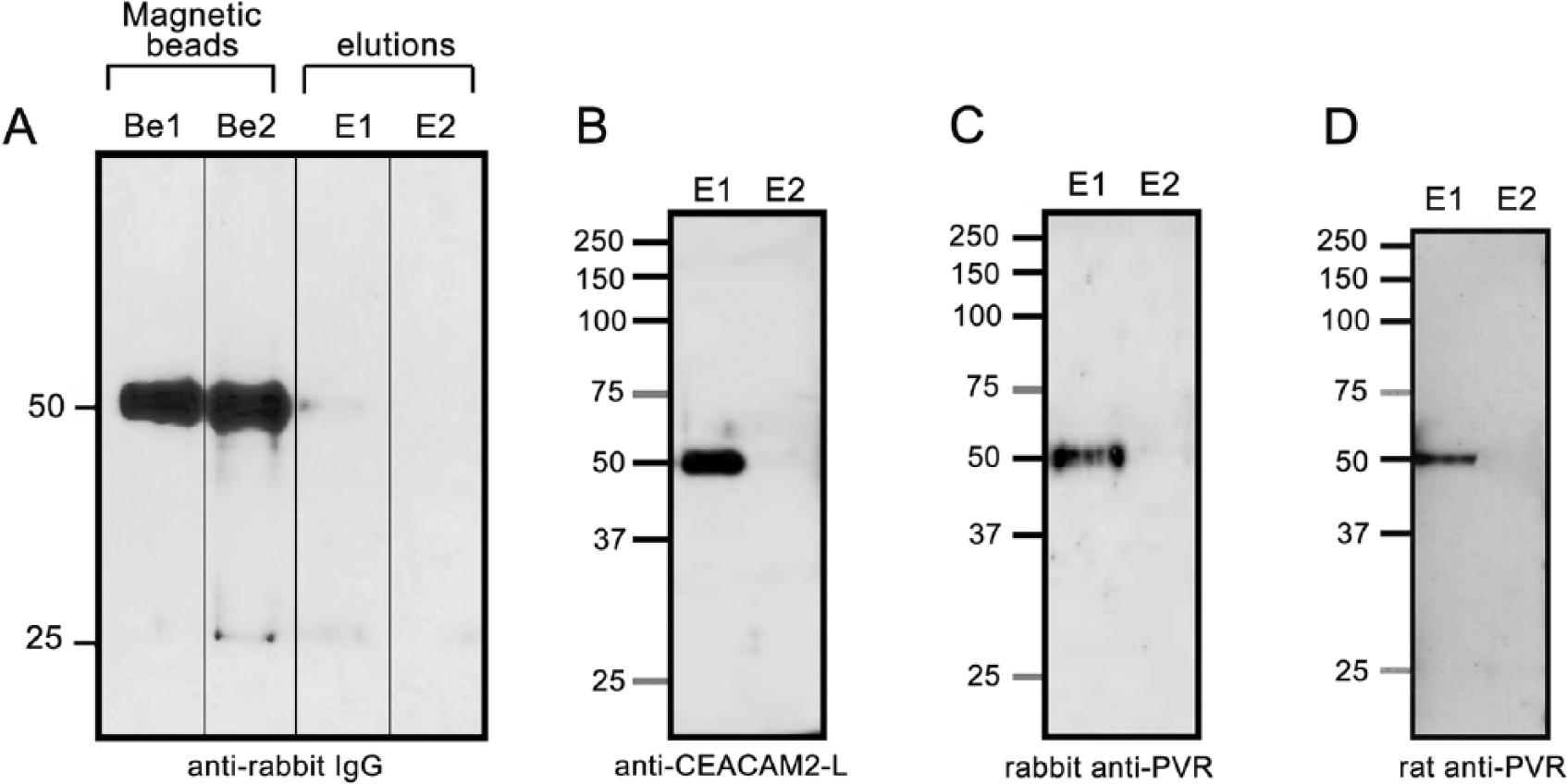
Direct interaction between CEACAM2-L and PVR in vivo. Proteins extracted from the membrane fractions by RIPA buffer were incubated overnight at 4C with either the anti-CEACAM2-L antibody or with pre-immune IgG, both of which were combined with magnetic NHS beads. After excessive washes, proteins bound to the beads were eluted by 0.1 M glycine-HCl (pH 2.9). (A) Proteins in both the magnetic NHS beads and the eluates were separated by SDS-PAGE, transferred to membranes and probed only with HRP-conjugated goat anti-rabbit IgG. The heavy chain (50 kDa) of IgG was detected in the beads (lanes Be1 and Be 2) but not in the eluates (lane E1 and E2), indicating that IgGs were retained on the beads after elution. Lane Be1: beads with anti-CEACAM2-L IgG; lane Be2: beads with pre-immune IgG; lane E1: elution from beads with anti-CEACAM2-L IgG; lane E2; elution from beads with pre-immune IgG. (B, C, and D) Eluates (lanes E1 and E2 in A) were subjected to immunoblot analysis with the anti-CEACAM2-L antibody (B), rabbit anti-PVR antibody (C), or rat anti-PVR antibody (C). A protein migrating at 50 kDa, which corresponds to CEACAM2-L, was detected in anti-CEACAM2-L-immunoprecipitated samples (B, lane E1), but not in the samples immunoprecipitated by pre-immune IgG (B, lanes E2). A protein migrating at 50 kDa, which is corresponds to PVR, was detected in anti- CEACAM2-L-immunoprecipitated samples (C and D, lanes E1), but not in the samples immunoprecipitated by pre-immune IgG (C and D, lanes E2). The results suggest a direct interaction between CEACAM2-L and PVR. Molecular standards are shown on the left in kDa.
Interaction between CEACAM2-L and PVR in Cultured Cells
To confirm the interaction between CEACAM2-L and PVR, we performed immunoprecipitation using transfected cultured cells. Full-length Ceacam2-L and Pvr were amplified by RT-PCR. CEACAM2-L was tagged with green fluorescent protein (GFP) (CEACAM2-L-GFP) in the pEGFP-N3 expression vector, and PVR was tagged with red fluorescent protein (RFP) (PVR-RFP) in the pTagRFP-N expression vector and the vectors transfected into COS-7 cells. CEACAM2-L-GFP (75 kDa) was expressed at the cell periphery or as cytoplasmic aggregation deposits in the transfected cells (Fig. 3A, left, top). PVR-RFP (75 kDa) was seen at the cell periphery as well as in the cytoplasm and nuclei (Fig. 3A, left, bottom). The transfected cells were detached from the dishes, mixed and co-cultured in one dish for a further 24 hr (Fig. 3A, right) and then lysed with RIPA buffer. Non-transfected cells were used as control. Proteins extracted by RIPA buffer from both non-transfected and transfected cells were first examined by immunoblotting with anti-CEACAM2-L or anti-PVR. As shown in Figure 3B, both CEACAM2-L-GFP and PVR-RFP were detected at 75 kDa in the transfected samples but not in the control. These samples were then used for immunoprecipitation with the anti-GFP antibody. As shown in Figure 3C, the anti-GFP antibody immunoprecipitated not only CEACAM2-L-GFP but also PVR-RFP, but the lysates from non-transfected cells were clear. These data indicate the direct interaction between CEACAM2-L-GFP and PVR-RFP in transfected co-cultured COS-7 cells.
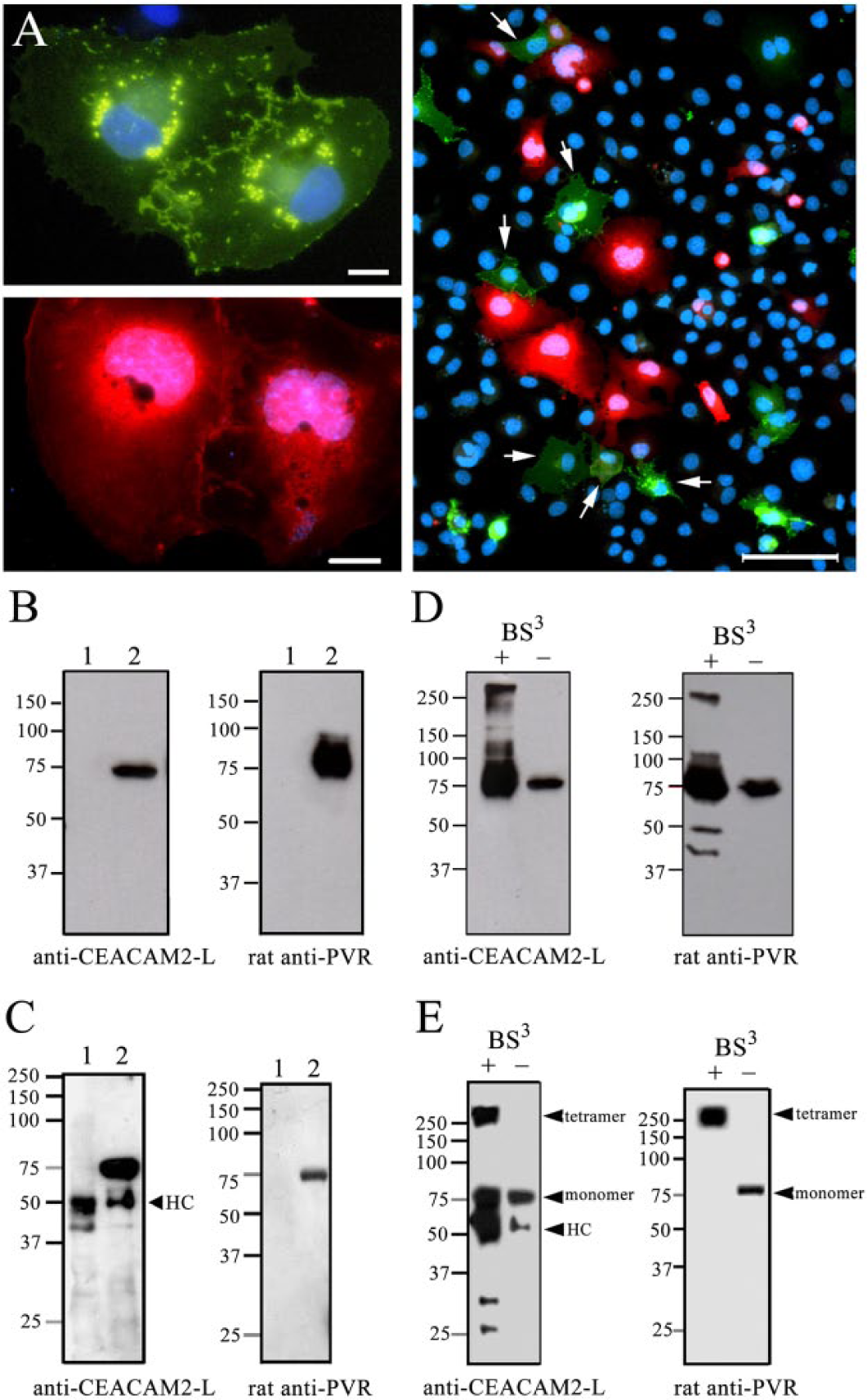
Interaction of CEACAM2-L and PVR in cultured cells. (A) COS-7 cells transfected with Ceacam2-L-GFP (left, top image) or Pvr-RFP (left, bottom image) were cultured separately. The cells were then trypsinized, mixed, and co-cultured together for 24 hr in the same dish (right). CEACAM2-L-GFP (75 kDa) expression was seen at the cell periphery and as small aggregates in the cytoplasm (left, top). PVR-RFP (75 kDa) was also seen at the cell periphery and in the cytoplasm and nucleus (left, bottom). In co-cultured cells (right), the arrows indicate CEACAM2-L-GFP-expressing cells in contact with cells expressing PVR-RFP. Bars: left 10 μm, right 100 μm. (B and C) Proteins extracted with RIPA buffer from non-transfected cells (lane 1) and transfected co-cultured cells (lane 2) were examined by immunoblot analysis either with the anti-CEACAM2-L antibody or with the rat anti-PVR antibody (B). Both CEACAM2-L-GFP (B, left panel) and PVR-RFP (B, right panel) were detected in the transfected samples (lane 2) but not in the control (lane 1). The same samples were used for immunoprecipitation with the anti-GFP antibody (C). Proteins precipitated with the anti-GFP antibody from non-transfected control COS-7 cells (C, lane 1) and co-cultured transfected cells (C, lane 2) were separated by SDS-PAGE. Lysates were subjected to immunoblotting with either the anti-CEACAM2-L antibody (C, left panel) or the rat anti-PVR antibody (C, right panel). Both CEACAM2-L-GFP (C, left panel, lane 2) and PVR-RFP (C, right panel, lane 2) appeared at 75 kDa on the blot, but not in the control samples (C, lane 1), indicating co-precipitation of PVR-RFP with CEACAM2-L-GFP by the anti-GFP antibody. HC: heavy chain of IgG. Molecular standards are shown on the left in kDa. (D and E) Proteins extracted with RIPA buffer from non-treated (lane -) or BS3-treated (lane +) transfected co-cultured COS-7 cells were examined by immunoblotting either with the anti-CEACAM2-L antibody (D, left panel) or rat anti-PVR antibody (D, right panel). Both CEACAM2-L and PVR were detected at 75 kDa in the absence of BS3 (D, lane -), and migrated at both 75 kDa and more than 250 kDa in the presence of BS3 (D, lane +). The same RIPA-extracted samples were used for immunoprecipitation with the anti-GFP antibody (E). Proteins precipitated using anti-GFP antibody from BS3-treated (lane +) or non-treated (lane -) co-cultured COS-7 cells were subjected to immunoblotting either with anti-CEACAM2-L (E, left panel) or rat anti-PVR (E, right panel) antibodies. BS3 treatment brought about molecular shift of the 75-kDa CEACAM2-L-GFP to more than 250 kDa (indicated by ‘tetramer’) with a concomitant similar shift of PVR-RFP. Monomers of CEACAM2-L-GFP (75 kDa) were also detected on the blot. HC: heavy chain of IgG. Molecular standards are shown on the left in kDa.
Next we investigated the effect of BS3 treatment on a molecular shift of CEACAM2- L-GFP and PVR-RFP on the blot. BS3 is a cell-surface cross-linker used to investigate the formation of protein complexes, such as dimers and tetramers. Proteins extracted by RIPA buffer from BS3-treated or non-treated transfected co-cultured COS-7 cells were first examined by immunoblot analysis either with the anti-CEACAM2-L antibody or with the rat anti-PVR antibody. As shown in Figure 3D, both CEACAM2-L-GFP and PVR-RFP were detected at 75 kDa in the absence of BS3, but migrated at both 75 kDa and more than 250 kDa in the presence of BS3. The data indicate effective cross-linking of the molecules by BS3. The same RIPA-extracted samples were then used for immunoprecipitation with the anti-GFP antibody. Precipitated proteins were subjected to immunoblot analysis either with the anti-CEACAM2-L antibody or with the rat anti-PVR antibody. BS3 treatment brought about molecular shift of the 75-kDa CEACAM2-L-GFP to more than 250 kDa, with a concomitant movement of the 75-skDa PVR-RFP to more than 250 kDa on the blot (Fig. 3E). A CEACAM2-L-GFP monomer (75 kDa) was also detected (Fig. 3E). In the absence of BS3, on the other hand, monomers of both CEACAM2-L-GFP (75 kDa) and PVR-RFP (75 kDa) were observed (Fig. 3E). These results suggest that BS3 forms heterophilic cross-links between CEACAM2-L-GFP and PVR-RFP and that dimeric CEACAM2-L forms trans-tetra complexes with dimeric PVR in the transfected cells.
Supplementary Figure 1A shows the effects of BS3 on the molecular shift of CEACAM2-L-GFP immunoprecipitated by the anti-GFP antibody from COS-7 cells transfected only with CEACAM2-L-GFP. In the single-transfected cells, a similar migration of CEACAM2-L-GFP (75 kDa) to more than 250 kDa was detected in the BS3-treated cells. This suggests that CEACAM2-L-GFP forms homophilic tetramer complexes between adjoining cultured cells, as seen in Figure 3A (left, top). We obtained a similar result for PVR-RFP expressed alone in transfected COS-7 cells (Supplemental Fig. S1B).
It is generally believed that N-terminal Ig domains of IgSF molecules are involved in protein-protein interactions (Momose et al. 2002; Takai et al. 2008; Narita et al. 2011). To investigate the involvement of the first Ig domain of CEACAM2-L in its interaction with PVR, we constructed an expression vector of CEACAM2-L (CEACAM2-LΔIg) that lacks the first Ig domain (Fig. 4A). COS-7 cells expressing either CEACAM2-LΔIg-GFP (Fig. 4B, top) or PVR-RFP (Fig. 4B, center) were separately cultured, and then mixed together as before and cultured for a further 24 hr (Fig. 4B, bottom) to ascertain whether CEACAM2-LΔIg-GFP could interact with PVR-RFP. Proteins extracted by RIPA buffer from transfected or non-transfected (control) co-cultured cells were first examined by immunoblot analysis either with the anti-CEACAM2-L antibody or with the rat anti-PVR antibody. As shown in Figure 4C (INPUT panel), both CEACAM2-L and PVR were detected in the lysates from transfected cells, but not from control cells. The same RIPA-extracted samples were then used for immunoprecipitation with the anti-GFP antibody. As shown in Figure 4C (IP panel), the anti-GFP antibody immunoprecipitated CEACAM2-LΔIg-GFP but not PVR-RFP. This suggests that the first Ig domain of CEACAM2-L is critical for its interaction with PVR.
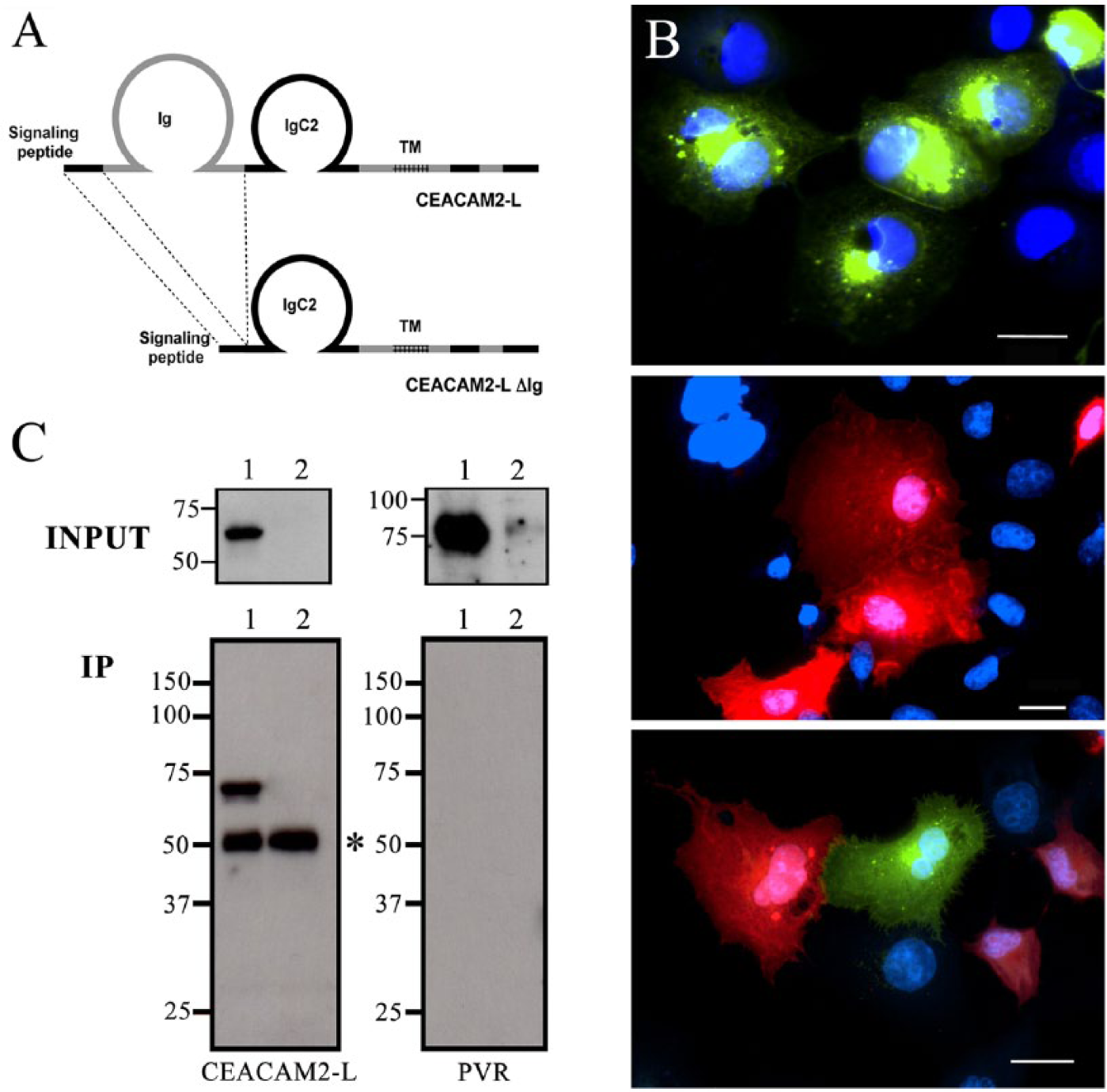
Mutated CEACAM2-L (CEACAM 2-LΔIg) failed to interact with PVR. (A) Schematic representation of CEACAM 2-L and CEACAM 2-LΔIg lacking the first Ig domain. CEACAM 2-L has one Ig domain, one IgC2 domain, a trans-membrane region (TM), and a cytoplasmic tail. (B) COS-7 cells transfected either with Ceacam2-LΔIg-GFP (top) or Pvr-RFP (center) were separately cultured, trypsinized, mixed, and co-cultured for a further 24 hr (bottom). Bars: 20 μm. (C) INPUT: Proteins extracted with RIPA buffer from non-transfected (control) or transfected co-cultured COS-7 cells were examined by immunoblot analysis either with the anti-CEACAM2-L antibody (left panel) or the rat anti-PVR antibody (right panel). Both CEACAM2- LΔIg and PVR were detected in transfected cell lysates (lane 1), but not control lysates (lane 2). IP: Proteins were immunoprecipitated from the same RIPA-extracted samples with anti-GFP, and lysates were immunoblotted with the anti-CEACAM2-L antibody (left panel) or the rat anti-PVR antibody (right panel). Although the anti-GFP antibody immunoprecipitated the 65-kDa CEACAM2-LΔIg-GFP (left panel, lane 1), it failed to precipitate PVR-RFP (right panel, lane 1). In the control (non-transfected COS-7 cells), the anti-GFP antibody did not precipitate any CEACAM2-LΔIg-GFP (left panel, lane 2). Asterisk: heavy chain of IgG. Molecular standards are shown on the left in kDa.
Glycopeptidase F Treatment
As shown in Figure 1D, the sizes of the proteins detected on the blot (50 kDa) were larger than the predicted molecular weights of full-length CEACAM2-L (37.4 kDa) and PVR (44.9 kDa). Since both CEACAM2-L and PVR have more than two potential N-glycosylation sites, we investigated by immunoblot analysis their migration shift after digestion with glycopeptidase F, which cleaves asparagine-linked high mannose as well as hybrid and complex oligosaccharides of glycoproteins. Proteins immunoprecipitated from the membrane fraction of the SE with the anti-CEACAM2-L antibody (Fig. 5A) as well as proteins extracted by RIPA buffer from transfected COS-7 cells (Fig. 5B) were processed for glycopeptidase F digestion and then subjected to immunoblot analysis.
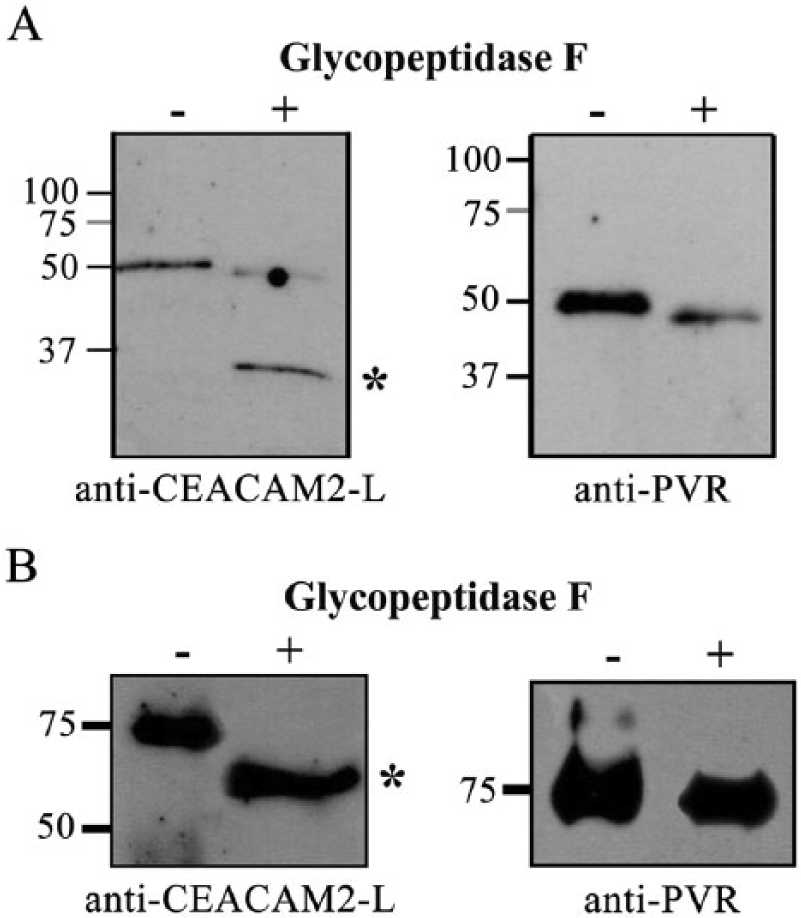
Effect of glycopeptidase F on the molecular shift of CEACAM2-L and PVR. (A) Proteins extracted by RIPA buffer from the membrane fraction of seminiferous epithelium (SE) were incubated with the anti-CEACAM2-L antibody immobilized on NHS magnetic beads. Immunoprecipitated proteins were incubated at 37C for 15∼20 hr in a reaction buffer in the presence or the absence of glycopeptidase F (2.5 mU/30 µl). Both undigested (-) and digested proteins (+) were subjected to immunoblot analysis either with the anti-CEACAM2-L antibody (left panel) or with the rat anti-PVR antibody (right panel). Asterisk (*) indicates 37-kDa CEACAM2-L after glycopeptidase F digestion. Digested PVR migrated at 48 kDa (right panel), a little faster than that of undigested PVR (50 kDa). (B) Proteins extracted with RIPA buffer from COS-7 cells transfected either with CEACAM2-L-GFP (left panel) or with PVR-RFP (right panel) were incubated at 37C for 15∼20 hr in the reaction buffer in the presence (+) or the absence (-) of glycopeptidase F, and both undigested (control) and digested proteins were subjected to immunoblot analysis either with the anti-CEACAM2-L antibody (left panel) or with the rat anti-PVR antibody (right panel). Glycopeptidase F digestion brought about a molecular shift of CEACAM2-L-GFP from 75 kD to 60 kDa (left panel, asterisk) but had little effect on PVR (right panel). Molecular standards are shown on the left in kDa.
After glycopeptidase F digestion, CEACAM2-L showed a molecular shift from 50 kDa to 37 kDa (Fig. 5A, left panel), and PVR showed a molecular shift from 50 kDa to 48 kDa (Fig. 5A, right panel). In the samples of the transfected COS-7 cells, glycopeptidase F treatment brought about a molecular shift from 75 kDa to 60 kDa for CEACAM2-L-GFP (Fig. 5B, left panel) but a distinct molecular shift of PVR-RFP was not observed by glycopeptidase F treatment (Fig. 5B, right panel). The results indicate that N-glycosylation of CEACAM2-L is responsible for the apparent molecular shift from 37.5 kDa to 50 kDa in electrophoresis.
Discussion
During spermatogenic differentiation, over half of differentiating spermatogenic cells undergoes apoptosis without maturing into spermatozoa (Huckins 1978; de Rooij 1973), although the underlying mechanism and functional significance of this phenomenon are unknown. Several studies of rodent testes have revealed that exposed phosphatidylserine groups on the surface of degenerating spermatogenic cells are recognized by class B scavenger receptor type I, which is expressed by Sertoli cells, thereby stimulating phagocytosis (Russell and Clermont 1977; Shiratsuchi et al. 1997; 1999).
Spermiation is initiated at the beginning of stage VII in the murine testes and terminates at the end of stage VIII with the release of spermatids from supporting somatic Sertoli cells into the lumen of the seminiferous tubule with concomitant degradation of the apical ES (O’Donnell et al. 2011). Before spermiation, residual bodies consisting of the excess cytoplasm of spermatids are engulfed and phagocytosed by Sertoli cells (O’Donnell et al. 2011). This last step is essential for the completion of spermiogenesis and involves cell-cell recognition between Sertoli cells and elongating spermatids, which is mediated by cell-surface proteins. The process is also highly specific, because Sertoli cells recognize and engulf only excess cytoplasm and never phagocytose the whole cell (O’Donnell et al. 2011). Although the structural and morphological aspects for removal of the excess cytoplasm of spermatids have been well documented (Russell et al. 1988; 1989 ; Weber and Russell 1987), details of the molecular mechanisms remain poorly understood. Several proteins, including Capza3 and Spem1, have been implicated in this process (Geyer et al. 2009; Zheng et al. 2007) but are not directly involved in cell-cell interaction. Since Capza3 regulates F-actin dynamics, it may contribute to the removal of cytoplasm from elongated spermatids by modulating cytoskeletal architecture.
In mouse testis, the expression of Ceacam2-L is first detected in the testes at 3 weeks postnatal development, with higher levels identified by 5 weeks, coinciding with the production of haploid, elongating spermatids in the SE. Immunohistochemical data revealed that CEACAM2-L is expressed at the spermatid surface (steps 13–16), and is integrated into residual bodies after stage VIII (Salah et al. 2012). Given that CEACAM2-L is localized exclusively to the plasma membrane overlying the excess cytoplasm of elongated spermatids, we previously suggested that CEACAM2-L is involved in recognition and/or phagocytosis of the excess cytoplasm by Sertoli cells.
PVR was originally identified as the receptor responsible for poliovirus infection (Mendelsohn et al. 1989), and is also known as tumor-associated antigen pE4 (Chadeneau et al. 1994) and nectin-like molecule 5 (Necl-5) (Ikeda et al. 2003). Based on its ubiquitous expression on the surface of Sertoli cells (Wakayama et al. 2007) and engagement in heterophilic interactions (Takai et al. 2003), here we investigated PVR as a potential binding partner of CEACAM2-L. The two proteins were closely disposed at the spermatid-Sertoli cell interface (Fig. 1), and PVR was co-immunoprecipitated with CEACAM2-L from seminiferous tubule membrane extracts (Fig. 2) and transfected COS-7 cells (Fig. 3). These results demonstrate the heterophilic binding of CEACAM2-L and PVR in vitro and in vivo, providing evidence that the excess cytoplasm of elongated spermatids is recognized by Sertoli cells through the interaction of the two proteins. Another IgSF member, CADM1—also known as IGSF4, spermatogenic IGSF, or Necl-2 (Shingai et al. 2003)—is expressed in the seminiferous tubules of mouse testis at the surface of zygotene and early pachytene spermatocytes as well as elongated spermatids, but not Sertoli cells (Wakayama et al. 2007; Wakayama and Iseki 2009). Since CADM1 can also engage in heterophilic interactions with PVR (Wakayama et al. 2009), a CADM1-PVR interaction could also be involved in the recognition of the excess cytoplasm of spermatids by Sertoli cells.
Several IgSF members function as adhesion molecules at the Sertoli cell-spermatid interface of the apical ES (Cheng and Mruk 2002). In general, they form cis-homophilic dimers and then trans-heterophilic or -homophilic tetramer complexes. For example, heterophilic Sertoli cell/spermatid interactions occur through nectin-2/-3 (Ozaki-Kuroda et al. 2002), JAM-B/-C (Gliki et al. 2004), and CAR/JAM-C (Mirza et al. 2006), whereas homophilic interactions are mediated by CAR/CAR (Wang et al. 2007). Recently, CEACAM6-L expressed by germ and Sertoli cells was observed to form homophilic trans-tetramers between these cells in the SE (Kurio et al. 2012). In the present study, chemical cross-linking experiments with BS3 in transfected COS-7 cells showed that CEACAM2-L forms trans-tetramer complexes with PVR (Fig. 3). Taken together, the results suggest that CEACAM2-L on elongated spermatids functions as an adhesion molecule, and forms heterophilic trans-tetramers with PVR expressed by Sertoli cells (Fig. 6).
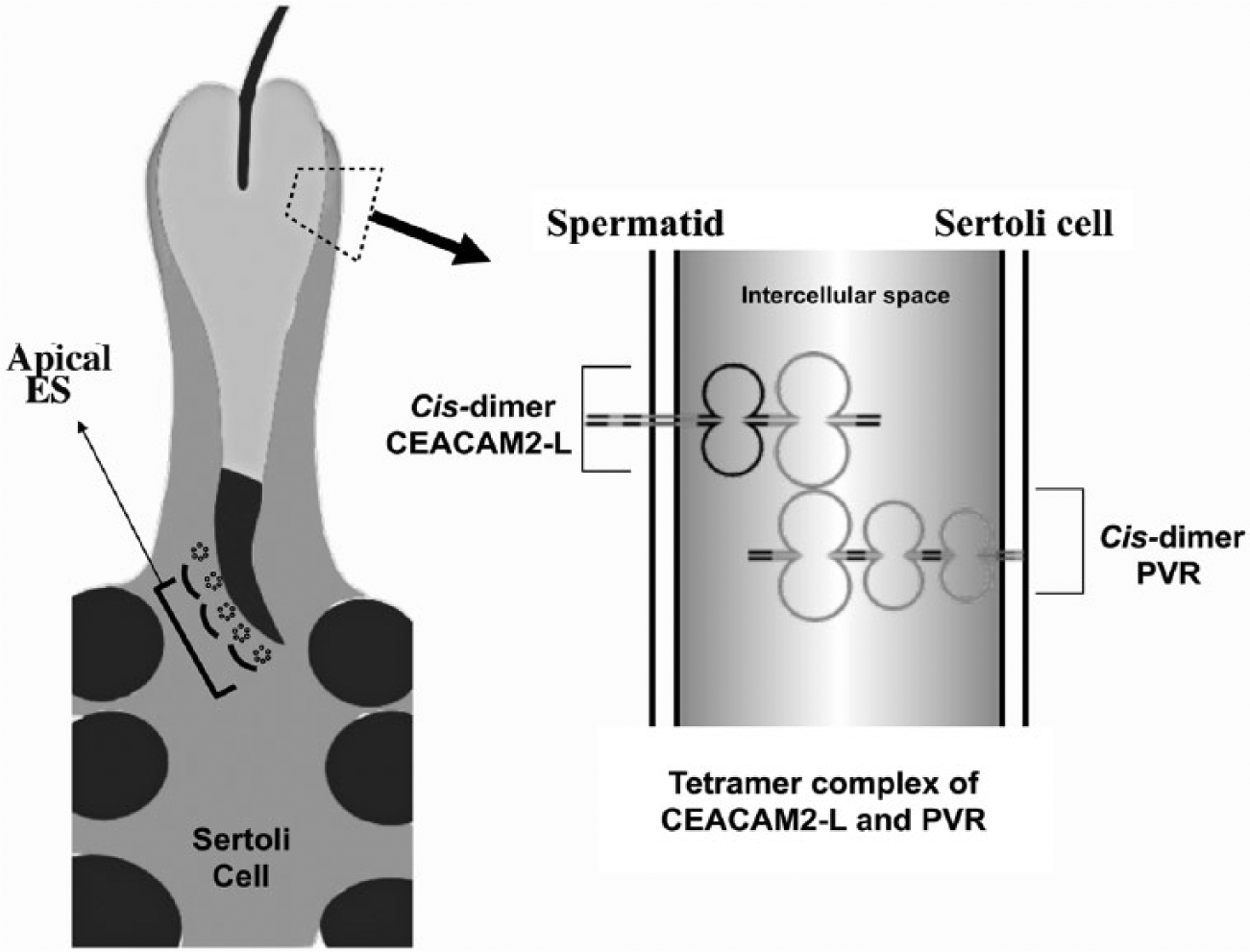
Schematic representation of interaction between PVR and CEACAM2-L. Cis-dimer of CEACAM2-L on elongated spermatids forms heterophilic trans-tetramers with dimeric PVR expressed by Sertoli cells. ES, ectoplasmic specialization.
Several recent studies have characterized the role of individual IgSF protein domains in cell-cell adhesion. Nectin-1 forms cis-dimers and engages in trans-interactions through its first Ig-like domain (Narita et al. 2011), whereas the first and second Ig-like domains of nectin-2 are essential for trans- and cis-dimer formation, respectively (Momose et al. 2002; Takai et al. 2008). In the present study, it was observed that CEACAM2-L lacking the first Ig domain failed to co-immunoprecipitate with PVR (Fig. 4), indicating that this domain is critical for the CEACAM2-L/PVR interaction.
The intracellular domain of CADM1 contains several motifs known to combine with actin filament-interacting proteins, suggesting that this domain may be involved in intracellular signaling. CEACAM2-L also has a large cytoplasmic domain. The identification of proteins binding to this region would therefore provide insight into the downstream factors that mediate the final step of spermiogenesis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Grant-in-Aid for
