Abstract
A well-known putative tumor suppressor WW domain–containing oxidoreductase (Wwox) is highly expressed in hormonally regulated tissues and is considered important for the normal development and function of reproductive organs. In this study, we investigated the cellular and subcellular localization of Wwox in normal testes during postnatal days 0–70 using Western blotting and immunohistochemistry. Wwox is expressed in testes at all ages. Immunohistochemistry showed that fetal-type and adult-type Leydig cells, immature and mature Sertoli cells, and germ cells (from gonocytes to step 17 spermatids) expressed Wwox except peritubular myoid cells, step 18–19 spermatids, and mature sperm. Wwox localized diffusely in the cytoplasm with focal intense signals in all testicular cells. These signals gradually condensed in germ cells with their differentiation and colocalized with giantin for cis-Golgi marker and partially with golgin-97 for trans-Golgi marker. Biochemically, Wwox was detected in isolated Golgi-enriched fractions. But Wwox was undetectable in the nucleus. This subcellular localization pattern of Wwox was also confirmed in single-cell suspension. These findings indicate that Wwox is functional in most cell types of testis and might locate into Golgi apparatus via interaction with Golgi proteins. These unique localizations might be related to the function of Wwox in testicular development and spermatogenesis:
Introduction
WW domain–containing oxidoreductase (
WWOX is ubiquitously distributed in various tissues and cell types 6 and exerts its physiological functions via interacting with different proteins in multiple cellular pathways. The first WW domain is a small protein module that interacts with partner proteins, and the second WW domain assists the first WW domain for binding as a chaperone. 7 It has been suggested that WWOX plays multiple roles via interacting with various proteins including apoptosis,8,9 cell signaling,10–15 cell adhesion,16,17 cell growth, 18 metabolic functions,19–22 regulation of transcription factors,23,24 and maintenance of genomic stability.3,5 The molecular function of SDR domain is to catalyze NAD(P)(H)-dependent oxidation–reduction (redox) reactions for cellular steroids and retinoids.25,26
Lethal dwarfism and epilepsy (LDE) rat strain was established in our laboratory. The
WWOX is expressed in various tissues except adipose tissues, connective tissues, lymphoid tissues, and blood vessels. The highest expression is observed in hormonally regulated tissue such as testis, prostate, ovary, mammary gland, adrenal gland, pituitary gland, and brain cells.6,28 In testis, Wwox is expressed in LC and cell layers of seminiferous tubules. But the details on cellular and subcellular localization of Wwox in developing testis and spermatogenesis have not been revealed. Therefore, we examined the localization of Wwox during postnatal development of testis and spermatogenesis.
Materials and Methods
Animals
Male Brown Norway (BN) rats were purchased from Charles River (Kanagawa, Japan). The inbred LDE strain27–29,33 was developed, and heterozygous female (+/
Protein Extraction From Whole Testis
The testes were homogenized in radioimmunoprecipitation assay buffer following the previously described method. 28 The homogenates were centrifuged at 12,000 × g for 10 min at 4C to separate the supernatant for Western blotting by decanting.
Cytoplasmic and Nuclear Extracts
The cytoplasmic and nuclear extracts were prepared according to a previously described modified method. 36 After preparation of single-cell suspension (SCS), the testicular cells were washed with PBS and incubated in cell lysis buffer (10 mM HEPES [pH 7.5], 10 mM KCl, 0.1 mM EDTA, 0.5% Nonidet P-40, 1 mM dithiothreitol [DTT], 0.5 mM phenylmethylsulfonyl fluoride, 1% protease inhibitor cocktail [Sigma-Aldrich, St. Louis, MO]) for 15 min. The cell membranes were disrupted by vortex, and cytoplasmic proteins were obtained by centrifugation at 12,000 × g for 10 min at 4C. The pelleted nuclei were washed thrice with cell lysis buffer and incubated in nuclear extraction buffer (20 mM HEPES [pH 7.5], 400 mM NaCl, 1 mM EDTA, 1 mM DTT, 1 mM phenylmethylsulfonyl fluoride, 1% protease inhibitor cocktail) for 30 min. Then the nuclei were vortexed and centrifuged to collect nuclear extract. BCA Protein Assay Kit (Thermo Scientific; Rockford, IL) was used to measure the protein concentration. 28
Isolation of Golgi Fractions
The isolation of Golgi fractions was performed using a Golgi isolation kit (Sigma-Aldrich) according to the manufacturer’s instructions. In brief, the fresh testes were collected, placed in ice-cold PBS, tunica albuginea removed, and then washed with 0.25 M sucrose solution containing 1% (v/v) protease inhibitor cocktail. The tissues were homogenized properly by Dounce homogenizer in 0.25 M sucrose solution. After centrifugation at 3000 × g for 15 min at 4C, the supernatant was transferred into a fresh tube and the sucrose concentration was adjusted to 1.25 M. A discontinuous gradient was prepared by adding 1.84 M, 1.25 M (sample), 1.1 M, and 0.25 M sucrose solution from bottom to top. High-speed centrifugation (Hitachi Micro Ultracentrifuge CS150GXL/CS120GXL) was performed at 120,000 × g for 3 hr at 2C, and Golgi-enriched fractions were collected from 1.1 M/0.25 M sucrose interphase. The fraction was stored at −80C until use.
Western Blotting
Equal amount of proteins were separated by SDS-PAGE and transferred to polyvinylidene difluoride membranes as described.27,28,37 After blocking, the membranes were incubated with rabbit anti-WWOX polyclonal antibody (1:2000 dilution; Sigma-Aldrich), rabbit anti-GM130 polyclonal antibody (1:2000 dilution; Abcam, Cambridge, MA), mouse anti-β-actin monoclonal antibody (1:5000 dilution; Applied Biological Materials Inc., Richmond, BC, Canada), mouse anti-α-tubulin monoclonal antibody (1:3000 dilution; Sigma-Aldrich), and mouse anti-p-Histone H2A.X antibody (1:400 dilution; Santa Cruz Biotechnology, Santa Cruz, CA), followed by incubation with Alexa Fluor 680–conjugated goat anti-rabbit IgG antibody (1:5000 dilution; Invitrogen, Carlsbad, CA) and IRDye 800–conjugated donkey anti-mouse IgG antibody (1:5000 dilution; Rockland Inc., Pottstown, PA). The Odyssey fluorescent imaging system (version 1.2; LI-COR Biotechnology, Lincoln, NE) was used to visualize signals, and densitometric analyses were performed by software ImageJ (version 1.46r; NIH, Bethesda, MD) as previously described. 28 The band intensities were quantified from three independent experiments from PNDs 13 to 42. The Wwox protein expression levels were normalized by α-tubulin, and correlation between expression and age was tested by standard linear regression analysis using a statistical software SPSS (version 21; SPSS Inc., Chicago, IL).
Immunohistochemistry
The freshly collected testes were embedded into 1:1 20% sucrose and Tissue-Tek O.C.T compound (Sakura FineTechnical Co., Ltd.; Tokyo, Japan) and flash-frozen by liquid nitrogen. The blocks were cut into 10 µm thickness and fixed with 4% paraformaldehyde (PFA) solution. After blocking with 2% bovine serum albumin (BSA) for 30 min, the sections were incubated for 2 hr with the following primary antibodies diluted into 2% BSA-PBS solution: rabbit anti-WWOX polyclonal antibody (1:500 dilution), mouse anti-3β-HSD monoclonal antibody (1:100 dilution; Santa Cruz Biotechnology), goat anti-GATA4 polyclonal antibody (1:1000 dilution; Santa Cruz Biotechnology, Dallas, TX), mouse anti-PLZF monoclonal antibody (1:100 dilution; Santa Cruz Biotechnology), mouse anti-DDX4/MVH monoclonal antibody (1:500 dilution; Abcam), mouse anti-SCP3 (1:200 dilution; Santa Cruz Biotechnology), mouse anti-giantin monoclonal antibody (1:500 dilution; Abcam), and mouse anti-golgin-97 monoclonal antibody (1:500 dilution; Santa Cruz Biotechnology). After washing in PBS, the cryosections were incubated with the following secondary antibodies: Alexa Fluor 488 donkey anti-rabbit IgG (1:2000 dilution; Invitrogen), Alexa Fluor 568 donkey anti-rabbit IgG (1:2000 dilution; Invitrogen), Alexa Fluor 568 donkey anti-goat IgG (1:2000 dilution; Invitrogen), and Alexa Fluor 568 donkey anti-mouse IgG (1:2000 dilution; Invitrogen). After 1-hr incubation, the sections were washed in PBS and mounted in ProLong Gold Antifade Reagent containing 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen) for counterstaining. The digital illustration was done by Biozero BZ-X710 fluorescence microscope using software BZ-X analyzer version 1.3.1.1 (Keyence; Tokyo, Japan). The stage of seminiferous tubules and the step of spermatids (ST) were determined by size, shape, and locations of SP and ST stained by DAPI in accordance with the stages of the cycle illustrated. 38
Isolation of Sperm
Mature sperm were collected following the previous methodology. 39 Briefly, the tail of the epididymis was cut into three pieces and put in prewarmed 5 ml DMEM medium (Nacalai Tesque, Inc.). The medium was incubated for 10 min at 37C to allow the sperm to come out from the epididymis. Then the medium was centrifuged (at 8000 × g for 10 min at 10C) and the supernatant discarded. The pelleted sperm were washed and resuspended with PBS. One drop of the sperm solution was put on a slide and allowed to dry in air. The sperm were fixed with methanol for 20 min at −20C for immunocytochemistry. Following blocking with 2% BSA, the sperm were incubated with rabbit anti-WWOX polyclonal antibody (1:500 dilution) and FITC-conjugated peanut agglutinin lectin (1:200 dilution; EY Laboratories Inc., San Mateo, CA). Appropriate secondary antibody and ProLong Gold Antifade Reagent containing DAPI were similarly used as described above.
Preparation of SCS
The SCS were obtained by enzymatic digestion described previously. 40 In brief, the testes were collected immediately after sacrifice and placed in ice-cold PBS. Tunica albuginea was removed carefully and the tissue placed into the digestion medium. The digestion medium was prepared with 1 mg/ml collagenase/dispase (Roche, Mannheim, Germany), 1 mg/ml hyaluronidase (Nacalai Tesque, Inc.), and 1 mg/ml DNAse I (Sigma-Aldrich) in DMEM/F12 (Life Technologies Corporation, Carlsbad, CA). The testicular tissue was minced and incubated for 25 min at 37C with slow continuous rotation, and additional mechanical disruption was performed by gentle pipetting. The digested cell suspension was filtered through 40-µm nylon mesh (Falcon, Durham, NC). The filtered sample was centrifuged at 400 × g for 10 min at 4C and resuspended with PBS. The resuspended SCS were plated onto glass slide and fixed with 4% PFA for immunocytochemistry. After washing, the SCS were visualized similarly as described in the “Immunohistochemistry” section.
Results
Age-related Expression of Wwox in Whole Testis
Both immunohistochemistry and Western blot showed that Wwox expression was absent in
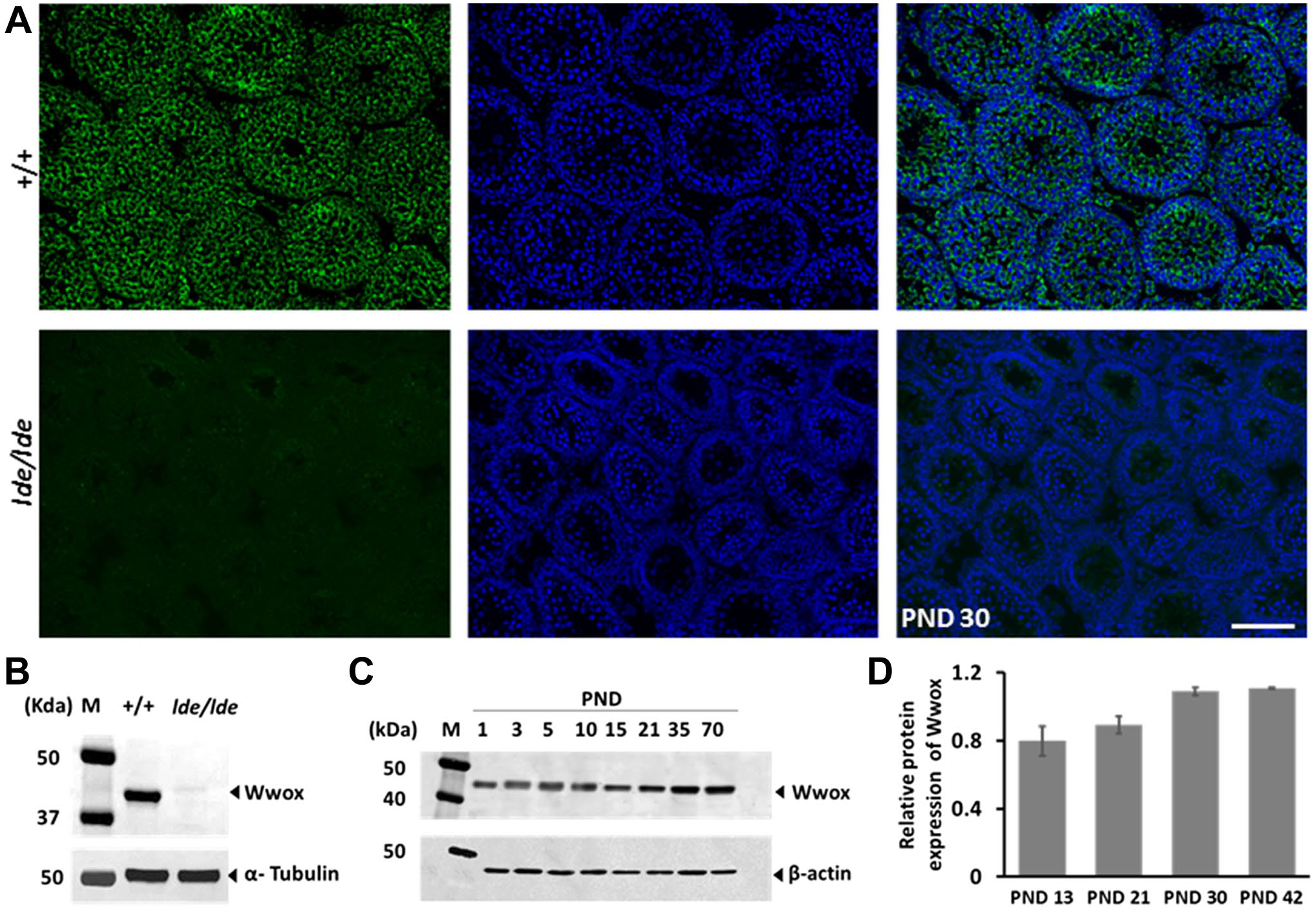
Wwox expression in normal and
Wwox Expression in Perinatal and Adult Testis
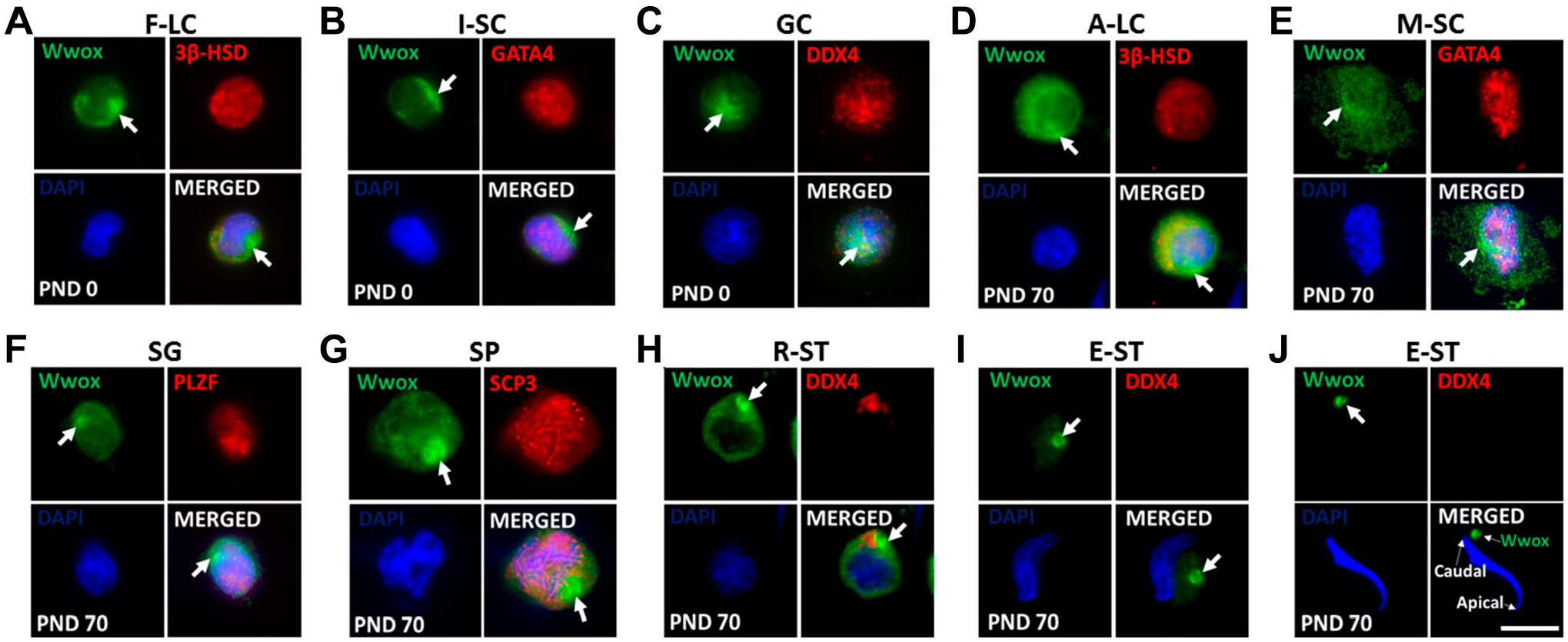
Our Western blotting revealed that Wwox is expressed at all days examined (PNDs 1–70). To identify the cellular localization of Wwox in immature and adult testis, immunohistochemistry was conducted at PNDs 3 and 70. Wwox was detected in gonocytes (GC) (Fig. 2A), fetal-type Leydig cells (F-LC) (Fig. 2C), and immature Sertoli cells (I-SC) (Fig. 2E) at PND 3, and developing germ cells (Fig. 2B), A-LC (Fig. 2D), and mature Sertoli cells (M-SC) (Fig. 2F) at PND 70. Wwox protein was diffusely distributed in the cytoplasm in all cell types. We also found focal intense signals of Wwox (FISWs) in the cytoplasm of germ cells and somatic cells (Fig. 2).
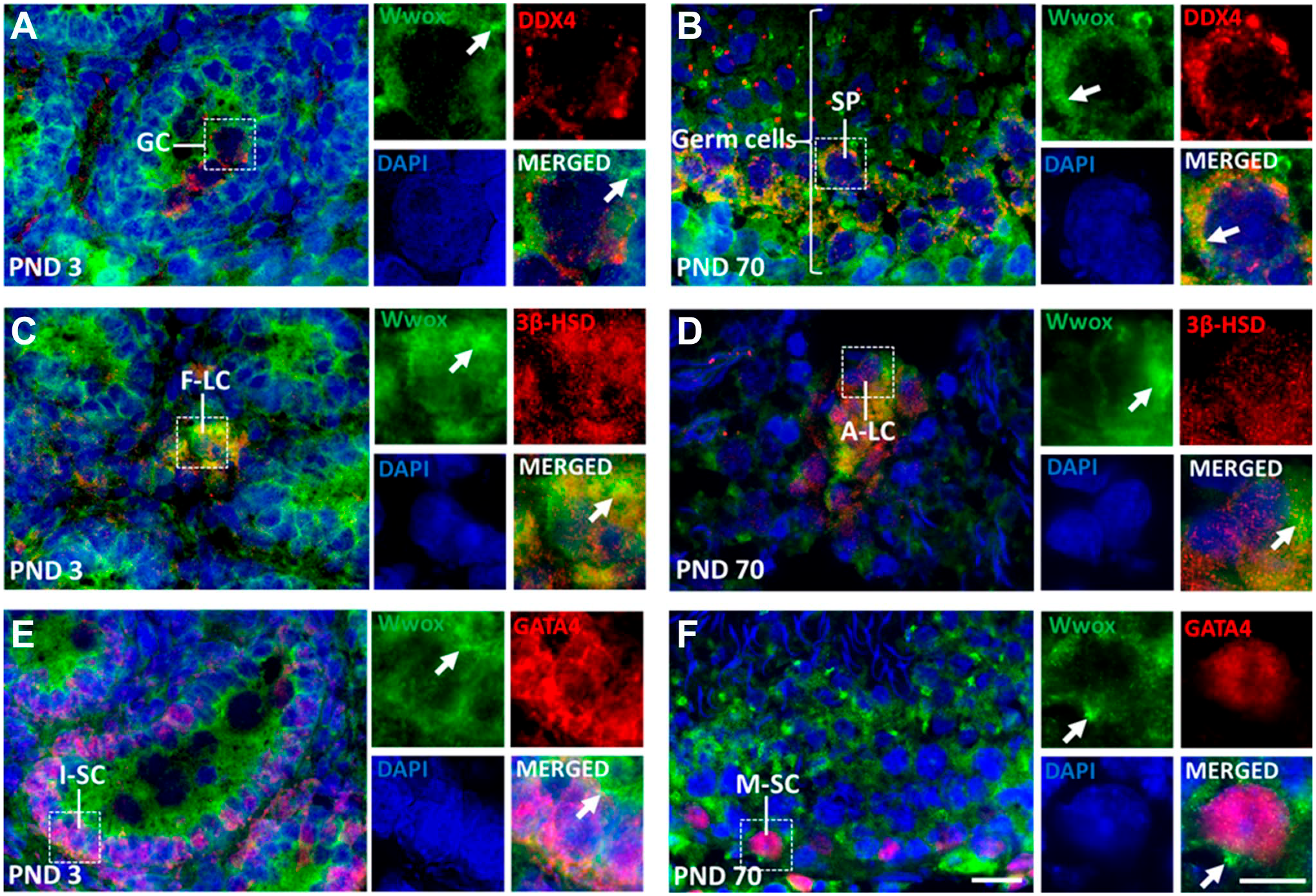
Wwox expression in testicular cells. (A, B) Double immunofluorescence staining of Wwox (green) with DDX4 (red), a germ cell marker; (C, D) 3β-HSD (red), a Leydig cell marker; and (E, F) GATA4 (red), a Sertoli cell marker (somatic cell marker in seminiferous tubules). The nucleus was stained with DAPI (blue). “Arrows” indicate focal intense signals of Wwox (FISWs) in the cytoplasm. Scale bar = 15 μm. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; GC, gonocytes; SP, spermatocytes; F-LC, fetal-type Leydig cells; A-LC, adult-type Leydig cells; I-SC, immature Sertoli cells; M-SC, mature Sertoli cells; PND, postnatal day.
Wwox Expression During the First Round of Spermatogenesis
Mouse and rat models for Wwox deficiency show a decreased number of germ cells and increased apoptosis of SP during the first round of spermatogenesis.27,31 In addition, our Western blotting revealed gradually increased expression of Wwox with testicular development during the first round of spermatogenesis. Therefore, we conducted immunohistochemistry at PNDs 5–52. The germ cells were identified during the first round of spermatogenesis by observing the developmental schedule 42 and differential DDX4 signals 43 and by observing the size, shape, position, and appearance of chromatin in nucleus stained with DAPI. 44 Wwox was detected in all types of developing germ cells, including GC at PND 5 (Fig. 3A), spermatogonia (SG) at PND 10 (Fig. 3B), leptotene spermatocytes (L-SP) at PND 13 (Fig. 3C), zygotene spermatocytes (Z-SP) at PND 17 (Fig. 3D), pachytene spermatocytes (P-SP) at PND 23 (Fig. 3E), diplotene spermatocytes (D-SP) at PND 26 (Fig. 3F), round spermatids (R-ST) at PND 36 (Fig. 3G), and elongated spermatids (E-ST) at PND 52 (Fig. 3H). Wwox was also detected in Sertoli cells and Leydig cells as shown in Fig. 2; no signal was detected in peritubular myoid cells (PM) (Fig. 3H). Wwox protein was diffusely distributed in the cytoplasm with FISWs (Fig. 3). These FISWs were gradually condensed, changed appearance, and increased in size from L-SP to D-SP (Fig. 3C–F). The FISWs were transformed as crescent shaped in Z-SP (Fig. 3D) and condensed spherical shaped in P-SP and D-SP (Fig. 3E and F). In R-ST, FISWs appeared as horse shoe–shaped. In spermatogenesis, cytoplasmic diffuse expression was reduced in ST compared with SP (Fig. 3C–H). These results indicate that Wwox is continuously expressed in developing germ cells, and cytoplasmic localization is changed during the first round of spermatogenesis.
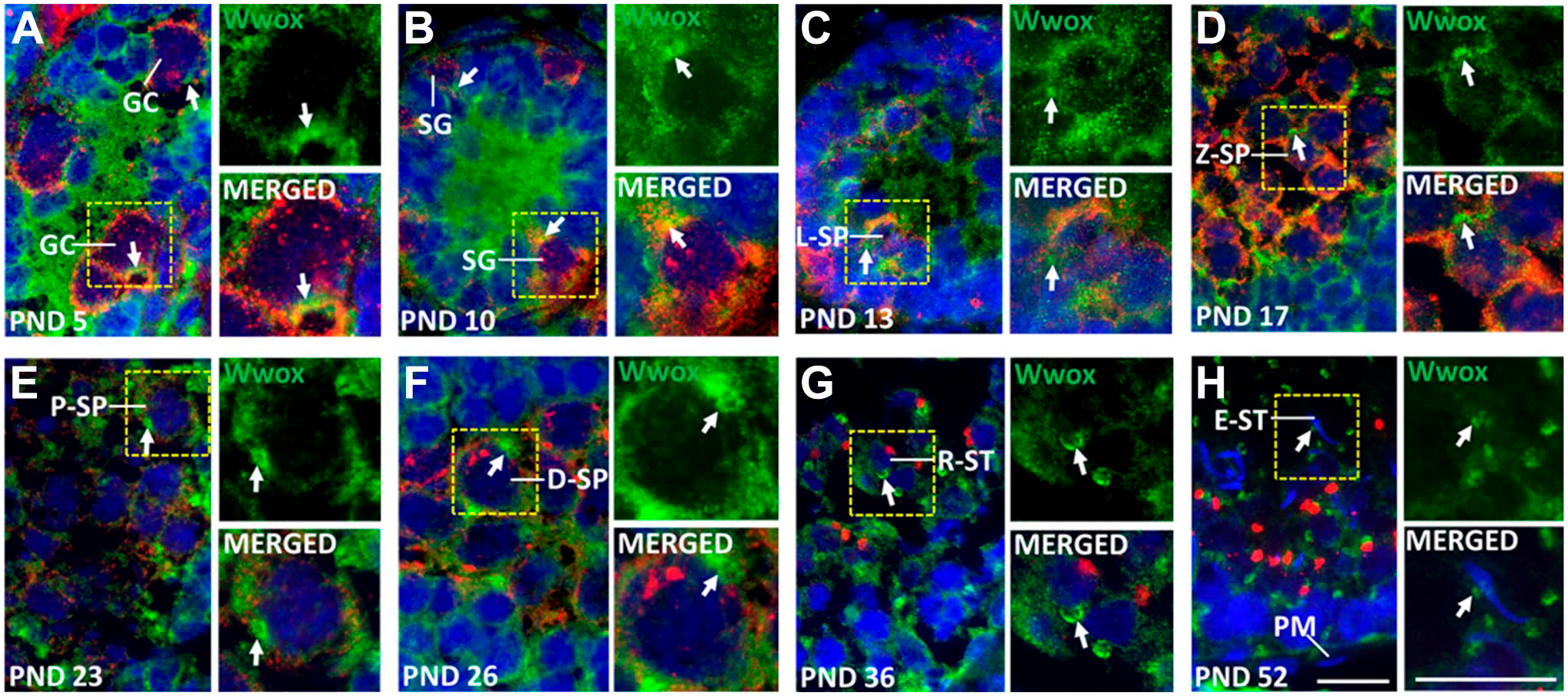
Wwox expression during the first round of spermatogenesis. Double immunofluorescence staining of Wwox (green) and DDX4 (red). The nucleus was stained with DAPI (blue). “Arrows” indicate focal intense signals of Wwox (FISWs) in the cytoplasm. FISWs were smaller and of irregular shape (A–C, H), crescent shape (D), condensed spherical shape (E, F) and horse shoe shape (G). Scale bar = 20 μm. Abbreviations: DAPI, 4’,6-diamidino-2-phenylindole; GC, gonocytes; SG, spermatogonia; L-SP, leptotene spermatocytes; Z-SP, zygotene spermatocytes; P-SP, pachytene spermatocytes; D-SP, diplotene spermatocytes; R-ST, round spermatids; E-ST, elongated spermatid; PM, peritubular myoid cells; PND, postnatal day.
Wwox Expression in Adult Spermatogenesis
Both mice and rat models with Wwox deficiency die soon after weaning.27,30,45 Therefore, the function of Wwox in adult spermatogenesis is unknown, and details on the expression of Wwox have not been revealed in adult spermatogenesis. In this study, immunohistochemistry was conducted in adult testis to reveal the localization of Wwox protein. Seminiferous tubules during spermatogenesis have been classified into 14 stages in rats. 38 At low magnification, Wwox was detected in seminiferous tubules and interstitial tissues, and the tubular expression of Wwox was variable depending on the stages of seminiferous tubules (Fig. 4).
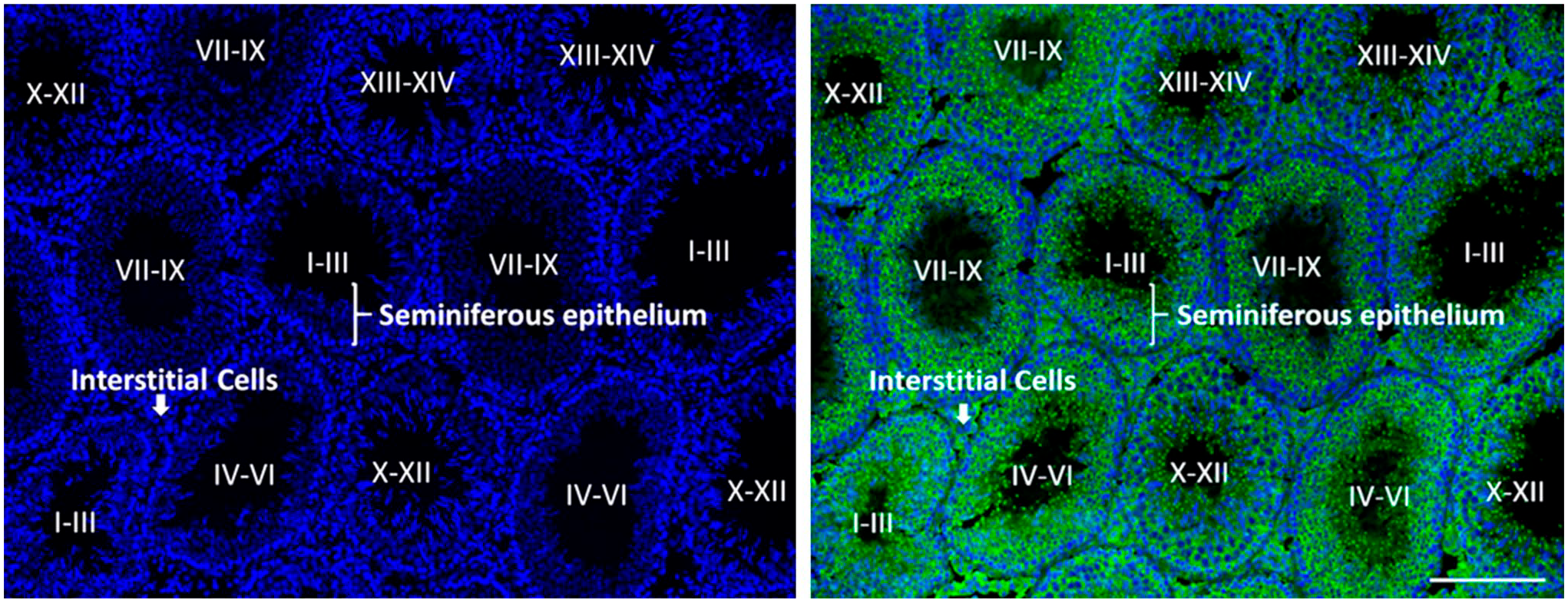
Wwox expression in adult testis. Immunofluorescence staining of Wwox (green) at PND 70. The seminiferous tubules were categorized into five stages (e.g., I–III, IV–VI, VII–IX, X–XII, and XIII–XIV) 38 based on the presence of different cell populations, location, size, shape, and appearance of the nucleus by staining the nuclei with DAPI (blue). Scale bar = 200 μm. Abbreviation: DAPI, 4′,6-diamidino-2-phenylindole; PND, postnatal day.
Wwox was detected in most types of adult testicular germ cells, including SG (Fig. 5A), PL-SP at VI–VIII stages (Fig. 5G–I), L-SP at IX–XI stages (Fig. 5J–L), Z-SP at XII–XIII stages (Fig. 5M and N), P-SP at I–XII and XIV stages (Fig. 5B–M and O), D-SP at XIII stage (Fig. 5N), and secondary SP at XIV stage (Fig. 5O). The appearance of FISWs in SP was similar to that during the first round of spermatogenesis. Step 1–17 ST were positive for the staining (Fig. 5B–O) but step 18–19 ST were negative during VI–VIII stages (Fig. 5G–I). Clear horse shoe–shaped FISWs were found in step 4–13 ST (Fig. 5E–N). No expression was detected in mature sperm collected from the epididymis (Fig. 5P). The expression pattern of Wwox protein in adult spermatogenesis was basically similar to that during the first round of spermatogenesis.
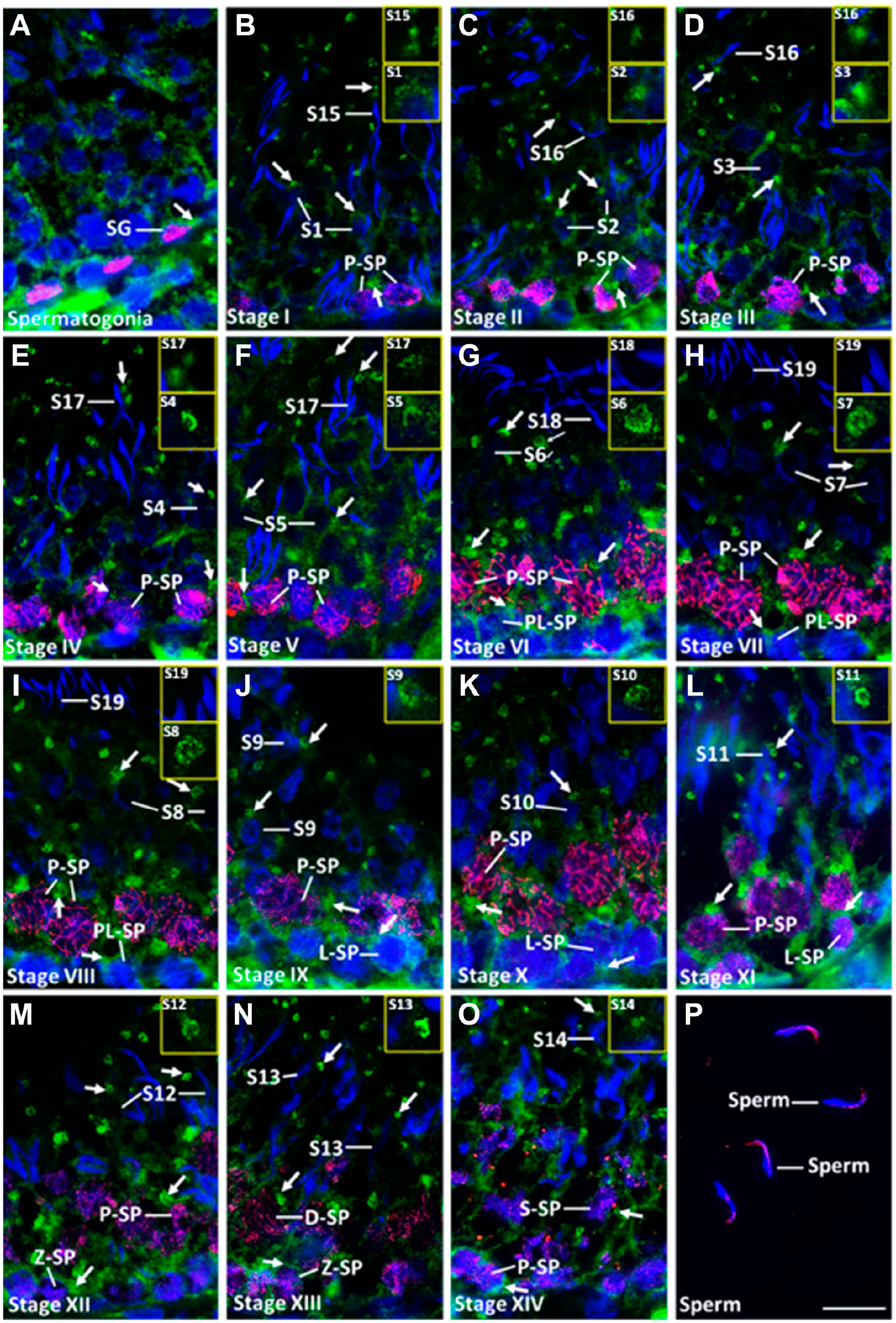
Wwox expression in adult testicular germ cells. Double immunofluorescence staining of Wwox (green) with (A) PLZF (red), (B–O) SCP3 (red), and (P) PNA lectin (red), an acrosomal marker. Nucleus was stained with DAPI (blue). “Arrows” indicate focal intense signals of Wwox (FISWs) in the cytoplasm. FISWs of spermatids were shown in large panels. Scale bar = 20 μm. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; SG, spermatogonia; PL-SP, preleptotene spermatocytes; L-SP, leptotene spermatocytes; Z-SP, zygotene spermatocytes; P-SP, pachytene spermatocytes; D-SP, diplotene spermatocytes; S-SP, secondary spermatocyte; S1-19, step 1–19 spermatids; I–XIV, stages of seminiferous tubules; PNA, peanut agglutinin.
Wwox Expression in Testicular Cells of SCS Samples
Because of the limitation of histochemistry for distinguishing signals between neighboring cells, we prepared SCS samples from testis, identified the specific cell type using markers, and checked the expression pattern of Wwox. Wwox was detected in F-LC, I-SC, GC, A-LC, M-SC, SG, SP, R-ST, and E-ST, and we found that Wwox was diffusely distributed in the cytoplasm with FISWs in all cell types (Fig. 6). In SCS, the FISWs became condensed spherical in SP (Fig. 6G) and horse shoe–shaped in R-ST and E-ST (Fig. 6H and I). The FISWs in early E-ST were located near the middle part (Fig. 6I), but in later E-ST, the signal was translocated to the caudal portion of the head (Fig. 6J).
Wwox expression in single-cell suspension (SCS). Double immunofluorescence staining of Wwox (green) with (A, D) 3β-HSD (red), (B, E) GATA4 (red), (C, H–J) DDX4 (red), and (F) PLZF (red), a spermatogonia marker; and (G) SCP3 (red), a spermatocyte marker. The nucleus was stained with DAPI (blue). “Arrows” indicate focal intense signals of Wwox (FISWs) in the cytoplasm. FISWs were smaller and of irregular shape (A–F, J), condensed spherical shape (G), and horse shoe shape (H, I). Scale bar = 10 μm. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; F-LC, fetal-type Leydig cells; I-SC, immature Sertoli cells; GC, gonocytes; A-LC, adult-type Leydig cells; M-SC, mature Sertoli cells; SG, spermatogonia; SP, spermatocytes; R-ST, round spermatid; E-ST, elongated spermatid; PND, postnatal day.
Subcellular Localization of Wwox in Testicular Cells
To identify the localization of Wwox (specifically FISWs) in testicular cells, immunohistochemistry and Western blotting were performed. Immunohistochemistry showed that almost all FISWs in the cytoplasm were colocalized with giantin for cis-Golgi marker in A-LC, M-SC, SP, and ST (Fig. 7A and B). But it partially colocalized with golgin-97 for trans-Golgi marker (Fig. 7C). To demonstrate the presence of Wwox in the cytoplasm and Golgi apparatus, Western blot was performed for cytoplasmic and nuclear fractions, and further for Golgi fraction. Wwox was detected in the cytoplasm (Fig. 7D) and enriched in Golgi fraction of testicular cells (Fig. 7E).
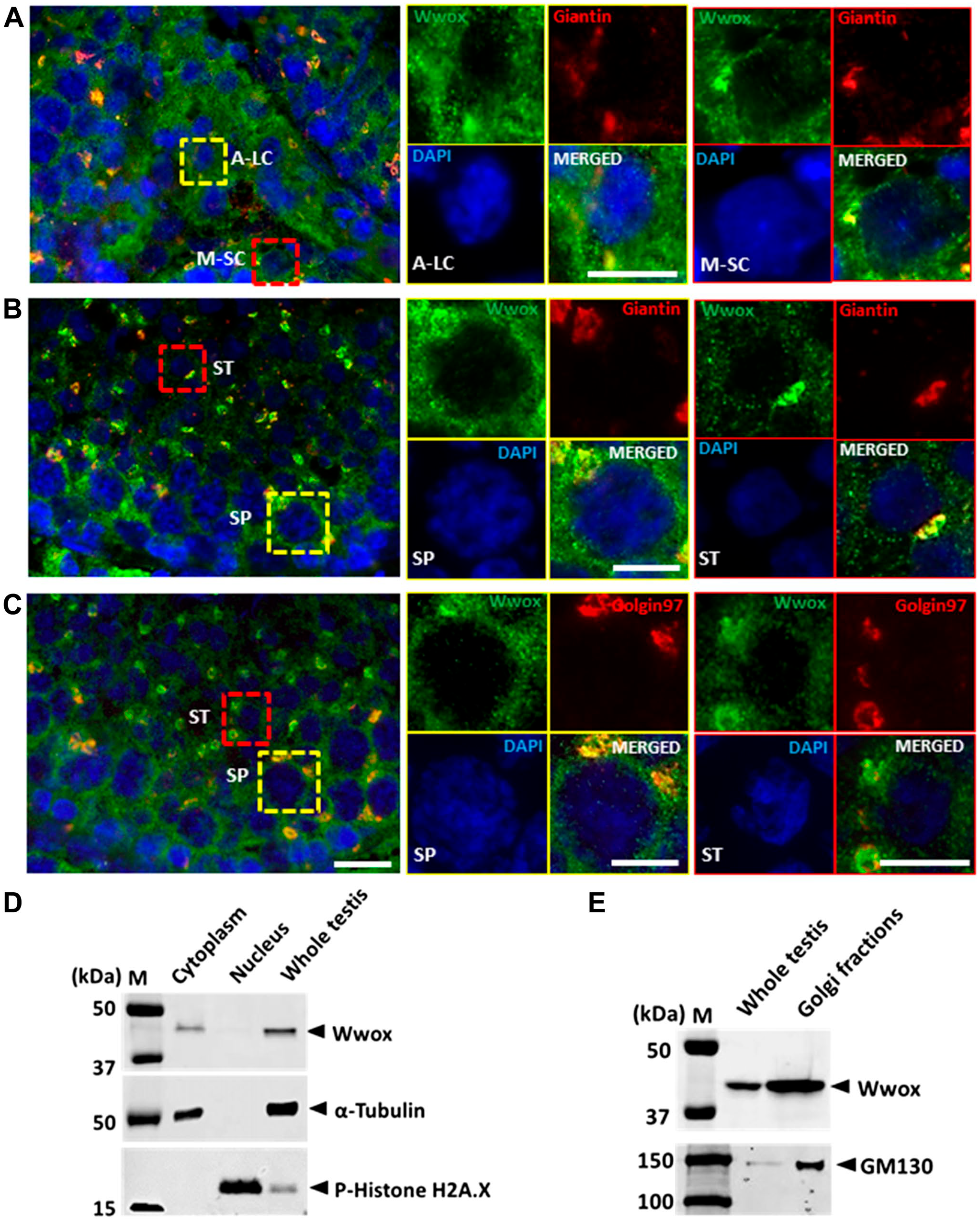
Subcellular localization of Wwox in testicular cells. Double immunofluorescence staining of Wwox (green) with (A, B) giantin (red), a cis-Golgi marker, and (C) golgin97 (red), a trans-Golgi marker. The nucleus was stained with DAPI (blue). (D) Western blot Figure 7. (continued) analysis of Wwox in testis homogenates, cytoplasmic extraction, and nuclear extraction. P-Histone H2A.x and α-tubulin were used as control for nuclear and cytoplasmic protein, respectively. (E) Western blot analysis of Wwox in testis homogenates and Golgi-enriched fractions. GM130 was used as control for cis-Golgi protein. Scale bar = 15 μm. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; A-LC, adult-type Leydig cell; M-SC, mature Sertoli cell; SP, spermatocytes; ST, spermatids.
Subcellular Localization of Wwox in SCS Samples
To confirm the localization of FISWs to cis-Golgi in each cell type, the SCS were subjected to immunofluorescence with giantin for cis-Golgi marker. The testicular cells in SCS were identified by observing the size, shape, appearance of chromatin in nucleus,38,44 cell-specific expression of giantin, 46 and Wwox expression pattern shown in SCS experiment of Fig. 6. The FISWs in the cytoplasm of F-LC (Fig. 8A), I-SC (Fig. 8B), GC (Fig. 8C), A-LC (Fig. 8D), M-SC (Fig. 8E), SG (Fig. 8F), SP (Fig. 8G), and ST (Fig. 8H and I) were exactly colocalized with giantin. In germ cells, condensed spherical and horse shoe–shaped FISWs were dependent on the shapes of cis-Golgi.
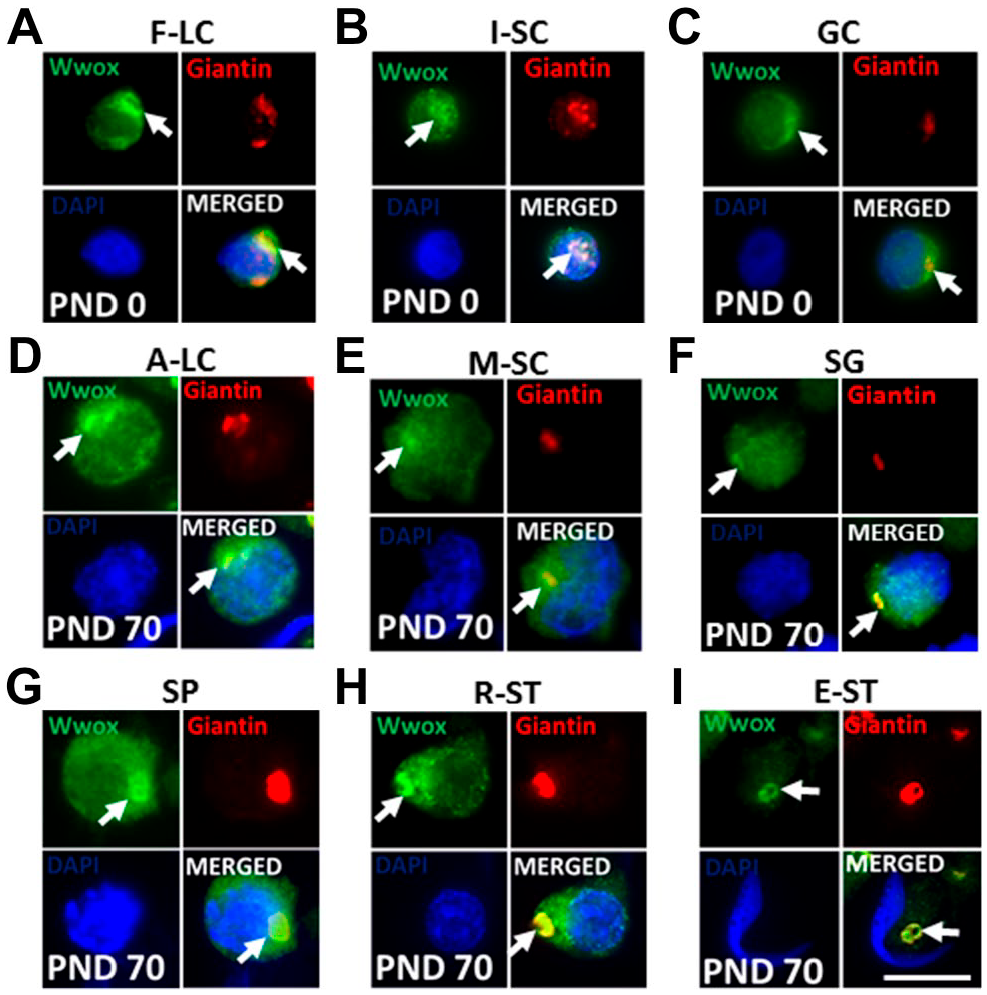
Subcellular localization of Wwox (green) and giantin (red) in testicular SCS. The nucleus was stained with DAPI (blue). “Arrows” indicate focal intense signals of Wwox (FISWs) in the cytoplasm. FISWs were smaller and of irregular shape (A–F), condensed spherical shape (G) and horse shoe shape (H, I). Scale bars = 20 μm. Abbreviations: DAPI, 4’,6-diamidino-2-phenylindole; F-LC, fetal-type Leydig cells; I-SC, immature Sertoli cells; GC, gonocytes; A-LC, adult-type Leydig cells; M-SC, mature Sertoli cells; SG, spermatogonia; SP, spermatocytes; R-ST, round spermatids; E-ST, elongated spermatids; PND, postnatal day.
Discussion
Similar to the previous reports, both immunohistochemistry and Western blot revealed that Wwox was undetectable in
It has been reported that Wwox protein is expressed in seminiferous epithelium, strongly expressed in A-LC, and moderately expressed in M-SC.6,30,31 However, age-related cellular expression and subcellular distribution in testicular cells have not been reported. In this study, initially, we showed that Wwox protein was expressed not only in seminiferous epithelium, A-LC, and M-SC but also in GC, F-LC, and I-SC, indicating that Wwox is expressed in most of the testicular cell types except peritubular myoid cells at all stages. Secondary, we were the first to find FISWs as well as cytoplasmic diffused distribution in all testicular cells, and this unique expression pattern was confirmed by SCS experiment.
During the first round of spermatogenesis, Wwox was expressed in all stages of germ cells (GC, SG, SP, and ST), and the expression pattern changed dynamically with germ cell differentiation. Interestingly, the FISWs gradually increased in size from L-SP to D-SP and became condensed spherical shaped in P-SP and D-SP. Similar expression of Wwox was observed in adult testicular germ cells with variable expression within seminiferous tubules at low magnification. In adult testis, we could identify the detailed expression pattern of Wwox in ST. The horse shoe–shaped FISWs were found in R-ST and E-ST, and the Wwox signals disappeared in step 18 to sperm, indicating that Wwox might be localized to a certain cytoplasmic organelle except mitochondria and lost during cytoplasmic elimination at the end of spermiogenesis. In
From the view of molecular structure, Wwox contains a nuclear localization signal (amino acids 50–55) in between two N-terminal WW domains and a mitochondrial targeting sequence (amino acids 209–273) mapping to the SDR domain region. 8 Many research groups using diverse experimental methodologies reported that Wwox/WOX1 localized in the cytoplasm,6,9,10,13,49 nucleus,9–11 perinuclear Golgi apparatus,3,50 endoplasmic reticulum, 51 mitochondria,8,52,53 lysosome and late endosome,50,54–56 and cell membrane. 57 However, because of unique horse shoe shape and disappearance in sperm, FISWs were suspected to be related to Golgi apparatus. Our double immunofluorescence analysis revealed that FISWs were colocalized with giantin of cis-Golgi markers. Biochemically, Wwox was also detected in Golgi-enriched fractions of adult testis. It has been reported that the Golgi complex appeared as spherical shaped in P-SP and increased significantly before completion of meiosis I. 58 In accordance with the report, the FISWs were also condensed spherical shaped and increased in size in P-SP and D-SP. The Golgi apparatus plays an important role in acrosomal biogenesis and gradually translocates from the acrosomal region to the caudal end of the head of E-ST and disappears after step 17 ST.59,60 Our SCS experiment and immunohistochemistry revealed almost complete colocalization of Wwox with cis-Golgi marker giantin in E-ST, the translocation of the FISWs from the middle in early E-ST to the caudal portion of the head in later E-ST, and disappearance of the signal after step 17 ST. After step 7 ST, the innermost Golgi cisternae and trans-Golgi networks disappear from the Golgi apparatus,59,60 but Wwox signals were detected until step 17 ST, indicating that Wwox was localized in cis-Golgi. However, Wwox does not have any membrane localization signals, which suggested that it might be localized into the cis-Golgi compartment via interacting with other Golgi protein.
This unique localization of Wwox in cis-Golgi might provide a clue to consider the function of Wwox during somatic and germ cells differentiation. The intact SDR domain is essential for localization of WWOX protein in Golgi apparatus,
48
and the SDR domain is considered important for steroid metabolism in testis.2,26,30 The
Footnotes
Acknowledgements
We thank all the people who worked hard to maintain the rats in our laboratory.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
HS and MAAM conceptualized the study. MAAM performed the experiments, analyzed the data, interpreted the results, and wrote the paper. MN, AD, YT, and KK supported the experiments. HS supervised the experiments and wrote the paper. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid for Scientific Research to H.S. (No. 20H03157) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
