Abstract
Cajal-Retzius cells in layer 1 of the developing cerebral cortex and their product of secretion, reelin, an extracellular matrix protein, play a crucial role in establishing the correct lamination pattern in this tissue. As many studies into reelin signaling routes and pathological alterations are conducted in murine models, we used double-labeling and confocal microscopy to compare the distribution of the cell-specific markers, calretinin and calbindin, in reelin-immunoreactive cells during postnatal rat and mouse neocortical development. In the rat, neither calretinin nor calbindin colocalized with reelin in Cajal-Retzius cells at P0-P2. From P5 to P14, the colocalization of reelin and calretinin was commonly found in presumptive rat subpial piriform cells. These cells progressively lacked calretinin expression and persisted into adulthood as part of the pool of layer 1 reelin-positive interneurons. Conversely, in the mouse, reelin-immunoreactive Cajal-Retzius cells colocalized with calretinin and/or calbindin. Subpial piriform cells containing reelin and calretinin were identified at P5-P7, but lacked calretinin expression at P14. In adult mice, as in the rat, reelin-immunoreactive cells did not colocalize with calcium-binding proteins. Our results reveal a complex neurochemical profile of layer 1 cells in the rat neocortex, which makes using a single calcium-binding protein as a marker of rodent reelin-immunoreactive cells difficult.
Introduction
The mammalian neocortex is a highly ordered structure. The earliest generated neurons in the cerebral cortex are preplate cells, which split to form the subplate and marginal zone cells when the cortical plate develops within the preplate (for a review, see Supèr et al. 1998; Marín-Padilla 1998). Cortical plate neurons are generated in an orderly progression, with cells of the deepest layers arising first, followed by cells of the middle layers and finally those of the upper layers. Each successively generated post-mitotic neuron must bypass the predecessor neurons that migrate along the same glial fiber before ultimately settling at the outermost level of the cortical plate just below the marginal zone (Angevine and Sidman 1961; Hicks and D’Amato 1968; Rakic 1972). Whereas radial migration is characteristic of pyramidal glutamatergic neurons, GABAergic interneurons migrate tangentially into the developing neocortex from the germinal zones of the basal ganglia primordium (for a review, see Marín and Rubinstein 2003).
Cajal-Retzius cells of the marginal zone, one of the most enigmatic and controversial classes of neurons, play a crucial role in radial migration given their capacity to synthetize and secrete reelin, an extracellular matrix protein (D’Arcangelo et al. 1995, 1997; Hirotsune et al. 1995; Ogawa et al. 1995). Reelin is absent in the reeler mutant mouse, which shows an inverted lamination plan (Caviness and Sidman 1973; Caviness 1982). Reelin binds to very low density lipoprotein receptor (VLDR) and apoE receptor 2 (ApoER2) (D’Arcangelo et al. 1999; Hiesberger et al. 1999) which regulate disabled-1 tyrosine phosphorylation (Howell et al. 1999) and cytoskeletal dynamics in post-mitotic migrating neurons (Chai et al. 2009; Meseke et al. 2013). Furthermore, reelin influences neuronal migration through radial glia regulation (Supèr et al. 2000; Förster et al. 2002) as there is evidence that lipoprotein receptors ApoER2 and VLDLR, as well as cytoplasmic adaptor protein Dab1, are also located in radial glia precursors (Luque et al. 2003). Reelin is expressed not only by Cajal-Retzius cells as the interneurons of the cortical plate express reelin from E18 onward (Alcántara et al. 1998). Even when cortical neurons terminate radial migration, reelin continues to be expressed into adulthood, preferentially in GABAergic interneurons (Pesold et al. 1998). In the adult brain, reelin can modulate glutamatergic neurotransmission and synaptic plasticity (Beffert et al. 2005) through the regulation of activity (Chen et al. 2005) and subunit composition (Groc et al. 2007) of postsynaptic glutamate receptors. Neuropsychiatric disorders, including autism, schizophrenia, bipolar disorder, major depression, Alzheimer’s disease and lissencephaly, share a common abnormal reelin expression feature in the brain (for a review, see Folsom and Fatemi 2013).
Establishing cell class-specific markers for Cajal-Retzius cells is a complex task. Besides reelin, calcium-binding protein calretinin is one of the best established markers of Cajal-Retzius cells (Verney and Derer 1995; Ogawa et al. 1995, Del Río et al. 1995; Alcántara et al. 1998). However in the rat brain, calretinin does not label the complete Cajal-Retzius cell population (Meyer et al. 1998). Furthermore, calbindin, another calcium-binding protein, is also widely distributed in the marginal zone (Enderlin et al. 1987; Huntley and Jones 1990; Sánchez et al. 1992; Verney and Derer 1995), although its expression is more restricted to the embryonic period and, in rodents, it is virtually absent in reelin-immunoreactive (reelin-ir) cells (Alcántara et al. 1998; Meyer et al. 1998). Given the widespread use of rats and mice as experimental models to study cerebral cortex development, comparative studies of cell-specific markers across species are required. A rigorous comparison of the preplate and its derivates during embryonic development of the rat and mouse brains has shown noticeable differences as compared to the cell-specific markers found (Espinosa et al. 2009).
In the present work, we focus on studying reelin-ir cells in layer 1 during neocortical developmental. We compared the expression of calcium-binding proteins calretinin and calbindin in reelin-ir cells in postnatal rat and mouse brains. We observed different colocalization patterns of calretinin and reelin across murine species in postnatal week 1. From P14 onwards, the pattern was similar between rat and mouse, with a predominant reelin-ir population that does not contain calretinin. Regarding calbindin, an unsuspected reelin/calbindin-ir subpopulation of layer 1 mouse cells was found in postnatal week 1. These results reveal noticeable differences between the rat and mouse layer 1 neocortex in postnatal week 1, with an evident diversity of cell subpopulations in the layer 1 rat neocortex.
Materials & Methods
Animals and Tissue Processing
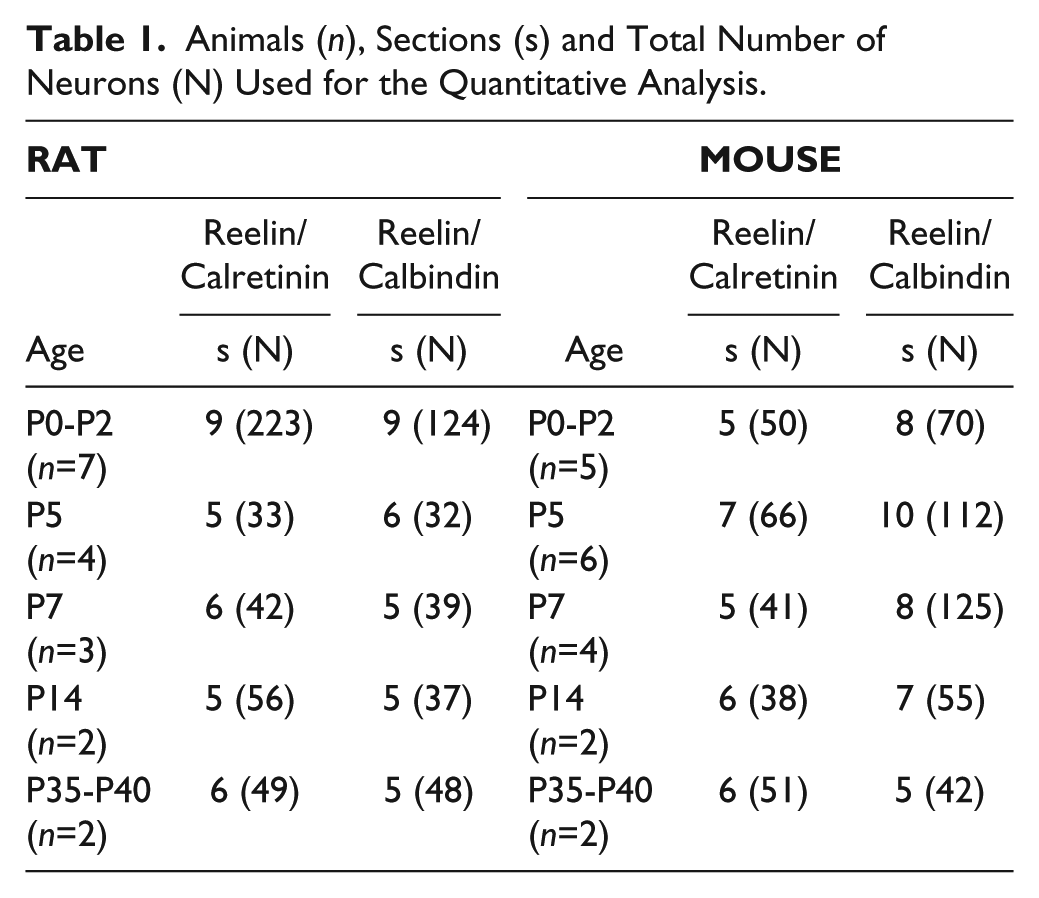
Thirty-seven rodents from postnatal day (P) 0 to P40 were used in this study (Charles River, Barcelona, Spain; Animal House of the Universidad Miguel Hernández, San Juan de Alicante, Spain and Animal House of the Universidad de Castilla-La Mancha, Albacete, Spain). Eighteen Wistar rats were grouped into the following developmental ages: P0-P2 (n=7), P5 (n=4), P7 (n=3), P14 (n=2) and P35-P40 (n=2). Nineteen postnatal C57BI mice were grouped as follows: P1-P3 (n=5), P5 (n=6), P7 (n=4), P14 (n=2) and P35-P40 (n=2). All procedures were approved by the authors’ institutions and conformed to Spanish (Royal Decree 1201/2005; Law 32/2007) and European Union (2010/63/UE) regulations for the use and care of animals in research.
Animals were deeply anesthetized with ketamine (75 mg/kg, Parke-Davis; Alcobendas, Spain) and xylazine (5 mg/kg, Dibapa; Barcelona, Spain). They were perfused through the left ventricle with 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4. Brains were dissected out, post-fixed in fresh fixative at 4C for 4 hr, and stored for 2 days in 30% sucrose for cryoprotection. Coronal 50-µm-thick sections of the neocortex were cut by a freezing microtome and were free floating-processed for double immunofluorescence.
Double Immunostaining and Confocal Microscopy
We used the following antibodies for double immunostaining purposes: 1) Rabbit anti-calbindin D-28k (1:5000) was purchased from Swant (#CB-38a; Bellinzona, Switzerland) and produced against recombinant rat calbindin D28k. The antibody recognizes a 28-kDa band in western blots in different species, including rat and mouse. No staining is observed in calbindin knock-out mice (Airaksinen et al. 1997). 2) Rabbit anti-calretinin (1:2000), from Swant (#7699/3H), was produced by immunization with recombinant human calretinin and characterized by western blotting and immunohistochemistry. The serum recognizes a single protein band of the appropriate molecular weight in different species, including rat and mouse (Schwaller et al. 1993). No immunolabeling has been found in the neocortex of the calretinin knock-out mice (Schiffmann et al. 1999). 3) Mouse monoclonal anti-reelin antibody (1:1000), clone G10, was produced against amino acids 164-496 of the recombinant reelin protein and purchased from Millipore (#MAB5364, Millipore; Temecula, CA). The antibody labels a band of approximately 388 kDa (de Bergeycket al. 1998) and two proteolytic reelin fragments of estimated sizes 300 and 180 kDa (Lambert de Rouvroit et al. 1999). The staining pattern in layer 1 rat and mouse neocortex corresponded to that described in the literature (de Bergeyck et al. 1998; Luque et al. 2003; Soria and Fairen 2000; García-Moreno et al. 2007). Rabbit polyclonal antibodies (anti-calretinin or anti-calbindin) were used as the first primary antibodies. Monoclonal anti-reelin antibody, G10 was used as the second primary antibody.
Free-floating sections were pretreated with 4% BSA, 3% normal goat serum and 0.1% Triton X-100 in phosphate-buffered saline (PBS) at room temperature for 30 min, and were then incubated with the primary antibodies at room temperature for 24 hr. All of the following steps were carried out in the dark at room temperature. After several rinses in PBS, sections were incubated with the following secondary antibodies: biotinylated goat anti-rabbit IgG (Vector Laboratories Inc.; Burlingame, CA) 1:100, for calcium-binding proteins, and Cy3-conjugated anti-mouse IgG (Amersham; Arlington Heights, IL) for reelin, diluted at 1:200, for 60 min. After rinsing, sections were incubated in Cy2-conjugated streptavidin (Amersham), diluted at 1:1000 for 60 min. After rinsing in PBS, sections were mounted and coverslipped with Cytoseal (Stephens Scientific; Wayne, NJ). The immunofluorescent sections were examined under a Leica TCS NT laser scanning confocal microscope equipped with an argon/krypton-mixed gas laser, with emission peaks at 488, 568 and 647 nm. A Zeiss LSM 710 laser scanning confocal microscope of continuous spectral detection, equipped with excitation laser lines at 405, 458, 488, 514, 561 and 633 nm, was also used in some experiments. A minimum of four images in each confocal image stack were recorded through separate channels for Cy2 (absorbance: 489, emission: 508 nm) and Cy3 (Cy3 absorbance: 550, emission: 570 nm).
Quantification and Statistical Analysis
To estimate the percentages of colocalization, manual counting was performed in confocal image series obtained from a minimum of two animals per postnatal age (Table 1). Differences in the distribution of cell labeling between rat and mouse at the same developmental ages were assessed using the chi-squared test. For this, a contingency table was created to compare the number of neurons immunoreactive to reelin, calretinin or calbindin, and double labeled (columns) between rat and mouse (files) for each age.
Animals (n), Sections (s) and Total Number of Neurons (N) Used for the Quantitative Analysis.
Results
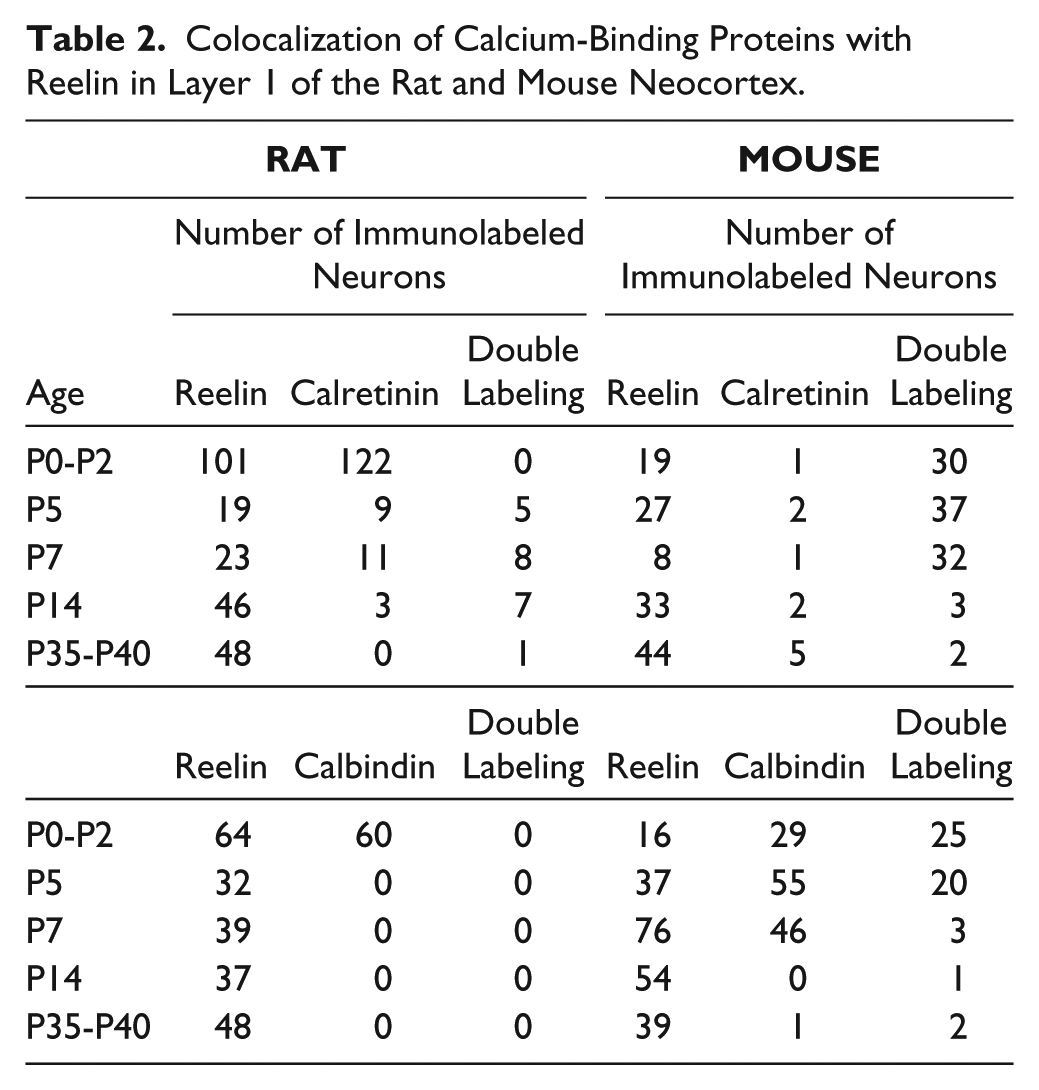
Table 1 provides the total number of animals and cells analyzed in this study. Table 2 shows the absolute number of counted neurons that were single or double labeled during the different postnatal ages.
Colocalization of Calcium-Binding Proteins with Reelin in Layer 1 of the Rat and Mouse Neocortex.
Colocalization of Calretinin and Reelin in Layer 1 of the Rat and Mouse Neocortex
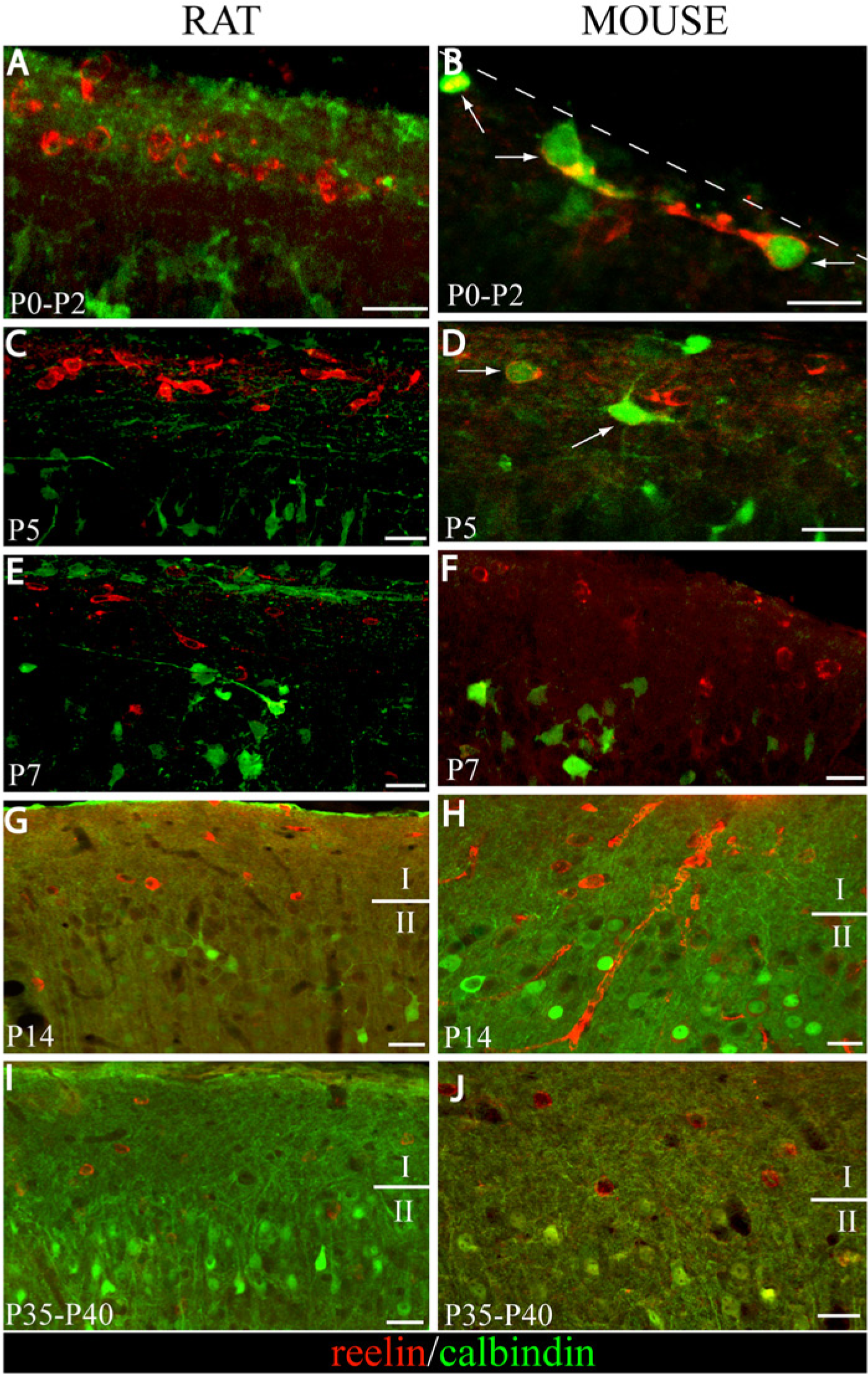
In the rat neocortex, no colocalization was found at P0-P2 (Fig. 1A). The colocalization percentages of reelin with calretinin for each stage are depicted in Figure 2. Whereas 45.3% of cells expressed only reelin at P0-P2, the remaining 54.7% were exclusively immunostained for calretinin. At P5, 15% of labeled cells were immunoreactive to both markers (arrow in Fig. 1C). This number hardly changed at P7, where 19% of cells were double-labeled (arrows in Fig. 1E). The distribution of cells immunoreactive only to calretinin or reelin was similar at P5 and P7. The presence of a number of cells that were immunoreactive only to calretinin at these ages is also noteworthy (27% at P5 and 26% at P7). The number of cells that were labeled only to reelin increased markedly at P14 (82.14%). Whereas the number of calretinin-ir cells (which were negative to reelin) was scarce (5.35%), some reelin-ir cells (12.5%) maintained calretinin expression (arrows in Fig. 1G). At P35-P40, the majority of cells expressed only reelin (97.95%).
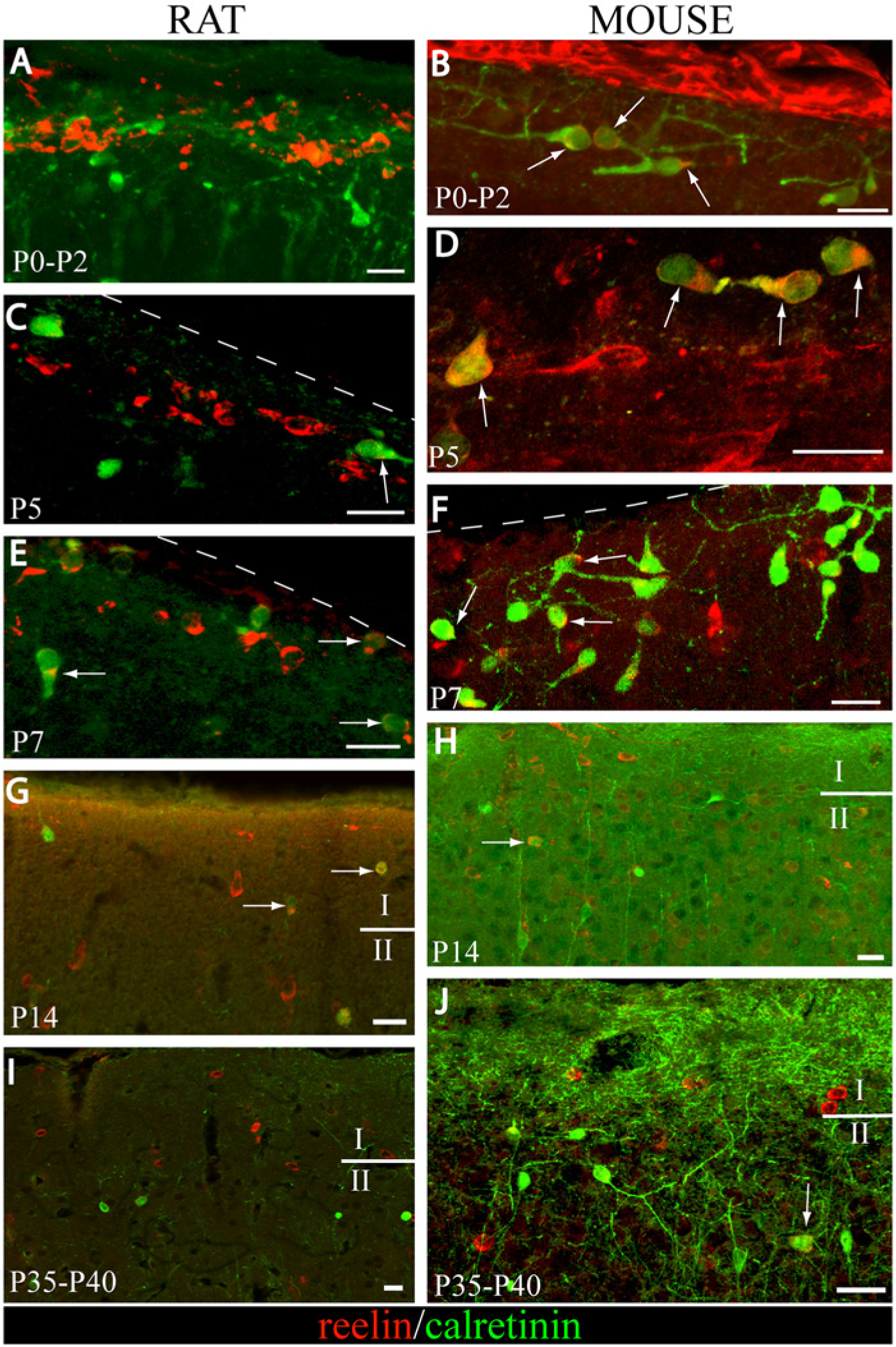
Expression of calretinin and reelin in layer 1 cortex during rat and mouse postnatal neocortical development (A-J). Confocal images of calretinin (Cy2, green) and reelin (Cy3, red) immunoreactivities in rat brains from P0-P2 to P35-P40 (A, C, E, G and I). Whereas reelin-ir cells do not express calretinin at P0-P2, a low level of colocalization is detected from P5 to P14 (arrows in C, E and G). Colocalization is scarcely found at P35-P40 (I). Confocal images of the calretinin (Cy2, green) and reelin (Cy3, red) immunoreactivities in mouse brains from P0-P2 to P35-P40 (B, D, F, H and J). Reelin-ir cells that express calretinin are the most abundant subpopulation (arrows in B, D and F). From P14 to P35-P40, reelin-ir cells hardly colocalize with calretinin. At these ages, double-labeled cells are frequently found in deeper layers (arrows in H and J). I, layer 1; II layer 2. Dashed lines indicate the pial border. Scale bars correspond to 20 µm.
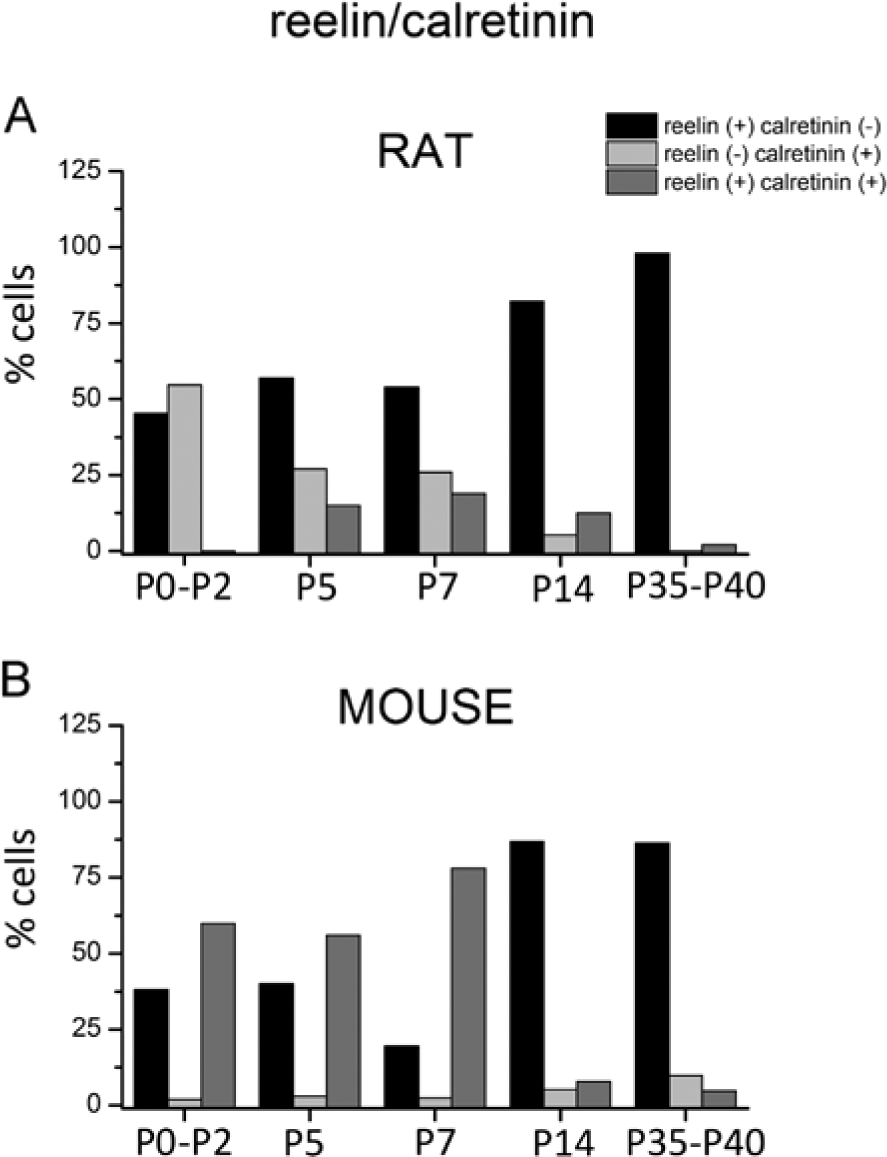
Colocalization of calretinin with reelin in layer 1 of the rat and mouse neocortex expressed as a percentage of the total number of cells. (A) No colocalization is observed in the rat neocortex at P0-P2, and there is a low degree of colocalization from P5 onwards. (B) Double-labeled cells are the main subpopulation in the mouse neocortex in postnatal week 1.
In the mouse neocortex, 60% of the colocalization between calretinin and reelin was found at P0-P2 (arrows in Fig. 1B). Reelin-positive layer 1 cells, which do not contain calretinin, represented 38% of the total population. At P5, double-labeled neurons represented 56% of the total cells (arrows in Fig. 1D). The remaining neurons expressed reelin (40%), and there were only a few calretinin-immunopositive cells (3%). At P7, the percentage of double-labeled cells (arrows in Fig. 1F) increased up to 78%, with the remaining 19.5% of reelin cells not immunoreactive for calretinin. Differences were statistically significant (Chi-square) from P0-P2 to P7, with respect to the distribution of both markers at the same ages in the layer 1 rat neocortex (Table 3). Reelin-ir cells, which do not contain calretinin, were the main subpopulation (Figs. 1H and 1J) at P14 (86.8%) and P35-P40 (86.2%). Besides, the double-immunolabeled interneurons were frequently found at lower layers (arrow in Fig. 1H). No differences were found across species from P14 to P35-40.
Statistical Analysis of the Distribution of Calcium-Binding Proteins in Reelin-ir Cells between Rat and Mouse Neocortex at Different Developmental Ages.
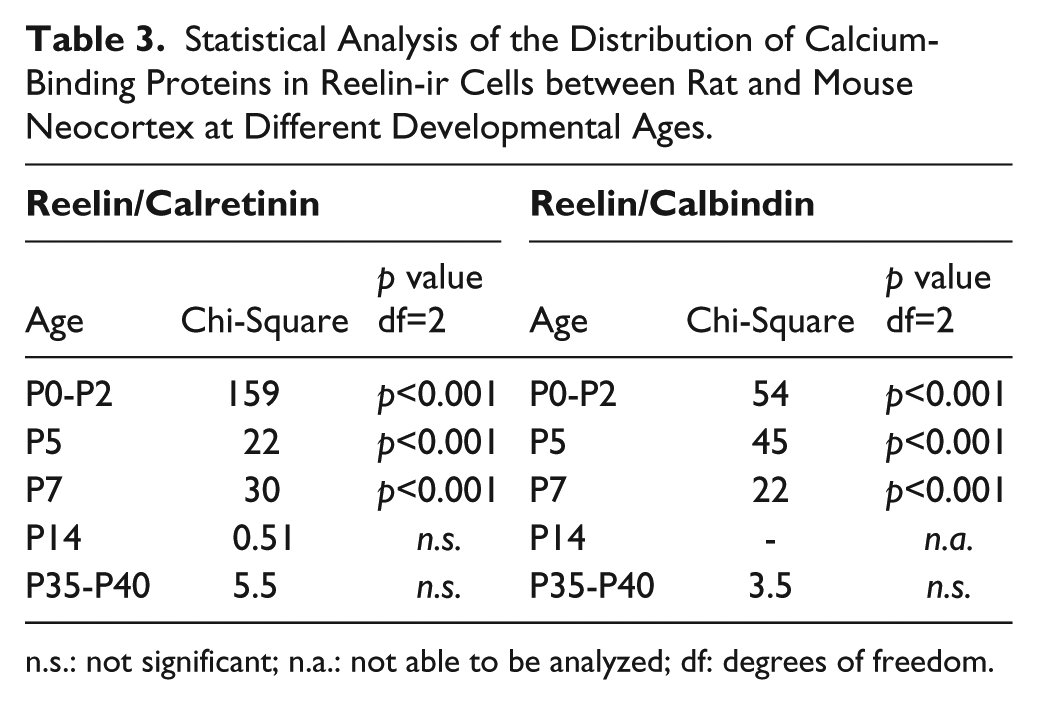
n.s.: not significant; n.a.: not able to be analyzed; df: degrees of freedom.
Colocalization of Calbindin and Reelin in Layer 1 of the Rat and Mouse Neocortex
In the rat neocortex, no double-labeling was observed at any of the ages studied (Fig. 3). The immunolabeling distribution is illustrated in Figure 4. At P0-P2, 51.6% of the cells were labeled for reelin, and the remaining 48.4% were immunoreactive to calbindin (Fig. 3A). At later postnatal developmental stages (P5 onward), calbindin-positive cells were completely lacking in layer 1 (Fig. 1C, 1E, 1G and 1I).
Expression of calbindin and reelin in layer 1 during rat and mouse postnatal neocortical development (A-J). Confocal images of the calbindin (Cy2, green) and reelin (Cy3, red) immunoreactivities from P0-P2 to P35-P40 in rat brains (A, C, E, G and I). The reelin-ir cells in layer 1 do not express calbindin during postnatal development. Furthermore, no cells are immunolabeled for calbindin from P5 onward (E, G I). Confocal images of the calbindin (Cy2, green) and reelin (Cy3, red) immunoreactivities in mouse brains from P0-P2 to P35-P40 (B, D, F, H and J). Two reelin-ir cells that colocalize with calbindin are shown at P0-P2 and P5 (arrows in B and D). Colocalization is barely detectable from P7 onward (F, H, J). I, layer 1; II layer 2. Dashed lines indicate the pial border. Scale bars correspond to 20 µm.
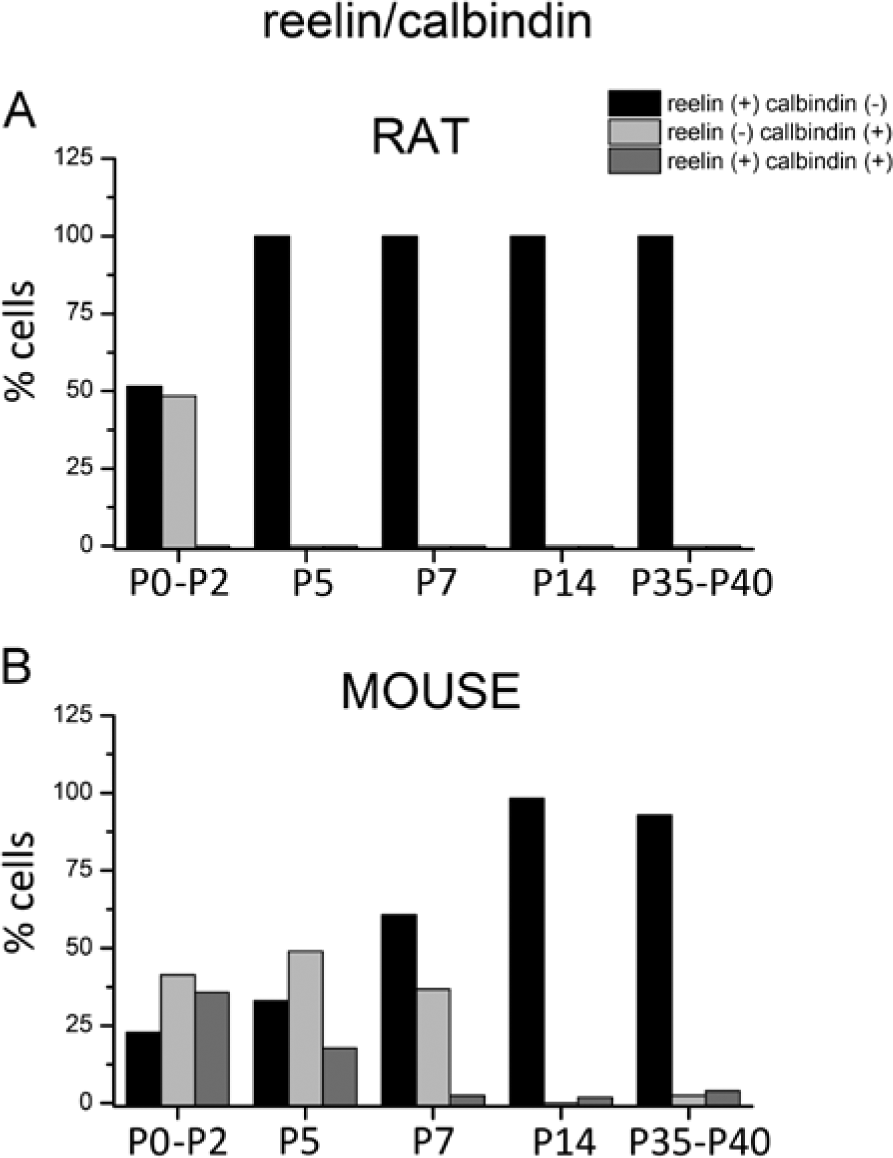
Colocalization of calbindin with reelin in layer 1 of the rat and mouse neocortex expressed as a percentage of the total number of cells. (A) No colocalization in the rat neocortex is observed at any age studied. (B) In the mouse neocortex, calbindin is expressed in layer 1 in week 1, with a moderate level of colocalization noted at P0-P2, which drastically drops with age.
In the mouse neocortex, in contrast, an unexpected colocalization (35.7%) between reelin and calbindin was observed at layer 1 at P0-P2 (Fig. 3B). Two horizontal double-labeled cells, indicated by arrows, are shown in Figure 3B. The number of reelin cells expressing calbindin lowered with age: 17.8% of cells were double-labeled at P4 (arrows in Fig. 3D) but only a very small percentage of reelin-ir cells (2.4%) expressed calbindin at P7. At these ages, a subpopulation of calbindin-ir neurons that was not immunoreactive to reelin was also present. Differences were statistically significant (Chi-square) at P0-P2, P5 and P7, with respect to the distribution of both markers at the same ages in the layer 1 rat neocortex (Table 3). At later developmental stages, from P14 and P35-P40, reelin-ir cells, which do not express calbindin, were the predominant subpopulation (Fig. 3H and J). At P14, the high percentage of cells immunoreactive only to reelin (98.18%) indicated that no differences existed between both rodent species. Furthermore, no significant differences were found across species at P35-P40.
Discussion
Here we have studied reelin, calretinin and calbindin in the neocortical layer 1 of rats and mice during postnatal development. Our objective was not to determine the origin and fate of the populations described herein but to compare the distribution of cell-specific markers in rats and mice during the postnatal period, for which numerous studies into the physiological properties of these cells have been done in pioneer studies (Hestrin and Amstrong 1996; Zhou and Hablitz 1996; Mienville and Pesold 1999). We used double-labeling experiments to colocalize reelin with calcium-binding proteins calretinin or calbindin. The major findings in this study are: (1) With relation to the reelin/calretinin double-labeling data, we observed two transient subpopulations of layer 1 cells in postnatal week 1 in the rat neocortex. Whereas one of the subpopulations was identified as reelin(+)/calretinin(-), the other one was reelin(-)/calretinin(+). Moreover, at the same ages, a significant number of cells in the mouse brain were only reelin-ir. These results contradict the concept that murine Cajal-Retzius cells commonly co-express calretinin and reelin. (2) In the mouse neocortex, we demonstrated the existence of a double-labeled reelin/calbindin cell subpopulation that was transiently observed in postnatal week 1. The proposed scheme in Figure 5 summarizes the comparative aspects of rat and mouse layer 1 development, based on our own results taken together with data published by different authors, as discussed below.
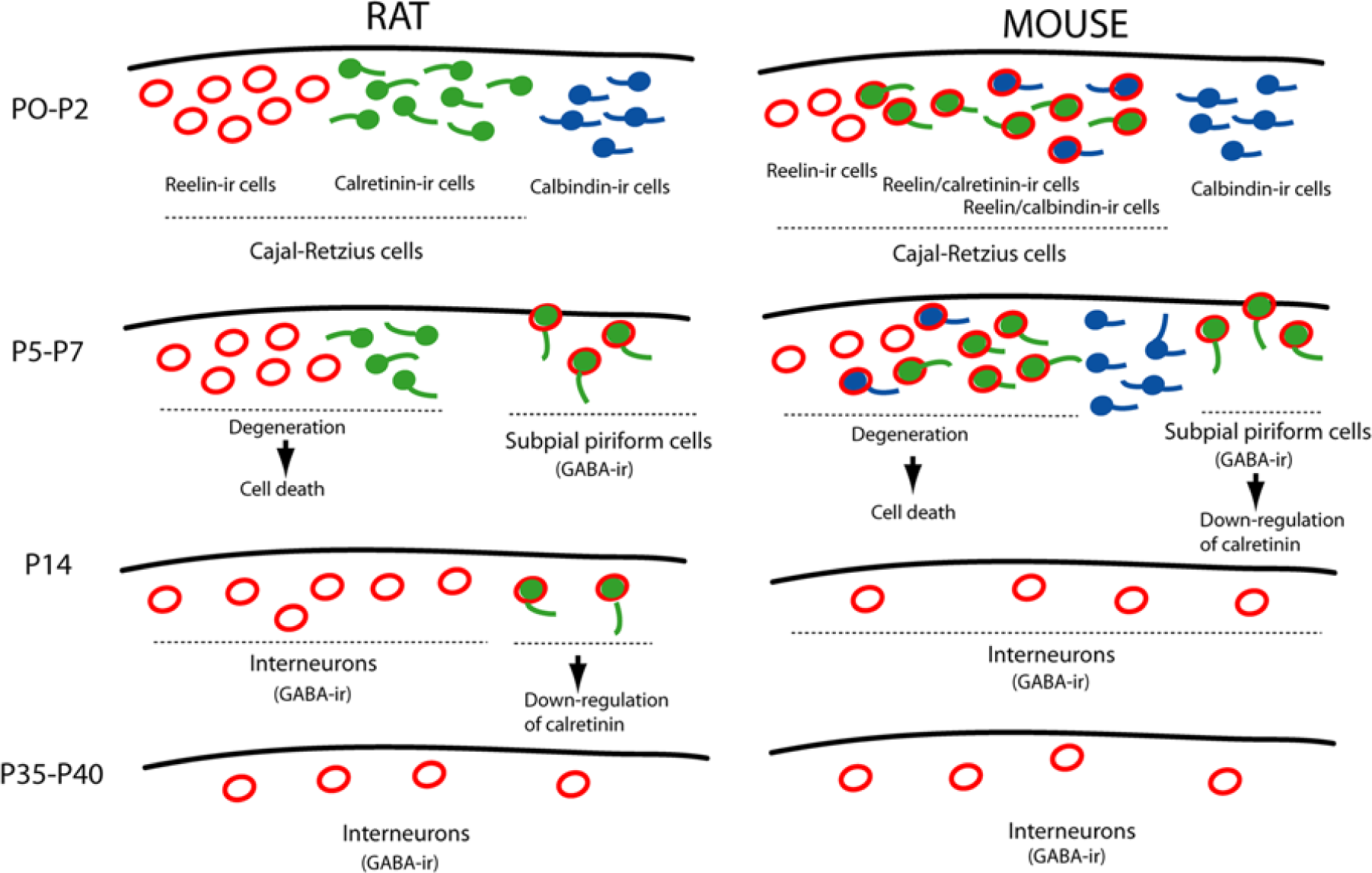
Proposed scheme of the layer 1 cells in postnatal mouse and rat neocortex. During the early postnatal period in the rat, two populations of Cajal-Retzius cells, reelin-ir and calretinin-ir, are found. At the end week 1, the colocalization of reelin and calretinin occurs in GABAergic subpial piriform cells. While Cajal-Retzius cells undergo cell death, subpial piriform cells lack calretinin expression and persist in adulthood along with tangential migrated GABA interneurons. In the early mouse neocortex, reelin colocalize with calretinin and/or calbindin in Cajal-Retzius cells. Whereas these cells degenerate and die, subpial piriform cells, as seen in rats, persist in adulthood along with interneurons. Although the calbindin-ir mouse neurons of layer 1 remain more days than in the rat brain, they probably down-regulate the calbindin expression, die or descend to deeper layers.
It is well-accepted that the calcium-binding protein calretinin is a specific marker of Cajal-Retzius cells in human (Verney and Derer 1995; Zecevic et al. 1999), monkey (Zecevic and Rakic 2001), mouse (Ogawa et al. 1995; Del Río et al. 1995; Alcántara et al. 1998) and rat brains (Weisenhorn et al. 1994). However, and previous to the present work, we had demonstrated that, in postnatal week 1, the picture of cell-specific marker calretinin was more complex in the layer 1 postnatal rat brain (Meyer et al. 1998). The results presented herein reinforce this idea: no double calretinin/reelin labeling was found at P0-P2. Moreover, the population of calretinin-ir cells, negative to reelin, was very large at this age and represented 54.7% of all of the labeled cells. Besides expressing different cell-specific markers, the morphological and physiological profiles allowed us to consider both populations to be Cajal-Retzius cells (Meyer et al. 1998; Schwartz et al. 1998). Later, from P5 to P14, the proportion of double-labeled cells increased. Previous work shows that the number of neurons, named subpial piriform cells, increase during this period (for a review, see Meyer et al. 1999). These interneurons express reelin, calretinin and GABA (Meyer et al. 1998). After Cajal-Retzius cells have gradually disappeared from layer 1, the subpial piriform interneurons, which lack calretinin, but maintain the reelin expression, persist during adult life, where they also contain GABA (Imamoto 1994; Pesold et al. 1998).
These results differ from those found in the mouse in postnatal week 1, where more than 50% of cells are reelin/calretinin-positive. However, it is also relevant that reelin-ir cells, which do not express calretinin, represent 38% of cells at P0-P2 and 40% of those at P5. This is also a large population of non-calretinin-positive Cajal-Retzius cells in the postnatal mouse neocortex. Later, the maximum colocalization percentage reached at P7 likely coincides with the increase in the GABAergic subpial piriform cells. This agrees with the transgenic mice experiments that demonstrated the progressive lack of reelin-ir Cajal-Retzius cells at this age and the increase in the GABA(+)/reelin(+) non-Cajal-Retzius cells (Soda et al. 2003; Chowdhury et al. 2010). Finally, at P35-P30, the cell composition of layer 1 is the same in both species.
In the present work, we also found differences in the layer 1 between rat and mouse in terms of the expression and distribution of calcium-binding protein calbindin. Whereas calbindin is not expressed in reelin-ir cells at any age in rats, 47% of the layer 1 mouse cells colocalize calbindin and reelin at P0-P2 and 17.8% at P5. As far as we are aware, this is the first time that the colocalization of calbindin with reelin has been described in the layer 1 mouse brain. Although this finding is somewhat unusual in other species, a modest number of reelin-ir cells, immunoreactive to calbindin, exist in the marginal zone of the embryonic macaque monkey (Zecevic and Rakic 2001). It is also noteworthy that we observed a transient population of reelin(-)/calbindin(+) cells in postnatal week 1, whose exact fate we cannot specify, and we cannot rule out classifying some of them as Cajal-Retzius neurons. Among the different fates for these cells, we can hypothesize among cell death, descending to deeper layers, or, simply, the down-regulation of calbindin.
The present work comparatively analyses for the first time the neurochemical differences in postnatal Cajal-Retzius cells in rat and mouse. For this study, no stereological methods were used. Although stereological methods, namely the dissector (Sterio 1984), would have allowed us to determine the total number of labeled cells at different ages, our interest was only focused on estimating the percentage of reelin-ir cells labeled for calretinin or calbindin. Whereas in some ages of the rat brain development there is a total lack of colocalization, it is possible that, in other developmental stages, the significant differences in proportions might be due, at least in part, to differences between both species in the morphology of the layer 1 cells. The level of statistical significance between both species during postnatal week 1 was less than 0.001.These differences suggest that the physiological properties of Cajal-Retzius cells cannot be exactly the same in both species. For instance, differences in spontaneous activity between rat and mouse have been described in layer 1 cells. Whereas Cajal-Retzius and non-Cajal-Retzius cells form part of the same correlated networks during mouse neocortex development (Aguiló et al. 1999), only non-Cajal-Retzius cells participate in the correlated networks during rat neocortical development (Schwartz et al. 1998). Differences in cell death mechanisms can also occur between rat and mouse Cajal-Retzius cells. NMDA receptors are important for regulating Cajal-Retzius cell death in postnatal weeks 2 and 3 in the rat (Mienville and Pesold 1999). Furthermore, the results obtained with organotypic mouse slices suggest that the blockade of action potentials and AMPA/kainate receptors, but not NMDA receptors, affect the survival of Cajal-Retzius cells (Del Río et al. 1996).
Cajal-Retzius cells, and their secreted gene product, reelin, are involved in neuronal migration, radial glia control, the formation of developmental synaptic networks and synaptic circuits, among other functions (for a review, see Soriano et al. 2005). Our results demonstrate that, in postnatal week 1, the colocalization of reelin with calcium-binding proteins calretinin and calbindin depends not only on the development stage, but also on the rodent species in which the experiments have been performed. The layer 1 cell population in the postnatal mouse is more uniform than that in the rat brain. In our opinion, experimental mouse models therefore present more advantages for studying the developmental events related to alterations in Cajal-Retzius cells, or abnormal reelin expression, which produce important young and adult neuropsychiatric disorders.
Footnotes
Acknowledgements
We thank Jose J. Cabanes and Jose R. Marín-Tébar for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Contract grant sponsor: Consejería de Educación JCCM; Contract grant number PII1I09-0056-7896 (to J.R.M.G.).
