Abstract
Oncomodulin (OM) is a small, acidic calcium-binding protein first discovered in a rat hepatoma and later found in placental cytotrophoblasts, the pre-implantation embryo, and in a wide variety of neoplastic tissues. OM was considered to be exclusively an oncofetal protein until its recent detection in extracts of the adult guinea pig's organ of Corti. Here we report that light and electron microscopic immunostaining of gerbil, rat, and mouse inner ears with a monoclonal antibody against recombinant rat OM localizes the protein exclusively in cochlear outer hair cells (OHCs). At the ultrastructural level, high gold labeling density was seen overlying the nucleus, cytoplasm, and the cuticular plate of gerbil OHCs. Few, if any, gold particles were present over intracellular organelles and the stereocilia. Staining of a wide range of similarly processed gerbil organs failed to detect immunoreactive OM in any other adult tissues. The mammalian genome encodes one α- and one β-isoform of parvalbumin (PV). The widely distributed α PV exhibits a very high affinity for Ca2+ and is believed to serve as a Ca2+ buffer. By contrast, OM, the mammalian β PV, displays a highly attenuated affinity for Ca2+, consistent with a Ca2+-dependent regulatory function. The exclusive association of OM with cochlear OHCs in mature tissues is likely to have functional relevance. Teleological considerations favor its involvement in regulating some aspect of OHC electromotility. Although the fast electromotile response of OHCs does not require Ca2+, its gain and magnitude are modulated by efferent innervation. Therefore, OM may be involved in mediation of intracellular responses to cholinergic stimulation, which are known to be Ca2+ regulated.
The PV family includes two sublineages, α and β (Goodman and Pechère 1977). The α PVs are less acidic (pI ≥ 5) than the β-isoforms and have an additional amino acid residue in the C-terminal helix. At present, the mammalian genome is known to encode only one α- and one β-isoform of PV (Föhr et al. 1993). Although it is most abundant in fast-twitch skeletal myofibrils (Heizmann et al. 1982) and GABAergic neurons (Celio and Heizmann 1981), the α PV is also expressed in a number of nonexcitatory tissues (reviewed in Heizmann 1988), including select cell types in the inner ear. It is believed to serve as a cytosolic Ca2+ buffer, facilitating myofibrillar relaxation and neuronal repolarization and preventing Ca2+ toxicity.
The mammalian PV β-isoform was discovered in a rat hepatoma (MacManus 1979) and was subsequently detected in the blastocyst and placental cytotrophoblasts (Brewer and MacManus 1985,1987). It has now been shown to occur in a wide variety of rodent and human tumors (MacManus et al. 1982; MacManus and Whitfield 1983; Brewer et al. 1984). The name oncomodulin (OM) was applied to the protein to reflect its common appearance in neoplasms and its putative Ca2+-dependent regulatory capacity. OM was previously believed to be an oncofetal protein, absent from any normal adult mammalian tissue. Recently, however, N-terminal sequence data for an acidic Ca2+ binding protein (CBP-15) expressed in the guinea pig organ of Corti (Senarita et al. 1995) was found to exhibit a very high degree of homology with the β PVs (Thalmann et al. 1995). Subsequent investigation has confirmed that CBP-15 is identical to OM (Henzl et al. 1997).
Previous work had demonstrated immunoreactivity for PV in inner hair cells (IHCs) of guinea pig and gerbil cochlea and in vestibular hair cells of guinea pig, rat, and mouse (Eybalin and Ripoll 1990; Deměmes et al. 1993; Pack and Slepecky 1995). Implicit in these earlier studies was the assumption that the antibodies employed recognized the α-isoform of PV, because this was the only isoform believed to be expressed in adult mammals. With the discovery of the β PV in the mature guinea pig organ of Corti, it became necessary to re-examine this issue. The availability of immunological probes against OM has provided an opportunity to unequivocally ascertain the distribution of the PV α- and β-isoforms in the inner ear. Using monoclonal and polyclonal probes raised against OM, we report here that the β PV is expressed exclusively in outer hair cells of the gerbil, rat, and mouse cochlea.
Materials and Methods
Tissue Collection and Processing
Inner ears were obtained from twenty Mongolian gerbils (
All animals were anesthetized with urethane (1.5 g/kg
Specimens processed for LM observation were dissected from the temporal bone and decalcified in either 0.12 M EDTA (pH 7.2) for 48 hr or 8 N formic acid overnight with gentle stirring. The ears were flushed with 0.1 M PBS, pH 7.2, dehydrated in a graded series of ethanols, cleared in Histoclear (National Diagnostics; Manville, NJ), and embedded in Paraplast Plus (Curtin Matheson; Marietta, GA).
A wide range of systemic organs from two adult gerbils were fixed in Carnoy's solution and embedded in composite paraffin blocks to survey at the LM level for OM in other tissues. The specimens included brain, alimentary tract, skeletal muscle, testis, ovary, thyroid, eye, thymus, lymph node, kidney, heart, pancreas, skin, fat, tongue, adrenal, liver, and spleen. Sections from the composite blocks were stained in each protocol with the immunoperoxidase procedure described below.
For electron microscopic (EM) immunostaining, inner ears were fixed identically to those described above for LM studies except that the fixative consisted of a mixture of 0.5% glutaraldehyde and 4% paraformaldehyde in 0.1 M PB, pH 7.2. After a 1-hr exposure to fixative the specimens were decalcified with EDTA as described above, sliced into half turns, dehydrated through a graded series of 50, 70, 90, and 95% ethanols, and infiltrated with Lowicryl K4M at −20C. The half turns were then oriented in Beem capsules containing fresh K4M resin and were polymerized under 360-nm wavelength UV for 24 hr at −20C and 48 hr at room temperature (RT) (Sugiyama et al. 1992; Nakazawa et al. 1995).
Preparation of Antibodies
A rabbit polyclonal antiserum and a mouse monoclonal antibody (MAb) were prepared against recombinant rat OM coupled to keyhole limpet hemocyanin, as described elsewhere (Hapak et al. 1989; Serda and Henzl 1991). For antiserum production, the antigen was emulsified with RIBI adjuvant (RIBI Immunochemicals; Hamilton, MT), to afford a final OM concentration of 0.25 mg/ml, and was injected at multiple sites, 0.05 ml per site, on the dorsal surface of a male NZW rabbit (Hurn and Chantler 1980). This procedure was repeated 5 weeks later. Thereafter, injections of recombinant OM alone were made as necessary to boost antiserum titer. Serum was collected by ear bleeding.
For the preparation of MAbs, female BALB/c mice received two 0.2-ml injections (
The polyethylene glycol-induced fusion of splenocytes with murine myeloma cells was performed using a modification of standard protocols (e.g., Hurn and Chantler 1980; Galfré and Milstein 1981; Goding 1985). Splenocytes from a single mouse were released from the splenic capsule with the aid of a tissue sieve, collected by centrifugation (1000 rpm, 5 min), resuspended in 5 ml of medium (RPMI-Hepes, 10% FBS), and counted. Splenocytes (2 × 108) were combined with actively dividing PAI-0 myeloma cells at a splenocyte: myeloma ratio of 10:1, collected by centrifugation, and incubated at 37C.
The PEG fusogen was prepared just before use by melting 30 g of PEG 1500 (Aldrich Chemicals; Milwaukee, WI) at 50–60C, mixing with 30 ml of warm RPMI-Hepes, adjusting the pH to 7.2 with NaOH, and filter-sterilizing. Two ml of this solution was added dropwise over the course of 1 min to the splenocyte-myeloma pellet. After an additional min at 37C, the PEG was gradually diluted with RPMI-Hepes, first with 2.0 ml added at a rate of 1.0 ml per min, then with an additional 8 ml added over the course of 1 min. After 2 more min at 37C, the cells were collected by centrifugation (1000 rpm, 5 min).
The cells were resuspended at 1.5 × 106 splenocytes/ml and dispensed into 96-well plates. The plating medium contained RPMI 1640 supplemented with NaHCO3, 10% heat-inactivated FBS, 2 mM
Hybridomas were assayed by ELISA and Western blot. The 1A10 antibody used for these studies belongs to the IgG1 subclass and harbors κ light chains, as determined with the Isotyper kit from Boehringer-Mannheim (Indianapolis, IN). Although its epitope has not been mapped, the antibody does not crossreact with rat muscle α PV or with either of the two avian thymic PVs, ATH and CPV3.
Immunoperoxidase Staining
Serial midmodiolar 5-μm-thick sections were mounted on chrome alum-subbed slides. Every twenty-fifth section was stained with hematoxylin and eosin. Selected sections were processed for immunostaining as described previously (Schulte and Adams 1989), using either the rabbit antiserum or the mouse MAb. Deparaffinized and rehydrated sections were immersed for 10 min in 3% H2O2 to block endogenous peroxidase. After washing with PBS, the sections were incubated for 20 min with PBS containing either 1% normal goat serum (NGS) for the antiserum or 1% normal horse serum for MAb and reacted overnight at 4C with antiserum diluted 1:200–500 or MAb diluted 1:5–10. The sections were rinsed with PBS and incubated for 30 min with either biotinylated goat anti-rabbit IgG (antiserum) or biotinylated horse anti-mouse lgG (MAb) (Vector Laboratories; Burlin-game, CA) at a dilution of 1:200 with 19% normal serum in PBS. The sections were incubated for 30 min with the avidin-biotin-horseradish peroxidase complex (Vectastain ABC kit), rinsed again with PBS, and reacted with monitoring for 1–3 min in substrate medium containing 3,3′ diaminobenzidine HC1 and H2O2 (Sigma Chemical, St Louis, MO) before dehydration and mounting.
Control sections were processed in parallel, substituting nonimmune rabbit serum (NRS) at a dilution of 1:200 in PBS for primary antibody. As a further control, sections were exposed either to polyclonal antiserum or to MAb that had been preincubated for 24 hr at 4C with 200 μg/ml recombinant OM.
To further investigate the inconsistent immunostaining of OHC nuclei in paraffin sections, inner ears from two adult gerbils were fixed as described above with a solution of 4% paraformaldehyde and 2% glutaraldehyde, decalcified in EDTA, sliced into half turns, and embedded in Epon. One-μm-thick radial sections from these specimens were etched with a 1:1 solution of alcoholic NaOH: absolute ethanol for 15 min, followed by four rinses for 5 min each in absolute ethanol (Baskin et al. 1979; Schulte et al. 1980). The etched sections were then immunostained using procedures identical to those described above for paraffin sections and were examined by LM.
Immunoelectron Microscopy
Ultrathin sections of cochlear half turns were cut and picked up on formvar- and carbon-coated nickel grids (Ted Pella; Redding, CA). The grids were incubated for 1 hr with 5% NGS in PBS and then reacted overnight at 4C with either the polyclonal anti-OM at a dilution of 1:800 or the MAb at a dilution of 1:10 with NGS in PBS. Control sections were processed in parallel but substituting primary antibody with a similar dilution of NRS. The grids were rinsed in PBS and then incubated for 2 hr at RT with a 1:20 dilution of goat anti-rabbit IgG adsorbed to the surface of 15-nm colloidal gold particles (Bio Cell Research Laboratories; Cardiff, UK) for the polyclonal antiserum or goat anti-mouse IgG absorbed to the surface of 10-nm gold particles (Sigma) for the MAb. After washes with PBS and distilled water, grids were stained with uranyl acetate for 15 min and lead citrate for 3 min and were examined in a JEOL-100S electron microscope.
Results
LM Immunohistochemistry
Fixation with Carnoy's solution provided the best retention of OM antigenicity with MAb 1A10 and the polyclonal antiserum. Inner ears fixed with the zinc-formalin solution showed weaker but still strong immunoreactivity with both probes. No differences in immunostaining were noted between the two decalcifying procedures.
In paraffin sections of gerbil inner ear, immunostaining for OM with MAb 1A10 was confined to OHCs in the organ of Corti (Figures 1 and 2). Reaction product was distributed diffusely throughout the OHC's somata, but stereocilia were unreactive (Figure 2). The immunostaining reaction was of similar intensity in all rows of OHCs throughout all turns of the cochlea.

Intense immunoreactivity for OM is confined exclusively to OHCs, as shown here in the upper basal turn of the gerbil cochlea. MAb 1A10.
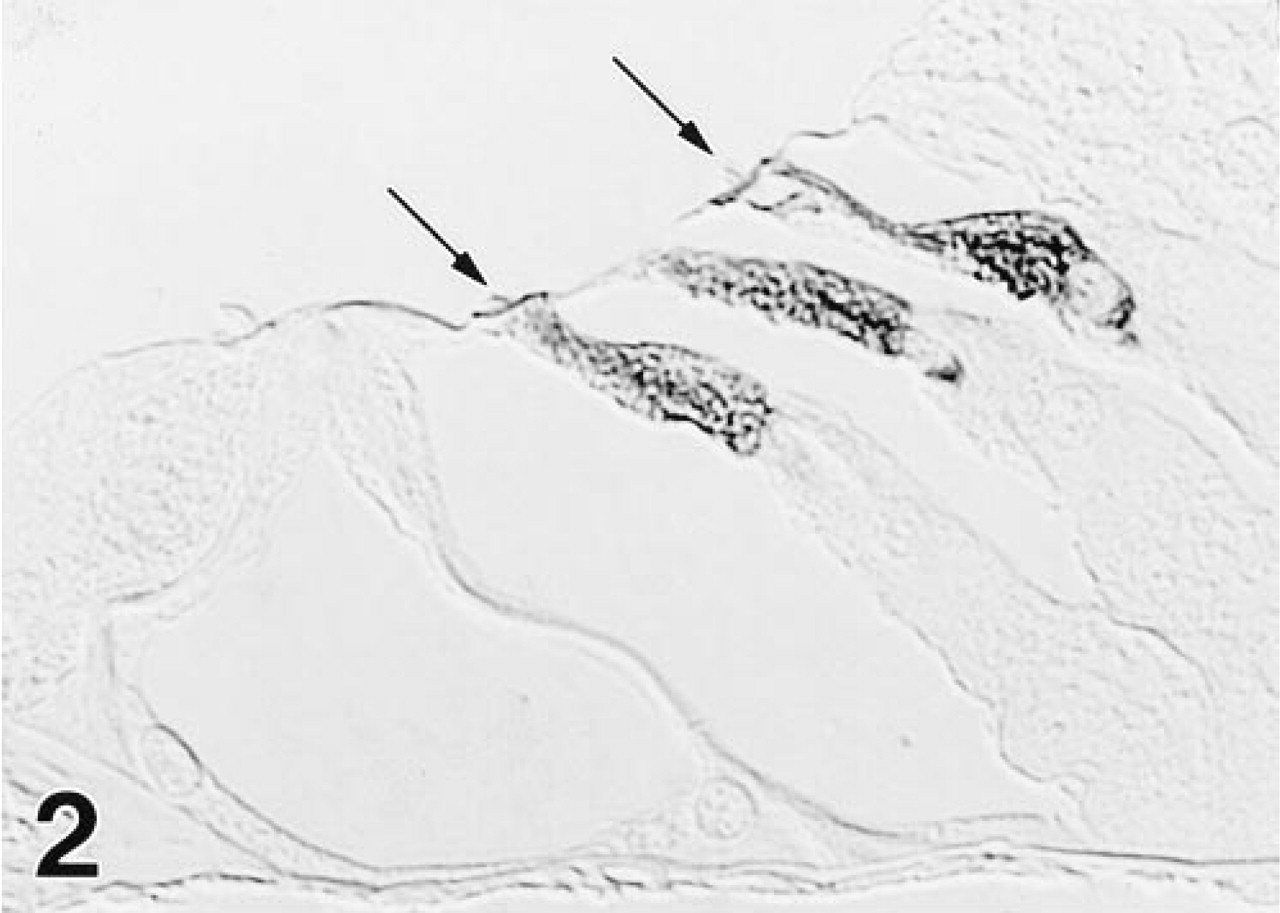
Reaction product is distributed diffusely throughout the cell body of OHCs. Stereocilia (arrows) are unstained, as are OHC nuclei in this section. MAb 1A10.
As with the MAb, the polyclonal antiserum imparted strong staining to all gerbil OHCs, but differed in reacting also with cochlear IHCs (Figure 3) and some vestibular hair cells (Figure 5). The IHCs showed a longitudinal staining gradient, with those in the apex staining intensely and those in the base only weakly. The immunolabeling pattern in the mouse and rat inner ear (Figure 6) was identical to that observed in the gerbil with both immunological probes.
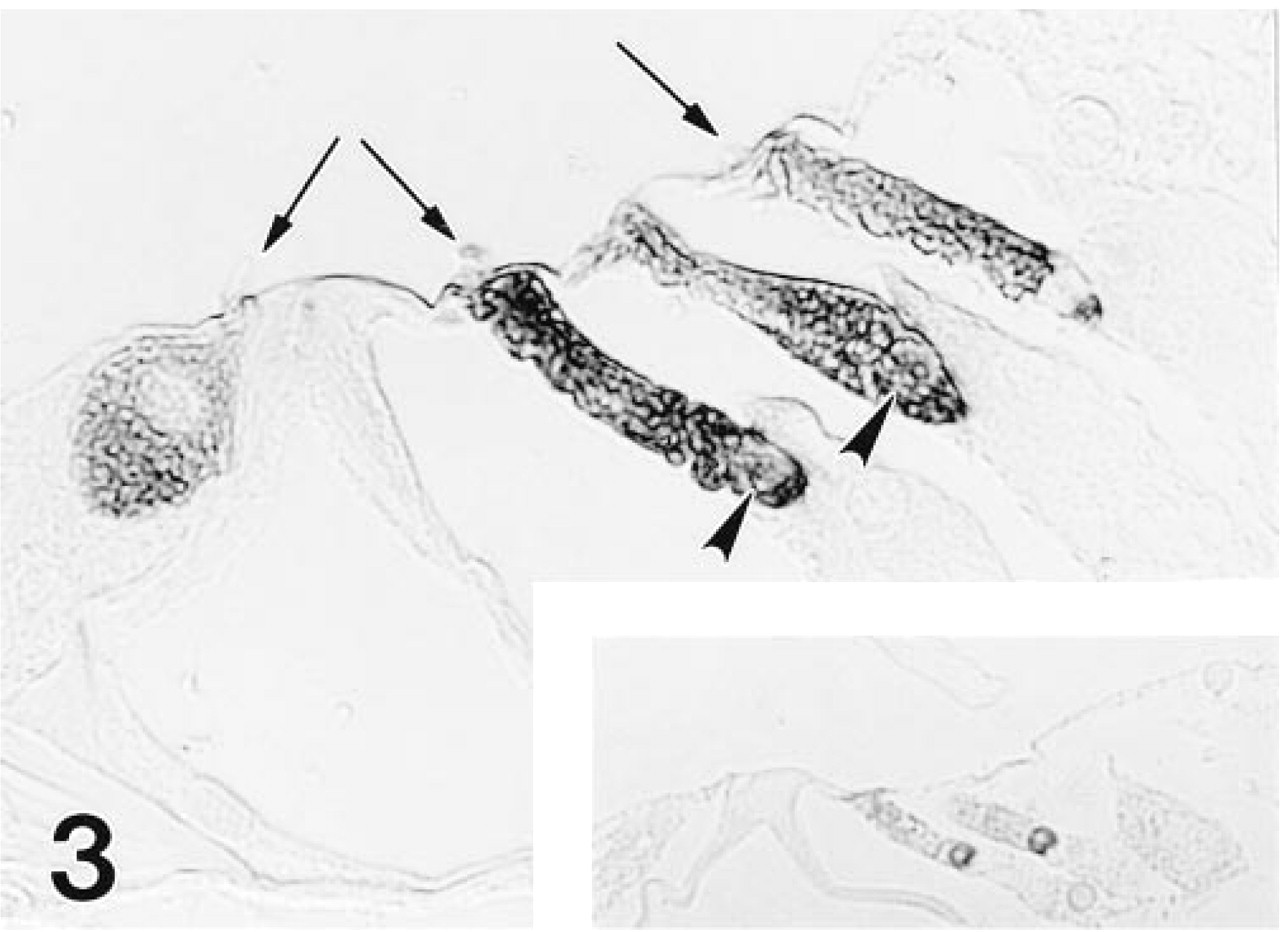
The polyclonal antiserum to OM also stains OHCs, but in addition reacts with IHCs, as shown here in the middle turn of the gerbil cochlea. HC stereocilia (arrows) are unstained and two of three OHC nuclei (arrowheads) are immunoreactive in this section. Nuclei of OHCs in pre-etched epoxy sections consistently showed strong immunoreactivity for OM (inset).
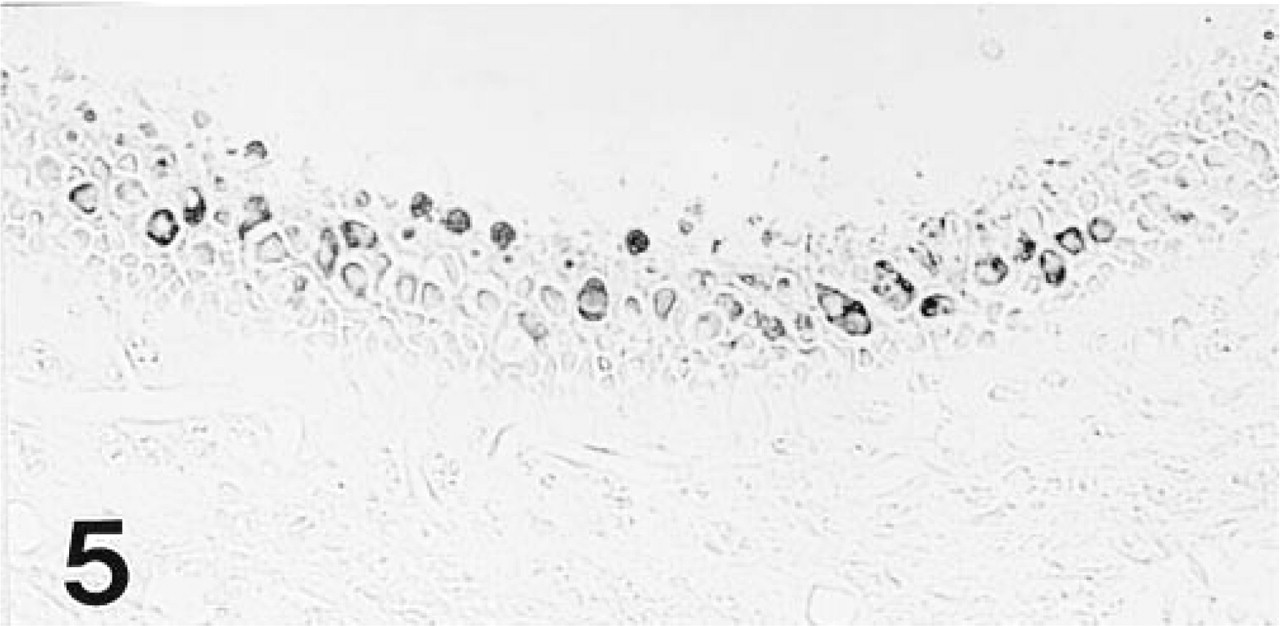
Hair cells (presumably Type I) in the central region of the gerbil's saccular macula show moderate reactivity with the polyclonal antiserum to OM.
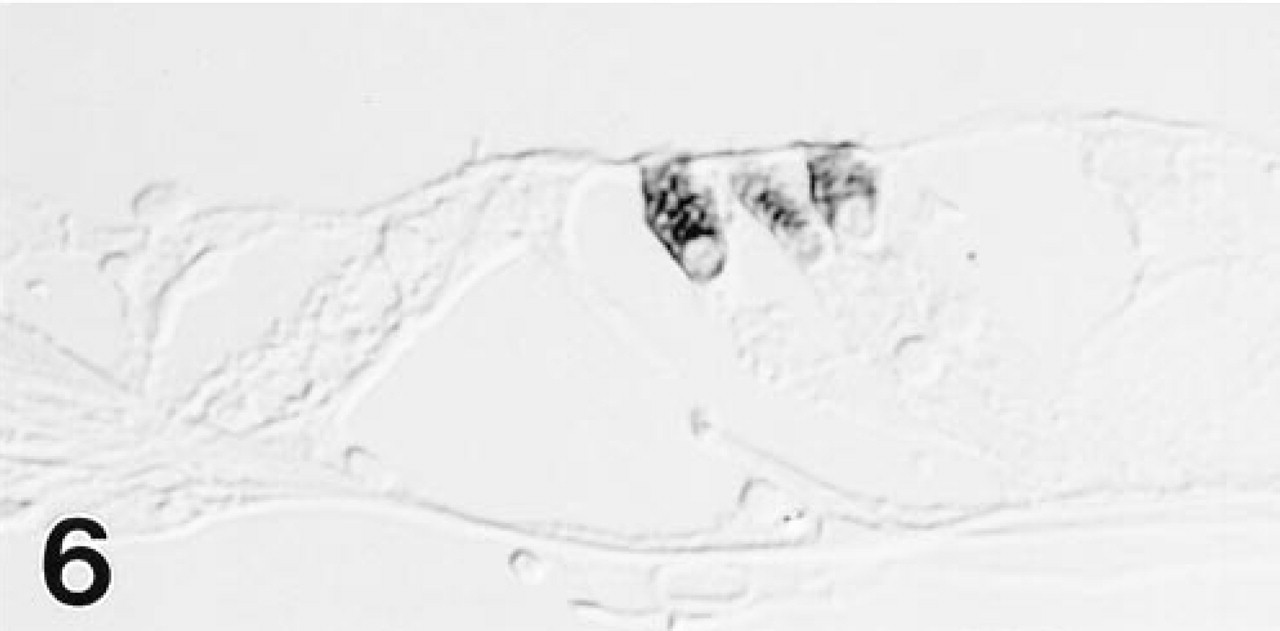
As in the gerbil, only OHCs in the rat organ of Corti are labeled with MAb to OM.
In paraffin sections, the nuclei of OHCs showed inconsistent staining with both the polyclonal antiserum and MAb 1A10 (Figures 2 and 3), most probably owing to failure of staining reagents to penetrate uncleaved nuclei. In contrast, in etched 1-μm-thick epoxy sections, OHC nuclei were consistently strongly reactive with both the polyclonal antiserum (Figure 3, inset) and the MAb (not shown).
No staining was seen on sections in which nonimmune rabbit serum was substituted at a similar dilution for primary antiserum. Preabsorption of a 1:5 dilution of MAb with 200 μg/ml of recombinant OM eliminated all specific staining in OHCs (Figure 4). Preincubation of a 1:200 dilution of the polyclonal antiserum with 200 μg/ml of recombinant OM also blocked staining in all sites, including IHCs and vestibular hair cells (not shown). Examination of composite blocks containing a wide range of gerbil organs revealed no immunopositive sites in any of the tissues studied.

Preabsorption of MAb 1A10 with 200 μg/ml of recombinant OM eliminated all specific staining of OHCs in the basal turn of the gerbil cochlea.
Immunoelectron Microscopy
At the EM level, immunogold labeling with MAb 1A10 again was confined exclusively to OHCs in the gerbil's organ of Corti. Colloidal gold particles were distributed diffusely over the OHC's cytoplasm and nucleus (Figure 7). Mitochondria failed to stain (Figures 7–10). The gold labeling in OHCs extended uniformly over the cuticular plate (Figures 10 and 11), but stereocilia were unlabeled (Figure 11). Gold particles also were present in the region of the lateral subsurface cisternae and the postsynaptic cisternae, but poor preservation of these structures made it impossible to ascertain if immunoreactive OM was present inside or occupied cytoplasm adjacent to the cisternae.
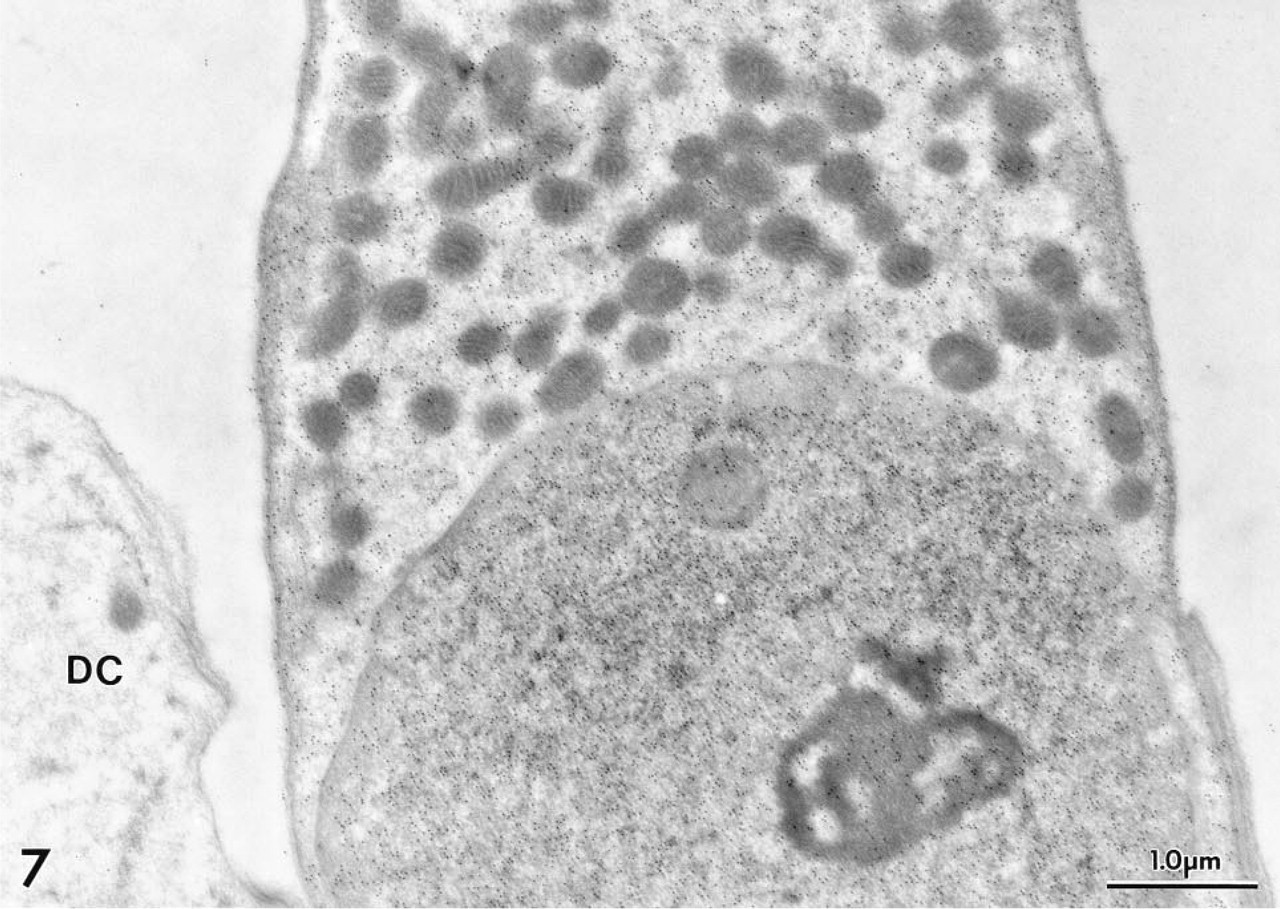
Gold particles denoting sites of MAb 1A10 binding to OM are distributed diffusely throughout the cytoplasm and nucleoplasm of a gerbil OHC. A few particles were seen overlying mitochondria. A portion of a Deiters cell (DC) is unreactive.
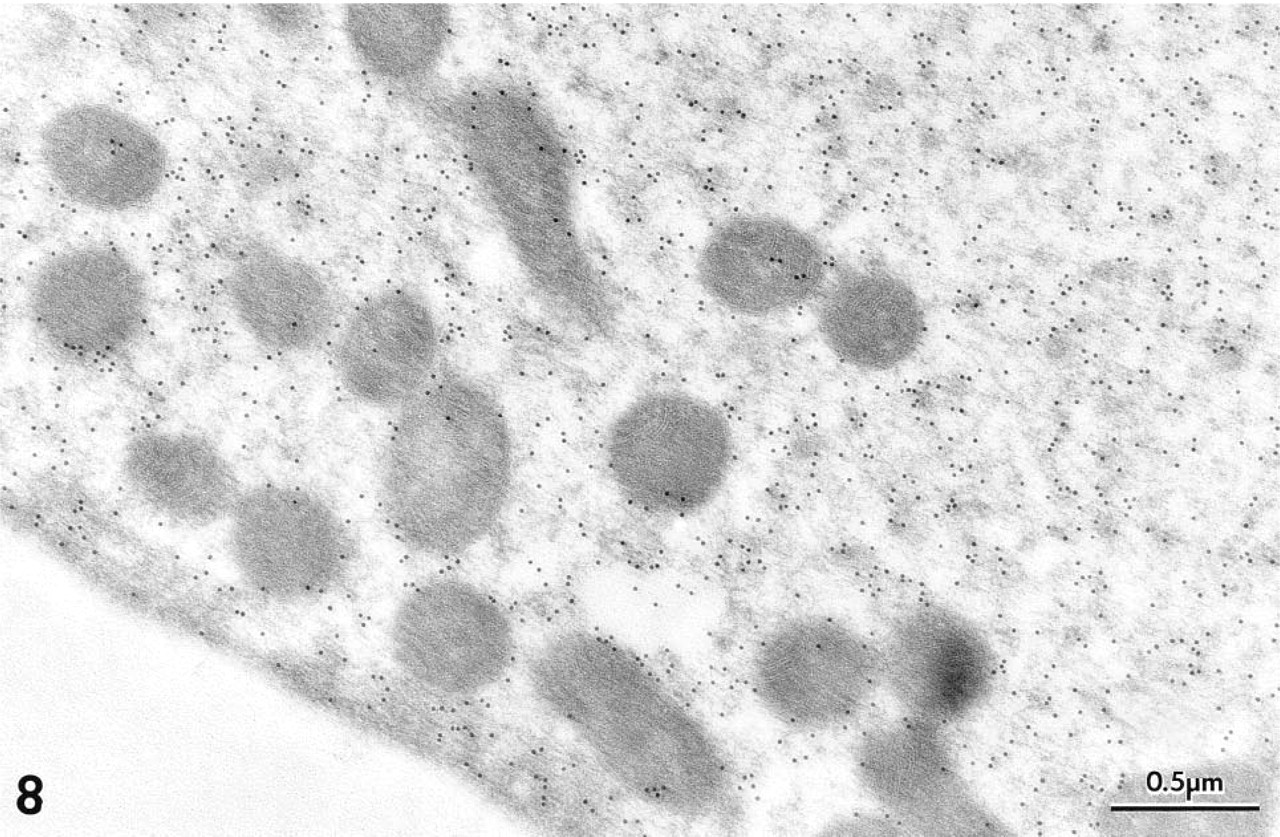
Diffuse labeling over the cytosol and lack of gold particles overlying mitochondria in a gerbil OHC is shown more clearly at higher magnification. Polyclonal antiserum.
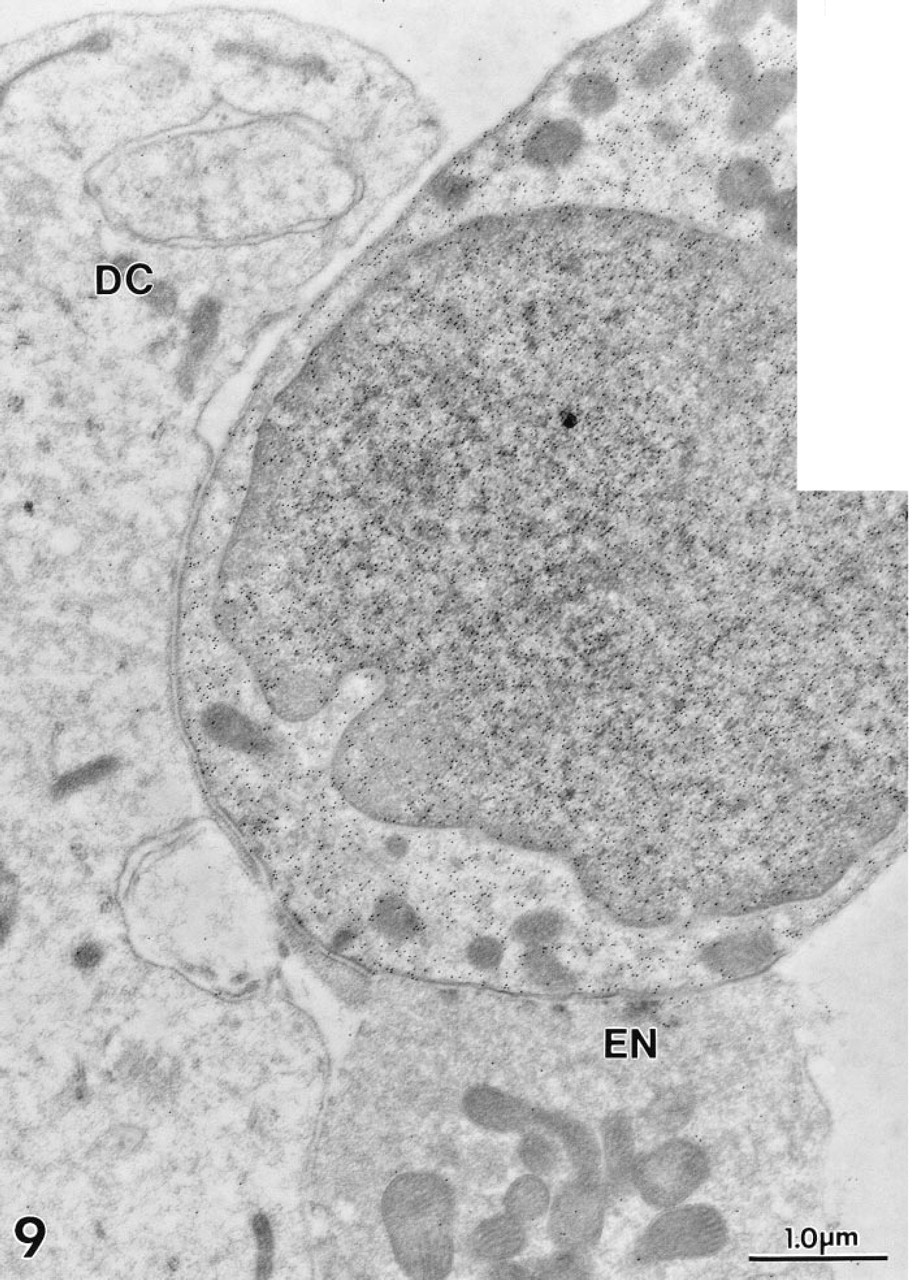
Cytosolic and nucleosolic gold labeling is clearly visible in this gerbil OHC. An adjacent efferent nerve terminal (EN) and Deiter's cell (DC) lack decoration. Polyclonal antiserum.
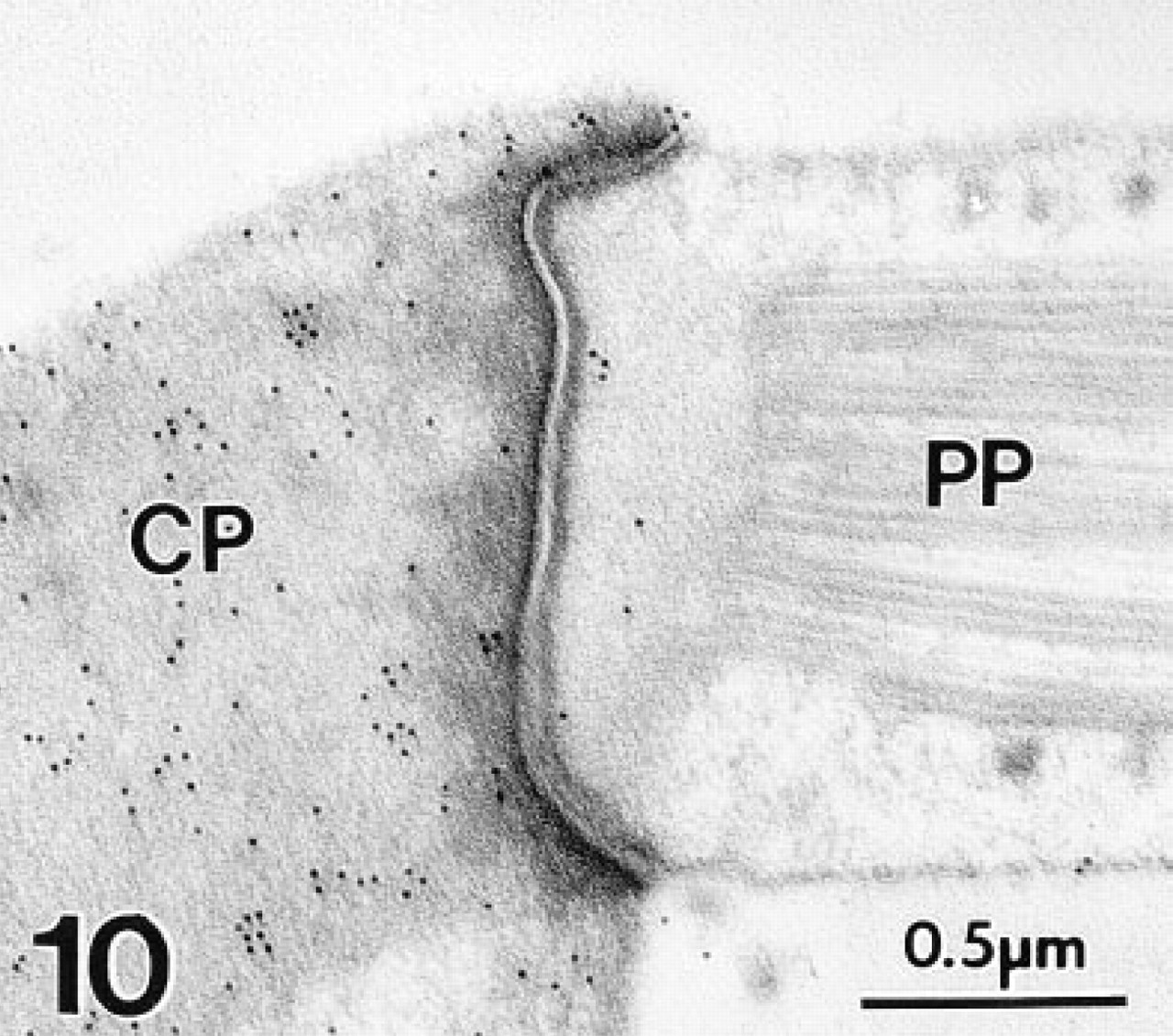
The cuticular plate (CP) of a gerbil OHC shows moderate labeling density with colloidal gold, whereas the tight junctionally-connected phalyngeal process (PP) of a Deiter's cell is unlabeled. Polyclonal antiserum.
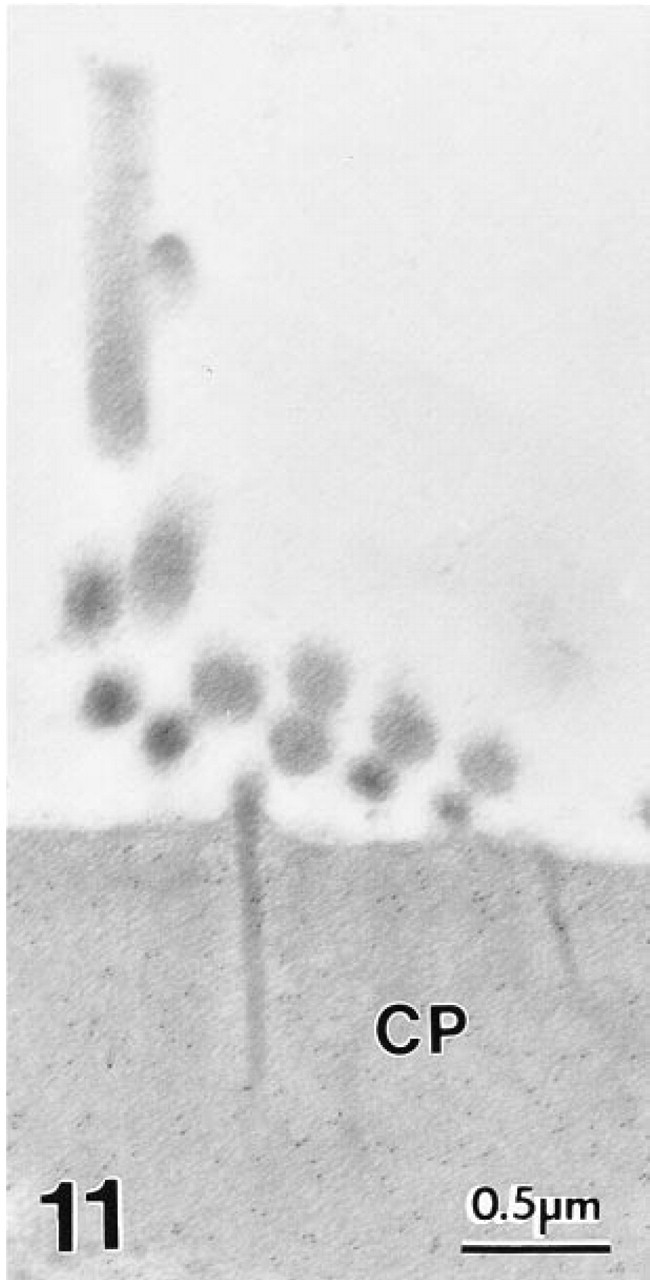
Stereocilia of a gerbil OHC are unlabeled with MAb to OM, although the cuticular plate (CP) shows moderate labeling.
With MAb 1A10, no positive staining was seen in any other site in the inner ear, including Deiter's cells and nerve endings (Figures 7 and 9) and IHCs (Figure 12). In agreement with the LM results, the polyclonal antiserum to OM showed weaker but consistent staining in the cytoplasm of cochlear IHCs (Figure 13). No staining was present on sections in which nonimmune rabbit serum was substituted for primary antibody.
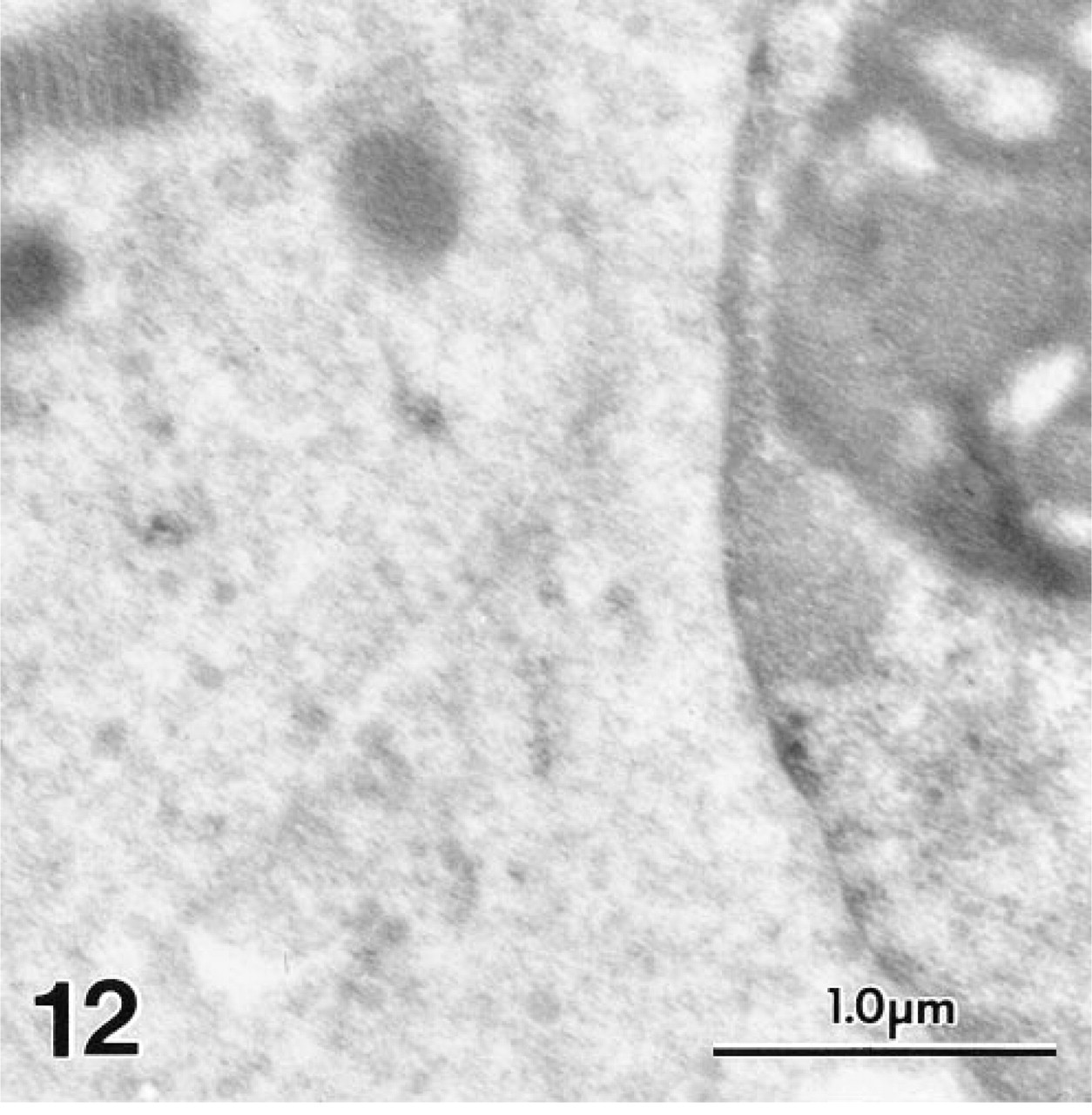
Gerbil IHCs failed to react with MAb to OM.
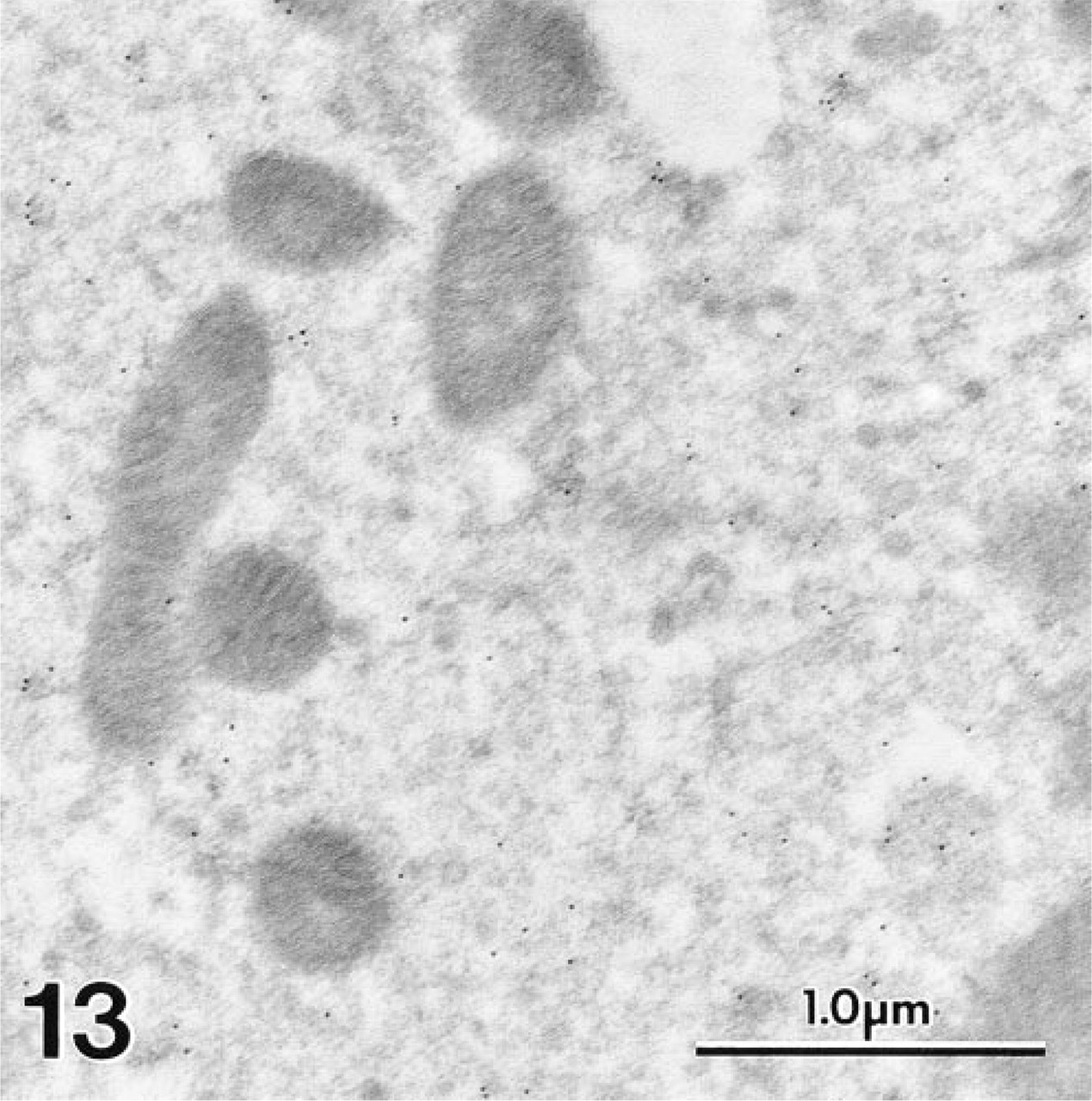
In contrast to Figure 12, the polyclonal antiserum to OM produced diffuse labeling over the cytosol of a gerbil IHC.
Discussion
In this study we examined the distribution of the mammalian β PV (OM) in three different species, using both monoclonal and polyclonal immunological probes. Our results provide an interesting counterpoint to previous work on PV in the inner ear (Eybalin and Ripoll 1990; Deměmes et al. 1993; Pack and Slepecky 1995). It is immediately apparent that the MAb employed by Pack and Slepecky (1995) did not recognize the PV β-isoform. Those authors reported that immunoreactivity for PV in the organ of Corti was confined to the IHCs. We obtained similar IHC immunoreactivity with the polyclonal antiserum to OM, including the previously noted longitudinal gradient in staining intensity from strong at the apex to weak at the base. Importantly, however, we also observed intense staining of OHCs with both the polyclonal antiserum and the MAb to OM. Because MAb 1A10 to OM recognizes the OHCs exclusively, we conclude that OM (β PV) expression in the inner ear is confined to the OHCs. We further conclude that the signal arising from the IHCs reflects crossreactivity of the polyclonal antiserum with the α-isoform of PV. Consistent with this hypothesis, the polyclonal anti-OM preparation likewise stains a subpopulation of vestibular hair cells, as previously reported (Eybalin and Ripoll 1990; Deměmes et al. 1993). These observations imply the existence of one or more epitopes common to OM and the α PV. Moreover, the OM epitope recognized by MAb 1A10 apparently is not shared by the α-isoform.
EF-hand Ca2+ binding sites can be divided into two categories on the basis of their affinities for Ca2+ and Mg2+ (Seamon and Kretsinger 1983). High-affinity, or Ca2+/Mg2+, sites exhibit very high affinity for Ca2+ (Kd < 10-7 M) and substantial affinity for Mg2+ (Kd < 10-4 M). These sites are continuously occupied under physiological conditions and, although inappropriate for Ca2+-dependent regulatory functions, are well suited to structural or Ca2+-buffering roles. By contrast, low-affinity (Ca2+-specific) sites exhibit moderate affinity for Ca2+ (Kd × 10-6 M) and negligible affinity for Mg2+ at physiological pH and ionic strength. These sites, unoccupied at resting-state intracellular Ca2+ concentrations, bind Ca2+ after an increase in cytosolic Ca2+ levels. Because the conformational change that accompanies Ca2+ binding can expose effector binding sites, Ca2+-specific sites are ideally suited for performing Ca2+-dependent regulatory roles.
The PV metal ion binding domains, known as the CD and EF sites, usually belong to the Ca2+/Mg2+ category (e.g., Haiech et al. 1979; Moeschler et al. 1980; Rinaldi et al. 1982; Serda and Henzl 1991; Eberhard and Erne 1994). OM, however, is an interesting exception to this rule. Despite 54% sequence identity with the rat α PV (Berchtold et al. 1982; MacManus et al. 1983), rat OM displays highly attenuated affinity for metal ions. In fact, with a Kd for Ca2+ of 0.8 μM and a Kd for Mg2+ in excess of 1 mM (Hapak et al. 1989; Cox et al. 1990), the CD site of OM qualifies as a Ca2+-specific site. Although this observation is consistent with a regulatory function, no physiological effector protein has been identified to date.
The literature regarding OM's putative regulatory capacity is both confusing and controversial. The preliminary reports that OM stimulates cyclic nucleotide phosphodiesterase (MacManus 1981a; Mutus et al. 1985) could not be confirmed by other laboratories (Klee and Heppel 1984; Clayshulte et al. 1990). Similarly, the suggestion that OM undergoes facile disulfide mediated dimerization (Mutus et al. 1988) has been refuted (Clayshulte et al. 1990). Reports that OM activates a nuclear protamine kinase (Boynton et al. 1982), that it inhibits glutathione reductase (Palmer et al. 1990), and that it stimulates DNA synthesis (Mac-Manus 1981b; Boynton et al. 1982) likewise remain unconfirmed. Blum and Berchtold (1994) have suggested that OM influences cell cycle progression in neoplasms in a manner similar to calmodulin (CaM). Presumably, however, this capacity would have little relevance to events in the nonmitotic OHCs.
The presence of the AB domain, the nonfunctional vestige of an EF-hand motif, in the PV tertiary structure prevents CaM-like interactions with helical peptide targets (Strynadka and James 1989; McPhalen et al. 1991). Intimately associated with the hydrophobic aspect of the paired CD and EF sites, the AB domain serves, in essence, as a built-in target sequence. Clearly, if OM serves in a regulatory capacity, then OM-effector interactions must differ fundamentally from those involving CaM.
The mammalian auditory system owes its unrivaled performance to the “cochlear amplifier,” a unique specialization wherein the receptor potential of the OHC does not serve to elicit a neural response, but rather, in a process of reverse transduction, is converted into a rapid motile response (Ashmore 1990; Dallos 1992). This phenomenon, manifested in vitro as electrically evoked sound frequency contraction/relaxation cycles (electromotility), leads to a sharply localized amplification of the passive Békèsy wave of the basilar membrane (Brownell 1983; Brownell et al. 1985; Ashmore 1987; Zenner et al. 1987; Dallos 1992, 1996). The vibratory response of the basilar membrane serves as the stimulus for the IHC, the sensory cell proper, and ultimately leads to transmitter release and excitation of the afferent synapses. It is the cooperative interaction between the two hair cell systems that produces the extreme sensitivity, wide dynamic range, and excellent frequency selectivity displayed by mammals (Dallos 1992,1996).
Detailed speculation regarding the role of OM in the OHC is premature. It is significant, however, that the OHC, undoubtedly the most highly differentiated hair cell in the entire acoustic-lateralis system, would be associated with such an unusual highly specialized molecule as OM.
It appears unlikely that OM is involved in the mechano-electrical transduction process or in the attendant adaptation phenomena in which the tension of the tip links of the stereocilia is being adjusted (Hacohen et al. 1989; Hudspeth 1989). The adaptation phenomenon depends on Ca2+ and has thus far been demonstrated only in frog saccular hair cells. However, it is generally assumed that these highly conserved processes occur in both mammalian cochlear hair cell systems and are mediated by abundantly expressed CaM (Walker et al. 1993). The present immunocytochemical data, which fail to localize OM to the stereocilia, are in agreement with these teleological considerations.
An involvement of OM in afferent transmission is also unlikely because of the absence of all but a minimal afferent innervation of the OHC. The small contingent of afferent fibers is unmyelinated and appears to convey proprioceptive-like rather than acoustic information (Dallos 1996).
Consequently, by elimination, we speculate that OM is involved in some way in the most specialized aspect of OHC function, the cochlear amplifier. Because electromotility continues in the absence of Ca2+, direct involvement of OM with the fast motor system can be discounted (Ashmore 1987; Santos-Sacchi 1989). Moreover, it is clear that the unprecedented rapidity of the motor element precludes intervening steps, depending instead on a biophysical process such as voltage-induced conformational changes. However, there is a considerable body of evidence that the massive cholinergic medial efferent system innervating the OHC exerts feedback control over the cochlear amplifier. Sziklai and Dallos (1996) and Sziklai et al. (1997) demonstrated a significant effect of acetylcholine on the gain and magnitude of OHC electromotility. Both the fast and slow intracellular effects of efferent stimulation are believed to be mediated by Ca2+ (Housley and Ashmore 1991; Doi and Ohmori 1993; Sridhar et al. 1995; Sewell 1996; Sziklai et al. 1997). It is conceivable that OM is in some way involved in the mediation of these processes, and thereby helps to adjust gain and amplitude of the cochlear amplifier.
In response to a variety of stimuli, OHCs also display slow motility, contractions and elongations occurring on a time scale of seconds and minutes (Brownell et al. 1985; Zenner et al. 1985; Schacht et al. 1995; Holley 1996). The physiological significance of slow motility is not completely understood. However, contraction of the OHC would bring the reticular lamina and the basilar membrane into closer proximity, thereby exerting an inhibitory effect upon basilar membrane motion (Schacht et al. 1995; Holley 1996). This phenomenon, which has been documented in situ after noise exposure (Harding et al. 1992), could represent the basis for temporary threshold shifts. Although slow motility is Ca2+-dependent, a role for OM again is unlikely because this type of motility is inhibited by classical calmodulin antagonists (Dulon et al. 1990; Schacht et al. 1995; Puschner and Schacht in press).
Existing data regarding the presence of OM in the nucleus are contradictory. Neither MacManus (1981b) nor Brewer et al. (1984) were able to detect OM in nuclear fractions of hepatoma cells by immunoassay. However, Brewer et al. (1984) observed variable nuclear staining in paraffin-embedded hepatoma and transformed NRK cells and reported that the distribution of immunoreactive OM varied with fixation conditions. Whereas acidic formaldehyde or acetone fixation yielded both nuclear and cytoplasmic staining, fixation with nonacidic formaldehyde, ethanol, or methanol yielded primarily cytoplasmic staining. At the ultrastructural level, we consistently observed many gold particles overlying the nucleoplasm in all OHCs. Because many nuclei are uncleaved in 5-μm paraffin sections, we speculated that the inconsistent staining seen in these specimens could be attributable to variable penetration of the staining reagents into nuclei. This hypothesis was confirmed by the consistent staining of all OHC nuclei on etched, 1-μm-thick epoxy sections. In all probability, OM enters the nucleus via diffusion, because the pores in the nuclear envelope are permeable to small proteins. Conversely, it also may diffuse out of unfixed nuclei during isolation and fractionation procedures.
The possibility that OM could function as a specialized cytosolic Ca2+ buffer within the OHC should not be dismissed. Roberts (1993,1994) has emphasized the need for a mobile Ca2+ buffer in the vicinity of Ca2+ channels to localize regions of high Ca2+ concentration and to expedite the return to resting state levels. Calbindin is conjectured to play this role in frog saccular hair cells (Roberts 1993). OM could serve a similar function in the mammalian OHC, but the seemingly sole reliance of mammalian species on the PV α-isoform for Ca2+ buffering activity renders this hypothesis unlikely. However, until an effector protein for OM is identified, a Ca2+ buffering role for OM cannot be excluded.
The cochlear hair cell is apparently the sole site of OM expression in postnatal mammals. In the interest of optimizing its acoustic response, the organ of Corti has retained only the most minimal metabolic machinery. Viewed against this austere backdrop, the recruitment of the PV β-isoform to the OHCs appears particularly significant. Whether it serves in a regulatory or an ion buffering capacity, it is likely that OM is vital to some unique aspect of OHC physiology or function.
Footnotes
Acknowledgements
Supported by Research Grants R01 DC00713 (BAS), R01 DC01414 (RT), and P01 DC00422 (BAS) from the National Institute on Deafness and Other Communication Disorders, National Institutes of Health.
We thank Ms Leslie Harrelson and Ms Nancy Smythe for editorial and technical assistance.
