Abstract
In this study, we analyzed vitamin D receptor (VDR) expression and survival in a breast cancer patient cohort of 82 breast cancer patients. Immunohistochemical analysis was possible in 91.5% of the patients (75/82). Staining was evaluated using the semi-quantitative assay according to Remmele and Stegner (immunoreactivity score [IRS]). IRS 0–1 was negative/very low, IRS 2–4 was moderate to high, and IRS 6–12 was high. Statistical analysis was performed by Spearman’s correlation test (p<0.05 significant). Overall survival was analyzed using Kaplan-Meier estimations. Only 6 patients had a negative IRS. Moderate IRS values were present in 20 patients. Most of the patients had a high IRS (49). For survival analysis, data were dichotomized (IRS 0–4: negative to moderate and IRS 6–12: high VDR expression). In univariate analysis, VDR expression showed significant differences in progression-free survival (PFS) and overall survival (OS). Patients with high IRS scores showed significantly better PFS and OS than patients with moderate/negative IRS scores for VDR expression. Tumor size was significantly correlated to PFS. When analyzed separately, the three different IRS groups showed significant differences in VDR expression. The present data suggest that VDR expression in breast cancer tissue may be of clinical significance, and the results provide evidence that VDR may be a factor with prognostic relevance.
The vitamin D receptor (VDR) belongs to the nuclear class II receptor family of two known families (Bettoun et al. 2003). The first one includes the estrogen, androgen, progesterone, and mineralocorticoid receptors the second one the thyroid (THR), vitamin D (VDR), retinoic acid (RAR), peroxisome proliferator-activated, and retinoid X receptors (RXR). It is a ligand transcription factor and mediates the actions of 1,25-dihydroxyvitamin D3, the active metabolite of vitamin D, which is involved in cell growth and differentiation in normal and malignant breast tissue, where it shows antiproliferative effects (Perez-Fernandez et al. 2007). VDRs are able to form heterodimers, for example, with RXR, THR, or RAR. These heterodimers have been thought to be nonpermissive because they neither bind nor show activation by RXR ligands (Forman et al. 1995).
In earlier studies, the expression of VDR was demonstrated in several tissues such as breast, bone, prostate, gut, activated B and T lymphocytes, monocytes, and keratinocytes (Gombart et al. 2006). VDR is expressed in epithelial, stromal, and immune cells of the normal mammary gland and is dynamically regulated in the epithelial compartment during hormonal changes in puberty and pregnancy (Zinser and Welsh 2004). Furthermore, it is present in malignant dividing cell types, responding to 1,25 vitamin D3 (Gombart et al. 2006). Beside VDR expression in most of the breast cancer cell lines (Frampton et al. 1982), there is an upregulation of VDR at the protein level in breast carcinoma as compared to normal breast tissue (Friedrich et al. 2002). A higher VDR expression in tumors seems to be correlated with a better prognosis in cancer patients (Berger et al. 1991). This could be dependent on the active forms of vitamin D, which is known to be an inhibitor of growth and is able to induce apoptosis (Narvaez and Welsh 2001; Friedrich et al. 2002; Matthews et al. 2010).
In the present study, immunohistochemical staining of VDR in paraffin-embedded malignant breast tissue was associated with the clinical pathological findings of these tumors. Furthermore, this investigation was performed to analyze the possible prognostic relevance of VDR in breast cancer patients.
Material and Methods
Study Population
Breast cancer samples from 82 patients of the Department of Gynaecology and Obstetrics of the Ludwig Maximilian University of Munich, Germany, were included in this study after permission from the local Ethics Committee. Paraffin-embedded tissues were sampled between 1990 and 2000 from patients with a first diagnosis of breast cancer. Patients fulfilling the inclusion criteria were chosen on the basis of equal distribution among certain clinical pathological findings such as lymph node involvement, histopathological type (invasive ductal), and as far as possible estrogen/progesterone receptors (Fig. 1). All tumor samples were classified by the TNM system (Boecker 2002). The histological classification was determined according to a modification of the Elston and Ellis grading proposed by Bloom and Richardson (Elston and Ellis 1991). The hormone receptor status was evaluated by immunohistochemistry. The tumor was classified as hormone receptor positive in case of positive staining in ≥10% of the tumor cell nuclei. Dissection of axillary lymph nodes was carried out in all of the patients. Only patients with immunostaining evaluable for VDR were chosen for further analysis. The age of the patients ranged between 54 and 95 years. All of the patients were postmenopausal.
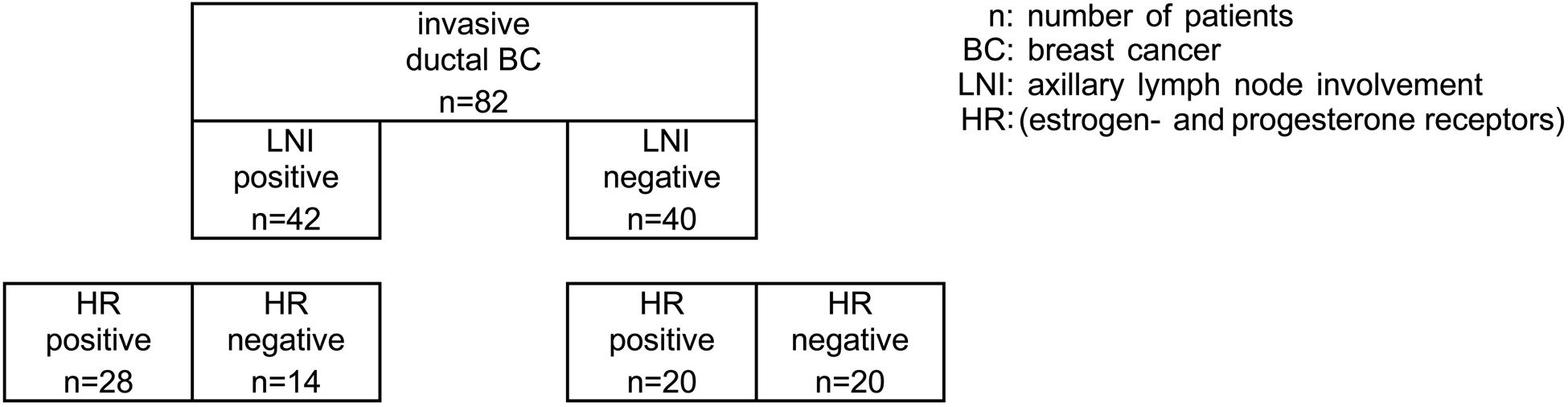
Patient cohort.
Clinical and Histopathological Data
The distribution of clinical and histopathological data such as tumor size, axillary node involvement, grading, estrogen-progesterone receptor (ER/PR) status, and Her-2 expression is presented in Table 1. Because of the tissue detachment in seven slides, data were evaluable only in 75 of 82 patients. Her-2 expression was reexamined by immunohistochemistry. Values of 0 and 1 were considered negative, values of 3+ were classified as positive, and in case of 2+, a fluorescence in situ hybridization was performed. As retrospectively analyzed, most patients had a negative Her-2 status. In six patients, it was not possible to determine Her-2 expression because of missing tissue.
Tumor Data of the Patient Cohort

p, pathological classification; LNI, lymph node involvement; ER, estrogen receptor; PR, progesterone receptor; n, number of patients.
Very small tumors were rare; most patients had a pT1c. The other clinical and pathological parameters were similarly distributed to data known from the literature.
All of the ER/PR-positive patients received antihormonal therapy.
VDR-positive results showed brownish staining of the nucleus and cytoplasm. Slides were considered negative when the immunoreactivity score (IRS) was 0–1, moderately positive when IRS was 2–4, or highly positive when IRS was 6–12. To simplify statistical analysis, data had to be dichotomized because the number of patients and events in the 0–1 IRS group was small.
Of 75 patients, immunohistochemical analysis revealed VDR-positive tumors in 69 (92%) of all tissues investigated (Fig. 2A,B). Six patients (8%) showed no expression of VDR (IRS 0–1). Moderate IRS values were presented in 20 patients (26.7%): IRS value of 2 was presented in 2 (2.7%), IRS of 3 in 8 (10.7%), and IRS of 4 in 10 (13.3%) of these patients. Most of the patients had a high IRS (65.3%)—an IRS of 6 was prevalent in 9 (12%), an IRS of 8 in 16 (21.3%), an IRS of 9 in 5 (6.7%), and an IRS of 12 in 19 (25.3%).
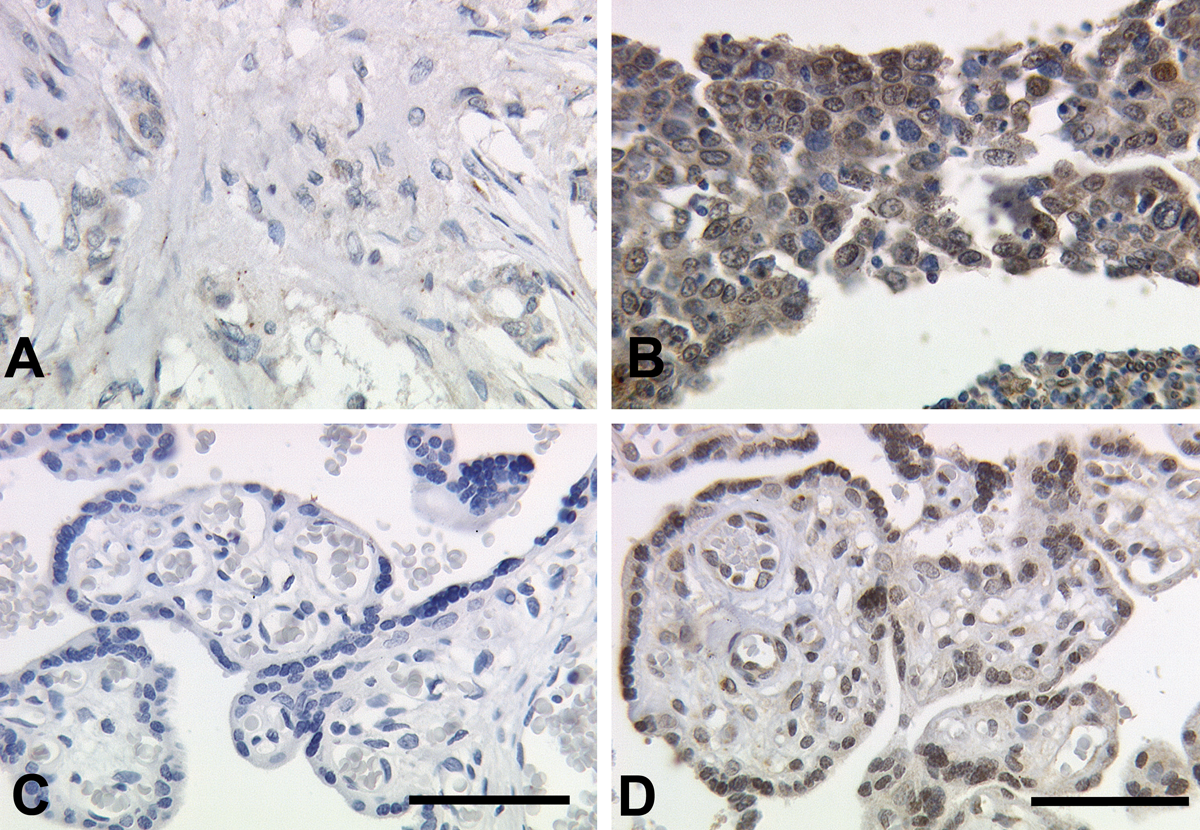
(A, B) Immunohistochemical staining of vitamin D receptor (VDR) in human breast cancer. The illustrations show immunoreaction after incubation with the primary antibody of the cells of the malignant breast tumors (25× lens). (A) Negative/low immunoreactivity score (IRS) and (B) high IRS. (C, D) Placental tissue serves as negative and positive control for VDR. For negative controls (blue), isotype-matching control antibodies of the same species were used (C). Positive control (D) shows VDR staining of villous trophoblast cells. Bars = 100 µm.
Correlations of VDR with Clinical Pathological Findings
On correlation analysis, VDR showed negative associations with tumor size (Spearman rho [SR] = −0.278, p=0.016) and lymph node involvement (SR = −0.411, p<0.01). No correlation was found with regard to grading (SR = −0.044, p=0.706), estrogen/progesterone receptor status (SR = 0.067, p=0.567), and Her-2 expression (SR = −0.006, p=0.959).
Progression-free and Overall Survival
Until 2010, we were able to collect current follow-up data for 65 of 75 patients (86.67%). Overall, 22 of 75 (29.3%) patients had recurrent disease since 1990; 18 of these patients had hematogenous metastatic disease, 12 had local recurrence, and 5 presented with contralateral breast cancer. Twenty-four (32%) of the breast cancer patients died, most of them due to metastatic disease.
Immunohistochemistry
Samples were processed for immunohistochemistry using a combination of pressure cooker heating and the standard streptavidin-biotin-peroxidase complex with the mouse IgG-Vectastain Elite ABC kit (Vector Laboratories; Burlingame, CA). Sections of 5 µm thickness were deparaffinized, hydrated, and incubated using xylol for 15 min; rehydrated in a descending series of alcohol (100%, 96%, and 70%); and, for antigen retrieval, incubated for 10 min using sodium citrate buffer (pH 6.0) containing 0.1 M citric acid and 0.1 M sodium citrate in distilled water. After washing in PBS, endogenous peroxidase activity was blocked by immersion in 3% hydrogen peroxide (Merck; Darmstadt, Germany) in methanol for 20 min. To prevent nonspecific binding of the primary antibody, the sections were incubated with diluted normal serum (10 ml PBS containing 150 µl horse serum; Vector Laboratories) for 20 min at room temperature. Sections were then incubated at room temperature for 60 min with the vitamin D antibody (monoclonal, clone 2F4, isotype IgG2a, AbD Serotec, MCA 3543Z) concentration of 0.5 mg/ml and diluted 1:100 in biotinylated serum (10 ml PBS containing 50 µl horse serum; Vector Laboratories) for 30 min at room temperature. Incubation was performed with the avidin-biotin-peroxidase complex (diluted in 10 ml PBS; Vector Laboratories) for 30 min. The chromagen 3,3′-diaminobenzidine (DAB; Dako, Glostrup, Denmark) was used for 8 to 10 min for visualization. Sections were counterstained with Mayer’s acidic haematoxylin, dehydrated, and covered.
Negative and positive controls (placental tissue) were used to assess the specificity of immunoreactions. For negative controls (colored blue), isotype-matched control antibodies of the same species (DAKO, Hamburg, Germany) were applied on breast cancer tissue that showed high VDR IRS expression (Fig. 2C). Negative control tissue showed neither nuclear nor cytoplasmic staining. On the other hand, it could be demonstrated that cytoplasmic staining was also specific. Positive cells showed a brownish color (Fig. 2D). Negative controls as well as unstained cells appeared in blue.
The IRS was assigned according to Remmele and Stegner (1987). The assessment of the degree of staining and distribution patterns of specific immunohistochemical staining were evaluated using a semi-quantitative assay as used for ER/PR receptors (Jeschke et al. 2005; Mylonas et al. 2005). The IRS was calculated by multiplication of the staining intensity. The percentage of cells with positive staining was scored as follows: 0 = no, 1 = weak, 2 = moderate, and 3 = strong staining; the percentage of positively stained cells was scored as follows: 0 = no staining, 1 = <10% of cells, 2 = 11% to 50% of cells, 3 = 51% to 80% of cells, and 4 = >81% of cells stained. The total score per sample therefore ranged from 0 to 12; 0 to 1 indicates no staining (e.g., negative results), 2 to 4 indicates moderate staining, and 6 to 12 indicates high staining. This evaluation was based on the original Remmele and Stegner (1987) characterization for hormone receptors in breast cancer. The slides were examined in a blinded way by the same experienced investigators using a CCD color camera (JVC, Victor Company of Japan; Yokohama, Japan) and a Leitz (Wetzlar, Germany) microscope.
Statistical Analysis
Statistical analysis was performed with PASW version 18.0 for windows (PASW; Chicago, IL) using Spearman’s correlation test. A 5% significance level was adopted. Independent variables were analyzed by the Mann-Whitney U test and Kruskal-Wallis test, respectively. Univariate analysis was done with encoding items. Progression-free and overall survival was analyzed using Kaplan-Meier estimations. Multivariate models were analyzed by Cox regression analysis.
Results
Data analyses included the evaluation of progression-free survival and overall survival. The univariate and multivariate model was used to show the prognostic relevance of the clinical and histopathological parameters. For Cox regression and Kaplan-Meier analysis, data were dichotomized (IRS 0–4 vs IRS 6–12). Furthermore, the intensity of scoring and the percentage of cells staining positive were assessed independently for correlation with outcome.
Progression-free Survival
Univariate analysis (Table 2) showed significant results for VDR (p=0.046) and of the known prognostic parameters for tumor size (pT) (p=0.001) and lymph node involvement (p=0.009). When separately analyzed for intensity and percentage of VDR-positive cells, significant differences were present only for the percentage of positive stained cells (p=0.001) in contrast to the intensity of VDR (p=0.131). For all other parameters, no significant differences could be demonstrated.
Univariate and Multivariate Analysis—Disease-free Survival
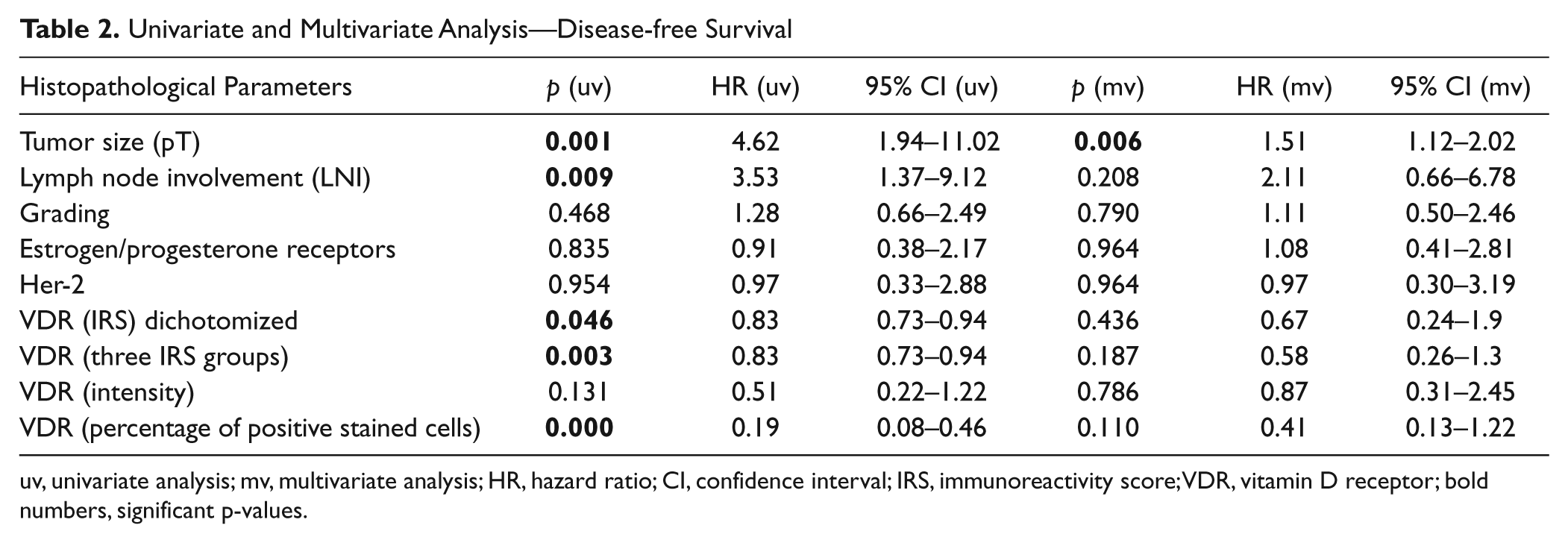
uv, univariate analysis; mv, multivariate analysis; HR, hazard ratio; CI, confidence interval; IRS, immunoreactivity score; VDR, vitamin D receptor; bold numbers, significant p-values.
As shown in Kaplan-Meier curve (Fig. 3A), patients with higher IRS (6–12) showed a significantly higher progression-free survival (PFS; log rank p=0.037 after dichotomization). Mean PFS differed between 8.4 years for the IRS 0–4 group and 14.3 years for the IRS 6–12 group. After a follow-up time of 12 years, about 70% of the patients with an IRS of 6–12 lived progression free in contrast to 50% of the patients with an IRS of 0–4. If data were analyzed separately for intensity of scoring and percentage of cells, survival rates differed (Fig. 3B,C). With regard to intensity, no significant differences for VDR expression could be seen between the groups of 0–1 or 2–3 (log rank p=0.119). In contrast, highly significant differences (log rank p=0.000) were demonstrated for a higher percentage of VDR expression (50%–100%) in contrast to results less than 50%.
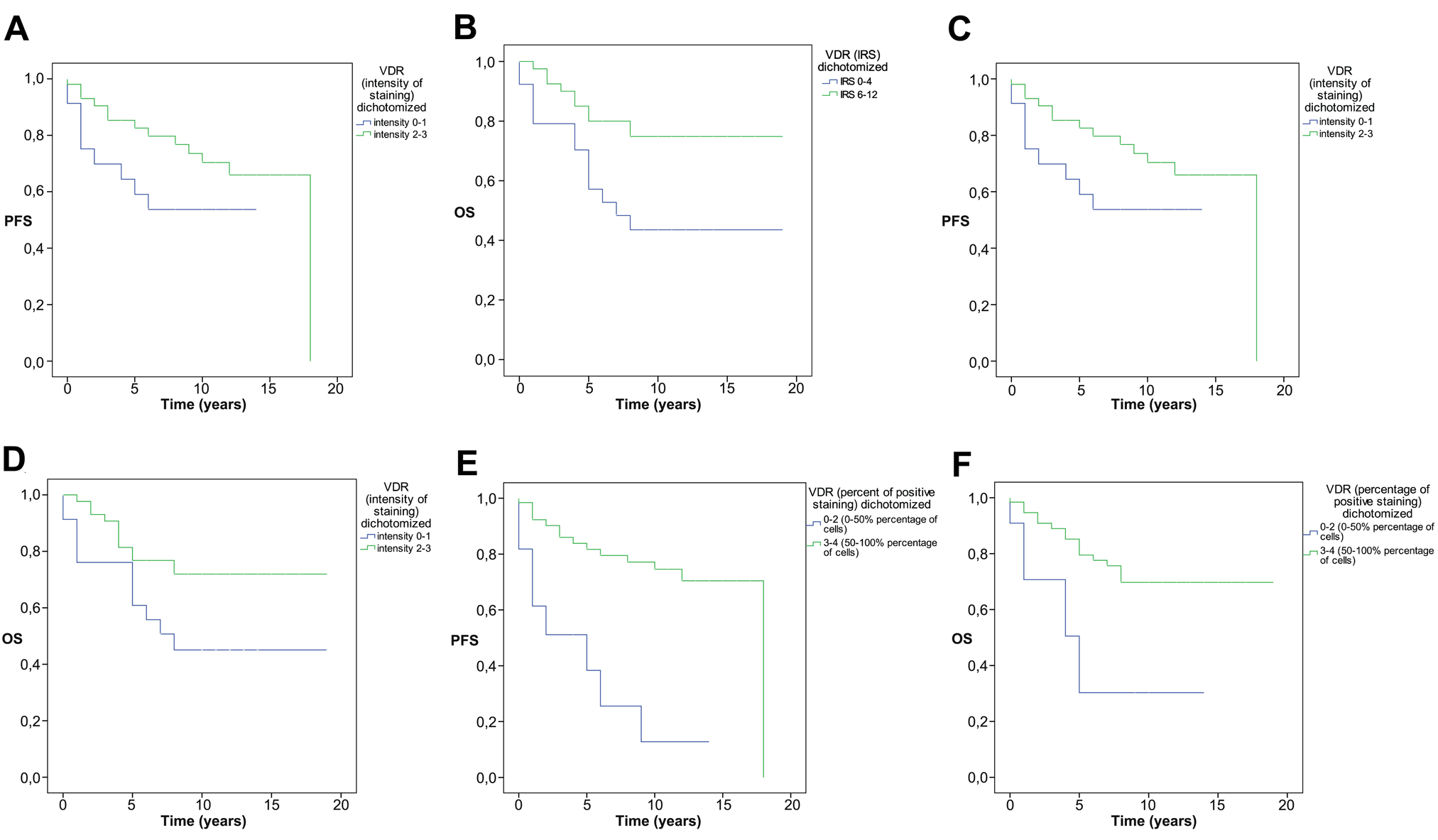
(A) Progressive-free survival (PFS) shown in Kaplan-Meier curves of two different vitamin D receptor groups classified as negative and moderately positive (0–4 immunoreactivity score [IRS]) or highly positive IRS (6–12 IRS). (B) Overall survival (OS) shown in Kaplan-Meier curves of two different vitamin D receptor groups classified as negative and moderately positive (0–4 IRS) or highly positive (6–12 IRS). (C) PFS shown in Kaplan-Meier curves of two different vitamin D receptor groups for intensity of staining (0–1 vs 2–3). (D) OS shown in Kaplan-Meier curves of two different vitamin D receptor groups for intensity of staining (0–1 vs 2–3). (E) PFS shown in Kaplan-Meier curves of two different vitamin D receptor groups for percentage of cells staining positive (0%–50% vs 50%–100%). (F) OS shown in Kaplan-Meier curves of two different vitamin D receptor groups for percentage of cells staining positive (0%–50% vs 50%–100%).
In multivariate analysis for the dichotomized IRS group, as for the selected analysis of intensity and percentage of cells stained positive (Table 2), only one of the parameters proved to be a prognostic factor for PFS (tumor size, p=0.006). VDR could not assert itself as a factor with prognostic importance (p=0.436; hazard ratio [HR] = 0.67; 95% confidence interval [CI], 0.24–1.9). This could also be demonstrated for separate analysis of intensity and percentage of positive-stained cells.
Overall Survival
As shown for PFS, VDR expression also showed significant differences in univariate analysis (p=0.014). When separately analyzed for intensity and percentage of positive cells, VDR showed significant differences (p=0.037 and p=0.007). Furthermore, tumor size (p=0.010) and lymph node involvement (p=0.028) showed significant differences. The other known prognostic factors showed non-significance (Table 3).
Univariate and Multivariate Analysis—Overall Survival
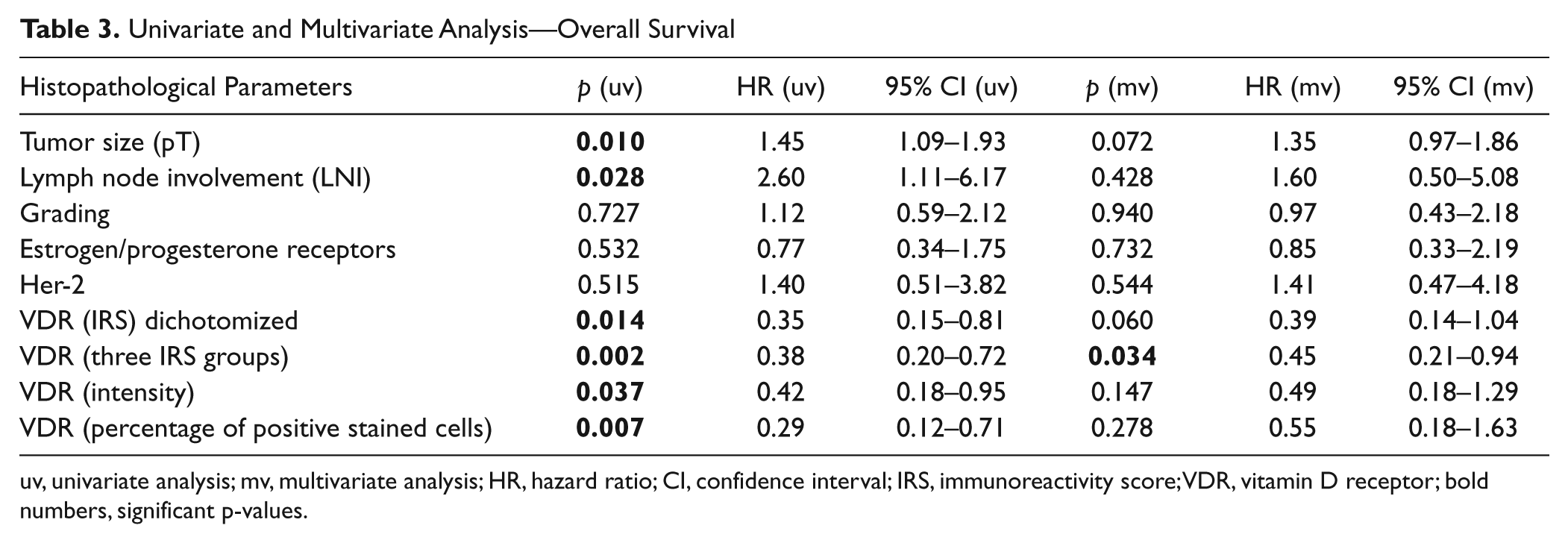
uv, univariate analysis; mv, multivariate analysis; HR, hazard ratio; CI, confidence interval; IRS, immunoreactivity score; VDR, vitamin D receptor; bold numbers, significant p-values.
Significantly better survival could be demonstrated separately for high IRS (log rank p=0.008; Fig. 3D), staining intensity (log rank p=0.028; Fig. 3E), and percentage of cells positively stained (log rank p=0.003; Fig. 3F). Mean survival was longer in the IRS 6–12 versus the IRS 0–4 group (15.3 vs 10.4 years).
None of the prognostic factors yielded significant differences (Table 3). For VDR, a trend for a better overall survival (OS) could be demonstrated (p=0.060; HR = 0.39; 95% CI, 0.14–1.04).
If analysis were done separately for the three groups named above (IRS 0–1, IRS 2–4, and IRS 6–12), significant results could be demonstrated for VDR (Table 3). Separate analysis of intensity and percentage of positive-stained cells of VDR showed non-significant results (Table 3).
Discussion
The results obtained in this study show that VDR expression is negatively associated with tumor size and lymph node involvement on bivariate correlation analysis. Furthermore, VDR expression was a factor with prognostic relevance on univariate analysis. A better outcome was seen in patients with high VDR-IRS than in patients with low IRS. However, after dichotomization in two VDR groups (IRS 0–4 and IRS 6–12), there was only a trend for a better OS. If the same analysis had been done for the three different IRS groups, significant results for OS would have been present. For this patient cohort, separate analysis of intensity, percentage of cells of VDR, and IRS results showed that results probably were influenced mainly by the percentage of positive-stained cells.
VDR, a nuclear transcription-regulating factor that belongs to the steroid hormone superfamily, is encoded by genes. The most well-studied gene is VDRG (vitamin D receptor gene). A few of its single-nucleotide polymorphisms (SNPs) such as Fok1, Bsm1, Taq1, and Poly (A) have been extensively studied (McCullough et al. 2009). All opposed data for associations with breast cancer were demonstrated, but in the case of Fok1, a large pooled analysis of 2009 associations with the Fok1 SNP was slightly stronger for localized invasive cases and did not vary by ER/PR tumor type or menopausal status (McKay et al. 2009). In contrast, data from a cohort of Caucasian women showed a higher breast cancer risk for the Bsm1bb genotype only for postmenopausal women (Trabert et al. 2007). On the basis of recent data, McCullough et al. (2009) stated an important role for vitamin D and its receptor for breast cancer in humans.
VDR is involved in cell growth and differentiation in normal and breast cancer tissue. VDR is expressed in epithelial, stromal, and immune cells of the mammary gland. In the epithelial compartment, it is dynamically regulated in special hormonal phases such as puberty and pregnancy (Zinser et al. 2002; Zinser and Welsh 2004). Animal models have shown that a lack of VDR expression is associated with alterations in proliferation and apoptosis of the epithelial cells (Zinser et al. 2005).
In a variable number of cases of infiltrative carcinoma, immunostaining of VDR occurred in the nucleus (Conde et al. 2004). Our data underline these findings with mainly nuclear VDR expression in breast cancer tissue samples.
We found a correlation between a better prognosis in patients with higher VDR IRS in contrast to patients with very low or no VDR expression. Only one study was found that investigated the immunocytochemical detection of the VDR in breast cancers: Berger et al. (1987) used an immunocytochemical technique to demonstrate the 1,25(OH)2D3 receptor in 80% of breast tumor cells. Our data are consistent with this result, with an approximately 90% positive VDR detection in breast cancer tissue by immunohistochemistry. The study by Berger et al. also demonstrated an association between higher tumor VDR expression and better prognosis (Berger et al. 1991), which was demonstrated by significantly earlier disease relapse in VDR-negative tumors than in VDR-positive tumors (p=0.045), but VDR was not related to overall survival (p>0.1).
Our study showed a trend for prediction of overall survival in contrast to previous studies (Freake et al. 1984; Eisman et al. 1986; Berger et al. 1991) and no effect on disease-free survival, in contrast to Berger et al. (1991). There is evidence (Freake et al. 1984; Eisman et al. 1986) that the effect could depend on the large, positively stained proportion of VDR and therefore the potential responsiveness of vitamin D (Berger et al. 1991). Otherwise, a link could exist between the established histopathological parameters and VDR expression (e.g., high levels of VDR correlated with small tumor sizes).
VDR is upregulated in breast carcinomas at the protein level as compared to normal breast tissue (Conde et al. 2004), indicating a possibly increased sensitivity to therapeutically administered vitamin D analogues (Friedrich et al. 2002). It is known that most dividing cell types expressing VDR respond to 1,25(OH)2D3 (Gombart et al. 2006). Preclinical and ecologic studies support a role of vitamin D in the prevention of breast and other cancer types (Welsh et al. 2002; Zhang et al. 2005). A prospective analysis of breast cancer incidence in relation to vitamin D intake in over 30,000 women in the Women’s Health Study indicated a moderate association between a lower breast cancer risk for premenopausal women and a higher vitamin D intake but was unable to confirm these findings for postmenopausal women (Lin et al. 2007). Further results of large cohort studies focused on postmenopausal status and a vitamin D intake of >800 IU/d could show a possible association with a decrease in risk of breast cancer (Robien et al. 2007). Previous data showed that a relatively noncalcemic analogue of vitamin D, 1-alpha-hydroxy-24-ethyl-cholecalciferol, is a promising chemopreventive agent in experimental mammary carcinogenesis models (Hussain et al. 2006).
Until now, the research data have been inconsistent and do not help to establish a cause-effect relationship (Colston 2008; Chung et al. 2009; Manson et al. 2011).
As shown in previous studies, we confirm that there is no correlation between the presence of VDR and ER/PR (Freake et al. 1984; Ulmann et al. 1984; Berger et al. 1987). However, in contrast to other studies (Clark et al. 1983; Mason et al. 1983; Howat et al. 1985), we were unable to demonstrate prognostic relevance for ER/PR expression, which might be due to the inclusion of the small number of patients. Data of vitamin D intake and breast cancer incidence or prognosis still showed heterogeneous results for postmenopausal patients (Robien et al. 2007; Chlebowski et al. 2008). The data also differed from results of premenopausal women (Lin et al. 2007). Therefore, ongoing research of VDR expression will include a larger cohort of patients, including premenopausal women. Furthermore, for confirmation of a “dose-response” with the IRS, a larger patient cohort that is separately evaluated for intensity and percentage of positive staining of cells is needed.
The strengths of our study are the consistent histopathology review by expert gynecologic oncology pathologists and the long follow-up time. The weakness of our study is the small number of patients, which could be one of the reasons for the different results in multivariate analysis after dichotomization. Therefore, caution is needed in the interpretation of the data because of the failure of VDR measurement in the peripheral blood of the breast cancer patients at the time of tumor biopsy due to the retrospective nature of the study.
Conclusion
VDR expression is a factor associated with clinical prognostic factors such as tumor size and lymph node involvement. Furthermore, VDR expression showed significant results on univariate analysis and a trend for being of prognostic relevance in multivariate analysis. The presence of VDR may be of clinical significance, and the results presented here may provide evidence that VDR is a factor with prognostic relevance that may therefore serve as a new target for innovative cancer therapies. Prospective studies are part of ongoing research, including measurement of vitamin D in peripheral blood.
Footnotes
Acknowledgements
We thank the patients for providing samples for this study. We also acknowledge the excellent technical assistance of M. Rübekeil, S. Kunze, and Ch. Kuhn and help with statistical analyses from A. Crispin. We thank Marion Glass for editorial assistance.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
