Abstract
Morphological data on heart damage and its mechanisms due to extremely severe course of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection are limited, as well as data on the correlation of damage and expression of vitamin D receptors (VDRs). In this study, we analyzed a series of myocardial samples obtained during postmortem autopsy of 48 critically ill patients with COVID-19 who died with SARS-CoV-2-associated pneumonia. The purpose of the study was to evaluate immunohistochemical VDR expression in the myocardium. The results showed the only minimal or no immunohistochemical expression of VDR in the nuclei of cardiomyocytes in most cases, along with the persisted strong expression in lymphoid cells. To the best of our knowledge, it is the first study and data provided were regarding myocardial VDR expression in COVID-19 patients. The results are of interest in terms of further study of the effects of ligand-associated VDR activation on the cardiovascular system.
Introduction
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) (2019-nCoV) infection is known to be a new beta-coronavirus infection, which belongs to the group of acute respiratory viral infections. 1 According to this, the organs of the respiratory system are mainly affected, 2 although recent studies, using the electron microscopy method, have demonstrated direct damage of the heart with the presence of viral particles in the myocardium.3,4 The described clinical and morphological patterns of heart damage include myocardial ischemic injury and acute coronary syndrome, acute myocarditis, or progressive heart failure by the left or right ventricular type up to cardiogenic shock with a prevalence of up to 10% among the general population of patients with COVID-19 and up to 41% among patients with an extremely severe course of infection.5,6 At the same time, the available information is often discordant in terms of the prevailing patterns of heart damage and its consequences. That may be associated with different mechanisms of SARS-CoV-2-associated lesions. 7
The study of 1,25-OH2-D3 (calcitriol, vitamin D3) and its receptors [vitamin D receptor (VDR)] looks promising from the standpoint of a comprehensive assessment of the effects of possible vitamin D deficiency and the relationship of VDR expression with the course and prognosis of coronavirus infection. To date, an extensive evidence base has been accumulated regarding the effectiveness of vitamin D medications in patients with COVID-19, although these data (even within the various meta-analyzes performed) are often contradictory, ranging from the absence of a proven effect of the use of vitamin D on morbidity, severity, and mortality rates in COVID-19 to a decrease in mortality rates up to zero when the target vitamin D3 level is above 50 ng/mL.8–12 The research interest in vitamin D and VDR is due, on one hand, to the fact that the estimated prevalence of severe vitamin D deficiency among people with coronavirus infection reaches 82.2%, 13 and, on the other hand, by the potential of ligand-associated VDR activation with the realization of its pleiotropic effects, both via genomic and non-genomic pathways. The identified VDR-modulated actions include regulation of immunological reactions and various metabolic pathways, as well as regulation of proliferation and differentiation of cells. 14
Data on the presence of VDR in cardiomyocytes, especially in terms of immunohistochemical confirmation of its expression, are extremely limited. The nuclear expression of VDR in cardiomyocytes of experimental animals (rats) and in cell culture was confirmed in several studies using the method of immunohistochemistry.15,16 According to the results obtained using VDR knock-out mice, the role of ligand-associated VDR activation in preventing the myocardial hypertrophy was demonstrated,17,18 as well as the involvement of VDR in the regulation of expression of metalloproteinases in the myocardium. 19
Despite the therapeutic and/or prophylactic potential of the effects (both genomic and non-genomic) of ligand-associated VDR activation in patients with COVID-19, including those aimed at mitigation of the damage to the cardiovascular system, data on the expression of VDR in the heart of the patients with COVID-19, as far as we know, are absent.
Materials and Methods
Patients and Autopsy Specimens
This study was performed using the samples of the left ventricular myocardium taken as a part of the standard postmortem pathomorphological study of 48 patients who died as a result of SARS-CoV-2-associated pneumonia. According to the current local recommendations, the diagnosis in all patients admitted to the hospital was confirmed on the basis of a set of clinical and laboratory data. As a control, autopsy samples of seven patients died for reasons not related to SARS-CoV-2 infection, as well as not related directly to the heart pathology (malignancies in three cases, ischemic stroke in two cases, and gastrointestinal bleeding due to peptic ulcer and liver cirrhosis—one case each). Autopsy sampling was carried out during period from February to May 2021.
This study complied with the Declaration of Helsinki, and was approved by the Ethics Committee on Research of the VI Vernadsky Crimean Federal University (No. A16-116072810135-5 dated January 20, 2021). The taking of autopsy samples was performed within the standard medical practice, regulated by Federal Law N 323-FZ dated November 21, 2011 (amended on July 2, 2021). All personal data of deceased patients were anonymized, and the personal data used within the study included gender and age of the patients.
Routine Histological and Immunohistochemical Study
After collection, samples of the left ventricular myocardium were fixed in neutral buffered 10% formalin for 24–48 hr, followed by histological processing using Logos Hybrid Histological Processor (Milestone Medical; Italy) and Leica EG1150 Modular Tissue Embedding Center (Leica Biosystems; Germany). Histological sections (~4 μm) were obtained from 10% formalin-fixed paraffin-embedded (FFPE) blocks using an automatic rotary microtome Leica RM2255 (Leica Biosystems) and stained with hematoxylin and eosin for provisional microscopic evaluation.
For immunohistochemical detection of VDR expression, FFPE histological 4-µm sections were used. Staining was performed using a BondMax Semiautomatic Immunohistostiner (Leica Biosystems). The staining protocol included dewaxing, heat antigen unmasking using Bond Epitope Retrieval 2 solution (Leica Biosystems) at pH = 9 for 20 min at 96°C, blocking peroxidase activity, incubation with the antibody for 15 min at room temperature, and visualization using the Bond Polymer Refine Detection system (Leica Biosystems). Rabbit IgG polyclonal anti-VDR antibodies at a dilution of 1:100 (VDR Antibody, CSB-PA060063; Cusabio, Houston, TX) were used. The specificity of VDR antibody was confirmed by the manufacturer (Cusabio) using Western blot analysis (unpublished data). For better visualization, counterstaining was performed with hematoxylin. Histological preparations were scanned using an Aperio CS2 Digital Pathology Slide Scanner (Leica Biosystems), followed by digital image analysis using Aperio ImageScope software.
The intensity of immunoreactivity (using 3,3`-Diaminobenzidine, DAB), if any, was assessed as the sum of the total intensity of nuclear and/or cytoplasmic brownish staining in the cell population as minimal or weak positive (± or +), moderately positive (++), or strong positive (+++) reactions. Samples of human kidneys were used as a positive control. In addition, lymphocytes were also used as an internal positive control with a strong positive immunohistochemical expression of VDR. Negative control was performed using the same samples by omitting the primary antibody.
Results
Basic Characteristics of Deceased Patients
Forty-eight patients died due to COVID-19 complications including 18 men (37.5%) and 30 women (62.5%). Median age was 76 years (range: 39–95 years). Median age of control group patients was 71 years (range: 59–89 years). In all cases, the diagnosis of SARS-CoV-2-associated infection was confirmed by the repeated positive PCR tests for viral RNA, as well as clinical and radiological findings. Some of the demographic, as well as anamnestic, clinical, and laboratory data were not available; therefore, only general data were summarized within this study.
Regarding all the cases, the primary diagnosis was coronavirus infection (COVID-19) with SARS-CoV-2-associated pneumonia and acute respiratory distress syndrome. The most common comorbidities in this group of patients included conditions such as atherosclerosis and coronary heart disease/coronary artery disease (38 cases; 79.2%), heart failure (33 cases; 68.7%), elevated blood pressure (22 cases; 45.8%), impaired glucose tolerance/diabetes mellitus, type 2 (8 cases; 16.7%), cardiac arrhythmias (5 cases; 10.4%), and obesity (4 cases; 8.3%). Other observed pathological conditions included urolithiasis (three cases; 6.25%), as well as chronic pyelonephritis, chronic pancreatitis, rheumatoid arthritis, peptic ulcer, HIV infection, prostate cancer, and lung neoplasm (one case for each; 2.1% each). In the control group, six of the seven cases (85.7%) were characterized by the presence of atherosclerosis and coronary heart disease as comorbidities.
Immunohistochemical Evaluation
Postmortem myocardial samples of the patients who died from COVID-19 were characterized by the absence of autolytic changes with the preservation of the general histological architecture of the myocardium, along with more or less pronounced ischemic or inflammatory changes (Fig. 1A).
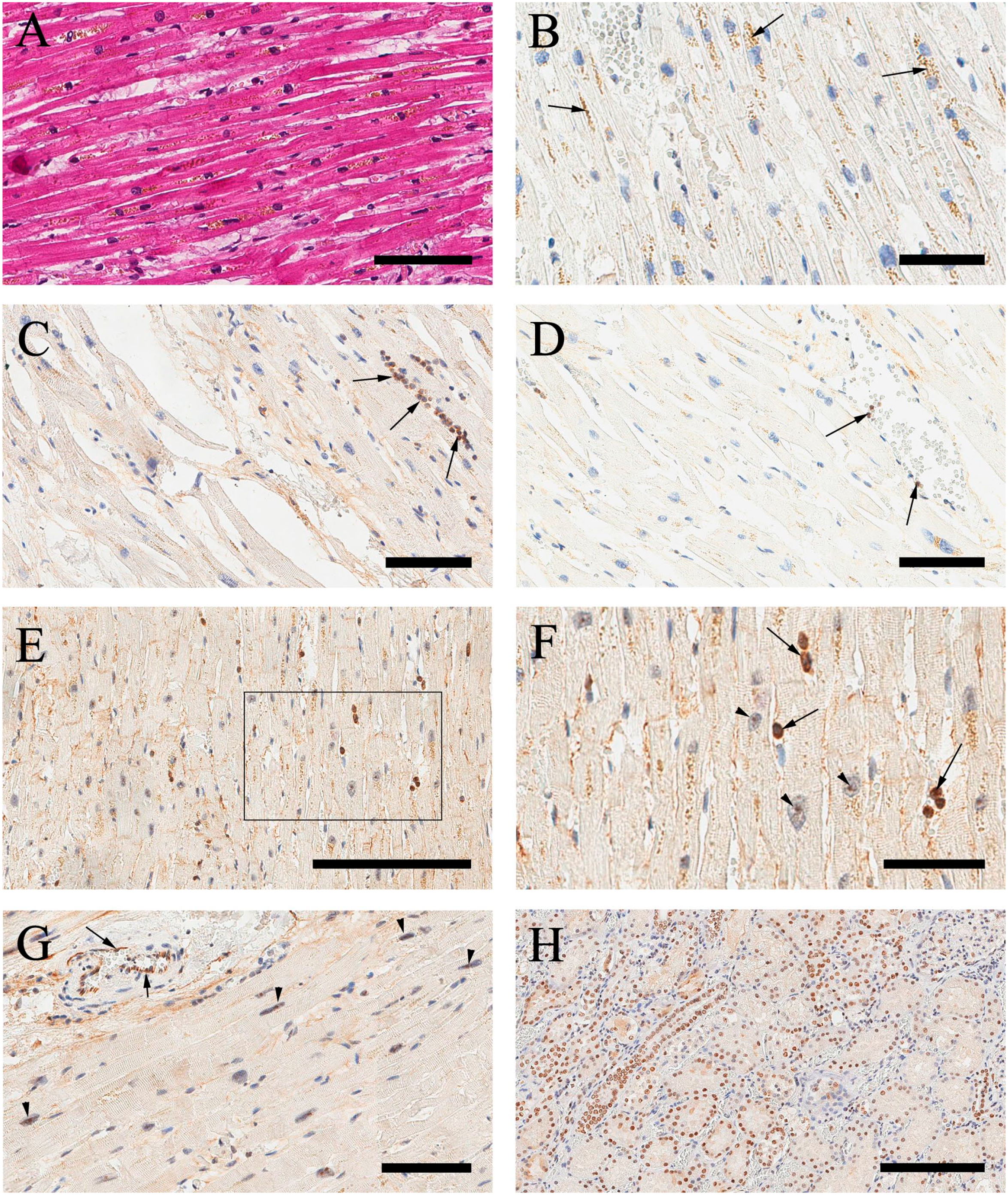
(A) Histological appearance of myocardium with preserved histoarchitectonics of contractile cardiomyocytes with mild ischemic changes and interstitial edema (representative case no. 23). H&E. Scale bar 80 μm. (B) Lack of immunohistochemical VDR expression in cardiomyocytes. Brown granules in the cytoplasm of cardiomyocytes (arrows) are due to the intracellular accumulation of lipofuscin pigment (representative case no. 23). Scale bar 50 µm. (C) Strongly VDR-positive lymphoid cells (arrows) against the background of the absence of immunoreactivity in cardiomyocytes (representative case no. 12). Scale bar 100 μm. (D) Lack of immunohistochemical VDR expression in cardiomyocytes and endothelial cells with isolated positive intravascular lymphocytes (representative case no. 24). Scale bar 100 μm. (E, F) Minimal anti-VDR immunoreactivity (nucleoli) (arrowheads) with positive lymphocytes (arrows) (representative case no. 21). Scale bar 200 μm and 60 μm, respectively. (G) Minimal IHC VDR expression in nucleoli of cardiomyocytes (arrowheads) with strong positive expression in endothelial cells (arrows) (representative case no. 15). Scale bar 90 μm. (H) Same case (no. 15) kidney with strong positive anti-VDR nuclear immunoreactivity in proximal tubules. Scale bar 200 μm. Abbreviation: VDR, vitamin D receptor.
In all the COVID-19 cases, with the exception of a small number of observations (discussed below), cardiomyocytes were characterized by only minimal (14 cases; 29.2%) or no (25 cases; 52.1%) immunohistochemical expression of VDR, both nuclear and cytoplasmic (Fig. 1B). Vascular endothelial cells also lacked any immunoreactivity with anti-VDR antibody. The presence of lymphoid cells with pronounced nuclear and/or cytoplasmic immunohistochemical VDR expression has confirmed the reactogenicity of the used anti-VDR antibody (Fig. 1C and D). In the cases of very low or minimal anti-VDR immunoreactivity, immunopositive regions were still present in the nucleoli of a number of contractile cardiomyocytes (Fig. 1E and F). In other cases, despite minimal IHC anti-VDR reactivity in cardiomyocytes, endothelial cells were characterized by strong positive expression (Fig. 1G). As a confirmation of anti-VDR reactivity, kidney specimens mounted on the same glass consistenly showed strong positive reaction with anti-VDR antibody (Fig. 1H).
In some cases, a pronounced perinuclear brown staining was noted in cardiomyocytes. The localization of staining coincided with accumulation of lipofuscin pigment. Taking into account the available information regarding nonspecific cross-reactivity of antibodies with the protein components of lipofuscin, 20 these areas were not considered as positive immunoreactivity and were excluded from the subsequent analysis of the obtained results.
In contrast to the above-mentioned cases, there were five cases (no. 9, 16, 20, 25, and 46) and four cases (no. 10, 17, 22, and 28) characterized by strong and moderate IHC reaction, respectively, featuring 18.7% in total of the analyzed cases (Table 1).
Summary of Immunohistochemical VDR Expression in Cardiomyocytes in Critically Ill Patients Decesead From SARS-CoV-2-associated Pneumonia.
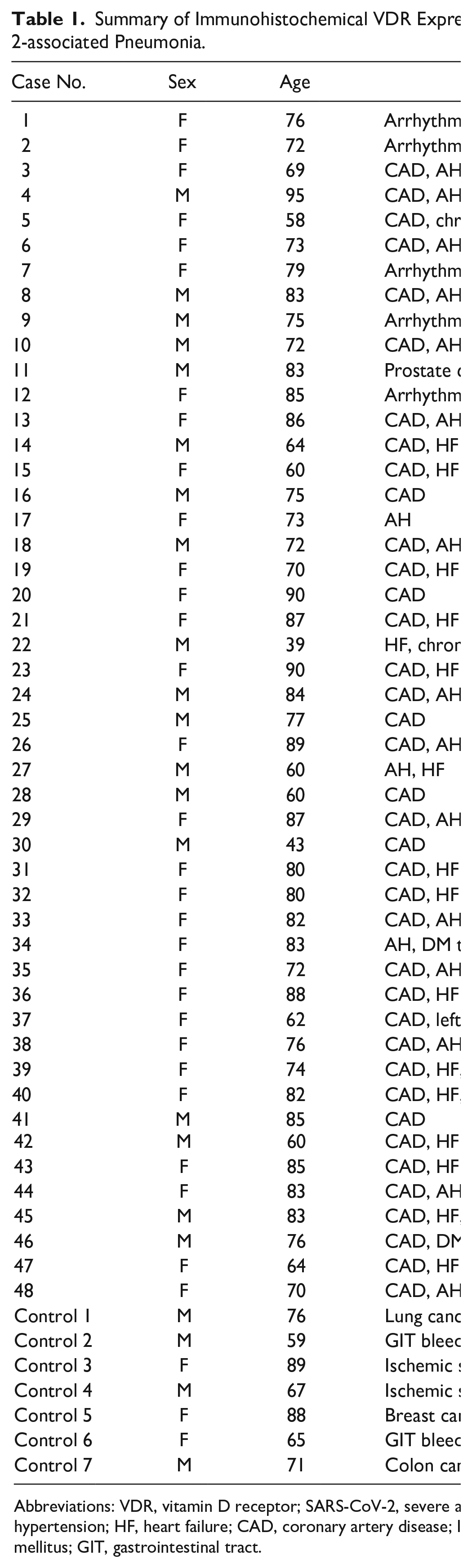
Abbreviations: VDR, vitamin D receptor; SARS-CoV-2, severe acute respiratory syndrome coronavirus-2; AH, arterial hypertension/essential hypertension; HF, heart failure; CAD, coronary artery disease; IGT, impaired glucose tolerance; HIV, human immunodeficiency virus; DM, diabetes mellitus; GIT, gastrointestinal tract.
In these observations, cardiomyocytes were characterized by moderate and/or strong immunohistochemical expression of VDR in the region of the cell nuclei (Fig. 2A and B). Notably, the most pronounced immunoreactivity was observed precisely in the region of the nucleoli against the background of moderate expression in the nucleoplasm. Anti-VDR immunoreactivity in the cytoplasm of cardiomyocytes was still absent. In addition, vascular endothelial cells were also characterized by the positive immunohistochemical expression of VDR (Fig. 2C and D).
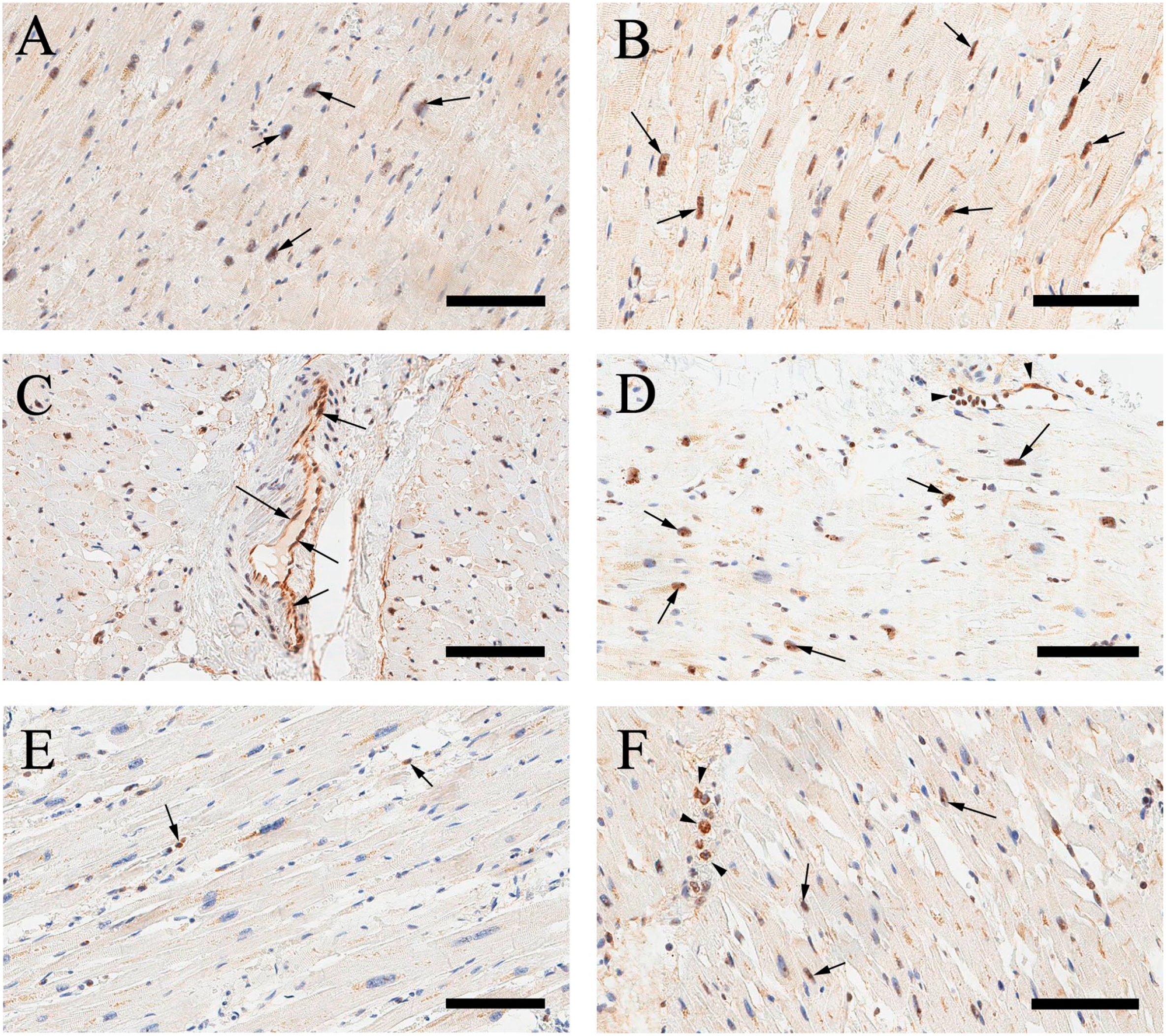
(A) Moderate immunohistochemical VDR expression in cardiomyocytes with predominantly nucleolar localization (arrows) (representative case no. 17). Scale bar 100 μm. (B) Strongly positive immunohistochemical VDR expression in cardiomyocytes (arrows) (representative case no. 16). Scale bar 80 μm. (C) Strong immunopositive reaction against anti-VDR antibody in endothelial cells of a blood vessel in the myocardium (arrows) (representative case no. 46). Scale bar 100 μm. (D) Strongly positive immunohistochemical VDR expression in cardiomyocytes (arrows), as well as endothelial cells and intravascular lymphocytes (arrowheads) (representative case no. 20). Scale bar 100 μm. (E) Lack of anti-VDR immunoreactivity in cardiomyocytes with few positive lymphocytes (arrows) (control no. 3). Scale bar 100 μm. (F) Minimal IHC VDR expression in nucleoli of cardiomyocytes (arrows) with strong positive cytoplasmic expression in leukocytes (arrowheads) (control no. 6). Scale bar 80 μm. Abbreviation: VDR, vitamin D receptor.
It is noteworthy that in the control group, the positive IHC VDR expression was detected only in one case (14.3%). Two cases were characterized by minimal anti-VDR immunoreactivity and four cases showed no anti-VDR immunoreactivity (Fig. 2E and F).
Discussion
Generally, the obtained study results using postmortem myocardial images of critically ill patients with COVID-19 showed a minimal or no immunohistochemical expression of VDR in cardiomyocytes in the vast majority of cases. These results were unexpected because a number of studies have demonstrated the presence of pronounced nuclear immunohistochemical VDR expression in cardiomyocytes. Indeed, Fraga et al. confirmed the nuclear VDR expression in rodent cardiomyocytes using the method of immunohistochemistry. The obtained data were also confirmed by the results of PCR analysis to identify the corresponding mRNA. 15 Nibbelink et al. demonstrated VDR expression in the cell culture of mice cardiomyocytes, as well as the role of ligand-associated activation of VDR on the functioning of cardiomyocytes. The authors also demonstrated an upregulation of VDR expression in response to 1,25(OH)2-vitamin D3. 16 Regarding the human heart, O’Connell and Simpson 21 were among the first who confirmed the presence of VDR in heart tissues using the Western blotting method. Subsequently, the VDR expression in heart tissues was confirmed in a number of studies; however, no data on immunohistochemical detection of VDR in human cardiomyocytes were found. In addition, it was found that the VDR expression in contractile cardiomyocytes is localized not only in the nuclei but also in the cytoplasm 22 and t-tubules of cardiomyocytes. 23
The available data on the role of ligand-associated VDR activation in cardiomyocytes can be arranged into two groups of effects: genomic and non-genomic. Presumably, genomic effects are associated, among other, with suppression of cell proliferation without induction of apoptosis, 24 stimulation of microtubule formation in cardiomyocytes, 23 and regulation of the WNT signaling pathway. 22 A group of non-genomic effects of ligand-associated VDR activation due to its translocation into the cytoplasm and cell membrane in cardiomyocytes is based on the regulation of the protein kinase C signaling pathway,25,26 cAMP, 27 and protein kinase A.28,29
The above-mentioned information is rather limited, in particular, in relation to the study of the human heart, both in normal conditions and in various pathologies. Nevertheless, it provides convincing evidence of the involvement of VDR in the regulation of the development and differentiation of cardiomyocytes during ontogenesis,15,22 as well as development of cardiac hypertrophy 18 and a number of other pathological conditions. 30
Concerning the VDR expression in patients with COVID-19, there are no available data. As far as we know, this study is one of the first that confirms immunohistochemical expression of VDR in human cardiomyocytes and the first regarding the VDR expression in the myocardium in patients with COVID-19. Considering observations in the control group, it is likely that the minimal or no IHC expression of VDR is not specific for SARS-CoV-2 infection, but rather associated with vitamin D deficiency in the population of critical ill and elderly patients.
According to the results of the analysis of a series of postmortem myocardial samples obtained during autopsy of 48 patients with an extremely severe course of COVID-19 who died due to SARS-CoV-2-associated pneumonia, immunohistochemical expression of VDR in cardiomyocytes was absent or was characterized by only insignificant immunoreactivity with localization in nucleoli of cells in most of the cases. The obtained data may significantly limit the potential for realizing the effects of 1,25(OH)2-vitamin D3 on cardiac muscles, particularly in critically ill patients with COVID-19.
Study Limitations
Despite the significance of the obtained results, the authors acknowledge the existence of a number of objective limitations of this study. This study was performed on a relatively small amount of autopsy material (48 samples) taken from deceased critically ill patients with COVID-19.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MAK conceived and designed the study. AVG obtained autopsy samples. TPM and EYZ performed the histological and immunohistochemistry methods. MAK, YAY, AAD, and AAB wrote the article. MAK, AVK, YAY, AAD, AAB, AAG, and AIZ analyzed the data and edited the draft manuscript. All authors read, reviewed, and approved the final manuscript before submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Russian Foundation for Basic Research (RFBR) and Republic of Crimea, project No. 20-415-910004, and had financial support of State Program No. FZEG-2020-0060 of the Ministry of Science and Higher Education of the Russian Federation.
