Abstract
Mucin O-glycosylation is characterized in cancer by aberrant expression of immature carbohydrate structures (Tn, T, and sialyl-Tn antigens). The UDP-N-acetyl-
T
All mucin-bound O-glycans are built up in a sequential step-by-step process in the Golgi apparatus, starting with the addition of a GalNAc to serine or threonine residues (synthesis of Tn antigen). Subsequently, elongation of O-linked sugars is achieved by transfer of additional sugar residues to the already glycosylated protein. These reactions are catalyzed by different glycosyltransferases whose specificities, sequential action, relative activity levels, and intracellular localization determine a cell-specific O-glycosylation profile. The initial key step in the regulation of O-glycosylation is the transfer of GalNAc from UDP-GalNAc to Ser or Thr residues on an acceptor polypeptide. This reaction is catalyzed by a family of UDP-GalNAc: polypeptide N-acetylgalactosaminyltransferases (ppGalNAc-T) (2.4. 1.41), of which 15 members have been identified in mammals (Ten Hagen et al. 2003; Wang et al. 2003; Cheng et al. 2004). Functional profiles of each component of the family have been characterized showing that these enzymes not only have different substrate specificities but also specific tissue-expression patterns (Sutherlin et al. 1997; Mandel et al. 1999; Young et al. 2003). One mechanism for mucin glycosylation changes in cancer cells could be a differential expression of ppGalNAc-Ts (Hanisch 2001). Immunohistochemical studies have shown an altered expression of some ppGalNAc-Ts in malignant transformation. Different expression patterns have been described in oral squamous cell carcinoma, in which decrease of ppGalNAc-T1 and increase of ppGalNAc-T2 and -T3 have been reported, compared with the expression pattern in normal oral mucosa (Mandel et al. 1999). In colorectal carcinoma, higher expression of ppGalNAc-T1 and ppGalNAc-T2 has also been described in comparison with normal colon epithelium (Kohsaki et al. 2000). ppGalNAc-T3 is highly tissue specific and is abundantly expressed in tumor cell lines arising from epithelial glands such as those from breast, colon, and prostate tissue (Nomoto et al. 1999). Several studies suggest that ppGalNAc-T3 could constitute a new tumor marker, applicable to the evaluation of patients with colorectal (Shibao et al. 2002), lung (Gu et al. 2004), pancreatic (Yamamoto et al. 2004), gastric (Ishikawa et al. 2004), gallbladder (Miyahara et al. 2004), and prostate (Landers et al. 2005) carcinomas.
We have recently reported mRNA expression of ppGalNAc-T6 in human breast cancer cell lines and breast tumors (Freire T, et al., unpublished data). The ppGalNAc-T6 mRNA evaluated by RT-PCR assay in bone marrow aspirates from breast cancer patients seems to be a specific marker, applicable to molecular diagnosis of tumor cell dissemination. In the present work we have produced a monoclonal antibody (MAb T6.3) against this enzyme, to evaluate the potential role of ppGalNAc-T6 as an immunohistochemical breast cancer tumor marker. We found that ppGalNAc-T6 is expressed in the majority of human breast carcinomas but not in normal breast epithelium and is sporadically found in non-malignant breast diseases.
Materials and Methods
Peptides
In selecting a strategy for antibody production, we considered the high sequence homology observed among ppGalNAc-Ts family members and chose a synthetic peptide whose amino acid sequence was observed only in ppGalNAc-T6 and not in any other ppGalNAc-T isoenzymes. The peptide EAQQTLFSINQSCLPGFYTPAELKP, used to generate the specific MAb, was designed according to the amino acid sequence of ppGalNAc-T6, accession number CAA69876 (Bennett et al. 1999), corresponding to residues 77–101. This is a region that displays very high variability among ppGalNAc-Ts family members. The peptide sequence contains a motif (CLPGFYTPAELKP) partially shared by ppGalNAc-T6 and ppGalNAc-T3 (the closest homolog of ppGalNAc-T6) and a motif (EAQQTLFSINQS) expressed by ppGalNAc-T6 only. Two protein conjugates of the same ppGalNAc-T6-derived peptide were synthesized using N-hydroxysuccinimide and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (Biosynthesis; Lewisville, TX): (a) KLH-conjugated peptide (used for mice immunizations) and (b) BSA-conjugated peptide (used in antibody specificity evaluation). The synthetic peptides EAQQTLFSINQS and CLPGFYTPAELKP were purchased from NeoMPS (Strasbourg, France).
Production of Monoclonal Antibodies
For anti-ppGalNAc-T6 MAb production, BALB/c mice were immunized four times with 100 μg of KLH-conjugated synthetic peptide at 2-week intervals. Isolated spleen cells were fused with mouse myeloma cells SP2/O. Screening of ppGalNAc-T6-positive hybridoma supernatants were evaluated by ELISA using the BSA-conjugated synthetic peptide and BSA alone. The microtiter plates were coated with 50 μl/well of 10 μg/ml antigens in freshly prepared bicarbonate buffer and incubated overnight at room temperature (RT). After washing with 0.1% Tween 20 in PBS (washing buffer), nonspecific sites were blocked with 1% (w/v) gelatin in PBS for 1 hr at 37C. After three washes, 100 μl of test supernatant was incubated for 2 hr at 37C. Secondary antibody (goat anti-mouse polyvalent antibody peroxidase conjugated; Sigma, St Louis, MO) was added (1 hr at 37C), and peroxidase activity was demonstrated by incubation with phosphate-citrate buffer, pH 5.0, containing ABTS [2,2-azin-bis(3-ethylbenz-thiazoline-6-sulfonic acid)] with hydrogen peroxide. The reaction was allowed to proceed for 30 min at RT and absorbance was read at 405 nm. Cells from positive wells were further cloned by limiting dilution three times. Immunoglobulins were typified using mouse monoclonal isotyping reagents (Sigma). MAb T6.3 (IgG1) was purified by affinity chromatography using a Protein A column (Sigma).
Molecular Cloning and Expression of Recombinant Human ppGalNAc-T6
A sequence of the coding region of the human ppGalNAc-T6 corresponding to the sequence 157–1019 of the mRNA (accession number NM_007210) was amplified by PCR from the cDNA of T47D breast cancer cell line using the primers 5′-CATATGGTCCTGGACCTCATGCTGGAGGCC-3’ and 5′-TGCTTCTCATGTGGAGGAAG-3′. PCR product was cloned in the pGEM T Easy Plasmid (Promega; Madison, WI) and then inserted into the pET22b plasmid (Novagen; Madison, WI) using the NdeI and EcoRI restriction sites. The resulting construct was used to transform competent Escherichia coli BL21 (DE3) cells (Novagen). Protein expression was carried out by growing in standard LB Broth medium until the OD 600-nm value reached 0.6. Induction of the recombinant protein was achieved by adding isopropyl β-
Preparation of Cell Lysates and Immunoblotting
Cell lysates were prepared in 10 mM Tris-HCl (pH 7.9), 150 mM NaCl, 1% NP40, and 1 mM phenylmethylsulfonyl fluoride. Nuclei were removed by centrifugation at 5000 X g for 5 min at 4C. Protein content was determined by the bicinchoninic acid method (BCA; Sigma) as recommended by the supplier. Cell lysates were resolved on SDS-polyacrylamide gel electrophoresis (Laemmli 1970) performed using a 10% gel under reducing conditions. Proteins were transferred to nitrocellulose sheets (Amersham; Aylesbury, UK) according to Towbin and coworkers (1979) at 60 V for 5 hr in 20 mM Tris-HCl, pH 8.3, 192 mM glycine, and 10% methanol. Residual protein-binding sites were blocked by incubation with 3% bovine serum albumin (BSA) in PBS overnight at 4C. The nitrocellulose was then incubated with the MAb anti-ppGalNAc-T6 for 3 hr at 37C. After three washes with PBS containing 0.1% Tween 20 and 1% BSA, the membrane was incubated for 1 hr at RT with goat anti-mouse immunoglobulin conjugated to peroxidase (Sigma) diluted in PBS 0.3% Tween 20 and 3% BSA. Reactions were developed by the enhanced chemoluminescence method (ECL; Amersham) according to the manufacturer's recommendations. The same procedure was performed omitting MAb anti-ppGalNA-T6 as a negative control.
Immunohistochemical and Immunocytochemical Analysis
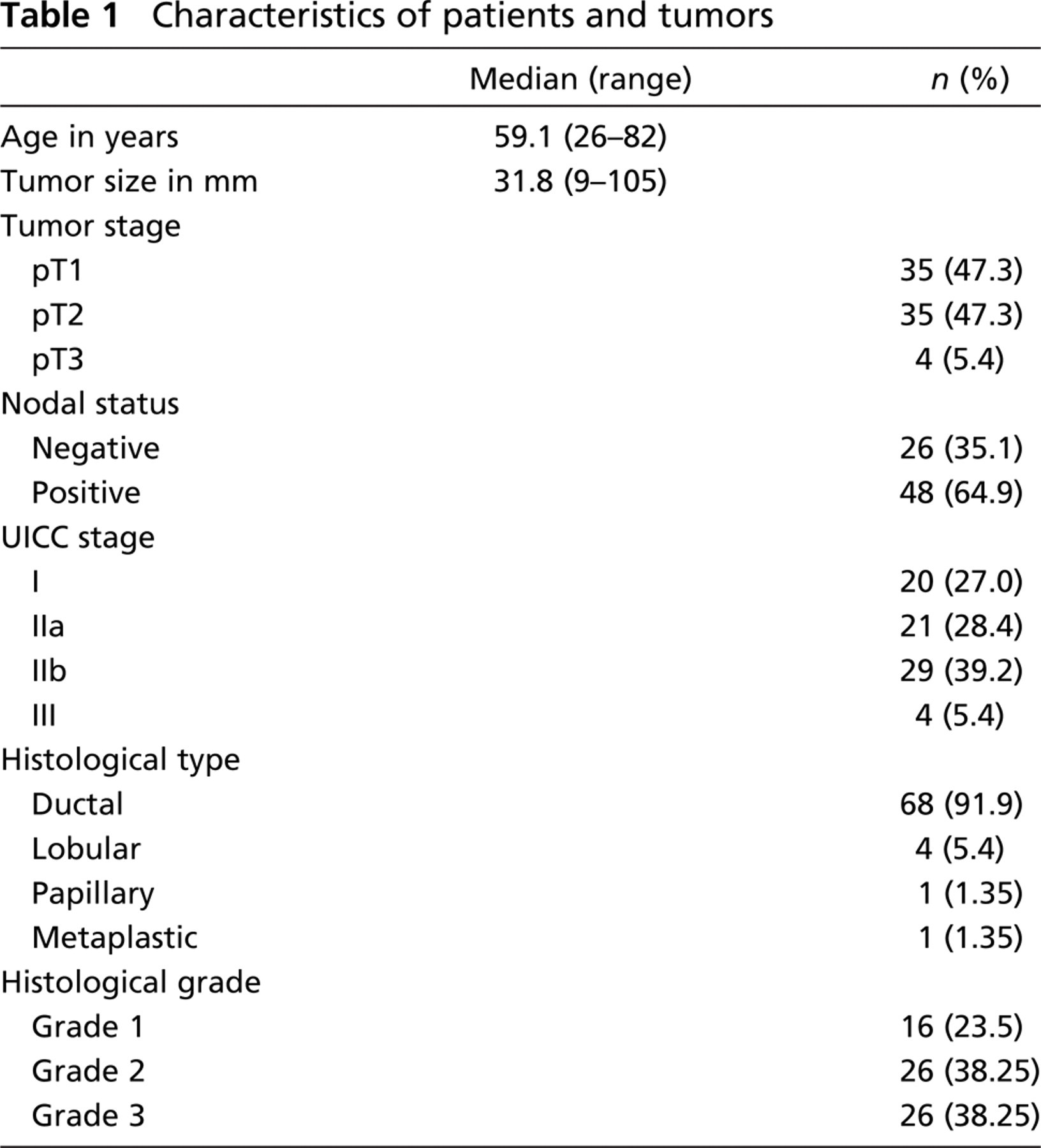
Immunohistochemical evaluation of ppGalNAc-T6 was performed in 94 randomly chosen patients who had undergone surgical treatment for a breast lump at Pereira Rossell Hospital (Montevideo, Uruguay) between 2002 and 2004 (74 patients with histopathological diagnosis of breast cancer and 20 patients with non-malignant diseases). The study was examined and approved by the ethical review board of the School of Medicine, Montevideo, Uruguay. Normal breast tissues were obtained from reduction mammoplasties (n = 5). All specimens were formalin-fixed (10%), paraffin-embedded tissues, and histological sections were subjected to hematoxylin and eosin staining (standard procedure) as well as immunohistochemical evaluation for ppGalNAc-T6 expression. All malignant tumors were conventionally classified by histological type and grading (Scarff–Bloom–Richardson system with Nottingham modification) (Frierson et al. 1995). For ppGalNAc-T6 immunostaining, the quenching of endogenous peroxidase activity was performed with 3% H2O2 in PBS for 20 min, then blocked with normal goat serum for 20 min (to decrease background staining). Anti-ppGalNAc-T6 MAb T6.3 was incubated overnight at 4C and peroxidase-conjugated goat anti-mouse polyvalent antibody (Sigma) was incubated for 60 min at RT. Reactions were revealed with diaminobenzidine (Sigma), washed in running water, counterstained in Mayer's hematoxylin, dehydrated in ethanol and xylene, and mounted. Between each step, sections were washed in PBS. For every assay, negative controls using PBS without primary antibody were included. The immunostaining frequency for each tumor was scored as follows: 0, for negative samples or <10% stained tumor tissue; 1, for samples stained between 10 and 39% of tumor tissue; 2, for samples stained between 40 and 79% of tumor tissue; and 3, for tumors with >80% of stained tumor tissue. Signal intensity was scored as strong (3), moderate (2), weak (1), and nul (0). Total immunostaining score results from the addition of both parameters. Scores were established jointly by four observers under a multi-head microscope. Clinicopathological information was masked to the observers.
Immunocytochemistry with MAb T6.3 was also performed in two human breast cancer cell lines (MCF-7 and T47D). Both cell lines were cultured in DMEM medium supplemented with 10% fetal bovine serum, 2 mM
Statistical Analysis
Correlation between ppGalNAc-T6 expression and various clinicopathological factors were analyzed using Fisher's exact test. Univariate analysis of disease-free survival was performed with the use of log-rank test; p values <0.05 were statistically significant.
Results
Generation and Characterization of Monoclonal Antibodies Anti-ppGalNAc-T6
A panel of MAbs specific for ppGalNAc-T6 was generated from mice immunized with a KLH-conjugated ppGalNAc-T6 peptide. Selection of specific hybridomas was performed by ELISA, screening against BSA-conjugated ppGalNAc-T6 peptide. One of the MAbs, T6.3, strongly reactive against the synthetic peptide, was used for further characterization. Considering that ppGalNAc-T6 expression in pancreas and placenta was previously reported (Bennett et al. 1999), we performed a preliminary immunohistochemical evaluation using MAb T6.3 on these tissues. A strong staining was observed in pancreas (Figure 1), where ppGalNAc-T6 was preponderantly detected in the apical perinuclear region, as expected for a glycosyltransferase localized in Golgi apparatus (Rottger et al. 1998) (Figure 1). In first-trimester placenta samples, ppGalNAc-T6 immunostaining was observed in subepithelial mesenchymal cells and in the syncytiotrophoblastic cells, whereas cytotrophoblastic cells were negative (Figures 1C and 1D). In Western blot, MAb T6.3 recognized a protein with an apparent molecular weight of 71 kDa in placental extracts (Figure 2) (expected size for native ppGalNAc-T6; Bennett et al. 1999), as well as the recombinant ppGalNAc-T6 expressed in E. coli (Figure 2). Considering the high homology in amino acid sequence between ppGalNAc-T3 and ppGalNAc-T6, both expressed in pancreas and placenta (Bennett et al. 1996), we evaluated more accurately the specificity of MAb T6.3 using two other synthetic peptides corresponding to amino acid sequences included in the peptide used as immunogen: EAQQTLFSINQS (a specific motif of ppGalNAc-T6 without any homology with ppGalNAc-T3 sequence) and CLPGFYTPAELKP (which has homologies with ppGalNAc-T3). MAb T6.3 reactivity was abolished by preincubation with the synthetic peptide EAQQTLFSINQS and evaluated by a competitive ELISA assay (Figure 2) and by immunohistochemical evaluation performed on pancreas and placental sections (data not shown). In contrast, the synthetic peptide CLPGFYTPAELKP was unable to inhibit MAb T6.3 binding in all assays performed. In addition, MAb T6.3 was unreactive in colonic normal mucosa by immunohistochemistry (data not shown), whereas ppGalNAc-T3 is strongly expressed in this tissue (Shibao et al. 2002).
ppGalNAc-T6 Expression in Breast
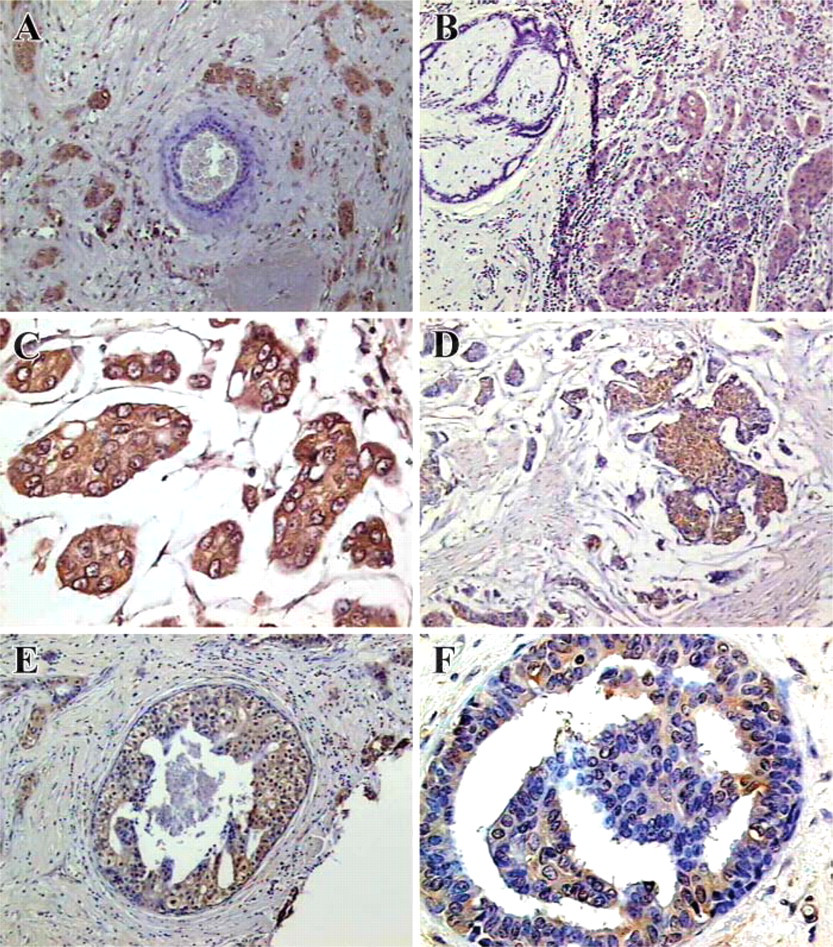
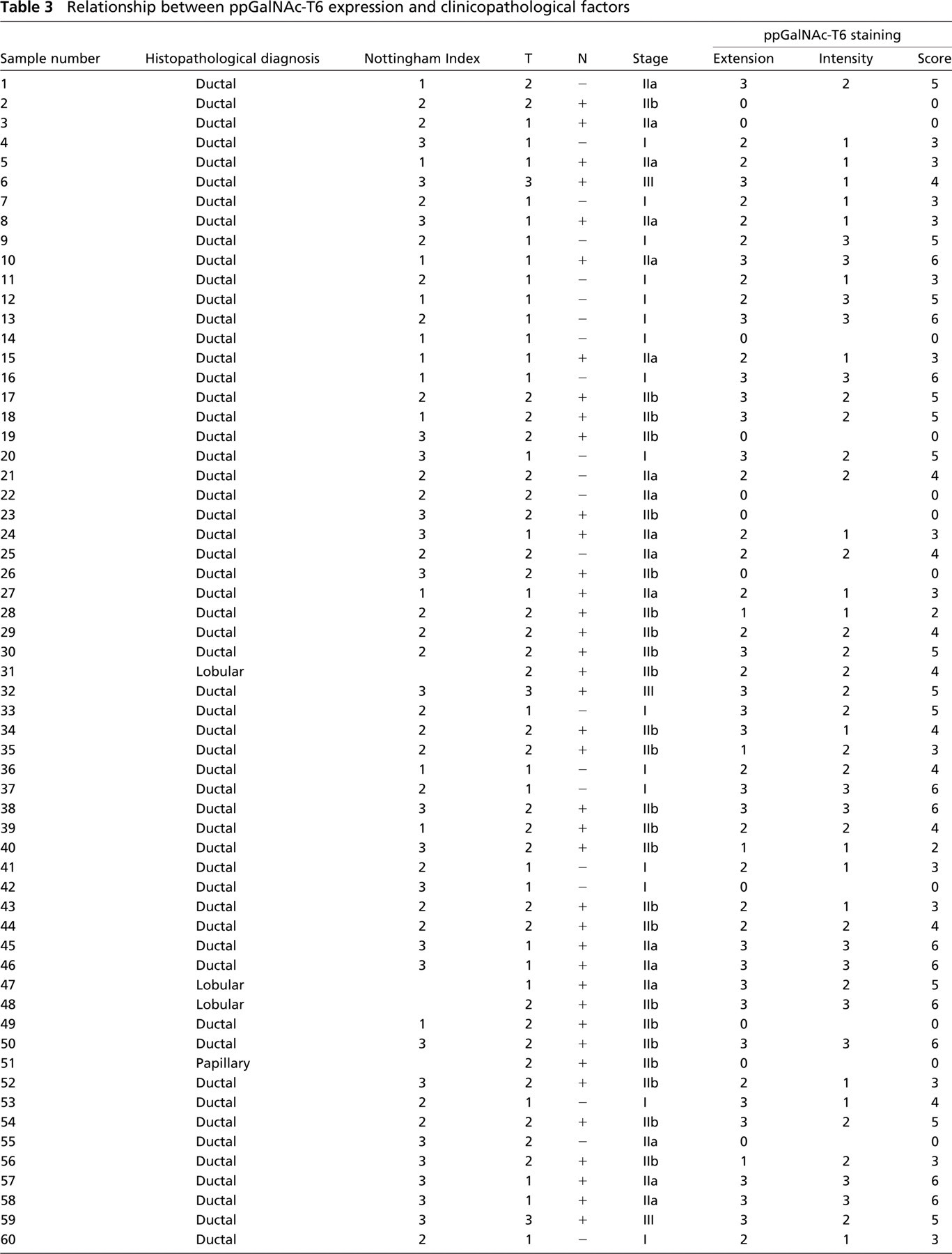
In immunocytochemical studies, MAb T6.3 was reactive with both MCF-7 and T47D breast cancer cell lines (data not shown). These results were in agreement with ppGalNAc-T6 expression revealed by RT-PCR in these cells lines (Freire T, et al., unpublished data). MAb T6.3 immunostaining was also evaluated in 74 primary tumors from breast cancer patients, 20 specimens from non-malignant breast diseases, and 5 normal breast tissues. Characteristics of breast cancer patients and tumors are indicated in Table 1. The majority of breast cancers were invasive ductal carcinoma and about one third of the patients were node negative. ppGalNAc-T6 was expressed in 60/74 (81%) invasive cancers (Table 2). Interestingly, MAb T6.3 always showed a diffuse cytoplasmic staining pattern (Figures 3A–3D). Microwave treatment strongly reduced immunostaining. The relationship between ppGalNAc-T6 expression and clinicopathological factors is shown in Table 3. T1 tumors were more frequently positive (32/35; 91.4%) than T2 tumors (24/35; 68.6%) (difference statistically significant, p = 0.04) and more frequently in N0 patients (22/26; 84.6%) than in those with metastatic lymph nodes (38/48; 79.2%) (difference not statistically significant) (Figure 4). Regarding disease stage, ppGalNAc-T6 was expressed in 18/20 (90%) tumors from patient stage I, in 18/21 (85.7%) stage IIa, and in 20/29 (69%) stage IIb (differences not statistically significant). No correlation was found between ppGalNAc-T6 expression and tumor grading, either between tumor staining intensity or the pathological parameter evaluated. Adjacent ductal carcinoma in situ (DCIS) was observed in 23/74 invasive cancers, and 21 (91.3%) showed ppGalNAc-T6 expression (Figures 3E and 3F).
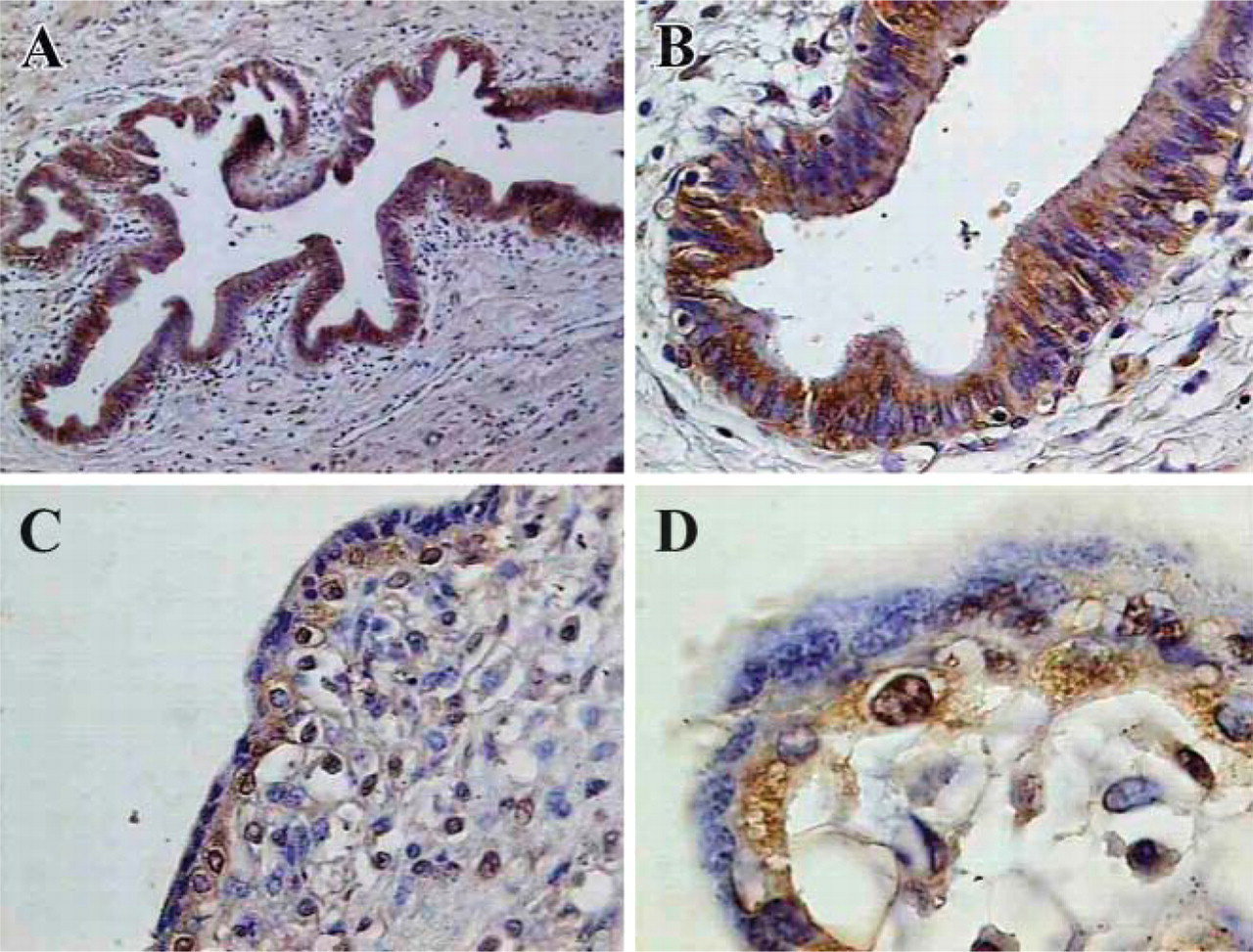
Immunohistochemical evaluation of ppGalNAc-T6 in pancreas and placenta. (
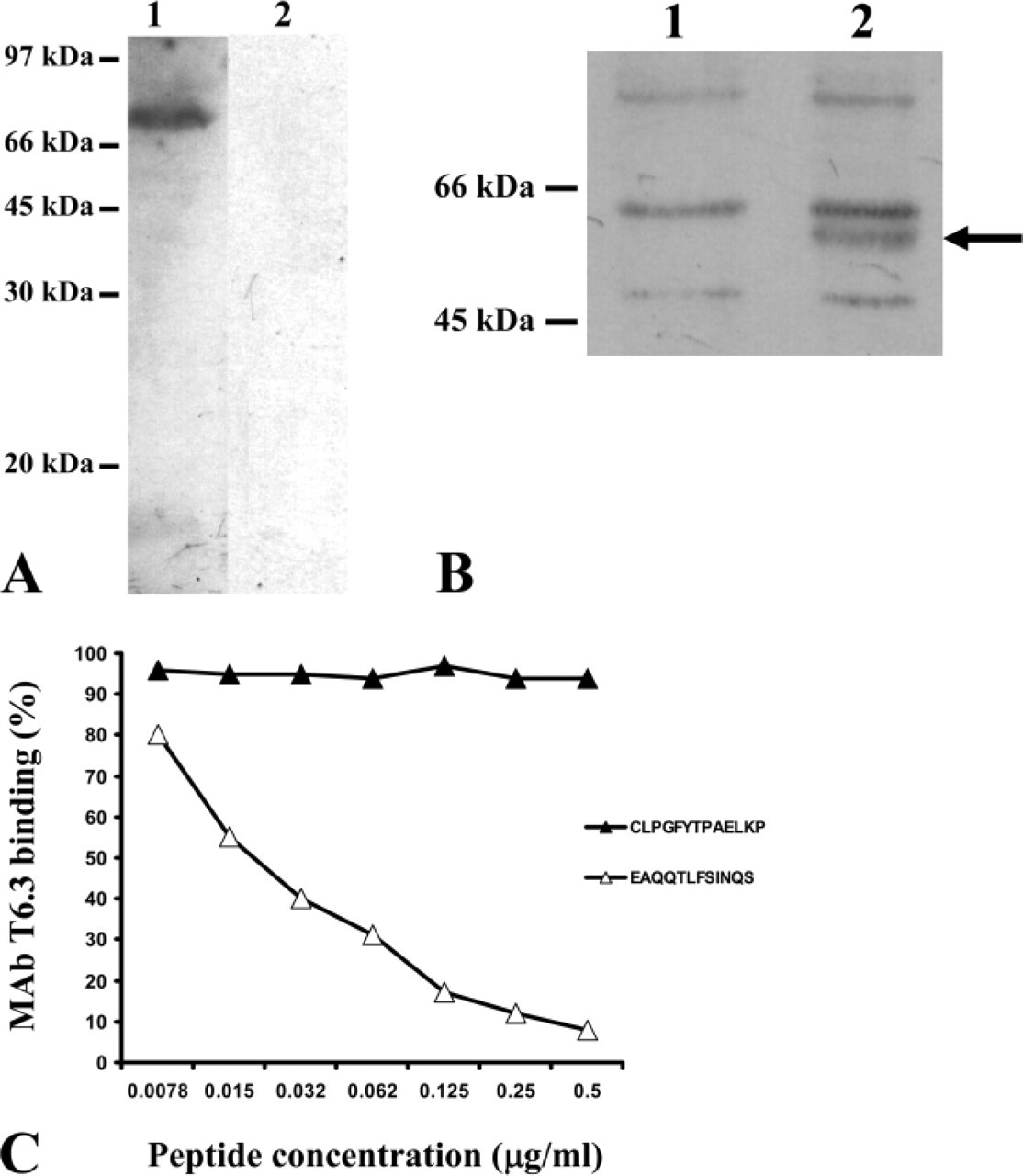
Characterization of anti-ppGalNAc-T6 MAb T6.3 specificity. (
ppGalNAc-T6 Expression in Normal Breast and Non-malignant Diseases
Morphologically normal epithelium adjacent to the pathological tissue was evaluated in 36 cases. Eleven (30.5%) were ppGalNAc-T6 positive (4/11 showed a strong immunostain and 7/11 immunostained weakly), whereas 25/36 (69.4%) were negative. All five normal breast tissues from reduction mammoplasties were also negative. In contrast to the frequent expression observed in breast cancers, ppGalNAc-T6 was rarely detected in non-malignant breast diseases. Only 4/20 (20%) showed ppGalNAc-T6 expression (Figures 5A–5D), distributed as follows: 2/2 sclerosing adenosis, 2/13 fibroadenoma, 0/3 fibrocystic disease, 0/1 galactocele, and 0/1 benign Phyllodes. The staining pattern was similar to that observed in invasive carcinomas.
Myoepithelial cells were clearly identified in normal lobules and ducts in 46 patients (29 cases of breast cancer and 17 samples with benign breast diseases). Interestingly, these myoepithelial cells were ppGalNAc-T6 positive in 13/29 (44.8%) breast cancer patients as well as in 4/17 (23.5%) samples with non-malignant diseases (sclerosing adenosis and fibroadenoma with focal hyperplasia). The staining was strong when these cells were associated with breast cancer (Figures 5E and 5F).
Discussion
Protein O-glycosylation is deregulated in breast cancer cells, leading to the accumulation of simple mucin-type tumor-associated antigens (Burchell et al. 2001). Because the initiation of mucin-type O-linked protein glycosylation is catalyzed and regulated by a family of ppGalNAc-Ts, we can hypothesize that the expression of these enzymes may be useful for diagnosis of breast cancer. In the immunohistochemical study presented here, we describe for the first time the expression of ppGalNAc-T6 protein in breast cancer. Our results state that ppGalNAc-T6 is not detected in normal breast tissues obtained from healthy women. It is expressed in only 4/20 samples (20%) of benign breast disease, whereas it is expressed in most breast carcinomas (60/74, 81%). These results are in agreement with recent mRNA data obtained in our laboratory using RT-PCR, where ppGalNAc-T6 expression mRNA was demonstrated in 22/25 breast tumors (Freire T, et al., unpublished data). Human GALNT6 gene, located on chromosome 12q13, contains 10 exons of 1869 base pairs that encode a type-II transmembrane protein (Bennett et al. 1999). This gene, highly similar to the one that encodes ppGalNAc-T3 (Bennett et al. 1996), is expressed in a restricted pattern, being mainly in normal placenta, trachea, brain, pancreas, and fibroblast cells (Bennett et al. 1999), as well as in conjunctival goblet cells (Argüeso et al. 2003). Protein expression of ppGalNAc-T6 in various organs has not been investigated thus far because of the lack of an appropriate antibody able to recognize this enzyme on formalin-fixed tissues. Considering the high sequence homology among different ppGalNAc-Ts, we have produced a MAb specific for ppGalNAc-T6, using a strategy based upon the immunization of mice with a synthetic peptide conjugated to KLH. The peptide sequence contains a motif partially shared by ppGalNAc-T6 and ppGalNAc-T3 (CLPGFYTPAELKP) and a motif expressed by ppGalNAc-T6 only (EAQQTLFSINQS). We selected the MAb T6.3 because its binding activity was inhibited by the peptide EAQQTLFSINQS and not by the peptide CLPGFYTPAELKP. The specificity of this antibody for ppGalNAc-T6 was confirmed by (a) Western blot using the recombinant ppGalNAc-T6, (b) its reactivity on tissues where this enzyme is expressed (pancreas and placenta), and (c) its non-reactivity on cells that normally express ppGalNAc-T3 but not ppGalNAc-T6 (normal colon).
Characteristics of patients and tumors
To our knowledge, ppGalNAc-T6 is the second member of the ppGalNAc-T family that is overexpressed in breast cancer. Previously, Nomoto et al. (1999) showed that ppGalNAc-T3 mRNA is expressed in cell lines derived from mammary gland adenocarcinomas. They observed by immunohistochemistry ppGalNAc-T3 expression in breast tumor tissues but not in normal mammary epithelium. However, this observation was not definitive because in that study a rabbit polyclonal antiserum raised against the synthetic peptide
Our results show a statistically significant association of ppGalNAc-T6 expression with T1 tumor stage but no correlation with nodal status or histological grade. This fact, together with the observation that ppGalNAc-T6 was strongly expressed in sclerosing adenosis and in most DCIS, suggests that deregulation of GALNT6 gene could be an early event during human breast carcinogenesis. The exact role played by the overexpression of ppGalNAc-T6 in breast carcinogenesis is not yet clear. Taking into account that a relationship between altered O-glycosylation and malignancy has been reported and that the ppGalNAc-T enzyme family plays a key role in O-glycosylation regulation (displaying site specificity and different kinetic properties toward O-glycosylation sites in mucins, such as the MUC1 tandem repeat) (Wandall et al. 1997; Hassan et al. 2000), its abnormal expression in breast cancer could determine changes in cellular functions such as adhesion, invasion, and metastasis. It will be important to evaluate the relationship between ppGalNAc-T6 and mucin-type cancer-associated antigen expression (such as Tn, T, and sialyl-Tn antigens) in breast tissues. These studies may also take into consideration the expression of other ppGalNAc-Ts (Bennett et al. 1998; Mandel et al. 1999; Schwientek et al. 2002), as well as core β1Gal-T and ST6GalNAc-I (Marcos et al. 2004). Marcos et al. (2003) observed that ppGalNAc-Ts expression was associated with different glycosylation forms of the MUC1 tandem repeat in gastric carcinoma cell lines.
Analysis of ppGalNAc-T6 a expression level in human breast tissues
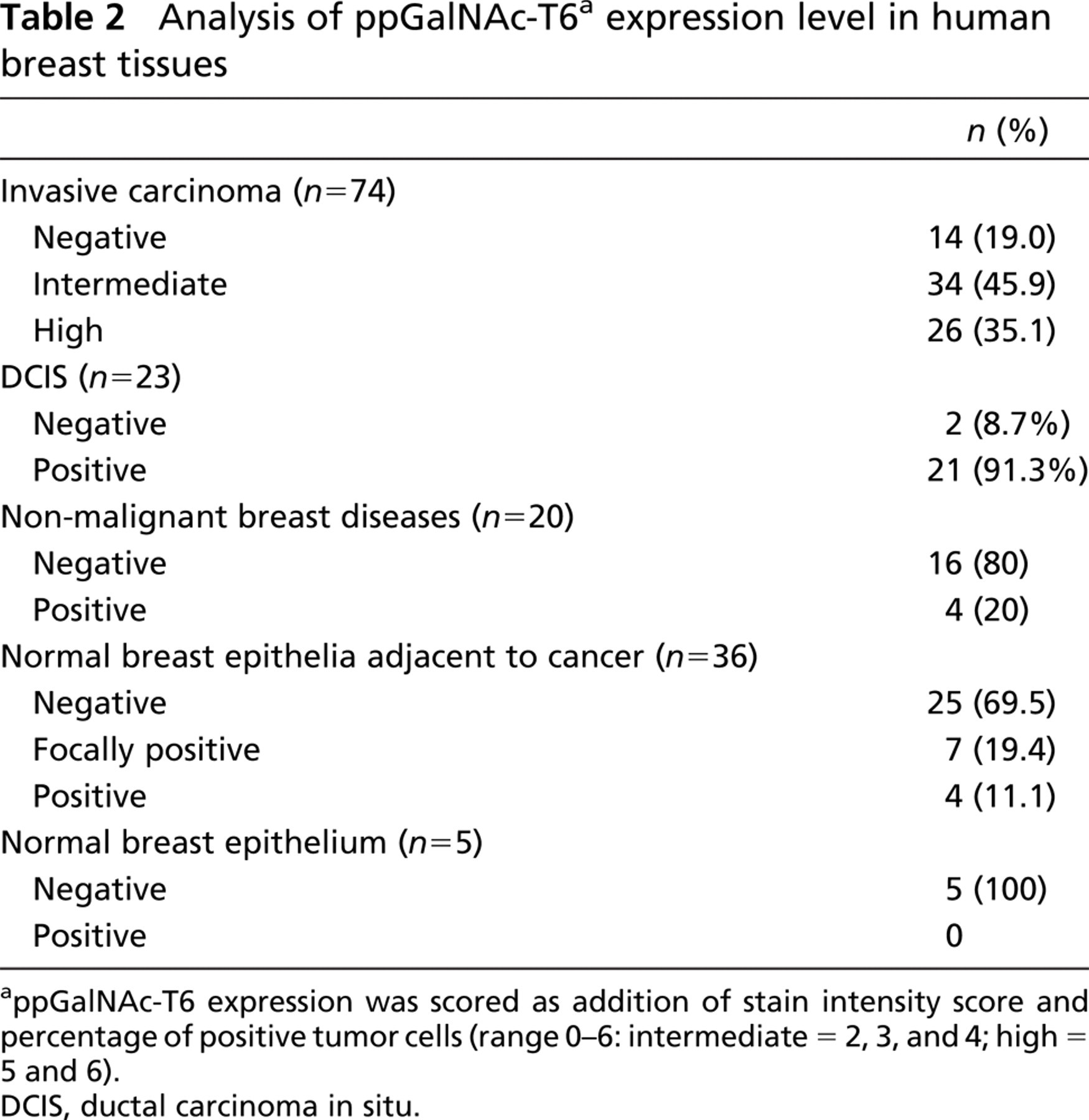
appGalNAc-T6 expression was scored as addition of stain intensity score and percentage of positive tumor cells (range 0–6: intermediate = 2, 3, and 4; high = 5 and 6).
DCIS, ductal carcinoma in situ.
ppGalNAc-T6 expression in breast cancer. (
The immunostaining pattern usually observed for glycosyltransferases (localized in the Golgi apparatus) corresponds predominantly to the perinuclear area (Yamamoto et al. 1999; Kohsaki et al. 2000). We observed this pattern for ppGalNAc-T6 staining on pancreatic tissues. In contrast, a diffuse-type localization of immunoreactivity was observed in the cytoplasm of breast tissues (benign diseases and cancer). One reason for the diffuse ppGalNAc-T6 immunostaining could probably be suboptimal tissue fixation, a commonly observed phenomenon in immersion-fixed surgical specimens. However, a similar diffuse cytoplasm distribution pattern has also been observed for other glycosyltransferases in cancer cells, for example, for a galactosyl- and sialyltransferase in cultured hepatoma cells (Taatjes et al. 1987), for β1,4-galactosyltransferase in endometrial cancer (Kubushiro et al. 1999), and for ppGalNAc-T3 in colorectal (Shibao et al. 2002) and gallbladder carcinomas (Miyahara et al. 2004). Enzyme localization is an important determinant of the carbohydrate repertoire expressed on the cell surface, and their relative positions are known to govern, in part, the structures of glycans produced by the cell (Grabenhorst and Conradt 1999). To explain the diffuse-type immunostaining pattern of ppGalNAc-T6 in breast tissues, a detailed study is necessary to determine the subcellular localization of this enzyme (e.g., assessing ppGalNAc-T6 subcellular immunostaining using organelle analysis applying confocal laser scanning microscopy, immunoelectron microscopy, etc).
Relationship between ppGalNAc-T6 expression and clinicopathological factors
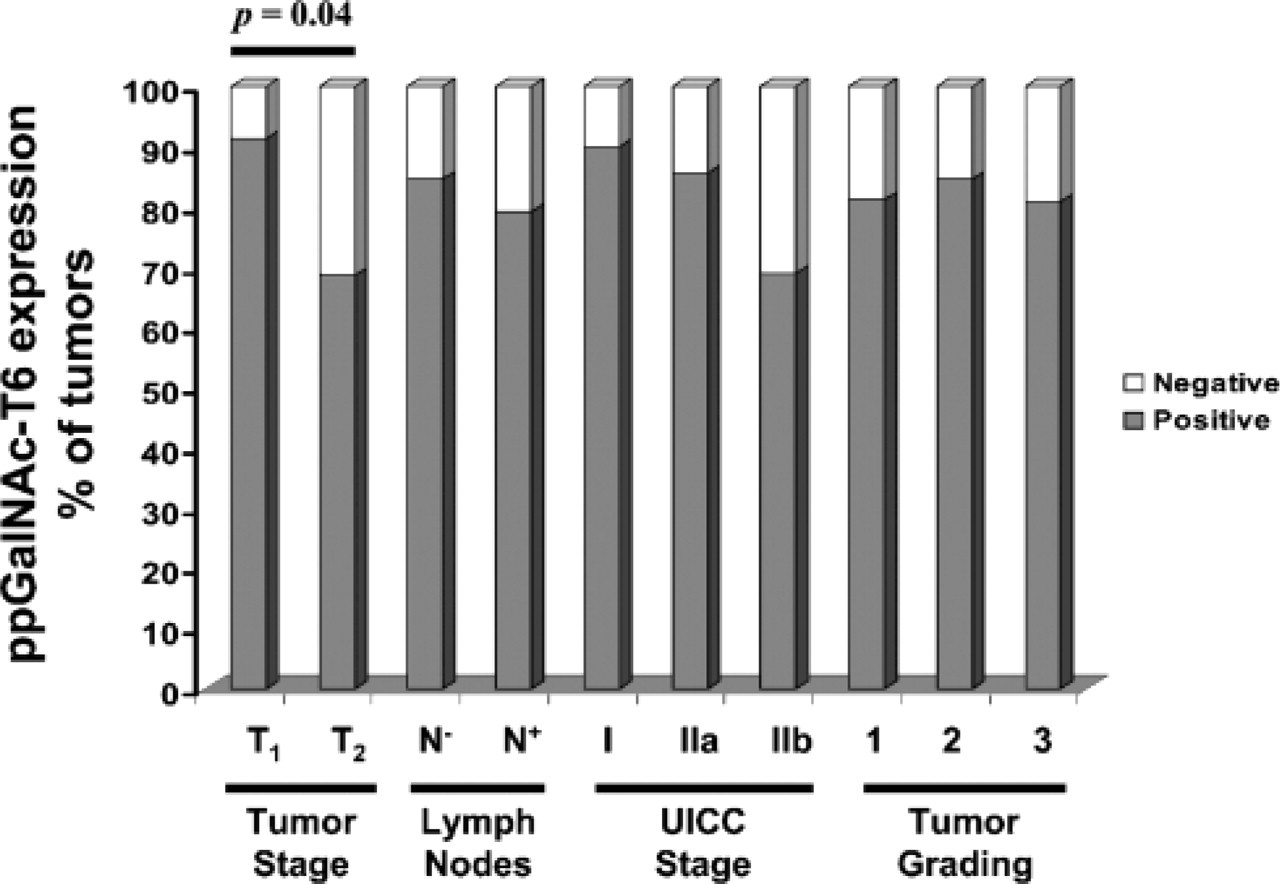
Relationship between ppGalNAc-T6 expression and different clinicopathological parameters.
We observed that myoepithelial cells strongly express ppGalNAc-T6 in some patients. This is interesting because myoepithelial cells can be considered as ‘natural tumor suppressors’ (Sternlicht and Barsky 1997) implicated in breast tumor progression, particularly in the transition of in situ to invasive carcinomas. Numerous in vitro and in vivo studies using diverse experimental systems have demonstrated that the growth (Xiao et al. 1999), polarity (Gudjonsson et al. 2002), angiogenesis (Alpaugh et al. 2000), and invasive behavior of breast cancer cells can be modulated by myoepithelial cells. Analysis of myoepithelial marker expression remains a commonly used approach to distinguish between benign and malignant tumors or to detect stromal invasion (Yaziji et al. 2000). Little information is available regarding the glycobiology of myoepithelial cells. Several questions are raised now about the biological significance of ppGalNAc-T6 in these cells, particularly related to the regulation of GALNT6 gene expression, its influence on myoepithelial cell phenotype, and its relation to normal and transformed ductal cells.
In summary, we have presented evidence that ppGalNAc-T6 is a novel immunohistochemical marker for breast carcinoma cells found in most ductal carcinomas but not in normal breast epithelium. To extend our observations, a follow-up study of a larger number of cases is necessary to determine the potential clinical value of this marker. Further studies are also required to determine the biological role of ppGalNAc-T6 in breast tumor development, growth, and invasion.
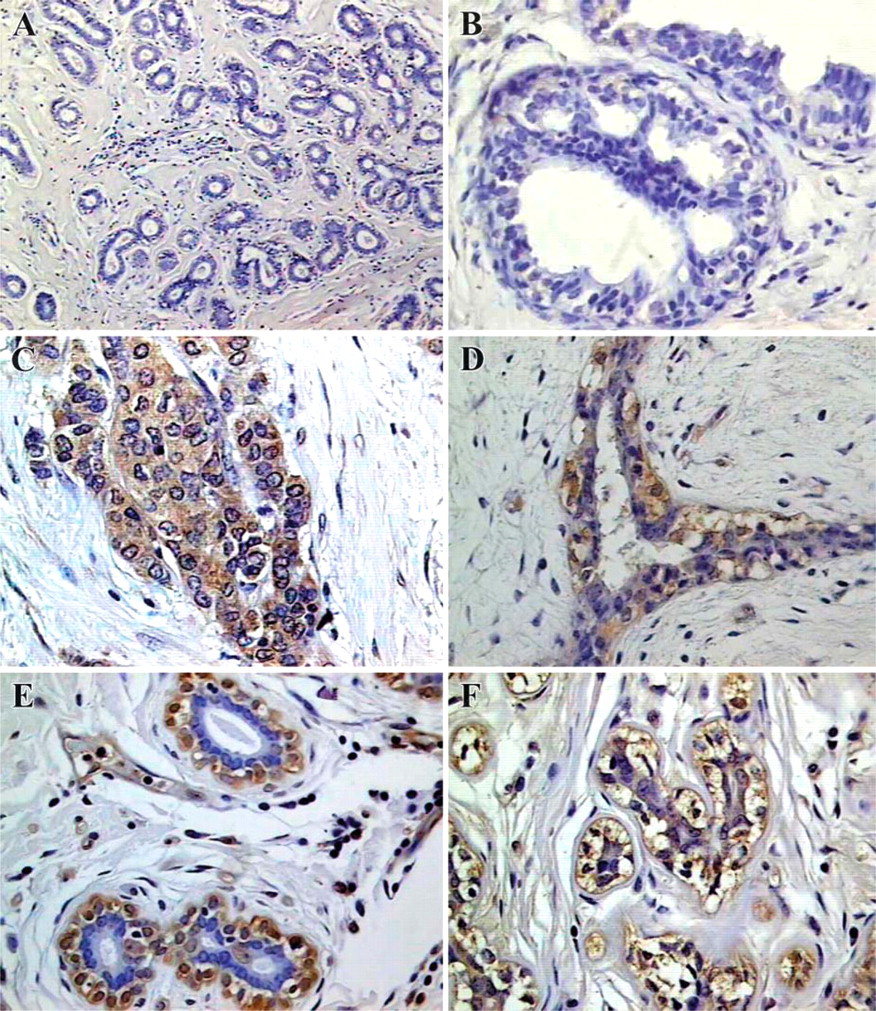
Immunohistochemical evaluation of ppGalNAc-T6 in non-malignant breast tissues. (
Footnotes
Acknowledgements
This work was partially supported by a grant from ECOS (France–Uruguay Program #03U02) and by a grant from Comisión Honoraria de Lucha Contra el Cáncer (Uruguay).
We thank Dr. Enrique Barrios for statistical analysis.
