Abstract
Introduction
Nocardiosis is reported in both captive and free-ranging marine mammals. Current
literature suggests that the systemic form is the most common presentation in cetaceans.
15,
39,
41,
45
There is a single report of pulmonary nocardiosis in a Pacific bottlenose dolphin
(
We report the results of postmortem examinations and describe the distribution of
histologic lesions in 8 hooded seals (
Materials and Methods
Case selection
Facilities in North America involved in diagnostic investigations of marine mammals were queried for suspected or confirmed cases of nocardiosis mortality in marine mammals. These facilities included zoos and aquaria holding marine mammals, facilities that respond to stranded free-ranging marine mammals, and diagnostic services that receive samples from these facilities.
Cases selected for inclusion and further evaluation were limited to those with
complete gross and histologic results and culture or polymerase chain reaction (PCR)
confirmation of nocardiosis. Cases that reported pyogranulomatous or granulomatous
inflammation but lacked culture or PCR confirmation were included after molecular
evaluations confirmed the presence of
Nine pinnipeds from 8 institutions met the criteria for inclusion. These consisted of
8 hooded seals and a leopard seal. All were free-ranging animals. Ten cetaceans from
7 institutions met the criteria for inclusion. These consisted of 3 species from wild
and captive populations including Atlantic bottlenose dolphin (
Histopathology
Tissues available for histologic review varied from animal to animal and included brain, skin, heart, lung, liver, kidney, spleen, lymph nodes, adrenal, stomach, intestines, thyroid, thymus, and pancreas. Tissues were routinely embedded in paraffin, sectioned at 4–7 µm, stained with hematoxylin and eosin (HE), and examined microscopically at the facility of origin. Slides were examined by 2 of the authors (LB and JAS) to assure continuity of descriptions.
To enhance visualization of the organisms, tissue sections were stained with Brown and Brenn (BB), Ziehl-Neelsen (ZN), Fite Faraco (Fites), Coates modified Fite stain (modified Fites), and Grocott's methenamine silver (GMS) according to published protocols. 1
Culture
Diagnostic cultures were performed for 6 pinnipeds and 7 cetaceans. Material was collected at necropsy and inoculated onto blood agar plates and incubated at 37°C under aerobic or microaerophilic conditions for periods ranging from 2 to 10 days.
Polymerase chain reaction
Formalin-fixed, paraffin-embedded tissues (FFPT) of pinniped Nos. 2, 4, and 6–9 and
cetacean Nos. 10–14 and 18 were processed for detection of
Results
Animals
Nine pinniped cases from 1974 to 2007 met the study inclusion criteria. The cases were limited to 8 hooded seals and a single leopard seal. Age, species, and sex distribution can be found in Table 1. The pinnipeds were all free-ranging animals in rehabilitation for periods ranging from 5 days to greater than 1 year before death. The affected hooded seals were stranded along the east coast of the United States—Florida (case Nos. 1 and 8), Connecticut (case No. 2), Virginia (case No. 4), North Carolina (case No. 6)—California (case No. 3), Puerto Rico (case No. 7), and the port of Montreal (case No. 5). The leopard seal (case No. 9) was stranded in Hawke's Bay on the north Island of New Zealand. The animal that was in hand only 5 days demonstrated both chronic and acute lesions, suggesting infection before entering rehabilitation. Three animals demonstrated signs only after being in rehabilitation for at least 3 weeks (case Nos. 1, 3, and 7). Six animals demonstrated signs related to nocardiosis on entry into rehabilitation (case Nos. 2, 4, 5, 6, 8, and 9) and were treated for a period of more than 1 year before death. One hooded seal was euthanatized (case No. 6); the other animals died spontaneously.
Distribution and severity of histologic
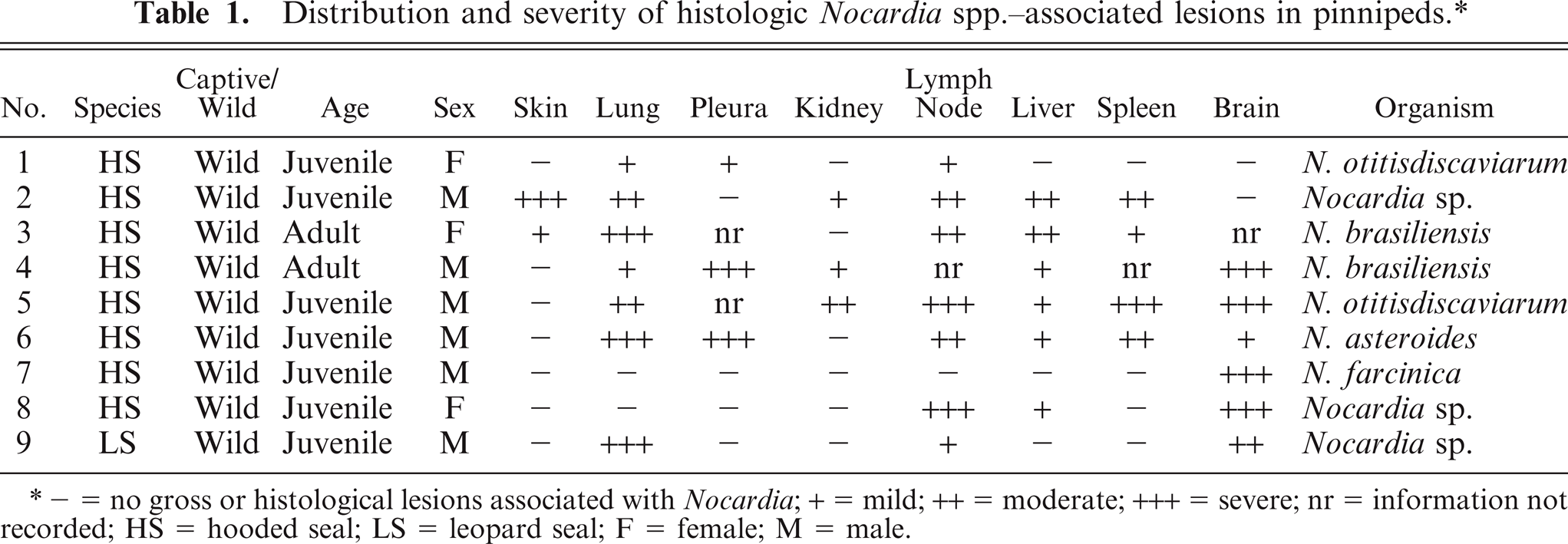
- = no gross or histological lesions associated with
Ten cetaceans from 1977 to 2007 met the criteria for inclusion. The species distribution included 3 Atlantic bottlenose dolphins, 4 beluga whales, and 2 killer whales. Age, species, and sex distribution of these animals can be found in Table 2. Eight cetaceans were from North American aquaria, and 2 were free-ranging animals stranded in Florida (case Nos. 10 and 11). One of the free-ranging dolphins was collected dead on the beach and thus must have become infected while in the wild. The second was in rehabilitation for 14 days (case No. 11). The duration of illness of the captive cetaceans ranged from 14 days to almost 2 years. One beluga whale was euthanatized because of the severity and progression of the disease (case No. 18); the other animals died spontaneously.
Distribution and severity of histologic
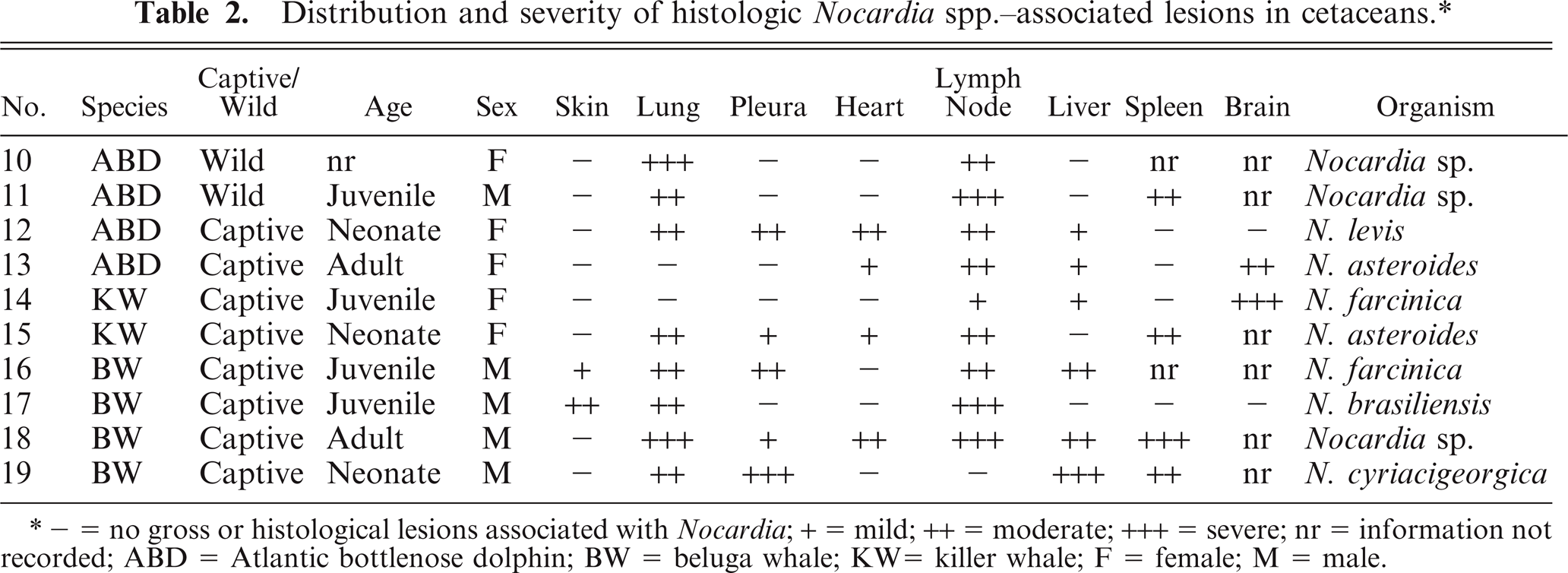
- = no gross or histological lesions associated with
Pathologic findings
Thoracic disease as a component of systemic infection was the primary presentation in hooded seals. Pyothorax was present in 3 hooded seals (Nos. 1, 4, and 6). Single or multiple pulmonary abscesses or pyogranulomas ranging from 2 mm to 10 cm were seen in 5 hooded seals (Nos. 2–6). All seals with pulmonary lesions had thoracic lymph node abscesses (Figs. 1, 2). Four of these 5 hooded seals had generalized lymph node abscesses (Nos. 2, 3, 5, and 6). Two hooded seals had pleuritis (Nos. 1 and 6), 2 had brain abscesses (Nos. 7 and 8), and 2 had abscesses or pyogranulomas in skin or subcutaneous tissues (Nos. 2 and 3; Fig. 3). The cervical skin lesion in hooded seal No. 2 extended diffusely into the musculature. In hooded seal No. 3, a lesion in the right nasal cavity extended into the surrounding muscle and bone. Pyogranulomas were found grossly in liver (Nos. 2, 4, and 5) and spleen (Nos. 5 and 6) of 4 animals. Three hooded seals were in good nutritional condition at necropsy (Nos. 3, 4 and 8) and 2 were thin or emaciated (Nos. 2 and 5). Distribution and severity of relevant microscopically recognized lesions in pinnipeds are summarized in Table 1. Six hooded seal cases had concurrent conditions, including injuries from shark bites (Nos. 6 and 7), bacterial valvular endocarditis (No. 2), a traumatic lesion in oral cavity (No. 4), and parasitic ulcers in the gastrointestinal tract (Nos. 2 and 3). One hooded seal had an eye removed because of glaucoma 2 weeks before death (No. 8). None of these animals had lesions suggestive of concurrent immune suppression or morbillivirus infection.
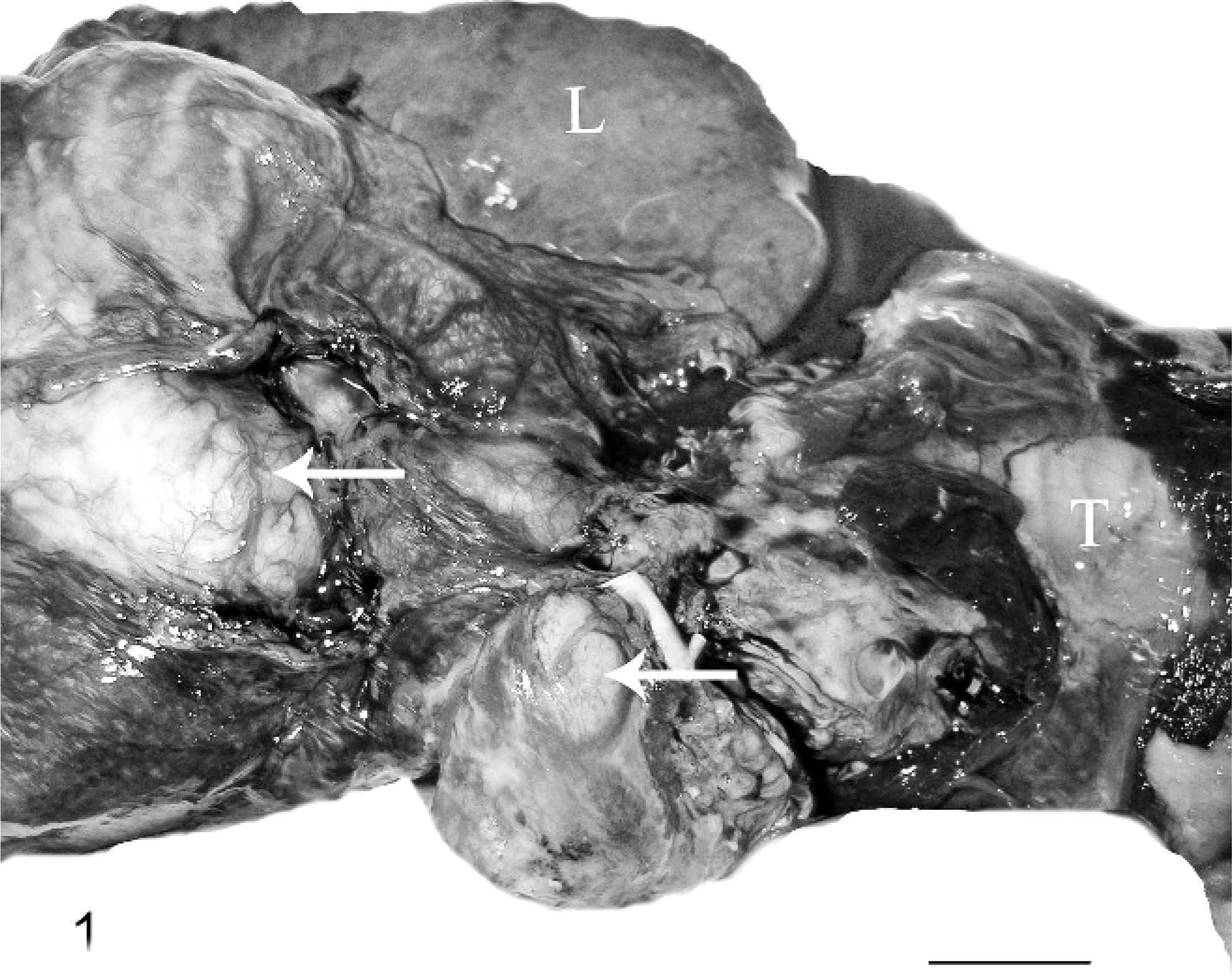
Thoracic viscera; hooded seal No. 1. The tracheobronchial lymph nodes (arrows) of this hooded seal with nocardiosis are massively enlarged and abscessed.
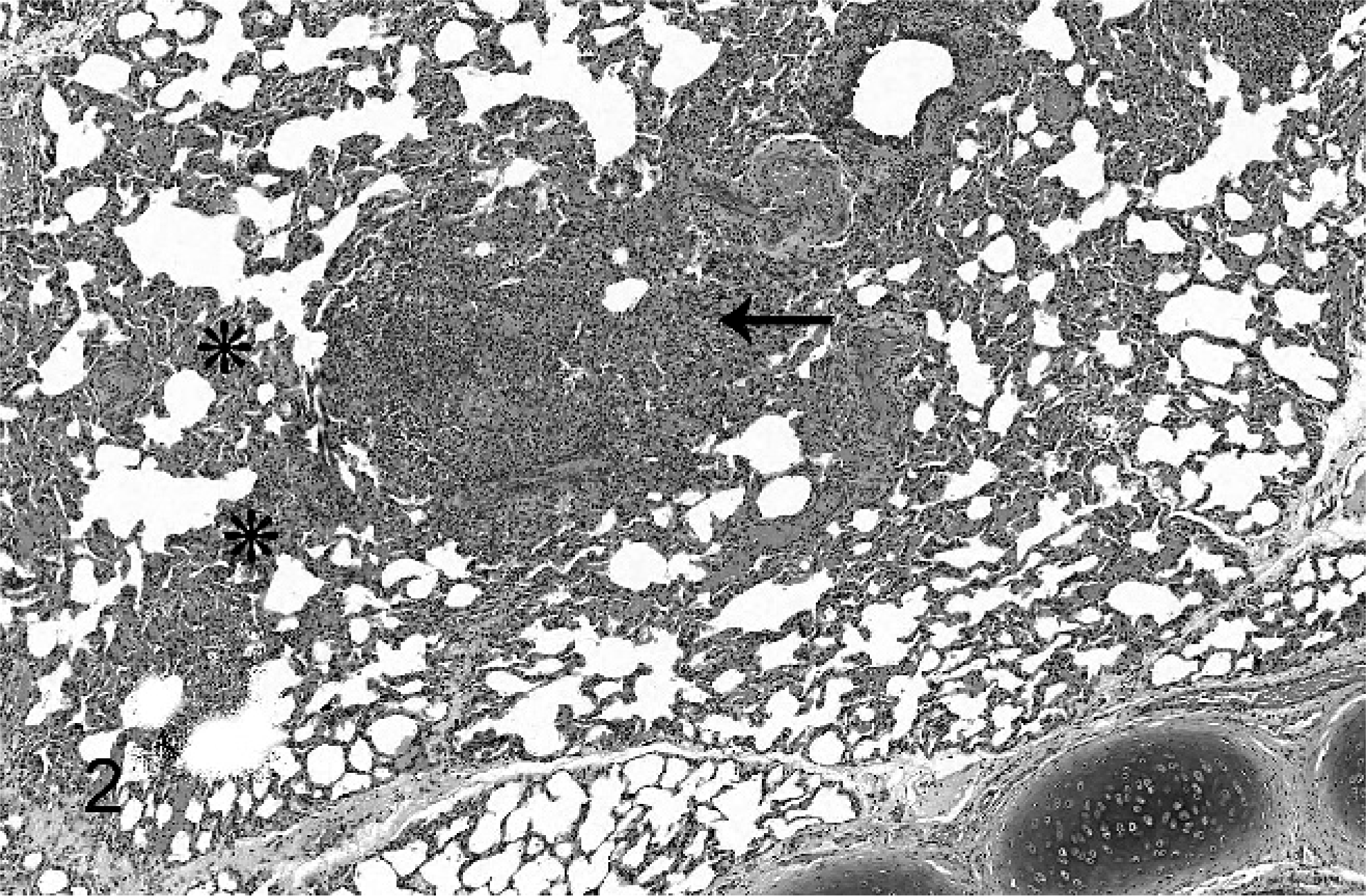
Lung; alveolar septa and a bronchiole from a hooded seal with nocardiosis. A granuloma (arrow). The alveolar septa are locally expanded with macrophages and lymphocytes (asterisks). No filamentous bacteria are seen with this stain. HE.
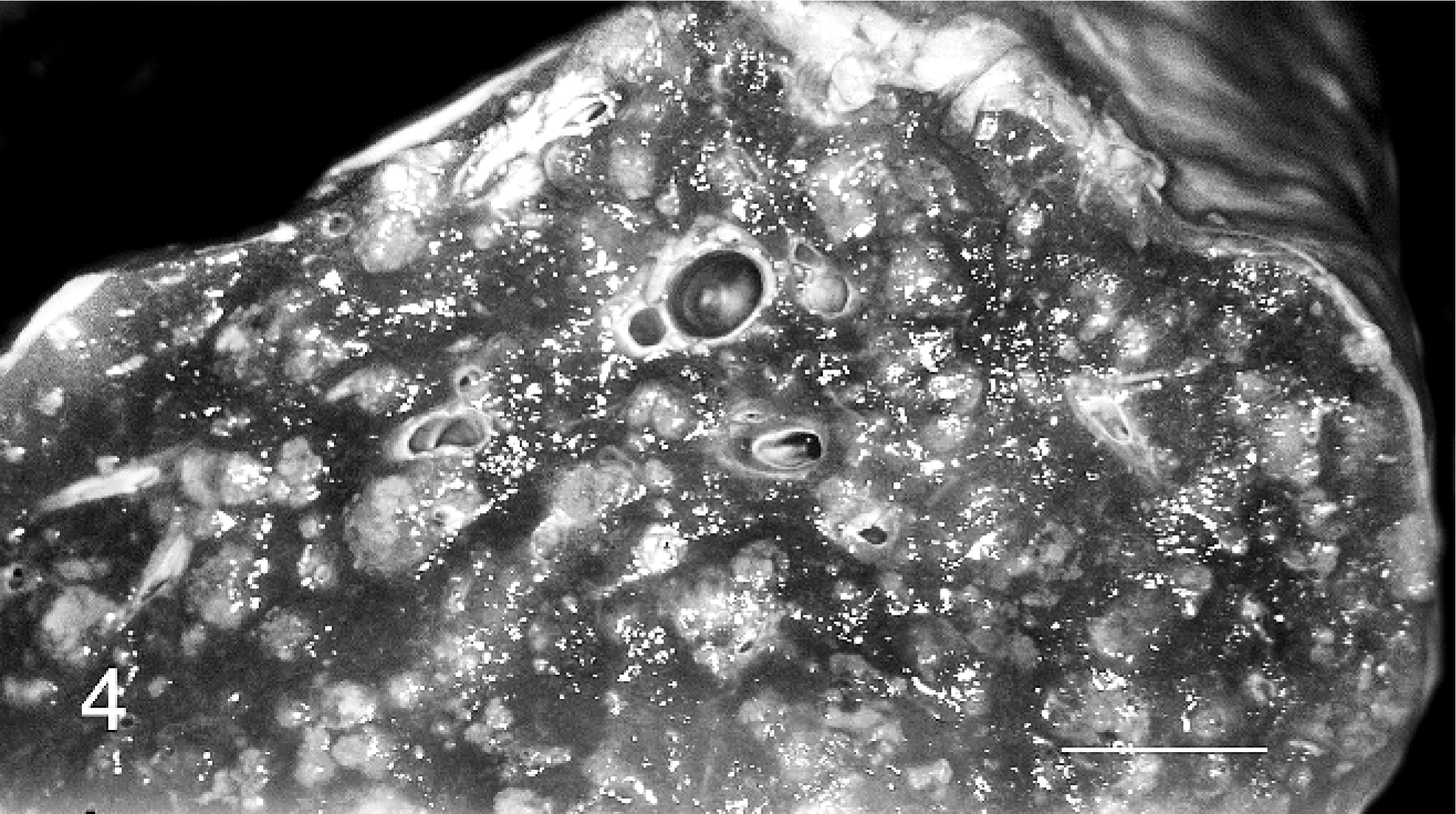
Skin; hooded seal No 2. Granulomatous focus of necrotic and viable neutrophils and macrophages in dermis with indistinct border. BB.
Gross and histologic evaluations in the leopard seal revealed multiple pyogranulomas in lung, thoracic lymph nodes, adrenals, and brain.
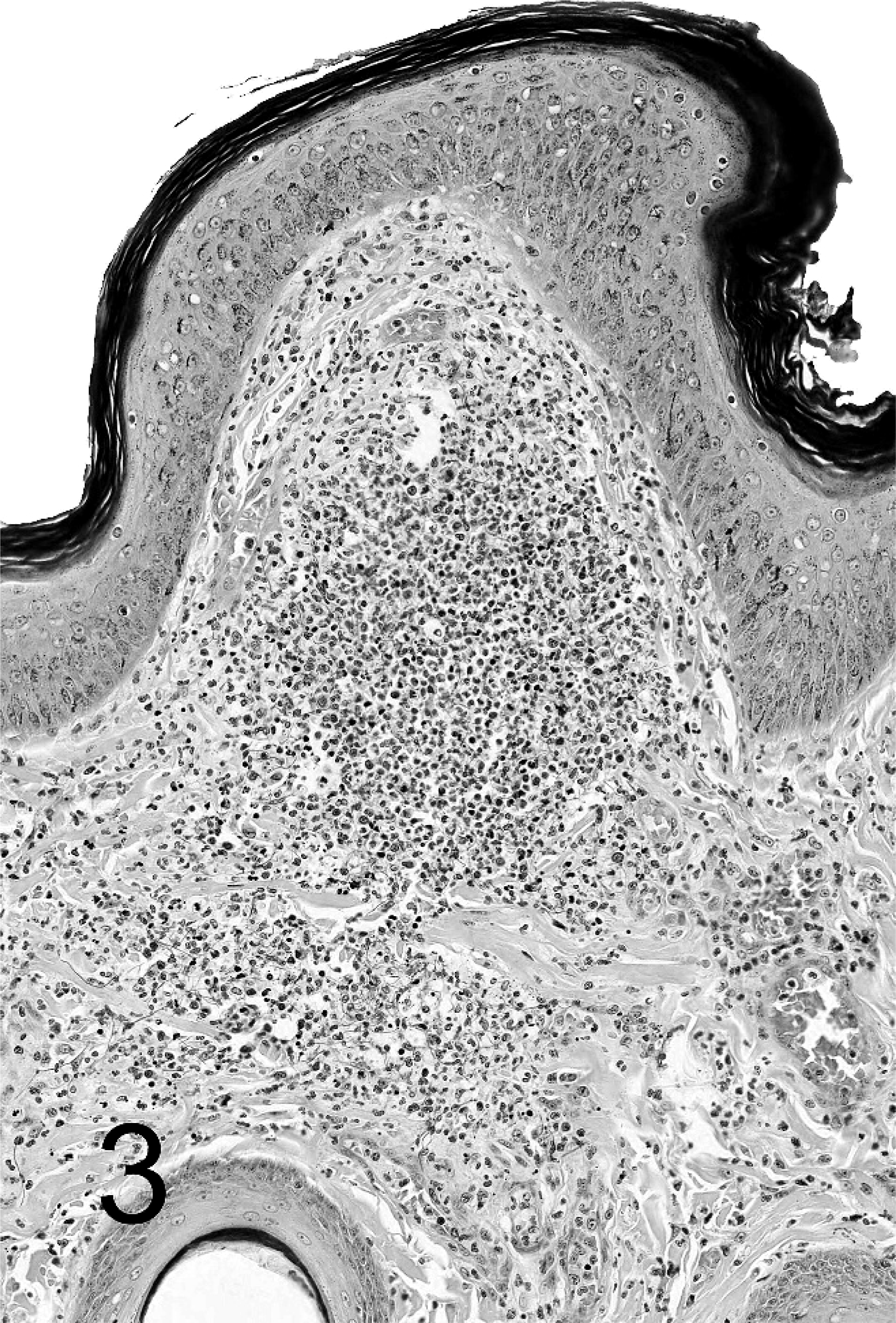
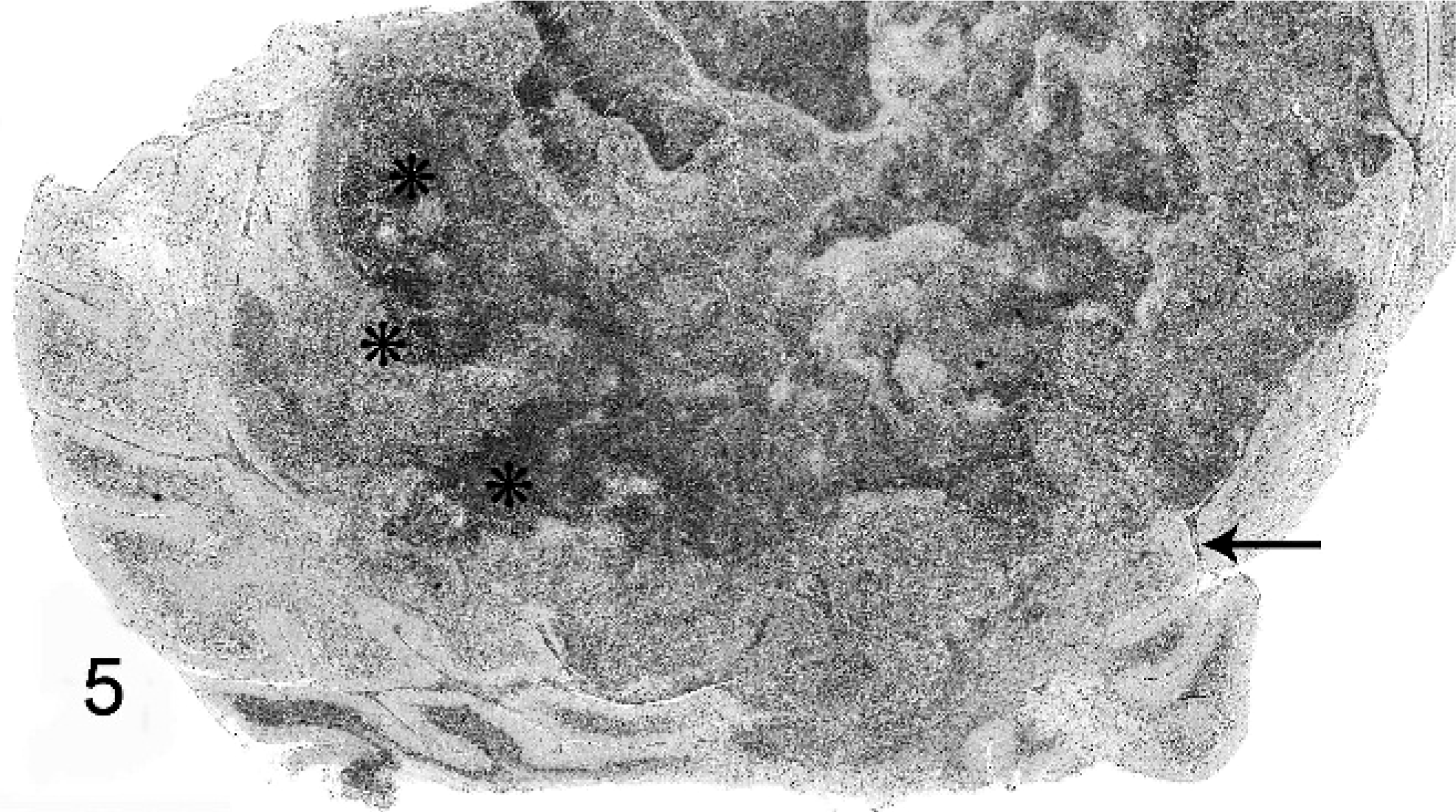
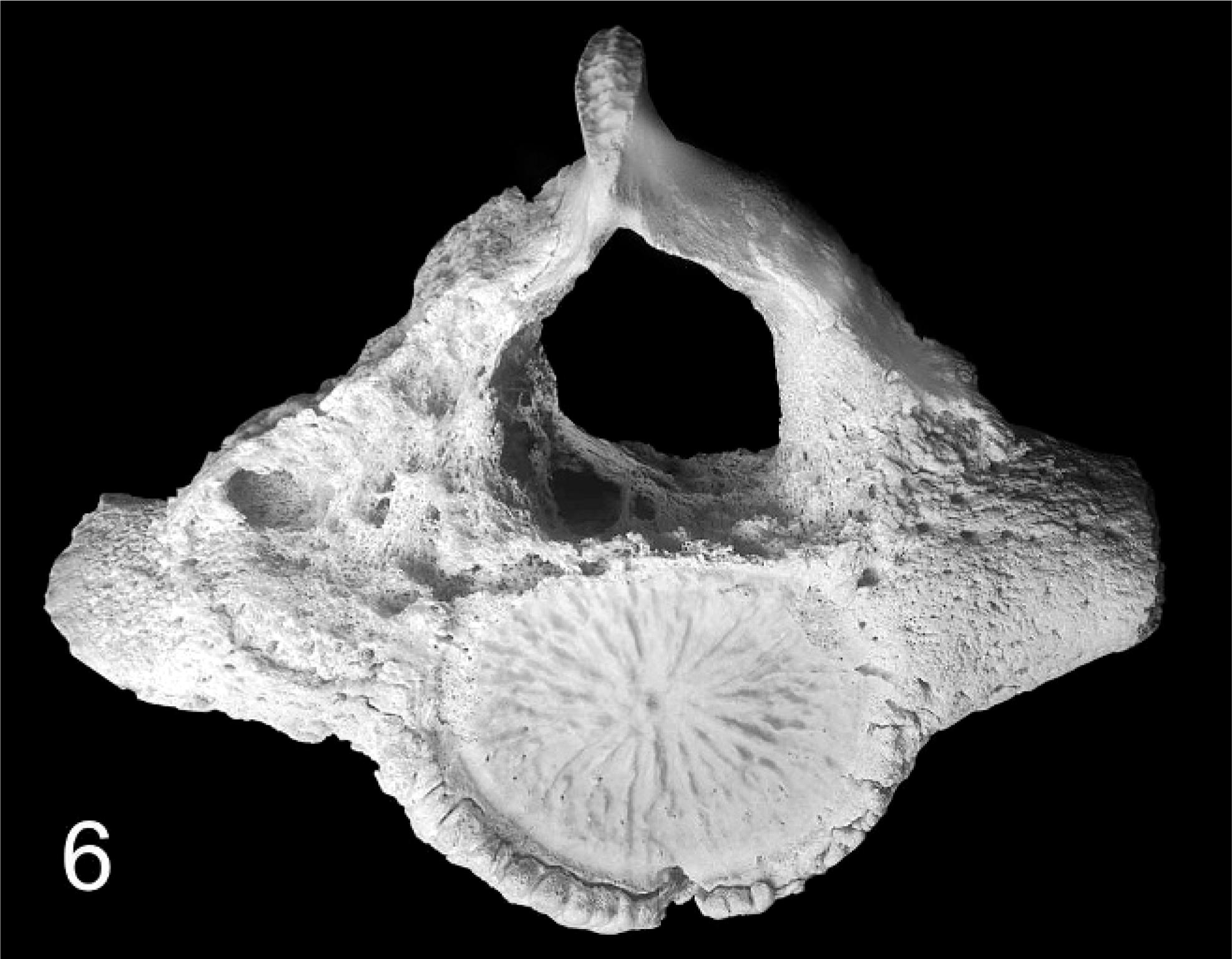
Distribution and severity of relevant lesions in the cetaceans are summarized in Table 2. Pulmonary pyogranulomas with thoracic lymph node abscesses were the most common presentation in cetaceans (Fig. 4). These were seen in 6 cetaceans (Nos. 12, 13, 15, 16, 18, and 19). Three of these animals had pleuritis and moderate amounts of clear yellow or serosanguineous thoracic fluid (Nos. 15, 18, and 19). The characteristics of the fluids are not recorded. Five of these 6 cases had generalized lymph node abscessation (Nos. 11, 15, 16, 18, and 19). Two cetaceans had brain abscesses (Nos. 13 and 14; Fig. 6). In cetacean No. 17, a pyogranulomatous dermatitis and cellulitis extended to the underlying vertebra. The vertebral body demonstrated focally extensive chronic osteonecrosis and remodeling (Fig. 5). Abscesses or pyogranulomas were also recognized in aortic endothelium (cetacean No. 18), spleen (Nos. 11, 15, 18, and 19), adrenals (Nos. 15 and 19), intestine (No. 11), humeroscapular joint (No. 14), kidney (No. 18), and liver (No. 19). Four cetaceans were in good nutritional condition at necropsy (Nos. 14, 15, 17, and 19), and 3 were thin or emaciated (Nos. 10, 12, and 19). Five of 10 cetaceans had concurrent conditions that included granulomatous dermatitis from lacaziosis (cetacean No. 13), ulcerative dermatitis containing ciliated protozoa (No. 10), skin lesions due to trauma (No. 11), and parasitic ulcers in the gastrointestinal tract (Nos. 11, 14, and 17). None of the animals had histologic findings suggestive of concurrent morbillivirus infection.
Lung; beluga whale No. 15. This lung of a beluga whale shows multifocal pyogranulomas scattered throughout the parenchyma.
Cerebellum; Atlantic bottlenose dolphin No. 13. The right area of the cerebellum demonstrates a severe suppurative encephalitis, the border is indicated by asterisks. Nonsuppurative meningitis (arrow). HE.
Thoracic vertebra; beluga whale No. 17. Focally extensive osteonecrosis with remodeling.
Histologic staining was evaluated for the ability to identify slender and filamentous
bacteria typical of
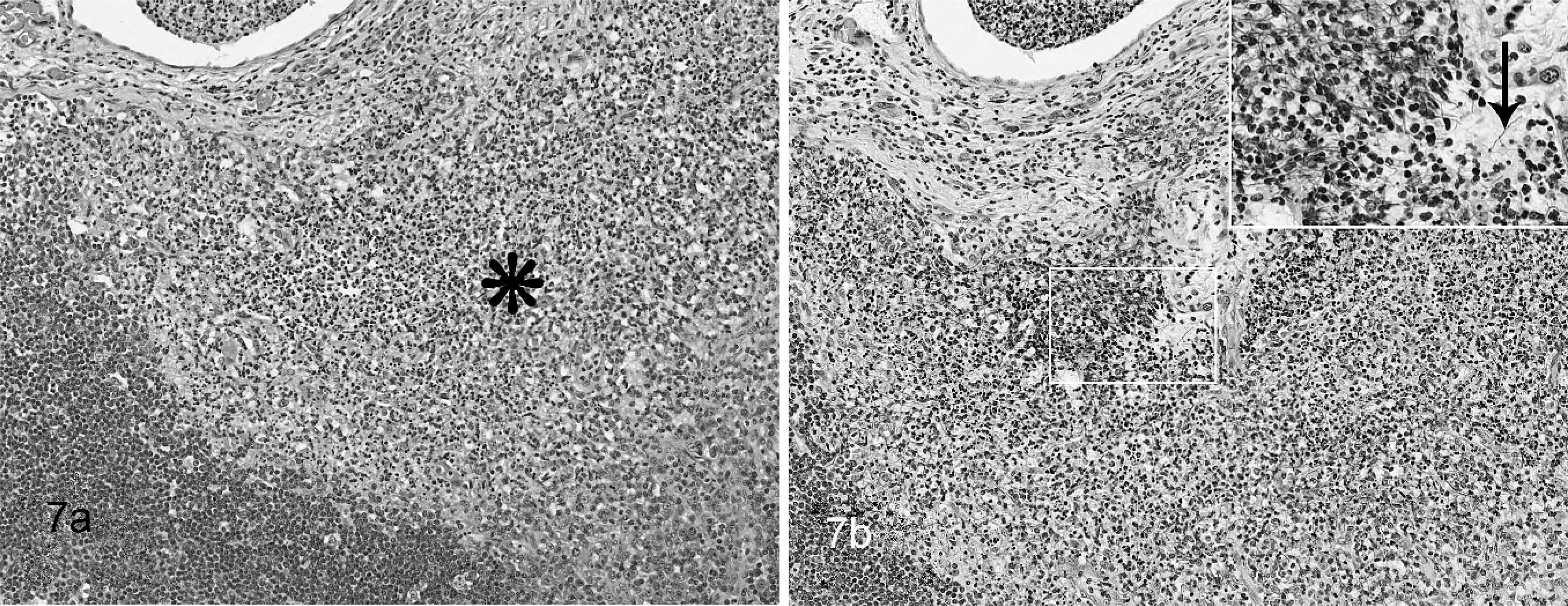
Lymph node; hooded seal No. 2. Granulomatous lymphadenitis Fig. 7a. The sinus is infiltrated by a
neutrophils and macrophages (asterisk). No bacteria can be seen with this
stain. HE. Fig. 7b. Multiple
filamentous
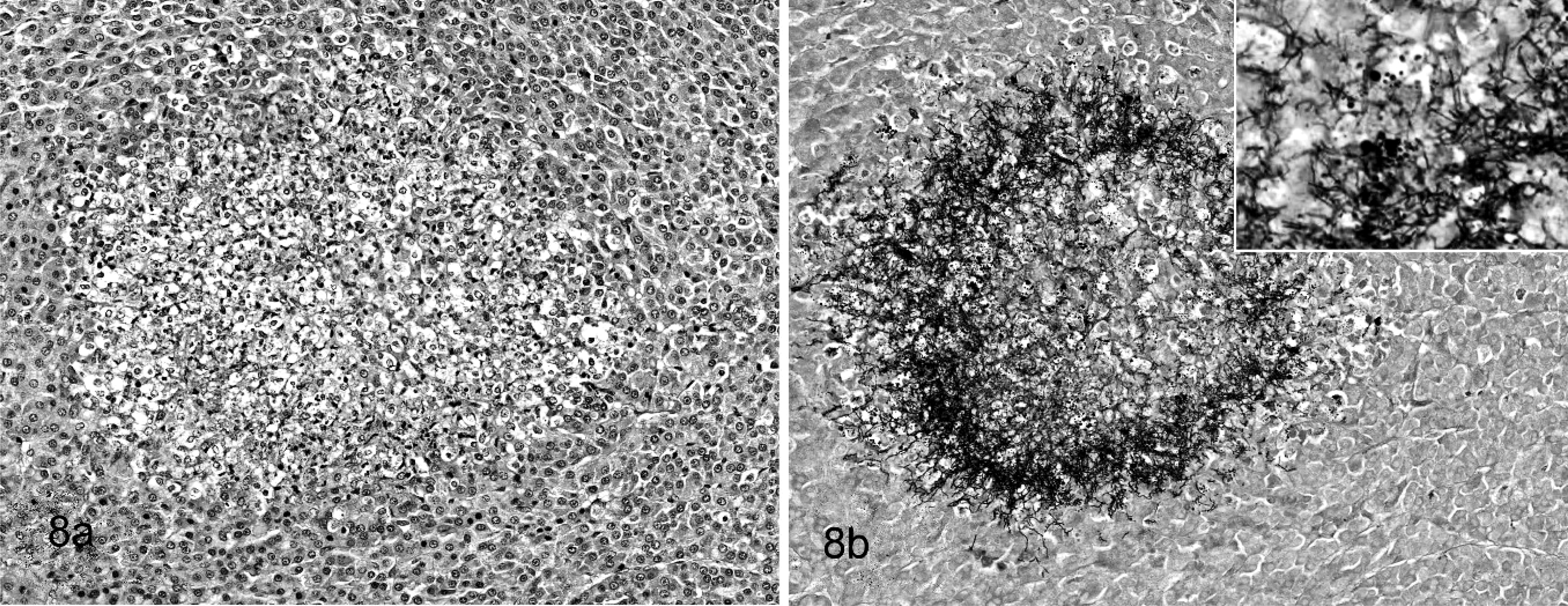
Adrenal gland; beluga whale No. 19. Fig. 8a. Large suppurative circumscribed nonencapsulated focus in
zona fasciculata of adrenal gland. HE stain shows no bacteria. Fig. 8b. Section adjacent to
8a. The stain shows filamentous argyrophilic
Bacterial culture and identification
Nocardiosis was confirmed by bacterial culture in 5 pinniped and 6 cetacean cases.
For confirmation and species identification, PCR with sequencing of amplicons was
performed on FFPE tissue in 5 pinnipeds and 7 cetaceans. In 13 of the 17 cases,
bacterial culture, PCR, or both were able to speciate the
A variety of
Discussion
The lesions caused by
Our findings support and further the current knowledge of nocardiosis in marine mammals.
The results of the currently published data are summarized in Table 3. A literature review demonstrates that the
systemic presentation occurs most commonly, with the lungs most frequently affected. A
single published case identifies involvement of the brain.
15
Brain tissues are often not examined, which could account for the infrequent
reporting of brain involvement in marine mammal nocardiosis. The current literature
describes an interesting
Literature cases with pathology results of marine mammal nocardiosis and
distribution of

+ =
A review of the staining characteristics in these cases proved surprising. Hematoxylin
and eosin staining failed to reveal the bacteria.
Nocardiosis has 3 main routes of infection: inhalation, inoculation, and ingestion.
2,
6,
13,
42
The high proportion of pulmonary involvement suggests that inhalation and
aspiration are the major routes of infection for both cetaceans and pinnipeds. However,
the route of infection is difficult to determine in most of our cases.
Current literature includes 16 cetacean cases of nocardiosis with variable demographic
data available.
15,
18,
25,
29,
41,
45,
49,
52
In previously reported and present cases, captive animals are in the majority (of
26 cases, 18 were captive and 8 were wild). This discrepancy is likely due to better
postmortem carcass condition and more thorough examination of captive cetaceans. The
medical history of the 2 free-ranging cetaceans and a single hooded seal suggests that
these animals were infected with
Hooded seals are dramatically overrepresented in this review. All affected hooded seals
were free-ranging animals. They are held in very low numbers in North America only for research.
35
Given the high levels of rehabilitation for sea lions and harbor seals and the
presence of these animals in captive collections, the lack of nocardiosis in these
species compared with the hooded seals is very unusual. An inherent species
susceptibility or immune deficit might be responsible for the high prevalence in this
species.
Nocardiosis is a significant cause of mortality in marine mammals. Fatal infections are
most typically systemic. In addition to the previously reported species, hooded seals
expand the list of affected marine mammals. Hooded seals appear to be particularly
vulnerable to this infection compared with other pinnipeds. Diagnosis can be facilitated
with GMS staining of granulomas. PCR is an effective way to confirm the diagnosis and
determine the species of
Footnotes
Acknowledgements
We are grateful to clinicians and pathologists that provided us with case materials, including Gregory Bossart, Paul Calle, Terry Campbell, Ronald Crowley, André Dallaire, Les Dalton, Fabio Del Pierro, Christopher Dold, Sam Dover, Lawrence Dunn, Kim Durham, Ruth Ewing, Michael Kinsel, Thomas Lipscomb, Dee McAloose, Richard Montali, Wendy Noke, Todd Robeck, René Roy, Todd Schmitt, Robert Schoelkopf, Megan Stolen, and Mike Walsh. We thank Robert Couey for graphic assistance as well as the staff of NOAA and US Marine Mammal Stranding Network for the stranding statistics, including Carley Lowe, Sarah Wilkin, Aleria Jensen, Joe Cordaro, and Elizabeth Tuohy-Sheen. We thank Stacy Schultz for technical assistance. Work in the laboratory of Dr. Terio was supported by an Office of Naval Research grant (N00014-06-1-0249).
