Abstract
Cerebral and disseminated encephalitozoonosis was diagnosed by histopathology, electron microscopy, and immunohistochemistry in 2 free-ranging South American fur seal pups found dead at Guafo Island (43°33′S 74°49′W) in southern Chile. In the brain, lesions were characterized by random foci of necrosis with large numbers of macrophages containing numerous microsporidial organisms within parasitophorous vacuoles. In addition, occasional histiocytes loaded with numerous mature and immature microsporidia spores consistent with Encephalitozoon sp were observed in pulmonary alveolar septa, splenic red pulp, glomerular capillaries, and proximal renal tubules by Gram and immunohistochemical stains. To our knowledge, microsporidial infection in a marine mammal species has not been previously reported.
The phylum Microsporidia includes obligate intracellular eukaryotic parasites that have rudimentary intracellular organelles (eg, mitochondria, Golgi bodies) and the presence of chitin and trehalose in their walls. 7 This parasitic group infects a wide range of hosts, from protists to humans; however, they are usually considered high-impact pathogens for invertebrates and fish populations. 7
Of the 14 species of microsporidia known to infect mammals, the most studied is Encephalitozoon cuniculi, which is reported to infect rabbits, canids, cats, primates, rodents, ruminants, horses, humans, birds, and bearded dragons. 5,10 Furthermore, E cuniculi and other congeners, such as Encephalitozoon intestinalis and Encephalitozoon hellem, are important human pathogens, especially in immunocompromised patients. 10 However, reports of natural microsporidial infection in marine mammals are lacking. We describe 1 case of fatal disseminated encephalitozoonosis and 1 case of localized cerebral infection in South American fur seal (Arctocephalus australis) pups.
Both animals were found dead at Guafo Island (43°33′S 74°49′W), southern Chile, in the austral summers of 2008 and 2005, respectively. Necropsies and tissue collection were performed following a standard protocol. 6 All collected samples were fixed in 10% neutral buffered formalin, processed by routine paraffin histology, and stained with hematoxylin and eosin (HE). In both cases, selected tissue sections (brain, kidney, lung, and spleen) were stained with Lillie Twort Gram, periodic acid–Schiff reaction, modified acid-fast, and Warthin-Starry stains. Sections from the same organs of case No. 1 were also immunostained with a rabbit hyperimmune anti–Encephalitozoon cuniculi antiserum, which has been shown to react with E. cuniculi, E. hellem, and E. intestinalis. 2 Two sections of brain from case Nos. 1 and 2 were stained with calcofluor white and observed with a fluorescent microscope using wide-field epifluorescence optics. Selected paraffin-embedded sections of brain from each case were processed for ultrastructural examination. Ultrathin sections were mounted on copper grids, stained with uranyl acetate and lead citrate, and examined and photographed using a transmission electron microscope.
Case No. 1 was a 2-week-old female pup in fair nutritional condition and with minimal postmortem autolysis. There was a moderate amount of hemorrhagic content within the jejunum and ileum, admixed with numerous adult hookworms (Uncinaria sp). There was a small focus of subdural hemorrhage on the dorsal aspect of the right parietal lobe and scant gastric content (milk) in the trachea and main bronchi.
Microscopic lesions were most severe in the brain. There were multiple small- to medium-size random foci of necrosis throughout the gray matter, with numerous foamy macrophages admixed with rare glial cells, lymphocytes, neutrophils, and small amounts of cellular debris. Neurons surrounding these areas had pyknosis and hypereosinophilic, sometimes contracted, cytoplasm. In some foci, macrophages were markedly enlarged, with a displaced nucleus and cytoplasm containing numerous oval, 2 × 1–μm lightly eosinophilic microorganisms sometimes surrounded by a narrow (0.5-μm) clear halo (Fig. 1). Larger numbers of microorganisms, some with a distinct marked polar staining, were observed in the same foci by Gram stain (Fig. 2). Numerous fungal spores were visualized with the calcofluor stain (Fig. 3), and fewer organisms were observed with a modified acid-fast stain. Rarely within the white matter, in areas free of inflammation, there were large (up to 30 μm in diameter) pseudocysts (xenomas) containing numerous 3 × 1–μm organisms with staining properties similar as in areas with inflammation. In a few sections, some of these pseudocysts were ruptured and surrounded by neutrophils and macrophages. Occasionally, moderate numbers of macrophages, lymphocytes, and fewer plasma cells and neutrophils expanded the leptomeninges and aggregated around blood vessels with plump endothelial cells.
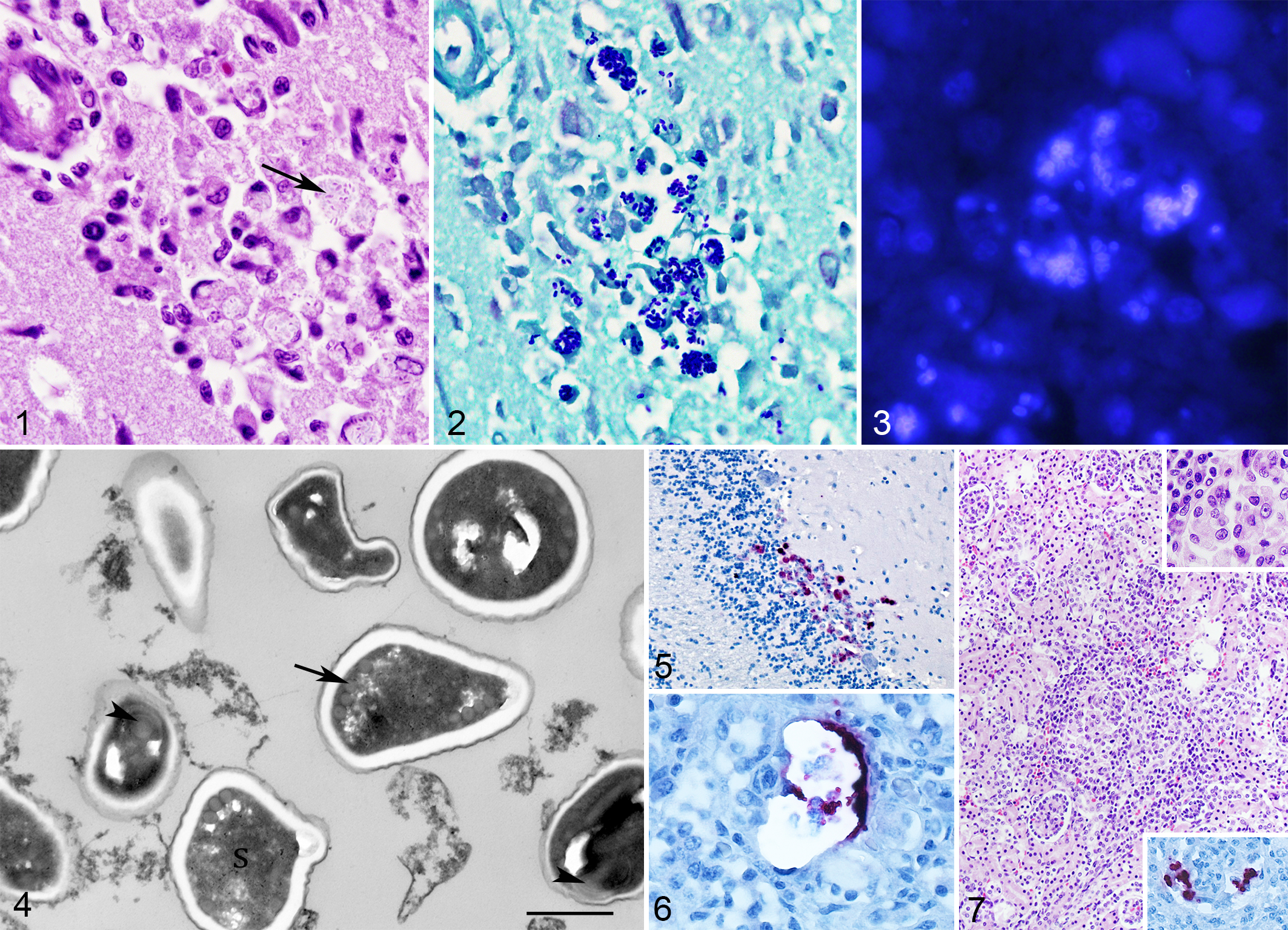
Encephalitozoon sp infection, South American fur seal, brain.
On ultrastructural examination, macrophages within areas of necrosis contained up to ten 2.2 × 1.25–μm spores within a large parasitophorous vacuole. Spores had a 2-layer wall with an external, up to 50-nm-thick, electron-dense membrane (exospore) contiguous with a 100-nm-thick electron-lucid layer (endospore). Occasional spores had a moderately electron-dense sporoplasm, while others were electron lucid (empty spores). In some spores, at the periphery of the sporoplasm, 4 to 5 cross sections of 50-nm diameter polar filaments were observed (Fig. 4). These filaments were lined by a 10-nm-thick, mildly electron-lucid membrane. These morphological characteristics were consistent with previously published descriptions for the genus Encephalitozoon. 8 The spores had marked, diffuse, positive immunostaining with the anti–E. cuniculi antiserum. In addition to the sites previously described, spores were also observed in the granular layer of the cerebellum (Fig. 5), lung alveolar macrophages, splenic red pulp, hepatic sinusoids and glomerular capillaries, Bowman’s spaces, and proximal renal tubules in the kidney (Fig. 6). Lesions in other tissues included a moderate, multifocal, histiocytic, interstitial nephritis with the presence of rare intrahistiocytic Encephalitozoon spores (Fig. 7). These spores were not observed in HE sections but were easily recognized with immunohistochemistry (Fig. 7, inset). In the lung, there was moderate, diffuse, histiocytic interstitial pneumonia and moderate, diffuse, lymphoplasmacytic enteritis in the jejunum and ileum. In the liver and spleen, there were occasional small foci of hematopoiesis.
Case No. 2 was a 3-month-old male pup in fair nutritional condition and with moderate postmortem autolysis. The small intestinal mucosa was reddened, and there were approximately 20 hookworms free in the intestinal lumen. The mesenteric lymph nodes were moderately enlarged. Microscopic lesions were restricted to the cerebrum. Foci of necrosis, as described in case No. 1, with occasional macrophages containing numerous cytoplasmic Encephalitozoon sp spores, were rarely observed throughout the gray matter of the parietal lobes. There was also mild lymphoplasmacytic enteritis and mild histiocytic interstitial pneumonia.
Additional diagnostic tests performed in formalin-fixed, paraffin-embedded sections of brain, lung, spleen, liver, and kidney included Sarcocystis neurona, Toxoplasma gondii, Neospora caninum, and canine distemper virus immunohistochemistry and pan-fungal, pan-bacterial, and pan-herpes virus polymerase chain reaction (PCR). All these tests yielded negative results. Efforts to amplify microsporidial DNA from formalin-fixed, paraffin-embedded tissues were unsuccessful. No fresh tissues were available for additional molecular analyses. However, the histopathological, ultrastructural, and immunohistochemical findings suggest that it is Encephalitozoon sp.
Disseminated encephalitozoonosis was the most likely cause of death in case No. 1, while the role of the Encephalitozoon encephalitis in the death of case No. 2 is uncertain. In mammals, disseminated fatal disease due to microsporidia has been reported in young foxes, dogs, and cats infected with E. cuniculi. 10 On the other hand, chronic subclinical infection with mild histiocytic encephalitis, as seen in case No. 2, is the hallmark of E. cuniculi infection in rabbits and rodents. 4 Epidemiological and experimental E. cuniculi infection studies suggest that chronic infection can develop in other species and that the outcomes of development of a chronic carrier state, elimination of the parasite, or disseminated fatal disease depend on the ability of the immune system to respond to the microsporidia infection. 3,9
The source of the infections in these young animals is unknown. It is not considered endemic in this population because, over a 7-year period with histopathologic examination of 85 South American fur seal pup brains, we detected only these 2 cases. Transmission from rats (Rattus rattus) that live at the fur seal rookeries is possible, since rats are considered chronic carriers that can disseminate E. cuniculi. 1 Other sources of a mammalian Encephalitozoon sp are unlikely since Guafo Island is an oceanic island with no other mammals besides introduced rats and cats.
To our knowledge, this is the first report of microsporidia infection in any marine mammal species. This expands the range of hosts affected by Encephalitozoon and highlights the need to consider microsporidia as etiological agents of disease in marine mammals.
Footnotes
Author Contributions
M. Seguel contributed to conception and design; contributed to acquisition, analysis, and interpretation of data; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. E. Howerth contributed to design, contributed to analysis and interpretation of data, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. J. Ritter contributed to design, contributed to acquisition and interpretation, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. E. Paredes contributed to conception, contributed to acquisition, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. K. Colegrove contributed to design, contributed to acquisition and analysis, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. N. Gottdenker contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Field work partially funded by grant SE-2012-53 from the Universidad Austral de Chile, Research and Development Direction; grant 2011-13 from The Society for Marine Mammalogy, small grants in aid of research award; and grant 13225-1 from The Rufford Small Grant Foundation.
