Abstract
Tissues of South American fur seal pups naturally infected with hookworms (
Hookworms are highly pathogenic nematodes that parasitize a wide range of mammals including most species of otariid seals. 14 In all hosts, adult hookworms live primarily in the small intestine, attach to the intestinal mucosa, and bite small portions of the epithelium and lamina propria, sucking blood from intestinal wounds. 10 This feeding behavior leads to chronic mucosal bleeding, in part due to the several anticoagulant proteins that the parasite secretes in the attachment site, resulting in hemorrhagic enteritis and chronic anemia that is characteristic of hookworm infection in most host species. 5,10 Experimental models show that pathogenic effects of hookworm infection depend on the species of hookworm, the host inflammatory response, and the number of nematodes in the intestine. 6
Hookworms have been described in 13 of 15 extant otariids and in 3 phocid species.
21
However, the taxonomy of marine mammal hookworms is probably incomplete, since to date there are only 4 fully described species:
The hookworm life cycle is similar in the 3 otariid species that have been investigated.
2,14,15
Neonates become infected most likely through the colostrum, and the prepatent period is between 12 and 18 days. Pups release embryonated eggs in the feces that hatch in the soil and larvate to the infective L3 stage, which penetrates the skin of most animals in the rookery. These larvae remain in subcutaneous tissues and mammary glands of females until the next reproductive cycle, when they are passed to the next generation of pups.
14
Contrary to other species of hookworms, otariid
Although most otariid species harbor hookworms, they can be an important cause of disease and mortality of pups in populations of Northern fur seals,
11
–13
California sea lions,
25
New Zealand sea lions (
In South American fur seals (SAFS), we have previously described the main pathologic findings and causes of pup death.
23,24
In these studies, we registered hookworm-related disease as the cause of up to 50% of the total pup mortality in some breeding seasons. In contrast to our findings, hookworm infection in SAFS rookeries in Uruguay is of low prevalence (< 5%) and has not been implicated in mortality based on gross postmortem examinations.
7
However, preliminary data indicate that SAFS in Uruguay are infected with
Materials and Methods
Animals
Necropsies were performed on 140 SAFS pups that were found dead on Guafo Island, Chilean Patagonia (43°36’S and 74°43’W), between December 28 and March 10 during the 2004 to 2008 and 2012 to 2015 breeding seasons. Histopathology was performed in 112 animals, and in 55 of these pups, peritoneal washing and bone marrow evaluation was also performed. These cases were selected based on minimal or mild postmortem autolysis and no signs of scavenging. The sex, rookery sector, total body length, and weight were recorded for all but 2 pups. Pups were between 2 and 11 weeks old, based on parturition peak date for the Guafo Island rookery (December 15).
20
A body mass index for the pups was calculated by dividing the weight (in kilograms) by the total length (centimeters).
23
After excluding animals that did not have a peritoneal wash and those that had incomplete samples for histology, inadequate intestinal fixation, or mild to moderate autolysis at histopathology, a total of 45 animals were included in the histologic statistical analyses. To test the significance of tissue changes in animals with different outcomes of hookworm infection, pups were divided into 3 groups: the first included pups that were considered to have died as a consequence of hookworm infection (termed hookworm enteritis with bacteremia [HEB]) but without hookworm peritoneal penetration (
Necropsies and Histopathology
Complete necropsies, including detailed gastrointestinal tract examination for nematodes, were performed in the field following a previously described protocol. 23 In addition, from 2012 to 2015 breeding seasons, examination of the peritoneal cavity for nematodes was conducted as described in otariid seals. 12 Tissues routinely sampled for histopathology included brain; lung; trachea; heart; spleen; submandibular, prescapular, bronchial, mediastinal, and mesenteric lymph nodes; colon; kidney; testis; ovary; adrenal gland; esophagus; stomach; liver; pectoral and masseter skeletal muscles; skin from the pectoral and maxillary regions; and at least 3 cross-sections from each small intestinal segment (duodenum, jejunum, and ileum). The sections of ileum were systematically sampled at 10 cm cranial to the ileo-cecal junction. Bone marrow was examined in the mainland lab (Universidad Austral de Chile) after formalin fixation and formic acid decalcification of the tibial epiphysis in samples collected between 2012 and 2015. Tissue samples were fixed in 10% neutral buffered formalin, embedded in paraffin wax, sectioned (4 µm), and stained with hematoxylin and eosin (HE). In selected cases, sections were stained with PAS-Alcian blue, Lillie Twort, or modified Brown and Brenn methods for Gram stain, Giemsa, acid fast, Gomori methenamine, and Warthin-Starry silver stains.
Tissue changes associated with hookworm infection were measured in hookworm-infected and hookworm-free pups with complete necropsies (including peritoneal wash) and tissue samples (
Differences in erythrocyte consumption in the hookworm intestinal tract were observed microscopically between pups with peritoneal penetration and those without. To test if there were actual differences in the intestinal content of hookworms between pups with different causes of deaths, a group of 30 to 40 hookworms per host was extended in a histology cassette, embedded in 2.5% agarose gel, and fixed in 10% buffered formalin, and two 0.3-cm-thick cross-sections were obtained from the mid-portion of the worms. The sections were processed routinely for histopathology, resulting in a 4-µm HE-stained section containing between 40 and 60 cross-sections of hookworms at the level of the intestine and reproductive organs. Thirty sections containing segments of the nematodes’ intestine were randomly selected, and the number of sections that contained at least 1 erythrocyte and the average number of erythrocytes in the sections were recorded.
Parasitology
All nematodes found at necropsy were retrieved, counted based on their location, and washed with sea water. Most were placed in 5% formalin for morphologic and histologic studies, and a small subset was placed in 70% ethanol for genetic studies. A subset of nematodes from each host was mounted on permanent glass slides and observed microscopically. Standard measurements were recorded and genus-level identification was performed according to the key for the identification of parasitic nematodes. 9
Bacteriology
In cases from 2012 to 2015, samples from the lung, liver, kidney, bronchial and mesenteric lymph nodes, distal jejunum, adrenal gland, and brain were collected in sterile plastic bags and stored at –19°C for up to 1 month in the field and later at –80°C in the mainland lab. In cases with gross evidence of peritonitis (peritoneal exudate and/or fibrin), sterile peritoneal swabs were taken. Carcasses with an estimated postmortem interval of less than 6 hours, based on absence of rigor mortis or previous observation of a sick pup found dead hours later, had blood swabs taken from the heart or cranial vena cava. Swabs were placed in a sterile plastic tube with Stuart medium and stored for up to 1 month at –19°C and later at –80°C until bacteriologic culture. Frozen tissues were defrosted at room temperature for 12 hours, and a sterile section obtained from the center of the sample was incubated in enriched medium at 37°C for 24 hours prior to plating on McConkey and blood agars. Swabs were defrosted at room temperature for 6 hours prior to plating. Isolated bacterial colonies were identified to the genus or species level by Gram stain and biochemical reactions using a BBL Crystal ID System for enteric/nonfermenting and gram-positive bacteria (BD, Sparks, MD).
Statistical Analyses
Statistical analyses and graphics used R-3.2.0® and GraphPad 6® software. To test histologic differences between animals that died because of HEB and those that died of trauma, Mann–Whitney tests were performed. Nematode burden and histologic differences between animals that died of trauma, HEB without peritoneal penetration, and HEB with peritoneal penetration were assessed by generalized linear models with negative binomial error structure (GLM.NB). Spearman rho correlation tests were performed in a matrix containing all histologic counting data and hookworm burden.
To test which variables were associated with hookworm peritoneal penetration, we used Firth’s logistic regression. Based on predictor correlations and histopathologic assessment, the predictor variables in the maximal (global) model were nematode burden, hookworm sections containing erythrocytes, intestinal villi length, number of leukocytes in the intestinal mucosa, and their interactions. The final models were selected based on the significance of the predictors, likelihood ratio, and the Akaike information criterion (AIC).
Results
Hookworm infection was found in 132 of 140 necropsied pups (94.2%; 68 males, 64 females). In 46 of 132 (34.8%) infected animals, the lesions caused by hookworms and secondary bacterial colonization, translocation, and likely sepsis were considered as the cause of death. This cause of death was designated as hookworm enteritis and bacteremia. The gross and histologic lesions observed in SAFS pups infected with
Lesions in SAFS Dying of Trauma
In all of the pups that died of trauma, hookworm-associated gross lesions were restricted to the distal jejunum, ileum, and mesenteric lymph nodes. Peyer’s patches were prominent and the mesenteric lymph nodes markedly enlarged (Supplemental Fig. 1). There were mild to moderate amounts of clear mucus on the jejunal and ileal mucosa admixed with small to moderate numbers (mean intensity = 163; bootstrap 95% confidence interval [CI], 102.76–240.71) of nematodes that were 1.0 (males) or 1.9 (females) cm in length. In some areas of the ileal mucosa, there were a few 0.1- to 0.2-cm diameter hemorrhages (hookworm attachment sites).
Microscopically, the mid and distal jejunum and ileum were filled with abundant PAS-Alcian blue–positive mucus and degenerate eosinophils (Supplemental Figs. 2a, 2b). The villi were long and large numbers of eosinophils and fewer lymphocytes and plasma cells expanded the lamina propria (Supplemental Fig. 3). The apical portion of the villi had rare areas with loss of epithelial cells (hookworm feeding sites) and exposure of the lamina propria. These areas were usually surrounded by numerous neutrophils, eosinophils, and occasional macrophages. At the base of the villi, there were numerous mitotic figures in the intestinal epithelium and large numbers of goblet cells. Peyer’s patches were prominent and populated by numerous lymphocytes and plasma cells and fewer macrophages. The mesenteric lymph node cortex was expanded by numerous secondary lymphoid follicles (Supplemental Figs. 4, 5). No bacteria were observed in the lamina propria or mesenteric lymph nodes with Gram, Giemsa, acid fast, and silver stains, and no bacteria were isolated from liver, mesenteric lymph node, spleen, brain, or blood. The hypercellular bone marrow contained approximately 60% of hematopoietic elements and at least 3 megakaryocytes per 400× section (Supplemental Figs. 6, 7; Supplemental Table 1).
Lesions in SAFS Dying of HEB
In pups that died of HEB (
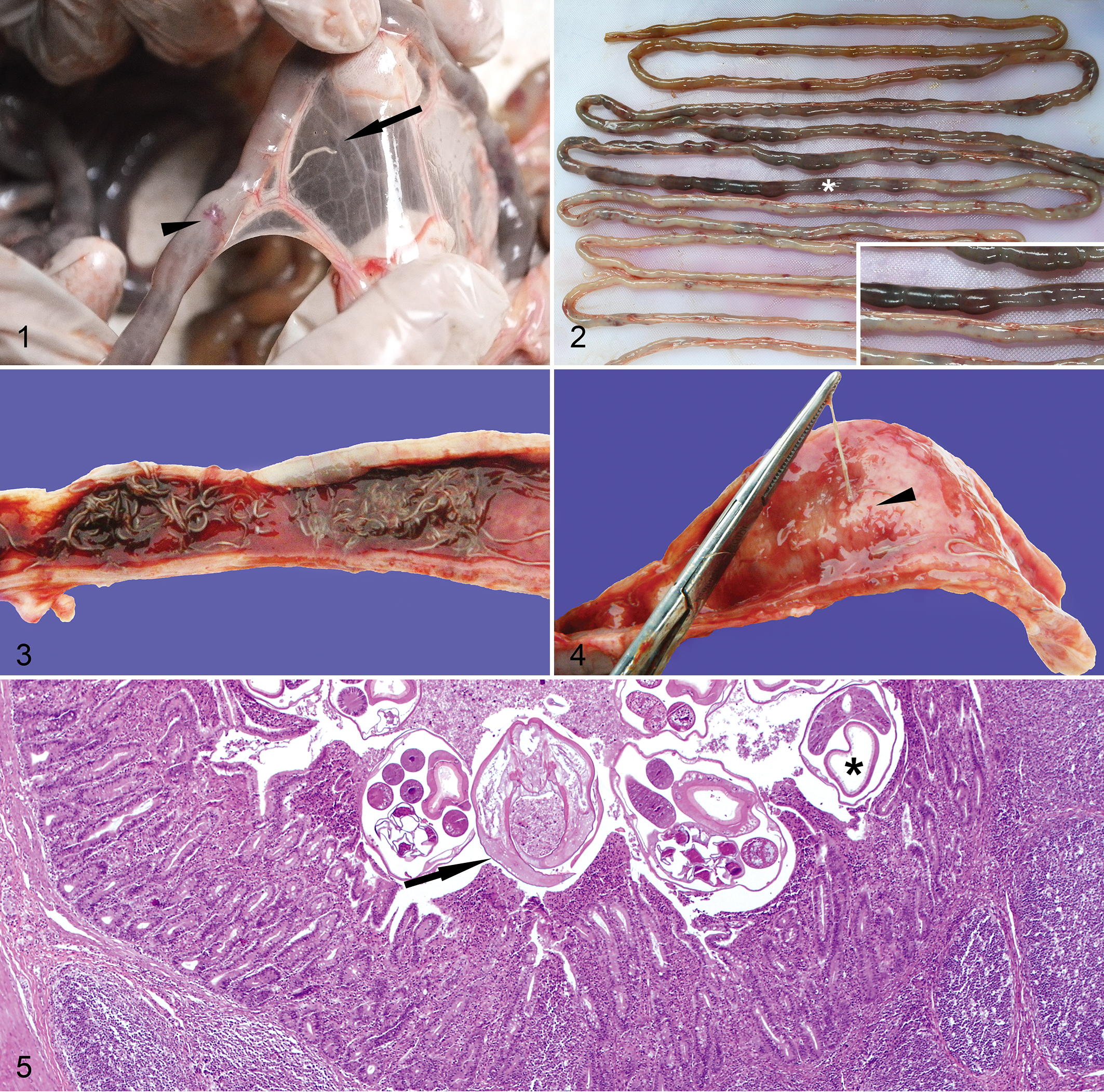
Microscopically, the lumens of the jejunum and ileum contained large amounts of cellular debris admixed with large numbers of bacteria, degenerate erythrocytes, and occasional 310- to 510-µm-diameter nematodes characterized by a thin cuticle; polymyarian, platymyarian musculature; a gastrointestinal tract lined by simple columnar epithelium with a prominent brush border; and reproductive tract with either numerous spermatids or 100-µm embryonated eggs (Fig. 5). In some nematodes, the cuticle was lined by numerous small gram-negative bacilli and the gastrointestinal tract brush border was occupied by a few gram-positive or gram-negative bacilli. Intestinal villi were short, blunt, and occasionally fused. There were multiple hookworm feeding sites with exposure of the lamina propria and numerous gram-negative short bacilli on the apical surface of adjacent epithelial cells. Macrophages, lymphocytes, plasma cells, fewer neutrophils, and rare eosinophils mildly expanded the lamina propria of the villi. The crypts were occasionally dilated and filled with cellular debris, degenerate neutrophils, and variable numbers of gram-negative short bacilli (Figs. 6a, 6b) and/or numerous gram-negative, silver-positive, thin, filamentous bacteria.
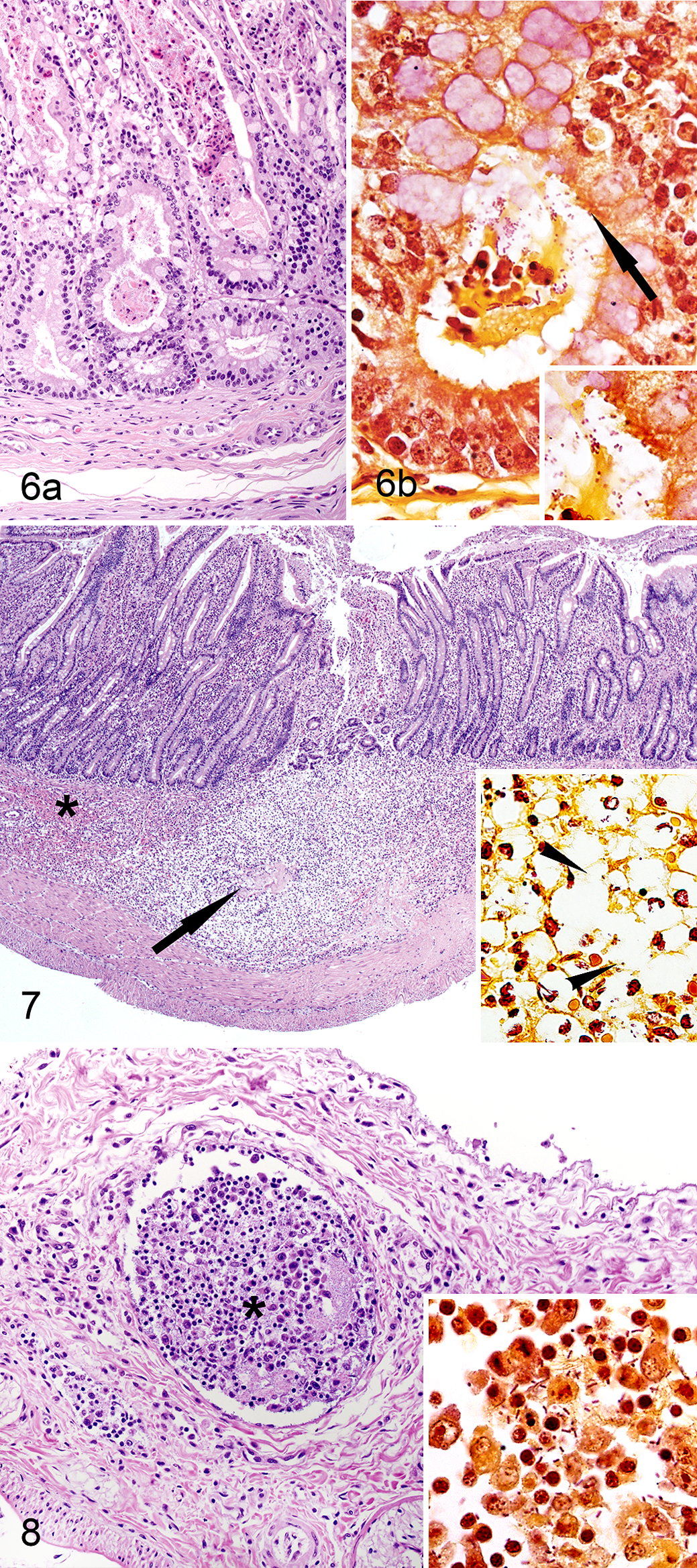
In pups with hookworm peritoneal penetration, multiple hookworm feeding tracks expanded the submucosa and muscularis (Fig. 7). These areas were characterized by a center of lytic necrosis surrounded by numerous macrophages, rare gram-negative short bacilli, and profuse hemorrhage. Rare macrophages, lymphocytes, and plasma cells slightly expanded the serosa and mesentery.
Peyer’s patches, mesenteric lymph nodes, and spleen were depleted of lymphocytes, and there was moderate erythrophagocytosis in the mesenteric lymph node. In 18 of 25 (78%) cases, the subcapsular and medullary sinuses contained a few intrahistiocytic or free colonies of gram-negative coccobacilli. Similarly, intrahistiocytic and free gram-negative bacilli were observed in blood vessels of the spleen (Fig. 8), liver, and lung. In some pups, there was moderate to marked splenic (14/25, 56%) and hepatic (8/25, 32%) extramedullary hematopoiesis. The liver had small to moderate numbers of macrophages, neutrophils, and rare lymphocytes within the portal areas and, in most pups (21/25, 84%), occasional small random foci of hepatocellular dissociation, microvacuolar degeneration, and necrosis. In rare animals (6/25, 24%), the tunica media of hepatic arteries was expanded by numerous neutrophils and macrophages admixed with scant fibrin. In most pups (23/25, 92%), there was mild to moderate interstitial histiocytic pneumonia. The bone marrow was mildly hypercellular with approximately 40% of hematopoietic components and few (< 1 per 400× field) megakaryocytes. Findings that occurred with less frequency included moderate purulent conjunctivitis in 6 pups, mild to moderate interstitial histiocytic nephritis in 6 pups, mild to moderate purulent meningitis in 4 animals, and fibrinous arthritis in 2 pups (Supplemental Table 1).
Bacteriology
Different species of enterobacteria were isolated from the small intestine, mesenteric lymph nodes, spleen, blood, liver, and other tissues (Table 1). The most commonly isolated species were nonhemolytic
Bacteria Isolated From 13 South American Fur Seals (
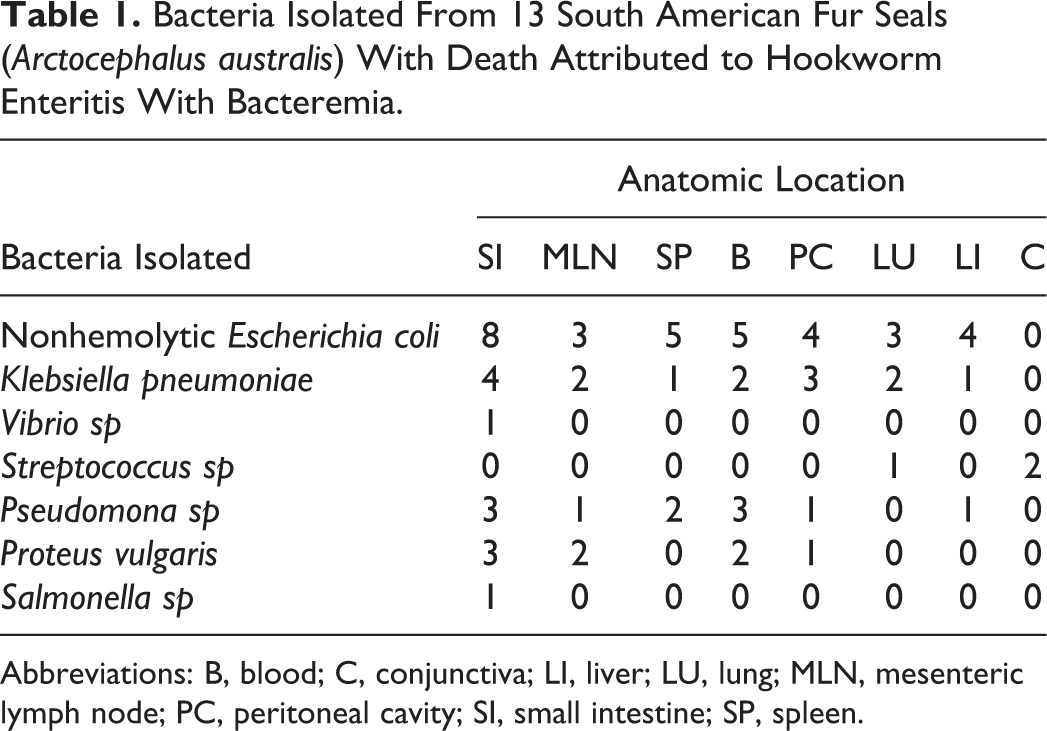
Abbreviations: B, blood; C, conjunctiva; LI, liver; LU, lung; MLN, mesenteric lymph node; PC, peritoneal cavity; SI, small intestine; SP, spleen.
Statistical Models and Differences Between Groups
The number of nematodes was higher in animals that died because of HEB (mean intensity = 761.8; bootstrap 95% CI, 517.73–1170.00) and in those that had hookworm peritoneal penetration (mean intensity = 1384.33; bootstrap 95% CI, 1112.56–1843.78) compared with animals that died due to trauma (mean intensity = 163; bootstrap 95% CI, 102.76–240.71) (GLM.NB, df = 44,
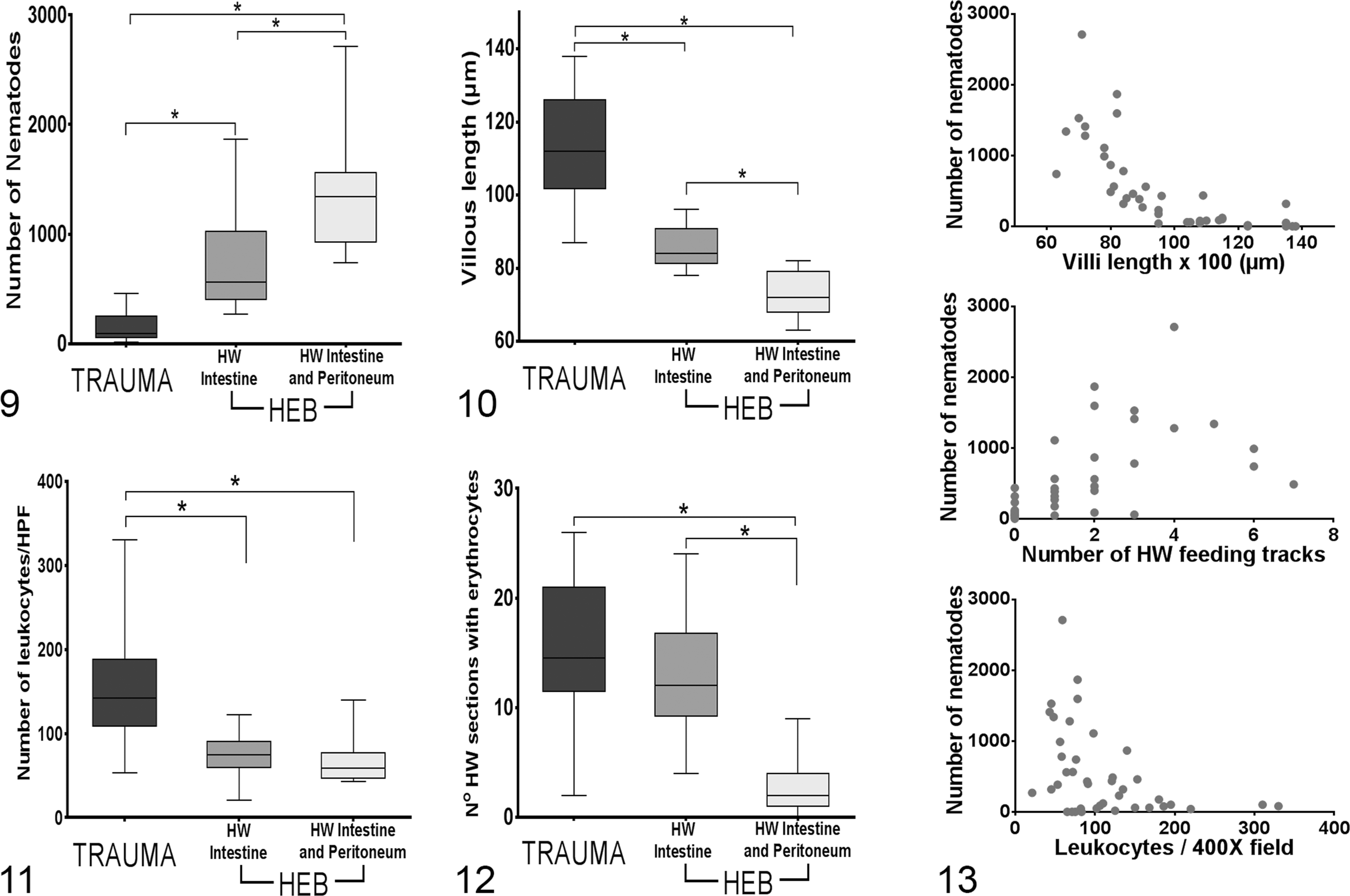
The number of nematodes was negatively correlated with the intestinal villous length (
Discussion
We describe detailed gross and histopathologic findings in SAFS infected with
In pups that died of causes unrelated to hookworm infection (trauma group), there was marked eosinophilic enteritis with a large amount of mucus. A similar type of enteritis has been described in rats infected with
The average hookworm burden in pups that died due to HEB (mean intensity = 762 nematodes) is smaller when compared with those reported in necropsied Australian sea lion pups (mean = 2138 nematodes),
16
New Zealand sea lions (mean = 824),
2
northern fur seals (means = 643, 1200),
11
–13
and California sea lions (means = 612, 1284).
11
–13
These differences could be due to population-specific factors such as overall prevalence, immunity, genetic susceptibility, and rookery substrate, or they could reflect species-specific differences, such as
In the HEB group, most observed lesions have been previously described in California sea lions infected with
In our population, as previously described in California sea lions,
25
hookworm-induced host death is most likely produced by a combination of marked anemia, intestinal damage with absorption of bacterial products, and translocation of bacteria into the vascular system and dissemination to major organs. It is likely that these events led to sepsis with the concurrent immune-metabolic disturbances that define this syndrome.
1
Nonetheless, since it is not possible to confirm sepsis by postmortem examination, we preferred to use the term introduced by Spraker et al
25
of HEB, despite the fact that the word
Bacterial infections found in most pups with HEB played an important role in their death; however, the variety and type of species isolated suggest that these bacteria were opportunistic pathogens, probably taking advantage of a disrupted intestinal barrier and a debilitated immune system. On the other hand, some of the isolated bacterial species are usually considered of low pathogenicity and common contaminants of biological samples (eg,
The parasitic burden was markedly different between trauma and HEB groups, suggesting that the number of nematodes is one of the most significant factors driving hookworm-induced tissue damage and mortality. In addition, hookworm burden was highly correlated with intestinal villous length, number of intestinal leukocytes, and hookworm feeding tracts. The short villi found in animals with high nematode burdens is probably caused by the “grazing” effect of hookworms on the intestinal villi, because to feed on blood, the nematodes have to bite and cut out pieces of the mucosa. Despite the large number of nematodes in pups with HEB, inflammation was minimal. This could be due to a primary lack of strong immune reaction in these pups allowing for the establishment of a larger number of larvae in the intestine. On the other hand, this weak inflammatory response could be the consequence of immunomodulatory hookworm secretion products. Both types of host–pathogen relationship have been shown to be significant in experimental models of hookworm infection, and it is very likely that these 2 mechanisms acted in our studied animals. 4,5,10,19 Another explanation for differences observed in the level of inflammation between pups with trauma and HEB could be the temporal variation in the process of hookworm disease, with an early strong eosinophilic response that later is overwhelmed, leading to mild inflammation by the time the hookworms kill a pup and we examine the tissues. However, given the marked synchronization of parasite transmission with the host reproductive cycle, we would expect to see differences in the type and level of mucosal inflammation in pups of different ages. This was not the case, and animals that died due to HEB were as young as 21 days old, whereas some pups dying of trauma with marked eosinophilic enteritis were up to 80 days old, making the temporal variation in the inflammatory process a less likely explanation.
To our knowledge, adult hookworm peritoneal penetration has not been reported in animal groups other than pinnipeds. This peculiarity could be related to the life strategy of marine hookworms, which are highly adapted to the reproductive cycle and lifestyle of their host. In general, pinniped hookworms infect neonates through the colostrum but are expelled from the intestine before their hosts are weaned between 2 and 6 months of age. 3,11 –15 This means that they have a short period of time to grow, mate, and release eggs. This probably is associated with aggressive feeding behavior to supply their high metabolic demands and could be one of the reasons for the high pathogenicity of hookworms in many pinniped species. 12,15,25 In addition, our histologic and statistical analyses indicate that in pups with hookworm peritoneal penetration, the parasites in the small intestine tend to have little blood in their gastrointestinal tract, suggesting that lack of food for the parasite could be one of the factors that make hookworms penetrate deeper into the intestinal wall and reach the peritoneal cavity. Another factor associated with increased likelihood of peritoneal penetration was the average intestinal villous length, which could indicate that a thinner mucosa is less of a barrier for deeper hookworm penetration or provides lesser possibilities for the nematode to feed.
In this study, we have shown that the parasitic burden is one of the main factors, but probably not the only factor, determining hookworm pathology and mortality. Large numbers of hookworms in the intestine lead to short intestinal villi, which, combined with primary or secondary mild intestinal inflammatory response, allow hookworms to feed deep into the mucosa, facilitating disruption of the intestinal barrier and bacterial translocation into the blood. In addition, in some animals, the large number of nematodes likely depletes the pups’ erythrocyte reserves and capacity for intestinal villous regeneration; therefore, there are decreased resources available for the parasites. This plus the proximity to the submucosa and muscularis due to shortened villi as well as the mild inflammatory response could be some of the factors contributing to hookworm peritoneal penetration.
Footnotes
Acknowledgements
We would like to thank the Quellon artisanal fishermen (Nautilus V, Marimar II, and Matias Jesus crews), the Chilean Navy, and the Guafo Island Chilean Navy lighthouse crews for their logistical support. We greatly appreciate the abnegate work of researchers, volunteers, and students who helped with necropsies and provided logistical support: Dr Hector Paves, Dr Roberto Schlatter, Dr Rodrigo Molina, Dr Felipe DeGroote, Dr Felipe Henriquez, Elvira Vergara, Amanda Hooper, Laura Martinelli, Camila Diaz, Dr Ignacio Silva, and Eugene DeRango. We thank the histology work of Aintzane Alberdi and Patricia Rowe. Research was performed under the authorization of several yearly permits issued by the Chilean sub-secretary of fisheries (SUBPESCA) (last permit number Resolucion exenta N88 del 15 de Enero del 2014).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The field and laboratory work was partially funded by Universidad Austral de Chile, Society for Marine Mammalogy, The Rufford Small Grants Foundation, and Sigma Xi Small Grants in Aid.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
