Abstract
The role of subgroup J avian leukosis virus (ALV J) infection profile in the development of histiocytic sarcomatosis (HS) in chickens was evaluated using retrospective analysis of 2 experiments involving in ovo and at-hatch inoculation of commercial meat-type and ADOL line 0 chickens with 100 or 10,000 TCID50 of various strains ALV J. HS was observed only in persistently viremic, meat-type chickens that were inoculated at hatch, but not in immunotolerized (persistently viremic, with no antibodies), in ovo inoculated chickens. However, the immunotolerized, in ovo inoculated chickens developed a high incidence of myeloid tumors. HS appeared to arise from the splenic ellipsoids and red pulp, and metastasized to liver, kidney, and other organs. The neoplastic cells were diffusely positive for ChL5, CD45, and MHC class II with multifocal infiltration of T and B lymphocytes. Expression of viral antigen gp85 within HS was very low compared with that noted in ALV J-induced myelocytomas. The above observations suggest that the mechanisms of oncogenesis of HS might be different from that of other ALV J-induced tumors.
Histiocytic proliferative lesions have been described in chickens since 1916. 14 The incidence of these lesions was sporadic and variable (up to 90%), and viral etiology was suspected although the exact nature of the causative agent was not identified. In the early 1990s, histiocytic proliferative lesions were observed with increasing incidence in broilers condemned in poultry processing plants as “leukosis” and were described as multicentric histiocytosis (MH). 10 Although MH was reproduced in broilers as well as in specific-pathogen-free (SPF) white leghorn chickens by inoculating homogenates of MH lesions, the etiology remained uncertain. 9
Arshad et al. reported rather similar gross and microscopic lesions in the context of ALV J infections from both field and experimental cases. 1 They characterized these lesions as histiocytic sarcomatosis (HS) based on histochemistry, immunohistochemistry, and transmission electron microscopy. Arshad and coworkers reported a very low incidence of HS in meat-type chickens infected with ALV J at hatch (1.1%). They suggested that the HS lesions may be specific to meat-type chickens and that the absence of HS lesions in meat-type chickens inoculated in ovo may be a result of the low incidence of this tumor type. 1 The objective of the present study was to evaluate the influence of the ALV J infection profile (viremia [V] and virus neutralizing antibody [NAb]) on the development of HS in meat-type chickens.
Information and samples used in the present study were obtained from 2 experiments aimed at studying various aspects of ALV J persistence. 13 In experiment 1, reported elsewhere, 13 174 commercial meat-type chickens were inoculated in ovo at the fifth day of embryonation (via yolk sac route) or at day of hatch (via intra-abdominal route) with either 100 or 10,000 TCID50 with 1 of the 3 ALV J strains (ADOL Hc1, ADOL 4817, ADOL 6803). 8 In experiment 2, 75 commercial meat-type chickens obtained from the same breeder used in experiment 1 and 100 ADOL line 0 white leghorn chickens were infected at hatch (via intra-abdominal route) with 1,000 TCID50 of a molecular clone ALV J termed ADOL pR5-4. 12 Forty-five meat-type chickens inoculated with tissue culture medium were used as negative controls.
In the present study, retrospective analysis of ALV J infection profile (V and NAb) of each chicken was performed using data from the previous 2 experiments, where chickens were tested for V and NAb at 9 time intervals (1, 3, 7, 11, 15, 19, 23, 27, and 32 weeks posthatch). Samples were tested by virus isolation (VI) and virus neutralization (VN) assays as described previously. 13 HS lesions were only observed in meat-type chickens that were inoculated at day of hatch (Table 1). None of the meat-type chickens inoculated in ovo or ADOL line 0 white leghorn chickens had evidence of histiocytic proliferative lesions. Further, 47/68 (69%) of meat-type chickens inoculated in ovo had other types of ALV J–induced tumors (i.e., myelocytomas, nephroblastomas, hemangiosarcomas, and erythroblastosis). All chickens that exhibited HS tested positive for viremia on most, if not all, of the samples collected at various time intervals; 6/15 chickens that had HS tested positive at least once for NAb against the inoculated ALV J strain (Table 2). HS lesions were observed in chickens that had succumbed to disease starting from 11 weeks posthatch until the study was terminated at 32 weeks posthatch; most of these chickens were stunted and appeared lethargic and anemic.
ALV J-induced HS lesions in chickens inoculated in ovo or at hatch with various ALV J strains and doses.
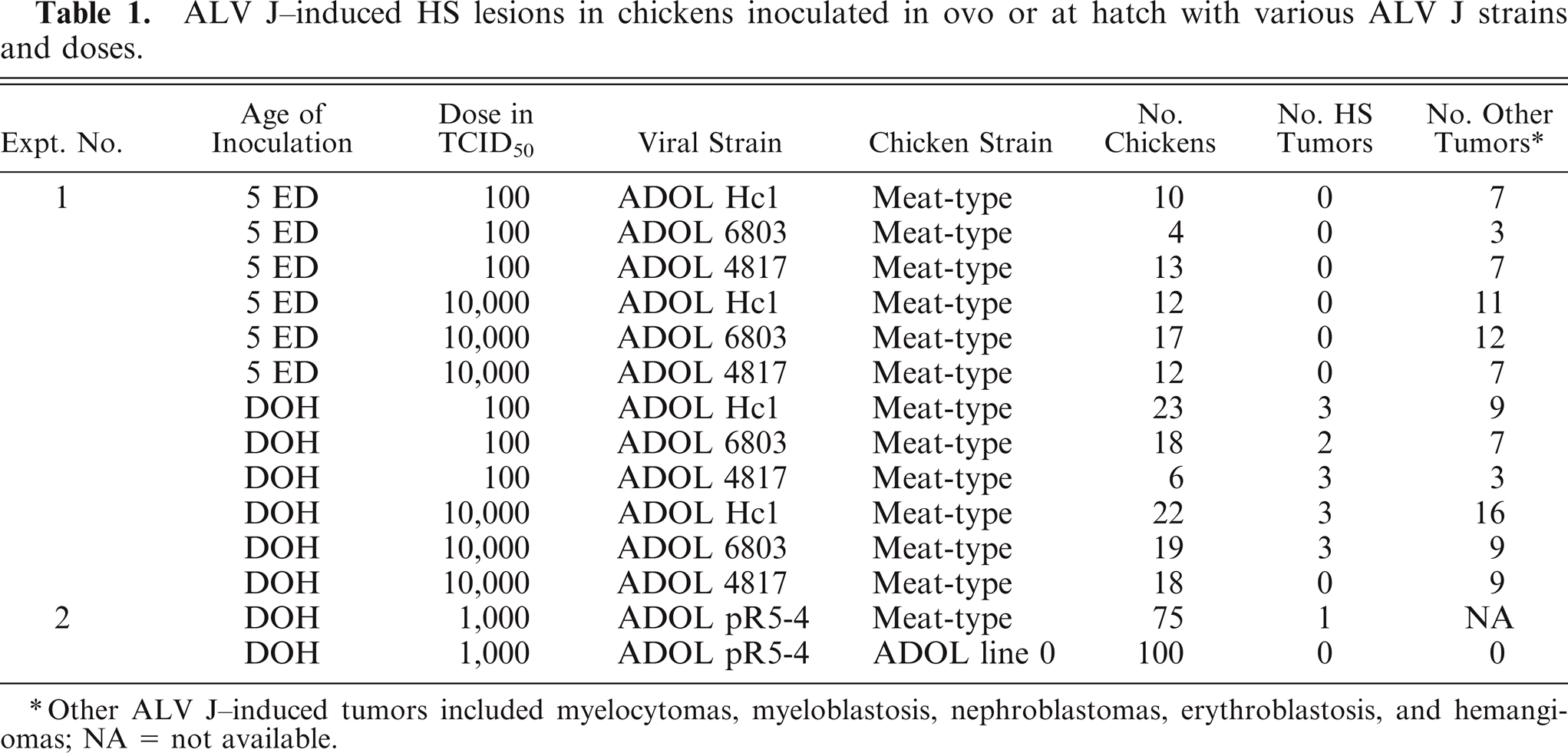
Other ALV J-induced tumors included myelocytomas, myeloblastosis, nephroblastomas, erythroblastosis, and hemangiomas; NA = not available.
ALV J-infection profile (viremia and NAb) in 15 meat-type chickens that developed HS following inoculation with ALV J at hatch.
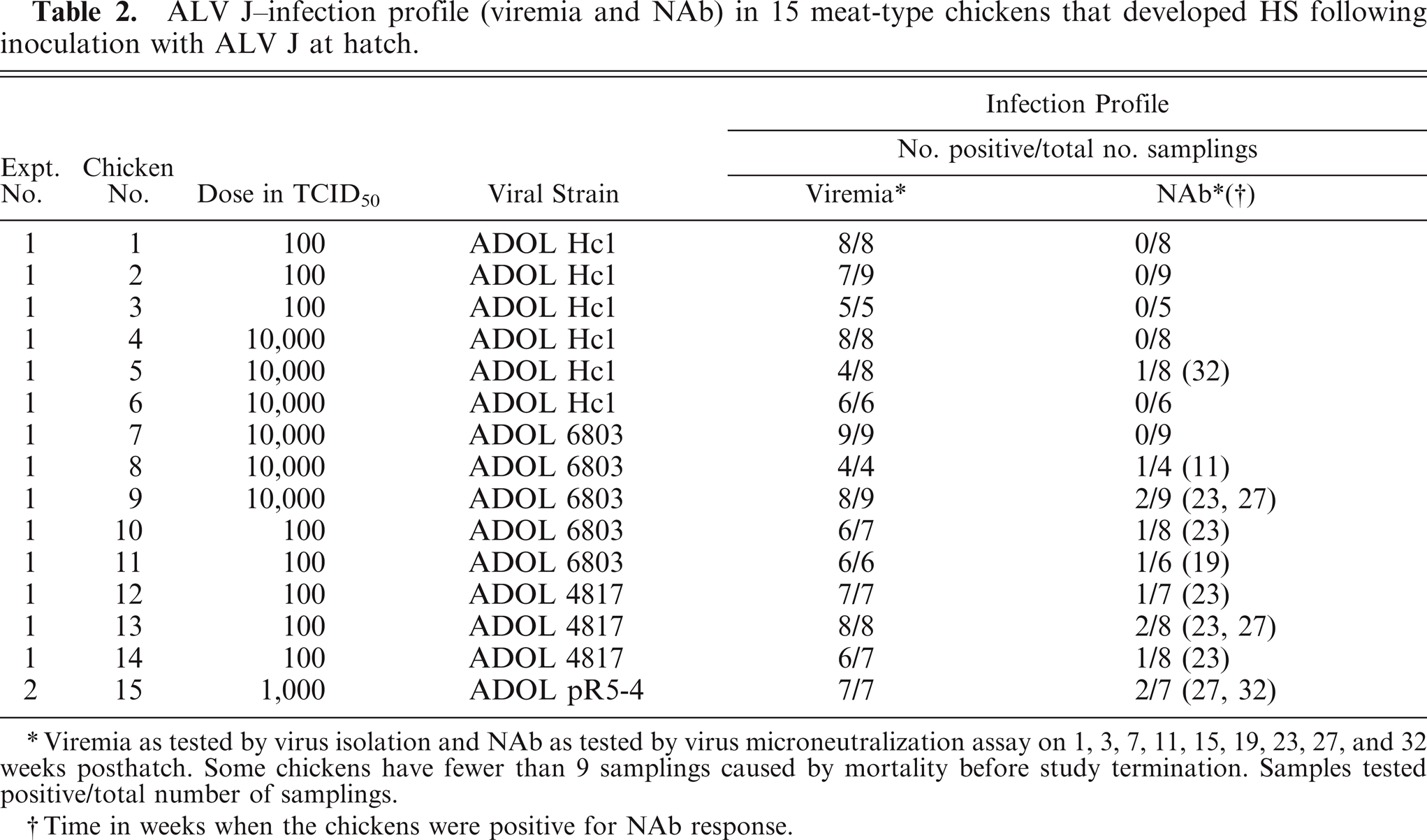
Viremia as tested by virus isolation and NAb as tested by virus microneutralization assay on 1, 3, 7, 11, 15, 19, 23, 27, and 32 weeks posthatch. Some chickens have fewer than 9 samplings caused by mortality before study termination. Samples tested positive/total number of samplings.
Time in weeks when the chickens were positive for NAb response.
In the present study, macroscopic HS lesions were observed in a total of 15 meat-type chickens. Tissues with macroscopic lesions were collected from chickens that succumbed to disease or at study termination at 32 weeks posthatch. Collected tissues were fixed in 10% neutral buffered formalin for microscopic evaluation and embedded in Tissue-Tek O.C.T compound (Sakura Finetek USA, Inc., Torrance, CA) and snap frozen in liquid nitrogen for immunohistochemistry studies. The spleen was consistently (100%) involved in all chickens with HS lesions. In addition, occasionally the liver (50%) and less frequently the kidney (21%), bone marrow (14%), and lungs (7%) exhibited HS lesions similar to those noted in the spleen. There was uniform splenomegaly (2–4×) characterized by diffuse miliary off-white 1–4 mm foci throughout the capsular surface as well as on the cut surface. Similarly, there was hepatomegaly (up to 2–3×) and renomegaly (up to 2×) with similar diffuse miliary off-white foci.
Collected tissues were cut at 5-µm thickness and stained by hematoxylin and eosin (HE), Snook's reticulin, and Van Gieson's trichrome methods. Microscopically, the splenic lesions consisted of multifocal to coalescing, unencapsulated circumscribed nodules that appear to expand the red pulp as well as the ellipsoid sheaths. In some cases, the splenic architecture was diffusely effaced by the proliferating neoplastic histiocytic population (Fig. 1). Within the liver, the neoplastic cells infiltrated numerous hepatic sinusoids and formed histiocytic nodules (Fig. 2). There were proliferating neoplastic emboli implanted on the intimal surface of the portal vein (Fig. 3). Similar neoplastic cells were also observed within other organs involved. The histiocytic proliferative lesions described in the present study were morphologically similar to earlier reports. 1 The neoplastic cells exhibited pleomorphic morphology ranging from plump spindle-shaped cells to large foamy epithelioid histiocytes that exhibited rare erythrophagocytosis (Fig. 4). In some sections, the histiocytic proliferations were accompanied by numerous neoplastic cells of myeloid lineage viz. myelocytes and myeloid stem cells. Infiltration of variable numbers of lymphocytes, plasma cells, and heterophils was also observed in some histiocytic proliferative lesions (Figs. 2, 4). Snook's reticulin stain depicted large open spaces devoid of reticulin within the proliferative areas (data not presented). Collagen as demonstrated by Van Gieson's trichrome stain was minimal to none in all cases (data not presented).
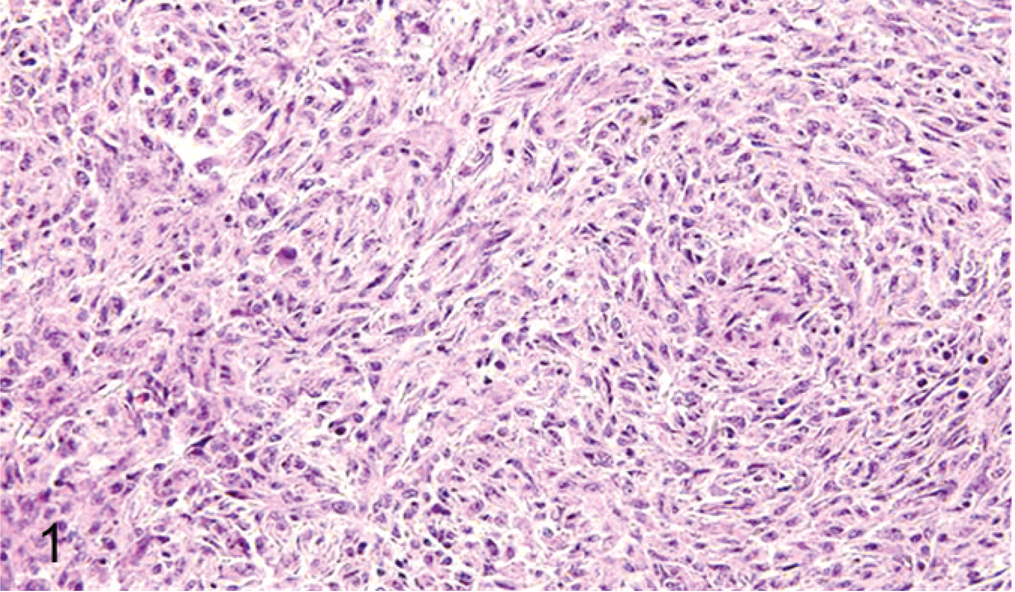
Spleen; the neoplastic histiocytic population with a prominent spindle cell morphology diffusely effacing the splenic architecture. HE stain.
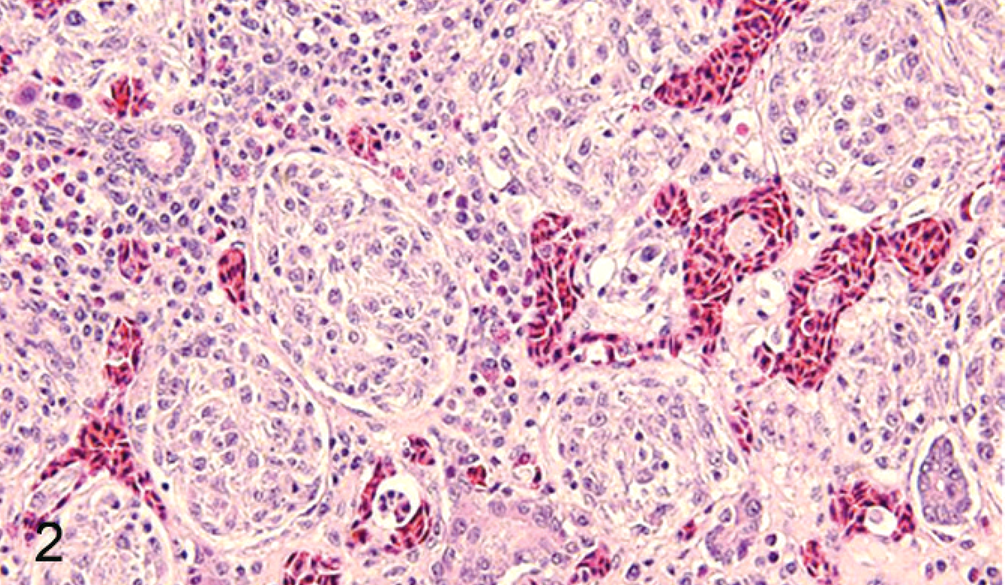
Liver; multifocal circumscribed, unencapsulated histiocytic nodules expanding the hepatic sinusoids in the periportal areas. In addition, there is mild to moderate infiltration of lymphocytes and plasma cells. HE stain.
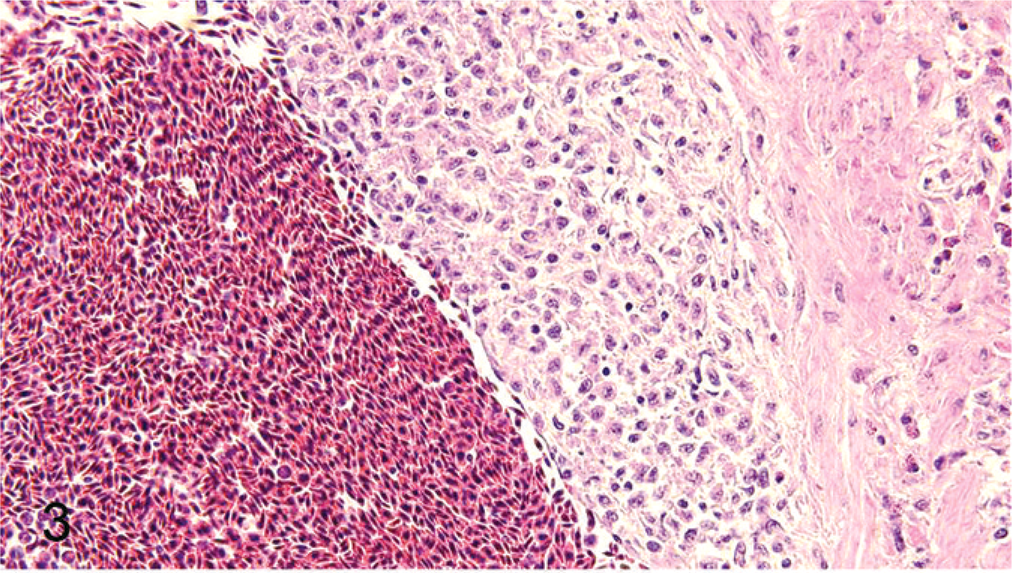
Liver; metastatic histiocytic emboli on the intimal surface of the portal vein. HE stain.
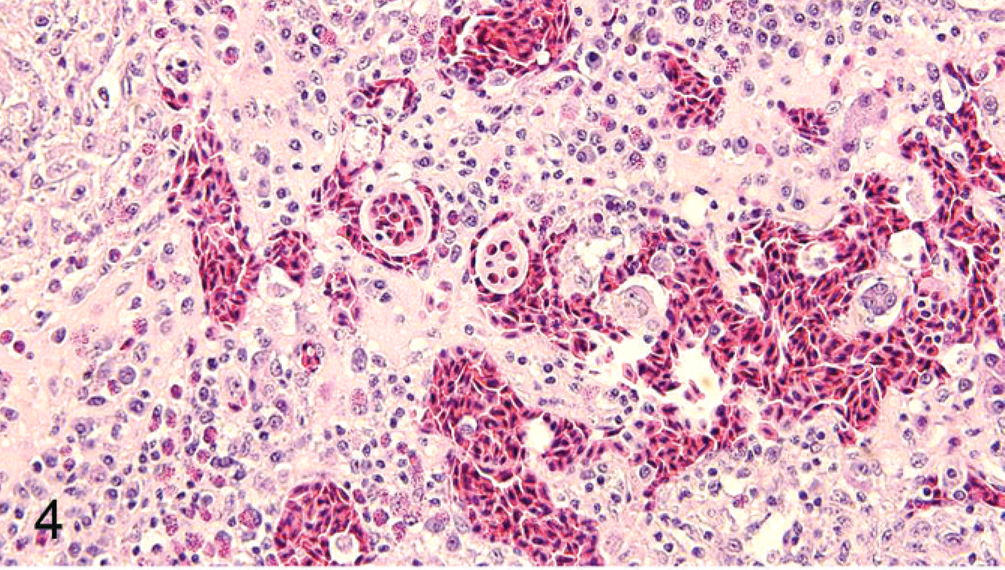
Spleen; the neoplastic histiocytes exhibit plump spindle shaped to large foamy epithelioid morphology accompanied by erythrophagocytosis. In addition, there is mild to moderate infiltration of lymphocytes, plasma cells, and heterophils within the section. HE stain.
Cryosections were stained with a panel of 10 monoclonal antibodies (MAbs) specific for various chicken antigens (Table 3). A modified avidin–biotin–peroxidase complex method using the Vectastain ABC kit (Vector Laboratories Inc., Burlingame, CA) was performed. Tissues were scored as 0 (no positive cells), 1 (a few scattered positive cells), 2 (moderate number of positive cells), and 3 (large number of positive cells). The proliferating cells stained strongly positive with antibodies ChL5 2 (myelomonocytic cells and activated T lymphocytes) within the spleen (Fig. 5) and liver (Fig. 6), with K55 6 (common leukocyte antigen CD45) within the spleen and liver, and with CIa 7 (MHC II) within the spleen and liver. The proliferating cells did not stain with CB4/CB5 5 (B lymphocytes) and p53 (Abcam, San Francisco, CA) antibodies. In addition, within the proliferating histiocytic nodules in the spleen, there was infiltration of variable proportions of cells staining positive with CT3 4 (CD3), CT8 3 (CD8), CT4 3 (CD4), and G2-3 15 (ALV J gp85) (Figs. 7, 8) MAbs.
Immunophenotype of the ALV J-induced histiocytic sarcoma tumor cells.*

Immunohistochemistry was done on frozen samples from 6 chickens, and the chicken number corresponds to the chickens in Table 2. Tissues were scored as 0 (no positive cells), 1 (a few scattered positive cells), 2 (moderate number of positive cells), and 3 (large number of positive cells). K55 = common leukocyte antigen CD45; ChL5 = myelomonocytic cells and activated T cells; CIa = major histocompatibility class II; CT3 = CD3 specific for all T cells; CT4 = CD4 T cells; CT8 = CD8 T cells; CB/CB5 = peripheral B cells; C2-3 = ALV J gp85 antigen; ML1/ML2 = myelocytoma tumor samples.
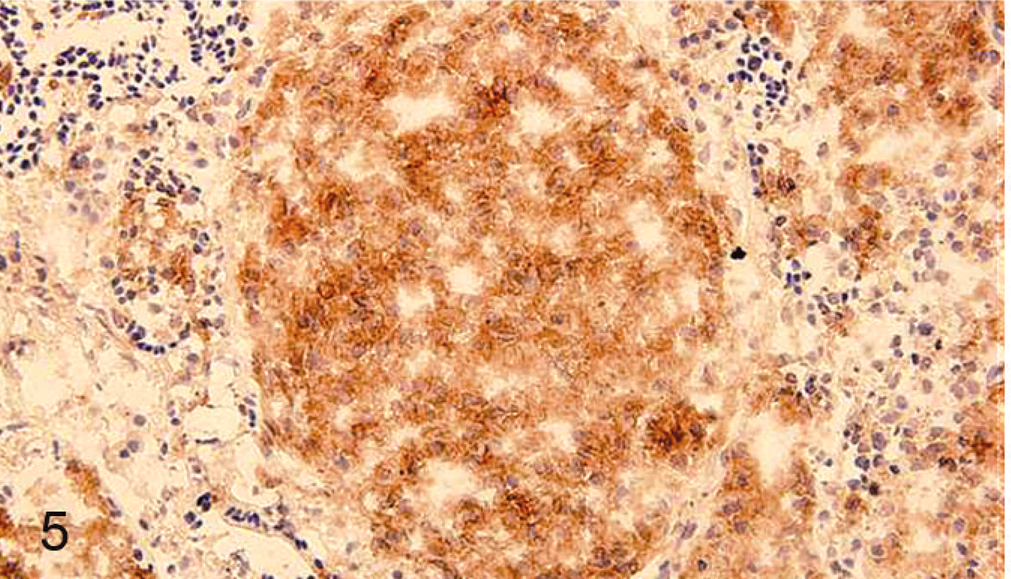
Spleen; the histiocytic nodules within the spleen are diffusely positive for ChL5. Immunoperoxidase staining for ChL5 on cryosection, DAB chromogen, hematoxylin counterstain.
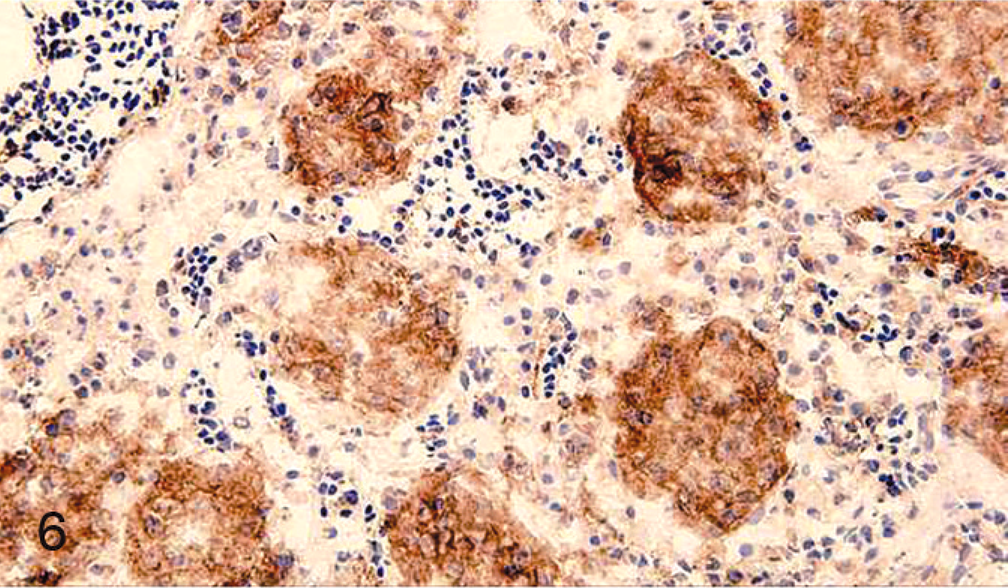
Liver; multifocal histiocytic nodules are diffusely positive for ChL5. Immunoperoxidase staining for ChL5 on cryosection, DAB chromogen, hematoxylin counterstain.
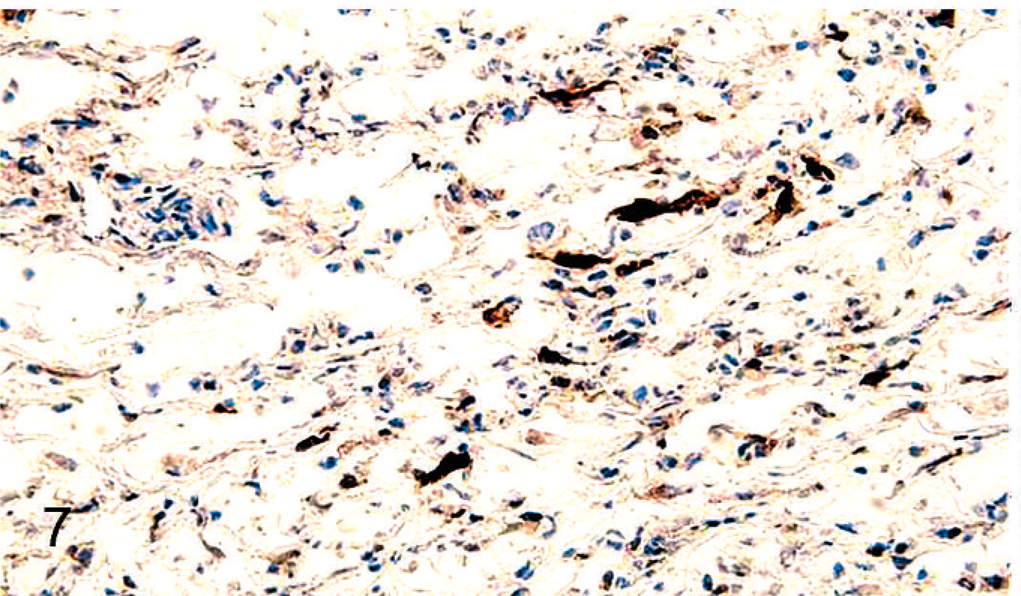
Spleen; scattered multifocal cells expressing G2-3 (ALV J gp85) antigen within the splenic histiocytic nodules. Immunoperoxidase staining for G2-3 on cryosection, DAB chromogen, hematoxylin counterstain.
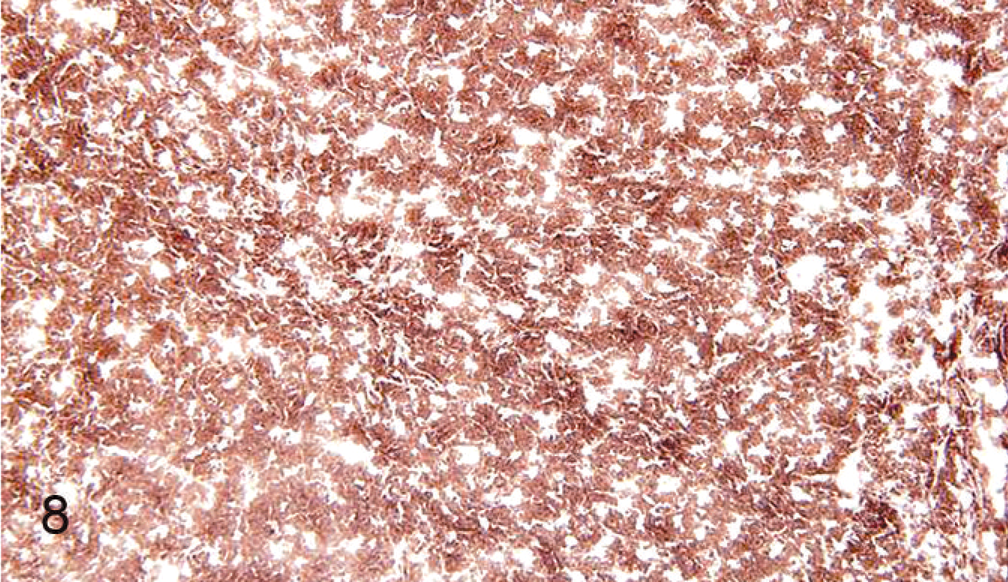
ALV J induced–myelocytoma; diffuse expression of G2-3 (ALV J gp85) antigen within the myelocytes in the myelocytoma. Immunoperoxidase staining for G2-3 on cryosection, DAB chromogen, hematoxylin counterstain.
Chickens inoculated in ovo with ALV J are usually immunotolerized, whereas chickens inoculated at day of hatch may be persistently viremic but are usually capable of responding immunologically against the inoculated virus. 13 In the present study, all the chickens with HS lesions were inoculated at day of hatch and were persistently viremic but were not immunotolerant to ALV J. They were able to mount an immune response, albeit an inefficient one, as evident by the presence of NAbs in 9/15 (60%) of chickens with HS; also, the infiltration of affected tissues with numerous plasma cells, lymphocytes and heterophils suggests mounting of an immune response. The reason that 6/15 (40%) of chickens with HS lesions did not develop any detectable NAb during the several sampling intervals is not known. It is possible that the VN assay used in the present study was not sensitive enough to detect low levels of antibody. It has been shown that although some ALV J–infected meat-type chickens have detectable antibody-forming cells within the spleen, they were unable to clear viremia. 16 At least in some chickens, the continued presence of the virus (or viral antigen) in the face of a patent immune system may cause reactive hyperplasia of splenocytes (reticular cells) that proliferate and may eventually transform. This hypothesis may be valid because HS has never been reported in immunotolerant, persistent viremia white leghorns, white rocks, or meat-type chickens that were infected in ovo with ALV J. In addition, HS lesions have not been observed in white leghorns, white rocks, or even meat-type chickens capable of clearing ALV J–induced viremia by mounting efficient NAb responses over a prolonged period of time against the inoculated virus strain. 1
ALV J–induced neoplasms in meat-type chickens include myelocytomas, erythroblastosis, myeloblastosis, nephroblastomas, hemangiosarcomas and HS. 13 The incidence of ALV J–induced tumors other than HS is usually higher (70%) in in ovo inoculated chickens than in chickens inoculated at hatch (50%). 13 In the present study, HS was not observed in any of the in ovo inoculated chickens (Table 1). In addition, HS lesions had very low expression of ALV J gp85 antigen (Fig. 7), compared with high gp85 expression in ALV J–induced myelocytomas (Fig. 8). Arshad et al. propose that ALV J–induced HS may be caused by a neoplastic transformation by a nonproductive infection of the histiocytes. 1 However, based on our results, we propose that reactive hyperplasia of splenocytes as a result of viral persistence is an important step before eventual neoplastic transformation in the pathogenesis of HS. Evaluation of HS tumors for upregulation of c-myc and other cellular oncogenes (as seen in other ALV–induced tumors 11 ) as well as prospective studies based on the above viral persistence data may help in elucidating the mechanism of oncogenesis of HS tumors.
Based on the immunophenotype, the neoplastic histiocytes were of myelomonocytic origin because they were positive for ChL5 (myelomonocytic cells and activated T cells), K55 (common leukocyte antigen CD45), and CIa (MHC II) and negative for CT3 (CD3), CT4 (CD4), CT8 (CD8), B4, and B5 (B cells). Arshad and coworkers, using a different set of monoclonal antibodies as well as transmission electron microscopy, demonstrated that the splenic histiocytic nodules comprised a predominance of cells of monocyte/macrophage lineage and of CD4- and CD8-positive lymphocytes. 1 Our results not only confirm the findings of Arshad and coworkers, but also provide evidence that the neoplastic cells within the liver are identical to that of the splenic HS lesions. The histiocytic proliferative lesions in the present study as well as in previous reports 1 consistently involved spleen along with frequent to occasional involvement of other organs. ALV J–induced HS appears to have originated in the splenic tissue as evident by the consistent splenic involvement as well as similar microscopic morphology and immunophenotype of neoplastic cells within the spleen and liver (Figs. 1, 2, 5, 6). In addition, invasion of the blood vessels by the neoplastic histiocytes and presence of neoplastic emboli within the liver (Fig. 3) suggest the possibility of metastasis of neoplastic histiocytes from spleen to other organs.
In summary, data from the present study expand the knowledge on factors that influence the pathogenesis of ALV J–induced HS. Only persistently viremic meat-type chickens that were infected at hatch developed ALV J–induced HS. The phenotype of HS tumor cells was confirmed to be histiocytes of myelomonocytic origin. Consistent splenic involvement with occasional neoplastic emboli within the liver as well as similar immunophenotype of the neoplastic cells within the spleen and liver suggest a primary splenic origin for these tumors. In addition, several differences were observed between HS and other ALV J–induced tumors that suggest different oncogenic mechanisms. Further studies are required to determine if the ALV J–induced HS lesions begin as a reactive process in the spleen against persistent viral load but later progress to neoplasia or are the result of a direct transformation event induced by ALV J similar to myelocytomatosis or myeloid leukosis. The ALV J–induced HS lesions seem to share several histologic features with some mammalian histiocytic proliferative diseases. These viral-induced HS may serve as a comparative model in humans and in other animals.
