Abstract
We investigated the histologic findings and viral antigen distribution in 3 cases of natural coinfection of layer hens with subgroup J avian leukosis virus (ALV-J), Marek’s disease virus (MDV), and reticuloendotheliosis virus (REV) in hens. At autopsy, diseased hens were found to have hepatosplenomegaly and thickened proventriculi, with white tumor nodules in the liver, spleen, lung, kidney, and ovary. Microscopically, most tissues had been infiltrated by neoplastic lymphocytes; the spleen, lung, proventriculus, heart, and liver had been infiltrated by both neoplastic lymphocytes and myeloblastic cells and/or primitive reticular cells. Fluorescence multiplex immunohistochemistry staining revealed ALV-J, MDV, and REV antigens co-expressed in the same tissue, even the same cell.
Keywords
Avian leukosis virus (ALV; family Retroviridae, genus Alpharetrovirus), Marek’s disease virus (MDV; family Herpesviridae, genus Mardivirus, species Gallid alphaherpesvirus 2), and reticuloendotheliosis virus (REV; family Retroviridae, genus Gammaretrovirus) are among the naturally occurring viruses that cause contagious, immunosuppressive, and oncogenic diseases in chickens. Coinfection with 2–3 viruses has become an important epidemiologic situation in poultry production.2,9,14,15,17 Many reports have described and compared histologic changes associated with single virus infection;1,7,8 however, histologic findings and antigen distribution from naturally occurring cases of coinfection have been reported only rarely.
Between October 2017 and April 2018, 3 flocks belonging to 3 different chicken layer farms from Anhui province in China experienced a mild increase in mortality. Sixteen diseased hens with depression, emaciation, white combs, and ruffled feathers from 3 flocks (5 or 6 diseased layer hens per farm, 130 or 180 d old; Table 1) were submitted to Anhui Agricultural University Animal Disease Diagnostic Laboratory and Animal Pathology Laboratory for autopsy and tumor diagnosis. All chickens from these 3 cases had been vaccinated with the MD CVI988/Rispens vaccine (attenuated serotype 1 MDV; Merial, Nanjing, China) at 1 d of age using the same standard operating procedures. Postmortem examinations were performed on 5 or 6 sick and deceased hens per case. Clinical signs in 3 cases presented for autopsy included extreme weight loss, prominent sternums, fat loss, and thin abdomens. Case 1 had white nodules in the liver, spleen, kidney, lung, pancreas, proventriculus, duodenum, ovary, and heart. Case 2 had marked hepatosplenomegaly (2–3 times normal size). Case 3 had white nodules in the liver and spleen (Table 1). Samples of liver, spleen, lung, kidney, heart, pancreas, duodenum, proventriculus, brain, sciatic nerve, and ovary were collected from all submitted birds. One portion of each tissue was fixed in 4% paraformaldehyde, and histologic sections were routinely prepared for hematoxylin and eosin staining and fluorescence multiplex immunohistochemistry (mIHC) staining to determine the expression of viral antigens. Another portion of each tissue was stored at −80°C for subsequent nucleic acid (viral RNA and DNA) extraction for PCR and reverse transcription PCR (RT-PCR) testing.
Histologic findings and viral antigen distribution in 3 cases of hens coinfected with subgroup J avian leukosis virus (ALV-J), Marek’s disease virus (MDV), and reticuloendotheliosis virus (REV).
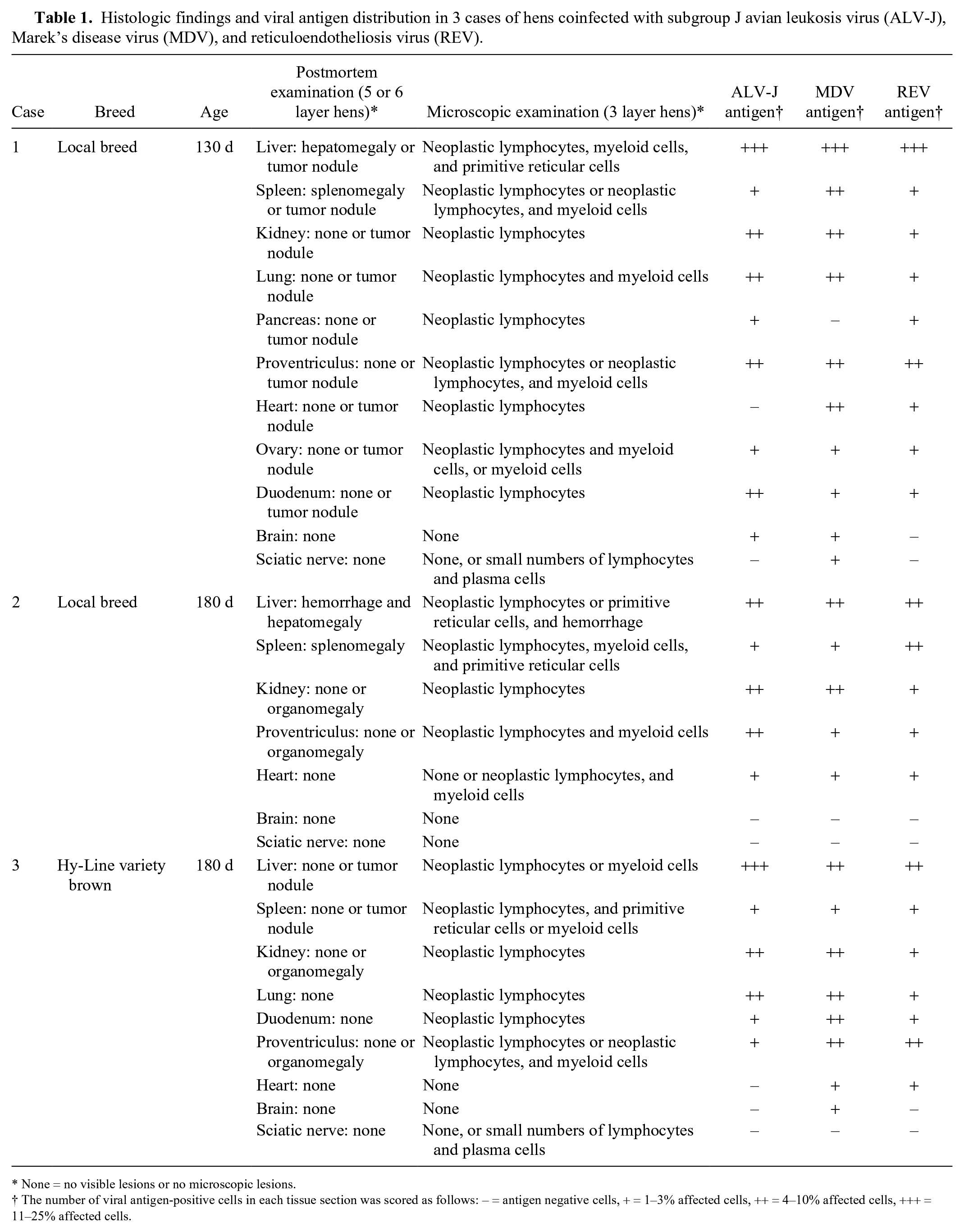
None = no visible lesions or no microscopic lesions.
The number of viral antigen-positive cells in each tissue section was scored as follows: – = antigen negative cells, + = 1–3% affected cells, ++ = 4–10% affected cells, +++ = 11–25% affected cells.
DNA and RNA were isolated from these samples (DNA and RNA extraction kit; Takara Biotechnology, Dalian, China) according to the manufacturer’s instructions. Specific primers3,5,11,14 (Supplementary Table 1) and viral nucleic acid templates were used to detect the suspected viruses, including ALV subtypes A, B, and J (ALV-A, ALV-B, and ALV-J), avian hepatitis E virus (aHEV; family Hepeviridae, genus Orthohepevirus, species Orthohepevirus A, B, C, and D; also known as big liver and spleen disease virus, BLSV), MDV, and REV, all of which may be associated with the observed clinical phenotype.
All chickens were found to be free of ALV-A, ALV-B, and aHEV, and were coinfected with ALV-J, MDV, and REV, based on PCR and RT-PCR results. Although 3 flocks had been vaccinated with CVI988 at 1 d of age, these flocks were still infected with very virulent MDV by phylogenetic analysis based on meq gene sequencing 11 (Supplementary Fig. 1).
Microscopically, most of the tissues had been infiltrated by neoplastic lymphocytes (Table 1). For example, the tumor nodules in the spleen of case 3 were composed of pleomorphic, neoplastic lymphocytes that effaced normal splenic structure (Fig. 1A); the ovary of case 1 had been infiltrated by myelocytes with eosinophilic cytoplasmic granules (Fig. 1B). The spleen, lung, proventriculus, and liver had been infiltrated by both neoplastic lymphocytes and myeloid cells (Table 1, Fig. 1C). Three types of tumor, including neoplastic lymphocytes, myeloid cells, and primitive reticular cells (relatively large cells with mitoses and round or oval nuclei), 10 were observed in the liver of case 1 and the spleen of case 2. Moderate-to-severe congestion of the liver and primitive reticular cells were observed in case 2 (Fig. 1D). Most of the neoplastic cell populations were almost “pure” (consisted of lymphoblastic, small lymphocytes with mitotic figures), but there were also some unidentified tumors (not included in Table 1). Although neoplastic invasion of nervous tissue was not observed in these birds, small numbers of inflammatory cells (lymphocytes and plasma cells) infiltrated the sciatic nerve in cases 1 and 3, consistent with MDV infection.
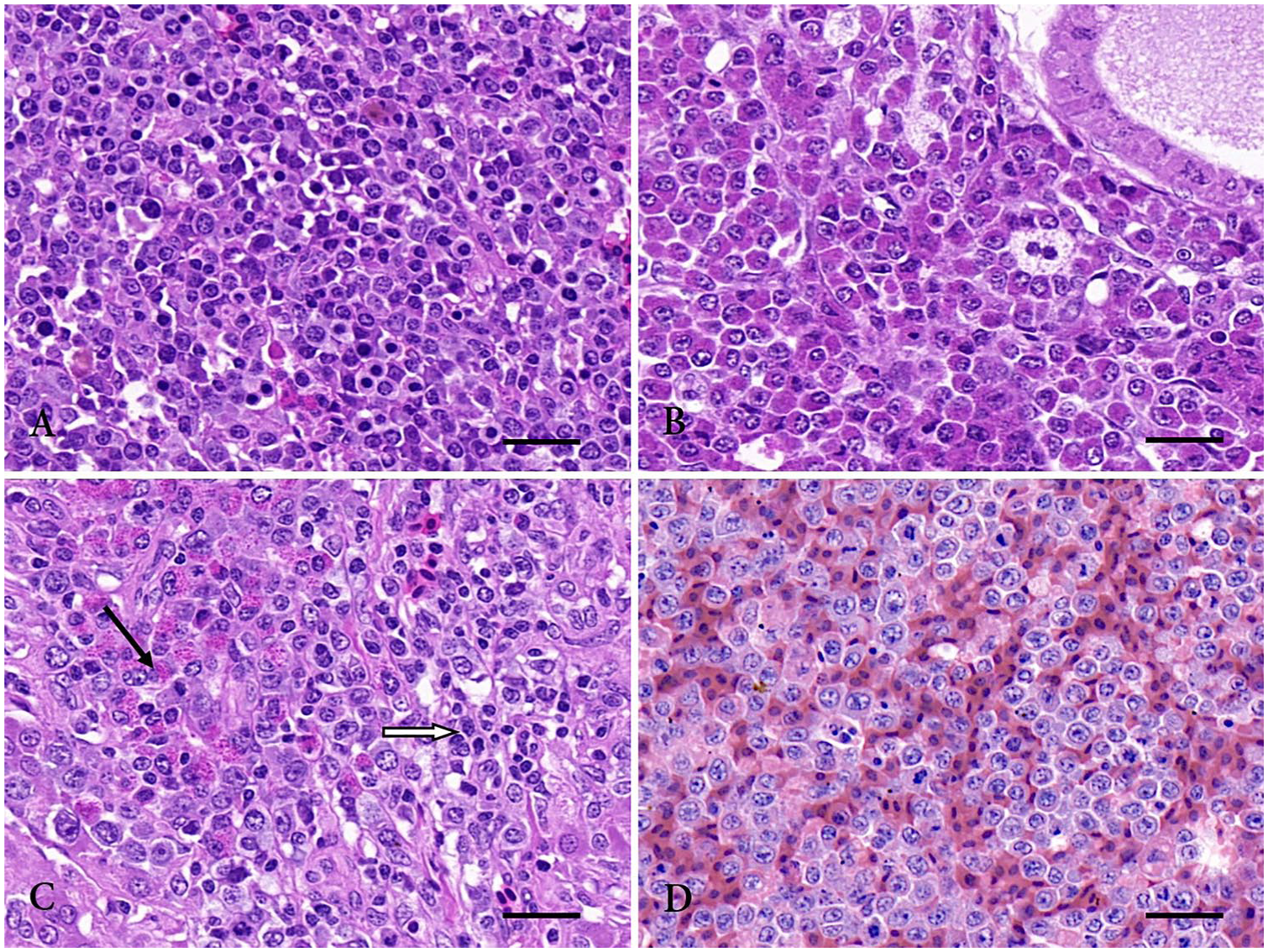
Histologic changes of layer hens coinfected with subgroup J avian leukosis virus (ALV-J), Marek’s disease virus (MDV), and reticuloendotheliosis virus (REV). H&E.
Previous studies have described histologic findings of coinfection with oncogenic viruses.2,5 However, few studies have reported the findings associated with ALV-J, MDV, and REV co-localization in the same tissue or cell.2,3,11,16 Instead of traditional IHC, we used fluorescence mIHC with tyramide signal amplification (TSA), a methodology that enables simultaneous detection of multiple proteins of interest in a given tissue section. To study the co-localization of antigens from ALV-J, MDV, and REV coinfection, all tissues examined by routine histology were also examined by fluorescence mIHC staining.6,12 Paraffin-embedded tissue sections were sectioned and successively incubated with the anti-MDV meq diluted 1:200 in blocking buffer, followed by application of a horseradish peroxidase–conjugated goat anti-mouse IgG secondary antibody (Sercicebio, Wuhan, China), and subsequently incubated with fluorescein isothiocyanate–conjugated tyramide reagent (Sercicebio; green fluorescence); sections were rinsed with washing buffer after each step. Following the heat-stable deposition of fluorescein isothiocyanate–conjugated tyramide around the MDV antigen sites, sections were again treated by microwave for heat-induced epitope retrieval to strip the primary and secondary antibodies bound to the tissue; the above steps were repeated with the anti–ALV-J gp85 (1:150) and Cy3-conjugated tyramide reagent (Sercicebio; red fluorescence); and with the anti-REV gp90 (1:200) and Cy5-conjugated tyramide reagent (Sercicebio; pink fluorescence). Finally, nuclei were counterstained with 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI; Sercicebio; blue fluorescence). Liver from an uninfected chicken from a healthy hen flock was used as negative control.
Fluorescence mIHC staining revealed that the highest number of cells positive for ALV-J, MDV, and REV in all tissues from all birds were present in the liver, followed by kidney, lung, and proventriculus (Table 1). Co-expression of ALV-J and REV antigen, MDV and REV antigen, ALV-J and MDV antigen, and of ALV-J, MDV, and REV antigen were observed in cases 1–3. For example, co-expression of ALV-J and MDV antigen were observed in the same population of lymphocytes that formed a large cuff around a medium-sized vessel; REV antigen was located in the hepatocytes of the liver from case 1 (Figs. 2A–D). ALV-J, MDV, and REV antigen were widely distributed in the same foci in the proventricular gland cells from case 2 (Supplementary Fig. 2). Our observations indicate that ALV-J, MDV, and REV coinfect the same tissue and cell, and the coexistence of 3 viruses in the same tissue and cell may provide conditions for the recombination and mutation of viruses in vivo.4,13
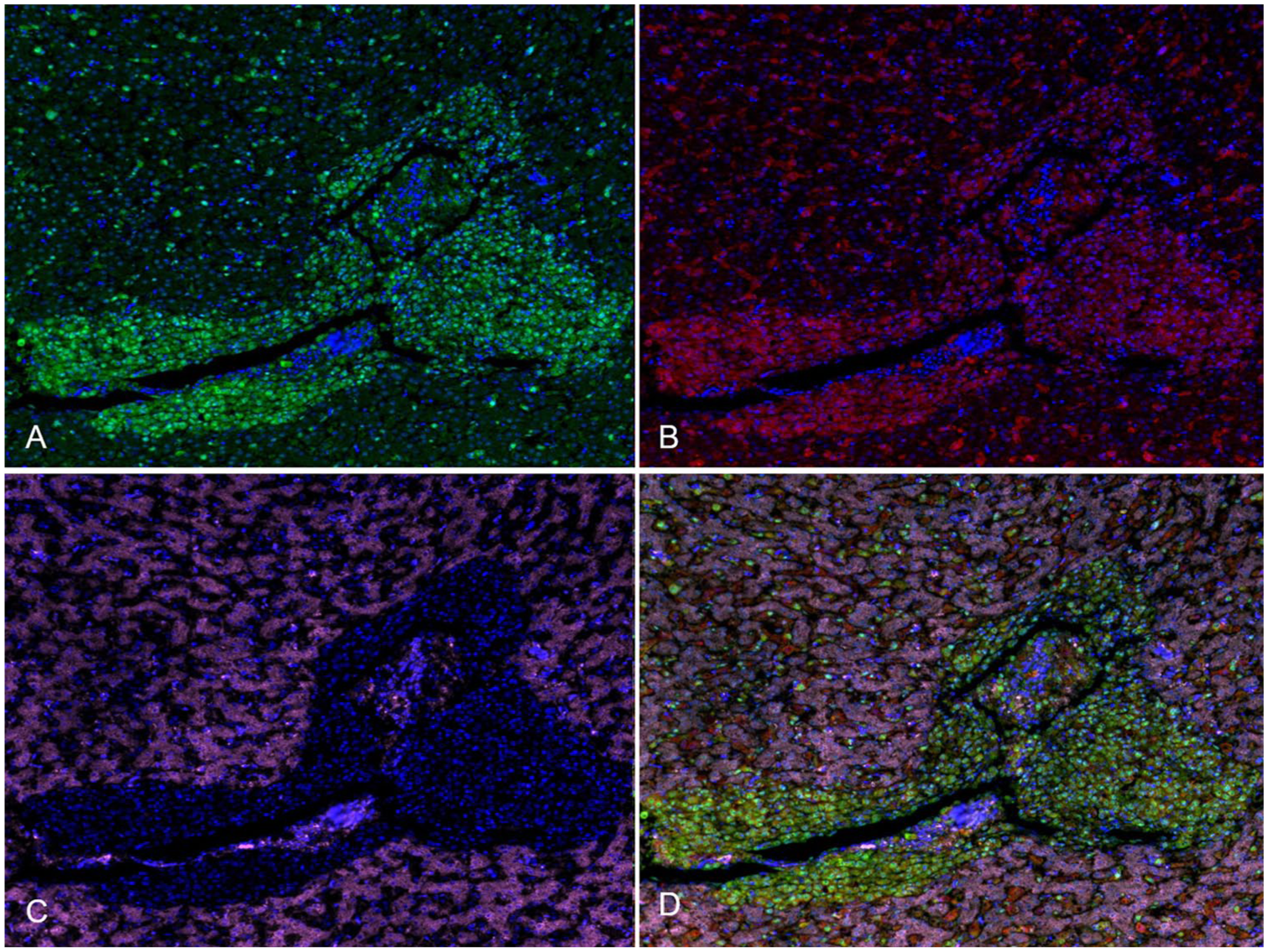
Fluorescence multiplex immunohistochemistry localization of subgroup J avian leukosis virus (ALV-J), Marek’s disease virus (MDV), and reticuloendotheliosis virus (REV) antigens in chicken liver.
Supplemental Material
DS1_JVDI_10.1177_1040638719868274 – Supplemental material for Histologic findings and viral antigen distribution in natural coinfection of layer hens with subgroup J avian leukosis virus, Marek’s disease virus, and reticuloendotheliosis virus
Supplemental material, DS1_JVDI_10.1177_1040638719868274 for Histologic findings and viral antigen distribution in natural coinfection of layer hens with subgroup J avian leukosis virus, Marek’s disease virus, and reticuloendotheliosis virus by Hongmei Liu, Kui Ma, Miaomiao Liu, Chengcheng Yang, Xueting Huang, Yu Zhao and Kezong Qi in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Drs. Aijian Qin and Hongjun Chen for kindly providing anti–ALV-J gp85, anti-REV gp90, and anti-MDV meq monoclonal antibodies.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the Academic Backbone Training Project of Anhui Agricultural University (grant 2014XKPY-25) and Natural Science Foundation of China (grants 31872445 and 31772707).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
