Abstract
The purpose of this study was to determine the immunophenotype and histologic classification of 50 cases of feline gastrointestinal lymphoma. Classification was determined using the National Cancer Institute Working Formulation and the Revised European and American Lymphoma/ World Health Organization classification system. Tissue sections were stained with HE, phosphotungstic acid hematoxylin, anti-CD3, anti-CD79a, and anti-BLA.36. Overall, B-cell tumors predominated at 54% (27/50), including 16 diffuse large with immunoblastic nuclear type, 2 diffuse large with centroblastic nuclear type, 3 small lymphocytic, 4 lymphocytic intermediate type, and 2 T-cell-rich large B-cell lymphomas. T-cell tumors comprised 38% (19/50), including 15 epitheliotropic small lymphocytic and 4 lymphoblastic. Three tumors (6%) were nonreactive for B- and T-cell markers and had eosinophilic cytoplasmic granules when stained with HE. Gastric tumors were diagnosed in 24% (12/50) of cats, and 18% (9/50) were present only in the stomach. All gastric lymphomas were of B-cell lineage. Small intestinal lymphoma predominated, with 74% (37/50) of cats affected: T-cell tumors comprised 52% (19/37); 38% (14/37) were B-cell tumors; 8% (3/37) were nonreactive for B- and T-cell markers; and 2% (1/37) expressed both CD3 and BLA.36. Of the 8 cats (16%) that had lymphoma of the large intestine, 88% (7/8) had B-cell tumors and 12% (1/8) had T-cell tumors. The strongest association between gastrointestinal lymphoma immunophenotype, histologic classification, and location occurred in the stomach, where there was a predominance of diffuse large B-cell lymphoma of immunoblastic nuclear type.
Lymphoma is the most common neoplasm in cats, 4, 9, 12, 19, 25, 28, 30, 38 accounting for 50 to 90% of all feline hematopoietic neoplasms. 37 Further, since the widespread implementation of test/euthanize and vaccination programs for the control of feline leukemia virus (FeLV), 21, 37, 38 the gastrointestinal tract (GIT) has been reported by several authors to be the most frequently recognized anatomic location of disease. 4, 12, 17, 19, 28 Cats with GIT lymphoma have a median age of 12 years, 37 and they are generally FeLV negative. 28, 37 Most authors report that within the GIT, B-cell tumors predominate over T-cell tumors and tumors of large granular lymphocytes (LGLs). 11, 18, 27, 38, 47 However, the literature is contradictory, and a predominance of T-cell lymphomas also has been reported. 52 Even so, to date, there are insufficient data to correlate tumor phenotype with clinical significance and outcome. Furthermore, to the best of these authors' knowledge, there are no published reports that correlate anatomic location and phenotype of feline GIT lymphomas.
The purpose of this study was twofold: to characterize feline GIT lymphoma with regard to histologic grade, phenotype, and anatomic location and thereby determine the types of lymphoma that occur within the GIT of cats, and to assess the correlation between tumor phenotype and GIT location.
Materials and Methods
Case Selection and Study Design
This was a retrospective, cross-sectional study. Case information was compiled from tissues submitted to the Auburn University surgical pathology service between 1993 and 2005. Only cats with a definitive histopathologic diagnosis of gastrointestinal lymphoma were included in the study. In this study, “gastrointestinal” refers to the stomach and the small and large intestine, with the large intestinal category also including the cecum. All tissues were fixed in 10% neutral-buffered formalin. Four-micron sections were cut and routinely prepared for HE staining. Pathology reports and glass slides were reviewed for all available cases. A total of 201 HE-stained slides from 58 cases were examined. Eight cases were excluded from the study because of insufficient data, insufficient tissue, or equivocal diagnosis upon review. For the 50 cases of GIT lymphoma chosen for study, representative sections and their corresponding paraffin blocks were selected for further assessment. Recorded data included submission date, case number, signalment, state of residence, feline immunodeficiency virus (FIV) and FeLV infection status, and all known sites of tumor invasion. In some cases, tissue samples were collected more than once from the same animal. In these cases, all samples were examined, and representative pretreatment sections were selected for assessment.
The study was divided into 3 parts. The first part involved characterization of each tumor using the National Cancer Institute Working Formulation (NCI WF) 1 and the dichotomous algorithm published by Carter et al. 5 Part 2 involved phenotypic analysis of tumors that was performed independently of data collected in part 1. Anatomic location of tumors within the GIT was determined predominantly through a combination of patient history and extensive clinical work-up, including physical examination, imaging, and exploratory laparotomy and/or laparoscopy with histopathology, and in 6 cases was confirmed by necropsy. Tumors that were determined to involve only 1 region of the GIT, whether or not there was local lymph node invasion, were categorized as solitary to that specific region of the GIT. In part 3, tumor morphology and anatomic location were combined with phenotype. By use of the criteria provided in the Armed Forces Institute of Pathology's International Histologic Classification of Hematopoietic Tumors of Domestic Animals, 41 each tumor was classified according to the Revised European and American Lymphoma/World Health Organization (REAL/WHO) classification system. In the category of diffuse large B-cell lymphoma, differentiation of the 2 subclassifications, centroblastic and immunoblastic, was based on established criteria. 10, 31 Briefly, diffuse large B-cell lymphoma with immunoblastic nuclear type was defined as being composed of at least 90% immunoblasts, which are characterized by a prominent, single, central nucleolus and abundant cytoplasm. Diffuse large B-cell lymphoma of centroblastic nuclear type is composed of sheets of centroblasts, which are characterized by one to several peripheral nucleoli and a narrow rim of cytoplasm; these tumors may contain variable numbers of immunoblasts. 10, 20, 31, 36
Immunohistochemistry
Monoclonal mouse anti-human CD3 (clone F7.2.38) and monoclonal mouse anti-human CD79αcy (clone HM57; CD79a) were purchased from DAKO (Carpinteria, CA). Monoclonal mouse anti-human B-lymphocyte antigen 36 (BLA.36) was purchased from Gene Tex Inc. (San Antonio, TX).
For all 50 cases, 4-micron sections were prepared from representative paraffin-embedded tissue blocks and processed for immunohistochemistry using antibodies against the pan–T-lymphocyte marker CD3, the pan–B-lymphocyte and plasma cell marker CD79a, 7, 46 and the human B-lymphocyte antigen 36 (BLA.36), which is expressed by early and activated B cells as well as Reed-Sternberg cells of Hodgkin's lymphoma in humans. 7, 8
Antigen retrieval for CD3 and CD79a was achieved by use of the manufacturer's protocol for steamer retrieval (Declere Ready-to-Use One-Step Procedure; Cell Marque, Hot Springs, AR) using a commercial steamer (model HS800; Black and Decker, Towson, MD). Control tissue was canine tonsil. Antigen retrieval for BLA.36 was conducted as follows. Four-micron sections were placed on charged slides and baked at 60°C overnight, and tissues were processed in a routine manner. Microwave enhancement was achieved as follows. Solutions for citrate buffer were prepared: solution A (0.1 M citric acid) and solution B (0.1 M sodium citrate). A working solution of 10 mM citrate buffer was prepared (18 ml solution A + 82 ml solution B brought to 1 liter with dH2O), and the pH was adjusted to 6.0. Slides were submerged in 10 mM citrate solution and processed in the microwave oven at full power (Sharp Carousel, 60 Hz, model R-209FW, Sharp Electronics Corporation, Mahwah, NJ) for 12 minutes. After processing the slides in the microwave, the cover was removed from the container, and the buffer solution was cooled to room temperature prior to removing slides. Slides were rinsed in 1× phosphate-buffered saline and then stained following the immunohistochemistry protocol. Control tissue was normal feline lymph node.
Following antigen retrieval, all slides were placed in the DAKO Autostainer Universal Staining System, submerged in Tris-buffered saline, and stained using the following protocol, optimized in preliminary studies. Each section was exposed to 100 µl primary antibody/Tris-buffered solution (dilutions of 1 : 400 for CD3, 1 : 100 for CD79a, and 1 : 1 for BLA.36) for the appropriate duration (30 minutes for BLA.36 and CD3, and 10 minutes for CD79a) and then rinsed with Tris-buffered saline. For negative controls the primary antibody was omitted. Sections then were exposed to 100 µl secondary reagent (Envision for BLA.36 [DAKO] and Link2HRP [DAKO] for both CD3 and CD79a) for 10 minutes and then rinsed as described previously. Finally, all sections were exposed to DAB chromogen for 5 minutes, rinsed as above, and then counterstained with hematoxylin (BLA.36 for 1 minute, and CD3 and CD79a for 5 minutes).
Statistics
A chi-square independence test was used to compare the proportions of B- and T-cell lymphomas located in 3 regions of the GIT tract (stomach, small intestine, and large intestine). A Fisher exact test (2-tailed) was used to perform pairwise comparisons between the stomach and small intestine, stomach and large intestine, and small intestine and large intestine. A Fisher exact test (2-tailed) was also performed to assess the relationship between tumor phenotype (B-cell vs. T-cell) and age of cats. For this assessment cats were divided into 2 groups: cats ages 10 years or younger and cats older than 10 years. In the current study, P < .05 was considered significant.
Phosphotungstic Acid Hematoxylin
To enhance visualization of cytoplasmic granules in large granular lymphocytes, paraffin-embedded tissue was sectioned at 4 microns and stained using the Mallory phosphotungstic acid hematoxylin (PTAH) method. 22
Giemsa
To identify mast cells, sections from the 3 tumors containing eosinophilic granules were stained with Giemsa (Newcomer Giemsa stock staining solution, catalog number 1120, 1 : 4 dilution; Newcomer Supply, Middleton, WI) following a modification of the manufacturer's protocol.
Results
The cats ranged in age from 4 to 16 years, with a mean of 10.8 years. Domestic Shorthair cats predominated at 25 (50%) of 50 cats. The remaining breeds included 7 Domestic Longhair, 7 mixed breed, 5 Siamese, and 1 Russian Blue. For 5 cats the breed was unrecorded. There was a predominance of male cats, at 62% (31/50), 16 cats (32%) were female, and for 3 cats the sex was unrecorded. FIV/FeLV testing was completed on 16 cats. All cats tested were negative. FIV/FeLV testing was performed using an ELISA SNAP test (IDEXX Laboratories Inc., Westbrook, ME) in 11 of 16 cases. In 5 cases the method of testing was unrecorded. The 50 cats in the study lived in the southeastern United States as follows: 28 (56%) from Alabama, 10 (20%) from Georgia, 6 (12%) from Florida, 4 (8%) from Kentucky and 1 (2%) each from Tennessee and Mississippi.
With regard to tumor location, 12 (24%) of 50 tumors were in the stomach. A total of 9 (75%) of these 12 tumors involved the stomach only. The remaining 3 stomach tumors also involved the small intestine. Small intestinal tumors comprised 37 of 50 tumors; 30 of these (80%) were in the small intestine only. Of the remaining 7 tumors in the small intestine, 3 tumors also involved the stomach and 4 tumors involved the large intestine. A total of 8 of 50 tumors involved the large intestine; 4 of these (50%) were in the large intestine only, and 4 involved the large and small intestine.
The NCI WF tumor characterization and designated grade 1 for the 50 cases are summarized in Table 1. The high-grade tumors identified by this system included 19 immunoblastic (Fig. 1) and 4 lymphoblastic (Fig. 2) lymphomas. Intermediate-grade tumors according to the NCI WF included 3 diffuse large, 4 diffuse small cleaved, and 2 diffuse mixed lymphomas (Fig. 3). Diffuse small lymphocytic lymphomas, 18 of which were identified in the 50 tumors studied, were the only type of low-grade tumor recognized using the NCI WF (Fig. 4). These tumors were composed of cells with uniform chromatin density and shallow nuclear indentations in some cells; such epithelial invasion is typical of low-grade T-lymphocytic lymphoma of the intestinal mucosa, and is sometimes best defined through analysis of clonality. 26 It should be noted that 3 tumors classified as immunoblastic by the NCI WF had cells with eosinophilic cytoplasmic granules detected microscopically at 1000× magnification. There is no allowance for this feature in the NCI WF algorithm; however, granulated lymphomas are incorporated into the REAL/WHO classification system.
Histologic classification of 50 cases of feline GIT lymphoma.

REAL/WHO classification system.
NCI WF classification system.
One tumor classified as “diffuse large cell lymphoma” by the NCI WF had cells positive for both BLA.36 and CD3. For this reason it was not assigned a REAL/WHO classification.
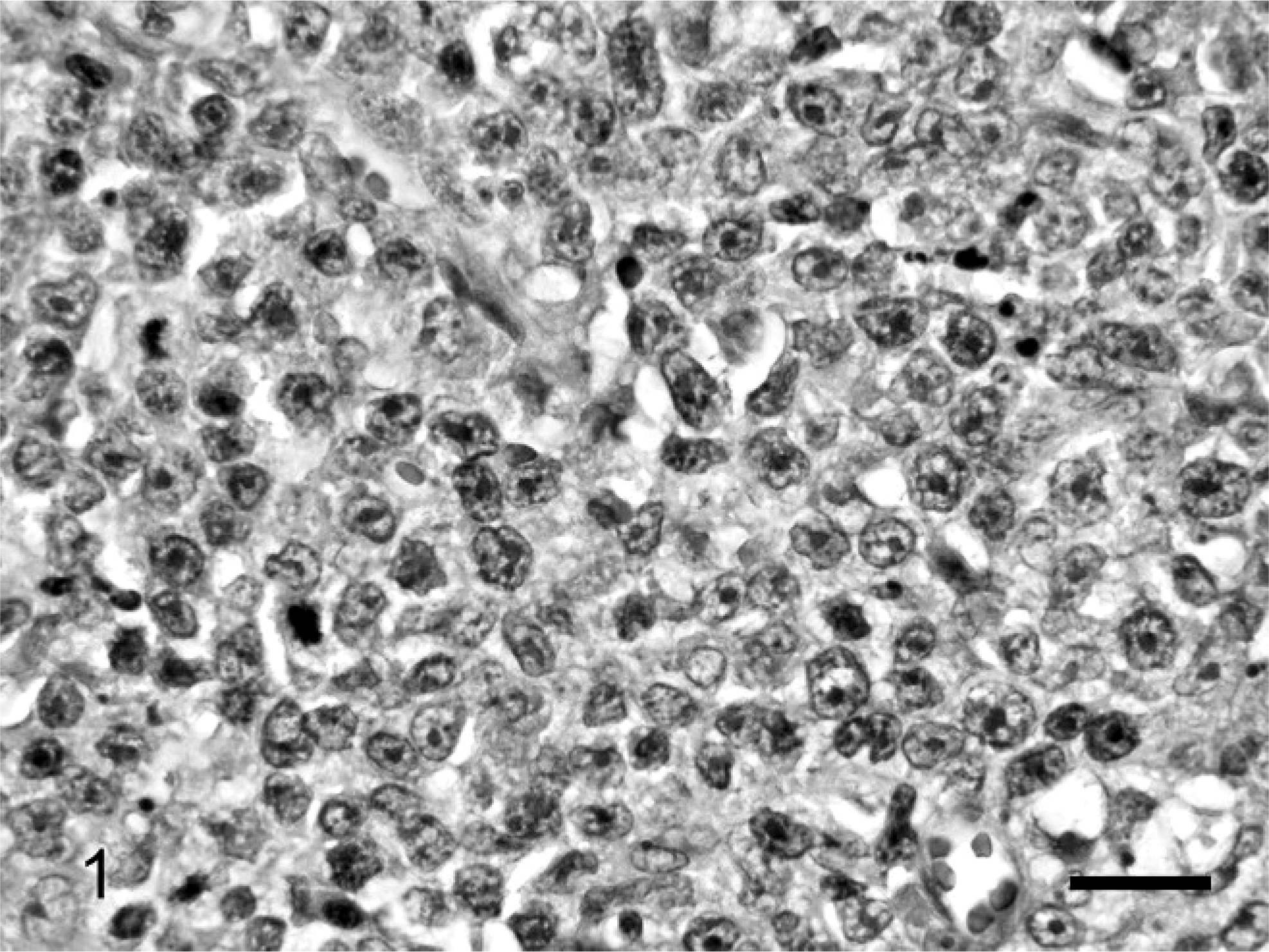
Stomach. The lymphoma is composed of large pleomorphic lymphocytes with large nuclei that are often cleaved, as well as large, prominent, single, central nucleoli. NCI WF classification: large cell immunoblastic lymphoma; REAL/WHO classification: diffuse large B-cell lymphoma of immunoblastic nuclear type. HE. Bar = 50 µm.
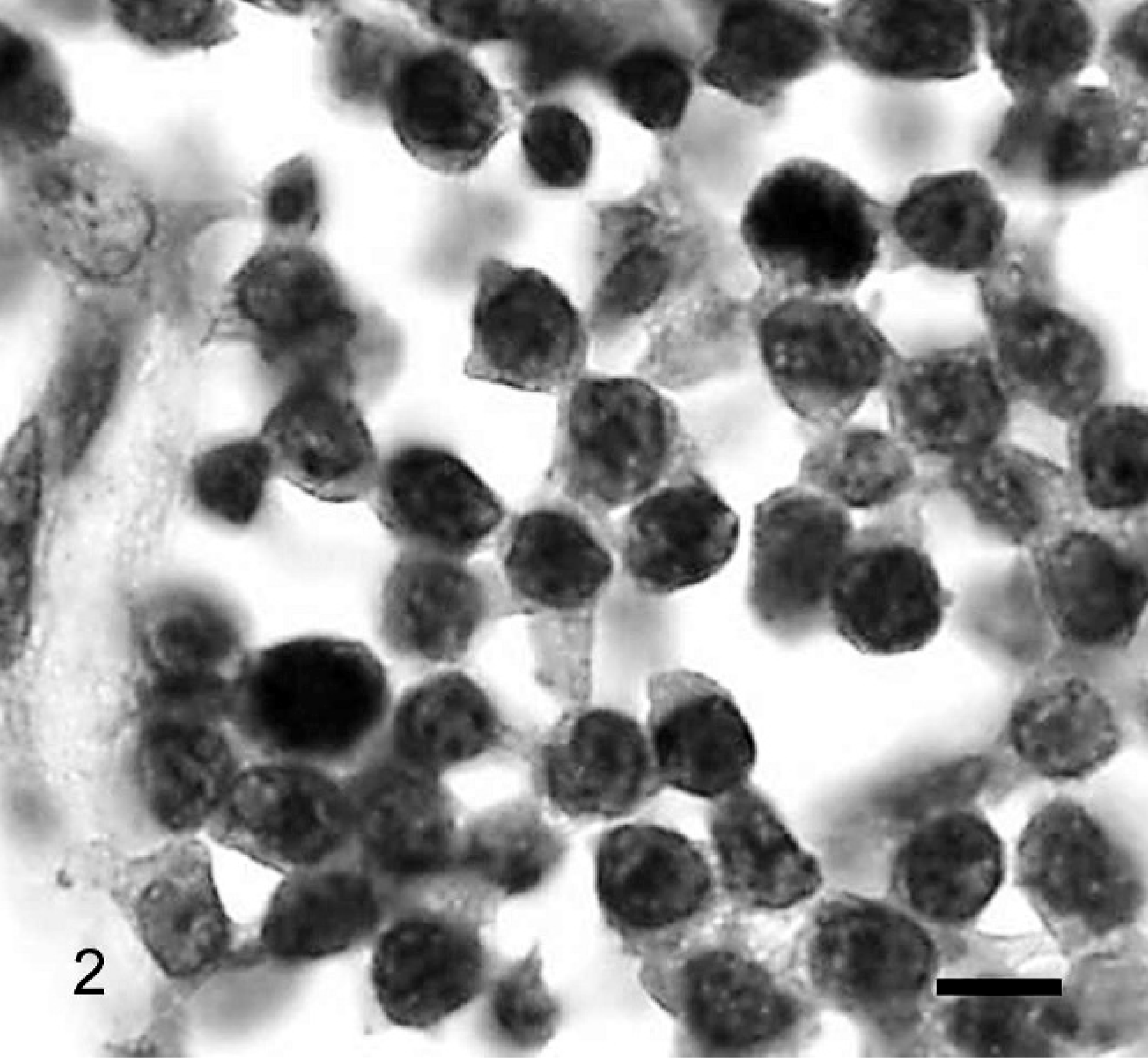
Small intestine. The lymphoma is composed of lymphocytes that have intermediate nuclear size, dispersed chromatin, indistinct nucleoli, and minimal cytoplasm. Note the mitotic figures, one in the upper right and one in the lower left region of the image. NCI WF classification: lymphoblastic lymphoma; REAL/WHO classification: T-cell lymphoblastic lymphoma. HE. Bar = 10 µm.
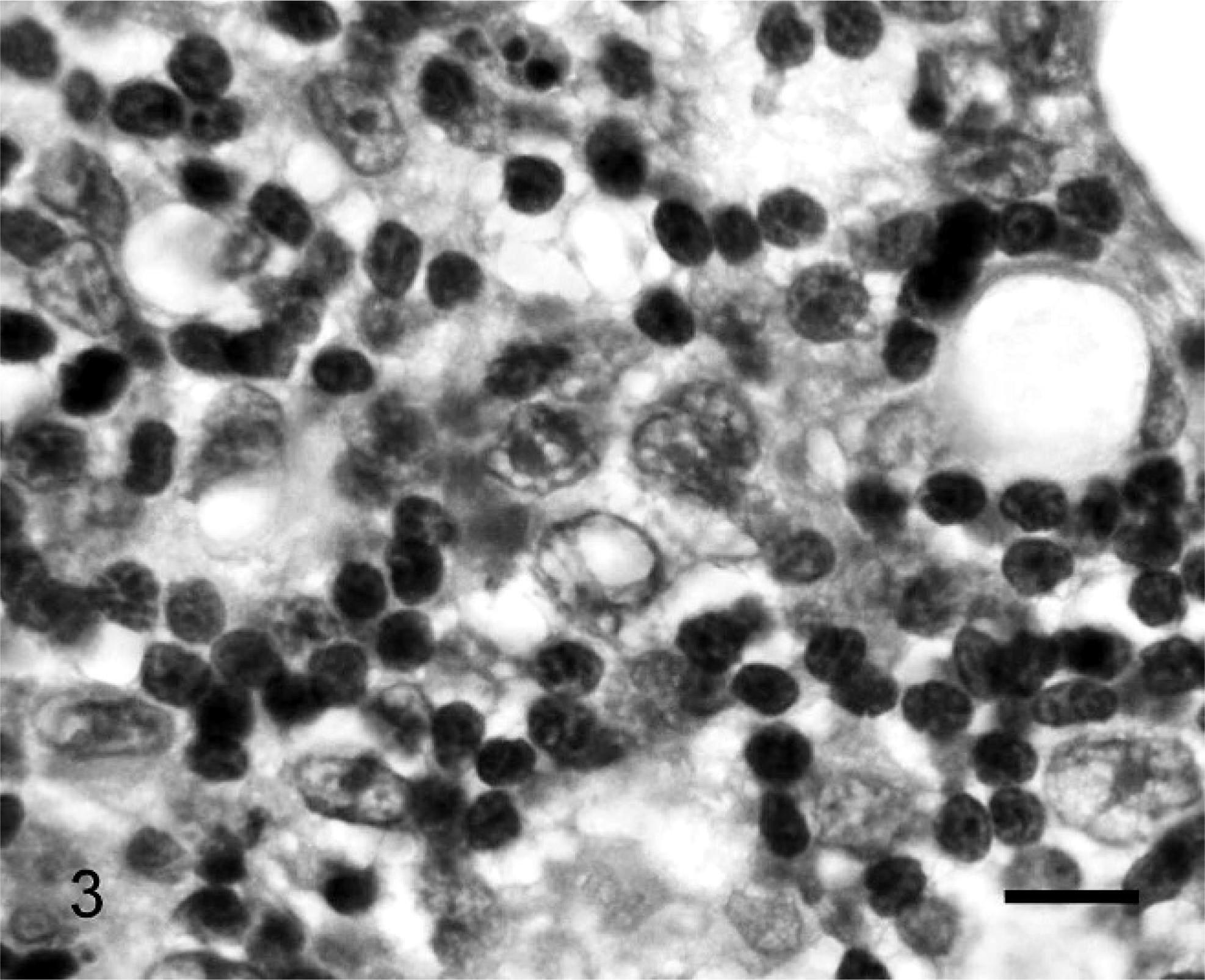
Small intestine. The lymphoma contains 2 distinct cell populations, including large atypical B cells, and small, uniform, reactive T cells with densely stained, compact chromatin, and scant cytoplasm. Note the characteristic fine fibrovascular sclerosis between the cells. NCI WF classification: diffuse mixed lymphoma; REAL/WHO classification: T-cell–rich large B-cell lymphoma. HE. Bar = 10 µm.
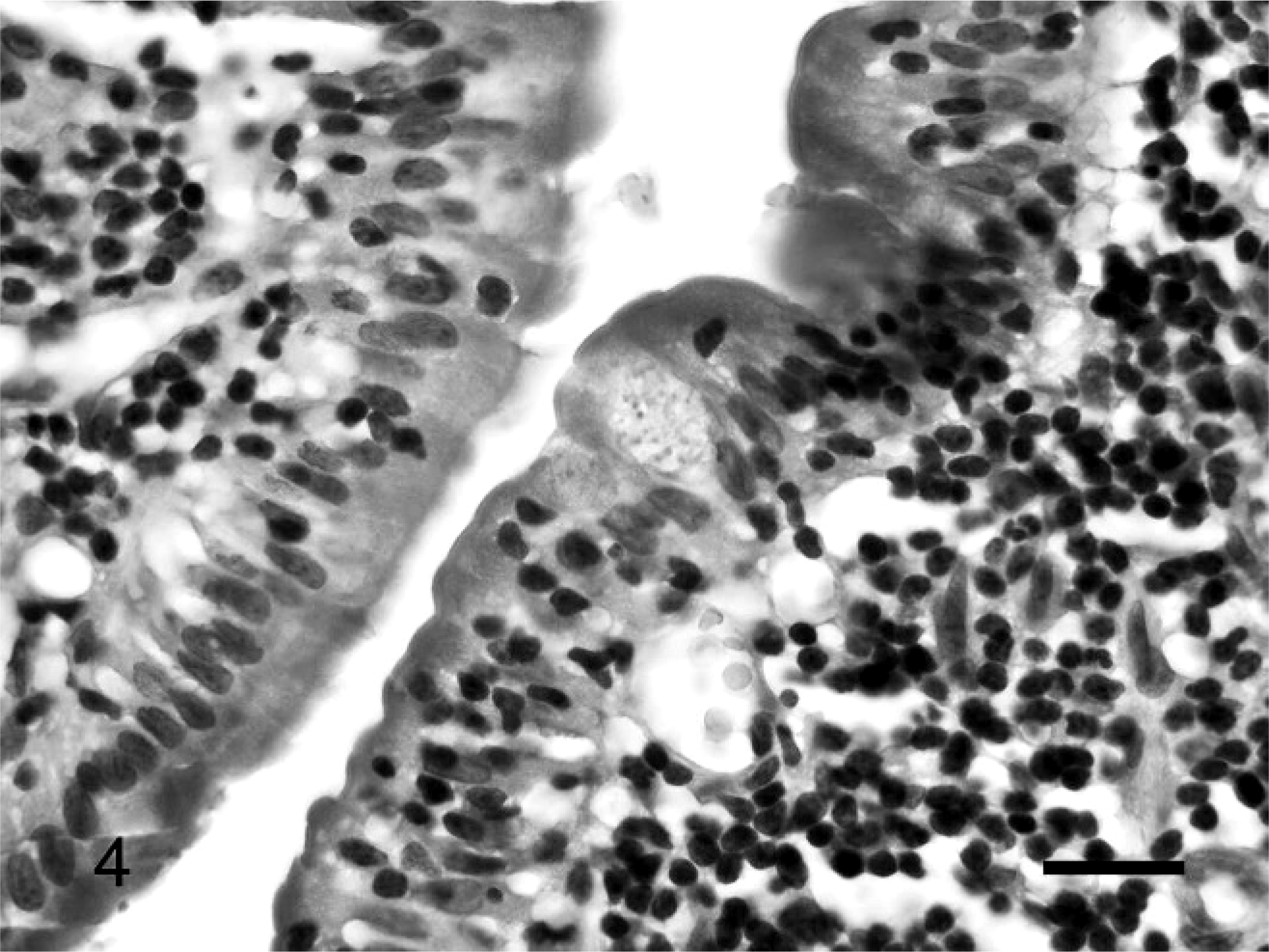
Small intestine. Small lymphocytes with uniform chromatin density and shallow nuclear indentations infiltrate villous epithelium. NCI WF classification: small lymphocytic lymphoma; REAL/WHO classification: epitheliotropic intestinal T-cell lymphoma. HE. Bar = 20 µm.
Overall, B-cell tumors predominated at 54% (27/50), whereas T-cell tumors comprised 38% (19/50). Three tumors (6%) were nonreactive to B- and T-cell reagents used and had eosinophilic cytoplasmic granules. One tumor stained strongly positive for both CD3 and BLA.36, and it was composed of large cells. When considering specific intestinal regions in relation to phenotype, all 12 gastric lymphomas were of B-cell lineage, and all solitary gastric lymphomas were of the large-cell type. T-cell tumors tended to predominate in the small intestine, at 52% (19/37), with 38% (14/37) being B-cell tumors. The 3 tumors that did not stain with B- and T-cell markers used in this study, as well as the 1 tumor positive for both BLA.36 and CD3, were located in the small intestine. In the large intestine the predominance was a B-cell phenotype, at 88% (7/8), with 1 tumor of T-cell lineage in the large intestine. A chi-square independence test was used to compare the proportions of all B- and T-cell lymphomas located in the 3 regions of the GIT. The test revealed that lymphoma phenotype was significantly (P < .01) associated with location. To specifically determine which areas of the GIT showed significant differences, pairwise comparisons of each region using a Fisher exact test (two-tailed) were performed. Results revealed significant differences in lymphoma phenotype between the stomach and small intestine (P < .01) and the small intestine and large intestine (P = .04). There was no statistical significance between the stomach and large intestine (P = .40).
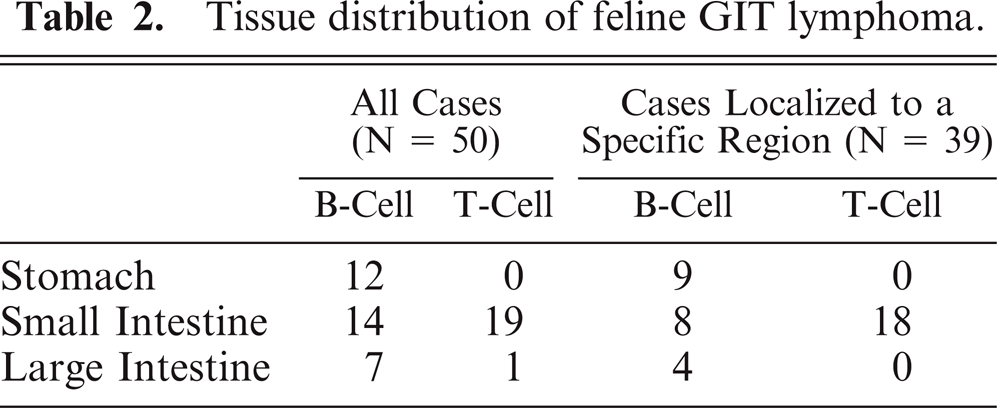
In an attempt to more clearly correlate tumor phenotype with location within specific regions of the GIT, tumors that appeared to be isolated to one region of the GIT (stomach, small intestine, or large intestine) were identified (43/50), and the data were analyzed (Table 2). Of the 9 tumors that were strictly isolated to the stomach, all 9 (100%) were B-cell tumors. Of the 30 tumors that infiltrated only the small intestine, 18 (60%) were T-cell tumors, 8 (27%) were B-cell tumors, 3 (10%) were non-B/non-T, and 1 (3%) was a dual-positive neoplasm. Of the 4 tumors that infiltrated only the large intestine, all 4 (100%) were B-cell tumors. A chi-square independence test was performed to compare the proportions of B- and T-cell lymphomas that were limited to specific regions of the GIT. The test revealed that lymphoma immunophenotype was significantly (P < .01) associated with location within the GIT. Pairwise comparisons of each region (stomach and small intestine, stomach and large intestine, and small intestine and large intestine) using a Fisher exact test (2-tailed) revealed a significant difference in lymphoma phenotype between tumors that localized exclusively to the stomach when compared to tumors that localized exclusively to the small intestine (P < .01). Likewise, a significant difference in immunophenotype was identified between localized tumors in the small intestine and large intestine (P = .02). There was no apparent difference in phenotype between tumors in the stomach and large intestine (P = 1.00).
Tissue distribution of feline GIT lymphoma.
To assess the potential association between tumor phenotype (B-cell vs. T-cell) and age of cats with lymphoma, cats were divided into 2 groups (≤10 years old and >10 years old), and a Fisher exact test (2-tailed) was performed. There was no significant association between tumor phenotype and age (data not shown).
The REAL/WHO classifications and diagnoses for all 50 cases are summarized in Table 1. The breakdown of the 27 B-cell tumors is as follows: 60% (16/27) were diffuse large B-cell of immunoblastic nuclear type (Fig. 1), 7% (2/27) were diffuse large B-cell of centroblastic nuclear type, 11% (3/27) were B-cell small lymphocytic, 15% (4/27) were B-cell lymphocytic intermediate type, and 7% (2/27) were T-cell rich large B-cell lymphoma (Fig. 3). Of the 19 T-cell tumors, 79% (15/19) were epitheliotropic small T-cell lymphoma (Fig. 4), and 21% (4/19) were T-cell lymphoblastic (Fig. 2).
Six percent (3/50) of the tumors were LGL type and were not labeled with B- or T-cell immunohistochemical markers. When viewed at 1000× magnification, the HE sections of these tumors were found to have cells with eosinophilic cytoplasmic granules. One of these tumors was positive for PTAH staining of granules. To rule out mast cell origin, the 3 tumors composed of cells with cytoplasmic granules were stained with Giemsa. In all 3 cases, although there were many neoplastic cells with granules, none of these cells had metachromatic granules. One of these tumors contained rare Giemsa-positive, morphologically normal, resident or infiltrating mast cells.
Discussion
The most significant finding in this study was the strong association between immunophenotype and GIT tumor location. The association was greatest in the stomach, where there was a predominance of diffuse large B-cell lymphoma of immunoblastic nuclear type. Likewise, B-cell lymphoma predominated in the large intestine. These findings are similar to those recently reported by Moore. 26 All large cell lymphomas in the GIT were of B-cell phenotype. T-cell lymphoma tended to predominate in the small intestine, and this finding is supported by a recent report that identified the origin of these tumors as the diffuse mucosa-associated lymphoid tissue of the small intestine. 26
Since its original development for the characterization of human non-Hodgkin's lymphoma, the NCI WF 1 has been used successfully to characterize lymphomas in cattle, 43– 45 dogs, 5 and cats 40 and to predict the clinical course of disease (indolent, intermediate-grade, and high-grade or rapidly progressive 1 ) with reasonable accuracy in these species. 3 Although the NCI WF is relatively simple to apply to histologic sections through a dichotomous algorithm, 5 the most accurate predictive models, particularly for tumors of small (mature) cell types, requires an integration of both mitotic rate and cell type. 24 For the purposes of this study, tumor grade was based on the published histologic criteria for the NCI WF; 1 accordingly, mitotic rate was used to distinguish between the high- and low-grade tumors of small cell types, as described. 5 For tumors of large (immature) cell types, mitotic index was not incorporated into the grading scheme, because most tumors of large cell types can be classified as high grade. 24 High-grade lymphoma must be distinguished from indolent tumors—notably marginal zone lymphoma—with intermediate-sized nuclei and low mitotic rates. 42 In the case of immunoblastic lymphoma of the current study, mitotic rate seldom exceeded 1 in most 1000× fields, but the nuclear diameter clearly exceeded the width of 2 red blood cells, indicating a large cell type. With the exception of the neoplasms that were composed of large cells with eosinophilic cytoplasmic granules, characterization using the NCI WF correlated well with the tumor classifications as determined using the REAL/WHO system. However, the very important difference between the 2 systems is that besides being developed prior to the implementation of immunohistochemical techniques and prior to the recognition of some types of lymphoid malignancies, the NCI WF is a classification of cell types, whereas the REAL/WHO system is a classification of diseases. 39 The latter is advantageous because it allows tailored management for a specific disease entity rather than a disease within a “treatment group.” 39
In the current study, the percent of diffuse large B-cell lymphomas of immunoblastic nuclear type is significantly higher than that of centroblastic nuclear type. These findings differ from a recent report indicating that the majority of diffuse large B-cell lymphomas in the GIT tract of cats were of centroblastic nuclear type. 26 One possible explanation for this difference is the criteria used for classification of immunoblastic versus centroblastic lymphoma. In the current study, diffuse large B-cell lymphoma with immunoblastic nuclear type was defined as being composed of greater than 90% immunoblasts, which were characterized by a prominent, single, central nucleolus and abundant cytoplasm. 20, 36 Diffuse large B-cell lymphoma of centroblastic nuclear type was assigned to tumors that were composed predominantly of centroblasts, which were characterized by one to several peripheral nucleoli and a narrow rim of cytoplasm; these tumors may contain variable numbers of immunoblasts. 20, 36 Using such criteria, a lymphoma classified as centroblastic could contain a cell population with features of immunoblasts. Precise criteria are therefore needed for histologic grading schemes, and differences between studies should be considered within this context. In humans, the centroblastic type accounts for approximately 80% of the cases of diffuse large B-cell lymphoma. This figure, however, includes all cases of diffuse large B-cell lymphoma, and not just those limited to the gastrointestinal tract. 36 Geographic location of the patients is another variable that should be considered when comparing different studies of lymphoma, and this factor warrants further study.
Prior to the routine application of immunohistochemical data, T-cell–rich large B-cell lymphoma was identified via the NCI WF as a “diffuse mixed” tumor together with another type of tumor composed of large and small B lymphocytes. 2, 39– 41 The latter tumor was not identified in this study. In the REAL/WHO classification system, T-cell–rich large B-cell lymphoma is a histologic variant of the diffuse, large B-cell lymphoma category. 2, 39, 41 In cats, T-cell–rich large B-cell lymphoma is a tumor that is most commonly reported as a mass in the ventral cervical or submandibular region, 7, 32 and some tumors isolated to the cervical/submandibular region have been classified as feline Hodgkin's-like lymphoma. 46 T-cell–rich large B-cell lymphoma is characterized microscopically by the presence of 2 distinct cell populations. There are scattered large atypical B cells, which may represent as few as 5% of the tumor population, 41 and numerous uniform, small- or medium-sized, reactive CD3+ T-cells that have densely stained, compact chromatin and scant cytoplasm. Most mitoses, often in atypical forms, are seen in the large neoplastic B-cells. Typically, the small reactive CD3+ T-cells comprise the majority of the cell population. 7, 32, 41 Admixed with the numerous CD3+ cells are fewer small lymphocytes that are positive for BLA.36 and CD79a. 7, 32, 46 The large neoplastic B-cells have large vesicular nuclei with prominent nucleoli, 41 and on the HE sections in the current study the individual neoplastic cells closely resembled those seen in the immunoblastic tumors. A 1999 study by Day et al. 7 reported that similar large, bizarre cells were consistently BLA.36 positive and were negative for CD79a. A 2001 report by Walton and Hendrick 46 found that the large neoplastic cells stained positive for both BLA.36 and CD79a. In the current study, the neoplastic cells were consistently positive for BLA.36 and variably positive for CD79a. Thus, the phenotypic staining patterns of the neoplastic B-cells may vary from case to case. In both cases in the current study, neoplastic cells were unevenly distributed throughout the mass, but overall the percentages of neoplastic cells in these 2 tumors were approximately 10 to 20% and 40 to 50%. Although a 40 to 50% neoplastic cell population in a T-cell–rich large B-cell lymphoma is somewhat high, percentages as high as 50% 33 and 60% 2 have been reported. Furthermore, it has been reported that over time, as T-cell–rich large B-cell lymphomas progress, the proportion of large neoplastic B-cells may increase to the point of predominance. 2, 39 Verification of the B-cell origin of these tumors by polymerase chain reaction as described by Werner et al. 50 was not attempted in the current study.
Another reported characteristic feature of T-cell–rich large B-cell lymphoma is the presence of a fine but dense fibrovascular sclerosis throughout the tumor, 39, 41 which is thought to be due to T-cell proliferation. 39 The presence of this fibrovascular network is the reason that these neoplasms do not fragment on sectioning, unlike high-grade lymphomas that tend to fracture, especially if the tumor section is large. 39 Consistent with the literature, 39, 41 a heavy fibrovascular network was identified in both T-cell–rich large-B-cell lymphomas identified in the current study (Fig. 3).
Three tumors were classified as immunoblastic lymphoma according to the NCI WF, yet under 1000× magnification the neoplastic cells were found to have eosinophilic cytoplasmic granules. Neoplastic cells in all 3 tumors lacked metachromatic granules with Giemsa staining (essentially excluding mast cell tumors) and did not express the B- or T-cell markers. Therefore, it is possible these tumors represented natural killer (NK) cell lymphomas. However, confirmation of an NK origin was not feasible because specific markers for feline NK cells are not available. 29 PTAH staining, which has been used in some studies for enhanced visualization of the granules in LGLs, 13– 15, 23 was performed on tissues of all 50 cases. One of the tumors that did not stain with B- and T-cell markers had PTAH-positive cytoplasmic granules, which is supportive of LGL lineage. 13– 15, 23 However, in another study of 11 cats, LGL granules did not stain with PTAH. 49 Therefore, in this study the lack of PTAH staining in 2 tumors containing cells with eosinophilic cytoplasmic granules does not exclude an LGL lineage. It is possible that tissue fixation time and/or age of paraffin blocks may have affected PTAH staining quality. However, it must also be considered that these tumors are of myeloid rather than lymphoid origin.
Tumors of LGL lineage in the GIT tract of cats are typically of T-cell phenotype. 29 However, in the current study the 3 LGL lymphomas failed to express any of the phenotypic markers. Because of the small to intermediate size of most cells in T-cell lymphoma, it is possible that some of the small- to intermediate-sized T-cell tumors might have contained indiscernible cytoplasmic granules, even when examined under 1000× magnification. In agreement with another study, 29 eosinophilic cytoplasmic granules were visible in histologic HE-stained preparations where the neoplastic cells had abundant cytoplasm, and all 3 LGL tumors in the current study were located in the small intestine.
The selection of 2 B-cell markers and 1 T-cell marker for phenotyping in this study was predicated on a 1999 study 11 in which CD79a was not expressed by 7% of the tumors that, based on other criteria, were concluded to be of B-cell type. These investigators also used 2 T-cell markers (CD5 and CD3) but reported that CD3 was sufficient to detect all of the T-cell tumors in their study. 11 B-cell markers were CD79a and CD79b in that study. 11 The latter antibody was not commercially available for the current study; therefore, the combination of CD79a and BLA.36 was chosen for the detection of B cells. Only 16 (59%) of the 27 B-cell tumors were positive for both BLA.36 and CD79a. Seven (26%) of the putative B-cell tumors were positive for BLA.36 but had extremely low expression of CD79a (so low that it was difficult to confidently conclude that these tumors were positive from the CD79a staining alone), and 4 were positive for CD79a but negative for BLA.36. Therefore, CD79a alone would have failed to identify 26% of the tumors, which by other criteria were determined to have been of B-cell origin. If BLA.36 had been used as the only marker, 15% of the putative B-cell tumors would have been undetected. It is possible that the different methods of antigen retrieval used for CD79a and BLA.36, the inability to control conditions of fixation, and the varying ages of paraffin-embedded tissues could have affected antigen integrity, and hence immunohistochemical reactivity. Furthermore, staining intensity of CD79a for tumors with low expression might have been enhanced with a prolonged incubation time or increased antibody concentration.
One tumor in the small intestine was composed of large lymphocytes that were strongly positive for both CD3 and BLA.36. The BLA.36 antigen is primarily known as a B-cell marker, but it has been reported to rarely label T-cell tumors. 6 Although some neoplastic lymphocytes express both B- and T-cell markers when evaluated by flow cytometry, 51 it is most likely that the tumor that stained positive for both BLA.36 and CD3, but not CD79a, is of T-cell lineage. Another possibility, albeit much less likely, is that this tumor represented a mixed cell tumor. Two-color immunofluorescence microscopy or flow cytometry could have distinguished between these 2 possibilities, but this was not feasible because of the use of paraffin-embedded tissue and the limited availability of differentially conjugated antibodies. The authors also considered the possibility of an inflammatory lesion; however, the uniform cell morphology and highly infiltrative pattern of growth were indicative of neoplasia.
It has been suggested that tumor phenotype in cats with GIT lymphoma is associated with age. 39 The reported hypothesis is that cats younger than 10 years will likely have B-cell lymphoma, whereas cats older than 10 years will likely have T-cell lymphoma. The findings in the current study do not support an association between age and phenotype.
There are several limitations to the current study. Additional B-cell markers, such as CD20, were not employed to provide further confirmation of B-cell lineage. CD20 is a nonglycosylated phosphoprotein expressed on the surface of pre–B cells and persists until plasma cell differentiation. 34, 35 However, CD20 also has been reported to be expressed at very low levels in some T-cell subsets (predominantly γδ T-cells) 16 and T-cell malignancies. 48 Future studies should incorporate a combination of antibodies, including anti-CD20, into the battery of immunohistological markers to enhance detection of B-cell neoplasms. Because of the retrospective design of this study and the broad distribution of treatment centers, no correlation could be drawn between the findings and survival time, response to therapy, and anatomic lesion distribution as confirmed by necropsy. Furthermore, the exclusive use of archival paraffin blocks limited the antibody selection to only those that could withstand formalin fixation under a variety of conditions.
In conclusion, this study identified a highly significant association between the location, classification, and immunophenotype of lymphoma within the feline GIT tract, where there was a predominance of diffuse large B-cell lymphoma of immunoblastic nuclear type within the stomach. The etiologic and pathogenetic significance of these findings is uncertain. Future studies should incorporate additional histochemical markers and address the role of infectious agents (for example, Helicobacter spp.) and resident lymphoid populations with respect to this unique distribution of feline lymphoma.
Footnotes
Acknowledgements
The project was funded in part by the Grace Kemper Research Fund. We wish to thank Beth Landreth, Uschi Wohlfert, Lori Carden, Abigail Carreño, and Lisa Parsons for their assistance with histologic techniques, and the faculty in the Auburn University Surgical Pathology Service for providing initial histologic evaluation of case material.
Findings presented at the 2006 Annual Meeting of the American College of Veterinary
Pathologists and the American Society for Veterinary Clinical Pathology in Tucson, AZ
(Abstract published in Vet Clin Path
