Abstract
Canine lymphoma is the neoplasm most often treated by chemotherapy, yet there are few data to correlate response to therapy with its different subtypes. This study is based on biopsy specimens from 992 dogs for which lymphoma was the clinical diagnosis. All cases were phenotyped by immunohistochemistry for CD3 and CD79alpha. Cases with histiocytic proliferation were evaluated immunohistochemically for CD18. Clonality was verified in 12 cases by polymerase chain reaction (PCR). Survival (event time) data and complete survival information (cause of death or time to last follow-up) were available on 456 dogs. Additional covariate information when available included size, age, sex, phenotype, stage and grade of lymphoma, mitotic index, and treatment protocol. Because of the many subtypes of B- and T-cell lymphoma, the cases were grouped into 7 diagnostic categories: (1) benign hyperplasia; (2) low-grade B-cell; (3) high-grade B- and T-cell; (4) low-grade T-cell; (5) centroblastic large B-cell of all mitotic grades (subdivided by clinical stage); (6) immunoblastic large B-cell of all mitotic grades, and (7) high-grade peripheral T-cell. Grouping was determined by histological grade (based on mitotic rate/400× field, with low-grade 0–5, intermediate 6–10, and high-grade >10) and stage for survival function estimation. No association with survival was found for size (based on breed of dog) or sex. All diagnostic categories of indolent or low-grade type had low mitotic rates, whereas those with clinically high grades had high mitotic rates. The diagnostic category with the most cases was centroblastic large B-cell lymphoma. Compared with dogs in this largest represented group of lymphomas, dogs with high-grade lymphomas had significantly higher mortality rates, and dogs with low-grade T-cell lymphomas had significantly lower mortality rates. Treatments for high-, intermediate-, and low-grade lymphomas were divided into 4 groups: absence of treatment, chemotherapy with or without hydroxydaunorubicin, and only prednisone. Dogs with low-grade T-cell (T-zone) lymphomas had the longest median survival (622 days), whereas the shortest median survival was in dogs with T-cell high-grade (peripheral T-cell) subtype (162 days). The dogs with centroblastic large B-cell lymphomas had a median survival of 127 days with low stage, 221 days with intermediate stage, and 215 days with advanced stage. Dogs with T-zone lymphoma were probably diagnosed in later stages of disease because of the lack of signs associated with progression. As with human lymphomas, a histological diagnosis with immunophenotyping is a minimal requirement for diagnosis of a specific subtype.
Malignant lymphomas are the most common canine tumor treated with chemotherapy and affect dogs of all ages and breeds. Diagnosis of lymphomas in dogs has followed systems of classifications developed for humans and has recently been accurately characterized by an adaptation of the latest World Health Organization (WHO) Classification for Neoplastic Diseases of the Lymphoid Tissues. 12 This classification was developed from recommendations of an international group of pathologists based on unique aspects of each neoplastic disease to provide an updated system of classification that did not reflect a single geographic origin. 11 This classification provided a system of categorizing lymphoid neoplasms according to their level of cellular maturation that also provided a level of prognostic indication. This system was tested by a group of physician pathologists who reviewed a large group of cases, admittedly with joint discussion on cases with disparities in interpretation, and achieved a high degree of consensus. 16
Subsequently, a group of 19 well-qualified veterinary general pathologists classified 300 cases of canine lymphomas, definitively characterized by 3 veterinary hematopathology specialists using the WHO criteria. 24 This test group of 19 veterinary pathologists achieved a high rate of accuracy (83%) of classification in the 300 individual cases without any follow-up group discussion. That study indicated that the WHO classification can be accurately applied to canine lymphomas by pathologists who are experienced but are not necessarily specialists in hematopathology, It remained to be demonstrated, however, that a specific diagnosis of lymphoma subtype had a similar impact on survival in dogs as it did in humans and whether a specific diagnosis of lymphoma in a dog impelled the specific chemotherapy required. There are few published studies that address the issue of individualized treatment and survival of dogs with specific types of lymphoma. 2,17,23 This report expands on the earlier study of 300 dogs examining WHO subtype classification by determining survival of dogs undergoing treatment for specific subtypes of lymphoma.
Materials and Methods
Study Population
Study subjects were recruited through a member solicitation in the newsletter of the Veterinary Cancer Society, in which most members are veterinary primary practitioners and oncologists. Accession of cases began in 2006 until 992 cases of canine tissues were examined from dogs with lymphadenopathy and suspected diagnosis of lymphoma (Table 1). An exhaustive effort was made to obtain follow-up information on all cases. Requests to the referring practice were sent for specific data on the stage of disease at diagnosis, the treatment protocol, and the date and cause of death or the last known date alive. Most practices could provide the stage of disease at diagnosis, if not already indicated at biopsy, and the treatment protocol. If required, and with the permission of the referring veterinarians, owners were contacted; they were typically forthcoming about providing further information on the patient’s medical management and, if appropriate, date and cause of death. Almost all the nonsurviving dogs had been euthanatized due to progression of lymphoma.
Descriptive Information About Suspected Lymphoid Tumors in 992 Dogs
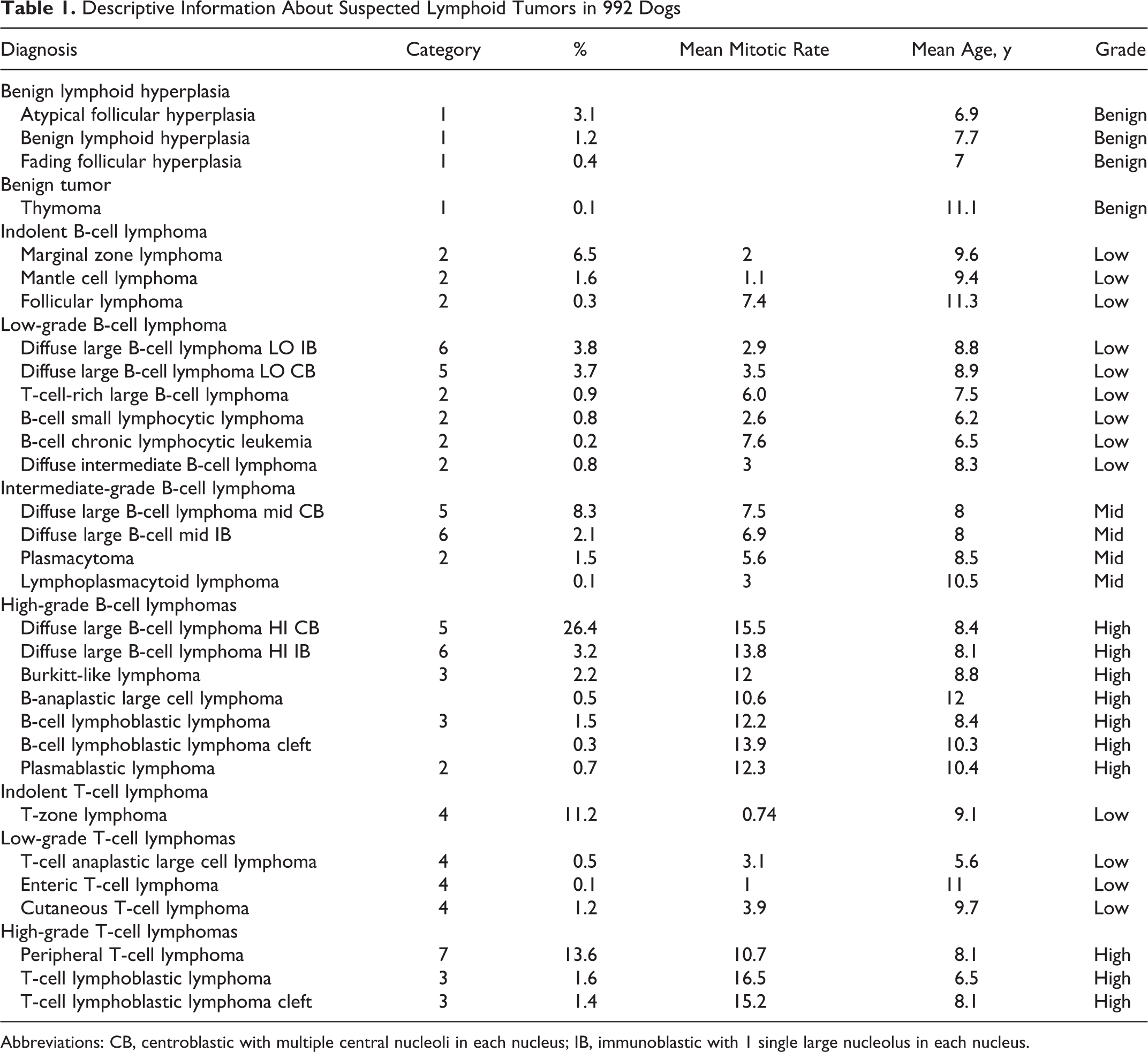
Abbreviations: CB, centroblastic with multiple central nucleoli in each nucleus; IB, immunoblastic with 1 single large nucleolus in each nucleus.
Collection of cases extended to 2008, with almost all cases coming from 27 US states and a few from Europe and Canada. Case information, including clinical case number, pathology case number, submitting practice and clinician, diagnosis, mitotic rate per 400× field, owner’s and dog’s names, sex (male castrate, male intact, female spayed, female intact), age in years, stage of disease, and treatment protocol, was recorded. Approximately one-third of the cases were broad Tru-Cut needle biopsy specimens; the remainder were incisional or excisional biopsy specimens. All cases routinely had immunohistochemistry (IHC) performed with CD3, CD79a, and additional IHC, including CD18, when needed to evaluate histiocytic proliferation. 23
Except for the first 300 cases examined by the review group, all remaining cases were diagnosed by a single pathologist (V.E.V.). Cases considered nondiagnostic (benign vs neoplastic or hyperplasia vs neoplasia) on initial evaluation (n = 12) were examined by PCR methods. 23 Histological examination began with assessment of tissue architecture; the general cell type was next determined. Because the mitotic rate was a critical part of many diagnoses, it was determined before the cells were described. The mitotic rate was counted in areas having the highest number of mitotic figures. In some lymphomas with high mitotic and cellular death rates, the presence of tingible body macrophages indicated areas of high mitotic rate. Counts of mitotic figures were continued until the same mitotic rate per 400× field was found in at least 3 areas.
Most types of lymphoma were recorded as a single diagnosis regardless of mitotic rate. For diffuse large B-cell lymphomas, the grade according to mitotic rate was included; for lymphoblastic lymphomas, the nuclear shape was included. Because the Revised European American Lymphomas Classification (REAL) group was concerned that large B-cell lymphomas of immunoblastic type with a large single central nucleolus were more aggressive than those with multiple central and peripheral nucleoli of the centroblastic type, these lymphomas were separately tallied, each with 3 levels of mitotic rates. 11 These additional data were collected to assist in interpreting differences in survival times. The lymphoblastic lymphomas of B- and T-cell types were tallied separately for nuclei of round or convoluted types to determine whether this morphologic detail was associated with a difference response to therapy and survival.
Lymphomas of mantle cell and marginal zone types appeared to arise as primary neoplasms in the spleen. Because these lymphomas were identified in peripheral areas by their relationship to a germinal center, cytological features, and low mitotic type, all tumors of these types were identified by specific diagnosis and not by location or topography.
Lymphomas Classification, Staging, and Grading
B- and T-cell lymphomas were grouped according to histological grade based on mitotic rate. This reduction in categories was considered necessary because the cases included 19 types of B-cell lymphomas and 16 types of T-cell lymphomas with too few cases in most categories for statistical analysis. By this classification, B- and T-cell lymphomas of lymphoblastic type were analyzed together because both by definition have a high mitotic rate and are similar clinically in response to therapy with difficulty in obtaining a remission. In contrast, the lymphomas with histological grades of low mitotic rate of B- and T-cell types were not combined to allow comparison of phenotype impact on survival of indolent lymphomas.
The cases of benign hyperplasia and of lymphomas were therefore placed by diagnosis into the following 7 categories:
Benign (BLH): fading follicular hyperplasia, atypical follicular hyperplasia, and benign lymphoid hyperplasia
Low-grade B-cell (LGBC): mantle cell lymphomas of node or spleen, marginal zone lymphomas (MZL) of node or spleen, follicular lymphomas, centrocytic lymphomas, lymphoplasmacytoid, and plasmacytic lymphomas 23
High-grade (LBL): T- and B-cell lymphoblastic lymphomas, including types with both round and convoluted nuclei and Burkitt-like lymphomas
Low-grade T-cell (TZL): T-zone lymphomas and T-cell anaplastic lymphomas
Centroblastic B-cell (DLBCL CB): large B-cell lymphomas of low, intermediate, and high mitotic rates
Immunoblastic B-cell (DLBCL IB): large B-cell lymphomas of low, intermediate, and high mitotic rates
T-cell high-grade (PTCL): peripheral T-cell lymphomas
For stage of disease (as distinguished from grade), the more detailed WHO system of Ia or Ib to Va or Vb was compressed (as is customary in the human staging system) to a 1–3 system. 15 WHO stages I and II were considered local disease in 1 node or region, with III and IV representing cases of generalized lymphadenopathy usually including some involvement of spleen and liver. Stage V was considered advanced disease (stage 3 in the compressed system) that included systemic involvement.
Combinations of lymphoma diagnostic categories were then divided into 3 major groups, determined by mitotic rate, of low, intermediate, and high grade. These 3 groups were further subdivided into 4 distinct subgroups based on treatment protocol:
High-grade lymphomas (LBL and PTCL) Protocols containing hydroxydaunorubicin (Doxorubicin), including L-asparaginase, vincristine, hydroxydaunorubicin, and prednisone L-asparaginase, vincristine, hydroxydaunorubicin, and prednisone (Wisconsin) Cyclophosphamide, hydroxydaunorubicin, vincristine, and prednisone (CHOP) Note that first 2 treatment protocols above (CTVX, Wisconsin) each contain doxorubicin and are based on the CHOP protocol. Variations in the order of dosage and timing occurred with the administration of the same drugs. Other protocols not containing hydroxydaunorubicin, including Mechlorethamine, vincristine, procarbazine, and prednisone (MOPP) Cyclophosphamide, vincristine, prednisone (COP) Prednisone and 1 other treatment, either chlorambucil or lomustine (CCNU) Only prednisone No treatment
Intermediate-grade lymphomas (DLBCL CB, DLBCL IB) Any hydroxydaunorubicin-containing protocol Any non–hydroxydaunorubicin-containing protocol Only prednisone No treatment
Low-grade lymphomas (LGBC and TZL) Any hydroxydaunorubicin-containing protocol Any non–hydroxydaunorubicin-containing protocol Only prednisone No treatment
Statistical Analysis
All data were recorded in a Microsoft Excel spreadsheet and later transferred to Stata/IC 12.1 (StataCorp LP, College Station, TX) for statistical analysis. Kaplan-Meier survival plots were generated to predict the course of disease progression (survival probability) in individual patients; log-rank tests were used to compare the distribution of survival across groups. Proportional hazards regression models were created to assess the relative rates of death among patient and treatment groups while controlling for confounding factors. 1 All regression models controlled for age as a confounder. Results are presented as hazard ratios (HRs) and 95% confidence intervals (95% CIs). Locally weighted scatterplot smoothing regression was used to graphically portray the relationship between mitotic index and survival and determine appropriate cut points for dichotomous analyses. P values less than .05 were considered statistically significant.
Results
The Study Population
Of the 992 dogs clinically suspected of having lymphoma, 944 had lymphomas, whereas 48 had benign (predominantly follicular) hyperplasia (category 1). Complete survival data on 456 dogs (48.3%) with lymphomas included either cause of death or time to last follow-up. Ninety-seven dogs were still alive or were lost to follow-up (ie, censored) at the end of data acquisition from veterinarians and owners; 95% of the censoring times fell between 10 and 937 days. Losses to follow-up were often due to relocation of the owner or primary veterinarian.
Of the 944 dogs with lymphoma biopsy specimens, 490 were male and 434 were female; sex was not recorded in 20 cases. There were 222 dogs (23.5%) of small breeds and 714 dogs (75.6%) of large breeds; 8 had no breed indicated. Of the 222 small breed dogs, 118 were male and 100 were female; of the 714 large breed dogs, 367 were male and 332 were female; sex or breed was not recorded in 27 cases. In the 456 dogs with complete survival data, the top 4 small breeds in descending order were terrier breeds, Cocker Spaniel, Beagle, and Shih Tzu. Of the large breeds, the 4 most common in descending order were Golden Retriever, Labrador Retriever, crossbred dog, and Boxer.
The median crude survival for all dogs with complete survival data was 221 days; for dogs with lymphomas, 218 days (n = 451); and for dogs with benign hyperplasia, 351 days (n = 5) (Fig. 1). There was a significant association between age and the rate of death in dogs with lymphomas when both linear (P = .005) and quadratic (P = .002) terms were used. All subsequent regression models included both age covariates as confounders.
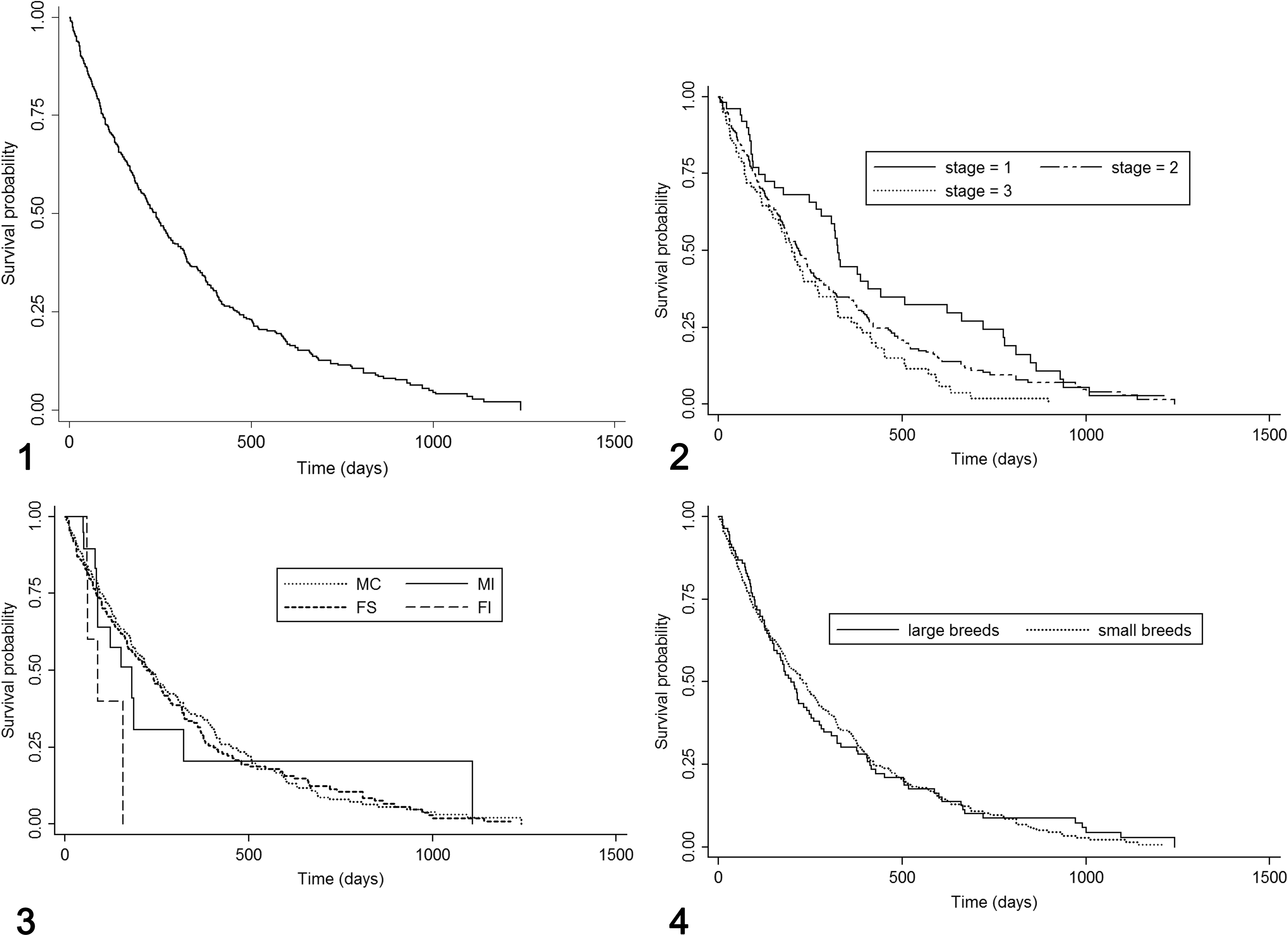
Overall survival in 456 dogs with canine lymphomas. Median survival = 221 days.
Impact of Stage and Signalment on Survival
Most case histories did not include a WHO “a or b” designation in staging to indicate systemic illness, but clinical signs were provided that assisted with that interpretation. Almost all dogs with clinically high-grade lymphomas presented with signs of systemic illness. This included lymphoblastic lymphomas and peripheral T-cell lymphomas and occasionally Burkitt-like lymphomas. In contrast, dogs presenting with a clinically indolent type of lymphoma, such as mantle and marginal zone, and particularly T-zone lymphoma, were free of clinical signs of disease at presentation. Dogs with lymphoblastic lymphomas were uniformly mildly depressed on examination; many were reported to be panting. Other signs of systemic involvement included loss of appetite and activity and the presence of systemic illness assessed on clinical examination.
In 394 dogs, the lymphomas were staged: 51 dogs in stage 1, 276 dogs in stage 2, and 77 dogs in stage 3. The impact of stage of disease of dogs treated for lymphomas is given in Fig. 2. Median survival in all cases was 324 days in stage 1, 218 days in stage 2, and 199 days in stage 3. The stage at which 75% of the dogs were predicted to die was also associated with survival: 720 days for stage 1, 424 days for stage 2, and 390 days for stage 3. The age-adjusted hazard ratio comparing stage 2 to stage 1 was 1.42 (95% CI, 1.00–2.01; P = .049), and the hazard ratio comparing stage 3 to stage 1 was 1.79 (95% CI, 1.19–2.70; P = .005). Median survival by stage was also calculated separately for category 5 dogs with diffuse large B-cell lymphomas: The median survival of dogs with stage 1 disease was 177 days (11 dogs), for stage 2 disease 221 days (141 dogs), and for stage 3 disease 215 days (34 dogs). However, analysis also showed that the stage at which 75% of the category 5 dogs were predicted to die was predictably associated with survival: 507 days for stage 1, 420 days for stage 2, and 360 days for stage 3. These survival curves were not significantly different after adjustment for age.
The association between sex and survival of 445 dogs with lymphomas is given in Fig. 3. Few dogs were sexually unaltered. There was no significant difference in survival between surgically altered males and females (P = .56), between intact males and females (P = .22), or between all males and females (P = .51) after age adjustment.
Figure 4 depicts the survival of large and small breed dogs with lymphomas. The median survival time of small dogs with lymphomas (197 days) did not significantly differ after age adjustment from that of the dogs of larger breeds (230 days) (P = .53), despite the longer expected lifespan of small breed dogs.
Survival by Lymphoma Diagnosis
Survival data for the 7 categories of canine benign hyperplasia and lymphomas are depicted in Fig. 5. The median survival in category 1 (BLH) was 351 days. There were 5 cases with complete data for this category. The submissions for this group totaled 47 cases, including 27 cases of benign atypical follicular hyperplasia, 11 cases of follicular hyperplasia, 5 cases of fading follicular hyperplasia, and 4 cases of atypical diffuse hyperplasia.
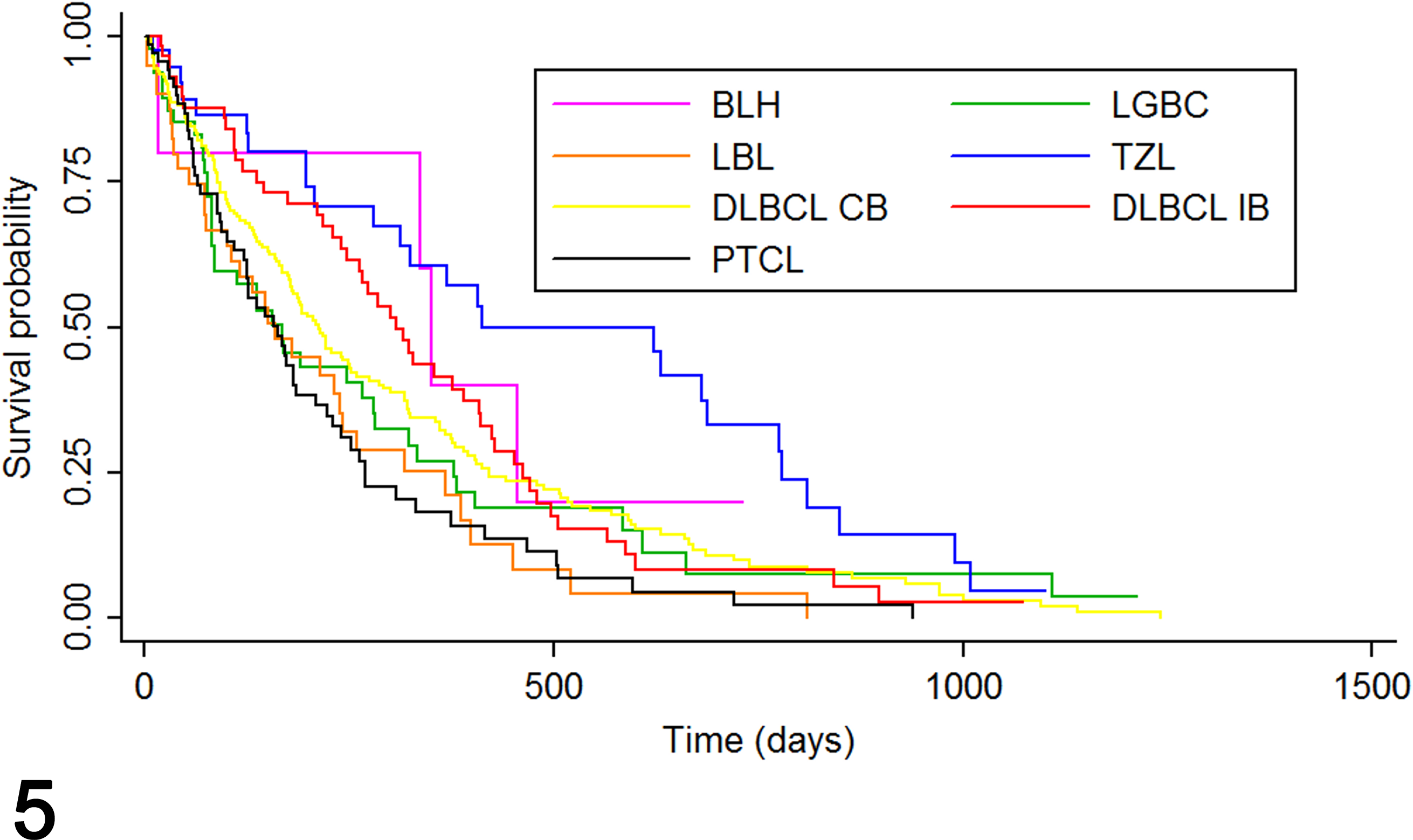
Survival probability in 7 distinct groups of 456 dogs with benign lymphoid hyperplasia or lymphoma. BLH, benign follicular hyperplasia (group 1); LGBC, marginal zone lymphomas (group 2); LBL, high-grade lymphomas (group 3); TZL, T-zone lymphomas (group 4); DLBCL CB, centroblastic large B-cell lymphomas (group 5); DLBCL IB, immunoblastic large B-cell lymphomas (group 6); PTCL, peripheral T-cell lymphomas (group 7).
The median survival in category 2 (LGBC) was 168 days. There were 47 cases with complete data. The total submissions for this group included 17 cases of mantle cell lymphomas (of which all but 1 were in spleen), 64 cases of marginal zone lymphomas (of which 10 were in spleen), 3 cases of follicular lymphomas, 7 cases of centrocytic lymphomas, and 24 cases of plasmacytoid type lymphomas. Centrocytic lymphoma was the diagnosis for diffuse lymphomas of low mitotic rate with cleaved or irregularly shaped nuclei and was interpreted as a tumor of the small nondividing cells of the germinal center. Marginal zone and mantle cell lymphomas are usually primary in the spleen with slow spread to other lymphoid organs. For this reason, the splenic and nodal types of these 2 lymphomas were not separated into splenic and nodal topographies as those in lymph nodes were assumed to have originated in the spleen.
The median survival for category 3 (LBL) high-grade lymphomas of intermediate cell size was 160 days. There were 40 cases in category 3 (LBL) with complete data. The total submissions for this group consisted of 18 cases of B-lymph-oblastic lymphomas, 32 cases of T-lymphoblastic lymphomas, and 21 cases of Burkitt-like lymphomas.
The median survival of category 4 (TZL) of low-grade lymphomas of T-cell type was 412 days. There were 39 cases in category 4 (TZL) with complete data. The total submissions for group 4 consisted of 32 cases of T-zone lymphomas and 5 cases of T-anaplastic large cell lymphomas.
The median survival for category 5 (DLBCL CB), which consisted of the high-grade diffuse large B-cell lymphomas of centroblastic nuclear type, was 213 days. There were 198 cases in category 5 (DLBCL CB) with complete data. The total submissions of this type of lymphoma included 252 cases of high grade, 80 cases of intermediate grade, and 35 cases of low grade. Those cases with complete data totaled 180 including all grades.
The median survival for category 6 (DLBCL IB), which consisted of diffuse large B-cell lymphomas of immunoblastic nuclear type (IB), was 308 days. There were 57 cases in category 6 with complete data. The total submissions of this type of lymphomas included 32 cases of high-grade lymphomas, 20 cases of intermediate-grade lymphomas, and 36 cases of low-grade lymphomas. Those cases with complete data totaled 56 including all grades.
The median survival for category 7 (PTCL) was 162 days. This category consisted of 70 cases of peripheral-cell lymphomas with complete data. The total submissions of this type of lymphomas totaled 130 cases.
Category-specific survival was compared with survival in category 5 (DLBCL CB), which was the most populous with 187 cases with complete data. The 2 categories characterized by high-grade lymphomas had significantly higher mortality rates: category 3 (LBL) (HR, 1.59; 95% CI, 1.07–2.36; P = .022) and category 7 (PTCL) (HR, 1.56; 95% CI, 1.14–2.13; P = .006). Category 4 (TZL) (low-grade lymphomas) had a significantly lower mortality rate (HR, 0.57; 95% CI, 0.37–0.87; P = .010). There was no significant difference between category 1 (BLH) and category 5 (HR, 0.77; 95% CI, 0.28–2.09; P = .61).
Impact of Mitotic Index on Survival of Treated Dogs
When all individuals were analyzed together using a LowESS scatterplot smoother, there was little evidence that mitotic index (MI) was associated with survival (Fig. 6). Based on this figure, mitotic index was used to divide patients into 2 groups: those with MI ≤ 20 per 400× field (n = 353) and those with MI ≥ 21 per 400× field (n = 26). Figure 7 demonstrates that survival was significantly less in the group with MI ≥ 21 per 400× field (P = .031).
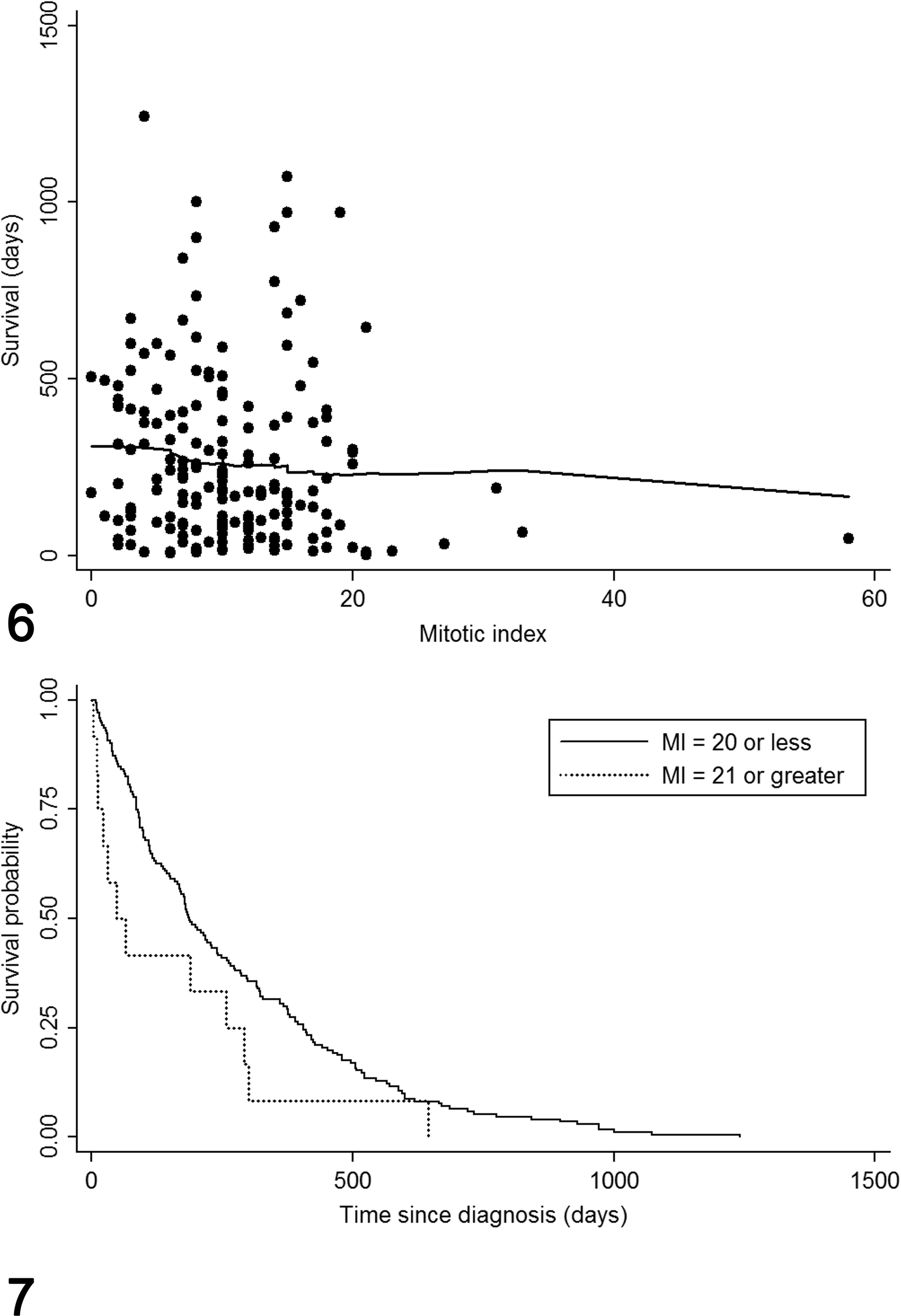
Mitotic index (MI) in 183 dogs with lymphomas. Regression line was obtained using a locally weighted scatter smoothing function.
Impact of Treatment Protocols
The dogs were treated not based on grade but based on choice of treatment protocol by the oncology practice. Some practices had their own ongoing research studies and gave all dogs with lymphoma the CVTX protocol. Because other studies have suggested that doxorubicin provides better survival, the many types of treatment protocols were condensed to those with and without that drug. There were too few cases in categories 1 (BFH) and 2 (LGBC) with sufficient survival data to make inferences concerning treatment. Table 2 contains associations between treatment and survival in categories 3, 4, and 5.
Association Between Treatment and Survival in 4 Groups of Dogs with Lymphoma.
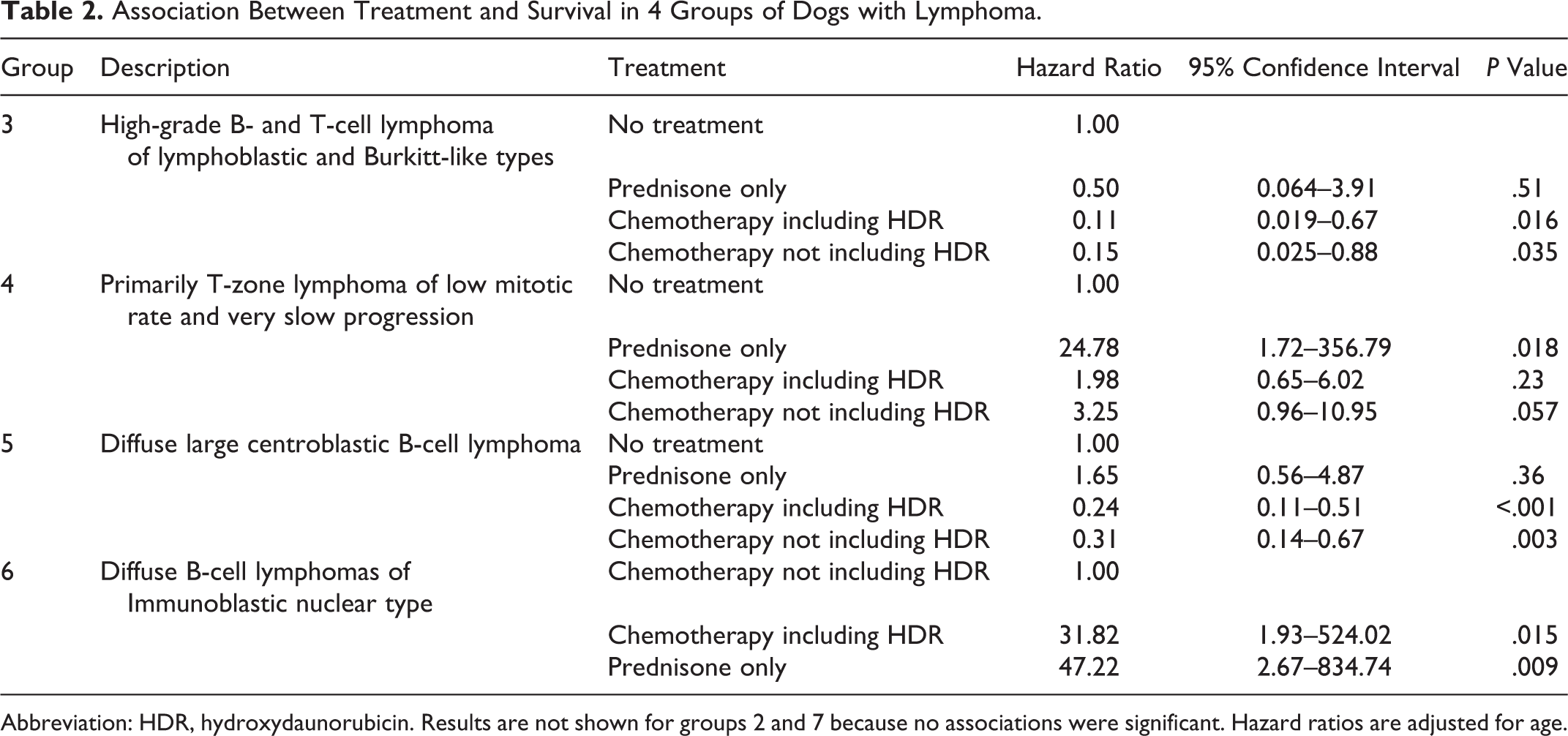
Abbreviation: HDR, hydroxydaunorubicin. Results are not shown for groups 2 and 7 because no associations were significant. Hazard ratios are adjusted for age.
Category 3 (LBL: high-grade B- and T-cell lymphomas of lymphoblastic and Burkitt-like types)
With 36 valid observations, there was little evidence that treatment with only prednisone was superior to no treatment at all (HR, 0.50; 95% CI, 0.064–3.90; P = .51). In contrast, chemotherapy with (HR, 0.11; 95% CI, 0.019–0.67; P = .016) or without (HR, 0.15; 95% CI, 0.025–0.88; P = .035) hydroxydaunorubicin was associated with a similar and significant decline in the rate of death compared with no treatment.
Category 4 (TZL: low-grade T-cell lymphomas)
Analysis of survival time for 36 dogs in category 4 of known ages and of low mitotic rates showed evidence that chemotherapy was associated with lower survival for this type of lymphoma. Compared with no treatment, the rate of death was higher in dogs that received chemotherapy with no hydroxydaunorubicin (HR, 3.25; 95% CI, 0.96–10.95; P = .057), chemotherapy with hydroxydaunorubicin (HR, 1.98; 95% CI, 0.65–6.02; P = .23), and prednisone only (HR, 24.78; 95% CI, 1.72–356.79; P = .018). There was no significant difference in survival between dogs receiving chemotherapy with or without hydroxydaunorubicin (P = .47), although survival was significantly worse in dogs receiving only prednisone compared with these 2 other treatment options (P = .011 and P = .022, respectively). In 13 dogs with TZL that were given no treatment, the median survival was 687 days. Complete survival data were present for 24 dogs with TZL that were given chemotherapy; median survival was 312 days without and 407 days with hydroxydaunorubicin. Of dogs receiving chemotherapy at diagnosis, 1 dog was in stage II, 3 were in stage IIa, 1 was in stage III, 6 were in stage IIIa, 2 were in stage IIIb, 1 was in stage IVa, 1 was in stage IVb, 2 were in stage Va, and 1 was in stage Vb; 6 had no staging recorded. The longest surviving dog in this group was diagnosed in stage IIIa, was given the CHOP protocol, and survived 990 days. The dog with the shortest survival in the treated group was in stage IVa and was given chemotherapy containing hydroxydaunorubicin; it survived for 10 days. The cause of death for these treated dogs was progression of disease for 15, ruptured urinary bladder for 1, splenic sarcoma for 1, and other causes for 5. In consideration of the slow rate of progression of TZL, it could be assumed that dogs with advanced stages of disease at diagnosis probably had lymphoma for 2 or more years. Because dogs with TZL characteristically do not present with loss of appetite or activity, it is likely that this aspect of their disease is part of the reason they tend to be diagnosed at an advanced stage of the lymphomas.
Category 5 (DLBCL CB: diffuse large centroblastic B-cell lymphomas)
The analysis of treatment for 186 dogs in category 5 indicated that chemotherapy, both with (HR, 0.24; 95% CI, 0.11–0.51; P < .001) and without (HR, 0.31; 95% CI, 0.14–0.67; P = .003) hydroxydaunorubicin, significantly extended survival. The rate of death in dogs treated only with prednisone was not significantly different from the rate in dogs without any treatment (HR, 1.65; 95% CI, 0.56–4.87; P = .36).
Category 6 (DLBCL IB: diffuse B-cell lymphomas of immunoblastic nuclear type)
In the analysis of treatment protocols on 57 dogs, there was no significant difference between dogs receiving chemotherapy that did or did not contain hydroxydaunorubicin (P = .95). Receiving only prednisone was associated with significantly poorer survival than receiving chemotherapy with (HR, 31.82; 95% CI, 1.93–524.02; P = .015) or without (HR, 47.22; 95% CI, 2.67–834.74; P = .009) hydroxydaunorubicin.
Category 7 (PTCL: peripheral T-cell lymphomas)
In the analysis of treatment protocols on 69 dogs, none of the treatment options (including no treatment) was significantly associated with survival (P = .43).
Associations between treatments and survival were also evaluated in dogs classified into the 3 categories based on histological grade (low, intermediate, and high; Table 3). In the low-grade category (n = 86 dogs, 81 of which had complete survival information), compared with dogs receiving no treatment, the dogs receiving chemotherapy with hydroxydaunorubicin, dogs receiving chemotherapy without hydroxydaunor-ubicin, and dogs receiving only prednisone had poorer survival. In the intermediate-grade category (n = 255 dogs, 242 of which had complete survival information), administering prednisone alone was not significantly different from no treatment, but treatment regardless of whether it contained hydroxydaunorubicin was significantly associated with improved survival. Finally, in the high-grade category (n = 110 dogs, 103 of which had complete survival information), compared with no treatment, administering only prednisone, chemotherapy with hydroxydaunorubicin, or chemotherapy without hydroxydaunorubicin did not appear to meaningfully affect patient prognosis.
Association Between Tumor Grade, Treatment, and Survival in Dogs With Lymphoma.

Abbreviation: HDR, hydroxydaunorubicin.
Discussion
This report is unique in that it combines the recognition of subtypes of lymphomas in dogs with survival data and response to varying types of treatment protocols. Each case in this study had been phenotyped, which has been shown to be essential for the diagnosis of all human lymphomas (except follicular types that can be recognized as B-cell type on the basis of tumor architecture). 3,11 Because follicular lymphomas are rare in animals, virtually all cases require determination of B- or T-cell type for a firm diagnosis of lymphoma subtype. Lymphoma in companion animals is no longer considered a generic disease. There is marked variation in the biology of untreated canine lymphomas that requires the management of each type of lymphoma to be carefully weighed. 8,22 The long survival of the low-grade lymphomas, most of which require conservative or no therapy, also makes a specific diagnosis imperative. One of the more surprising findings of this study is the relatively short survival of dogs with benign lymphoid hyperplasia, many of which had follicular hyperplasia that must be differentiated from true follicular lymphoma. 23 Thus, for a dog to have a florid follicular hyperplasia, there must be a strong immune stimulation that indicates a systemic response in most patients.
It is possible to diagnose lymphomas by cytological examination, but it is not yet possible to give a specific diagnosis of lymphoma subtype by interpretation of a fine needle aspirate. 4,6 In contrast, it appears that a cytological preparation (if made like a blood smear) of a uniform population of smaller cells that have a “hand mirror” type of cytoplasmic extension is almost certainly derived from a node with T-zone lymphomas. An imprint of a T-zone lymphoma does not have the hand mirror cytoplasmic features. A further study that combines examination of the cytological and histological features of this subtype of lymphomas is needed to verify this apparent association. Although flow cytometry can be rapid and informative and may provide a very high likelihood of lymphoma and even phenotype, it is unable to provide a diagnosis of specific lymphoma subtype. 9,18,25 The practice of lymphoma diagnosis in human medicine is solidly based on histology, and this practice needs to be followed in veterinary oncology as well. Currently, most lymphoma therapy in animals is largely palliative, suggesting that collaboration is needed with oncologists to test specific protocols on single subtypes of lymphoma with a goal of therapy that is closer to curative.
This study, by separating the treatment protocols for high-grade lymphomas into those with or without hydroxydaunorubicin (Doxorubicin), has given further definition to lymphoma therapy. With due consideration for toxicity, this study has shown that the inclusion of hydroxydaunorubicin to other intensive protocols showed high probability for prolonging survival of dogs with intermediate-grade lymphomas. The survival of dogs with low-grade T-zone lymphomas deserves further study because many of the dogs with that diagnosis were given treatments of various kinds. However, there were sufficient numbers of untreated cases to demonstrate that chemotherapy was associated with reduced survival for dogs with that diagnosis. Alternatively, it may be that the dogs treated for TZL presented in very late stage with prominent lymphadenopathy, whereas those that were untreated were apparently asymptomatic with minor lymphadenopathy. Dogs with T-zone lymphomas may have a dual presentation because some lived for years with no therapy and others reached a stage at which therapy was indicated. It may be that the cases with the larger cells termed “intermediate” type are those that tend to progress, whereas those of small cell type may be a cohort that does well with no therapy. There is very little difference in the mitotic rates of these types of T-zone lymphomas, with usually none in a 400× field of the small cell type and 0–1 in the tumors with cells of intermediate size.
The management of diffuse large B-cell lymphomas (DLBCL) of immunoblastic nucleolar type (IB) may not be sufficiently different from management of the more common type with centroblastic nucleolar configuration (CB) to warrant treating these as separate diagnoses. In the older classification of the NIH Working Formulation, the DLBCL of IB type was classed as high grade and the DLBCL of CB type was considered to be of intermediate grade; that has since been questioned in human studies. There appears to be considerable heterogeneity within the morphologic types of human large B-cell lymphomas that relates to significant differences in survival. Some of the subtypes associated with shorter survival are distinguished morphologically, as in this study, whereas others are identified by specific immunohistochemical markers. 3,10,14,19
There is marked variation in the survival of dogs with DLBCL of varying mitotic rates. The median survival in dogs with fewer than 20 mitoses per 400× field was found to be 188 days; in contrast, the median survival of dogs with 21 or more mitoses per 400× field was 31 days. Because most chemotherapeutic drugs act on cells at some stage of mitosis, it is important to recognize the level of cell proliferation and how those lymphomas can most effectively receive targeted treatment. The assessment of mitotic rate is essential for obtaining a specific diagnosis, and the most efficient manner of determining this parameter is from a histological section.
A remaining problem in diagnosis of canine lymphomas is that with disease progression, marginal zone lymphomas have cells with a single large nucleolus that may appear as a diffuse proliferation with a low mitotic rate and can be confused with low-grade lymphomas of DLBCL-IB type, which also have a single large nucleolus. Usually, there is sufficient architectural evidence of nodular late-stage MZL that this entity can be identified on that basis if sufficient tissue is available for examination. In addition, MZL has a nucleus of intermediate cell size (1.5 red cell diameters) with abundant cytoplasm, but that of the IB type of DLBCL is truly of large cell type (2–2.5 red cell diameters), with this difference in nuclear size sufficient to suggest one diagnosis over the other.
A further area of diagnostic differentiation identified by this study is whether it is advantageous to differentiate between lymphoblastic lymphomas (LBL) of convoluted (LBC) or smooth round (LBL) nuclear types in terms of treatment or survival. There were only 30 cases of T-cell LBL total and 18 cases of B-cell LBL in this study, too few to determine meaningful survival differences.
Like the exfoliation of cells of T-zone lymphomas in cytological preparations, the appearance of the cells of lymphoblastic lymphomas is also distinctive to the experienced observer. In both cytological and histological examination, the dispersed chromatin of LBL of B- or T-cell type is characteristic and appears more densely stained with obscured nucleoli. A report on the diagnostic criteria of LBL by both cytological and histological examination is needed together with a collaborative trial of intensive therapy, which now appears to increase survival times in humans. 21 At present, these appear to be the most difficult subtype of canine lymphoma to bring into remission and to attain long-term survival with results similar to human cases of lymphoblastic leukemia/lymphomas. 7,13
Most of the cases for this study were derived from areas within the continental United States, with a few from Canada and Europe. There did not appear to be any difference in the distribution of various types of lymphomas by geographic location. However, there does seem to be a higher level of Burkitt-like lymphomas (BKL) from one high-level oncology practice in Arizona. Whether by experience or location, the affected dogs in that area also appear to have longer survival, with some living for 2 to 3 years following diagnosis. In humans, there is similar concern that some cases may be difficult to distinguish from DLBCL with a high mitotic rate. 5,20 This is another area that deserves further detailed study.
Finally, the difficulty of conducting a study of this type needs to be recognized. In the accession of cases, a copy of the submitting document was retained as well as the slides and the pathology report. This meant that address as well as the phone and fax number of nearly all cases was retained. Initially, all practices were sent a mail request for follow-up of their case submissions. This request yielded results on about 250 cases. Following this, a fax request was sent 6 months later for information that pertained to animals still alive at the initial request or that was simply not received at that time. Subsequently, 4 or 5 requests were sent to some practices and many phone calls were made with specific requests for information on stage of disease at diagnosis, treatment protocol, and outcome, with date and cause of death. With all of these efforts, about one-quarter of the cases were lost to follow-up; together with those still living, these totaled about half of the targeted database of 1000 cases. In addition, retrospective analyses of treatment efficacy must be interpreted with great caution given the likely problem of confounding by treatment indication: The choice of treatment could have been influenced by the clinician’s perception of patient prognosis. This study attempted to address this source of bias by controlling for age and evaluating stage of disease, but the potential for residual confounding of the hazard ratios and survival functions by unknown factors cannot be ruled out.
In conclusion, in animals, as in humans, a histological diagnosis of lymphomas remains essential. 16 Lymphomas in animals and humans require a specific diagnosis for selecting the most appropriate chemotherapy. The diagnosis of lymphomas can be difficult and may require multiple modalities, including clonality testing. Next-generation collaborative studies by pathologists and oncologists are required for each subtype of lymphoma to determine the optimal treatment for each entity.
Footnotes
Acknowledgements
Support of this study by the American Kennel Club Canine Health Foundation is gratefully acknowledged. The following oncologists and practices are gratefully recognized for providing case materials and staff to phone owners and review files for data relating to treatment protocols and survival of the dogs. Without their continued participation this study could not have been completed: Veterinary Cancer Group, Tustin, CA: Dr M. Rosenberg and associates and L. Shapiro Integrative Veterinary Oncology, Phoenix, AZ: Dr B. Hershey Center for Specialized Veterinary Care, Westbury, NY: Dr G. Post Animal Medical Center, New York, NY: Dr A. Hohenhaus Southeast Veterinary Oncology, Orange Park, FL: Dr T. LaDue Red Bank Veterinary Hospital, Tinton Falls, NJ: Dr C. Clifford College of Veterinary Medicine, University of Illinois: Dr T. Fan
These veterinarians and hundreds more that are not named have our most sincere thanks. We also thank J. Jopling for assistance with tabulation and extraction of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
