Abstract
Transmission of sheep scrapie and some other prion diseases, including variant Creutzfeldt-Jakob disease of man, probably occurs via the oral route. A disease-associated variant of the host-coded prion protein (PrPd) accumulates in germinal center follicles of lymphoid tissues, including Peyer's patches of the gut, where it can be detected before its accumulation in the central nervous system. To investigate the potential role of lymphoid tissue nerves in neuroinvasion, we used immunohistochemical methods to study the frequency and distribution of nerves and PrPd accumulation in Peyer's patches and other lymphoid tissues from scrapie-affected and unaffected sheep. Nerves were infrequently found in secondary follicles of Peyer's patches, but never in germinal centers of the other lymphoid tissues tested. No differences in the frequency or distribution of nerves were found in relation to the presence or absence of PrPd accumulation. PrPd accumulation and nerves were only infrequently present together in Peyer's patches. These results suggest that, even if amplification of infectivity in lymphoid tissues facilitates neuroinvasion, nerves within lymph nodes and germinal centers of Peyer's patches do not play a primary role in transport of infectivity to the central nervous system. However, sheep between 2 and 4 months of age had significantly more nerve fibers within follicles than older groups. It is therefore possible that a general increase in nerve density of the intestine during early phases of life may contribute to an increased susceptibility of young animals to oral prion infection.
Introduction
Scrapie, a disease naturally affecting sheep and goats, belongs to a family of progressive neurodegenerative disorders, consisting of the infectious, familial, and sporadic forms of transmissible spongiform encephalopathy (TSE) or prion disease of animals and man. The TSEs also include bovine spongiform encephalopathy (BSE) of cattle and exotic ungulates, chronic wasting disease of cervids (CWD), and Creutzfeldt-Jakob disease (CJD) of humans. TSEs can be naturally, experimentally, or iatrogenically transmitted to several mammalian species. They are characterized by the accumulation of abnormal disease-specific isoforms of a host-encoded cell-surface glycoprotein called prion protein (PrPd) within tissues of the lymphoreticular system (LRS), peripheral and central nervous systems, and some other organs. TSEs can be experimentally induced by several routes, although natural sheep scrapie, BSE, CWD, and variant CJD are generally thought to be acquired orally. It is often proposed that neuroinvasion takes place after amplification of infectivity in LRS tissues followed by an ascending infection along nerves of the autonomic nervous system (ANS). This assumption is based on several lines of evidence: first, that infection can be demonstrated in secondary follicles of LRS tissues prior to its demonstration in the central nervous system (CNS) 1, 4, 22, 23 ; second, that nerves have been found within germinal centers of Peyer's patch (PP) follicles 7 in direct contact with both follicular dendritic cells (FDCs) and tingible body macrophages (TBMs), 8 both of which are known to amplify or accumulate PrPd and infectivity; third, that infectivity can be identified within the ANS including the enteric nervous system (ENS) and celiac-mesenteric ganglia (CMG) 1, 9, 24 ; and finally, that the initial sites of PrPd accumulation in hamsters, 1 sheep, 24 and cattle 9 orally infected with TSEs are the dorsal motor nucleus of the vagus nerve (DMNV) and the intermediolateral tracts of the spinal cord, sites which connect with the ENS and CMG via the vagal and splanchnic nerves, respectively. 1, 9
Although the hypothesis that infection may be amplified in lymphoid tissues and then undergo retrograde transportation to the CNS via peripheral nerves is supported by good evidence, there are some studies that shed doubt on the role of PP or other lymphoid tissue germinal centers in neuroinvasion. When homogenates containing scrapie infectivity and PrPd are inoculated directly into the intestinal lumen, PrPd can be seen to cross the villous epithelium. From here, PrPd may be transported directly into submucosal lymphatics and bypass the PP. 14 In addition, sheep of some particular PrP genotypes show inconsistent evidence of LRS involvement when compared with other genotypes 6, 4, 23 ; and naturally occurring BSE of cattle, though putatively infected via the oral route, lacks LRS PrPd accumulations.
For lymphoid tissues to have a central role in neuroinvasion, there must be a consistent relationship between the presence of infectivity in lymphoid tissues and nerves of the ANS, but there is almost no data on the degree to which LRS germinal centers are innervated. While previous descriptions suggest that PPs are innervated by the submucosal enteric plexus, 18 it has been generally accepted that germinal centers of lymph nodes do not have nerve terminals. 3 Moreover, recent studies suggest that neuroinvasion can also occur through the hematogenous route. 20
For several years our laboratory has had the opportunity to perform necropsies of large numbers of sheep exposed to natural and experimental scrapie. From across these studies we have identified sheep in which PrP genotype, age, and infection status was known in order to study the possible role of nerves within LRS tissues in neuroinvasion. Here we determine the frequency and distribution of nerves within selected lymphoid tissues of scrapie-exposed and control sheep at the preclinical and clinical stages of disease.
Materials and Methods
Animals
Archived formalin-fixed, paraffin wax–embedded samples of distal ileum, retropharyngeal, mesenteric, and jejunal lymph nodes were sourced over a 6-year period from TSE-affected and control sheep. Affected sheep (n = 41) were defined as those in which PrPd could be detected by immunohistochemistry in CNS and at least one visceral tissue and were obtained from both natural disease and from experimental oral challenge studies. Of these, 62% of sheep from the 7- to 20-month-old group, 80% of sheep from the 22- to 40-month-old group, and 94% of sheep from the 53- to 90-month-old group were at clinical stages of disease. Controls were (1) scrapie-free sheep of New Zealand origin (n = 27), (2) sheep not exposed to scrapie (n = 6), and (3) sheep potentially exposed to natural or experimental scrapie but lacking detectable PrPd accumulation in any tissue (n = 53).
Tissue blocks were examined from sheep aged from 2 to 90 months and divided into 4 groups (Table 1). Several PrP genotypes were present in the animals studied and are listed in Table 1. The ARR/ARR and ARQ/ARR genotypes are classified as resistant to scrapie in the UK national sheep scrapie plan (where letters represent the single letter code for amino acids on each allele of the PrP gene at codons 136, 154, and 171) and were represented only in the PrPd negative control groups; all other genotypes were represented in both PrPd positive and PrPd negative groups. Of the animals studied, 76 were AA homozygous at codon 136 (of these 33 were of the ARR/ARR or ARR/ARQ genotype and fell into the control group), 35 were VA heterozygous, 14 were VV homozygous, and 2 were of unknown genotype. With the exception of the earliest age group, in which there were no VV homozygotes, all genotypes were represented in the age groups studied albeit at numbers too low to provide accurate statistical analysis.
Ileal Peyer's patch and lymph node sections studied (n = 1 per animal): PrPd status, numbers of ileal Peyer's patches and lymph nodes studied and genotypes of sheep are stated in relation to age.*

PrPd = abnormal form of the host-coded prion protein; LN = lymph nodes.
Ileal PPs were studied from all sheep and retropharyngeal, mesenteric, or distal jejunal lymph nodes from a selection of sheep.
Immunolabeling
Formalin-fixed tissue samples were embedded in paraffin wax and cut according to standard procedures for light microscopy. Immunohistochemistry was performed on serial sections from all tissue blocks containing secondary follicles using the monoclonal PrP-antibody R145 (VLA, Weybridge, UK) 16 and the monoclonal antibody protein gene product (PgP) 9.5 (Dako UK Ltd, Cambridgeshire, UK) against the neuron-specific PgP 9.5. Standard immunohistochemical techniques have demonstrated the presence of PgP 9.5 in neurons and nerve fibers at all levels of the central and peripheral nervous system. Sections were labeled as per the protocol in routine use in our laboratory, 6 R145 at a dilution of 1 : 2,000 and PgP 9.5 at a dilution of 1 : 1,000. When pretreated with formic acid and citrate autoclaving, R145 antibody labels only disease-associated forms (PrPd) of prion protein.
Double immunolabeling was also carried out on an ileal PP section from a 30-month-old clinically affected sheep. Standard immunohistochemistry was initially performed on sections incubated with R145 (see above) using di-amino benzidine as a chromogen. Without allowing slides to dry, the anti-PgP 9.5 antibody was applied, followed by further standard immunohistochemistry. Vector VIP substrate kit was used as a substrate as per kit instruction (Vector labs, Peterborough, UK). In order to clearly visualize both staining patterns, sections were very lightly counterstained with Meyer's hematoxylin.
Scoring
Numbers of immunolabeled follicles within each section were counted. Labeling of PrPd and nerves was scored using similar criteria with a score of 0–3 given for each of the light zones and dark zones (maximum available score = 6). The definitions of scores are listed below. Tissues identified as containing PrPd within one or more secondary follicles and with a score of 0.5 or above were classified as scrapie-affected. Although previous studies have shown that a proportion of sheep at preclinical stages of infection may have PrPd confined to LRS tissues, 5, 26 in this series, all animals with PrPd accumulations within follicles were also found to have accumulations within the CNS.
Within the light zones of the follicles, both FDC and TBM labeling were separately scored.
Nerves.
0 = no labeling in any follicles
0.5 = less than 3 fine nerve branches labeled in less than 5% of light/dark zones
1 = multiple fine nerve branches in less than 20% of light/dark zones and/or less than 3 thick-branched nerve fibers in less than 5% of light/dark zones
1.5 = multiple fine nerve branches in less than 50% of light/dark zones and/or less than 3 thick-branched nerve fibers in less than 20% of light/dark zones
2 = multiple fine nerve branches in less than 50% of light/dark zones and multiple thick-branched nerve fibers in less than 20% of light/dark zones
2.5 = multiple fine nerve branches in less than 70% of light/dark zones and multiple thick-branched nerve fibers in less than 50% of light/dark zones
3 = multiple fine nerve branches and multiple thick-branched nerve fibers in most light/dark zones
PrPd
0 = no labeling in any light/dark zones 0.5 = fine FDC or light TBM labeling in less than 5% of light/dark zones 1 = fine FDC or light TBM labeling in less than 20% of light/dark zones 1.5 = fine FDC or moderate TBM labeling in less than 50% of light/dark
zones 2 = moderate FDC or moderate TBM labeling in less than 50% of light/dark
zones 2.5 = moderate to intense labeling of FDC or intense TBM labeling in less
than 70% of light/dark zones 3 = intense labeling in most light/dark zones
Statistical analyses
Statistical tests were performed using GraphPad InStat (GraphPad Software, La Jolla, CA, USA). All tests were nonparametric Mann-Whitney tests, where 2 groups of figures were being considered and Kruskal-Wallis test for 3 or more.
Results
PrPd
PP secondary follicles from all sheep in the 2- to 4-month-old group were negative for PrPd (Fig. 1), which is broadly consistent with previous studies of natural disease in AA136 17 and VA136 sheep. At clinical stages of disease, the majority of follicles were positive for PrPd, although the actual proportions of positive follicles varied substantially between animals (55–100%). The precise time of exposures, and therefore incubation period, was not available for field cases, although the percentage of positive follicles and the mean magnitude of follicular PrPd accumulation did not vary significantly between the 3 older age groups (Fig. 1). Where present, labeling within the follicle was consistent with a linear FDC-type pattern, and a multi-granular intracellular TBM accumulation of PrPd as previously described. 15, 23
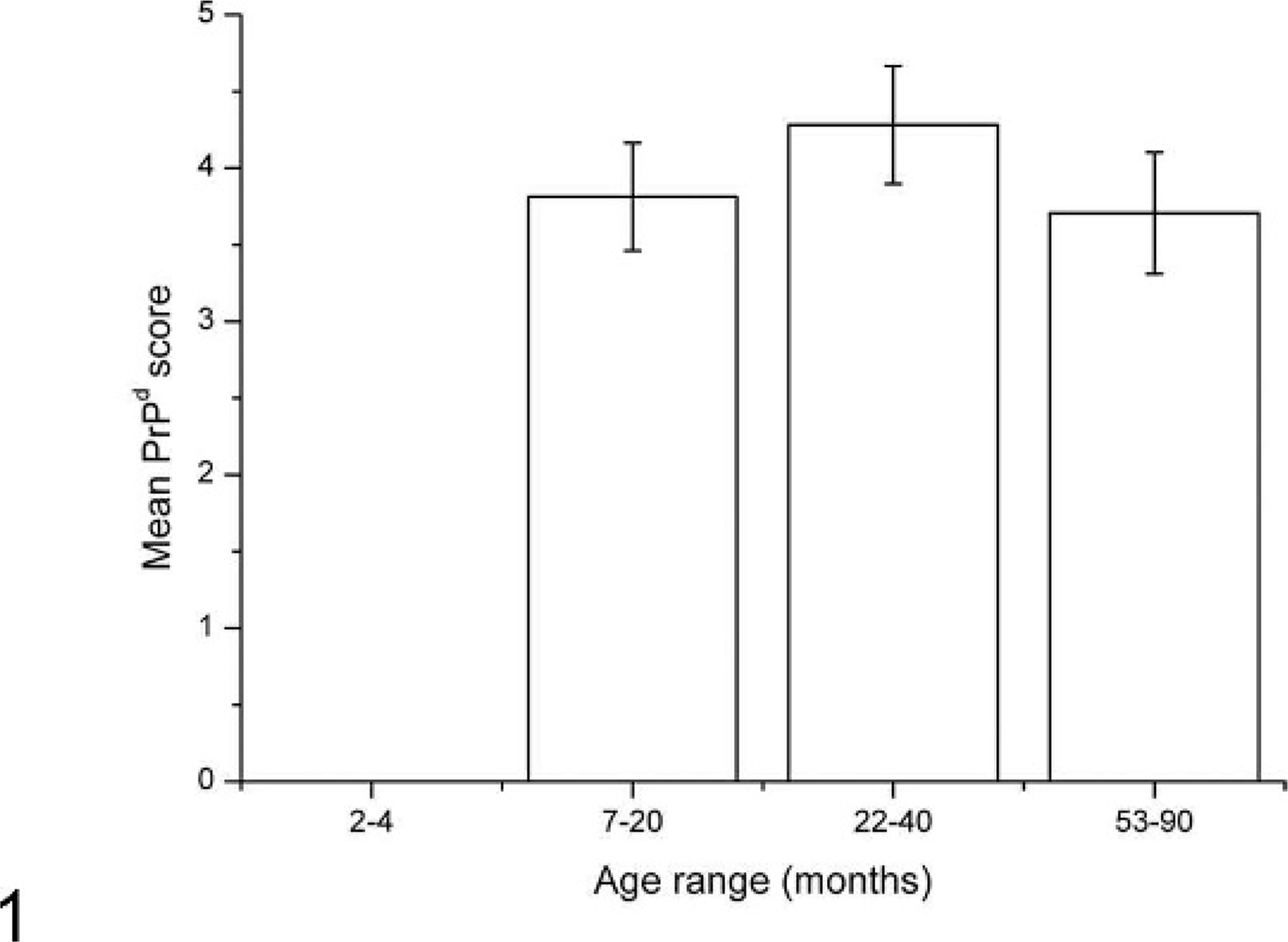
Magnitude of PrPd accumulation in scrapie-affected sheep in each age group in ileal Peyer's patch follicles. Within the 2- to 4-month-old group, no sheep showed PrPd within follicles. Within the 3 later age groups, there was no statistically significant difference in levels of PrPd between groups. Results are expressed as mean ± SEM for each age group.
Nerves
Innervated follicles were only identified within ileal PPs; all other lymph nodes examined did not contain nerve fibers within follicles.
Within the ileum, nerves were found in the mucosal and submucosal plexi, beneath the follicle-associated epithelium of the dome, in the T-cell dependent interfollicular zones, and within the lamina propria of villi and in secondary follicles of PPs. The capsule of the PP follicle was found to contain a network of nerve fibers sometimes in substantial numbers, along with the dark and light zones of the germinal centers. Within the 3 oldest age groups (7 to 90 months), when the PrPd positive sheep were compared with the scrapie-unexposed and PrPd negative sheep, there were no statistically significant different patterns of innervation, either in numbers of innervated PP secondary follicles or in the PgP 9.5 nerve score. Only 15% of PrPd accumulating follicles were PgP 9.5 positive, and the magnitude of follicular PrPd did not correlate with the magnitude of follicular innervation (data not shown). Thus, amplification of infectivity, as measured by presence of PrPd, did not influence the patterns of innervation.
No significant differences were found in the mean magnitude of innervation of follicles between the 3 oldest age groups (Fig. 2).
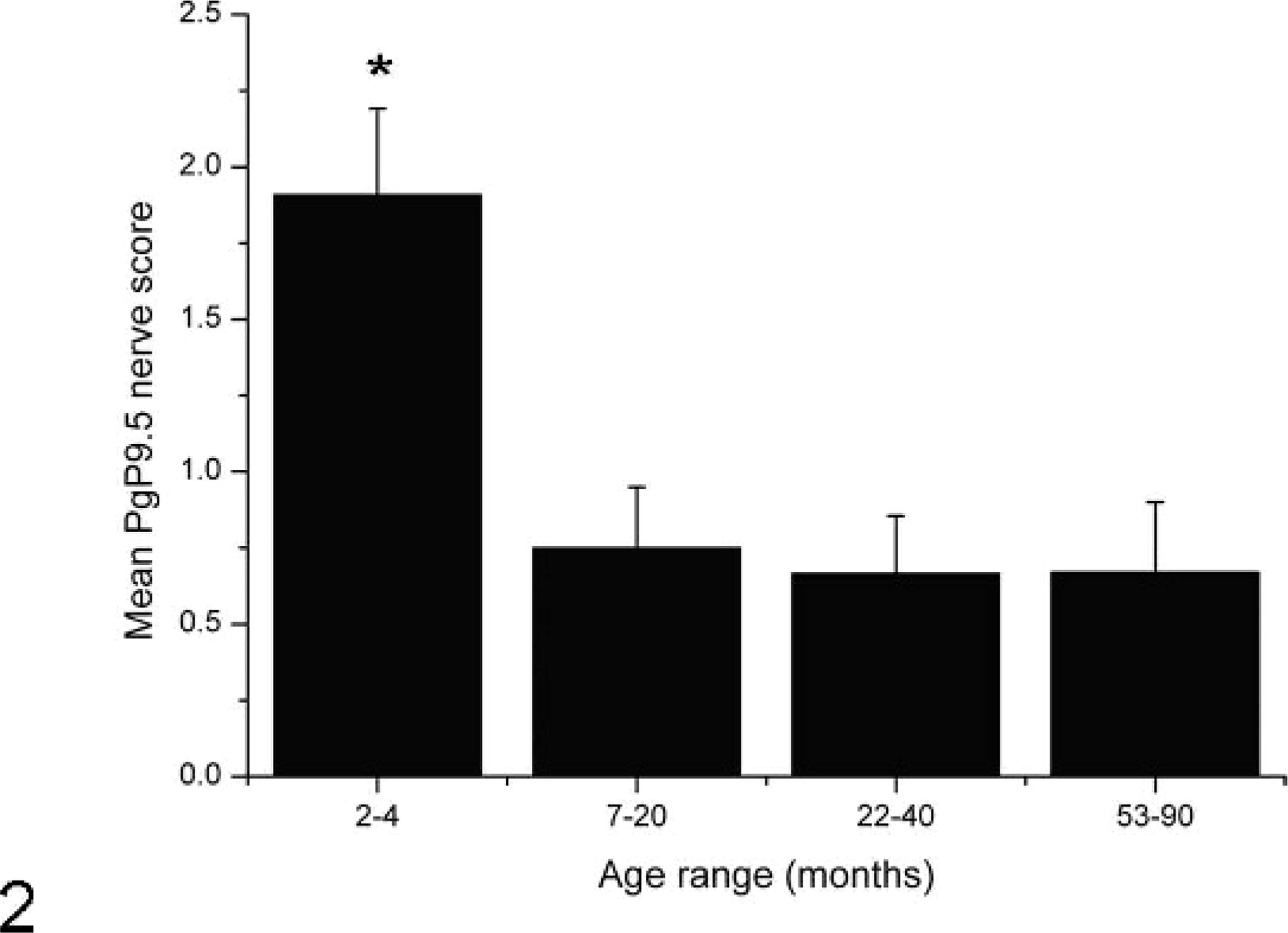
Magnitude of PgP 9.5 nerve labeling in each age group in ileal Peyer's patch follicles. Follicles from sheep aged between 2 and 4 months showed significantly higher nerve scores than at later time points (asterisk). Levels of innervation at the later time points did not vary significantly. Results are expressed as a mean ± SEM for each age group.
Within the PPs of PrPd positive sheep, 28 of the 41 sheep (68%) did not have any innervated follicles. Amongst the remaining 13 sheep, the average number of follicles that also contained nerve fibers was 42%. There was no significant difference in numbers of sheep with both PrPd and PgP 9.5 labeling between age groups.
As PrPd accumulation did not appear to affect the frequency or distribution of nerves within secondary follicles, the data for determining the effects of age on the frequency and magnitude of innervation were pooled for PrPd positive and PrPd negative tissues. Sheep within the 2- to 4-month age group showed significantly more nerves within the secondary follicles (Fig. 2) than older sheep (P < .05, using Kruskal-Wallis test, nonparametric analysis of variance [ANOVA]). It is well known that the number of lymphoid follicles within the gut decreases with age. 18, 21 Neither the absolute numbers of follicles across the whole intestine, nor the numbers of follicles per unit length were determined in this study; therefore the values are shown as proportions of innervated to noninnervated follicles. We did however determine that numbers of follicles within our studied sections decreased with age as expected (the average number of follicles per tissue section in the 2- to 4-month-old group = 44; 7- to 20-month-old group = 31; 22- to 40-month-old group = 18; and the 53- to 90-month-old group = 13). There were no significant differences in mean nerve score between the 3 older age groups (Fig. 2).
In the 2- to 4-month-old group, patterns of PgP 9.5 labeling varied greatly, from limited fine nerves within the dark zone of the follicle (0.5 score; Fig. 3a, b), to intense patterns of labeling consistent with both fine, unbranched nerves and thicker, branching nerves in the light zone, dark zone, and mantle areas of the follicle (4.5 score) (Fig. 3c). In the 3 youngest groups (2 to 40 months), innervation was significantly more frequent within the dark zone of the secondary follicles than within the light zones (P = .0217 using the Mann-Whitney test) (Fig. 4); in the oldest group (53 to 90 months), nerves were less abundant within the dark zones and more frequent within light zones. No association was detected between nerve frequency and genotype. In particular, VV sheep, which are putatively the most susceptible to scrapie, did not have conspicuously greater numbers of innervated follicles or more extensively innervated follicles when compared with those of the other genotypes, though insufficient numbers of sheep were available for meaningful statistical analysis.
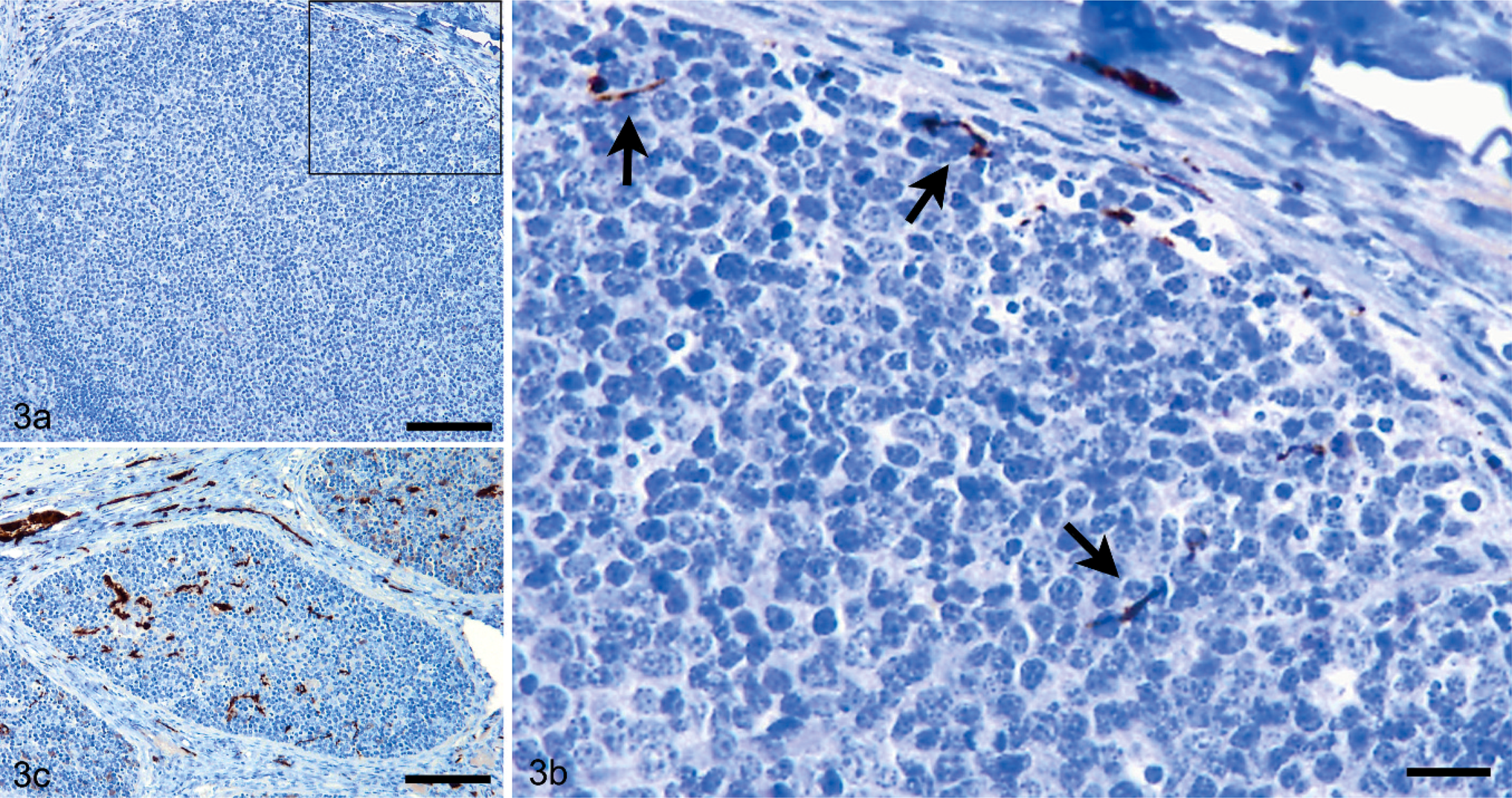
Immunolabeling for PgP 9.5 in the ileal Peyer's patch. Fig. 3a, b. Limited fine nerve branches are present within the dark and mantle zones of the follicle and in the capsule, giving an overall PgP 9.5 nerve score of 0.5. Bar = 250 µm (Fig. 3b is a higher magnification of the area outlined in Fig. 3a, where Bar = 45 µm). Fig. 3c. Abundant nerves are labeled within both the light, dark, and mantle zones of the follicle, giving a nerve score of 4.5. Bar = 250 µm.
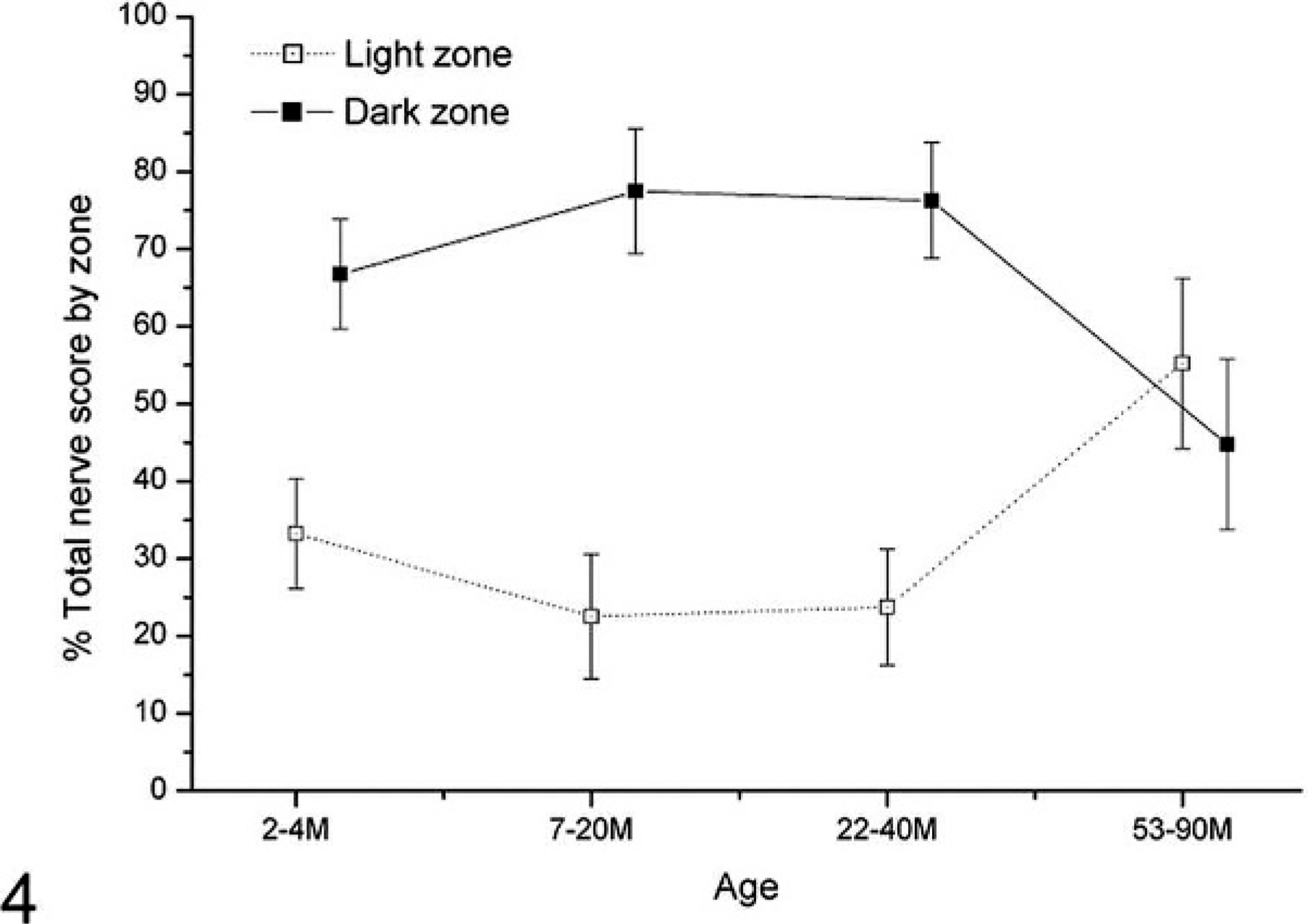
Ileal Peyer's patch light and dark zone PgP 9.5 nerve score between age groups. Patterns of labeling are altered within follicles of sheep in the 53–90 month age group—nerves are significantly more frequent within the light zones of follicles in the 53–90 month age group when compared with follicles of sheep between 2 and 40 months of age.
Within follicles of sheep from the 3 older age groups, a considerably greater number of follicles did not contain nerves when scores were compared with the youngest age group. The distribution of nerve scores of 0–4.5 were relatively consistent between animals in the 2- to 4-month-old group, while more than 40% of follicles from animals from each of the 3 older groups did not contain nerves.
Dual labeling was performed to show the relationship between nerves and cells accumulating PrPd within follicles. Where PrPd was present in follicles it was demonstrated that nerve fibers were in close contact with both FDCs of the light zone and TBMs of the dark and light zones, which accumulate PrPd (data not shown).
Discussion
In this study we have shown that scrapie infection and the associated PrPd accumulation do not influence the magnitude, frequency, or distribution of nerves within secondary follicles of sheep PPs, or of the other lymphoid tissues examined in this study. We have further shown that the apparent absence of nerves in secondary follicles of many scrapie-affected sheep and the low percentage (15%) of innervated PrPd positive follicles argues against the suggestion that infection of the ENS occurs subsequent to amplification of infectivity in LRS tissues. Furthermore, a significant number of scrapie-affected sheep (68%) did not show any innervated follicles and a further fraction had nerves only within the mantle zone; an area of the follicle in which contact with the cell type that sustains infection, the FDC, is absent. The availability of tissues from scrapie-affected and control sheep of known PrP genotypes and ages from a wide range of different studies made this study possible, but the large numbers of possible sheep genotypes precluded statistically meaningful comparisons in all ages of sheep. Nevertheless our studies show that the putatively most susceptible sheep genotype VRQ/VRQ does not have widely different numbers or distributions of nerves from those of intermediate susceptible sheep or even sheep of the most resistant ARR/ARR genotypes.
This study did not investigate all PPs of the intestine. With regard to scrapie-affected sheep lacking nerves in follicles, it is possible that small numbers of secondary follicles at other sites within the gastrointestinal tract could also contain both innervated follicles and PrPd accumulations. However, the readiness with which scrapie can be transmitted orally argues that uptake of infection will involve widely distributed sites and not be dependent on a chance uptake of infectivity in infrequent, randomly distributed innervated follicles. Furthermore, although this study focused on the PPs, we have no evidence that innervation is more frequent in other lymphoid structures; indeed, our evidence is to the contrary: that other LRS tissues do not have any innervated secondary follicles. Thus, with or without prior amplification of infectivity in germinal centers, neuroinvasion is unlikely to be the result of retrograde transportation of infectivity via nerves in secondary follicles of PPs or other LRS tissues.
It has been considered likely that the ENS, and its connections either to the vagal nerve and the DMNV, or the CMG, which connects to the splanchnic nerves followed by the intermediolateral tracts of the spinal cord, are the initial sites of CNS PrPd accumulation and thus represent the sites at which neuroinvasion occurs. 1, 25 Following oral infection, PrPd accumulates around mature FDCs and in TBMs of lymphoid nodules of the gut-associated lymphoid tissues. 1, 13 FDCs, present within all LRS tissues, are the cell types necessary for prion propagation within the LRS. 2 Ultrastructural immunocytochemical studies of PPs of sheep indicate that these cells are in close contact with nerve fibers and that this site could be important for neuroinvasion following oral infection with a TSE agent. 8 However, nerve fiber endings are also found adjacent to lacteals and to the villous epithelium. 8 It is therefore possible that infection may reach the ENS directly and without the intervention of PPs.
As described earlier, both natural and experimental situations are recognized in which neuroinvasion occurs in the absence of lymphoid PrPd accumulation or infectivity. 6, 23 A recent study has now shown that the initial sites of PrPd accumulation in the brain of scrapie-affected sheep are the same irrespective of the route of inoculation, including subcutaneous and intravenous routes of challenge, 20 an observation that is difficult to reconcile with retrograde spread of infectivity to the CNS from nerves of LRS tissues. In the same studies, the circumventricular organs (CVOs) were consistently affected at early stages of disease; as these CVOs contain fenestrated blood vessels, they may distribute blood-derived infectivity to target regions within the brain suggesting that neuroinvasion may occur through the hematogenous route. 20 Thus, field and experimental data suggest that neuroinvasion may not invariably be associated with prior amplification of infectivity in secondary follicles and subsequent retrograde transportation of infectivity to the brain. Several studies have shown that infectivity is present in the blood of scrapie-affected sheep 11, 12, 19 and, as mentioned above, recent studies have provided evidence for a hematogenous neuroinvasion pathway. 20 Besides factors such as PrP genotype and agent strain, young lambs appear to be more susceptible than adult sheep to oral TSE infection. 10 This may be in connection with a higher permeability of the gut mucosa, or a higher abundance of lymphoid follicles in the PPs when compared with older sheep. While these factors could result in higher levels of circulating infection, which could access the brain through the CVOs, it may be argued that young lambs also appear to have more frequent and abundant innervation of PP follicles, which could result in nerve-mediated neuroinvasion. However, this increased innervation is true by comparison with older age groups, but in absolute figures, neither the proportion of innervated follicles (an average of 30% per animal), nor the level of innervation of nerve-containing follicles (scores of less than 2 in 50% of follicles), or the distribution of those nerves (mostly dark zone) argue for a frequent nerve-mediated neuroinvasion.
In conclusion, scrapie infection did not appear to affect the frequency or distribution of innervation within PPs or of the other LRS tissues examined in this study. The low proportion of follicles that contain both PrPd and nerves particularly within the youngest age group, and the significant fraction of sheep that lacked nerves in any PrPd containing follicles, is not consistent with nerve-mediated neuroinvasion after LRS amplification of infectivity. Indeed this study argues that LRS amplification of infectivity may serve principally to provide increased infectivity within blood. Nevertheless, a proportion of secondary follicles in sheep PPs contained nerve fibers, which were more numerous in young animals, declining after 4 months of age.
Footnotes
Acknowledgements
The authors would like to thank Callum Donnelly, Elaine Craig, Lynne Fairlie, and Maria Oliva for technical work carried out during this study.
