Abstract
To investigate the relative involvement of the olfactory region in classical bovine spongiform encephalopathy (BSE), immunohistochemical labeling of prion protein scrapie (PrPSc) was scored in the brainstem, frontal cerebral cortex, and olfactory bulb of cattle with natural and experimental clinical cases of BSE in Great Britain. The intensity of immunolabeling was greatest in the brainstem, but PrPSc was also detected in the olfactory bulb and the cerebral cortex. A diffuse, nonparticulate labeling, possibly due to abundance of cellular PrP, was consistently observed in the olfactory glomeruli of the cases and negative controls. Involvement of the olfactory bulb in BSE and other naturally occurring TSEs of animals raises speculation as to an olfactory portal of infection or a route of excretion of the prion agent.
Bovine spongiform encephalopathy (BSE) is a progressive, fatal neurologic disease of cattle that was first discovered in Great Britain (GB). The BSE epidemic in GB resulted from a foodborne infection with a major strain of a transmissible spongiform encephalopathy, or prion agent. Recently there have been geographically widespread reports of small numbers of so-called atypical BSE in cattle, designated principally H and L types, based on the molecular weight of the unglycosylated band of the disease-specific isoform of prion protein scrapie (PrPSc) on Western blotting (WB). Only two cases of atypical BSE have been reported from GB; both were H type and were diagnosed on molecular characterization. 10 Detailed neuropathologic description of atypical BSE is limited to the L type, originally named bovine amyloidotic spongiform encephalopathy (BASE). 3 In classic BSE, the greatest extent of PrPSc detected by immunohistochemical labeling is observed in the brainstem, whereas there is far less PrPSc accumulation in the cerebral cortex. In contrast, the brainstem in BASE cases is reported to show only weak PrPSc immunolabeling, and prion protein (PrP) amyloid plaques are present and most numerous in the thalamus, subcortical white matter, the deeper laminae of the cerebral cortex, and the olfactory bulb. 2 In GB BSE cases, PrP amyloid plaques have been inconsistently observed in the thalamus, 12 but the olfactory bulb has not been studied previously. Here we report an immunohistochemical study of the relative PrPSc accumulations in brainstem, frontal cerebral cortex, and olfactory bulb of seven arbitrarily selected cattle with BSE to determine pathologic features that might reflect phenotypic or pathogenetic variations.
Medulla oblongata at the level of the obex, frontal cerebral cortex, and olfactory bulb, inclusive of olfactory glomeruli, were examined from five naturally occurring cases of BSE and two experimental cases (Table 1). The BSE cases were detected as clinical suspects among a GB population born after the reinforced ban on the feeding of meat and bone meal to farmed animals. The experimentally exposed cattle were infected orally, one with 1 g and the other with 100 g of pooled brain homogenate from clinical cases of BSE. 1 To provide a range of negative controls, 26 additional animals were examined. Eleven were healthy calves, 5–6 months old, sourced from a farm that had had no BSE suspect cases since 1993. Ten brains were sourced from healthy slaughtered cattle, approximately 2 years old. Four were adult cattle culled at the termination of an experiment at ages 7 years 8 months to 7 years 9 months, after having been inoculated intracerebrally as calves with salivary gland from cattle orally dosed with BSE agent and killed 18 months after exposure. Three of these displayed neurologic signs that, on pathologic examination, were not referable to BSE. The remaining control was an animal reported as a clinical suspect that was born after the reinforced ban but was diagnosed negative.
Distribution and scoring of PrPsc immunolabeling (mAb R145) in selected brain areas of cattle with confirmed cases of BSE. ∗
Scoring scale of PrPSc abundance: 0 = no PrPSc, + = focal/infrequent, ++ = moderate occurrence, +++ = abundant.
For the immunohistochemical examinations, the monoclonal antibody (mAb) R145 was used. 1 R145 is an anti-PrP rat mAb raised against the bovine peptide sequence 217–231. The abundance of PrPSc deposition was assessed subjectively on a four-point scale (Table 1). In negative controls, two additional anti-PrP mAbs, 12F10 and P4, and a nonimmune rat IgG were applied to olfactory bulb and frontal cortex; 12F10 is raised to a synthetic peptide corresponding to 142–160 of human recombinant PrP and targets the C-terminal of the PrP and P4 is an anti-PrP mouse mAb raised against the ovine peptide sequence 89–104 of the N-terminal of the PrP.
An immunoblotting method (WB) was applied to brainstem samples of BSE cases to confirm PrPSc detection and molecular characterization independent of the immunohistochemical approach. 9 This method was also applied to olfactory bulb of six animals selected from adult and subadult negative controls (two 5- to 6-month-old calves, two of the abattoir-slaughtered healthy cattle, and two experimentally inoculated cattle).
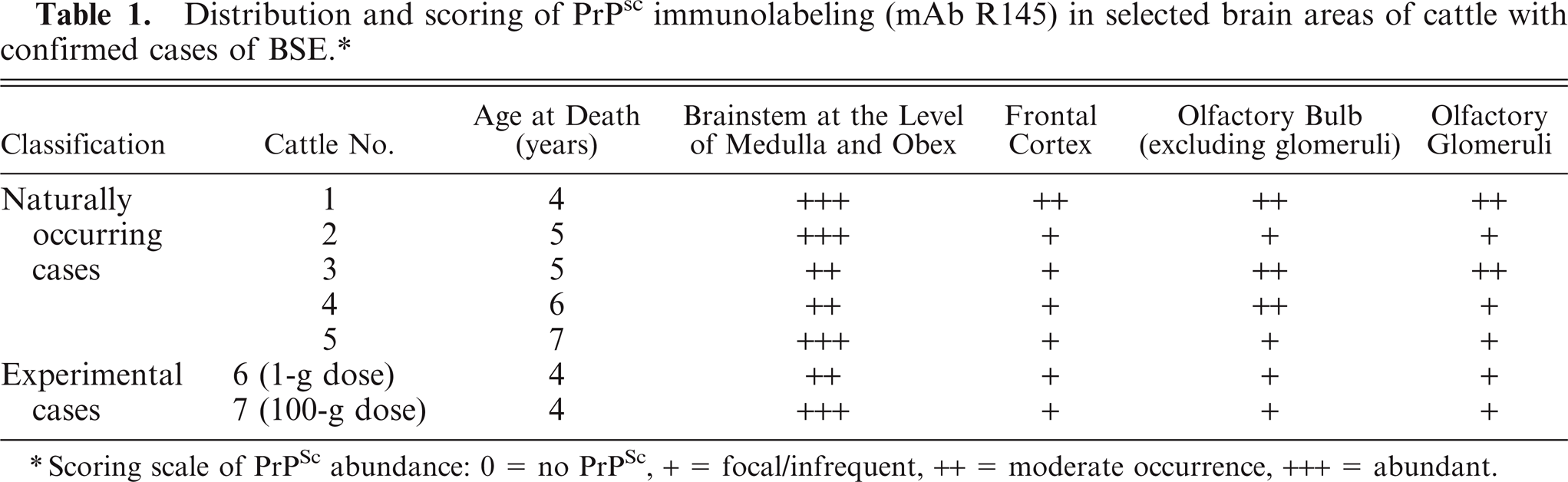
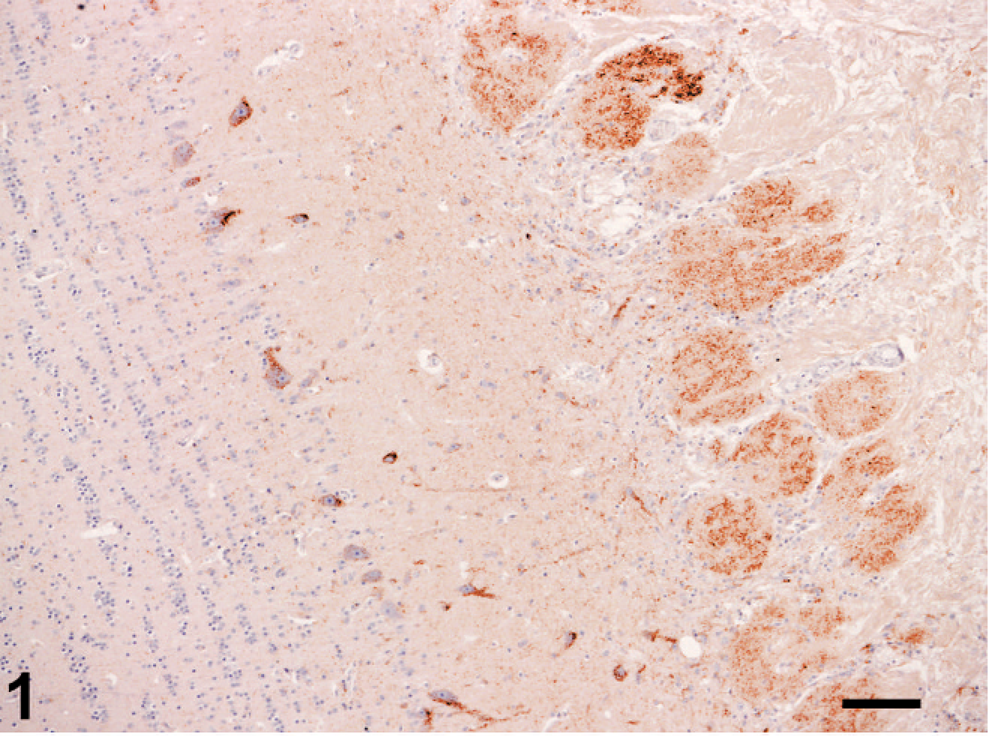
In the confirmed clinical naturally occurring cases and the two confirmed experimental cases examined, PrPSc immunolabeling was in general more marked in the brainstem at the level of obex than in the forebrain structures examined (Table 1). In two cases, in which only moderate labeling was scored in the brainstem, similar levels of labeling were detected in olfactory bulb. The morphologic appearances of PrPSc labeling identified were consistent with other published observations on BSE. 2 Particulate and/or granular PrPSc immunolabeling was consistently observed in the olfactory glomeruli of the olfactory bulb in all BSE cases (Figs. 1, 2) but varied in extent. Outside the glomeruli in the olfactory bulb and the frontal cortex, particulate neuropil, stellate glial, and granular neuronal cytoplasmic labeling was sparse and unevenly distributed or focal.
Olfactory bulb; cattle 1. PrPSc labeling of olfactory glomeruli and neurons. Immunolabeling with mAb R145 and hematoxylin counterstain. Bar = 100 µm.
Olfactory bulb; cattle 1. Detail of PrPSc labeling of olfactory glomeruli. Immunolabeling with mAb R145 and hematoxylin counterstain. Bar = 50 µm.
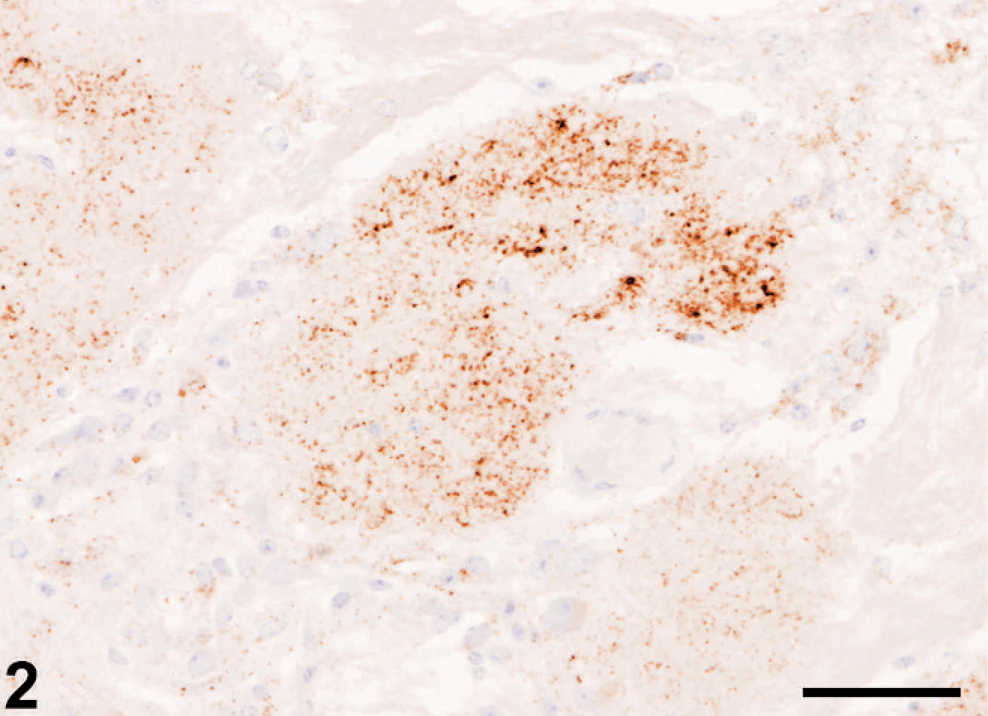
A faint, diffuse, nonparticulate labeling was a constant feature in the olfactory bulb glomeruli of all BSE cases (labeled with mAb R145) and negative control cattle (Fig. 3) when labeled with mAbs R145, 12F10 or P4, but it was not observed in olfactory bulb sections immunolabeled with rat IgG. No labeling was detected in olfactory bulb outside the glomeruli or frontal cortex areas of negative controls labeled with mAbs R145, 12F10, or P4. In all BSE cases vacuolar changes featured in the medulla but not in the forebrain structures examined. WB examination of brainstem in all of the positive cases gave a uniform molecular weight with the mAb 6H4, and none were detected by the mAb P4, providing the characteristic molecular profile for classical BSE. Olfactory bulbs from negative control animals, including cattle aged 7 years, examined by WB, gave negative results.
Olfactory bulb; negative control, cattle 8. Faint diffuse, nonparticulate background labeling confined to glomeruli (∗), with occasional more intense foci (arrow). Immunolabeling with mAb R145and hematoxylin counterstain. Bar = 50 µm.
This study records the involvement of the olfactory bulb as a feature of natural cases and of experimental cases of classical BSE after oral exposure. Immunolabeling in the frontal cortex scored similarly or only slightly less than that of the olfactory glomeruli or adjacent olfactory bulb consistent with olfactory involvement being secondary to the propagation of PrPSc in other brain regions. In no cases were PrPSc plaques observed. Accumulations of PrPSc were, in general, greatest in the brainstem, consistent with previous accounts of the pathology of classic BSE and, indeed, with proposed primary infection of the central nervous system, via alimentary innervations and spinal and medullary pathways, after oral exposure. 1
In BASE, in addition to PrP-amyloid plaques in the olfactory bulb, the highest levels of PrPSc were recovered from the thalamus and olfactory regions. 2 Also, in both captive and free-ranging mule deer with chronic wasting disease, the olfactory cortex has been found to be among the most severely affected areas of the brain. 8 Such patterns may be solely a reflection of selective vulnerability of certain neuroanatomic loci and, in end-stage disease, a reflection of phenotype, but equally, they arouse speculation as to possible olfactory portals of infection or excretion of agent. In human transmissible spongiform encephalopathies (TSEs), PrPSc immunolabeling has been reported in the olfactory tract of a variant Creutzfeldt-Jakob disease patient, and in sporadic Creutzfeldt-Jakob disease (sCJD), selective deposition of PrPSc in olfactory glomeruli, olfactory tracts, and olfactory cortex is recorded. 6 In the latter study, PrPSc was also reported in the cilia of olfactory receptor neurons and basal cells of the olfactory epithelium but not in the respiratory epithelium. Detection of PrPSc in an olfactory mucosa biopsy, performed 45 days after disease onset in a sCJD patient, led to the suggestion that the involvement of olfactory epithelium might be an early event in sCJD. 6
PrPSc accumulation is also reported in the olfactory and vomeronasal epithelium of hamsters after intracerebral inoculation of the HY strain of the transmissible mink encephalopathy agent, 4 implicating centrifugal spread from the brain as the most likely mechanism. The olfactory/cribriform plate/nasal lymphatic pathway has a well-recognized important role in the drainage of cerebrospinal fluid (CSF) from the cranial vault. 7 That this might also be a means of transport of TSE agents from the intracranial compartment and ultimately to the blood is a possibility. However, infectivity in CSF in naturally occurring TSEs of animals has been investigated rarely and detected only in scrapie of sheep and goats. 5 Demonstration of PrPSc by immunohistochemistry is dependent on a knowledge of disease-specific labeling forms and their distribution patterns in relation to the particular prion disease, species, or experimental model under consideration. As seen in this study, apparently immunologically specific but unwanted labeling is sometimes encountered and requires careful interpretation. In this instance such unexpected labeling may be due to the abundant expression of cellular prion protein (PrPc) in olfactory neurons. 6 Because each olfactory glomerulus contains the convergent axons of olfactory receptor neurons and the dendrites of the secondary-order neuron, the mitral cell, the PrP-labeling at this site is not associated with neuronal perikarya. Further approaches are required to determine the subcellular location of PrP in the olfactory glomerulus.
The present study demonstrates that, in clinical cases, involvement of the olfactory lobe is a consistent phenotypic feature of classic BSE but, in contrast to BASE, 3 it is not preferentially affected compared with the cerebral cortex or brainstem. In studies of the pathogenesis of classical BSE after oral exposure, infectivity has not been demonstrated in nasal mucosa, 11 but it has been shown, albeit at low titer, in this tissue in clinical cases of scrapie of sheep and goats. 5 The detection of PrPSc in olfactory bulb in this study and indeed in other TSEs in terminal disease suggests that olfactory pathways cannot be excluded as a secondary or ancillary route of infection.
Footnotes
Acknowledgements
We thank the many support staff of the Department of Pathology and the Animal Services Unit, Veterinary Laboratories Agency-Weybridge, UK. Thanks also to Dr. John Spiropoulos for his assistance with the illustrations. The experimental study from which tissues of two animals were used in this study was funded formerly by Ministry of Agriculture, Fisheries and Food and latterly by Department for the Environment, Food and Rural Affairs and the Food Standards Agency. The study was carried out with the helpful support of Danny Matthews, formerly TSE programme manager, VLA. The travel opportunity for Yoon Hee Lee was sponsored by the Korean Civil Service Commission and National Veterinary Research and Quarantine Service.
