Abstract
Scrapie and bovine spongiform encephalopathy (BSE) are both prion diseases affecting ruminants, and these diseases do not share the same public health concerns. Surveillance of the BSE agent in small ruminants has been a great challenge, and the recent identification of diverse prion diseases in ruminants has led to the development of new methods for strain typing. In our study, using immunohistochemistry (IHC), we assessed the distribution of PrPd in the brains of 2 experimentally BSE-infected sheep with the ARQ/ARQ genotype. Distribution of PrPd in the brain, from the spinal cord to the frontal cortex, was remarkably similar in the 2 sheep despite different inoculation routes and incubation periods. Comparatively, overall PrPd brain distribution, evaluated by IHC, in 19 scrapie cases with the ARQ/ARQ, ARQ/VRQ, and VRQ/VRQ genotypes, in some cases showed similarities to the experimentally BSE-infected sheep. There was no exclusive neuroanatomical site with a characteristic and specific PrPd type of accumulation induced by the BSE agent. However, a detailed analysis of the topography, types, and intensity of PrPd deposits in the frontal cortex, striatum, piriform cortex, hippocampus, mesencephalon, and cerebellum allowed the BSE-affected sheep group to be distinguished from the 19 scrapie cases analyzed in our study. These results strengthen and emphasize the potential interest of PrPd brain mapping to help in identifying prion strains in small ruminants.
Prion diseases, or transmissible spongiform encephalopathies (TSEs), are neurodegenerative diseases that include classical and atypical types of scrapie in sheep and goats, chronic wasting disease (CWD) in deer and elk, as well as Creutzfeldt-Jakob disease (CJD) in humans. In 1986, bovine spongiform encephalopathy (BSE) emerged in cattle, 32 and 10 years later, the appearance of variant Creutzfeldt-Jakob disease was assumed to result from BSE transmission to humans.7,10,16 This increased the attention given to other potential sources of human infection such as sheep and goats, which were shown to be experimentally susceptible to BSE by oral route. 12
The possibility of transmission of this agent within small ruminant herds was of considerable concern, as some flocks could have been exposed to BSE-contaminated meat-and-bone-meal (MBM).8,33 Indeed, natural BSE infection in a goat in France was demonstrated by a wide range of discriminatory methods, 11 and a potential similar case in a goat was found in a retrospective analysis of archived material in Scotland. 19
Historically, TSE strain identification has been done by transmission to different mouse lines by analyzing the incubation period and the distribution of spongiform lesions in affected brains. 5 This approach requires several sub-passages to adapt the agent to the host before the incubation times and lesion profiles can be established and is therefore extremely laborious and time consuming. 5 More recently, immunochemical techniques that analyze either the protease sensitivity and cleavage site of PrPd in vitro 28 or its intracellular truncation pattern in situ 20 and PrPd brain mapping methods14,26,15 have been used to discriminate TSE strains. We have previously shown the potential of PrPd brain mapping analyses to discriminate TSE strains in mice. 21 In this study, we investigated whether this approach is also suitable for discriminating, by the use of immunohistochemistry (IHC), between BSE and scrapie in sheep. The results indicate that a detailed IHC analysis of the types and intensity of PrPd deposits in different brain areas may be a suitable approach to distinguish the BSE infection from scrapie in formalin-fixed tissues.
Materials and Methods
Animals and Tissue Samples
All sheep samples used in this study are recorded in detail in Table 1 . Brains from 2 sheep with the ARQ/ARQ genotype for the prion protein, which had been experimentally infected with the French BSE case no. 26, developed clinical signs of TSE, respectively 2 years post-inoculation (SB1, intraperitoneal inoculation) and 4 years post-inoculation (SB3, intrasplenic injection). 23
Details on Sheep Clinically Affected With Spongiform Encephalopathies
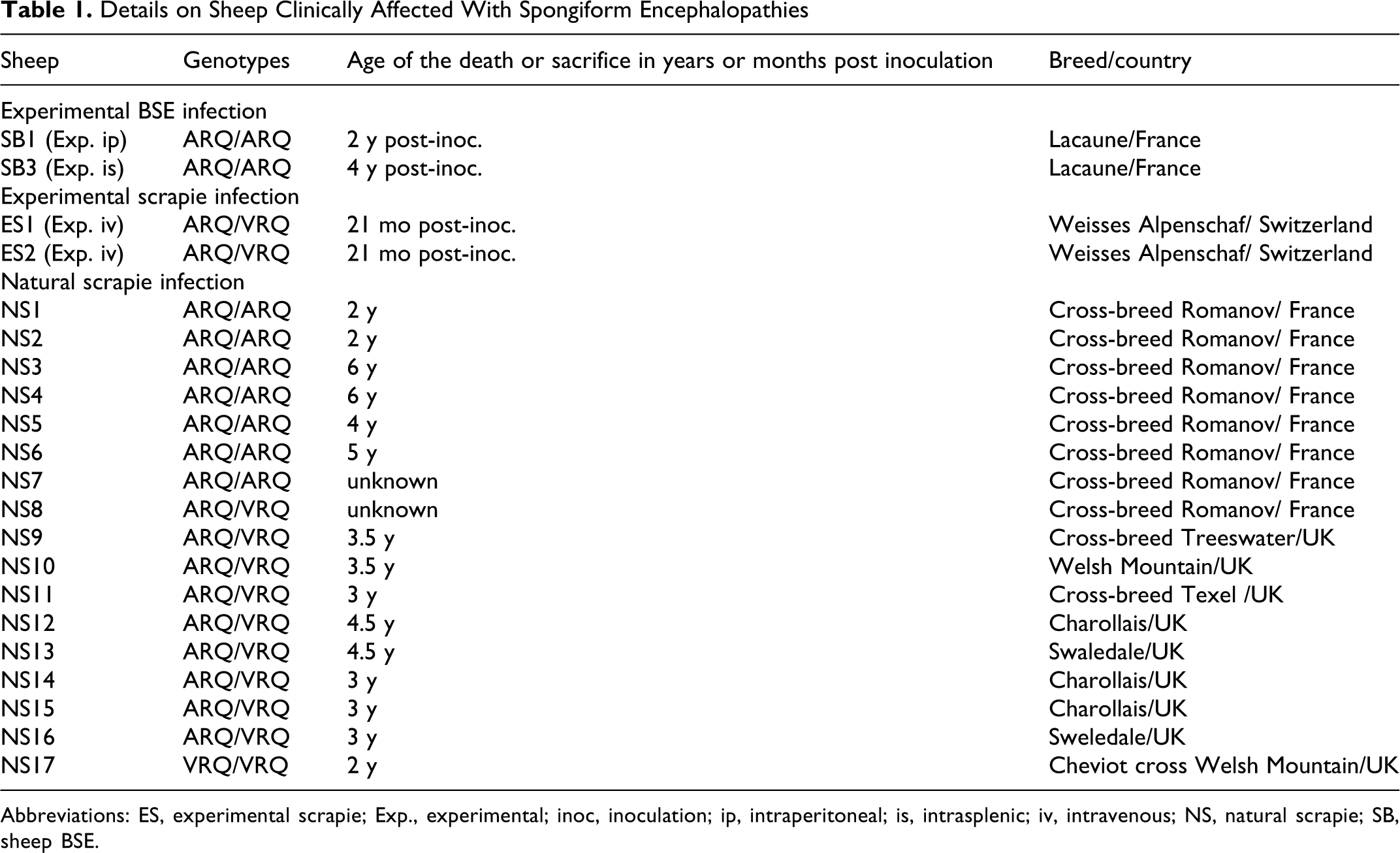
Abbreviations: ES, experimental scrapie; Exp., experimental; inoc, inoculation; ip, intraperitoneal; is, intrasplenic; iv, intravenous; NS, natural scrapie; SB, sheep BSE.
Experimental and natural scrapie cases with clinical signs of TSE were used for comparison of the neuroanatomical PrPd profiles (details in Table 1). The 2 sheep infected by the iv route with a scrapie VRQ/VRQ field case (20 mL of a 1% brain homogenate) had developed clinical signs of TSE and were killed 21 months post-inoculation.
Sections of the frontal cortex, basal nuclei, thalamus and hypothalamus, mesencephalon and hippocampus, medulla oblongata, and cerebellum were analyzed for PrPd brain mapping. For each of the experimental and natural scrapie cases, a possible BSE-like infection was ruled out by analyzing either the PrPd cleavage sites using a differential ELISA assay 28 or the intracellular truncation pattern of PrPd in IHC.20,23
Immunohistochemistry
Formaldehyde-fixed brain samples were routinely dehydrated and embedded in paraffin. To ensure adhesion, sections (5 µm) were placed onto pretreated glass slides (StarFrost, Medite Histotechnic, Burgdorf, Germany) and dried. The slides were dewaxed and rehydrated in water, then used for PrPd immunohistochemical analysis following a previously described procedure.2,22,24 Sections were incubated overnight with the primary antibody, SAF84 Mab (Spi-Bio, Massy, France), and then rinsed. The primary antibody was detected using the avidin-biotin-peroxidase complex (ABC) system (Vector Laboratories, Burlingame, CA, USA) and 3,3′-diaminobenzidine DAB or DAB-nickel (Zymed, San Francisco, CA, USA) as chromogens, which produce brown or black deposits at the site of a positive reaction. After slight counterstaining using aqueous hematoxylin, slides were covered and analyzed under a light microscope (Olympus, Rungis, France) coupled with an image analysis workstation (Biocom, Les Ulis, France). Nonspecific binding was controlled by omitting the primary antibody and using a normal mouse serum (Zymed, San Francisco, CA, USA) with the same pretreatments.
PET-Blot
The Paraffin-embedded tissue (PET)-blot method was used to visualize the proteinase K-resistant protein in situ (PrPres) and to confirm the specificity of PrPd deposits in the brains of both BSE-infected sheep. The detailed procedure has been described in previous articles.21,27 Illustrations of PET-blot results are available online in the supplementary data, illustrations 1 to 11 (si1 to si11), available at http://vet.sagepub.com/supplemental (for the identification of neuroanatomical sites of the supplemental illustration see table 2).
Identification of Neuroanatomical Sites (see supplemental illustrations)
Scoring System
A scoring system that reflected the intensity of PrPd immunolabeling in IHC on the brain tissue sections of BSE- and scrapie-infected sheep was applied in 5 levels of PrPd accumulation (1: minimal, 2: slight, 3: moderate, 4: marked, 5: intense). Schematic brain sections I and II (frontal cortex), III (basal nuclei), IV (thalamus), V (thalamus/hypothalamus), VI (anterior mesencephalon, hippocampus, temporal cortex), VII (posterior mesencephalon, occipital cortex), VIII (pons, cerebellum), IX and X (medulla oblongata, cerebellum), and XI (cervical spinal cord) illustrate the PrPd distribution in BSE-infected sheep (supplementary data, figure plate).
Results
Neuroanatomical Distribution and Deposition Types of PrPd in BSE-Infected Sheep
The neuroanatomical distribution of PrPd was easily assessed using the PET-blot as a complementary approach to IHC analyses and was found to be similar in the 2 BSE-infected sheep. The largest amount of PrPd accumulation was detected in the basal nuclei (section III), thalamus, hypothalamus (sections IV and V), mesencephalon (sections VI and VII) and the medulla oblongata (section X). PrPd IHC staining appeared moderate in the frontal cortex and was minimal in the parietal and occipital cortex. In the cerebellum, PrPd labeling was minimal to slight. Using IHC, different types of PrPd deposits (intraneuronal, intraglial, stellate, subpial, perivascular, subependymal, linear, fine punctate, coarse particulate, coalescing, and perineuronal) were distinguished.
Both BSE-infected sheep had very similar PrPd brain profiles, although some discrete differences were identified. In sheep SB1, the overall PrPd accumulation was more intense in all the structures analyzed when compared to sheep SB3. Furthermore, in sheep SB1, the subpial labeling was more intense in the frontal cortex. In the midbrain, the main PrPd accumulation was of a stellate type in both sheep, but there was less intraglial labeling in sheep SB3.
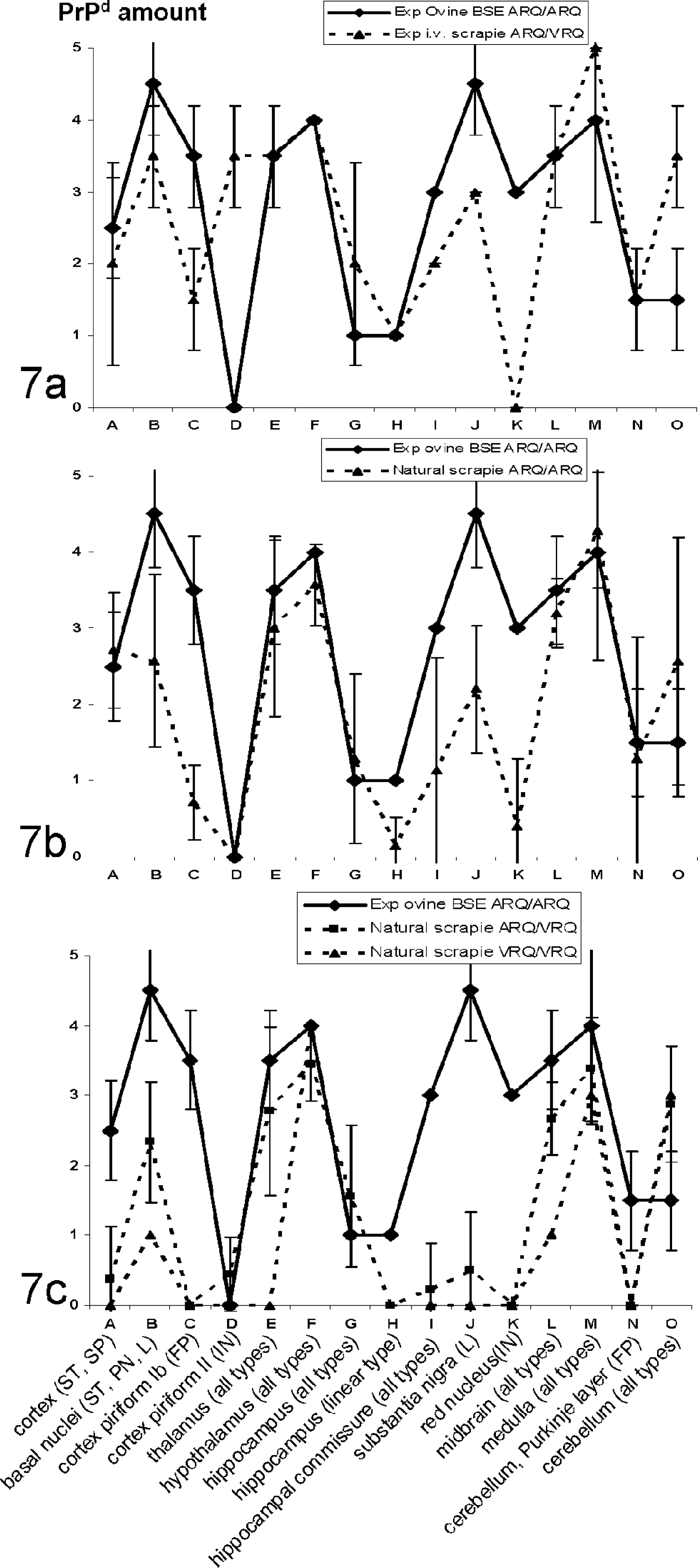
A preliminary evaluation of the PrPd brain distribution in all sheep in the study and data reported in the literature 15 prompted us to analyze the types and amount of PrPd deposits in specific brain areas in all the TSE cases. Figures 7a-c, which illustrate the profiles of PrPd brain deposits, were generated by quantifying either specific types of PrPd deposits in particular brain areas (eg, linear type in the substantia nigra) or all types of PrPd in larger structures (eg, medulla).
Cortex
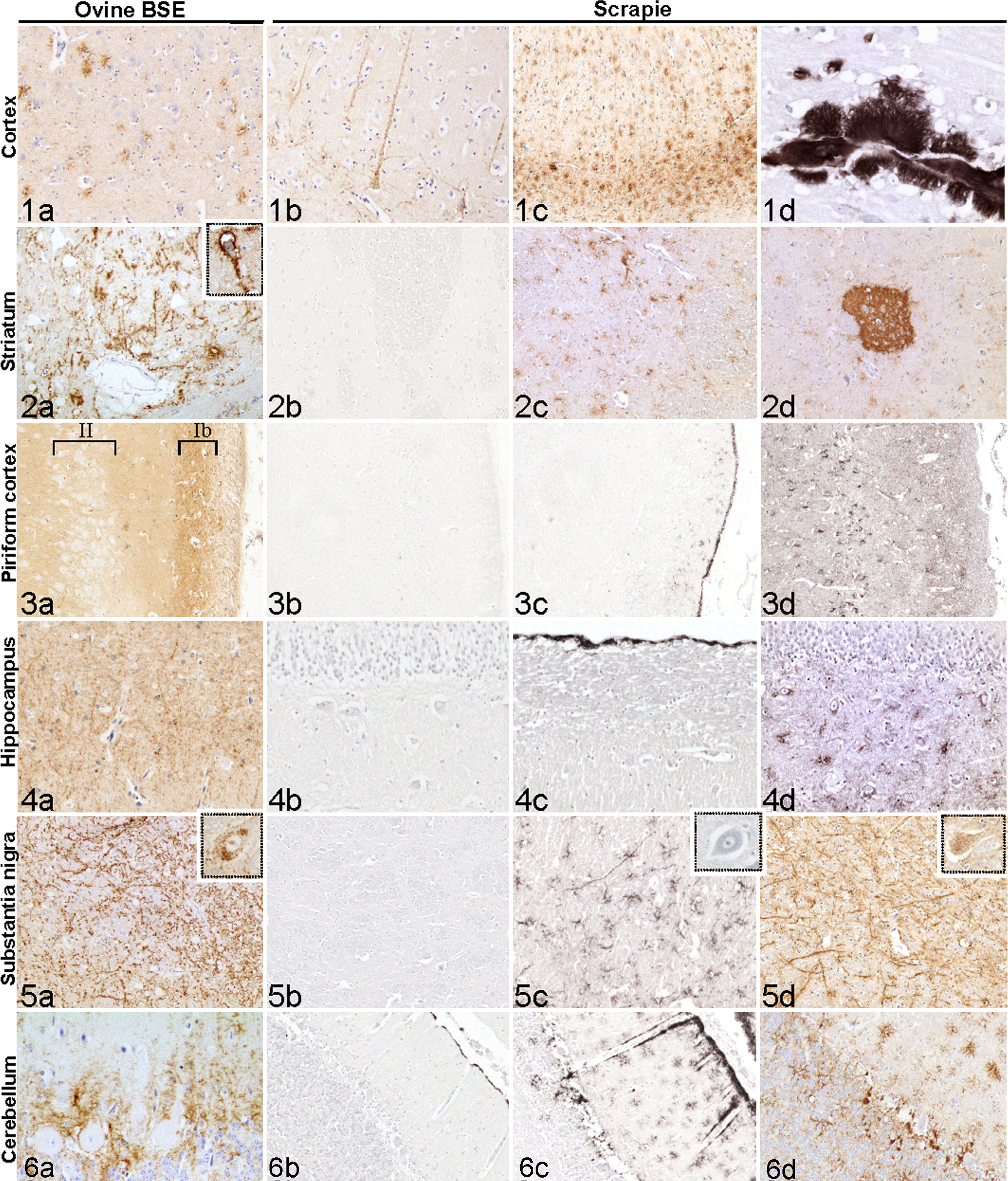
Slight to moderate PrPd accumulation was observed in the frontal cortex (particularly in the rhinal fissure). In more caudal cortical areas such as the parietal and occipital cortex, minimal traces of PrPd were noted. Stellate and subpial types predominated in the grey matter (Figs. 1a and Si1) and were associated with less frequent perivascular and stellate types in the white matter.
Basal nuclei (striatum)
A marked to intense PrPd accumulation was observed in the putamen and globus pallidus, whereas its intensity was less in the caudate nucleus. Stellate, perineuronal, and linear types of PrPd accumulation predominated in these nuclei (Figs. 2a and Si3).
Piriform cortex
A moderate to marked fine punctate type of PrPd accumulation, together with traces of glial and stellate types, was identified in the plexiform layer (mostly in the Ib layer) of the piriform cortex (Figs. 3a and Si2). This type of PrPd deposit was noticed from section III to section VI of the entorhinal cortex (schematic brain sections). Neurons in layer II of the piriform cortex did not present any trace of intracytoplasmic PrPd accumulation (Fig. 3a, layer II).
Hippocampus
A minimal fine punctate/linear type of PrPd accumulation was restricted to the outer molecular layer of the CA neurons (Fig. 4a).
Hippocampal commissure
A moderate fine punctate labeling was identified in this brain area together with other types (Fig. Si6).
Mesencephalon
A marked to intense and conspicuous linear type of PrPd accumulation was observed at the anterior and posterior mesencephalon in the substantia nigra in BSE-affected sheep (Figs. 5a and Si7). In the red nucleus area, moderate intraneuronal PrPd accumulation was observed (Fig. 5a, inset).
Cerebellum
PrPd staining was relatively faint when compared to some scrapie cases. A fine punctate type of PrPd accumulation (diffuse fine punctate labeling) was observed between the granular and molecular layers (Fig. 6a ) that was confirmed by the PET-blot analysis, which showed a strip of PrPd deposits at the Purkinje cell layer (Fig. Si9). There was no trace of intracytoplasmic PrPd deposits in the Purkinje neurons. The stellate type was also present in the area of the cerebellum, whereas coarse particulate and coalescing types were not observed.
Neuroanatomical Distribution and Deposition Types of PrPd in Scrapie-Infected Sheep: Comparison With Ovine BSE
In sheep affected with scrapie carrying the ARQ/ARQ genotype, as in experimental ovine BSE, the distribution of PrPd in the brain was recorded from the frontal cortex to the medulla oblongata (Fig. 7b). Despite interindividual variability in the intensity of deposits in scrapie-infected sheep, PrPd accumulation was observed most intensively in the frontal cortex, basal nuclei, thalamus, hypothalamus, mesencephalon, medulla, and cerebellum. PrPd labeling was less intense in ARQ/VRQ sheep compared to ARQ/ARQ sheep infected with scrapie, notably in the frontal cortex, basal nuclei, and mesencephalon (Figs. 7b and 7c), except in the 2 experimentally iv-infected sheep (Fig. 7a). PrPd labeling was systematically observed in 19/19 scrapie-infected sheep in the hypothalamus (moderate to marked), medulla oblongata (slight to intense), and mesencephalon (slight to marked).
When compared to scrapie cases with the same or different genotypes, the PrPd profile obtained in BSE-infected sheep (Fig. 7a-c) revealed noteworthy differences; for example, the absence of intraneuronal PrPd deposits together with moderate to marked fine punctate PrPd deposits in layer Ib of the piriform cortex in ovine BSE (Fig. 3a) were not observed in scrapie cases (Fig. 3b-d, Fig. 7a-c, sites C and D). Similarly, the amount of intraneuronal PrPd deposits in the red nucleus area (Fig. 7a-c, site K) and the linear type in the substantia nigra were also higher in ovine BSE when compared to scrapie (Fig. 7a-c, site J).
All the different types of PrPd deposits that were observed in BSE-infected sheep were also identified in scrapie cases. The vascular amyloid type was noticed in only 3/19 ARQ/VRQ scrapie-infected sheep in the striatum, thalamus/hypothalamus, and/or parietal cortex (Fig. 1d). A dense fine punctate accumulation was observed in 7/19 scrapie cases in the Islands of Calleja (minimal to intense labeling; Fig. 2d), but was absent in ovine BSE cases.
The different types and intensities of PrPd deposits in the cortex, striatum, piriform cortex, hippocampus, substantia nigra, and cerebellum in scrapie-affected sheep illustrated in Figs. 1b-d and 6b-d show the variability of PrPd deposits among these sites.
Discussion
In sheep, strain characterization by analyzing the variations in the distribution of histopathological lesions in natural scrapie is well recognized. 34 However, it seems that lesion profiles in sheep as a means of identifying scrapie strains may pose great difficulties and are influenced by several factors, especially when dealing with natural disease. 1 More recently, analysis of the disease-associated prion protein PrPd, for example, the extent of glycosylation,10,16 protease sensitivity in vitro and in situ,20,13,18,28,31 and the topographical distribution of PrPd 6,21 has facilitated prion strain identification.
In our study, in spite of a difference of 2 years in the incubation period between the 2 experimentally BSE-infected sheep, the distribution and nature of PrPd deposits were remarkably similar between the 2 animals. The detailed PrPd brain mapping results indicate that the examination of basal nuclei, piriform cortex, frontal cortex, hippocampal commissure, hippocampus, substantia nigra, red nucleus, and cerebellum (brain sections III, VI, and VIII) are the most interesting combination of neuroanatomical sites to take into account in order to discriminate BSE from scrapie agents among the TSE-affected sheep we studied. Similarly, a consistent PrPd profile, reported by another group and consistent with our ovine BSE cases, was described in sheep carrying ARQ and/or VRQ alleles, experimentally infected with BSE with different breeds, incubation periods, and inoculation routes. 15 Indeed, in that study, the topographical description of the PrPd deposits in the brain was characterized mainly by stellate and subpial types in the grey matter of the cortex; perineuronal, linear, and stellate types in the basal nuclei; intraneuronal accumulation in the red nucleus area; linear type in the substantia nigra in the midbrain, and extracellular deposits at the Purkinje cell layer in the cerebellum. 15 In our study, we identified additional neuroanatomical region candidates for discrimination of BSE and scrapie on the basis of types of PrPd deposits: the hippocampal commissure, hippocampus, and particularly the piriform cortex, in which a dense fine punctate PrPd accumulation occurred in the plexiform layer extending from the olfactory tubercle (section III) to the entorhinal cortex (section VI). The specific combination of particular PrPd deposits in these brain areas in sheep with BSE has not been seen in any of the scrapie cases, even though individual similarities with scrapie cases can exist. However, these observations need further validation using larger numbers of BSE-exposed animals, particularly those dosed by the oral route.
In BSE-infected sheep, we observed the most widely reported deposition types (intraneuronal, fine punctate, linear, coarse particulate, coalescing, stellate, intraglial, subpial, and perineuronal) as described in another study in sheep with ARQ and/or VRQ allele genotypes. 15 In sheep with scrapie, we also identified all the PrPd types usually described,14,26 whereas the amyloid vascular plaques were observed in only a few scrapie cases but not in BSE-infected sheep, as has been mentioned by others.14,15,26
In sheep affected with scrapie, it seems that the pattern/types of PrPd deposits in the obex are strongly associated with the PrP genotype, 30 but its relationship with regard to the identification of TSE strains/isolates is not clear. In our study, the types and pattern of PrPd deposits also seem to be more consistent within animals of the same genotype (Figs. 7(a-c)). However, we found a considerable degree of variation in the PrPd deposition pattern, even in scrapie-infected sheep of the same genotype. This finding may reflect scrapie strain differences or most likely, only variations in the degree of PrPd accumulation. Indeed, in natural scrapie cases it is impossible to assess the incubation times and duration of clinical disease that may extend for some months, 17 both of which likely have an impact on its phenotypic presentation. 29 In contrast, in the 2 iv-infected sheep in our study, the PrPd brain pattern was highly consistent between the 2 animals, which might be owing to better control of the progression of the disease over time. Finally, concerning the naturally infected sheep with scrapie, we clearly cannot determine the significance of these variations in the PrPd pattern. Mouse transmission studies of these cases will offer interesting complementary data with regard to the possible identification of different strains.
To conclude, although we analyzed only 2 animals experimentally infected via different inoculation routes, our results support the consistency of the PrPd brain profile in sheep clinically affected with BSE when compared with those profiles reported in the literature. 15 There is a weakness with our approach, which is the requirement of entire sheep brains for analysis, which may be difficult to obtain in surveillance programs for TSEs. Furthermore, the greatest limitations are likely to be related to the need to analyze clinically affected sheep with scrapie. Yet it can be difficult to clearly identify signs of TSE; the duration of the disease that may extend to some months 17 is not known precisely, and we do not know the evolution and degree of PrPd accumulation in the brain during the clinical phase. 29 Thus, our work should be complemented by analyzing more TSE-affected sheep, but it seems that this approach likely offers another complementary approach for strain identification. With the description of atypical forms of scrapie in sheep 3 and in cattle4,9 (displaying a phenotype different from classical scrapie when transmitted to sheep 25 ), the complexity of strain identification in ruminants argues for the development and validation of tools for accurate TSE strain identification.
Footnotes
Acknowledgements
We gratefully acknowledge the VLA TSE Archive, Weybridge for providing brains of sheep that were clinically affected with scrapie, and K. Zellweger, NeuroCenter, Berne for conducting the iv experimental scrapie infection study. We are also grateful for the excellent histotechnical assistance of Céline Raynaud and Mikaël Leboidre (Anses, Lyon). We wish to thank Jackie Godfrey, a professional translator and English native speaker, who carefully read and reviewed the present article.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sector.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
