Abstract
This study aimed to clarify the timing and infectivity of equine herpesvirus 9 (EHV-9) infection in BALB/c-nu/nu mice and their immunocompetent counterpart (BALB/c). Following intranasal inoculation with 105 PFU of EHV-9, specimens from 8 mice per group were collected at different times postinoculation (PI) and assessed using histopathology, immunohistochemistry for viral antigen, and quantitative real-time polymerase chain reaction for ORF30 gene expression. In BALB/c-nu/nu mice, EHV-9 antigen was abundant in olfactory epithelia of all inoculated animals, and in the olfactory bulb of 1 animal. In contrast, only 1 BALB/c mouse per time point had rhinitis, with mild to moderate immunopositivity starting from 12 to 48 h PI, followed by a gradual virus clearance at 72 h PI. Statistically, significant differences were noted in the immunohistochemistry reactions between the 2 mouse strains, indicating that BALB/c-nu/nu is more susceptible to infection. Relative expression levels of ORF30 gene in olfactory epithelia were significantly different between the 2 groups, with the exception of 12 h PI, when BALB/c-nu/nu animals showed dramatic increases in ORF30 gene expression level until 48 h PI, followed by a decline in expression level until the end of experiment. In contrast, the expression level in brains showed no differences between mouse strain except at 96 h PI. In both strains, the highest messenger RNA expression was detected at 48 h PI, followed by a decline in BALB/c mice, proving a rapid clearance of virus in BALB/c and a gradual slowing down of the increased expression levels in BALB/c-nu/nu.
Keywords
In previous studies, we reported that equine herpesvirus 9 (EHV-9) could induce encephalitis in hamsters using different routes of inoculation (oral, peritoneal, and ocular), but the intravenous route failed to induce encephalitis. In these experiments, the virus was predominantly transmitted through neurogenic pathways, including the olfactory and trigeminal nerves following nasal inoculation, 12 the trigeminal and vagal nerves following oral inoculation, 14 the spinal cord and abdominal nerves following intraperitoneal inoculation, 15 and the optic nerve after ocular inoculation. 13 None of these studies examined the infectivity of EHV-9 in immunocompromised nude mice and their immunocompetent counterparts. The pathology of other viruses experimentally studied in BALB/c and the BALB/c-nu/nu mice, such as equid herpesvirus type 1, 29 herpes simplex virus type 1, 19 hepatitis E virus, 22 Sindbis virus, 21,35 murine leukemia virus, 20 HSV-1 ICP34.5 mutant, 27 murine K papovavirus, 18 and Theiler’s virus, 41 has been described in the literature.
Congenitally athymic nude mice, which are immunologically characterized by the absence of functionally mature T lymphocytes, exhibit marked differences in susceptibility to infection, in comparison with immunocompetent BALB/c mice. This is characterized, as expected, by an increase in the severity of histologic lesions, as in Q fever, 23 sporotrichosis, 9 coccidiosis, 2 and Cryptococcus neoformans infection, 6 or by an unexpected decrease in severity, as in mucormycosis, 7 systemic candidiasis, 8 and listerial infections. 30
The purpose of this study was to compare the infectivity of EHV-9 in BALB/c-nu/nu and BALB/c mice and to investigate whether adult BALB/c-nu/nu mice inoculated intranasally with EHV-9 develop clinical signs of encephalitis or have an anomalous decrease in susceptibility following infection, as previously reported in other pathogens. 29,37
Materials and Methods
Virus Culture
Madin-Darby bovine kidney (MDBK) cells were used for the propagation of EHV-9. The inocula were prepared by culturing the virus from the original seed stocks of EHV-9 (P19, fifth passage in MDBK cells). The virus was titrated by a plaque-forming assay on MDBK cells.
Animals
Forty-eight 4-week-old male BALB/c-nu/nu (BALB/cSlc-nu/nu; SLC, Hamamatsu, Japan) mice were divided into 6 groups (representing the different time points of sacrifice). Each group had 8 mice: 6 were inoculated with EHV-9 virus, and 2 were kept as control. A similar pattern of inoculation was performed in the BALB/c mouse cohort. All animals were purchased from a known breeder (SLC, Hamamatsu, Japan). The animals were housed in plastic cages and kept in an isolated biohazard cabinet for approximately 14 days of acclimatization. They were provided with a basal pellet diet (Oriental MF; Oriental Yeast Co., Tokyo, Japan) and bottled water ad libitum. The experiment was conducted in accordance with laws and standard operating procedures related to the use of laboratory animals. The experimental protocol was approved by the Animal Experiment Committee of the Faculty of Applied Biological Science at Gifu University, Japan.
Virus Inoculation
Each experimental animal of both mouse strains was intranasally injected with a single dose of 105 plaque-forming units (PFU) of EHV-9 virus. Mice were sacrificed at 12, 24, 36, 48, 72, and 96 h PI. The animals were assessed at least 3 times daily for clinical signs previously reported in EHV-9 infection. The signs included weight loss, crouching posture, nasal and lacrimal discharges, and salivation. By the second day, animals became hyperreactive to stimuli and displayed occasional tremors associated with uncoordinated movements of the limbs.
Pathological Investigations
At necropsy, the heads (including olfactory epithelia and brain tissues) were bisected on the sagittal plane. Sections of the spinal cord, lungs, liver, spleen, and small and large intestines were fixed in 10% neutral buffered formalin. Samples were routinely processed and embedded in paraffin wax. Sections (5 μm) were stained with hematoxylin and eosin (HE).
Immunohistochemistry
Fixed tissue sections were also analyzed via immunohistochemistry (IHC) by the avidin-biotin-complex (ABC) method as previously described. 38 The primary antibody was a rabbit antibody specific for EHV-9 (1 in 800 dilution; Veterinary Microbiology Laboratory, Gifu University, Japan), and the secondary antibody was biotinylated anti–rabbit immunoglobulin (Ig) G (DAKO Cytomation, Fort Collins, CO) with visualization using liquid 3,3′-diaminobenzidine substrate chromogen system (DAKO Cytomation) and hematoxylin counterstain. Tissue sections from the EHV-9–infected hamsters and sera from nonimmunized rabbits and goats were used as controls.
RNA Isolation and Quantitative Real-Time PCR
Total RNA from formalin-fixed, paraffin-embedded (FFPE) tissue blocks of the olfactory epithelia and brain was isolated from both mouse strains using the RNeasy FFPE Kit (QIAGEN,Valencia, CA) according to the manufacturer’s instructions. Subsequently, 1.5 μg of isolated RNA was combined with reverse transcriptase master mix consisting of 2 μl of 10× RT buffer (TOYOBO, Osaka, Japan), 5 mM dNTP (TAKARA Bio, Otsu, Japan), 25 pmol random primer (TOYOBO), 40 U RNase inhibitor (TOYOBO), and 50 U reverse transcriptase (TOYOBO). The reaction mixture was incubated at 30°C for 10 minutes and subsequently at 42°C for 40 minutes, followed by incubation at 99°C for 5 minutes to stop the reaction. Real-time PCR assays were carried out using 12.5 μl SYBR Premix EX Taq (TAKARA Bio), 10 μl specific primers, and 10 ng complementary DNA in a thermal Cycler Dice Real Time System (TAKARA Bio). An ORF30 gene primer set (ORF30A, 5′-GTC AGG CCC ACA AAC TTG AT-3′; ORF30B, 5′-ACT CGG TTT ACG GAT TCA CG-3′) was used. PCR conditions were 37°C for 15 minutes and 98°C for 1 minute. Relative quantities were measured by the ▵▵Ct method (crossing-point method). Amplification products were checked by 3% agarose gel electrophoresis and stained with ethidium bromide. The value of each sample was normalized by β-actin as an internal control gene. 39
Statistical Analysis
A t test (2 samples assuming equal variances) was used to compare values of body weight in both BALB/c and BALB/c-nu/nu mice per h PI. Assumptions of normality of the IHC findings were tested using the Anderson-Darling test. Because of the highly non-normal distribution of IHC data and findings of the expression-level values of the ORF30 gene, we performed a nonparametric Mann-Whitney test to compare the IHC mean value of the BALB/c-nu/nu group with that of the BALB/c group per hour after scarification. The Kruskal-Wallis test was used for statistical analyses of all possible pairwise comparisons (controlled at a family error rate of 0.2) revealed from the IHC findings and quantitative PCR (qPCR) results in each mouse relative to time (hours). Analytical programs were conducted using Minitab statistical software (MTW13; Minitab, State College, PA) and Microsoft Excel (Microsoft, Redmond, WA). 31 P < .05 was considered statistically significant.
Results
Clinical Signs and Body Weight
None of the inoculated animals in either group exhibited any significant clinical signs. Body weights are summarized in Fig. 1. No significant differences could be detected except at 72 and 96 h PI (P < .05) between the 2 groups.
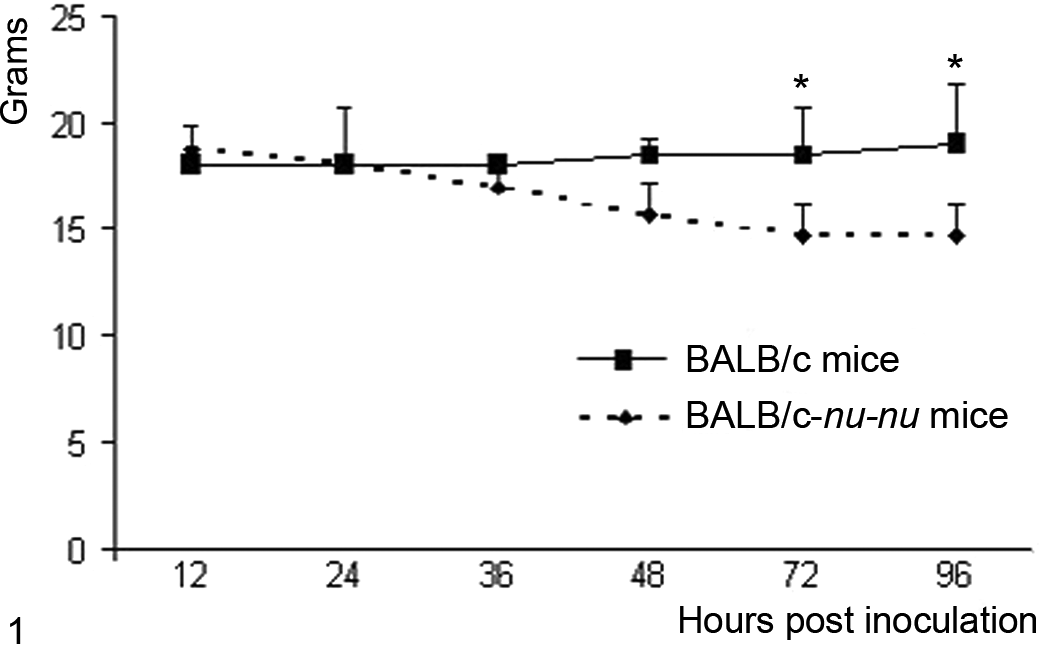
Mean body weights of BALB/c and BALB/c-nu/nu mice intranasally inoculated with equine herpesvirus 9 virus. Error bars indicate standard deviations. *t test, P < .05.
Pathological Findings
At necropsy, no gross abnormalities were observed in the organs or tissues of either mouse strain. Histopathological examination of tissues from the BALB/c mice noted 1 animal per time point in which a mild rhinitis was observed in the olfactory epithelium at 12 h PI, consisting of a focal area of necrosis of the olfactory epithelium and infiltration of a mixed population of inflammatory cells, dominated by neutrophils and lymphocytes, within the mucosa and admixed with desquamated epithelial cells extending between 24 and 36 h PI. Rhinitis was noted to be moderately increased in all inoculated animals at 48 h PI. However, the olfactory epithelium was within normal limits in all inoculated BALB/c animals at 96 h PI (Fig. 2a). In BALB/c-nu/nu mice, however, the nasal cavities of all infected animals exhibited necrotizing rhinitis that increased in severity over time up to 96 h PI. Lesions consisted of multiple necrotic foci within the olfactory epithelium with epithelial intranuclear inclusion bodies and mixed inflammatory cell infiltrates (Fig. 2b).
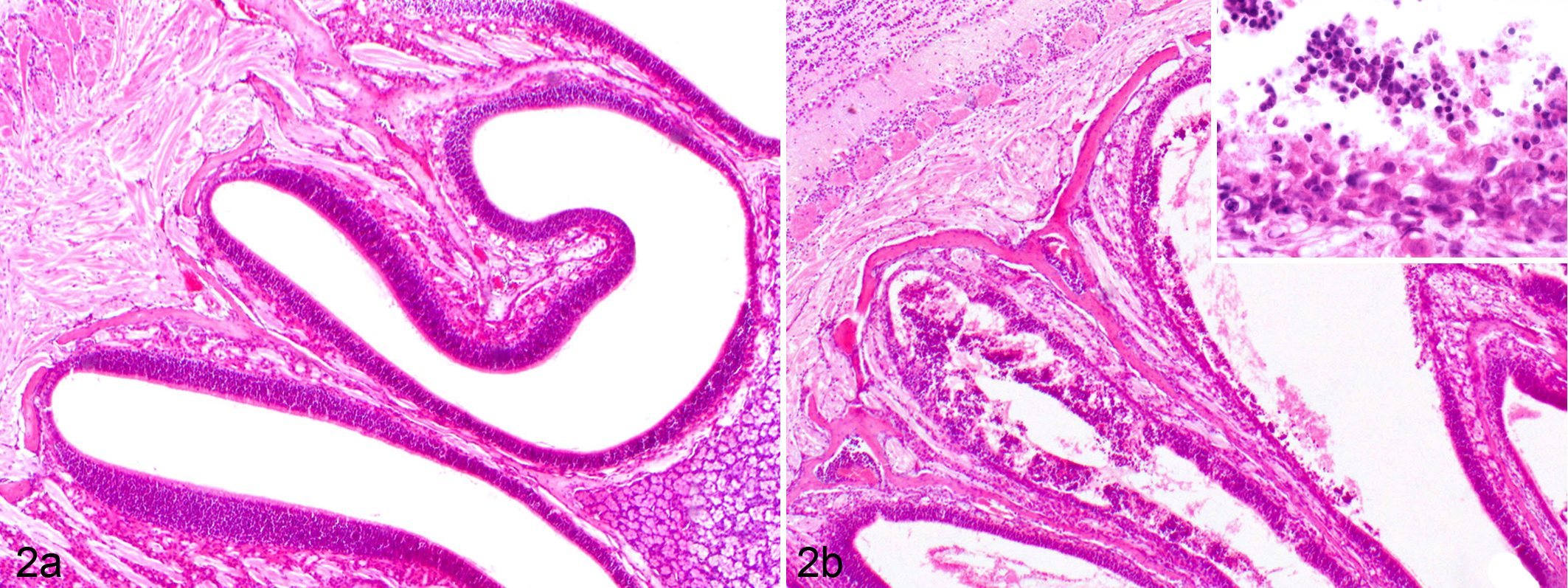
(a) Olfactory epithelium; BALB/c mouse infected with equine herpesvirus 9 (EHV-9) via the nasal route 96 hours postinoculation (h PI). Normal olfactory epithelium. Hematoxylin and eosin (HE) stain. (b) Olfactory epithelium; BALB/c-nu/nu mouse, infected intranasally with EHV-9. Necrosis, erosion in the mucosa, and presence of inflammatory cells in the lumen. (Inset) Higher magnification shows mucosal erosion and inflammatory cells. HE stain.
Mild interstitial pneumonia was observed in both mouse strains from 24 to 96 h PI. Pulmonary changes consisted mainly of diffusely thickened alveolar septa with variable numbers of macrophages and neutrophils. No microscopic abnormalities were noted in other organ systems, including the central nervous system, in either BALB/c or BALB/c-nu/nu mice.
Immunohistochemistry
The distribution patterns of EHV-9 antigen in the olfactory epithelia, olfactory nerves, and brains of all mice, as well as the number of infected animals, are summarized in Table 1.
Immunohistochemical Detection of EHV-9 Antigen in Different Infected Tissues of Intranasally Inoculated Animals in BALB/c and BALB/c-nu/nu Mice.

EHV-9, equine herpesvirus 9; h PI, hours postinoculation; +, presence of viral antigen; –, absence of viral antigen. Numbers in parentheses represent number of infected animals/total number of animals.
Characterization of immunohistochemical reactions in both mouse groups revealed a strongly significant difference (P < .001) in EHV-9 immunoreactivity between the 2 strains at 12 to 36 h PI (Figs. 3c–5c). In BALB/c mice, only 1 animal per time point demonstrated positive immunoreactivity to the EHV-9 antigen, whereas in BALB/c-nu/nu mice, EHV-9 immunoreactivity was detected in all inoculated animals during this period. In BALB/c mice, the positive immunohistochemical staining consisted of focal superficial areas of viral antigen within the olfactory epithelium at 12 h PI (Fig. 3a) and 24 h PI (Fig. 4a), with mild immunolabeling at 36 h PI (Fig. 5a). In nude mice, the amount of viral antigen increased over time to reach a peak at 36 h PI (Figs. 3b–5b). In contrast, at 48 h PI, the intensity of EHV-9 immunoreactivity increased in the BALB/c mice (Fig. 6a) but was relatively decreased in BALB/c-nu/nu mice (Fig. 6b). No significant difference in the quantitative analysis of immunoreactivity could be detected between the 2 strains (Fig. 6c; P = .1074). At 72 h PI, the immunohistochemical reactivity could be seen not only in the olfactory epithelium but also moderately within the olfactory nerve bundles in all inoculated BALB/c-nu/nu mice (Fig. 7b). However, BALB/c mice showed decreased level of viral antigen within the olfactory epithelium (Fig. 7a). There was a significant difference in the immunoreaction intensity between the 2 groups (Fig. 7c; P = .0018). Finally, at 96 h PI, BALB/c mice exhibited a complete clearance of viral antigen from the olfactory epithelium (Fig. 8a). On the other hand, in BALB/c-nu/nu mice, viral antigen extended more deeply into the olfactory nerve in all inoculated animals, and EHV-9 antigen was observed in the olfactory bulb in 1 animal (Fig. 8b). Statistically, there was a difference in the degree of immunoreactivity between the 2 groups (Fig. 8c; P = .0171).
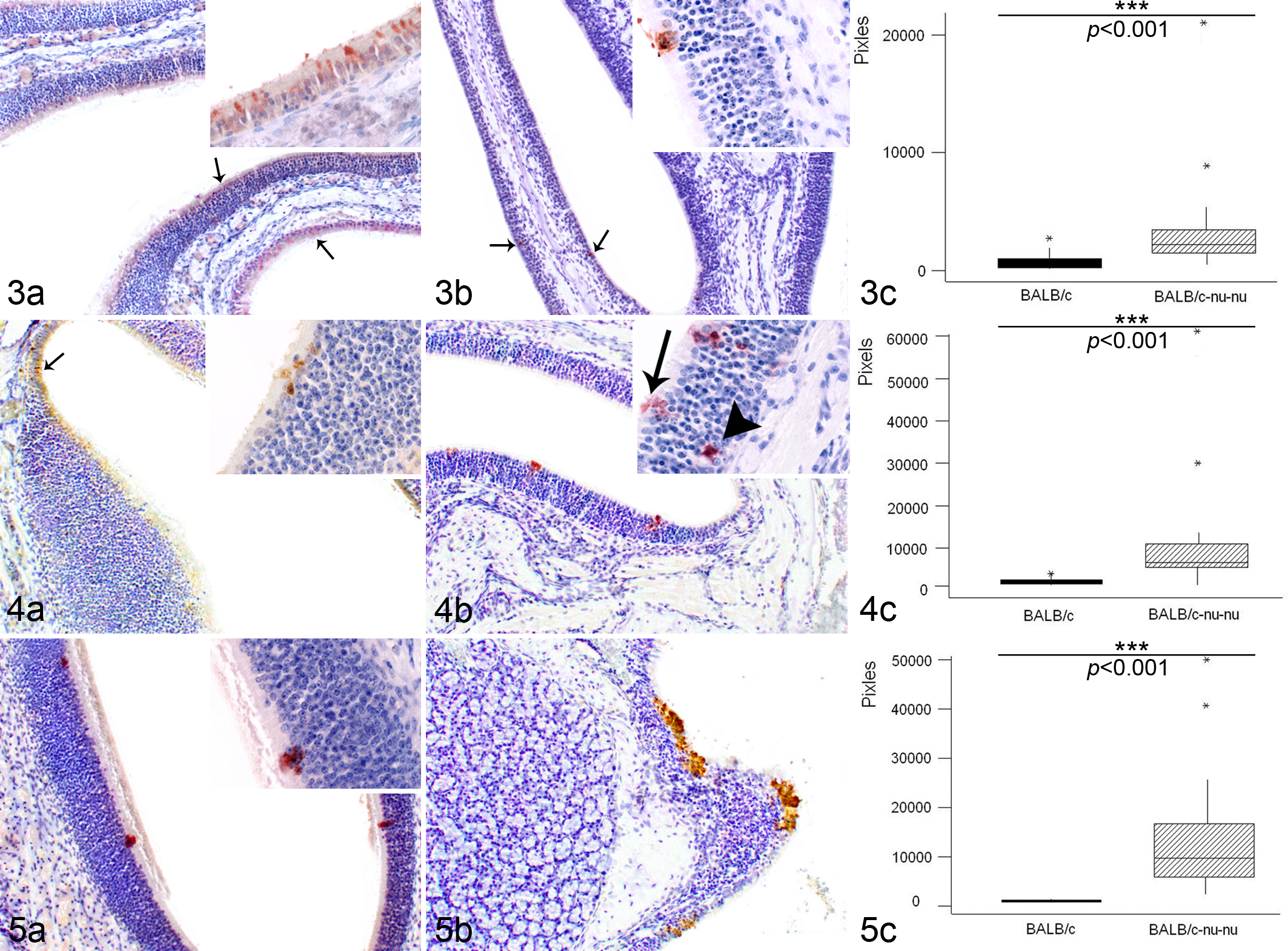
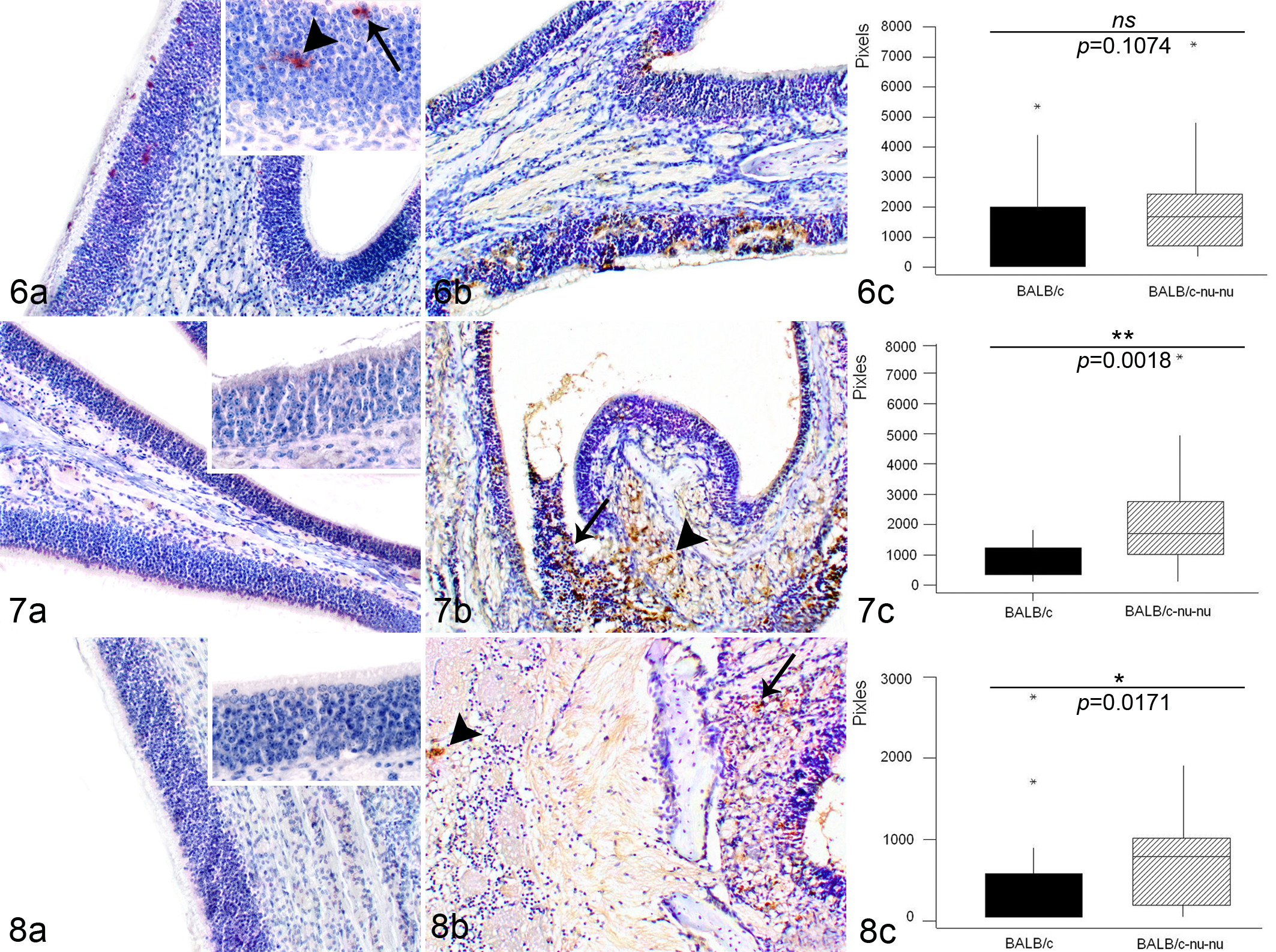
The pixel intensity of the IHC reactions at the different time points after inoculation was measured in each mouse strain (Fig. 9a,b), with the Kruskal-Wallis test revealing a significant difference (P < .001) between the nude and normal mice. The highest intensity within the olfactory epithelium was from 24 to 36 h PI in BALB/c-nu/nu mice and at 48 h PI in normal mice.
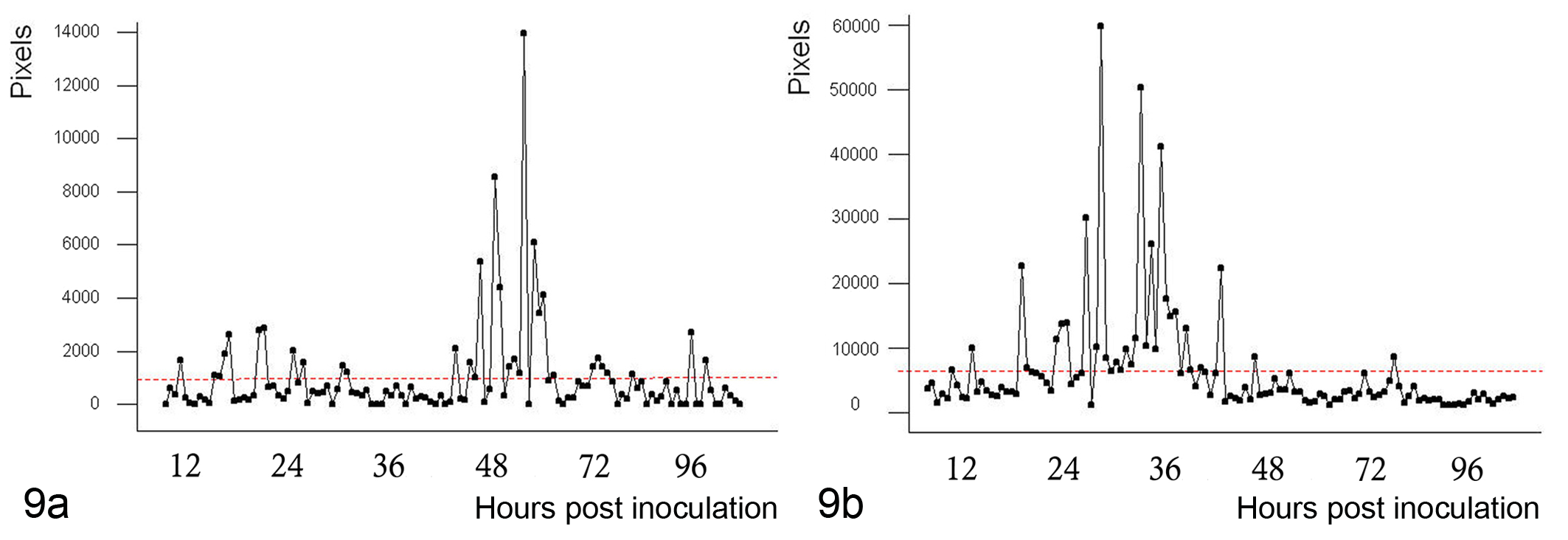
(a) Intensity of immunohistochemistry (IHC) reactions at different hours postinoculation (h PI) in BALB/c mice. Maximum intensity was seen at 36 to 48 h PI (Kruskal-Wallis test, P < .001). (b) Intensity of IHC reactions h PI in BALB/c-nu/nu mice. Maximum intensity was seen at 24 to 36 h PI (Kruskal-Wallis test, P < .001).
Quantitative Real-Time PCR
Relative expression levels of the ORF30 gene in both the olfactory epithelia and the brains of BALB/c and BALB/c-nu/nu mice, using quantitative RT-PCR, are summarized in Figure 10.
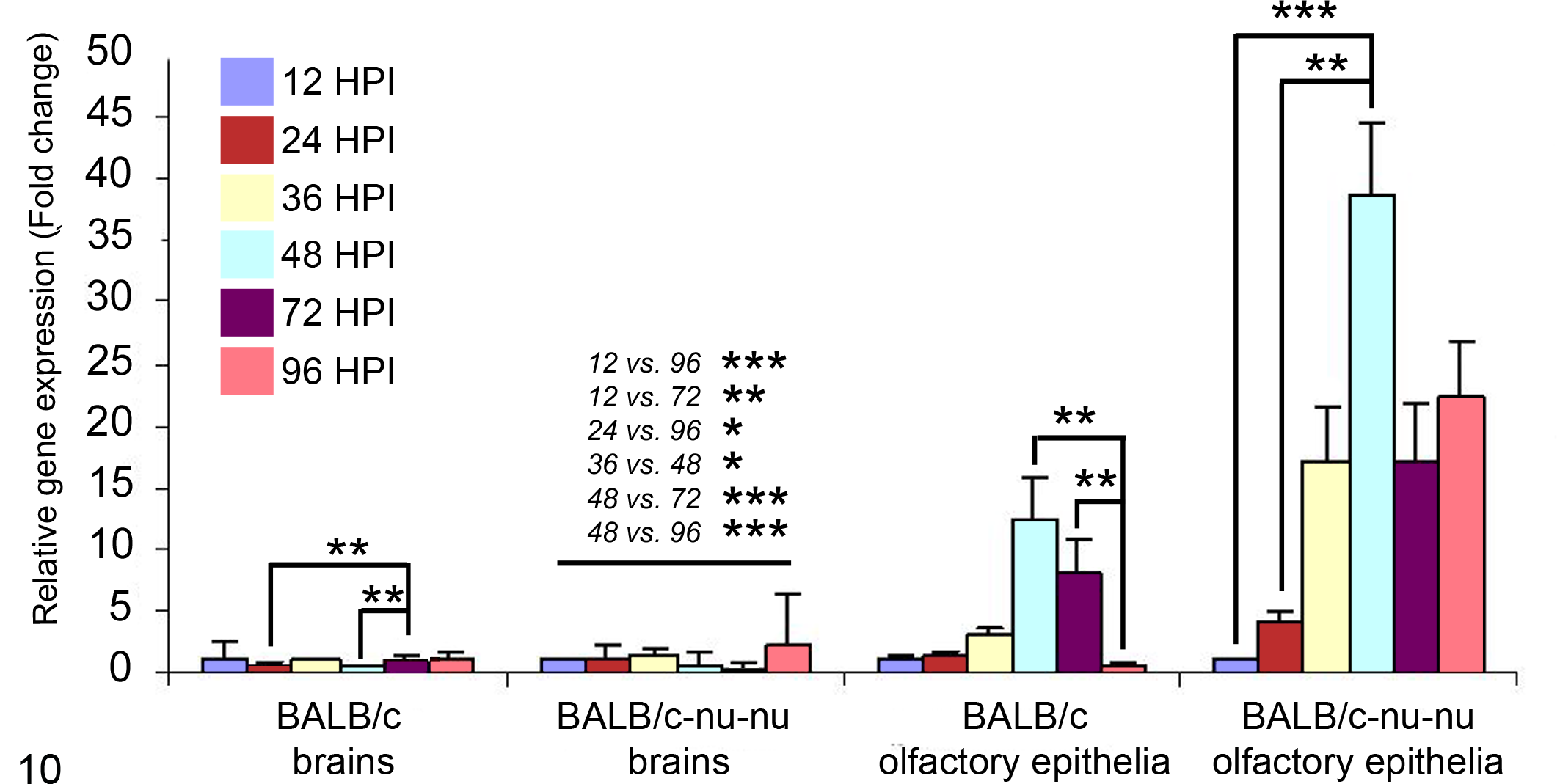
Analysis of relative transcription activity of the viral gene ORF30 in olfactory epithelia and brains of BALB/c and BALB/c-nu/nu mice by real-time reverse transcription polymerase chain reaction. The figure compares the transcription levels with the crossing-point method using β-actin gene control. (*P < .05, **P < .01, ***P < .001, Kruskal-Wallis test).
Analysis of possible pairwise comparisons of values for gene expression during different h PI (using the Kruskal-Wallis test) showed the following: Significant differences in messenger RNA (mRNA) expression levels in the olfactory epithelia of BALB/c mice between 48 and 96 h PI (P = .0017) and between 72 and 96 h PI (P = .0058). In this strain, the relative ORF30 gene expression level was positively increased from 12 to 48 h PI (12-fold compared with 12 h PI), followed by a dramatic sharp decrease to the lowest level at 96 h PI (less than 1-fold compared with 12 h PI), providing clear evidence of viral clearance in BALB/c mice. Significant differences in mRNA expression levels in brains of BALB/c mice between 24 and 72 h PI (P = .0028) and between 48 and 72 h PI (P = .0028). Finally, the transcription activity of the ORF30 gene 96 h PI indicated EHV-9 clearance from the brain. In BALB/c-nu/nu olfactory epithelia, there was a proportional increase of mRNA expression levels until 48 h PI (40-fold difference in comparison with 12 h PI), followed by a gradual slowing to reach 20-fold at 96 h PI (compared with 12 h PI). Significant differences were observed between 12 and 48 h PI (P = .0006) and between 24 and 48 h PI (P = .0058). In brains of BALB/c-nu/nu mice, there were many differences in the relative qualitative expression of the ORF30 gene at different times. These were 12 vs 96 (P = .0004), 12 vs 72 (P = .0015), 24 vs 96 (P = .0116), 36 vs 48 (P = .0116), 48 vs 72 (P < .0001), and 48 vs 96 h PI (P < .0001).
Comparisons of the relative quantity of ORF30 gene expression (crossing-point method) at each time point between BALB/c and BALB/c-nu/nu mice strains were carried out using the t test. There were no statistically significant differences in relative gene expression values of ORF30 in the brain tissues at any time point except at 96 h PI. In the latter, expression of the ORF30 gene significantly increased in nude mice (P = .002475). On the other hand, gene expression was significantly increased in olfactory epithelia of BALB/c-nu/nu mice compared with BALB/c mice at 24, 36, 48, 72, and 96 h PI (P = .026665, .008277, .0309, .026782, and .007453, respectively).
Discussion
In the current investigation, congenitally athymic (BALB/c-nu/nu) mice were more susceptible to the intranasal inoculation of EHV-9 than phenotypically normal mice (BALB/c), which were relatively resistant to acute infection with EHV-9. This finding was confirmed by histopathological and quantitative immunohistochemical analyses as well as qPCR findings at different times after infection. The findings are consistent at times ranging from 24 to 96 h PI, with the exception of the 48-h PI time point. That specific time point represents the peak of virus propagation in both experimental groups. The nonsignificant differences at this point might be attributed to the increase in virus propagation in the olfactory epithelia of BALB/c mice and to the slight decline of the virus antigen level within the olfactory epithelia in nude mice. Furthermore, using qPCR as a sensitive method for measuring the expression level of the ORF30 gene within olfactory epithelia, it was revealed that at 48 h PI, there was a significant difference between the 2 strains. The expression level in BALB/c-nu/nu mice was twice that noted in BALB/c mice, and at 96 h PI, it was 20-fold that of BALB/c mice.
Predominant striking features following intranasal inoculation of EHV-9 in normal BALB/c were the scarce histologic lesions comprising minimal mononuclear cell inflammatory reactions in the olfactory epithelium and a complete absence of lesions in the brain. In parallel, IHC examination revealed a negative immunoreactivity to viral antigen in the brain at all time points PI. In addition, a low expression level of the mRNA ORF30 gene was observed throughout the experiment. These results were quite similar to those obtained by LeBlanc et al, 28 who studied the infection of normal mice with a virulent Venezuelan encephalitis virus.
The present study demonstrated a weak immunohistochemical reaction to EHV-9 antigen within olfactory epithelia and a sharp decrease in mRNA levels at 96 h PI (less than that found at 12 h PI in the same mice strain), providing evidence of viral clearance from the olfactory epithelia in BALB/c mice. This clearance was lacking in the BALB/c-nu/nu cohort.
Specifically sensitized T cells may act directly to lyse virally infected target cells. 10,11 Cell-mediated lysis of viral antigen–expressing cells may be beneficial because the sensitized T cells may function solely to eliminate virus-transformed cells (such as in the rejection of allogenic tumors) 17,32 or in the association with other cells, especially macrophages, to limit the growth of cytolytic viruses in different tissues. 4 Moreover, it has been reported that these cells might directly act to lyse virus or infected target cells (as in vaccinia virus) or in the inhibition of viral growth and spread, therefore possibly representing an important antiviral effector mechanism. 5,40 Conversely, the absence of these cells may lead to an extensive virus-induced cytopathology. 3 Other investigations reported that the acute intranasal infection of BALB/c mice with murine gammaherpesvirus produced a robust chemokine response. This plays an important role in the host defense by recruiting leukocytes to sites of inflammation and thereby assisting in the control or clearance of the infection from the lungs of infected animals. 36
It has been established that the most efficacious immune response against a given viral pathogen is highly dependent on the life cycle of the virus and the interaction of the virus with the target host cells. For equine herpesviruses, both antibody- and cell-mediated immune responses are likely to be important. 24 Humorall immunity is represented by production of both neutralizing and complement-fixing antibodies starting approximately 2 weeks after field or experimental infection with EHV-1, 1 whereas the cell-mediated immune (CMI) response is considered a more important defense against herpesvirus dissemination than humoral immunity. This occurs since CD8+ cytotoxic lymphocytes (CTLs) form an essential part of the immune defense against herpesviruses, including equine herpesvirus 1 (EHV-1). 27 Recently, the number of EHV-1–specific CTL precursors (CTLp) has been identified as the most accurate and measurable correlate of immunity to EHV-1 infection. A low frequency of CTLp is accompanied by an increased severity of EHV-1 infection and vice versa. A high frequency of CTLp reduces the duration and level of viremia and, in turn, the pathology associated with the period after EHV-1 infection. 26 Smith et al 33 suggested that lymphocyte subpopulations, including CD4+ and CD8+ T cells, contribute to the viral clearance during EHV-1 infection. However, other studies on EHV-1 in hamsters demonstrated that an antiviral humoral immune response might play a role in viral clearance, following both intranasal and intraperitoneal inoculation of EHV-1 virus. 34
Congenitally athymic nude mice are known to have deficient CMI and impaired T-lymphocyte production, explaining the high susceptibility to EHV-9 infection and their inability to clear the virus from their tissues. This phenomenon was observed, in the present study, by IHC detection of the virus at late time points after inoculation (72 and 96 h PI) in the olfactory epithelium and olfactory nerve, with extension of the virus to the olfactory bulb at 96 h PI. The high expression level of the ORF30 gene indicated the persistence of the virus until the end of the experiment. It is noteworthy that the propagation of the virus within the olfactory nucleus may occur at 168 h PI (data not yet published). From the data presented in the current study and that reported within the literature, 4,11,16,25,33,40 it has been demonstrated that T lymphocytes are required in recovery following nasal inoculation of EHV-9 due to the almost complete clearance of the virus from the olfactory epithelia in BALB/c mice in contrast with the persistence of the virus in nude mice.
In summary, BALB/c mice are less susceptible to acute infection with EHV-9 infection in comparison to BALB/c-nu/nu mice, but further investigation of their apparent resistance to the infection may yield important immunological insights. In contrast, BALB/c-nu/nu mice can be readily infected with EHV-9. The nude mouse may therefore be a useful animal model for studying the acute pathogenesis and immunological aspects of EHV-9. Further studies to evaluate the dynamics of cytokine, immunoglobulin, and cellular responses are needed in both mouse strains to further expand the knowledge base of EHV-9 infection in these mouse strains.
Footnotes
Acknowledgments
This study was supported in part by a grant-in-aid (emerging—general) for scientific research from the Ministry of Health, Labor and Welfare of Japan; a grant from Hokkaido University; and a grant from the Ono Pharmaceutical Co, Ltd. We sincerely thank Ms C. Swift for proofreading the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hokkaido University and Ono Pharmaceutical Co, Ltd.
