Abstract
Papillomavirus infections are responsible for plaques and papillomas in various locations on the skin and in mucous membranes. The aim of this report was to describe morphologic features of a viral pigmented conjunctival plaque and 2 conjunctival squamous papillomas in 3 dogs, and to investigate these lesions for the presence of papillomavirus DNA by polymerase chain reaction (PCR), DNA sequence analysis, and in situ hydridization (ISH). Histopathology revealed in all neoplasms various degrees of epithelial hyperplasia, acanthosis, and hyperkeratosis with koilocytosis. In all lesions E6, E7, and L1 gene fragments of canine oral papillomavirus (COPV) DNA were detected by PCR and sequencing analysis. ISH revealed COPV DNA in a highly specific pattern within nuclei of the hyperplastic epithelium. The presence of canine papillomavirus in ocular conjunctival plaques and papillomas suggests these benign lesions may have the potential for malignant transformation. This is the first time that the lambdapapillomavirus COPV has been detected in ocular epithelial hyperplastic lesions.
In dogs, papillomavirus infection can cause single or multiple epithelial plaques and papillomas in a variety of locations on the skin, including the mucosa and mucocutaneous membranes of the oral cavity, conjunctiva and cornea, lips, penis, and vulva. 1, 2, 6, 12
Canine oral papillomavirus (COPV) is classified as lambdapapillomavirus and associated with papillomas, or warts, in the oral cavity of both domestic dogs and wild canids. 12 After an incubation period of 1 or 2 months, the benign lesions usually do not elicit any serious problems, and even severe oral papillomatosis shows spontaneous immune-mediated regression. However, some cases are refractory to treatment, 11 and, rarely, squamous cell carcinomas of the skin and gingiva have also been associated with canine papillomavirus infections. 2, 18
Histomorphologically, cutaneous papilloma virus–induced lesions have been classified as exophytic papilloma, inverted papilloma, and canine viral pigmented plaque. 5 Upon conjunctiva-only squamous papilloma is described so far.
Recently, a suggestion was made to standardize the nomenclature for cutaneous pigmented nevus or pigmented papule in canine viral pigmented plaque. 5
Apart from COPV, which was originally isolated from papillomas of the oral cavity, lips, and skin, 15, 18 novel canine cutaneous papillomaviruses (CPV3 and CPV4) were isolated from a cutaneous canine viral pigmented plaque. 7, 10, 16, 19, 20 Partial sequencing of the DNA of 1 cutaneous papillomavirus showed about 64% nucleotide sequence similarity to human papilloma virus and less than 57% sequence similarity to the L1 gene of COPV. 16, 19
The major objective of this study was to describe distinct and different morphologic features of conjunctival epithelial hyperplastic lesions in 3 dogs and to investigate the hypothesis that these lesions were associated with COPV by means of polymerase chain reaction (PCR), DNA sequence analysis, and in situ hybridization (ISH).
Dog No. 1, a 2.5-year-old intact male Cavalier King Charles Spaniel, was presented with a pigmented circular elevated plaque on the limbal conjunctiva, 0.4 cm in diameter (Fig. 1A). The dog was otherwise healthy. The lesion was removed surgically, and no recurrence was reported 6 months after removal.
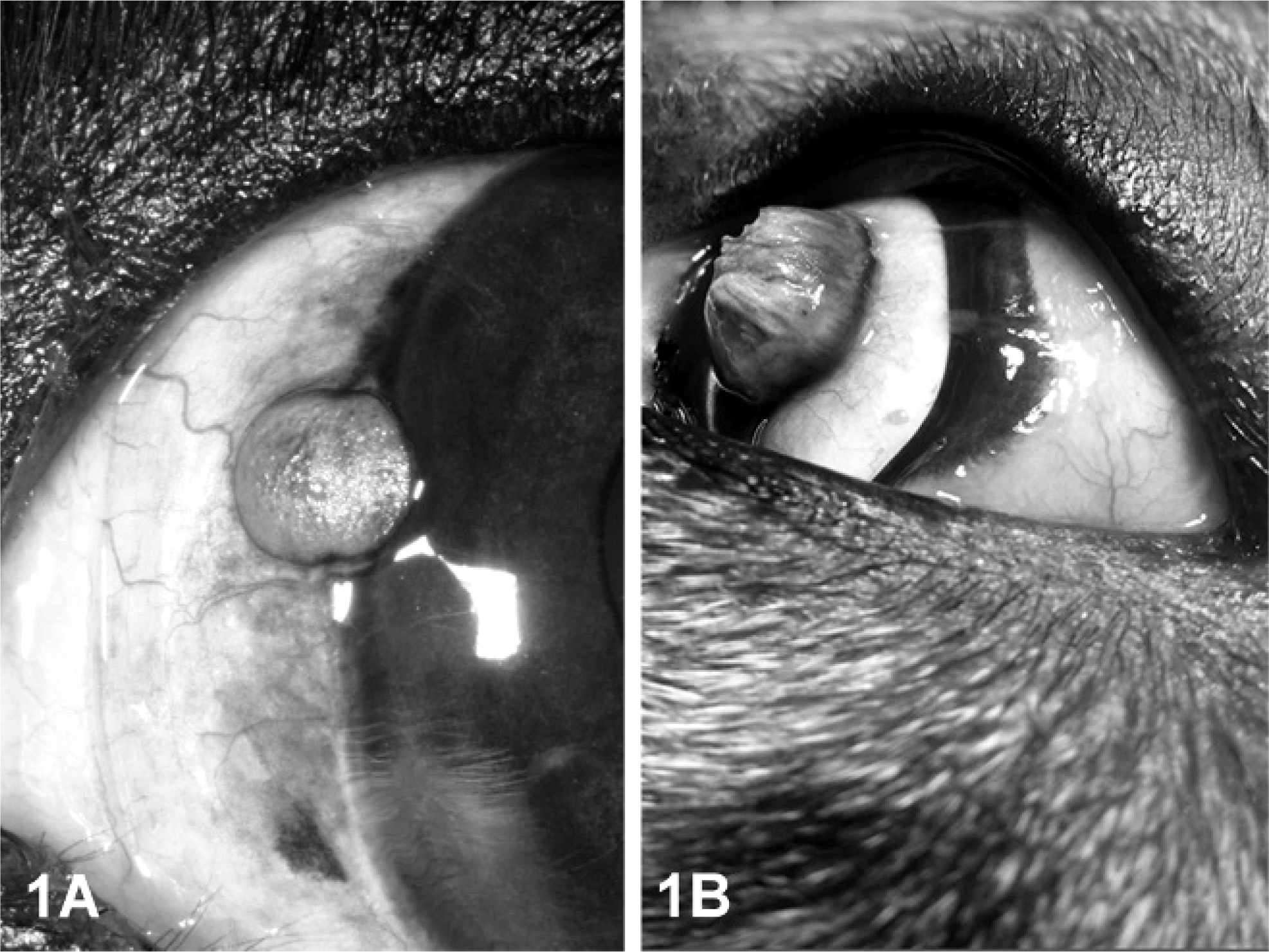
Limbal conjunctiva; dog No. 1. Unilateral, there is a round, sharply demarcated, slightly elevated, pigmented plaque on the corneal limbus.
Dog No. 2, a 2-year-old intact male Parson Russell Terrier mix breed dog showed a raised neoplasm with verrucous fronds on the limbal conjunctiva, 0.5 cm in diameter (Fig. 1B). The neoplasm represented the second relapse, but after the third surgical excision no further recurrence was reported over a 6-month observation period.
Dog No. 3, a 2-year-old female Newfoundland, was presented with a flat round exophytic hyperkeratotic mass on the conjunctiva of the third eyelid, 0.7 cm in diameter. According to the history given by the owner, the neoplasm developed during a 1- to 2-week period and was removed by surgical excision with preservation of the third eyelid. After 2 months, a slow-growing relapse was reported and subsequently completely removed.
The entire conjunctival neoplastic tissue of all 3 dogs was removed, fixed in 4% formaldehyde, and submitted for histopathology. Samples were routinely processed, embedded in paraffin wax, cut at 3 µm, and stained with HE.
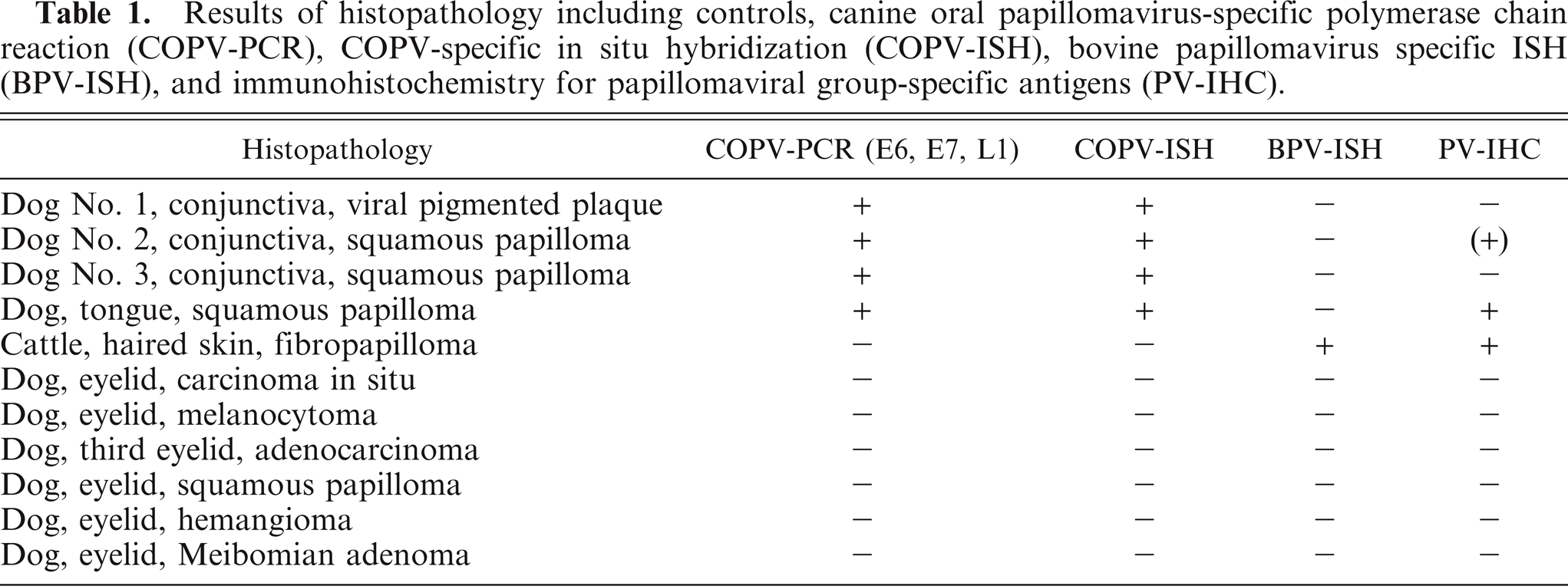
To investigate the paraffin-embedded material for the presence of papillomavirus antigens, immunohistochemistry was applied, and analysis for COPV and bovine papillomavirus (BPV) DNA by PCR and ISH was performed as described previously. 17 Briefly, immunohistochemistry was performed using the avidin–biotin–peroxidase complex method and a commercial rabbit polyclonal antibody against PV group-specific antigens (Dako Diagnostika, Hamburg, Germany). The DNA of the paraffin-embedded tissues was extracted, taking special care to avoid cross-contamination. The dewaxed sections were digested in 0.2 mg/ml proteinase K solution overnight at 55°C. The solution was boiled for 10 minutes and used for DNA amplification by PCR. Six oligonucleotide primers were chosen, complementary to the DNA sequence of COPV (Genbank: D55633): E6+ nucleotides (nt) 113–132 (5′-GGCACTGTTATCAATGGAGC-3′), E6− nt 443–462 (5′-CACATAGTTCTTTGTCCGCC-3′), E7+ nt 559–578 (5′-GGATATTGTGCTGACAGAGC-3′), E7− nt 683–702 (5′-TTCCCACAAAATCCACAGGC-3′), L1+ nt 7131–7150 (CTTGTTTGGGGCTTAAGAGG-3′) and L1− nt 7372–7391 (TGCAGTGTGTACCTGTCCTG-3′). PCR was carried out using Taq DNA polymerase (Promega, Mannheim, Germany) and initial heating (2 minutes/95°C), denaturation (1 minutes/95°C), annealing (1 minute/52.5°C), and primer extension (2 minutes/72°C). As controls for specific amplification, tissue from a COPV-induced canine oral papilloma and various cutaneous and mucocutaneous neoplasms were included, as summarized in Table 1. ISH was performed essentially according to the previously described protocol. 17 Dewaxed sections of the conjunctival lesions were digested for 15 minutes in 10 µg/ml proteinase K at 37°C in a humidity chamber. Hybridization was performed overnight with 500 ng/ml of digoxigenin-labeled COPV probe or BPV probe after simultaneous denaturation of the target DNA and probe. To visualize the hybridization signals, 5-bromo-chloro-3-indolyl phosphate and 4-nitro blue tetrazolium chloride were used according the manufacturer's instructions (Roche Diagnostics GmbH, Mannheim, Germany). Sections of BPV-induced bovine fibropapillomas and a canine COPV-induced canine oral papilloma served as controls.
Results of histopathology including controls, canine oral papillomavirus-specific polymerase chain reaction (COPV-PCR), COPV-specific in situ hybridization (COPV-ISH), bovine papillomavirus specific ISH (BPV-ISH), and immunohistochemistry for papillomaviral group-specific antigens (PV-IHC).
PCR amplicons were sequenced using the Prism BigDye Terminator v1.1 Cycle Sequencing Kit on the DNA sequencer 3130 Genetic Analyzer (both Applied Biosystems, Darmstadt, Germany). To this end, 2 µl (3–5 ng) DNA were eluted from 1.5% agarose gels and purified with the QIAquick Gel Extraction Kit (Qiagen, Hilden, Germany), and the sequencing reaction was conducted using the BigDye Terminator v1.1 Cycle Sequencing RR-100. The sequence was analyzed using a ABI3130. Sequence data were assembled and edited with GCG, Version 11.1 (Accelrys Inc., San Diego, CA).
Histologic examination of the 3 conjunctival specimens revealed various morphologic aspects. In dog No. 1, the focally thickened conjunctiva showed strong similarities to a viral pigmented plaque of haired skin, which is characterized by moderate hypertrophy of stratum basale, progressing through a thickened stratum spinosum (acanthosis) and orthokeratotic and parakeratotic hyperkeratosis. The basal epithelial cells formed variable widely spaced digitations expanding into the underlying stroma. Moderate to marked hyperpigmentation was found preferentially within the lower epithelial layers and to a lesser extent in the stratum spinosum and stratum corneum. A marked hypergranulosis with mildly enlarged and irregularly shaped keratohyalin granules and few koilocytes with a clear cytoplasm and pyknotic nuclei were present in upper epithelial layers (Fig. 2A).
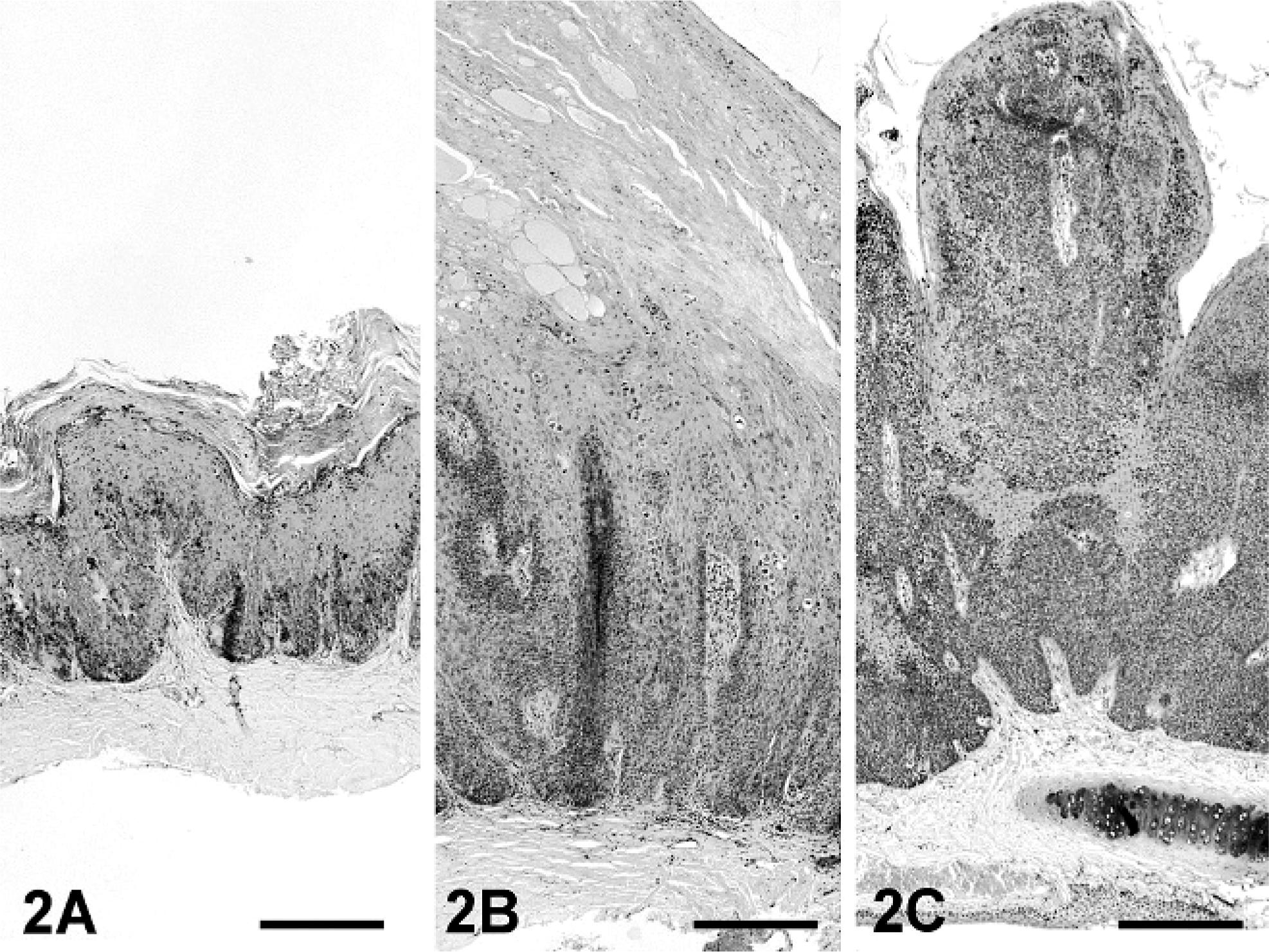
Conjunctiva, viral pigmented plaque; dog No. 1. The conjunctiva shows mild to moderate hyperplasia with acanthosis and hyperkeratosis. Note the undulating pattern of the basal layer. HE. Bar = 250 µm.
Dogs No. 2 and No. 3 presented sharply demarcated viral exophytic squamous papillomas characterized by multiple fingerlike projections of hyperplastic squamous epithelium and congested blood vessels in the connective tissue cores. The epithelial cells progress from a hypertrophied stratum basale through a thickened stratum spinosum (acanthosis). Cells of the basal layer are often elongate and aligned perpendicular to the stroma. Mitotic figures average was 1 per 1 hpf. Multifocally, subcorneal epithelium showed large and irregularly shaped keratohyalin granules and a variable number of koilocytes occasionally with basophilic intranuclear inclusions (Fig. 2B, C). There is prominent orthokeratotic and parakeratotic hyperkeratosis that often contains ghost cells.
In all 3 lesions PCR for COPV DNA yielded amplification products each with the predicted size for the E6, E7, and L1 genes. Sequencing of the amplicons revealed for all 3 fragments 100% identity with the published DNA sequence of COPV (Genbank: D55633). In all samples, in situ hybridization showed that COPV DNA was located in a highly specific pattern within nuclei of the hyperplastic epithelium. The black hybridization signals were especially present, with increasing intensity in the outermost layers and koilocytes (3a, b). Immunohistochemistry revealed only small numbers of positive cells within the keratinized layers and few koilocytes of the papilloma in dog No. 2 (data not shown).
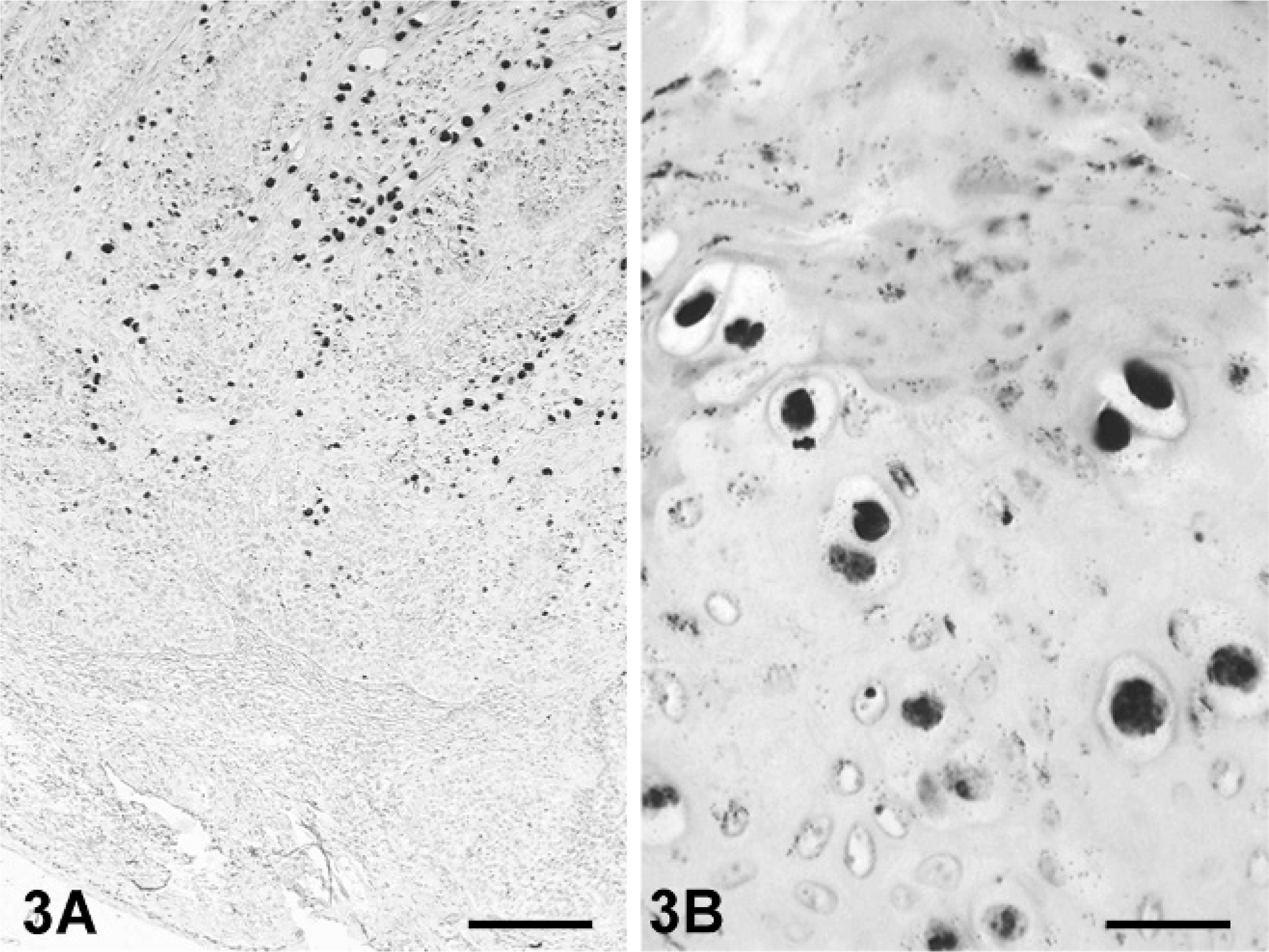
Squamous papilloma; dog No. 2. COPV DNA is present in the basal layer and with increasing intensity toward the outer cornifying layers. ISH. Bar = 200 µm.
Papillomaviruses are known to induce cutaneous plaques or papillomas in various locations on the body. Canine oral papilloma virus was isolated from papillomas in mucocutaneous sites such as the oral cavity and skin. 15, 18 However, viral conjunctival papilloma and pigmented plaque of the third eyelid or conjunctiva are not included in the proposed World Health Organization histologic classification of ocular tumors of domestic animals. 21 In this case study, we present 2 dogs with a classic exophytic papilloma on the conjunctiva of sclera and third eyelid. They were characterized by fingerlike projections of hyperplastic epithelium with giant keratohyalin granules within the subcorneal layers, intranuclear inclusion bodies, and koilocytes as a sign of cytopathic viral effects. Both papillomas were shown to be positive for COPV DNA by PCR and ISH. Only 1 papilloma was weakly positive by immunohistochemistry for group-specific papillomaviral antigen. This is not an unusual finding, as earlier studies have shown only between 38.2% 14 and 61.1% 13 of canine papillomas were positive for papillomaviral antigens by immunohistochemistry. The low sensitivity, together with a presumably low expression rate of COPV antigens in an abnormal location, might be responsible for this apparent discrepancy with PCR and findings by immunohistochemistry.
The conjunctival pigmented plaque of dog No. 1 presented distinctive histologic features, different from commonly encountered exophytic papilloma found on oral or conjunctival mucosa. This conjunctival plaque showed morphologic similarities to cutaneous plaques described elsewhere. 7 Apart from hyperplastic epithelium with hyperkeratosis, hypergranulosis, and koilocytes, only few intranuclear inclusion bodies were detected in the upper layers of the epithelium. The conjunctival plaque was shown to be positive for COPV DNA as demonstrated by PCR and ISH, but group-specific papillomaviral antigen could not be detected by IHC. In contrast, the cutaneous plaques were induced by a papilloma virus other than COPV. 7, 16
The nomenclature of viral pigmented plaque is little confusing because different authors used various terms for the same lesion. There have been described canine epidermodysplasia verruciformis, 19 pigmented plaque, 10 pigmented papule, 7 or lentiginosis profusa. 3 Recently, a suggestion was made to standardize the nomenclature in viral pigmented plaque. 5
Based solely on morphology, consideration must be given to whether the conjunctival plaque is a preliminary stage of an exophytic papilloma or a separate entity. Molecular results, however, support the assumption that the plaque is the initial lesion that progresses to papilloma or squamous cell carcinoma with time. Progression of cutaneous pigmented plaque into squamous cell carcinoma has been already reported. 10 Because COPV-DNA has also been detected in squamous cell carcinomas, a possible progression from virus-induced papilloma or plaque to this malignant variant can not be excluded.
Clinically, only 1 eye was affected in all 3 dogs and no further papilloma virus-induced lesions were detected. None of the 3 dogs had further papillomas or plaques in the oral cavity or skin, and an immunosuppressed condition was not reported in contrast to other studies described. 7, 8 Possible sources of infection might include contact with an infected dog or genetic or acquired disorders of immunity, which lead to increased susceptibility of PV-induced lesions in dogs. 8, 9, 15 In this study no immunosuppression, other diseases or treatments were reported in any of the 3 dogs. It is not known at this time if the dogs suffered from oral papillomatosis as puppies or at a younger age. A genetic predisposition—at least for the conjunctival plaque—could be assumed but not be proven in these cases.
Footnotes
Acknowledgement
We are grateful for the excellent technical assistance provided by G. Czerwinski.
