Abstract
Actinic keratosis (AK) and Bowenoid in situ carcinoma (BISC) are two distinct forms of in situ squamous cell carcinoma in felines. They usually occur on different locations and present with specific clinical and histologic features. However, in some cases, these diseases cannot be distinguished either clinically or histopathologically. The aim of the present study was to determine the accuracy of diagnosis based on clinical or histologic criteria alone, and whether immunohistochemistry for papillomavirus or p53 can improve the accuracy of diagnosis. A series of in situ squamous cell carcinoma cases (
Keywords
Introduction
Actinic keratosis (AK), Bowen's disease (BD), Bowenoid papulosis, erythrodysplasia of Queyrat, and epidermodysplasia verruciformis are regarded as various forms of squamous cell carcinoma (SCC) in situ in man. 2 AK and BD have also been described in felines, although the latter condition does not match exactly its human counterpart. 3, 6, 13 The term Bowenoid in situ carcinoma (BISC) has recently been coined to describe this condition and will be consequently used in this article. 7 Feline AK presents as plaquelike to papillated, solitary or symmetrical, scaly lesions that occur mostly on the pinnae, nose, and eyelids of white cats. 6 BISC lesions are often multifocal, crusted plaques that occur in any location, including dark pigmented and non-sun-exposed haired areas. 3, 6, 13 Histologically, BISC lesions are characterized by irregular epidermal hyperplasia with broad rete ridges and full-thickness epidermal dysplasia. Hair follicle infundibula and isthmi may be involved in the hyperplastic process. 3, 6, 7, 13 Feline AK lesions are usually less hyperplastic, and hair follicles are less deeply affected. Nonetheless, the histologic features are not always conclusive in order to distinguish both diseases, and the pathologist may render a differential diagnosis in such cases. The final diagnosis then has to be made based on clinical features such as the affected area and the color of the skin/hair coat. 6, 7
AK lesions have been attributed to excessive sun exposure, and published data suggest that BISC may be induced, at least in some cases, by papillomavirus (PV) infections. 5, 12, 14 UV-induced mutations of the p53 gene are frequently associated with impaired degradation and abnormal intracellular accumulation of the expressed protein and, consequently, with positive p53 immunostaining. 15 UV-induced conditions such as actinic keratosis may therefore show p53 overexpression. 17– 19 However, high-risk papillomaviruses induce p53 ubiquitinization, and degradation and PV-induced conditions are usually characterized by negative p53 immunostaining. 1, 21 P53 and PV immunohistochemistry (IHC) may consequently be useful to differentiate the 2 forms of feline SCC in situ and might provide valuable insight into their pathogenesis.
The aim of the present study was to determine the accuracy of diagnosis based on clinical or histologic criteria alone and to determine whether further diagnostic steps such as IHC for PV or p53 can improve the accuracy of diagnosis.
Materials and Methods
Study material and clinical features
The study has been carried out on 45 archival formalin-fixed, paraffin-embedded samples of feline SCC in situ obtained by one author (CF) from several experienced dermatopathologists. A previous histologic diagnosis of AK, BISC, or a differential diagnosis AK/BISC and sufficiently detailed clinical information were prerequisites for inclusion in this study. The exact criteria used to make these diagnoses are not known to the authors. Based on the clinical information, a tentative distribution in one of the two following study groups was made. Samples were allocated to the AK group (
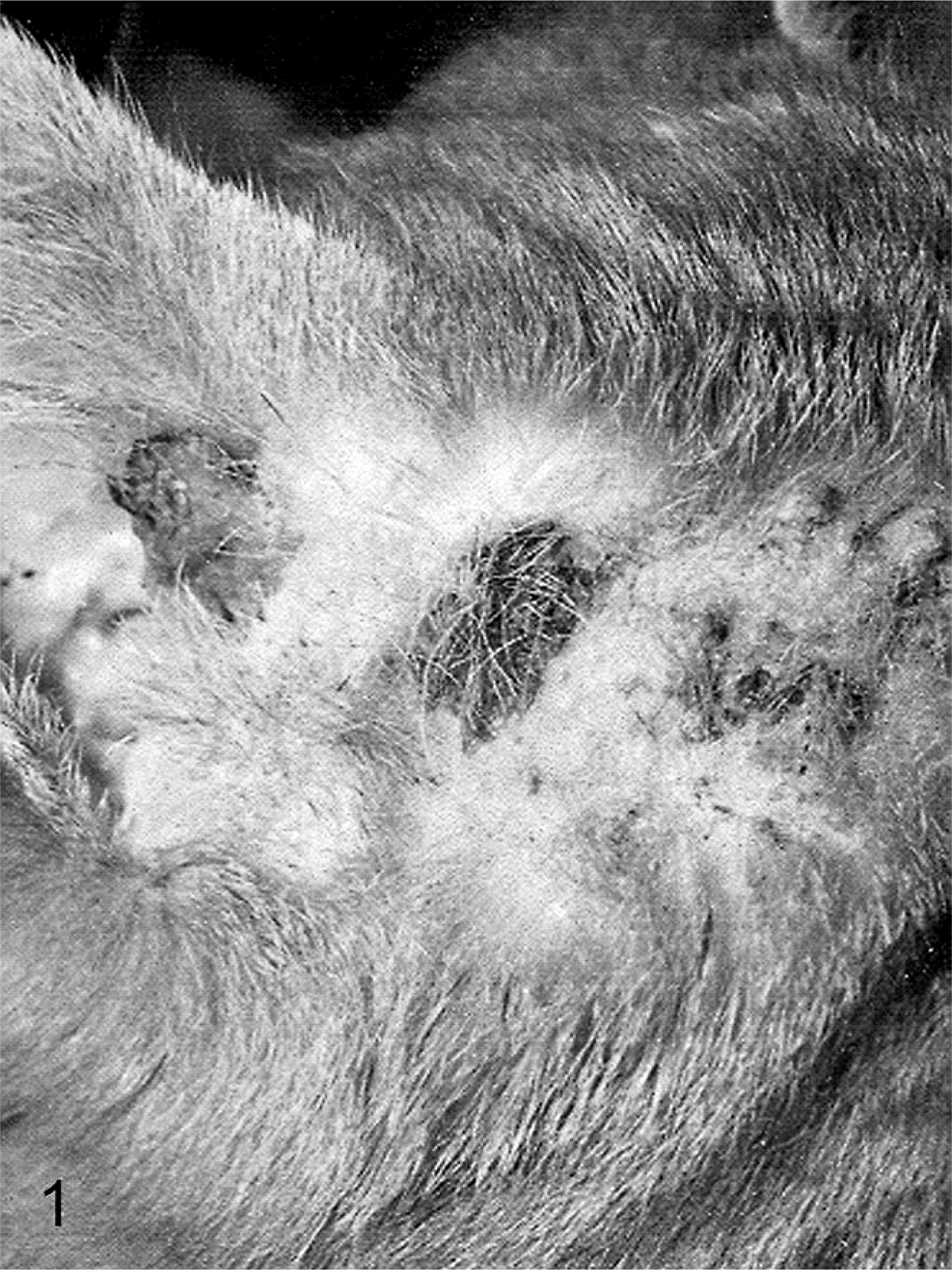
Clinical appearance of BISC lesions (case No. 45). Note the multiple, roughly circular crusty and erosive lesions on an haired area of the head (the area has been shaved).
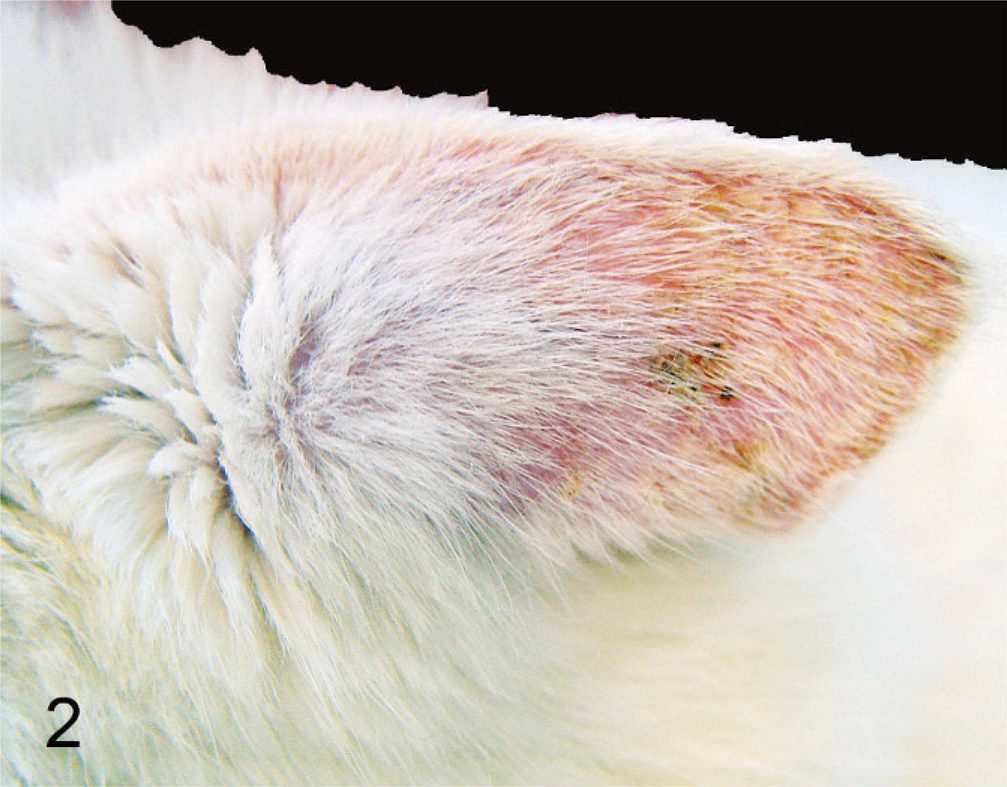
Clinical appearance of AK (case No. 43). Note the generalized erythema of the convex aspect of this white-haired pinna and the poorly demarcated crusty lesions.
Histological examinations
Skin samples had been previously fixed in 4% neutral buffered formalin and embedded in paraffin wax. Sections (3–5 µm) were cut and stained with hematoxylin and eosin or used for immunostaining. The cases were numbered from 1 to 45 by one of the authors (CF) and were evaluated blinded and independently by 2 experienced dermatopathologists (MW, MH), who were not aware of the clinical context. Evaluations were carried out using current criteria adapted from Hargis (Lies of lesions. Personal communication, ISVD Meeting 2003, Monterey, CA) and Gross and coworkers. 7 Briefly, both BISC and AK lesions are characterized by moderate to severe irregular epidermal hyperplasia and dysplasia without invasion through the basement membrane. The following criteria were used to diagnose a lesion as BISC: an abrupt transition between the lesional and nonlesional epidermis; dysplasia in all layers of the epidermis, mostly involving the entire infundibular epithelium and characterized by marked loss of nuclear polarity and epithelial stratification; frequent basaloid appearance and variable size of the keratinocytes; nuclei varying from ovoid hyperchromatic to large and multilobular; multifocal groups of cells with an elongated nucleus tilted in one direction (wind-blown appearance), scattered koilocytes, especially in early stages, with clear cytoplasm, large round keratohyalin granules, pycnotic nuclei, and apoptotic cells. A typical example is shown in Fig. 3.
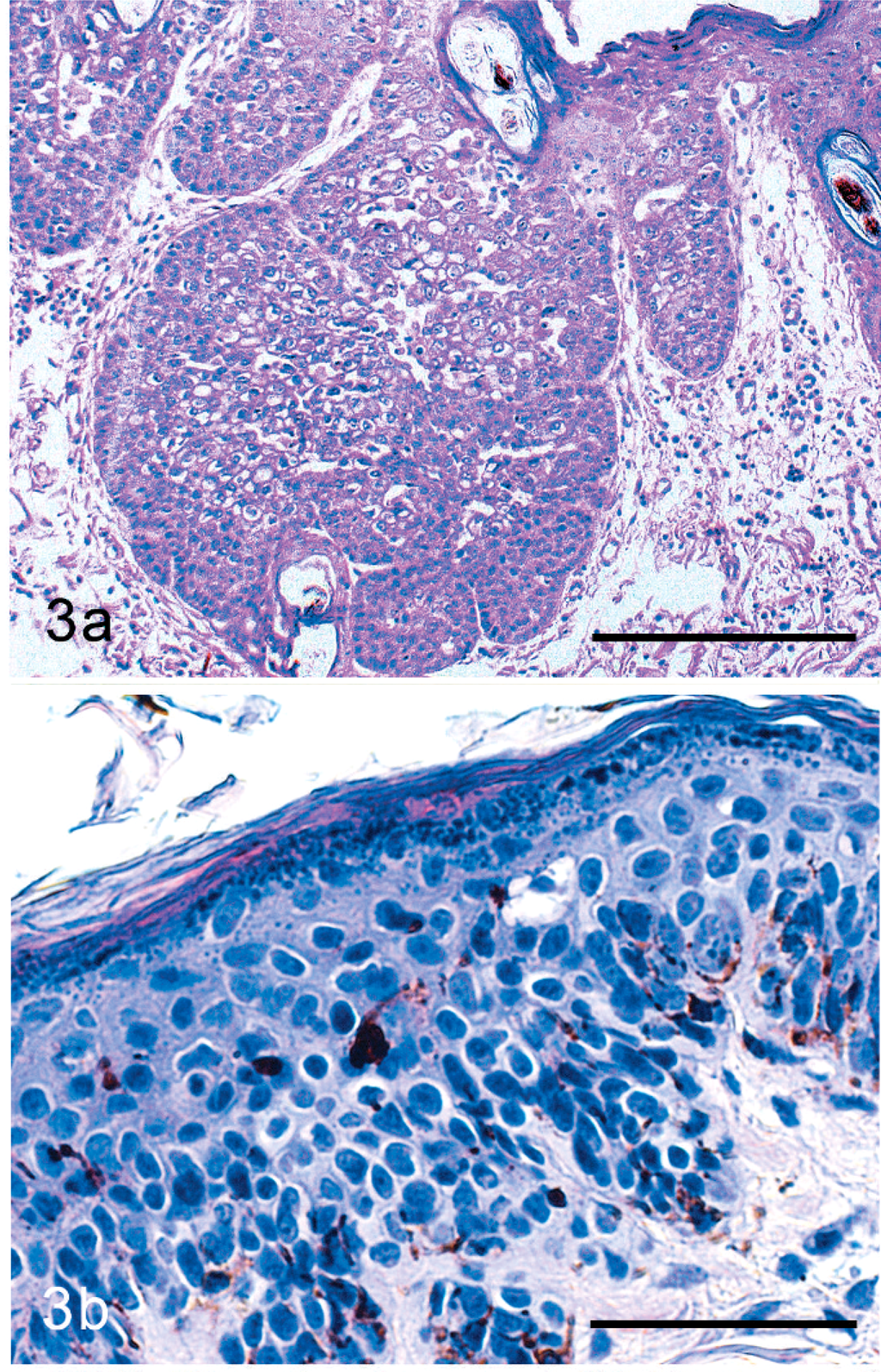
Histologic appearance of BISC (case No. 14). Note the thick irregular hyperplasia of the epidermis with the broad rete ridges and the follicular involvement. HE stain. Bar = 200 µm.
The following criteria were used to classify a lesion as AK: smooth transition between the lesional mild to marked hyperplastic epidermis and the healthy epidermis; loss of cell polarity and thus loss of the normal architecture confined to the basal and spinous layers and involvement of the hair follicle only very superficially, if at all; cytoplasm slightly paler than in the adjacent normal epidermis; mild to moderate nuclear atypia and mild increase in the number of mitotic figures; and apoptotic cells. An example is shown in Fig. 4. Using these criteria, the 2 dermatopathologists classified the 45 cases as BISC, AK, or AK/BISC. The last term indicated a differential diagnosis and was used if a definitive diagnosis was not possible because of overlapping features. The histologic diagnoses of each case were subsequently compared with each other, with the initial histologic diagnosis, and with the tentative clinical classification. The histologic diagnosis was considered clear cut when at least 2 histologic diagnoses were identical and the third diagnosis was either identical with the 2 other diagnoses or AK/BISCr. The cases were considered unambiguous when histologic diagnoses were clear cut and identical to the clinical diagnoses. In all other instances, they were considered ambiguous.
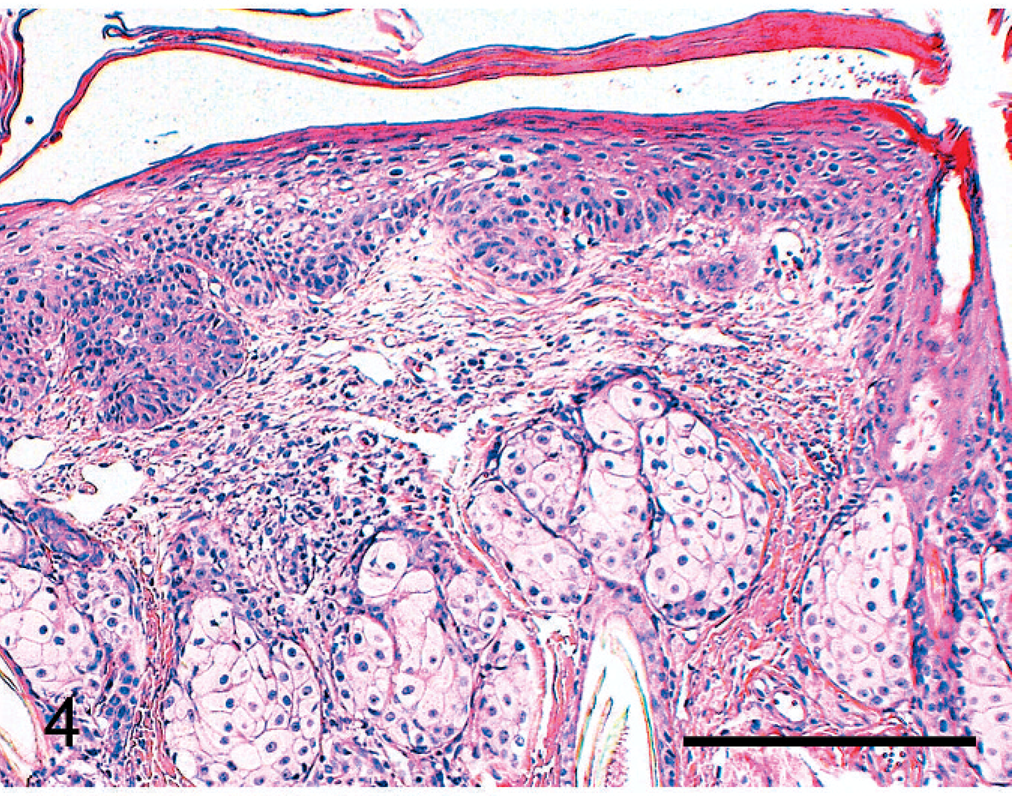
Histologic appearance of AK (case No. 39). Note the moderate hyperplasia of the epidermis, the dysplasia affecting the basal and suprabasal layers of the epidermis, and the discrete hyperplasia of hair follicle (arrow). HE stain. Bar = 200 µm.
Immunohistochemical analyses
Immunohistochemical detection of papillomavirus antigen was carried out by one of the authors (DG) at the Immunology Laboratory of Prairie Diagnostic Services, Saskatoon, Canada, using an avidin–biotin–peroxidase complex technique adapted for an automated slide stainer (CodeOn Histomatic Stainer, Fisher Scientific, Edmonton, AB, Canada) as previously described. 10 A rabbit anti-bovine papillomavirus type-1 antibody (Dako Diagnostics Canada Inc., Mississauga, ON, Canada) was used as the primary antibody, and both diaminobenzidine (Electron Microscopy Sciences, Fort Washington, PA, USA) and Nova Red (Vector Labs Inc., Burlington, ON, Canada) were used as chromogens on 2 different sections for each sample.
As negative controls, replicate sections were stained with a normal rabbit antiserum as the primary antibody. Sections from a canine cutaneous papilloma were included in each run of the immunohistochemical assay as a positive control for the staining procedure.
Immunohistochemical detection of p53 was carried out by one of the authors (FG) using a rabbit polyclonal antibody (Clone CM-1; Novocastra) and following a previously described immunoperoxidase-based protocol. 9 A canine tumor known to carry a mutation of the p53 gene was used as a positive control. Normal epidermis served as an internal negative control in each case. As an additional negative control, consecutive sections of all tumors were treated with the procedure described above, with the exception that the primary antibody was omitted. Samples were regarded as positive when a substantial proportion (exceeding at least 10%) of keratinocytes in regions with hyperplasia/dysplasia showed a positive nuclear signal (Fig. 5). Furthermore, cases were considered negative when positive staining was confined only to a small part of the lesion or to the healthy skin.
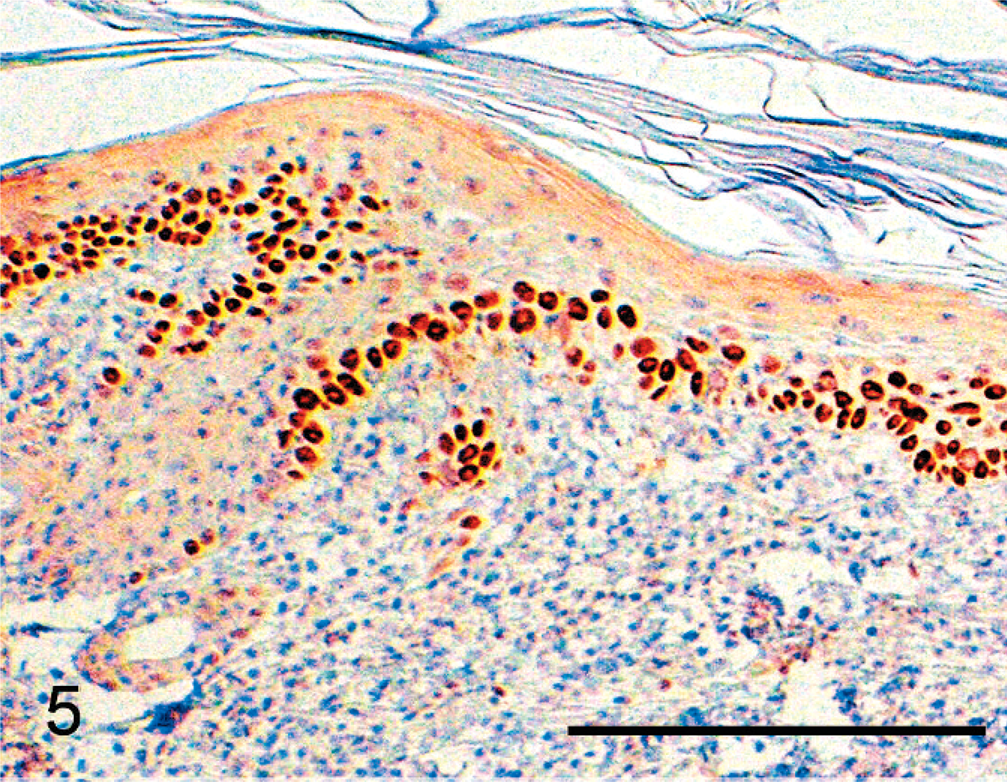
P53 detection in AK (case No. 23). Note the presence of numerous strongly p5-positive nuclei in the stratum basale and stratum spinosum involving the superficial epidermis and part of the hair follicle. p53 immunostain. Bar = 200 µm.
Statistical evaluations regarding positivity for both PV and p53 antigens in both AK and BISC groups were carried out using Fischer's exact test.
Results
Clinical features
BISC lesions occurred in any location on the body and mostly presented as big, crusted, hyperpigmented, and roughly circular lesions with a diameter ranging from several millimeters to 2 cm (Fig. 1). AK lesions were erythematous, scaly to crusty, with a variable degree of erosion/ulceration, depending on the clinical stage (Fig. 2). Clinical features of all cases are summarized in Table 2. They largely reflect the inclusion criteria. Based on the 38 cases, which were clinically clear cut, no breed or sex predisposition was apparent in the 2 groups. Cats that presented with BISC were significantly older than those with AK (mean age AK: 9.5, mean age BISC: 13.1,
Criteria for distribution of samples in AK or BISC clinical groups. ∗

AK = actinic keratosis; BISC = Bowenoid in situ carcinoma.
Clinical, histopathologic, and immunohistochemical data of study cases.
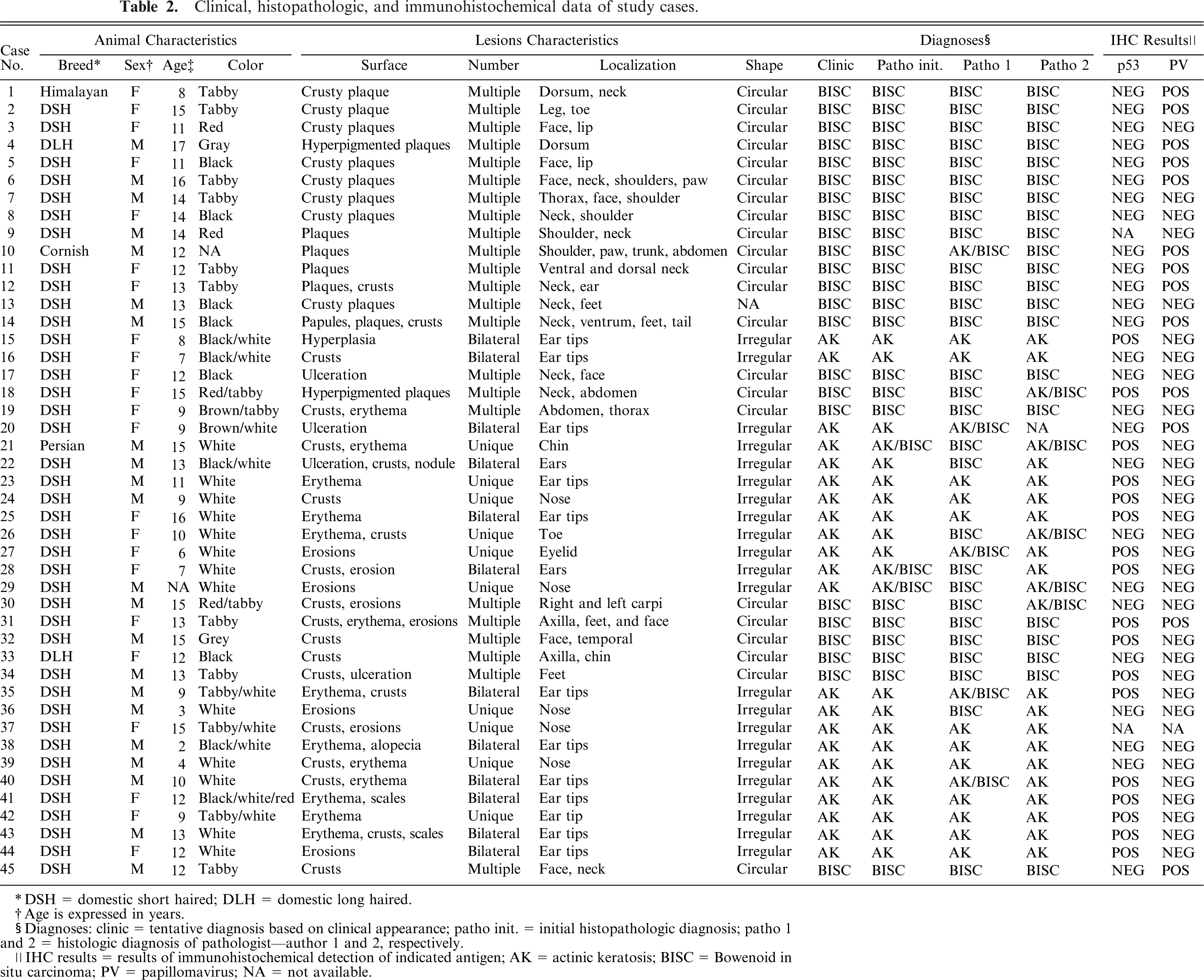
DSH = domestic short haired; DLH = domestic long haired.
Age is expressed in years.
Diagnoses: clinic = tentative diagnosis based on clinical appearance; patho init. = initial histopathologic diagnosis; patho 1 and 2 = histologic diagnosis of pathologist—author 1 and 2, respectively.
IHC results = results of immunohistochemical detection of indicated antigen; AK = actinic keratosis; BISC = Bowenoid in situ carcinoma; PV = papillomavirus; NA = not available.
Histological examination
The major criteria used by the 2 pathologist authors to distinguish between BISC and AK are described in Material and Methods. The initial histologic diagnosis and the diagnoses of the 2 pathologist authors are presented in Table 2. The histologic diagnosis was clear cut in all but 7 cases (No. 20, 21, 22, 26, 28, 29, and 36). Interestingly, in all these cases, the clinical diagnosis was AK. In case No. 20, one of the authors rendered a differential diagnosis, while the second considered the material inadequate for examination. In 3 other cases (No. 22, 28, and 36), one of the pathologists made the diagnosis BISC, while the second one made the diagnosis AK. In 2 of these cases (No. 22, 36), the initial histologic diagnosis was AK, while in case No. 28 the initial pathologic diagnosis was AK/BISC. Two out of 3 pathologists rendered a differential diagnosis in case Nos. 21 and 29, while in case No. 26, one of the pathologists made the diagnosis BISC and the other rendered a differential diagnosis. Analyses of the features leading to conflicting diagnoses in these particular cases revealed that the 2 pathologists were weighing the involvement of deep infundibular portions differently. In addition to the cases with non-clear-cut diagnosis presented above, pathologist authors rendered a differential diagnosis in 4 and 2 cases, respectively. This was usually associated with the presence of features of both conditions (i.e., smooth margins and deep follicular involvement). Interestingly, IHC results were positive in 5 out of these 6 cases and thus helpful for further diagnosis (see below).
Comparing the clinical diagnoses with the histologic diagnoses revealed a large number of unambiguous cases (
Immunohistochemical analyses
p53 immunoreactivity was observed in 11 out of the 14 unambiguous AK cases (78.6%) (case No. 37 was not tested). In all these p53-positive AK cases, labeling involved the nuclei of basal and suprabasal keratinocytes, while most cells in the superficial layers were negative (Fig. 5). Most but not all regions with epidermal hyperplasia/dysplasia were labeled independently of their thickness, conferring a patchlike appearance to the positive areas. In these regions, the superficial epidermis was predominantly affected, and occasional involvement of the hair follicle infundibulum was observed (Fig. 5). Within positive regions, in most of the cases the majority of positive cell nuclei was intensely labeled; focally labeling intensity was variable. In a few cases, labeling intensity of positive nuclei was variable or weak. In most of the cases considered negative based on the 10% cutoff, labeling involved rare or few single scattered basal cells, or small clusters of cells within areas with epidermal hyperplasia/dysplasia.
In the BISC group, only 4 out of 22 (18%) samples (case No. 9 was not tested) showed positive p53 immunostaining. In p53-positive BISC cases, the labeling pattern was not distinguishable from that observed in the AK cases. The basal cell layer was involved in all 4 cases. An important finding in the BISC cases considered negative for p53 based on the 10% cutoff was the presence, in approximately half of the cases, of areas of small to moderate size containing clusters of cells with p53 labeling of variable intensity involving preferentially suprabasal cells and leaving the majority of basal cells negative (Fig. 6). The staining results are presented in Table 2.
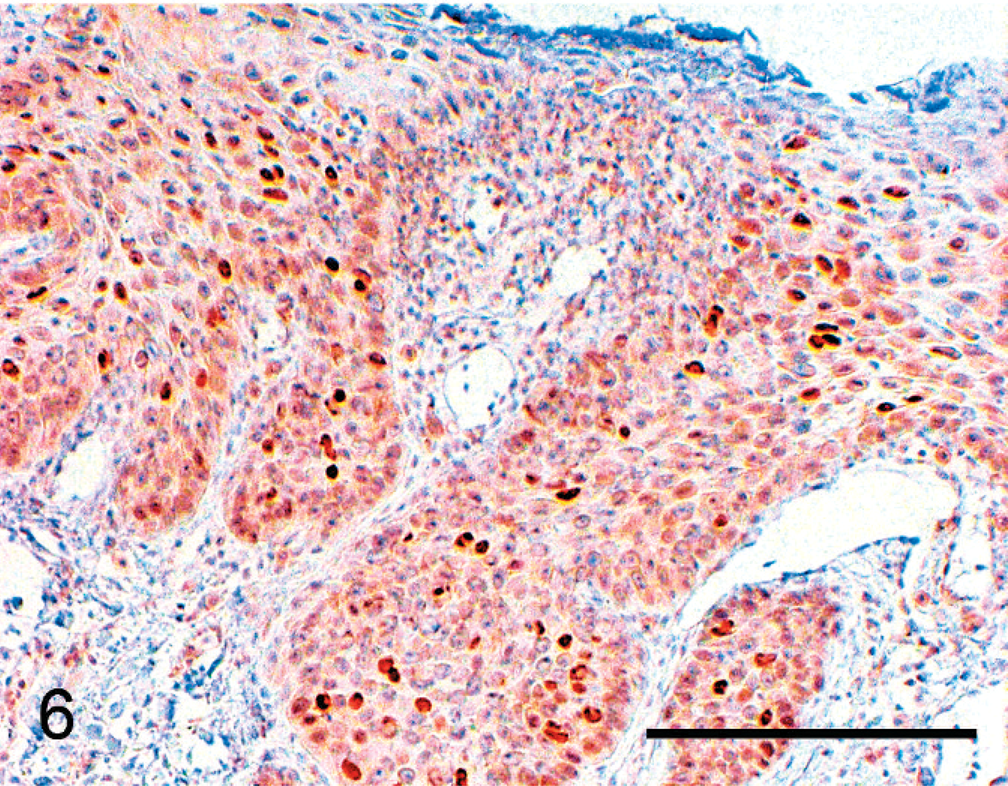
Peculiar pattern of p53 expression in a BISC (case No. 22). Note the presence of scattered p53-positive nuclei mostly located above the basal layer. This case was deemed negative because this pattern was focal, and the overall number of p53-positive nuclei across the whole lesion was below 10%. p53 immunostain. Bar = 200 µm.
The difference between the AK and BISC groups pertaining to p53 positivity is significant (
Eleven out of 23 BISC (47.8%) cases were deemed positive for PV antigens, whereas no positive case was found in the group of unambiguous AK cases. The results are shown in Table 2. The difference pertaining to PV positivity between the 2 groups is significant (
Regarding ambiguous cases, p53 IHC did not provide any further information in 5 cases (Nos. 20, 22, 26, 29, 36), but was positive in 2 cases (Nos. 21, 28). Interestingly, in these 2 cases, cells from the deep infundibulum stained positively. PV IHC suggested BISC in 1 case (No. 20). In this last case, changes occurred on the white-haired part of the ear, which clinically suggested AK. One pathologist author rendered a differential diagnosis, while the other one considered the sample inadequate for evaluation.
Discussion
Based on histologic criteria, an accurate classification into either AK or BISC was made in 84% of the cases of feline in situ SCC. The present study shows, however, that neither clinical nor histologic examinations alone provide sufficient information to make an accurate classification in all cases. This does not mean that the histologic criteria applied are inadequate, but that some cases share criteria suggestive of both conditions or lose the typical features of their initial condition. In fact, it is not surprising that hair-follicle involvement or full-thickness dysplasia is present in some advanced cases of AK. Moreover, it has been shown that koilocytes are rare in advanced cases of BISC, which in some instances render the diagnosis more difficult. 20 Based on the results of this study, major problems can arise in the diagnosis of cases with clinical features suggestive of AK. As 7 out of 22 (32%) such tentative clinical diagnoses were not confirmed by the histology, clinical features cannot be considered sufficient for this diagnosis. PV antigen was detected in only 1 of these ambiguous cases, and BISC cannot be ruled out completely in the remaining cases because this condition may in fact occur as solitary lesions in nonpigmented or nonhaired areas of the skin. However, the histologic diagnosis of clinically typical BISC cases appeared to be reliable. Our data suggest that cases clinically presenting as AK might sometimes and for unknown reasons carry features suggestive of BISC. It must be noted that a bias may have affected to some extent these results because of the fact that samples of ear pinna are easily recognized. This may have led the pathologists to render a diagnosis of AK in some of these cases. Another possible bias might have ensued from visible pigments in most BISC cases.
This study supports the association of PV replication and BISC lesions. This was already shown by other authors and our study confirmed that not all BISC samples are positive for PV antigens in immunohistochemistry. 12, 20 Similarly, a recent PCR-based study demonstrated presence of PV-DNA in only 11 of 18 samples of BISC. 14 The likely causes of this phenomenon have been extensively discussed by Wilhelm and coworkers. 20 Briefly, even if one cannot exclude that BISC may have 2 different causes, one viral and another unknown one, the likely explanation of the negativity of some BISC is the loss of PV protein expression during the malignant transformation process. 4
It was not possible to demonstrate PV replication in any confirmed case of AK in this study, and, from a practical point of view, positivity for PV antigens seems to be a hallmark of BISC lesions and not of AK lesions. Nevertheless, the presence of PV in these lesions cannot be ruled out definitively because of the relatively low sensitivity of the detection technique.
This study shows that AK lesions are often associated with positive p53 immunohistochemical labeling, which suggests that mutations of the p53 gene are frequent in this condition. The widespread occurrence of patches of p53-positive cells in the sunlight-exposed skin of cats is, to the authors' knowledge, a previously unreported finding. Such patches have been shown to frequently occur in human sun-exposed skin and to be associated with mutations of the p53 gene. 8 Sequencing of p53 will be helpful to confirm that mutations are indeed UV-induced based on their typical signature. In addition, such sequencing could contribute to explain why some cases of BISC are also positive for p53 IHC. One hypothesis would be that this is a result of UV-induced mutations, but the presence of p53-positive nuclei deep in the hair follicles could also suggest other mechanisms. For example, it has been shown that an increase in proliferative activity of the epidermis is associated with an overexpression of p53. This overexpression is not associated with a mutation. 16 This mechanism could explain our finding of a focal p53 overexpression in some BISC cases. A study carried out in humans has shown that PV infections are, in some instances, associated with inactivated and stabilized p53. 11 Our results are not conclusive for this point, and further investigations are warranted.
Some cases of AK were deemed negative in IHC for p53. As some UV-induced mutations are not associated with p53 stabilization, these findings do not rule out the presence of such mutations. Alternatively, alterations of other genes might be associated with this condition.
The present study confirms that close cooperation between clinicians and pathologists is mandatory for accurate diagnosis of feline in situ SCC. Nonetheless, some cases, especially late stages, might not be accurately classified as either AK or BISC solely on the basis of clinical and histologic criteria. In some of these cases, IHC, especially for PV, may be helpful to distinguish both conditions. The frequent occurrence of p53 overexpression in AK in this study supports the notion that this condition is UV-induced. In addition, p53 overexpression of unclear origin was occasionally demonstrated in some cases of BISC. Finally, the study confirms the presence of PV in a substantial number of BISC cases. This work contributes to a better characterization of the causes and diagnoses of these conditions; this could be the basis of clinical studies aiming to improve the treatment of AK and BISC.
Footnotes
Acknowledgement
The authors wish to thank Ms. Sabina Wunderlin, Institute of Veterinary Pathology, Zurich, for excellent technical help.
