Abstract
A 2-month-old mongrel dog had multifocal necrotizing hepatitis, interstitial pneumonia, and hemorrhagic enteritis. Immunohistochemistry detected antigens of Clostridium piliforme in the intestine and liver, and antigens of canine distemper virus within the lung, urinary bladder, brain, spleen, and liver. Furthermore, uncharacterized intralesional coccidian protozoa were observed within the intestine.
History, Gross Findings, and Laboratory Results
A 2-month-old, male, mongrel dog, which originated from Estonia, was brought to Finland soon after birth and was admitted to the Veterinary Teaching Hospital, University of Helsinki, due to bloody diarrhea, vomiting, and progressive weight loss. Serum laboratory findings were increased aspartate aminotransferase (479 U/liter; reference range 33–251 U/liter), hypoalbuminemia (8 g/liter; reference range 30–41 g/liter), and hypoproteinemia (19 g/liter; reference range 58–77 g/liter). At necropsy, there was emaciation, sunken ocular globes, and icterus of visible mucous membranes and of the subcutaneous tissue. There were several 0.5- to 1.5-cm-diameter pale areas randomly distributed throughout the capsular and sectioned surfaces of the liver, multifocal hemorrhage of intestinal segments (jejunum, ileum, and colon), and heavy, wet lungs.
Differential Diagnoses
The principal differential diagnoses for hemorrhagic enteritis in dogs includes canine parvovirus, bacterial infections (Neorickettsia helminthoeca, Salmonella spp., Clostridium piliforme, or C. perfringes), or protozoan-induced hemorrhagic enteritis. 2 The main differential diagnoses for infectious hepatic diseases of dogs would be canine leptospirosis, Tyzzer's disease, canine infectious hepatitis, and canine herpesvirus. 9
Microscopic Findings
There was multifocal, necrotizing hepatitis, with intrahepatocytic filamentous bacteria, oriented in crisscross patterns or in parallel formations, which were gram-negative, pale by the routine HE stain, purple with Giemsa, and distinctly identified by the Warthin-Starry (WS) silver impregnation method (Fig. 1). Intestinal lesions included areas of severe sloughing of superficial mucosa, blunting and fusion of intestinal villi, severe cryptal dilation and necrosis, moderate-to-severe hemorrhage associated with intralesional bacteria, and various forms of a coccidian protozoan parasite (Fig. 2). In some intestinal segments, intralesional bacteria had similar morphology as those observed within the liver, whereas bacteria appeared in cross sections in other fragments. Additionally, there were accumulations of bacteria within the intestinal lumen. Pulmonary alterations included degeneration and necrosis of epithelial bronchial and bronchiolar cells, interstitial pneumonia, and pulmonary edema associated with eosinophilic intracytoplasmic inclusion bodies within epithelial cells (Fig. 3). Neurologic lesions consisted of cerebellar white matter vacuolization. There was ballooning degeneration of transitional epithelial cells of the urinary bladder associated with eosinophilic intracytoplasmic inclusion bodies (Fig. 4) and severe depletion of splenic lymphoid tissue.
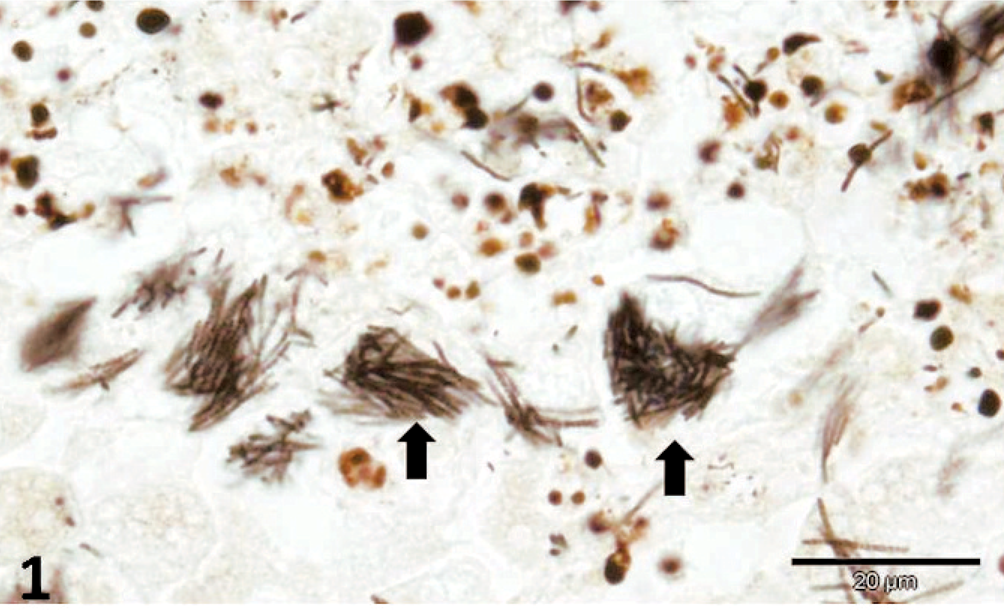
Liver; dog. Hepatocellular necrosis with intracytoplasmic bacilli (arrow) arranged in stacks. Warthin-Starry stain. Bar = 20 μm.
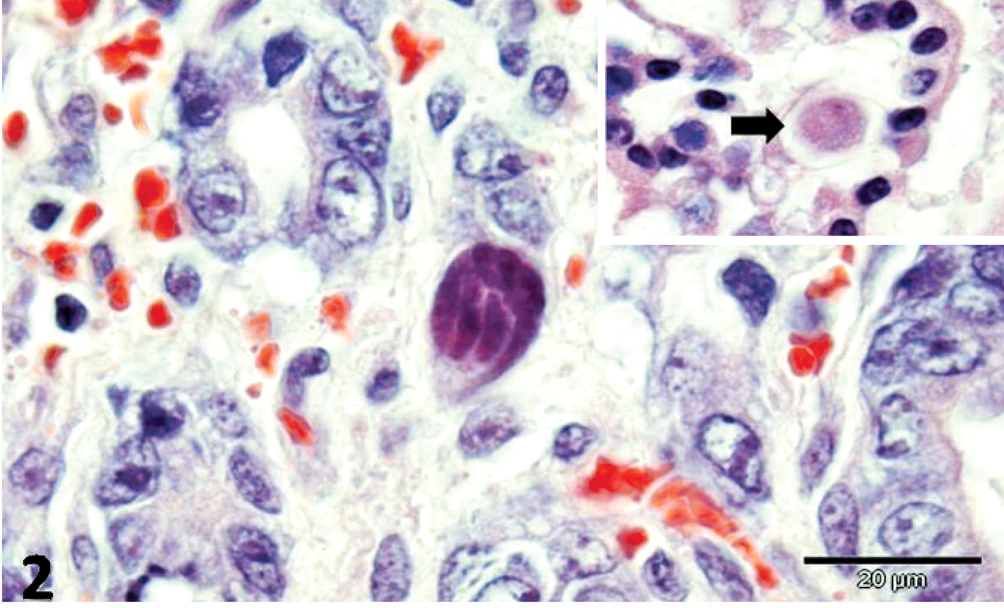
Jejunum; dog. Intralesional trophozoite of coccidial protozoan. Insert: Macrogamete (arrow) of intestinal protozoa. HE. Bar = 20 μm.
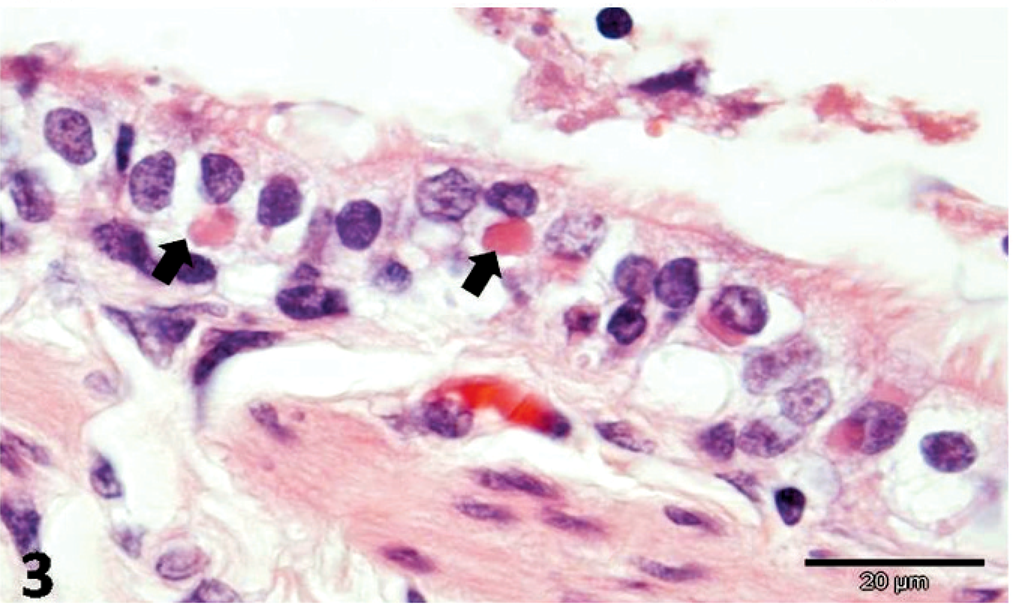
Lung; dog. Distemper viral intracytoplasmic eosinophilic inclusion bodies (arrow) within epithelial cells. HE. Bar = 20 μm.
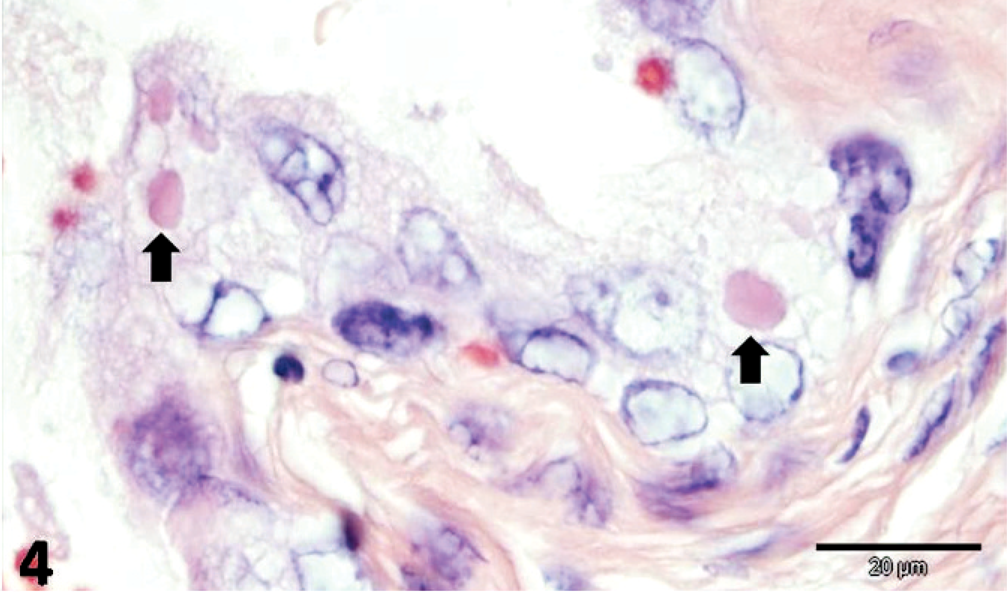
Urinary bladder; dog. Hydropic degeneration of transitional epithelial cells with eosinophilic distemper viral inclusion bodies (arrow). HE. Bar = 20 μm.
Immunohistochemistry
Immunohistochemistry (IHC) was done to detect antigens of canine parvovirus (CPV; Serotec, Oxford, UK) and canine distemper virus (CDV; Serotec), using the streptavidin-biotin technique in an automated staining system (LabVision, Fremont, California). IHC was also used to detect antigens of the RT strain of C. piliforme, as previously described. 4 Tissue samples from previous cases of CDV and CPV (A. Sakura, unpublished data) and of Tyzzer's disease 4 were used as positive controls; for negative controls, the primary antibody was substituted by phosphate-buffered saline (PBS) for CDV and CPV, and by normal mouse serum for C. piliforme.
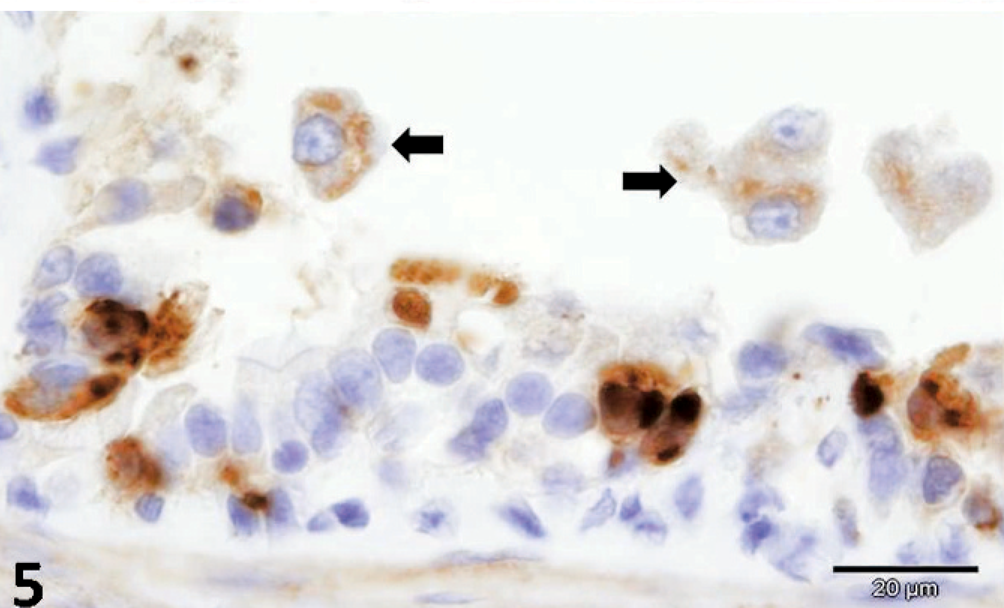
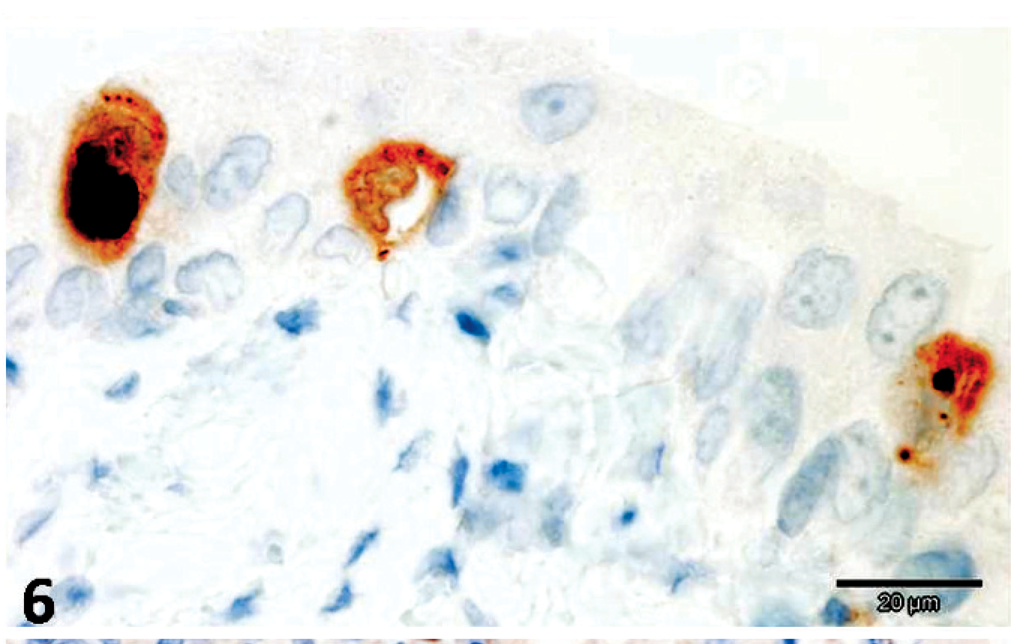
CDV-positive antigens were demonstrated within pulmonary epithelial cells and alveolar macrophages, transitional epithelial cells of the urinary bladder, splenic lymphocytes, astrocytes and neurons of the brain, and within hepatic macrophages (Figs. 5, 6). Additionally, the intracytoplasmic bacilli of hepatocytes and intestinal epithelial cells were positive to anti-C. piliforme RT strain antisera by IHC (Figs. 7, 8). Furthermore, antigens of CPV were not demonstrated in the tissues evaluated, whereas free bacteria within the intestinal lumen were negative for the anti-RT antibody of C. piliforme.
Lung; dog. Positive immunoreaction to antigens of canine distemper virus within pulmonary epithelial cells and macrophages (arrow). Immunohistochemistry; streptavidin-biotin method. Bar = 20 μm.
Urinary bladder; dog. Positive staining of canine distemper virus antigens within transitional epithelial cells. Immunohistochemistry; streptavidin-biotin method. Bar = 20 μm.
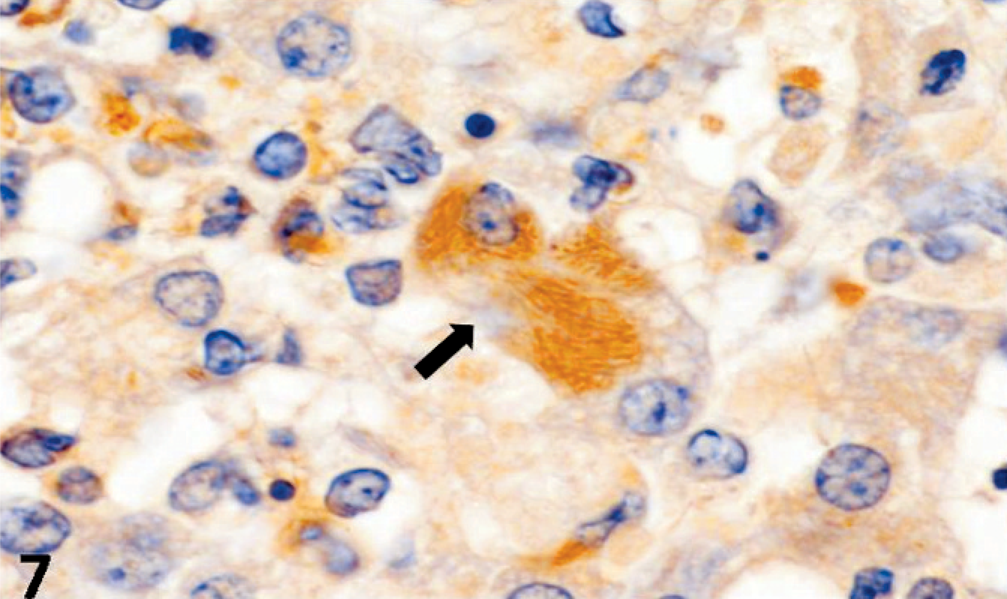
Liver; dog. Intracellular bacilli (arrow) reacting positively to antisera of C. piliforme RT strain. Immunohistochemistry; avidin-biotin peroxidase method. Bar = 10 μm.
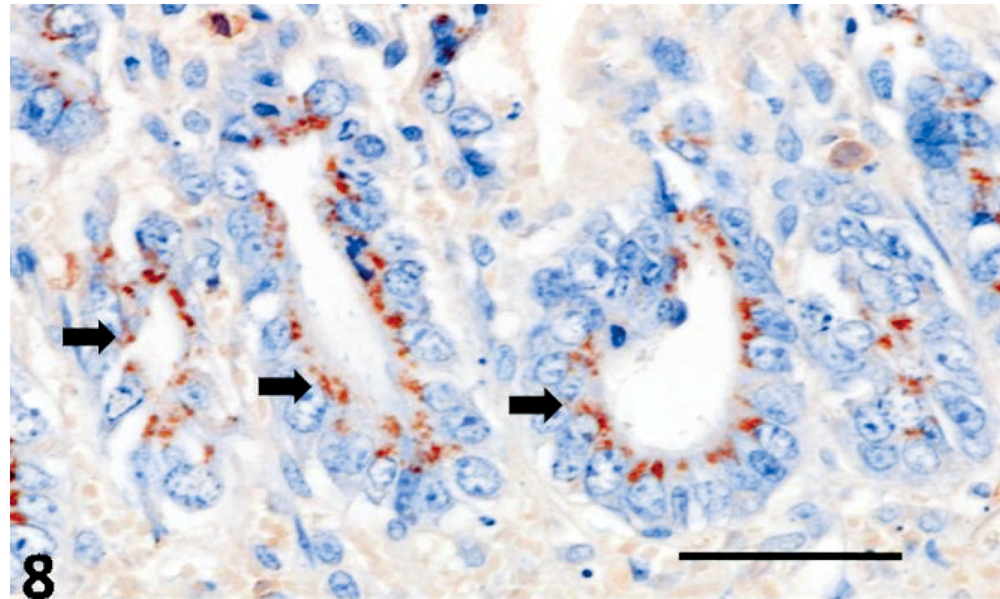
Jejunum; dog. C. piliforme RT strain antiserum (arrow) within the intestinal mucosa. Immunohistochemistry; avidin-biotin peroxidase method. Bar = 50 μm.
Diagnoses
Canine distemper; Tyzzer's disease; intestinal coccidiosis.
Discussion
Tyzzer's disease is an acute spontaneous infection of young animals caused by the spore-forming, gram-negative bacterium C. piliforme. 5, 9, 10 The gross, histopathologic, and histochemical findings observed, principally within the liver of this dog, are consistent with that induced by C. piliforme. 5, 7, 9– 11 The protozoan intestinal parasite was not characterized due to the absence of fecal samples, and the difficulty of clearly characterizing the agent based only on the histologic sections. Nevertheless, since there were various forms of the coccidian parasites, it may be an Isospora species of coccidian. These are the most common cause of enteric coccidiosis in dogs. 3 This is the first report of Tyzzer's disease in a dog from Finland, and probably Northern Europe. There is a report of Tyzzer's disease occurring in a litter of puppies from the UK. 6
In this case, the primary infection was probably due to CDV, which might have predisposed the dog to secondary Tyzzer's disease and intestinal coccidiosis; similar simultaneous infections of CDV and C. piliforme have been previously described in dogs 7 and raccoons. 11 Furthermore, feline panleukopenia virus (FPLV) produced a similar effect in cats with Tyzzer's disease. 4
The pathogenesis of Tyzzer's disease is not fully understood. 5 However, Tyzzer's disease is commonly associated with stress due to weaning, overpopulation, inadequate sanitary conditions, or transport, or with immunosuppression due to corticosteroid, 5 anticancer treatment, 9 or viral-induced infections, 1, 4, 5, 11 as was the case of this dog. Susceptible dogs might be infected by C. piliforme upon ingestion of rodent feces contaminated with bacterial spores or by contact with a previously infected animal. 5 Thereafter, the bacteria enter and proliferate within intestinal epithelial cells, and after immunosuppression, leave the intestine by the portal circulation to colonize the liver, 5 resulting in characteristic multifocal necrotizing hepatitis, as was observed in this dog. Myocardial necrosis is also described in Tyzzer's disease. 8 The intestinal C. piliforme-associated lesions do not form a definite pattern, are not constantly observed, and are unspecific, but the hepatic lesions are diagnostic for Tyzzer's disease, 9 being observed in all mammalian species.
