Abstract
Concurrent Clostridium piliforme and canine distemper virus (CDV) infection was diagnosed in 2 canine littermates and 1 gray fox kit from Texas, USA. In all 3 animals, intracytoplasmic, filamentous bacteria, consistent with C. piliforme, were present along the margins of foci of hepatic necrosis. Additional histologic findings included intracytoplasmic and intranuclear inclusion bodies in bile duct and bronchial epithelial cells of the fox kit, and mild intestinal necrosis in 1 puppy. PCR assays confirmed the presence of C. piliforme in all 3 animals, CDV in both puppies, and canine parvovirus in 1 puppy. Fluorescent antibody testing confirmed the presence of CDV in the fox kit. Concurrent canine distemper and Tyzzer disease in canine littermates and the gray fox has not been reported previously, to our knowledge.
Clostridium piliforme, the agent of Tyzzer disease, is a gram-negative, spore-forming bacterium that is known to infect a variety of species; the infection is particularly fatal for laboratory animals, lagomorphs, and foals. 7 Disease is most common in young, immunocompromised animals, and immunosuppressive factors that can precede infection include stress, poor environmental conditions, and/or diet. 1 Additionally, certain bacterial, fungal, and viral etiologies have been reported to contribute to immunosuppression in animals diagnosed with concurrent Tyzzer disease. One of these viral agents is canine distemper virus (CDV; Paramyxoviridae, Canine morbillivirus), which has been reported as a coinfection with Tyzzer disease sporadically in dogs and in a raccoon.5,6,9
Infection of canids with CDV, an enveloped RNA virus, is more commonly diagnosed than Tyzzer disease, and CDV infection is usually controlled with vaccination. CDV infects dogs and a wide range of wildlife carnivores, such as raccoons, skunks, wolves, coyotes, and foxes. 8 In dogs, secondary infections associated with the immunosuppressive effects of CDV include, among others, toxoplasmosis, mycotic pneumonia, infectious canine hepatitis, and Tyzzer disease.4,6 Here we present 3 cases of concurrent Tyzzer disease and canine distemper in 2 littermate puppies and 1 gray fox kit.
Cases 1 and 2 were intact female, 8-wk-old, mixed-breed littermate puppies that were presented to a veterinary clinic after being housed at a foster home in east Texas, USA for 2–3 d. Both animals had been diagnosed and treated previously for coccidiosis. They had been vaccinated with a multivalent vaccine (Nobivac Canine 1-DAPPv; Merck) that included CDV, adenovirus, parainfluenza virus, and canine parvovirus (CPV; Carnivore protoparvovirus 1), 5.5 wk prior to presentation for autopsy. Case 1 received a booster vaccination 3 wk following the initial vaccination; however, case 2 received only the single, initial dose. Upon arrival at the veterinary clinic, both animals were malnourished, in lateral recumbency, and had icteric mucous membranes and seizures. Case 1 was euthanized, and case 2 died spontaneously shortly after arrival. Acute hepatotoxicity was suspected in both cases based on clinical signs and an antemortem chemistry profile performed on case 1 (moderate hypoalbuminemia, markedly elevated alkaline phosphatase activity, severely elevated alanine aminotransferase [ALT] activity, marked hyperbilirubinemia, and moderate-to-marked hypoglycemia). The owner reported that 2 other littermates that were housed at the same foster home had died shortly after developing hemorrhagic diarrhea. Parvovirus SNAP tests performed for those 2 littermates and for case 1 were negative.
Case 3, a male gray fox, estimated to be 8-wk-old, was found dead outdoors in Austin, TX. It was part of a family of wild foxes with a litter of 7 kits, of which 4 were found dead; 2 of the 4 kits had been observed to be thin, stressed, and lethargic for a short period of time. One other dead kit was also submitted for autopsy.
All 3 cases were submitted for autopsy to the Texas A&M Veterinary Medical Diagnostic Laboratory in College Station, TX. The 3 carcasses were in very poor nutritional condition, with no fat reserves and generalized muscle atrophy. Grossly, similar changes in the livers from the 3 animals included diffuse pallor, friability, and pale, pinpoint foci (Fig. 1A, 1B). In addition, case 2 had slightly over-expanded lungs that were mildly firm, with rib impressions; the small intestine had prominent Peyer patches and segmental areas with red liquid-to-mucoid contents. Case 3 also had severe flea infestation and a small, dark, consolidated area in the right caudal lung lobe. Samples of liver, lung, heart, brain, sciatic nerve, spleen, kidney, adrenal gland, urinary bladder, small intestine, large intestine, esophagus, trachea, thyroid gland, tongue, pancreas, lymph node, and bone marrow from the 3 cases were taken and fixed by immersion in 10% neutral-buffered formalin for 4 d. The tissues were processed routinely for the production of H&E-stained sections. Fresh tissues from all 3 cases were also collected at autopsy for additional testing.
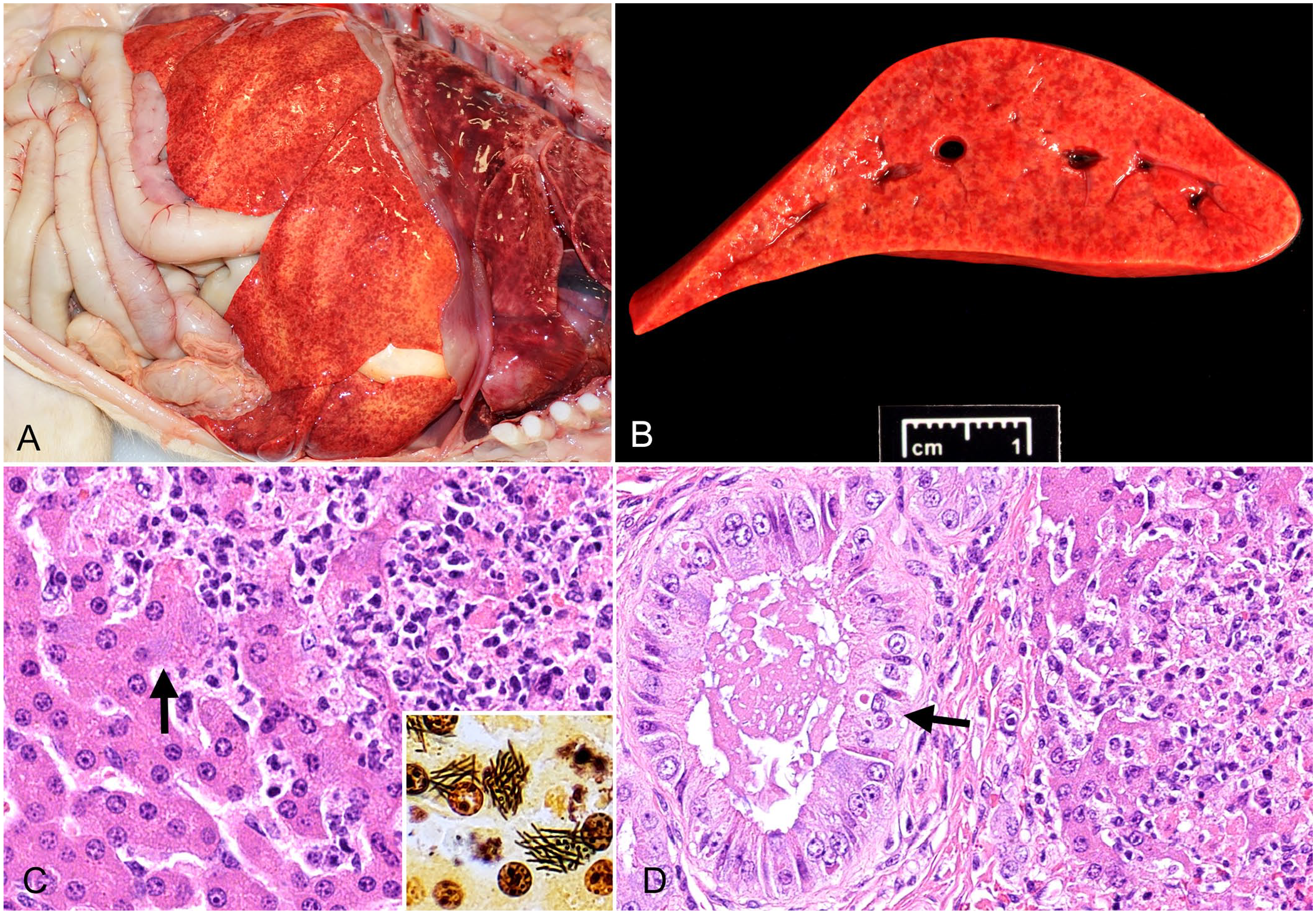
Tyzzer disease and canine distemper in an 8-wk-old puppy, case 1 (A, B); and in a gray fox kit, case 3 (C, D).
Microscopically, the livers of the 3 animals had similar lesions consisting of randomly distributed, discrete, approximately round, multifocal-to-coalescing areas of necrosis and neutrophilic inflammation. Within the cytoplasm of many hepatocytes, and more rarely in the extracellular space, most often along the periphery of these necrotic areas, were rare, faintly basophilic, filamentous bacteria arranged in crisscrossed or stacked bundles (Fig. 1C). These bacteria stained strongly positive with Steiner stain (Fig. 1C). Orange plugs of bile were frequently found in bile canaliculi. Additional histologic findings in the liver of case 3 included numerous eosinophilic intracytoplasmic and intranuclear CDV inclusion bodies in the bile duct epithelium (Fig. 1D).
In the lungs, cases 1 and 2 had mild-to-moderate pulmonary edema, fibrin, and foamy macrophages found in occasional alveoli. No viral inclusion bodies were found in the lungs of cases 1 or 2. Case 3 had a focal area of severe, necrosuppurative, bronchointerstitial pneumonia with numerous eosinophilic intracytoplasmic and intranuclear inclusion bodies similar to those described in the biliary epithelium.
In case 1, a small, focal, subendocardial infiltrate of a few lymphocytes and plasma cells was observed in the left ventricle. In case 2, the small intestine had occasional mildly to moderately dilated crypts that contained small amounts of necrotic cell debris. No viral inclusion bodies were observed in the small or large intestines of all cases, and no other microscopic abnormalities were observed in any of the other tissues from the 3 animals examined. Steiner stains performed on sections of heart from case 1 and small and large intestine from case 2 did not reveal any positively staining bacteria.
To confirm the presence of C. piliforme, nucleic acid from paraffin sections of liver from all 3 cases were subjected to PCR testing as described in the literature. 2 All 3 cases were positive for the 16S rRNA gene of C. piliforme.
Fresh brain tissue from cases 1 and 2 was pooled and processed for CDV reverse-transcription real-time PCR (RT-rtPCR) as described previously. 2 Target nucleic acid for CDV was detected with a cycle threshold (Ct) of 28.5, indicating that moderate-to-large amounts of CDV nucleic acids were present. Real-time PCR for the detection of CPV was also performed on paraffin curls of the small intestine from cases 1 and 2 and was detected in case 2 with a Ct of 34.6.
For case 3, fresh and fixed samples of lung, liver, and kidney were processed for fluorescent antibody (FA) testing for CDV. The fresh tissues were frozen in cryomolds containing optimal cutting temperature embedding medium (Tissue-Tek; Sakura), and ~5-mm thick sections were cut using a cryotome before the sections were mounted on positively charged slides. Once dried, the slides were placed in ice-cold acetone for 10–15 min and allowed to air dry. For fixed tissues, antigen retrieval was done with proteinase K solution at room temperature for 90 s. The slides were then washed 3 times in PBS, 5–10 min per wash. Fresh and fixed sections were covered with anti-CDV fluorescein isothiocyanate antibody (1:25 and 1:15, respectively; VMRD) and incubated for 3–4 h at 37°C in a humidified chamber. Following incubation, slides were washed 3 times in PBS (5–10 min per wash), covered with FA mounting medium (1% glycerol in PBS) and coverslipped, then all sections were examined with a fluorescence microscope. All sections tested, both fresh and fixed, had positive CDV fluorescence.
A presumptive diagnosis of concurrent C. piliforme and CDV infection was established based on gross and microscopic changes and confirmed by detection of C. piliforme by PCR and CDV by RT-rtPCR and FA testing in all 3 cases.
C. piliforme, like other Clostridium spp., is ubiquitous in the environment and is most likely transmitted through ingestion of its spores, which can survive in the environment for periods of at least one year. 1 C. piliforme most commonly infects young and juvenile animals and/or those that are immunocompromised. Environmental factors most likely played a role in the infections of all 3 cases described here. For cases 1 and 2, the rescue group reported that the foster caretaker housed these puppies, as well as their littermates, outdoors in unsanitary conditions. This caretaker had a history of previous issues of infectious disease on the property. Case 3 was one of a wild fox litter; itself and its littermates were observed to be malnourished and distressed before being found dead.
When cases 1 and 2 died and were submitted for autopsy, hepatotoxicity was suspected based on clinical presentation and biochemical assays. Given the acute nature of Tyzzer disease, the clinical presentation observed in these cases has not been reported commonly in dogs. A dog with Tyzzer disease with clinical signs similar to that of hepatotoxicity, including increased ALT activity, icteric mucous membranes, and icteric subcutaneous tissue upon autopsy was reported previously. 3 Additionally, in the previous report, the dog was also diagnosed with CDV infection and coccidiosis, and the authors proposed that immunosuppression from CDV predisposed the dog to Tyzzer disease and coccidiosis. 3
Before death, both cases 1 and 2, and 2 of their littermates, had hemorrhagic diarrhea and were tested for CPV using a SNAP test; all test results were negative. However, histologic findings in the small intestine of case 2 and the detection of CPV by rtPCR were compatible with parvoviral infection. Unsanitary conditions and the coinfections previously mentioned are factors that likely contributed to a weakened immune status in case 2, and thus it is not surprising that this puppy was also infected with CPV.
It is interesting that cases 1 and 2 were related and from the same litter, as typically Tyzzer disease is a sporadic disease with individual animals being affected. For example, a second fox kit from the same litter as case 3 was also submitted for autopsy, and this kit was diagnosed with CDV but did not have Tyzzer disease. We believe this signifies that cases 1 and 2 were more severely immunocompromised, perhaps because of various genetic, gestational, environmental, or other infectious influences. Notably, the CPV infection mentioned above was an additional immunosuppressive factor in this dog litter.
Although Tyzzer disease and CDV coinfection has been reported in the domestic dog, to our knowledge, this coinfection in related dogs and in the gray fox has not been reported previously. These cases demonstrate the significance of environmental factors as a risk for C. piliforme infection and highlight the contribution of the immunosuppressive effects of CDV, which has been proposed in previous cases of coinfection with Tyzzer disease.3–6,9 Our cases also emphasize the importance of considering antecedent CDV infection in any canid species diagnosed with Tyzzer disease.
Footnotes
Acknowledgements
We thank Dr. Brian Porter for his assistance with these cases, and Dr. Andrés de la Concha-Bermejillo for his assistance with autopsy of another fox kit from the litter. This manuscript is dedicated to the memory of Dr. Bob Dittmar.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
