Abstract
Canine distemper virus (CDV) has long been recognized as a cause of myocarditis; however, cases of myocarditis caused by naturally acquired CDV infection have been reported only rarely in dogs. We describe here our retrospective study of naturally acquired systemic CDV infection in 4 dogs, 4–7 wk old, that had myocarditis, with myocardial necrosis and fibrosis. One of the 4 dogs had intracytoplasmic eosinophilic inclusion bodies in cardiomyocytes. Other lesions included bronchointerstitial pneumonia (4 of 4), necrotizing hepatitis (2 of 4), splenic lymphoid necrosis (2 of 4), encephalitis (1 of 3; brain was not submitted in 1 case), and necrotizing gastroenteritis (1 of 4). The presence of CDV in the heart was confirmed by immunohistochemistry in all 4 dogs.
Infectious myocarditis, an uncommon and often fatal disease in dogs, can be caused by various microorganisms including viruses, protozoa, bacteria, and fungi.6,7,11,13 A retrospective study using a PCR assay reported that canine adenovirus (Canine mastadenovirus A), canine respiratory coronavirus, canine distemper virus (CDV; Canine morbillivirus), canine parainfluenza virus, canine herpesvirus 1 (Canid alphaherpesvirus 1), West Nile virus, and canine parvovirus 2 (CPV-2), were detected in cases of canine viral myocarditis. 11 CDV was detected in 5 of 54 (9%) dogs with myocarditis by PCR assay, but immunohistochemistry (IHC) for CDV was negative in all of the cases.
Although CDV has been described as a cause of canine myocarditis in textbooks, 13 a literature search found few cases of naturally acquired CDV infection with myocarditis. An experimental CDV infection study demonstrated myocardial necrosis and inflammation in young puppies.7,8 We describe here 4 cases of naturally acquired CDV infections with myocarditis.
A database search of archived diagnostic cases of CDV infection from January 2005 to February 2020 at the University of Missouri, Veterinary Medical Diagnostic Laboratory (Columbia, MO) yielded 135 cases, and among those, 4 cases had myocarditis. The diagnoses of all 4 cases were supported by histopathology, IHC, and PCR assay.
All 4 dogs were 4–7 wk old. Cases 1–3 had clinical signs of lethargy and neurologic signs of seizures and circling. Case 4 died suddenly without any significant clinical signs. A local veterinarian performed the autopsy on case 1 and submitted major organs, except brain, for histologic examination. Cases 2–4 were autopsied at the University of Missouri. CDV vaccination status of the 4 dams was not available. Case 1 was from California; cases 2–4 were from Missouri.
On histopathology, all 4 dogs had multifocal mild-to-moderate myocarditis, primarily with lymphocytes and lesser numbers of neutrophils and macrophages. The inflammation was accompanied by myocardial necrosis and fibrosis (Fig. 1A). Dog 1 had eosinophilic intracytoplasmic inclusion bodies (Fig. 1A, inset). Other lesions (Table 1) included bronchointerstitial pneumonia (4 of 4), necrotizing hepatitis (2 of 4), splenic lymphoid necrosis (2 of 4), encephalitis (1 of 3), and necrotizing gastroenteritis (1 of 4).
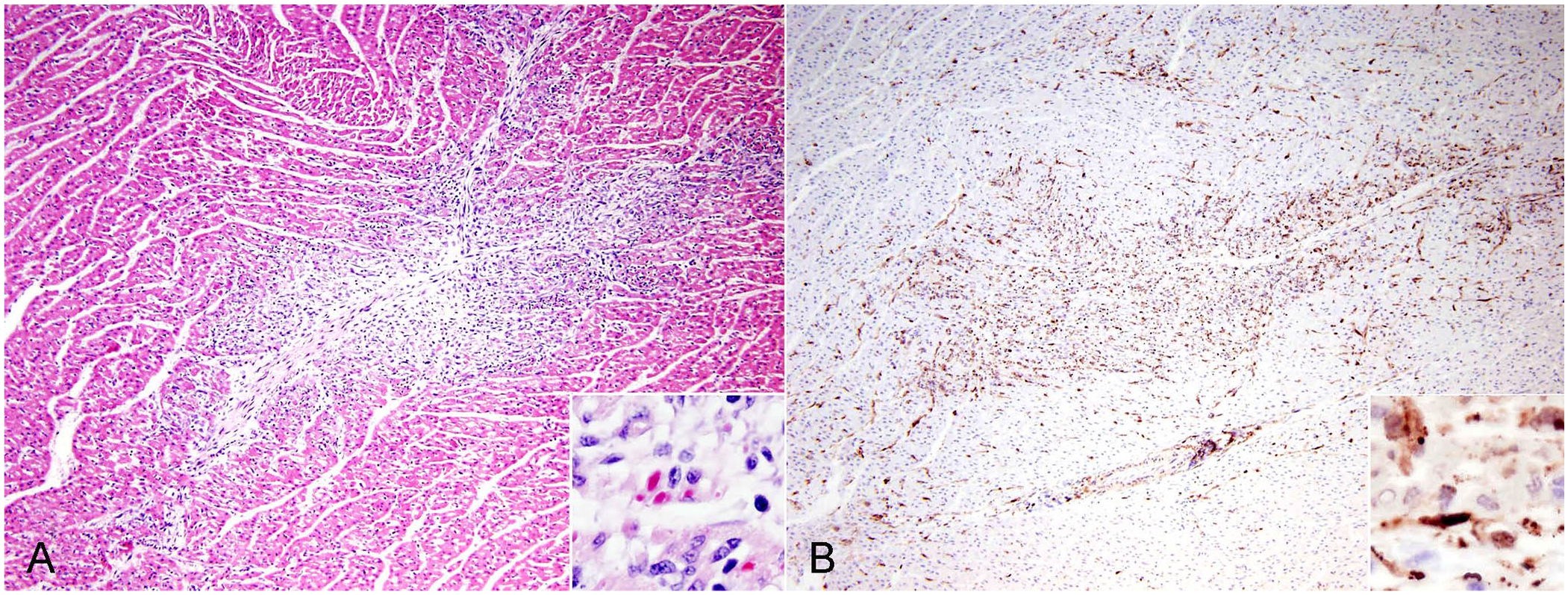
Myocarditis caused by canine distemper virus infection in a dog, case 1.
Naturally acquired canine distemper virus infection in 4 dogs with myocarditis.

NA = tissue was not available; + = positive.
All 4 dogs had positive immunoreactivity for CDV in the myocardium and were negative for CPV-2 (Fig. 1B). For IHC, mouse anti–distemper virus monoclonal antibody (clone DV2.12; Custom Monoclonals International) and mouse anti–CPV-2 monoclonal antibody (A3B10; VMRD) were used as the primary antibodies. The MACH 2 system (Biocare Medical) was used for antigen detection; immunoreactivity was visualized (Romulin AEC chromogen; Biocare Medical). Hematoxylin was used as counterstain.
PCR assays for CDV and CPV-2 were performed in cases 1, 3, and 4. All 3 dogs were positive for CDV but negative for CPV-2. A PCR assay for canine herpesvirus was performed only in case 4, which was also negative. Amplification of CDV was carried out in a single reaction (OneStep RT-PCR kit reagents; Qiagen). Primers CDV-F and CDV-R were employed under PCR conditions described previously. 4 Testing for canine herpesvirus was carried out (Applied Biosystems 7500 Fast real-time PCR system, Thermo Fisher Scientific; QuantiTect Probe PCR master mix kit, Qiagen). The TaqMan primer pairs and probes were described previously. 2 For the CPV-2 PCR assay, a primer pair that amplified a 529-bp fragment of CPV-2 was employed: 5′-CATCATTGGGGAAAAGTACC (Parvo F) and 5′-GGGTTAGTTGGTTCTCCTTGGT (Parvo R). The PCR conditions for the amplifications were: hold at 95°C for 12 min, followed by an initial touchdown step consisting of 14 cycles at 95°C for 30 s, 72°C for 30 s, and 72°C for 1 min—the annealing temperature was decreased by 1°C every cycle. This initial step was followed by 35 cycles at 95°C for 30 s, 58°C for 30 s, and 72°C for 1 min. Following final extension at 72°C for 7 min, PCR products were analyzed on 2% agarose gel and visualized on a UV light table.
CPV-2 infection has been recognized as the most common cause of myocarditis in young dogs.5,11 Conventional and real-time PCR assays detected CPV-2 in 12 of 40 dogs (30%) <2 y old with myocarditis, and in 2 of 41 (5%) control young dogs.5,11 In addition, 2 of 26 (4%) dogs >2 y old with myocarditis were positive for CPV-2 by PCR assay, but viral quantity was low and not detected by in situ hybridization in these cases. 11 CDV was detected in 5 of 54 (9%) dogs with myocarditis, of which 28 were young dogs <2 y old and 26 were adult dogs; no positive case was detected among the 26 adult dogs. Interestingly, the PCR assay detected CDV in 2 of 35 control young dogs and 5 of 22 control adult dogs; however, no CDV antigen was detected by IHC in all PCR-positive, diseased and control cases. The authors suggested that CDV in myocarditis mainly plays an immunosuppressive role and not a primary etiology, because in all PCR-positive cases, the CDV nucleic acid was co-detected with that of canine adenovirus, CPV-2, canine parainfluenza virus, Mycoplasma cynos, and/or Neospora caninum. 11
CDV infection can cause a highly contagious systemic disease that affects a wide range of animals including domesticated and wild dogs, ferrets, raccoons, big cats, and pinnipeds.9,10 CDV is a single-stranded RNA virus that belongs to the genus Morbillivirus, family Paramyxoviridae, along with measles virus (Measles morbillivirus), cetacean morbillivirus, feline morbillivirus, peste-des-petits-ruminants virus (Small ruminant morbillivirus), phocine distemper virus (Phocine morbillivirus), and rinderpest virus (Rinderpest morbillivirus). 10 CDV initially enters the host via the upper respiratory or oral mucosal epithelium, locally infecting lymphocytes, macrophages, and dendritic cells, prior to spreading to the tonsils and regional respiratory lymph nodes within 24 h. Within 2–4 d, all of the lymphoid system becomes infected, resulting in severe immunosuppression. By 6–9 d post-infection (dpi), the virus spreads to other epithelial cells, such as those of the gastrointestinal system, liver, and kidney by cell-associated viremia.9,11
An experimentally induced CDV infection caused myocardial degeneration, necrosis, and mineralization with mild inflammation in 11 of 30 (36%) gnotobiotic Beagle puppies.7,8 The puppies were inoculated with CDV at 5–7 d old, and gross or microscopic lesions of the heart developed as early as 16 dpi. However, when the virus was inoculated into puppies 10–21 d old, 0 of 27 had myocardial lesions. The authors described eosinophilic viral inclusions in sarcoplasm of affected hearts but did not specify how many dogs had inclusion bodies in the cardiomyocytes. All infected dogs in our study were 4–7 wk old at the time of autopsy, although it is uncertain when CDV infection occurred. Only case 1 had several eosinophilic intracytoplasmic inclusion bodies, which is a rare finding. CDV inclusions can be found in the epithelial cells of airways, stomach, bile duct, urinary tract, skin, and central nervous system. 10
Measles virus, a morbillivirus that infects people, can cause systemic disease, similar to that seen in dogs infected with CDV. 12 In humans, myocarditis associated with measles viral infection is also rare. A report of a case of measles myocarditis and an accompanying review of the literature supported the rarity of measles-associated myocarditis. 3
CDV vaccination has significantly reduced the number of CDV infections in domestic dogs worldwide; however, despite intense vaccination, the disease continues to be globally recognized, including in the United States. 1 A 2018 study reported that there are 3 main CDV lineages circulating in the United States: America-3, America-4, and America-5. These lineages are different from those traditionally identified in the United States, including America-1, which is included in the majority of current distemper vaccines. 1 The discrepancy in vaccine strains and circulating wild-type viruses, together with increased interstate and international travel, are believed to be important factors contributing to the recent cases of CDV infections.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
