Abstract
Burkholderia mallei causes the highly contagious and debilitating zoonosis glanders, which infects via inhalation or percutaneous inoculation and often culminates in life-threatening pneumonia and sepsis. In humans, glanders is difficult to diagnose and requires prolonged antibiotic therapy with low success rates. No vaccine exists to protect against B. mallei, and there is concern regarding its use as a bioweapon. The authors previously identified the protein BpaB as a potential target for devising therapies due to its role in adherence to host cells and the formation of biofilms in vitro and its contribution to pathogenicity in a mouse model of glanders. In the present study, the authors developed an immunostaining approach to probe tissues of experimentally infected animals and demonstrated that BpaB is produced exclusively in vivo by wild-type B. mallei in target organs from mice and marmosets. They detected the expression of BpaB by B. mallei both extracellularly and within macrophages, neutrophils, and epithelial cells in respiratory tissues (7/10 marmoset; 2/2 mouse). The authors also noted the intracellular expression of BpaB by B. mallei in macrophages in the regional lymph nodes of mice (2/2 tissues) and MALT of marmosets (4/5 tissues). It is interesting that B. mallei bacteria infecting distal organs did not express BpaB (2/2 mice; 3/3 marmosets), suggesting that the protein is not necessary for bacterial fitness in these anatomic locations. These findings underscore the value of BpaB as a target for developing medical countermeasures and provide insight into its role in pathogenesis.
Keywords
Burkholderia mallei is an immotile, host-adapted, Gram-negative bacillus that does not survive for extended periods of time in the environment outside of its natural equine reservoir. The organism causes the highly fatal zoonotic disease glanders, which primarily affects horses, mules, and donkeys. Other animal species and humans are susceptible to B. mallei infection, and the organism is readily transmitted via punctured skin and the inhalation of contaminated aerosols upon contact with tissues and biofluids from diseased equids. Glanders was eradicated from North America and Western Europe through systematic large-scale culling of infected animals during the first half of the 20th century. However, areas of endemicity still exist in many parts of the world such as Central and South America, the Middle East, Asia, and Africa. Infected animals can be asymptomatic but remain extremely contagious, which enables persistence of B. mallei in the equine population. In addition, the organism is difficult to identify due to lack of reliable modern diagnostic tools. For these reasons, glanders is considered a significant biosafety and biosecurity risk and is reportable to the World Organization for Animal Health. 2,7,11,12,17,18,21 –23,30,31,33,42,43,48,50,51
The clinical progression and pathology of glanders in animals and humans are similar, although the presentation of any 2 cases in the same species, even those resulting from direct transmission, can differ substantially. The most common manifestations are life-threatening pneumonia and bacteremia. Other clinical signs may initially include fever; myalgia; swollen and painful joints; prostration; headache; swelling of the face, limbs, and lymph nodes; increased respiratory effort; and development of painful nodules involving the face, arms, and legs. Later in the course of the disease, signs may progress to inappetence, diarrhea, weight loss, mucopurulent nasal and/or ocular discharges, and necrosis of the tracheobronchial tree. Pustular skin lesions, and development of abscess-forming ulcerative nodules known as farcy, are also common on the arms, legs, and abdomen. 2,7,12,17,18,21,23,34,39,43,45,48,50,51 In humans, glanders is almost always fatal if untreated. Even with aggressive antimicrobial therapy, the mortality rate nears 50%. Response to treatment is slow, elimination of the infection is difficult, and patients are prone to debilitating bouts of remission and exacerbation. Burkholderia mallei is also resistant to many antibiotics, which restricts therapeutic options. 20,29,36,47,49 Treatment of glanderous animals in nonendemic countries is prohibited, and strict quarantine and cull reportable programs are enforced.
The pathogenicity of B. mallei is complex and involves the coordinated expression of a large number of virulence factors that support extracellular and intracellular replication of bacteria as well as the seeding of deep tissues. 6,10,15,16,26,40,54 The organism uses type 3 and type 6 secretion systems to inject its own effector proteins inside host cells and exploit eukaryotic cellular functions. Once internalized, B. mallei escapes endocytic vacuoles and enters the cytoplasm of infected host cells to replicate. The bacilli then recruit and polymerize host actin to one of their poles, forming tails that propel bacteria from one cell to another. Burkholderia mallei can infect many different cell types in this manner including epithelial and dendritic cells, macrophages, and neutrophils. This ability to thrive intracellularly helps protect bacteria from host immune responses and antibiotics, and it aids in dissemination to target organs where the organism forms chronic lesions and granulomas that are difficult to treat. Hallmark histopathologic features include pyogranulomatous lesions composed of a central core of degenerate neutrophils covered by fibrinohemorrhagic exudate, which can mature to consist of a core of liquefactive or caseous necrosis, surrounded by epithelioid macrophages, multinucleated giant cells, lymphocytes, and granulation tissue. Glanderous lesions develop primarily in the liver, spleen, lungs, and lymph nodes, but they can also be found in any tissue throughout the body.
Although natural cases of glanders are uncommon in humans due to control measures, B. mallei represents a bona fide global health risk due to its prior (and repeated) use in biological warfare. 1,7,27,38,44,50,52,53 Glanders was involved in the first modern use of microbes as weapons during World War I when German agents targeted equids and other livestock in the United States, Romania, Russia, France, and Mesopotamia, which affected movement of Allied troops. During World War II, the Imperial Japanese Army also intentionally infected equids, civilians, and prisoners of war with B. mallei at the Ping Fan Institute in Manchuria. For these reasons, the US Federal Select Agent Program classifies the organism as a Tier 1 Select Agent, and the development of medical countermeasures is a pressing need as no vaccine currently exists and treatment options are inadequate.
With this in mind, our group previously identified the protein BpaB as a potential target for devising pretreatments and/or therapies. 55 We demonstrated that BpaB is located on the surface of B. mallei bacteria and is therefore readily accessible for recognition by the host immune system. We showed that the protein mediates adherence to human lung epithelial cells and promotes the formation of biofilms in vitro, which are key mechanisms of bacterial pathogenicity. Furthermore, we discovered that a mutant lacking expression of BpaB is attenuated in virulence using a mouse model of aerosol infection. Hence, BpaB displays many properties of an excellent candidate for developing countermeasures, and targeting the protein may interfere with B. mallei’s ability to establish itself and persist in the host, which in turn could reduce the risk of infection. While characterizing BpaB, we also made the discovery that the protein is not produced in vitro by wild-type B. mallei bacilli cultured in the laboratory, but it is expressed in vivo during infection, as evidenced by the presence of BpaB-specific antibodies in murine, horse, and human serum samples. The goal of the present study was to develop an immunostaining method and directly investigate the selective in vivo production of BpaB by B. mallei.
Materials and Methods
Strains, Plasmids, Cell Line, and Growth Conditions
The B. mallei strains and plasmids used in this study have been described in detail elsewhere. 35,55 Briefly, B. mallei ATCC 23344 is a wild-type isolate from a human patient who died of glanders. Burkholderia mallei bpaB KO is an isogenic mutant strain of B. mallei ATCC 23344 in which the genomic copy of the bpaB gene has been inactivated with a Zeocin antibiotic resistance cassette. The plasmid pBpaB encodes a kanamycin antibiotic resistance marker and contains a cloned, intact copy of the bpaB gene; the B. mallei bpaB KO mutant strain carrying this construct constitutively produces the BpaB protein. The plasmid pBHR1ΔDra, also encoding the kanamycin resistance marker, is a vector control that does not contain a cloned DNA insert, and B. mallei bpaB KO carrying this plasmid does not produce the BpaB protein.
Burkholderia mallei strains were cultured at 37°C using Brucella medium (BD) supplemented with 5% (vol/vol) glycerol and 1.5% (wt/vol) Bacto agar (Becton, Dickinson and Company). The medium used to grow the B. mallei bpaB KO mutant carrying plasmids pBpaB and pBHR1ΔDra was also supplemented with the antibiotics Zeocin (Life Technologies) and kanamycin (MP Biomedicals) at final concentrations of 7.5 µg/ml and 5 µg/ml, respectively.
The cell line J774A.1 (murine macrophages; ATCC TIB-67) was cultured in DMEM medium (Corning) supplemented with 10% (vol/vol) fetal bovine serum (HyClone) at a temperature of 37°C and atmosphere containing 7.5% CO2, as outlined by others. 3
In Vitro Experiments With J774 Murine Macrophages
Bacteria were grown on agar medium for 40 hours and suspended in phosphate-buffered saline (PBS) to a concentration of 109 colony forming units/ml. The suspensions were used to infect monolayers of J774 murine macrophages seeded into the wells of Upcell 24-well tissue culture plates (Nunc) at a multiplicity of infection of 10 bacteria to 1 macrophage; control wells were inoculated with PBS. The tissue culture plates were then centrifuged (5 minutes, 165 × g) and incubated for 10 hours at 37°C to maximize intracellular replication of bacteria. Following this, the monolayers were washed twice and the plates were transferred to 25°C for 10 minutes, after which the macrophages were dissociated from the plastic support of the wells and collected in suspension. Aliquots were lysed with PBS containing 0.5% saponin (wt/vol) and serial dilutions were spread onto agar plates to enumerate bacteria and verify intracellular replication of the B. mallei strains. The remaining macrophages were pelleted by centrifugation (5 minutes, 165 × g) and fixed for 24 hours in 50 ml of 10% (vol/vol) neutral-buffered formalin (Fisher Chemical). After fixation, the macrophages were pelleted by centrifugation (5 minutes, 165 × g) and embedded in 3% (wt/vol) agarose. These agarose cell pellets were bisected, placed in histology cassettes (Leica BIOSYSTEMS), subjected to automated tissue processing, embedded in paraffin, and then cut into 3-µm-thick sections for immunohistochemical analysis.
Tissues From Experimentally Infected Animals
Archival tissue sections from n = 2 BALB/c mice and n = 5 marmosets (Callithrix jacchus) infected with the B. mallei wild-type strain ATCC 23344 (and matching uninfected control animals) were generated in the context of previously published studies. 19,25 Briefly, the tissues were submersed in 10% neutral-buffered formalin for 1 to 3 weeks, dissected, and processed into 3-µm-thick sections as described above.
Antibodies
BALB/c mouse immune serum against whole B. mallei organisms was produced in a previous study 25 and consisted of pooled serum from mice infected via the aerosol route with the B. mallei wild-type strain ATCC 23344. The BpaB-specific murine IgG monoclonal antibody BpaB-MAb#4 has been described elsewhere and is commercially available (Kerafast, catalog number EGA074). 55 Naïve BALB/c mouse serum was purchased from Charles River Laboratories. These antibody probes were used in immunohistochemical analyses at concentrations of 0.5% (vol/vol) for the naïve and immune mouse sera and 10% (vol/vol) for the BpaB-MAb#4 monoclonal antibody.
Immunohistochemical Analyses
Sample sections (J774 macrophages, mouse tissues, marmoset tissues) were mounted onto positively charged glass slides, deparaffinized at room temperature by soaking overnight in xylene (EMD Millipore), and immersed in a series of room temperature baths (100% ethanol for 5 minutes, 95% ethanol for 5 minutes, 75% ethanol for 5 minutes, double distilled water for 5 minutes). Antigens were then unmasked by incubation in a 10-mM sodium citrate buffer (pH 6.2) for 10 minutes at a temperature of 120°C and a pressure of 10 psi. The samples were washed 3 times with Tween-Buffered Saline (TBS; Dako Agilent Technologies), blocked for 10 minutes at room temperature using TBS supplemented with 10% (vol/vol) normal goat serum (Sigma-Aldrich), washed 3 times with TBS, and then probed for 16 hours at 4°C with naïve mouse serum, mouse immune serum against whole B. mallei organisms, or the mouse monoclonal antibody BpaB-MAb#4 diluted in Antibody Diluent reagent (Dako Agilent Technologies). Next, the samples were washed 3 times with TBS to remove excess probes and incubated at room temperature for 1 hour with a goat anti-mouse IgG (H+L) antibody conjugated to alkaline phosphatase (Dako Agilent Technologies) diluted 1:500 in Antibody Diluent reagent. After this incubation, the slides were washed 3 times with TBS to remove unbound goat anti-mouse antibody and incubated for 2 to 10 minutes at room temperature with Vulcan Fast Red solution per the manufacturer’s recommended procedure (Biocare Medical). Following this, the samples were washed 3 times with double distilled water to remove excess chromogen, counterstained at room temperature for 2 minutes with hematoxylin (Sigma-Aldrich), dipped 5 times in Bluing reagent (Sigma-Aldrich), immersed in a series of room temperature baths (75% ethanol for 1 minute, 95% ethanol for 1 minute, 100% ethanol for 1 minute, xylenes for 2 minutes), and cover-slipped for microscopic examination. Histologic evaluations were performed by board-certified veterinary pathologists using an Olympus BX41 microscope (Olympus Corporation), and images (40× and 100× magnification) were captured with an Olympus DP71 camera and software (Olympus Corporation).
Research Compliance Statement
All experiments with live B. mallei were performed inside a Class II Biosafety Cabinet in a BSL3 laboratory and in compliance with the rules and regulations of the US Federal Select Agent Program. The University of Georgia’s Institutional Biosafety Committee approved all experiments prior to initiation. Archived animal tissues were obtained from previously published studies, which were performed with approval from the University of Georgia’s Institutional Animal Care and Use Committee. All animal experiments were carried out in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals by the National Institutes of Health.
Results
Validation of Antigen-Retrieval and Immunostaining Methods
Our group previously developed an immunofluorescence staining approach to visualize B. mallei in tissues of experimentally infected animals using immune mouse serum against the whole organism. 19 In the current studies, we expanded the procedure to detect B. mallei by immunostaining with the chromogen Vulcan Fast Red and examine in vivo production of the virulence factor BpaB by probing infected tissues with a monoclonal antibody that specifically recognizes the protein.
Prior to analyzing tissue sections of infected animals, we validated the antibody probes and staining reagents using murine J774 macrophages infected in vitro with a panel of B. mallei wild-type and recombinant mutant strains. Upon infection, macrophages were incubated for 10 hours to maximize intracellular replication of bacteria. The cells were then fixed in formalin, paraffin-embedded, and sectioned for immunohistochemical staining in order to mimic the process used to prepare tissue samples from infected animals and optimize antigen-retrieval conditions.
Immune mouse serum against whole B. mallei organisms identified bacilli within macrophages infected with recombinant bacteria constitutively producing BpaB (Fig. 1), a mutant lacking expression of the protein (Fig. 4), and the wild-type strain ATCC 23344 (Fig. 7). In contrast, the infected macrophages did not react with naïve mouse serum (Figs. 2, 5, 8), and control uninfected macrophages did not show reactivity with either immune (Fig. 10) or naïve (Fig. 11) serum. When probed with a murine monoclonal antibody specific for BpaB, only J774 cells infected with recombinant bacteria constitutively producing the BpaB protein showed immunoreactive intracellular bacilli (Fig. 3). Although macrophages infected with the mutant lacking expression of the bpaB gene product and macrophages infected with wild-type B. mallei clearly contain bacteria that react with immune serum against the whole organism (Figs. 4, 7, respectively), these intracellular bacilli did not show reactivity with the BpaB-specific monoclonal antibody (Figs. 6, 9, respectively). The lack of reactivity of macrophages infected with the mutant was expected, as this strain contains an inactivated copy of the bpaB gene in its genome. The lack of reactivity of macrophages infected with the wild-type strain ATCC 23344, which specifies an intact bpaB gene, is consistent with our previously published findings that the BpaB protein is not produced at detectable levels by the organism cultured under any in vitro conditions tested to date. 55 Taken together, these data demonstrate the sensitivity and specificity of our antibody probes and staining reagents and establish our ability to detect B. mallei bacteria that produce the virulence factor BpaB in formalin-fixed samples.
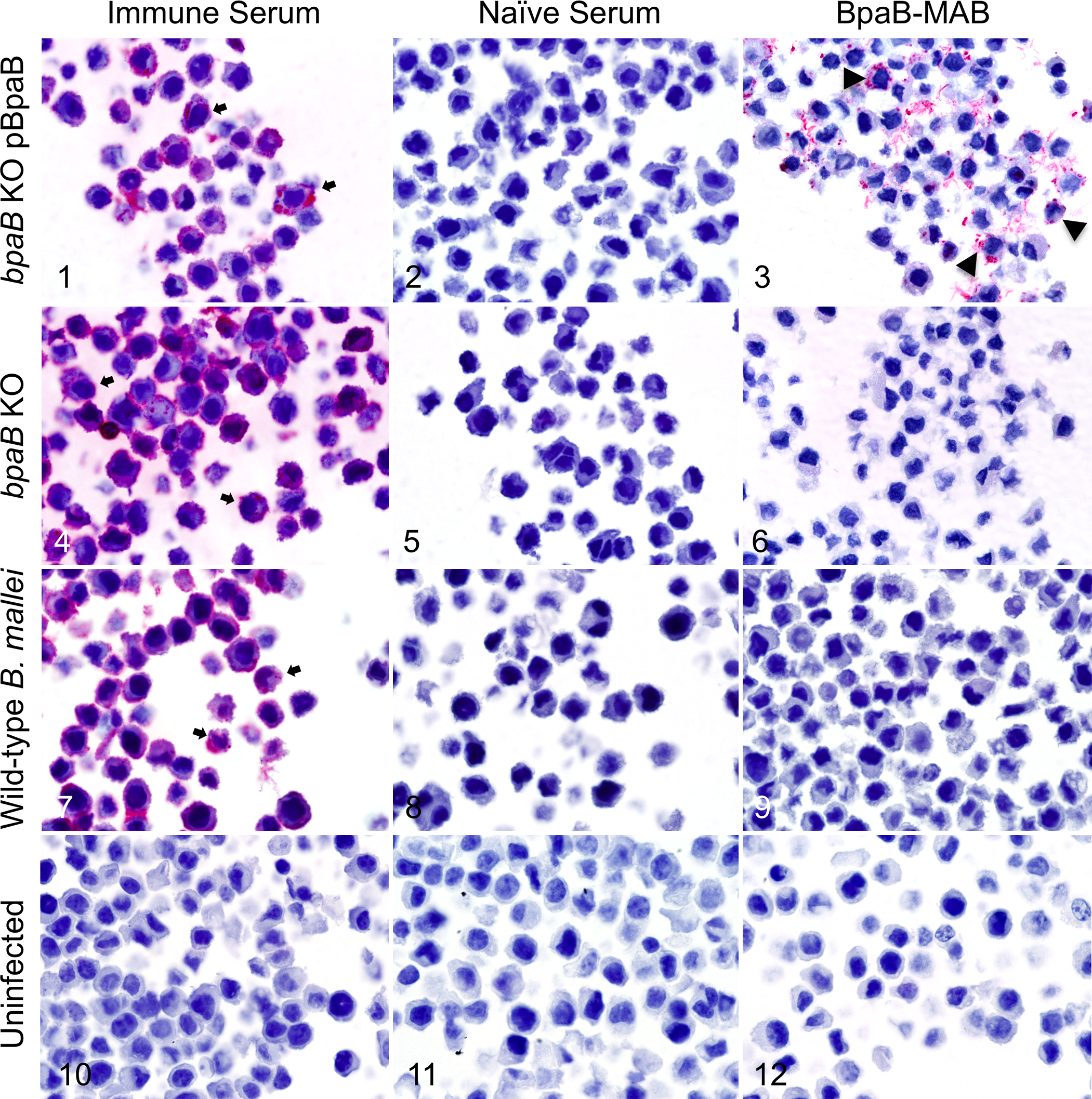
Immunohistochemical analysis of J774 murine macrophages infected in vitro with wild-type (WT) and recombinant mutant strains of B. mallei.
Immunohistochemical Examination of Tissues From Infected Mice
Aerosol infection is one of the most common routes of inoculation in naturally occurring cases of glanders, and the lung is a likely portal of entry for B. mallei in the event of adversarial use. The current benchmark animal model in the field to study the disease and evaluate pretreatments or therapies is the mouse, especially BALB/c and C57BL/6 strains. 9,13,14,16,28,32,46 The model produces hallmarks of glanders (low infectious and lethal doses, rapid replication of the agent in the lungs and dissemination to target organs [lymph nodes, liver, and spleen] where they form chronic lesions, and bacteremia), and infected mice produce antibodies against antigens that are known to be targets of the human and equine immune responses, thus demonstrating immunological parallels and relevance. 24,25,55 Our group has developed a noninvasive and highly reproducible inoculation method to study aerosol infection by B. mallei. 24,25,55 The model entails the use of a handheld nebulizer to deliver bacteria directly into the lungs. The device, termed Microsprayer, generates aerosols from the tip of a thin tube that is attached to a high-pressure stainless steel syringe containing the agent. A modified pediatric otoscope is used to introduce the tube inside the trachea of anesthetized mice, and a 50-μl volume of bacterial suspension is aerosolized in the airways.
With this in mind, we used our validated Vulcan Fast Red immunostaining method to analyze archival tissue sections from BALB/c mice infected with the wild-type B. mallei strain ATCC 23344 via the Microsprayer platform. The samples from n = 1 uninfected control and n = 2 infected animals were probed with immune mouse serum against the whole organism (to visualize B. mallei in tissues), naïve mouse serum (negative control), and the BpaB-specific murine monoclonal antibody BpaB-MAb#4 (to detect B. mallei bacteria that produce BpaB). The findings are summarized in Table 1 and representative images are shown in Figs. 13 to 16.
Vulcan Fast Red Immunostaining of Archival Tissue Samples From BALB/c Mice Infected With the Wild-Type B. mallei Strain ATCC 23344.a
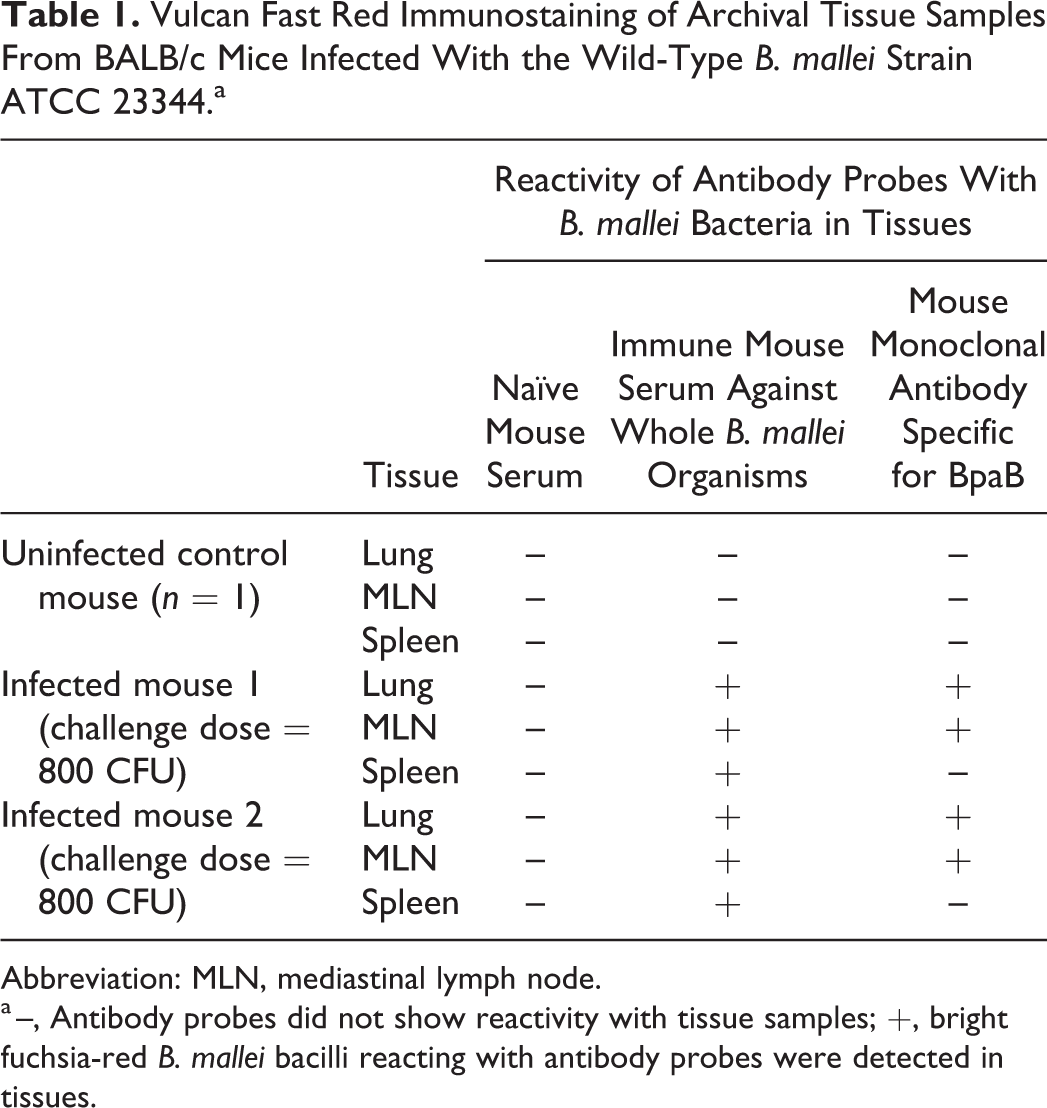
Abbreviation: MLN, mediastinal lymph node.
a –, Antibody probes did not show reactivity with tissue samples; +, bright fuchsia-red B. mallei bacilli reacting with antibody probes were detected in tissues.
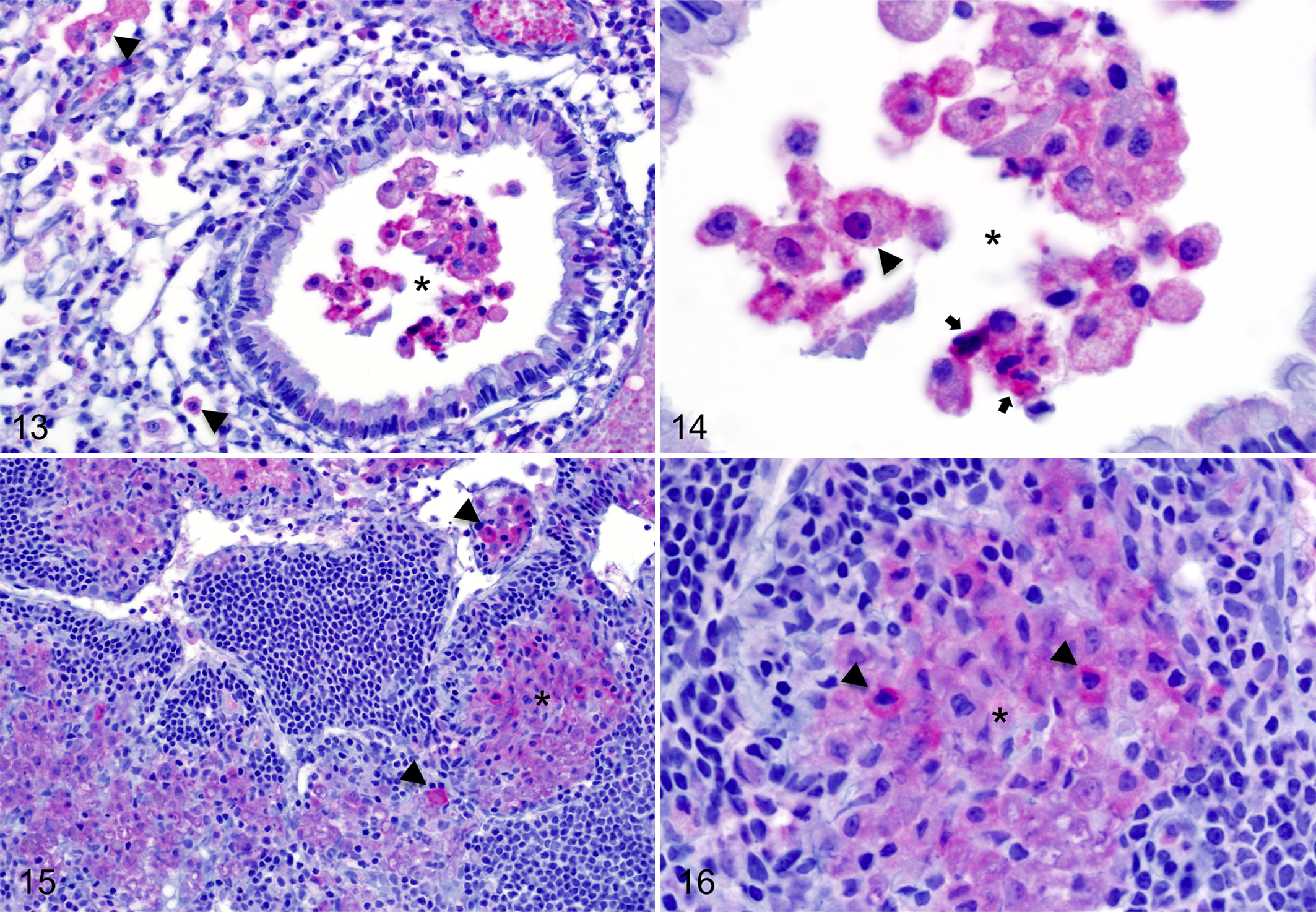
Immunohistochemical analysis using BpaB-specific mouse monoclonal antibody (BpaB-MAb#4) in tissues from BALB/c mice infected with wild-type B. mallei strain ATCC 23344 (challenge dose = 800 CFU).
The lungs of the uninfected control mouse consisted of histologically unremarkable bronchi, bronchioles, alveoli, and their associated blood vessels. Likewise, the mediastinal lymph node and spleen sections demonstrated normal tissue architecture. As expected, the uninfected control samples did not show reactivity with any of the antibody probes (Table 1).
Target organs from mice infected with B. mallei ATCC 23344 demonstrated lesions consistent with glanders. Pulmonary lesions consisted of variably sized, necrotic, parenchymal pyogranulomas. Burkholderia mallei bacteria expressing BpaB were found predominantly within macrophages and neutrophils in the airway and alveolar lumens (2/2 mice; Figs. 13, 14) and in the cytoplasm of epithelial cells that line the mucosa of bronchioles and alveoli (2/2 mice; data not shown). Extracellular bacilli reacting with the BpaB-specific monoclonal antibody were also observed in the lumen of blood vessels (2/2 mice; Fig. 13), associated with the surface of bronchiolar and alveolar respiratory epithelial cells (2/2 mice; data not shown), and in both unremarkable and inflamed alveolar spaces (2/2 mice; data not shown).
Change accepted. Mediastinal lymph nodes were disrupted by either large, discrete, necrotic pyogranulomas or by multifocal to coalescing regions of granulomatous inflammation, both containing intralesional B. mallei bacteria (2/2 mice; Table 1). Bacilli expressing BpaB were found within macrophages and, to a lesser extent, neutrophils (2/2 mice; Figs. 15, 16). Similar to the lungs, the infected spleens were littered with large, necrotic pyogranulomas. Although extracellular and intracytoplasmic B. mallei bacteria reacting with immune serum against the whole organism were detected in splenic tissue sections, these bacilli did not show reactivity with the BpaB-specific monoclonal antibody BpaB-MAb#4 (2/2 mice; Table 1). Taken together, the data demonstrate that the virulence factor BpaB is produced by wild-type B. mallei bacteria in target organs of mice during infection and that the protein is expressed in some, but not all, diseased tissues.
Immunohistochemical Examination of Tissues From Infected Marmosets
Murine models have been instrumental to our understanding of pathogenicity by B. mallei and provide an excellent platform for the development of medical countermeasures (ie, identification and characterization of high value targets). However, due to the organism’s classification as a Tier 1 Select Agent and the nature of glanders infection, it is unlikely that licensure of products can be achieved exclusively through traditional Phase III clinical trials in individuals at risk of exposure. Burkholderia mallei is extremely infectious via the aerosol route, causes severe clinical symptoms, and is resistant to most antibiotics. Furthermore, glanders is almost always fatal if untreated and requires prolonged therapy with low success rates in eliminating the infection. For these reasons, the FDA Animal Rule is the most relevant path for licensing countermeasures against B. mallei and will rely on the use of additional, nonrodent models of human glanders to validate lead candidates. To address this, our group developed a model of intranasal B. mallei infection using the common marmoset (Callithrix jacchus) and demonstrated that this nonhuman primate develops an illness similar to that described in other established models for the organism. It is important that the disease onset, progression, and lesions in the marmoset resembled that reported for human glanders. 19
With this in mind, we used our panel of murine antibodies to probe archived tissue sections from uninfected (n = 2) and infected (n = 5) marmosets and immunostained the samples with Vulcan Fast Red. The nasopharynx, lungs, liver, and spleen of uninfected control animals were within normal limits and did not react with any of the antibody probes (2/2 marmosets; Table 2). In contrast, multiple tissues from marmosets infected with B. mallei ATCC 23344 showed lesions characteristic of glanders, and these organs all contained intralesional bacilli reacting with immune serum against whole B. mallei organisms (5/5 marmosets; Table 2). The lesions in the lungs ranged from small, scattered, foci of necropurulent or pyogranulomatous inflammation to severe necropurulent bronchopneumonia. Bacilli reacting with the BpaB-specific monoclonal antibody BpaB-MAb#4 were detected primarily in the cytoplasm of respiratory epithelial cells in the bronchi (3/5 marmosets; Fig. 17) and bronchioles (3/5 marmosets; data not shown), and within macrophages and neutrophils located in the parenchyma and alveolar spaces (3/5 marmosets; data not shown). Extracytoplasmic B. mallei bacteria producing BpaB were also observed inside pulmonary blood vessels (3/5 marmosets; data not shown), bound to the apical surface of bronchiolar and alveolar respiratory epithelial cells (3/5 marmosets; data not shown), and in the airway lumens of the bronchi, bronchioles, and alveoli where they aggregated, admixed with mucopurulent exudate (3/5 marmosets; Fig. 17).
Vulcan Fast Red Immunostaining of Archival Tissue Samples From Marmosets Infected With the Wild-Type B. mallei Strain ATCC 23344.a
Abbreviation: NP, nasopharynx.
a –, Antibody probes did not show reactivity with tissue samples; +, bright fuchsia-red B. mallei bacilli reacting with antibody probes were detected in tissues.
b The tissue samples were generated in the context of a previously published study and the numbers correspond to unique identifiers used for the marmosets while conducting the study.
The lesions in nasopharyngeal tissues consisted of necrosis of the mucosa, purulent to pyogranulomatous rhinitis, and large amounts of mucopurulent exudate within the nasal cavity. As noted for the lungs, the exudate in the nasopharynx was frequently intermixed with clusters of extracellular B. mallei bacteria reacting with the BpaB-specific antibody probe (4/5 marmosets; data not shown). Bacilli producing BpaB were also detected attached to the nasopharyngeal mucosa, inside the cytoplasm of ciliated nasopharyngeal respiratory epithelial cells (4/5 marmosets; Fig. 18), in the lumen of blood vessels (4/5 marmosets; Fig. 18), and within macrophages in lymphoid tissues associated with the mucosa of the nasal cavity (4/5 marmosets; Fig. 19).
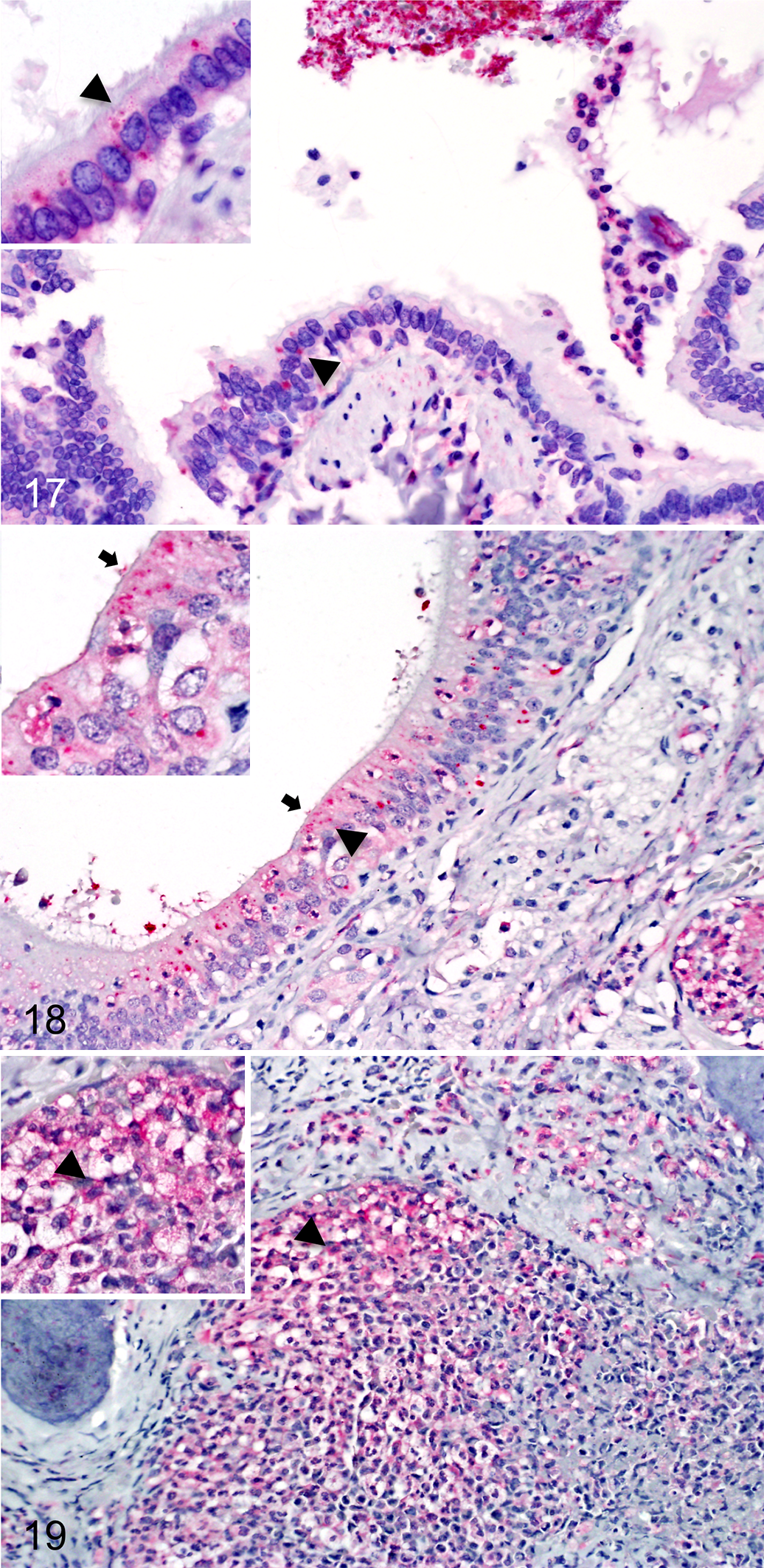
Immunohistochemical analysis using BpaB-specific mouse monoclonal antibody (BpaB-MAb#4) in tissues from marmosets infected with the wild-type B. mallei strain ATCC 23344.
Lesions in the spleen consisted of small, scattered microabscesses and pyogranulomas, whereas lesions in the liver ranged from scattered microabscesses to multifocal necrosis. In both the liver and spleen, intralesional bacteria were detected with immune serum against whole B. mallei organisms (5/5 marmosets; Table 2). However, these bacilli did not show reactivity with the BpaB-specific monoclonal antibody (5/5 marmosets; Table 2). Based on these findings, we conclude that BpaB is produced by wild-type B. mallei ATCC 23344 colonizing target organs of marmosets and that expression of the protein varies in individual tissues.
Discussion
This study demonstrates in vivo production of the virulence factor BpaB by wild-type B. mallei bacteria upon respiratory infection of mice and marmosets, 2 relevant animal models to study glanders. We also report development of an immunohistochemistry staining method for not only detecting the presence of B. mallei organisms in host tissues but also visualizing in situ expression of an individual antigen. In accordance with previously published guidelines for developing and validating immunohistochemistry techniques, our method is highly specific and sensitive for distinguishing BpaB from other molecules expressed by bacteria and host cells. 37
The ability to survive and replicate inside host cells is a key aspect of pathogenesis by B. mallei, and macrophage infection assays represent a benchmark in vitro model system to study virulence of the organism. 4 –6,8,15,16,26,40,41 With this in mind, we discovered that wild-type B. mallei bacilli do not produce detectable amounts of the BpaB protein during in vitro infection of murine macrophages (Fig. 9). However, immunostaining of infected tissues from mice (Figs. 13–16) and marmosets (Figs. 17–19) using the BpaB-specific monoclonal antibody probe revealed that BpaB is produced by B. mallei bacteria within host cells including macrophages, neutrophils, and epithelial cells. These data underscore our previously published discovery that BpaB is not noticeably expressed by the organism grown under any laboratory conditions tested to date and demonstrate selective in vivo production of the protein during infection. 55 The presence of extracellular BpaB+ bacilli in diseased tissues (Figs. 13, 17, 18) also suggests that the intracellular niche is not essential for upregulating expression of the protein in vivo.
The ability to colonize mucosal tissues of the airways is another key aspect of pathogenesis by B. mallei. Airborne transmission is one of the most common routes of infection in natural cases of glanders, and the lung is a likely portal of entry if the organism were to be used during a biological attack. Burkholderia mallei adheres to and invades the respiratory epithelial cells in the airway, prior to disseminating directly into the bloodstream and/or indirectly via phagocytosis by macrophages and dendritic cells, which then traffic to regional lymphoid tissues (draining lymph nodes, MALT). Pyogranulomatous inflammation ensues in these lymphoid tissues, and B. mallei further disseminates by draining into the thoracic duct for redistribution via leukocytes trafficking in lymph and/or bacteremia, which ultimately leads to the seeding of distal organs such as the spleen and liver where the organism forms lesions and granulomas that are difficult to treat. Previous work characterizing BpaB suggests that the protein contributes to colonization of the respiratory airways, as it was shown to mediate adherence to human alveolar epithelial cells (A549, ATCC® CCL-185™) and to promote the formation of biofilms using in vitro model systems. 55 Our in vivo findings confirm these prior studies by demonstrating that B. mallei bacilli attached to the apical surface of airway epithelial cells produce BpaB (Fig. 18) and that bacteria expressing the protein form large aggregates akin to biofilms in the respiratory tract (Fig. 17). In addition, BpaB+ bacilli were observed in the cytoplasm of airway epithelial cells and professional phagocytes (Figs. 13, 14, 17, 18), in the lumen of blood vessels of the lungs (Fig. 13) and nasopharynx (Fig. 18), and in inflamed regional lymphoid tissues (mediastinal lymph nodes in Figs. 15, 16; nasopharyngeal MALT in Fig. 19). Taken together, these data indicate that BpaB represents a potentially high value antigen for developing countermeasures against respiratory infection. Targeting the protein during acute infection may interfere with disease progression by blocking B. mallei adherence to the mucosa of the airways and/or promoting opsonophagocytic clearance of the organism before bacteria disseminate to deep tissues.
In addition to supporting a role for BpaB in colonization of the respiratory tract, our in vivo findings revealed that the protein does not appear to be necessary for bacterial fitness in distal organs. Although the spleen and liver of infected mice and marmosets were clearly colonized with B. mallei bacteria reacting with immune serum against the whole organism, these bacilli did not express BpaB (Tables 1, 2). To our knowledge, this is the first report demonstrating not only selective in vivo production but also differential expression of a B. mallei virulence protein at distinct anatomic foci of infection. The molecular mechanisms and factors driving in vivo production of BpaB are currently being investigated, as this will provide novel insights in the complex biology and host–pathogen interactions during glanders and will yield useful information for the development of countermeasures and diagnostics for B. mallei.
Footnotes
Acknowledgements
We thank Dr Elizabeth Howerth, Deb Carter, and Patricia Rowe (University of Georgia) for their invaluable advice regarding the development of immunohistochemical techniques, the Histology Laboratory (University of Georgia) for their assistance with tissue processing, and Drs Kaori Sakamoto (University of Georgia), Teresa Shaffer (University of South Alabama), and Jennifer Willingham-Lane (University of Georgia) for their help reviewing this manuscript.
Declaration of Conflicting Interests
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported, in part, by the National Institutes of Health National Center for Research Resources (05 T35 OD010433-09) to MEL and by a Zoetis-Morris Animal Foundation Veterinary Fellowship for Advanced Study (D12ZO-902) to SMZ. The funders had no input in the study design, data collection and analysis, the decision to publish, or the preparation of the manuscript.
