Abstract
Six 3-month-old BALB/c Rag2-/- mice developed dyspnea 10 days after intravenous injection of wild type BALB/c CD45RB(high) lymphocytes to induce colitis as a model of inflammatory bowel disease. The lungs of all 6 mice were diffusely gray-purple and did not collapse completely. Microscopic findings were extensive coalescent patchy to diffuse alveolitis, characterized by macrophages and multinucleate giant cells, lymphocytes in alveolar lumina and septa, alveolar luminal of neutrophils, and alveolar proteinic material containing small black vesicular bodies characteristic of Pneumocystis sp. in methenamine silver stained sections. The morphologic diagnosis was diffuse granulomatous pneumonia with intra-alveolar organisms consistent with Pneumocystis sp., with an unusually aggressive inflammatory response related to the experimental procedure and possibly to the BALB/c genetic background.
Keywords
Background
In most cases, information about a research model readily is obtained by searching PubMed (http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed) for the investigator's name, research topic, or both. The search statement ‘“Inflammatory Bowel Diseases”[Mesh] AND “Antigens, CD45”[Mesh]’ produces citations from which one finds that colitis is induced in severe combined immune-deficient (SCID) mice or recombinase-activating gene (Rag1 or Rag2) knockout mice by transfer of CD45RB(high) T cells from immunocompetent, histocompatible donor mice. CD45 functions in regulation of T-cell activation, and different CD45 isoforms are expressed to different degrees in different T-cell subsets. High expression of CD45RB is characteristic of CD4+ effector cells that, in the absence of regulatory T cells, induce colitis in response to commensal intestinal bacteria. 7
Inbred mouse strains, and even substrains, have important phenotypic differences that can contribute to the observed disease problem. Information about the phenotypes of mouse strains can be obtained from sources such as Festing's Inbred Strains of Mice on The Jackson Laboratories Mouse Genome Informatics web site (http://www.informatics.jax.org/) and Brayton's descriptions of disease susceptibilities of common mouse strains. 1 According to these sources, BALB/c mice in general are highly susceptible to a variety of infectious agents. Obtaining information about strain characteristics via PubMed searching is somewhat more difficult, but with some experimentation with different search strategies, it usually is possible to obtain a list that is both informative and manageable.
The mutant phenotype can be obtained similarly. However, a frequently encountered problem is that multiple mutant alleles exist that confer different phenotypes, depending on such factors as whether the mutant gene is tissue or developmentally specific or whether a partial gene product is produced, but the specific mutation present in the submitted mice is not identified in the history one is initially given, as in this case. Searching the Mouse Genome Informatics web site for Rag2 alleles, one finds 3 different knockout alleles listed. From purchase records showing that the mice had been obtained from Taconic and listing the catalog number, and in turn from the Taconic web site (http://www.taconic.com/), the genotype of the mice was more precisely established as C.129S6(B6)-Rag2tm1Fwa N12, a congenic line established by backcrossing the Rag2tm1Fwa mutant allele from B6;129S6 mixed background mice onto the BALB/cAnNTac strain to the N12 generation. With the mutant Rag2 allele identified as Rag2tm1Fwa , one can then refer to the Mouse Genome Informatics web site for specific information about it. In this case, it is sufficient to know that loss of function of this gene confers a severe combined immunodeficiency phenotype.
History, gross findings, and laboratory results
Six 3-month-old mice of both sexes and identified as BALB/c Rag2–/– were submitted for diagnosis. The mice had been purchased from a commercial vendor. Two weeks previously, the mice were intravenously injected with wild-type BALB/c CD45RB(high) lymphocytes to induce colitis as a model of inflammatory bowel disease (IBD). The mice began to become ill 10 days after the injection. All 6 mice were lethargic and had rough coats, hunched posture, and rapid, deep respiratory efforts. The mice were euthanatized and subjected to postmortem examination. The lungs of all 6 were diffusely gray-purple, did not collapse completely, and were difficult to infuse with fixative (Fig. 1). There were no other gross lesions. No pathogens were identified in bacterial cultures of respiratory tract lavage samples. Health surveillance results for the specific pathogen–free (SPF) source colony, with the use of dirty bedding sentinels, were negative except for Helicobacter bilis and unidentified Helicobacter spp.
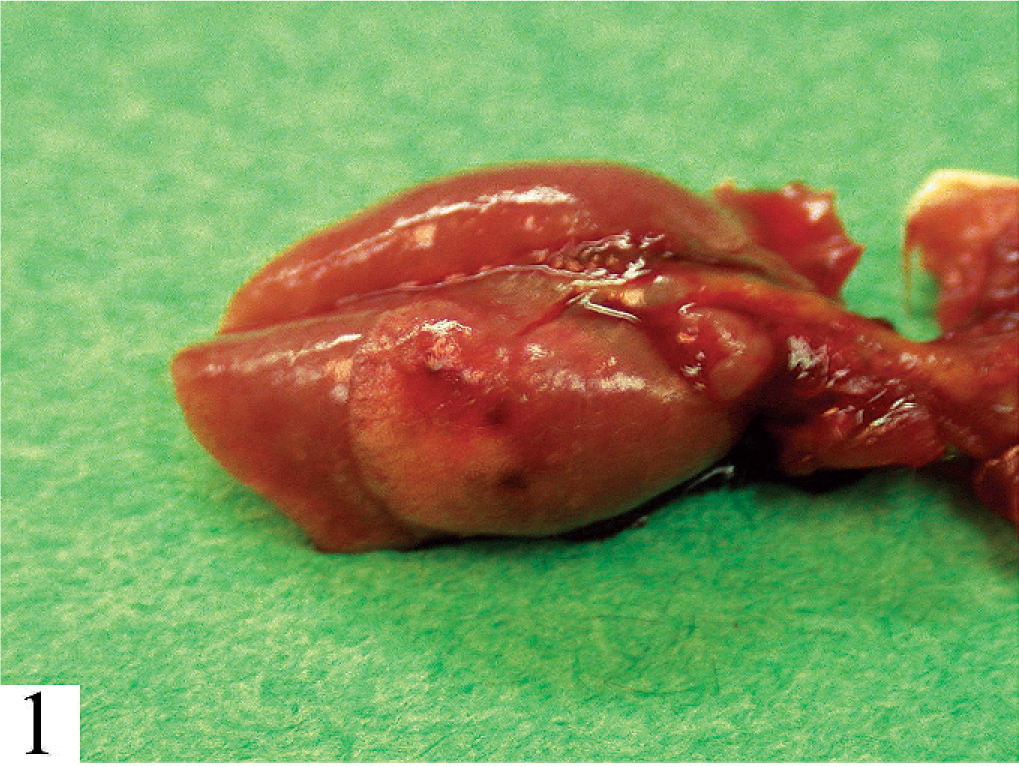
Lung. All lobes are diffusely gray-purple and only partially collapsed.
Differential diagnoses
The “classic” differential diagnoses for clinically evident pneumonia in mice include Mycoplasma pulmonis, cilia-associated respiratory (CAR) bacillus, and Sendai virus. 6 In immunodeficient mice, pneumonia virus of mice (PVM) and Pneumocystis murina also should be considered, as should the possibility of exacerbated disease because of combinations of pathogens such as M. pulmonis and Sendai virus in immunocompetent mice and PVM or Pasteurella pneumotropica with P. murina in immunodeficient mice. 6 The clinical and gross findings probably are most consistent with Sendai virus disease, but “classic” pathogens such as M. pulmonis, CAR bacillus, Sendai virus, and PVM rarely are found among contemporary SPF laboratory mice. Most instances of disease are associated with experimental treatment, genetic factors, or both.
Microscopic findings
Findings were identical in all 6 mice. The lungs contained extensive coalescent patchy to diffuse alveolitis, characterized primarily by macrophages and multinucleate giant cells, lymphocytes in alveolar lumina and septa, and alveolar luminal collections of neutrophils (Fig. 2). Also within alveoli were collections of proteinic material containing multiple, uniform, tiny basophilic dots within small clear spaces. Lymph nodes were populated with lymphoid cells of various sizes, but lacked cortex, medulla, and other lymph node structure. The thymus was composed mostly of stroma and vessels, with very few lymphocytes. Other organs and tissues were normal. In methenamine silver–stained sections of lung (Fig. 3), small black vesicular bodies characteristic of Pneumocystis sp. were evident. Morphologic diagnoses were diffuse granulomatous pneumonia with intra-alveolar organisms consistent with Pneumocystis sp. and thymic and lymph node hypoplasia consistent with partially reconstituted severe combined immunodeficient mice.
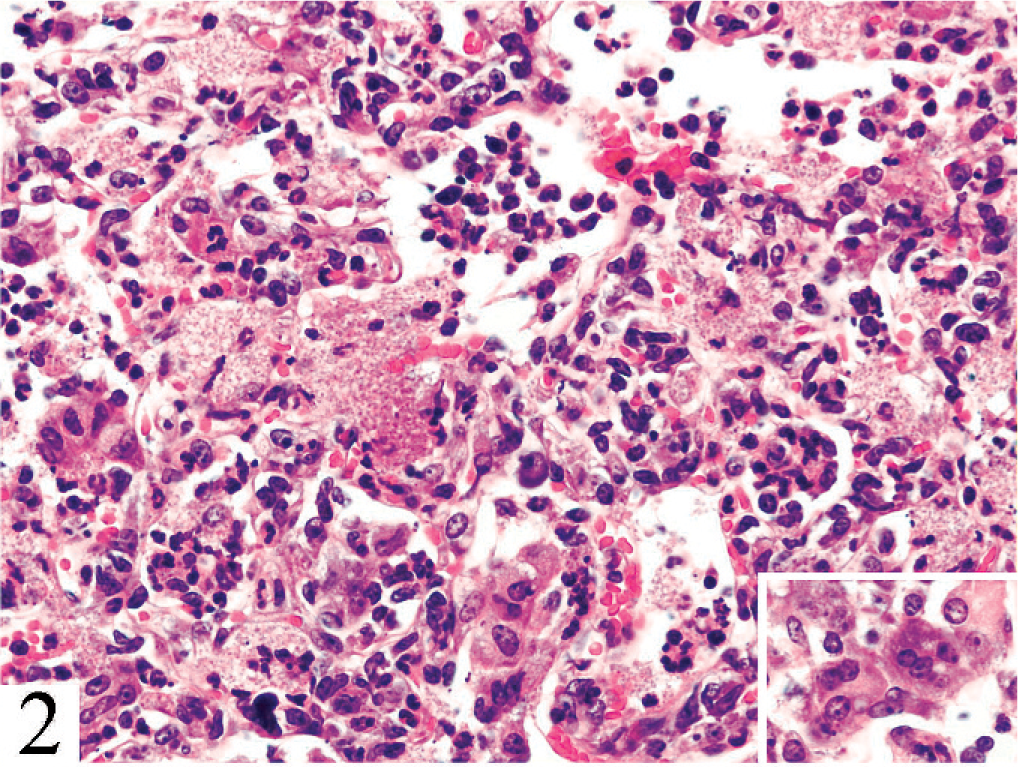
Lung. Alveolitis is characterized by macrophages, lymphocytes, neutrophils, multinucleate giant cells (inset), and granular proteinic material containing multiple tiny clear spaces. HE.
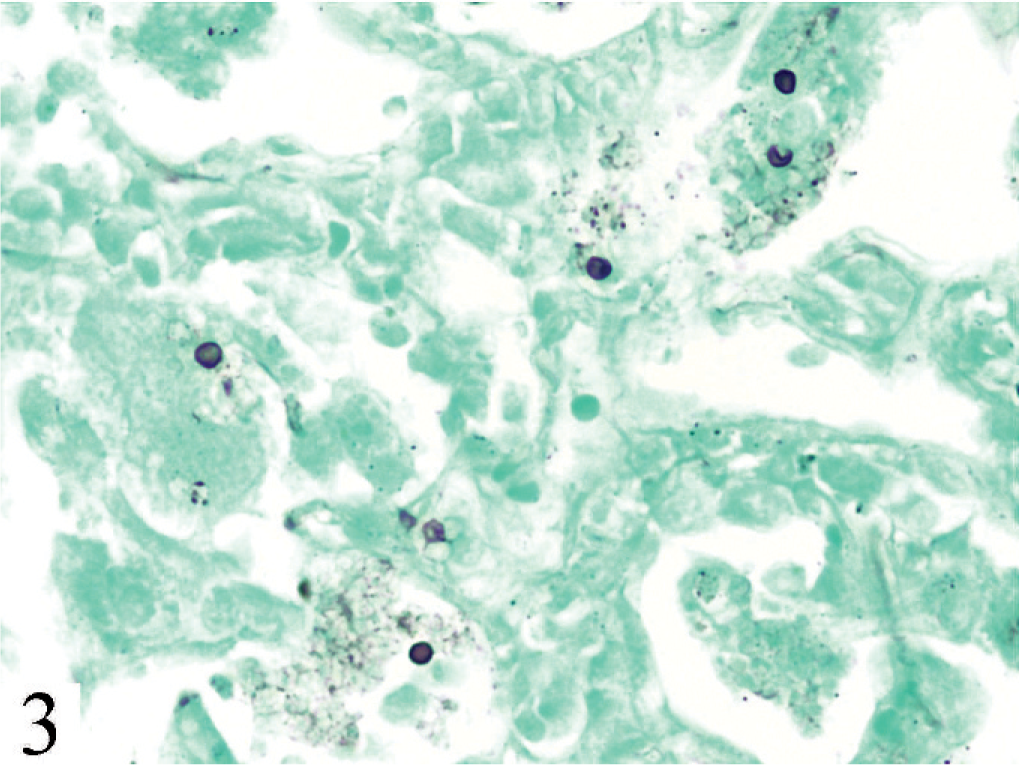
Lung. Vesicular bodies characteristic of Pneumocystis sp. Gomori methenamine silver.
Diagnosis
Pneumocystis pneumonia.
Discussion
Pneumocystis pneumonia is common in nude, SCID, and other severely immunodeficient mice, but this case is unusual in the rapid and simultaneous development of pneumonia in all of the mice and in the intensity and character of the inflammatory response compared with that typical of pneumocystosis in mice (Fig. 4). Because of the absence of necrotizing respiratory mucosal lesions characteristic of Sendai virus or PVM, negative results of respiratory tract bacterial cultures, and lack of evidence of CAR bacillus infection in silver-stained sections, we concluded that P. murina was the only pathogen present. (Subsequent health surveillance results for the colony also produced no evidence of viral infection.) Inasmuch as transfer of CD4+CD25− T cells into Rag2–/– mice known to be infected with P. murina induced similar fatal pneumocystosis, 5 we concluded that the observed disease was related to the experimental procedure, with the transferred CD45RB(high) cells inducing pneumonia in response to P. murina and with a possible contribution to increased susceptibility by the BALB/c genotype. The problem has not recurred, which could reflect that P. murina, rather than being widespread as a latent infection among mice, probably occurs as an active infection. 4
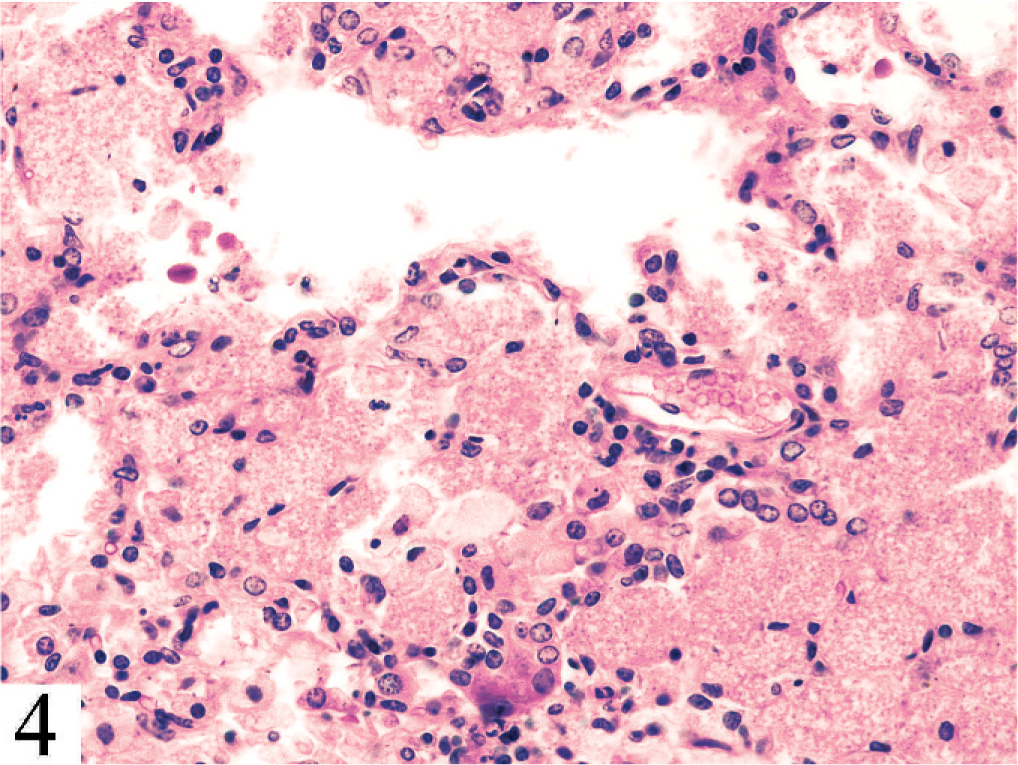
Lung. Alveolitis in spontaneous pneumocystis pneumonia in a C.B-17 Prkdcscid severe combined immunodeficiency mouse, with extensive intraalveolar accumulation of proteinic material and Pneumocystis murina organisms and mild inflammatory response. HE.
It is important to be aware of the role of Helicobacter spp. in mice used in IBD research. Helicobacter hepaticus, H. bilis, and other species can alter colitis expression in IBD models and can cause colitis in immunodeficient mice. 3 Because mice from the investigator's colony are used for experimental colitis studies, it is monitored by commercial polymerase chain reaction testing for Helicobacter spp. and is known to harbor H. bilis and unidentified Helicobacter spp. H. bilis was detected in fecal samples from all 6 of the submitted mice, but none had colitis. In our experience, expression of colitis associated with Helicobacter spp. in immunodeficient mice is highly variable. This probably is at least partly due to other members of the bowel flora, as it is both reported 2 and our experience that gnotobiotic C.B-17 SCID mice colonized with Altered Schaedler Flora® and with H. hepaticus develop little or no colitis.
