Abstract
Forty-two ewes had an intravenous catheter sewn in place in a prepared area over the jugular vein and beginning at 60 days of gestation received an infusion 3 times daily. The infusion consisted of sterile saline or sterile saline containing arginine. Twenty-six ewes in both control and treatment groups aborted between 81 days of gestation and term. Fetuses from 16 ewes that aborted were examined. Most were autolyzed or had early mummification. Macroscopic placentitis and noncollapsing lungs were noted. Large numbers of coagulase-positive Staphylococcus were isolated from fetal abomasal content, lungs, brains, or placentas. Histologically, suppurative placentitis with necrosis and pulmonary aspiration of meconium and amniotic debris often with suppurative bronchopneumonia were observed in abortuses. Four ewes euthanized and examined after abortion had suppurative endometritis. Three ewes had severe, chronic, jugular thrombophlebitis from which coagulase-positive Staphylococcus was isolated. The fourth ewe had mild phlebitis, and Staphylococcus aureus was isolated from both the catheter and the blood. Catheter-associated staphylococcal abortion was diagnosed.
Staphylococcus aureus is a pathogen that causes mastitis and dermatitis in sheep 11, 17, 26, 27, 30, 34, 35 and a syndrome of lamb pyemia/septicemia. 10, 32 Although S. aureus is only a sporadic cause of abortion in ewes, it has been reported to cause an abortion-stillbirth syndrome. 10, 13, 21 Staphylococcus spp. is not found commonly in the vaginal flora of sheep but has been cultured from spontaneous, ovine endometritis and from the cervical flora after harvest of embryos. 24, 29 In human beings, S. aureus colonizes the skin and nares and is a common pathogen associated with vascular access devices. 31 It also is found in the skin and upper respiratory tract of ruminants. 34 This manuscript describes an outbreak of coagulase-positive Staphylococcus (Cp-S)–induced abortion associated with long-term, jugular vein catheterization in a group of pregnant ewes.
Materials and Methods
Forty-two ewes were confirmed as pregnant by ultrasound and put into groups for a study. Ewes were housed individually and manipulated in their pens. The experiment involved long-term, jugular vein catheterization with a polyurethane catheter (16-gauge, 13-cm, Milacath, Mila International, Inc., Florence, KY). Wool at the catheterization site was clipped (No. 40 blade, Oster, McMinville, TN), and the skin was cleaned with an iodine-containing soap (povidine-iodine, Vedadine Surgical Scrub, VEDCO, St. Joseph, MO) and then flooded with 70% ethanol prior to insertion of the catheter. Catheters were inserted and sewn in place 1 to 3 days before the first infusion was to be done at 60 days of gestation (DOG). Ewes were infused 3 times daily with either a dose of 80 mg/kg of
After 3 ewes aborted between 81 and 97 DOG, the investigator began to administer once-weekly, prophylactic antibiotics to all ewes on the study (oxytetracycline 20 mg/kg, Oxymycin 200, Agripharm Products, Grapevine, TX). Sporadic abortions continued; however, when 4 abortions occurred over a weekend, 6 102 to 134 DOG fetuses were presented with their placentas to the diagnostic laboratory at Texas A&M University, Department of Veterinary Pathobiology. Three were recently aborted fetuses, and 3 had been frozen.
Necropsies were performed on the abortuses, and placentas were examined. Samples of placenta, lung, and abomasal content were submitted for bacterial culture. Usually, lung, abomasal content, or placenta was cultured, and placenta and lung were examined histologically from excessively autolyzed or mummified specimens. If autolytic or mummified twins were presented, the fresher of the 2 was tested. Because of the macroscopic appearance of the placenta, the first 4 placentas presented also were cultured for fungi. Placenta, lung, liver, brain, kidney, skeletal muscle, and adrenal gland were fixed in buffered formalin, embedded in paraffin, sectioned at 5 µ, and stained for routine light microscopy. Upon being told that the macroscopic appearance of the initial specimens was from probable infectious abortions, the investigator increased the antibiotic treatments to twice weekly; however, abortions continued. Subsequent, aborted fetuses and placentas were examined as above. Infusion solutions in use were submitted for aerobic bacterial culture. Because newborn lambs with staphylococcal septicemia frequently have Staphylococcus isolated from the brain in pure culture, the fetal brains instead of placentas were cultured in later abortions.
Four ewes were euthanized and necropsied after abortion. Lung, liver, kidney, adrenal gland, spleen, uterus, urinary bladder, and jugular vein were fixed and processed for routine light microscopy. The uterus, any jugular vein thrombi, and catheters from the last 2 ewes were aerobically cultured. Clotted blood samples taken from 2 ewes while they aborted had been collected from the jugular vein of the noncatheterized side and aerobically cultured. The last ewe necropsied after abortion had a second fetus in the uterus, and it was examined as above.
Tissues submitted for bacterial culture were surface sterilized, and samples were inoculated onto 5% sheep blood agar (BVA Scientific, San Antonio, TX) and incubated at 37°C in 5–10% CO2 for up to 72 hours. Clotted blood samples were cultured directly as above. Sterile saline was flushed through catheters, and the effluent was cultured. Colonies with typical Staphylococcus spp. morphology were tested for coagulase production by inoculating rabbit plasma (BVA Scientific, San Antonio, TX), incubating overnight at 37°C in air, and observing for coagulation. Two C-pS isolates were speciated (Vitek Gram positive identification card, BioMerieux, Durham, NC) according to the manufacturer's recommendations. The final C-pS isolate that had been speciated as S. aureus was tested for antibiotic susceptibility by disk diffusion following Clinical and Laboratory Standards Institute–recommended methods (BBL disks- Sensidisk, Becton Dickenson & Co., Sparks, MD). Fungal culture protocol included inoculation of Sabouraud-dextrose and potato-dextrose agar media (Remel, Lenexa, KS) and incubation at room temperature for up to 3 weeks.
Placentas from 8 normal births and a normal lamb that died subsequent to a dystocia were examined by histology and bacteriology as above. The catheter from a ewe that delivered a small, term stillborn lamb was cultured as above.
Results
Although the ewes remained afebrile and showed no clinical signs, 26 ewes aborted between 81 and 135 DOG. Sixteen aborted fetuses were examined, and the results of the testing are presented in Table 1. None of the ewes were noted to have dermatitis, mammary abscesses, or mastitis. Most abortuses were not fresh and often were expelled in their membranes. Many were autolyzed or had early mummification, indicating that they had died long before being expelled. Chorioallantoic membranes were thick, tan/grey, and opacified and had dry, nonviable cotyledons (Fig. 1). Fetal lungs were slightly firm and expanded (Fig. 2). Histologically, all lungs were noted to contain meconium and aspirated cell debris in airways and alveoli (Fig. 3), indicating in utero fetal stress and an increase in swallowing of allantoic fluid, but in fetuses aborted after 114 DOG, neutrophils also infiltrated alveoli to some degree (Fig. 4). More intense, pulmonary inflammation was evident in the older fetuses. Fetal lungs and occasionally a liver contained numerous Gram-positive cocci. Chorioallantoic surfaces were thickened by necrotic debris, fibrin, and degenerating neutrophils with a mild to moderate, pleocellular, inflammatory infiltrate within the chorioallantois (Figs. 5, 6). Some placental vessel walls were necrotic and infiltrated by neutrophils. Large numbers of Gram-positive cocci were on membranes, within some macrophages, and occasionally inside distended placental vessels. Fungal elements were not noted in placentas. Inflammation was only observed in lungs and placentas.
Results of examination of aborted lambs.
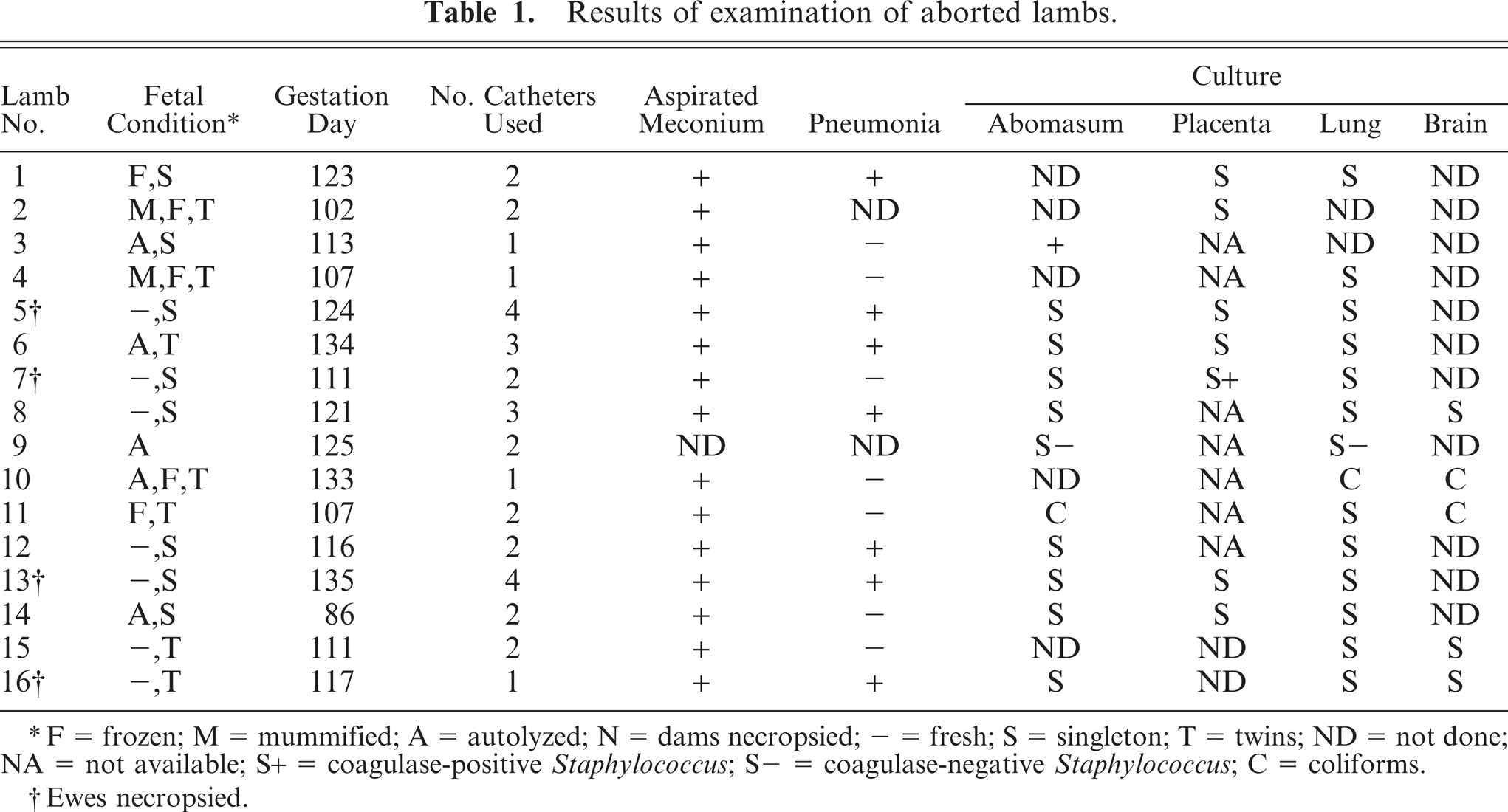
F = frozen; M = mummified; A = autolyzed; N = dams necropsied; - = fresh; S = singleton; T = twins; ND = not done; NA = not available; S+ = coagulase-positive Staphylococcus; S- = coagulase-negative Staphylococcus; C = coliforms.
Ewes necropsied.

Placenta; ovine abortus No. 5. The chorioallantois with chronic placentitis due to a coagulase-positive Staphylococcus infection has a thickened, opacified intercotyledonary zone. Cotyledons are dry, thick, and nonviable.
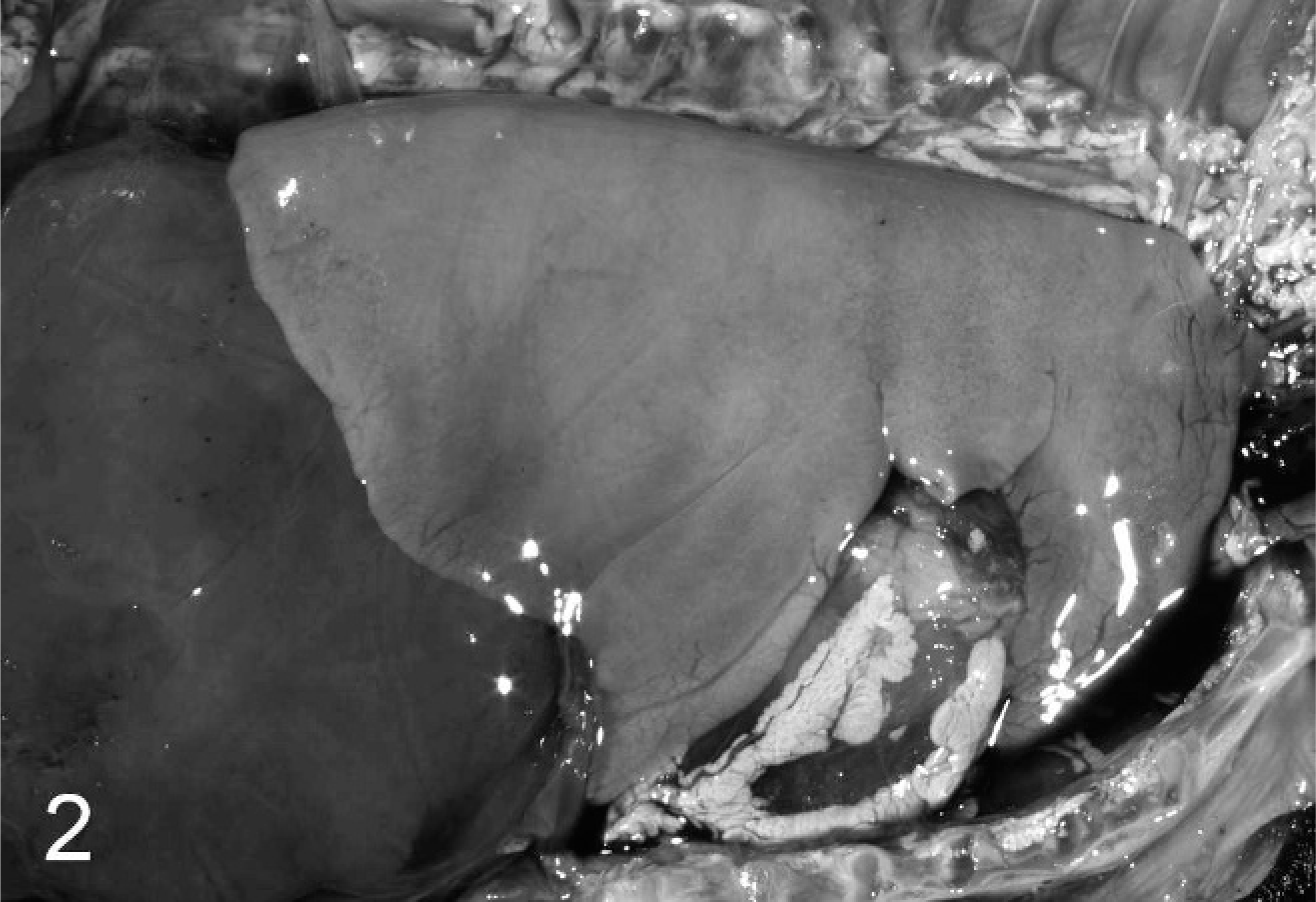
Thorax; ovine abortus No. 5. The lung is expanded by aspirated cell debris and meconium such that it fills the thorax.
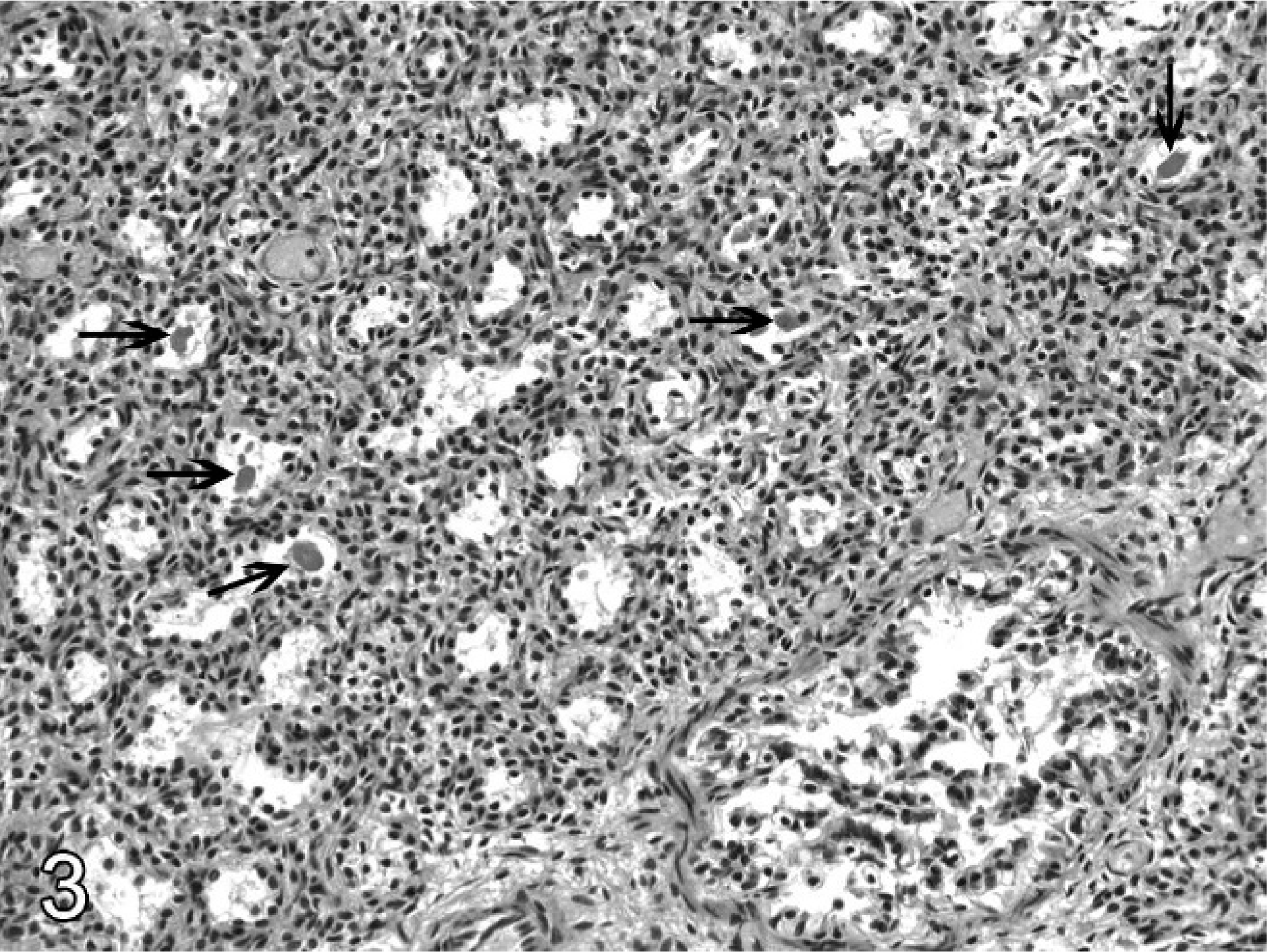
Lung; ovine abortus No. 12. Meconium is in the air spaces (arrows), but the alveoli have no inflammatory infiltate. HE.
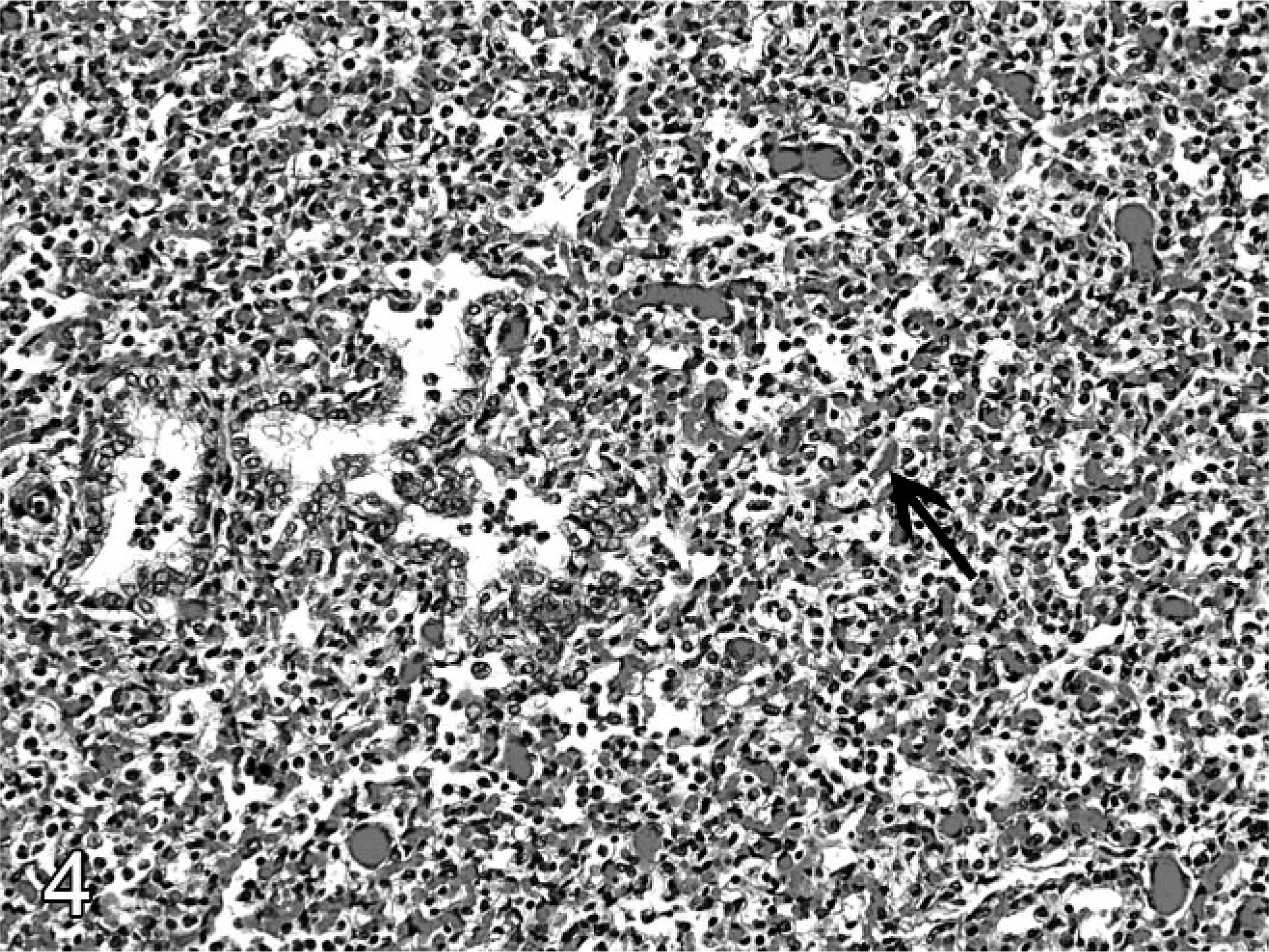
Lung; ovine abortus No. 13. The lung has neutrophils in alveoli and bronchioles. A piece of meconium is in an alveolus (arrow). HE.
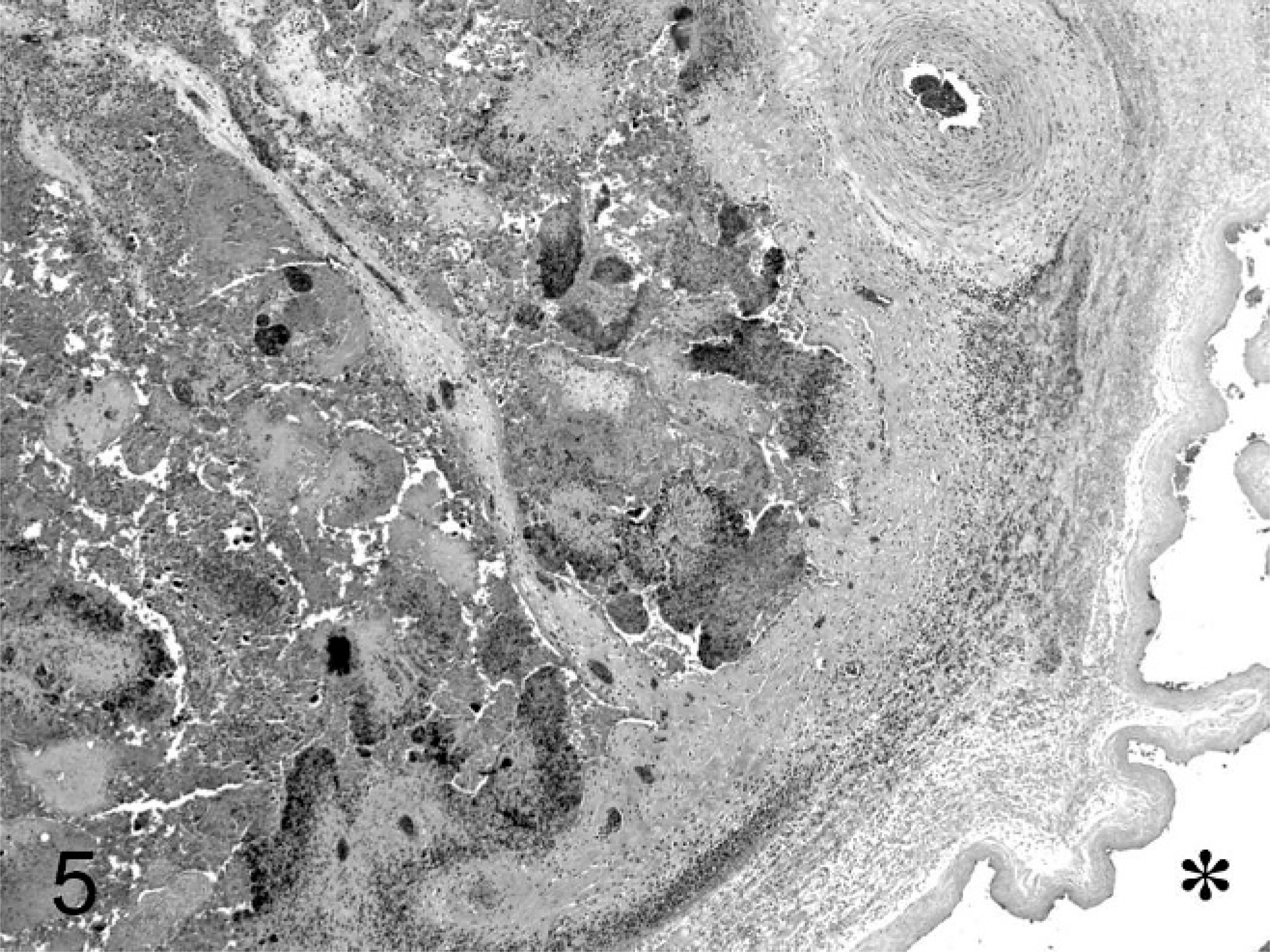
Chorioallantois; ovine abortus No. 6. Fibrin, cell debris, and exudate are on the surface, and a pleocellular, inflammatory infiltrate is in the interstitium of the chorioallantois. An asterix is on the allantoic side of the membrane. HE.
Chorioallantois; ovine abortus No. 13. Necrotic debris, edema, and fibrin thicken the membrane, and numerous colonies of cocci are both on the surface and within vessels (arrows). An asterix is on the allantoic side of the chorioallantois. HE.
High numbers of C-pS were cultured from lung, abomasal content, placenta, or brain of 14 of 16 aborted fetuses. Two C-pS isolates from lesions of jugular thrombophlebitis of ewe Nos. 13 and 16 were speciated as S. aureus. The coagulase-positive S. aureus isolate from ewe No. 16 was resistant to tetracycline. In addition to growth of C-pS, placental cultures resulted in growth of lesser numbers of Escherichia coli, Enterococcus spp., Bacillus spp., and Aeromonas spp., organisms thought to be fecal contaminants. Infusion fluids cultured during the outbreak were sterile with the exception of growth of 1 colony of Ochrobacterium antropi isolated from saline.
A coagulase-negative Staphylococcus spp. (C-nS) was cultured from tissues of a 125 DOG abortus (No. 9) with bronchopneumonia and placentitis. A 133 DOG abortus (No. 10) that had been frozen for 15 days prior to presentation had large numbers of E. coli, Proteus mirabilis, Aeromonas spp., and Corynebacteruim spp. isolated from brain and lung samples cultured. This fetus had no placentitis or bronchopneumonia.
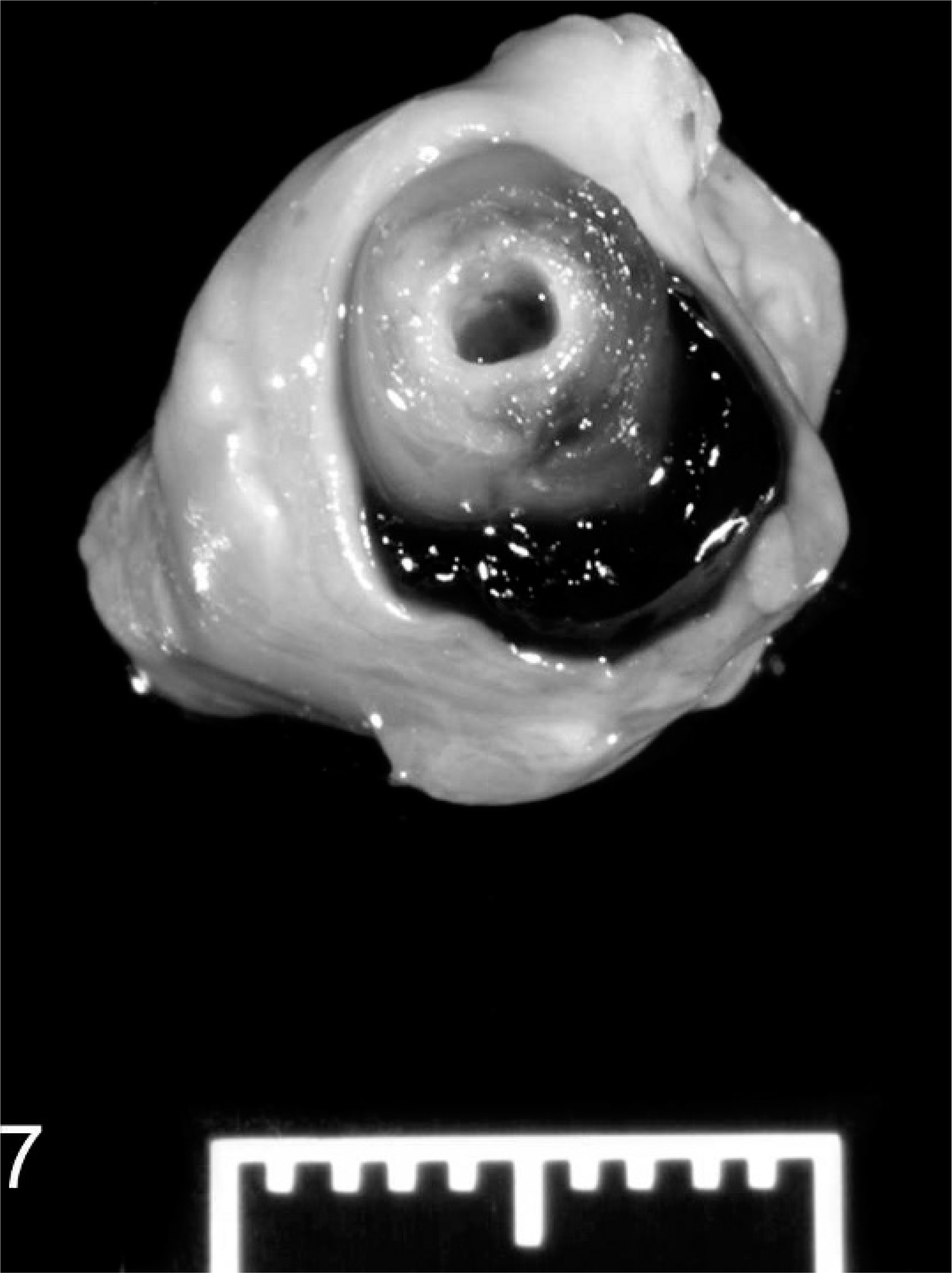
Four ewes were necropsied after aborting (Table 2). These ewes had no macroscopic lesions except in the uterus or jugular vein. All had severe suppurative endometritis. Histologically, Gram-positive cocci were noted in the uteruses, and C-pS were isolated in mixed culture from 3 of 4 uteruses. One ewe (No. 16) was euthanized and examined 1 day after aborting a C-pS–positive fetus and had a second, C-pS–positive fetus in the uterus. Ewe No. 7 was necropsied 4 days after abortion and did not have C-pS isolated from the uterus, although C-pS in pure culture had been isolated from the ewe's fetus. The first 3 ewes examined had severe, chronic, jugular thrombophlebitis with organized thrombi extending toward the heart from the point of catheter insertion. In 2 cases (ewe Nos. 5 and 13), the center of the thrombus contained a channel that had been maintained by the catheter (Fig. 7). In 1 case (ewe No. 13), the entire right jugular vein was occluded (Fig. 8), and the opposite jugular vein had been catheterized in order to complete the experimental procedure.
Results of examination of aborting ewes. ∗
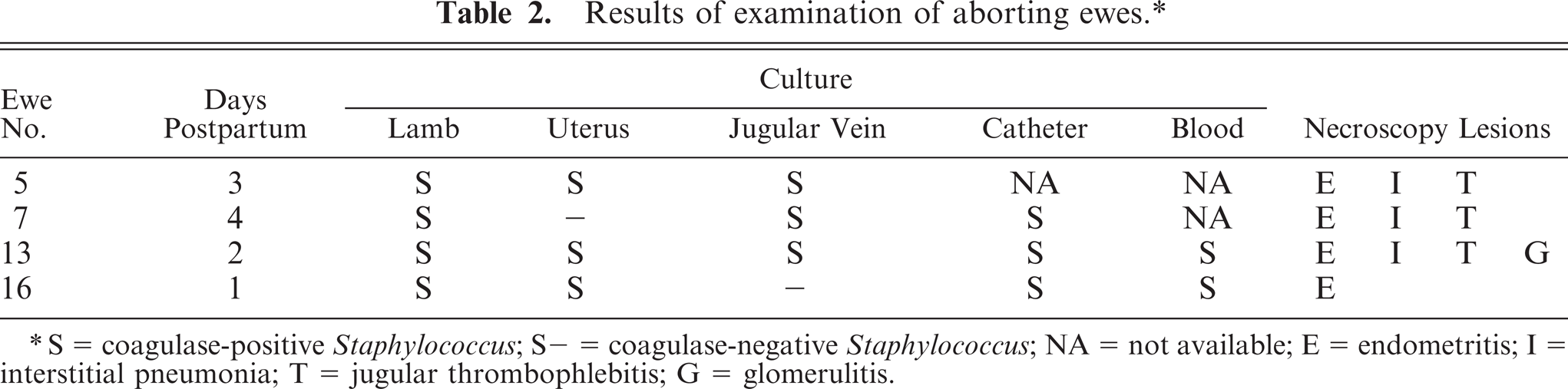
S = coagulase-positive Staphylococcus; S- = coagulase-negative Staphylococcus; NA = not available; E = endometritis; I = interstitial pneumonia; T = jugular thrombophlebitis; G = glomerulitis.
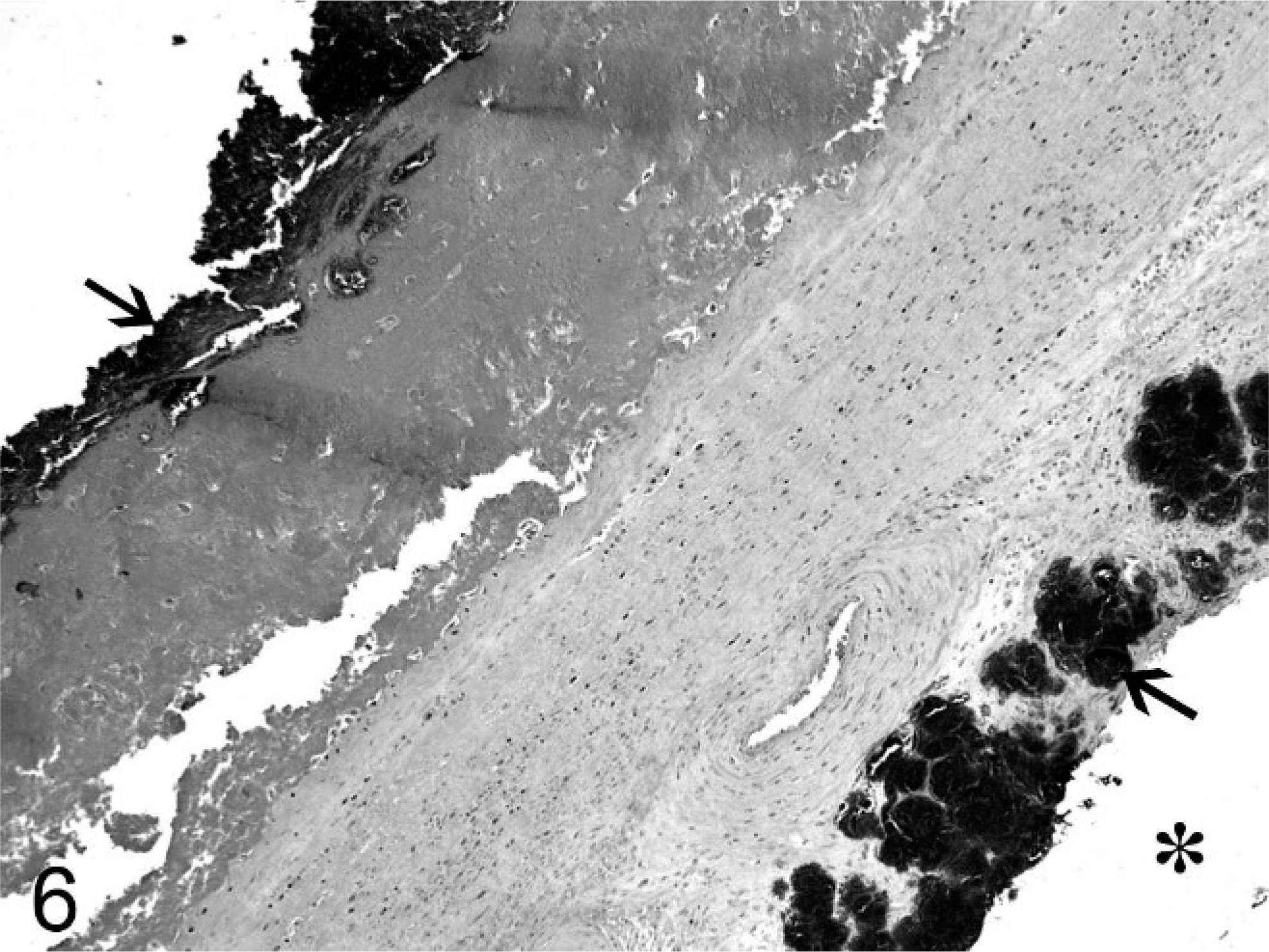
Jugular vein; ewe No. 7. The area of thrombophlebitis segmentally adheres and thickens the vein. Laminae of fibrin and inflammation are around the channel maintained by the catheter that has been removed. Clotted blood fills the remaining lumen.
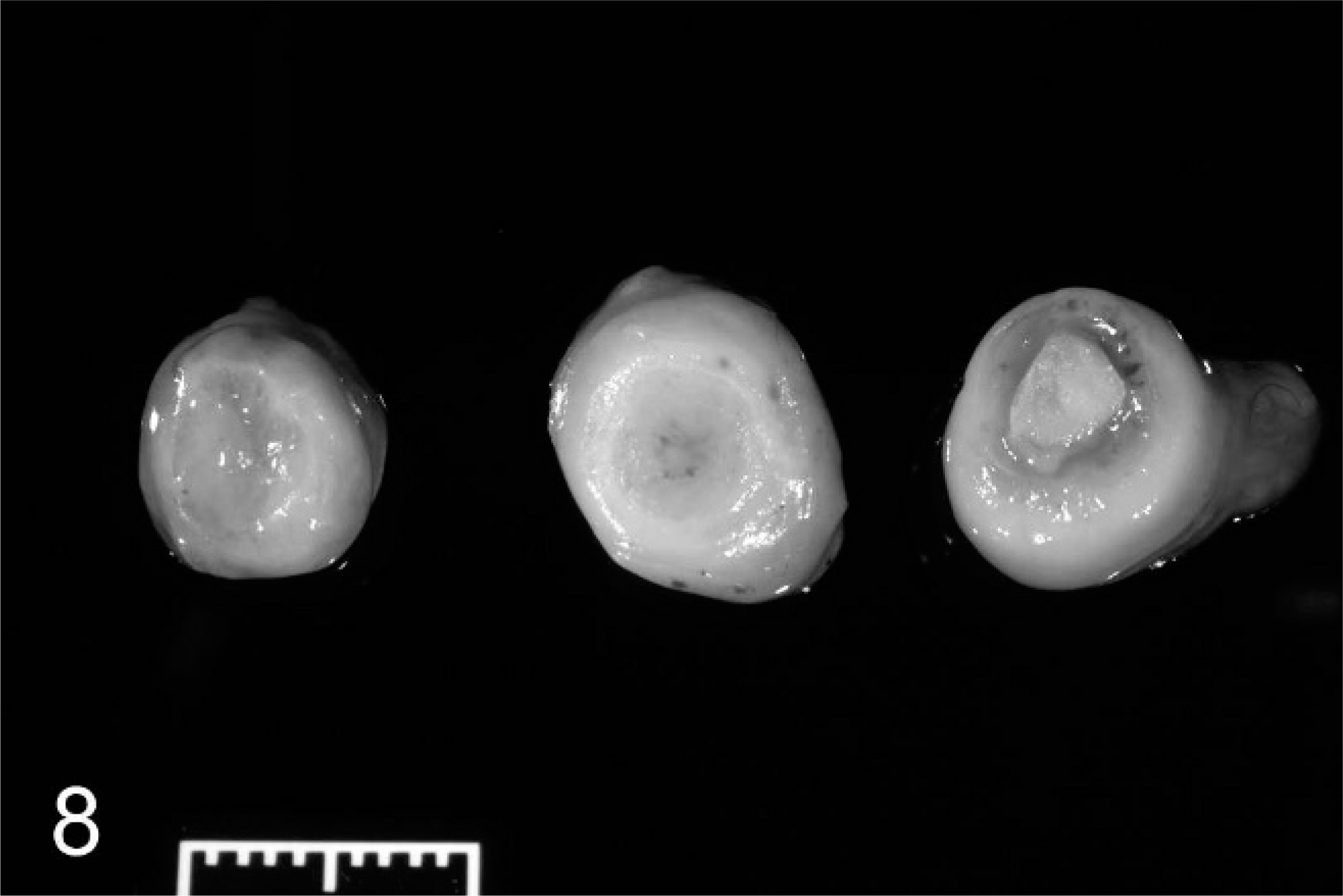
Jugular vein; ewe No. 13. Cross-sections of chronic, occlusive thrombophlebitis show the nature of the material occluding the jugular vein. At different points, the vein has a complete, fibrous occlusion (left), an occlusion of fibrosis and inflammation (center), or fibrosis with a central core of exudate (right).
Histologically affected venous walls consisted of mature, fibrous connective tissue peripherally with proliferating, small blood vessels having cuffs of plasma cells and a few lymphocytes (Fig. 9). Moving adlumenally, mature connective tissue was replaced by granulation tissue and aggregates of lymphocytes and histiocytes. The center of the thrombi consisted of either islands of neutrophils and histiocytes separated by an immature, fibrovascular stoma or segmentally along the length of the thrombi or islands of fibrin, neutrophils, and histiocytes (Fig. 10). In affected veins that had channels maintained by catheters, the venous wall lesions were similar; however, laminae of fibrin and neutrophils with colonies of Gram-positive cocci were compacted against the catheter channel by surrounding granulation tissue. The fourth ewe had mild roughening of the venous endothelial surface due to fibrin deposition, and the venous adventitia had hemosiderin deposits and vessels cuffed by lymphocytes and plasma cells.
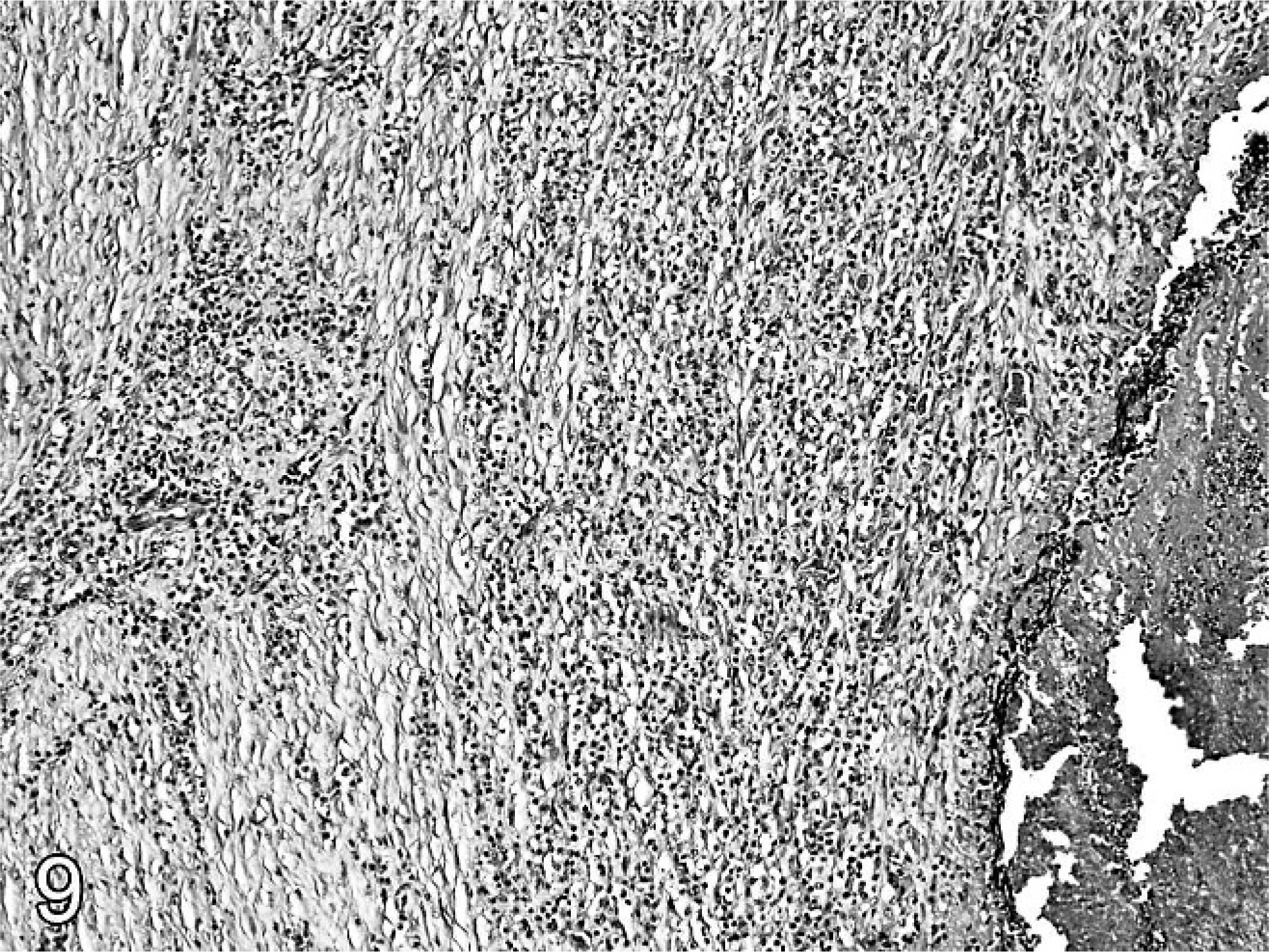
Jugular vein; ewe No. 13. The periphery of a chronically contaminated, inflamed, and thrombosed jugular vein has mature connective tissue surrounding vessels cuffed by mononuclear cells separating aggregates of lymphocytes and histiocytes. HE.
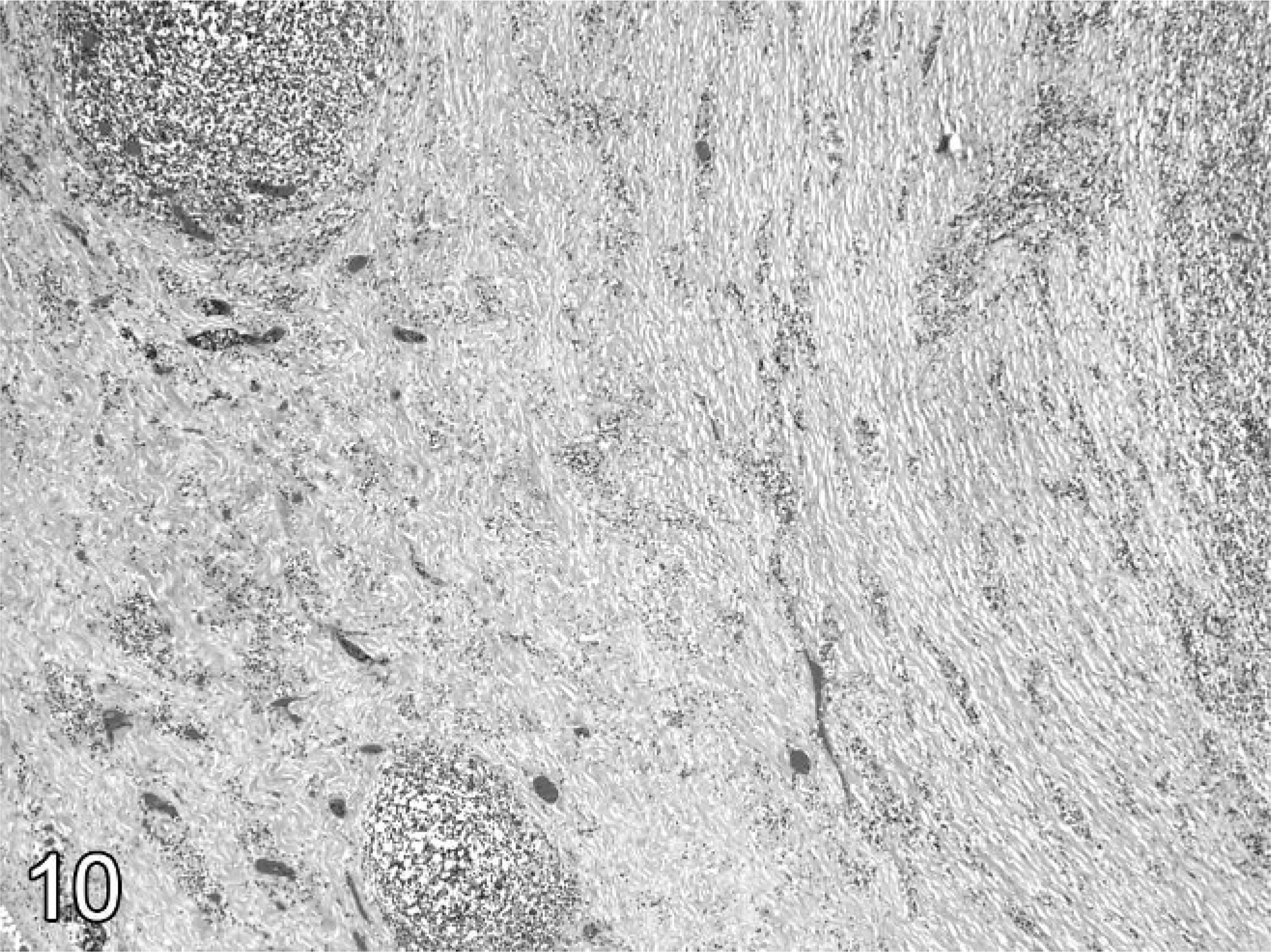
Jugular vein; ewe No. 13. The character of the thrombus changes from mature connective tissue to organizing granulation tissue. The center (at right) has fibrin and neutrophilic debris. HE.
C-pS were isolated from all thrombi. Bacterial culture of clotted-blood samples taken during abortion from the last 2 ewes necropsied and the catheters from the last 3 ewes necropsied resulted in isolation of C-pS in pure culture. Two C-pS isolates cultured from the thrombophlebitis were typed as S. aureus. Histologically, ewe Nos. 5, 7, and 13 had mild, multifocal, hemorrhagic interstitial pneumonia. Ewe 13 had mild, acute glomerulitis.
Approximately 2 weeks after increasing the dose of antibiotics, some live lambs were delivered in the study, and ultimately 16 of the original 42 ewes delivered term lambs. Placentas from 10 lambs born at term had no placentitis, but small numbers of C-pS were isolated from 2 of 3 catheters from ewes delivering live lambs. The lung and brain from a term lamb that died in dystocia did not have organisms isolated from lung, abomasal content, or brain, and no histologic lesions were observed in tissues. Another term lamb was stillborn and small but was not presented for study. Although the average weight for live, term, singleton lambs was 4.8 kg (range 3.5–7.5 kg), this lamb weighed 1.4 kg. C-pS were cultured from its dame's catheter.
Discussion
It was obvious from gross and histologic examination of the initial placentas that the abortions were the result of chronic bacterial infection. Differential diagnoses for spontaneous, chronic placentitis in sheep include infections by Brucella, Salmonella, Actinomyces, Campylobacter, Chlamydophila, Coxiella, Listeria, and fungi. 13, 21 Other infectious etiologies do not characteristically result in intercotyledonary areas thickened by exudate with bacterial colonies and intracellular bacteria. In this study, the bacteriologic cultures and histologic observations of the cases identified Staphylococcus as the etiology. After negative culture of the infusion solutions, contamination of the catheters by common skin organisms was the presumed cause of the Staphylococcus abortions. The jugular thrombophlebitis in conjunction with C-pS–contaminated veins and catheters are findings critical to confirm catheterization and C-pS sepsis in the pathogenesis of the outbreak. Unfortunately, few blood samples and catheters from ewes were presented for culture. The catheters tested and the clotted blood samples taken from the last 2 ewes necropsied contained C-pS, and these findings further supported the diagnosis. 28
Because the final ewe necropsied was one of the final ewes to abort, the lack of severe jugular vein lesions was attributed to the increased antibiotic treatment schedule, and the investigators improved the care of catheters and injection technique after the diagnosis was made.
Although not often reported in domestic animals, venous catheter infections in human beings are a well-studied phenomenon. Catheter contamination can occur from the exit site, the catheter hub, contaminated infusion solutions, or, least common, septicemias originating at noncatheter sites. Catheter sepsis most commonly occurs from skin contamination at the catheter exit site. The breakdown of the natural cutaneous barrier, the piston-like movement of the catheter, and the tissue damage caused by the catheter predispose the site to infection. Further, chronic infections are predisposed because biofilms made by certain bacterial pathogens aggregating and secreting carbohydrates, 18 as well as the muco-polysaccharide in heparin used to maintain catheter patency, protect organisms from antibiotics used to treat the infection. 1, 12, 18
A number of measures are used to minimize contamination of catheters. 2, 3, 7, 12, 16 In all cases, preparation of the catheter site and use of sterile barrier precautions (use of a drape, gloves, mask, cap, and gown) while installing a catheter is critical. Aseptic handling and disinfection of the hub before handling are essential precautions for long-term catheter placement. Tunneling of catheters to limit capillary movement of bacteria from the skin also is a useful precaution. Catheter composition and use of catheters impregnated with antibacterial substances will affect the infection rate. Indeed, the composition of the catheter and the types of infused material can induce an aseptic phlebitis at the catheterization site. 4, 5, 7, 33 When an infection is suspected, filling of the catheter and maintaining it full of an antibiotic solution between infusions, the antibiotic lock technique, 15, 28 is recommended as a means of managing contamination. However, because of the high rate of complications and antibiotic resistance related to C-pS infections, prompt catheter removal is the preferred treatment when C-pS is detected. 12, 28 The above measures often are not reported to be employed long-term in routine animal research units.
Staphylococci have been shown to produce a device-associated biofilm. These biofilms may increase the antibiotic resistance of sessile, bacterial colonies as much as 1,000-fold when compared with the susceptibility of planktonic bacteria. 1, 31, 43 C-pS do not induce systemic illness in mature sheep and are an uncommon abortifacient. It is believed that skin contamination caused the catheter infection and a bacteremia that infected the fetuses in this report. Resistance to tetracycline was a further complication in these abortions. The dose of tetracycline used was that recommended for prophylaxis and not treatment of Staphylococcus infections; therefore, selection for resistant C-pS may have been favored. 14, 34 It is not known if all C-pS isolates were resistant to tetracycline or if the environment became increasingly contaminated with resistant C-pS during the experiment.
The isolates were identified as Staphylococcus and typed as coagulase positive or negative. Few C-pS were speciated. Although there are 3 C-pS species, S. aureus, S. intermedius, and S. hyicus, most ruminant C-pS isolates are S. aureus, whereas most C-pS from dogs and cats are S. intermedius. S. hyicus is isolated most frequently from swine. 6, 20, 22 Hence, it was presumed that most of our C-pS were S. aureus, a common skin contaminant of sheep.
Although most aborted fetuses tested were infected by C-pS, 2 did not have C-pS isolated from their tissues. In one case, large numbers of C-nS were isolated from a fetus with suppurative placentitis and bronchopneumonia. 15, 23 In human beings, and occasionally in dogs, C-nS are isolated from catheter-associated infections. S. epidermis, a C-nS species, is the most common, human contaminant of nontunneled catheters and causes septicemia with no clinical signs. 9, 23 C-nS are associated with the skin, mammary gland, and sheep pens, 6, 31 but our C-nS isolate was not typed. The other, non–C-pS–infected abortus was contaminated with E.coli, P. mirabilis, Aeromonas spp., and Corynebacteruim spp. Presumably, fecal contamination of the catheter was the source of these organisms.
An interesting observation was the change in the pulmonary lesions observed in fetuses of different ages infected with the same organism. Although bacteria were seen in all lungs from which C-pS were isolated, neutrophils were seen only in the lungs of older fetuses (from 114 DOG onward). Mature neutrophils and complement activity are reported to appear in ovine fetal blood at approximately 123 DOG, 8, 19, 25 and neutrophil chemotaxis to endotoxin is reported to reach adult levels by 131 DOG. 19 It was presumed that pulmonary inflammation occurred in the infected fetuses only after adequate maturation of the fetal nonimmune defense systems.
Complications from catheter infection must always be considered when contemplating protocols using central venous catheters. The Staphylococcus-associated abortion outbreak in this report was a unique occurrence predisposed by an opportunistic, common bacterium of the skin and a common, but inadvertent, breach of the host's natural defenses that exposed the fetuses to infection.
