Abstract
The economically important effects of Tritrichomonas foetus infection in cattle are abortion and infertility, yet there has not been an animal model to examine the parasite-host interactions during gestation. In this study, 5- and 7- to 8-week-old BALB/cAnNCr, BALB/cJ, and SCID/NCr mice on a BALB/c background were intravaginally infected with T. foetus. All BALB/ cAnNCr and BALB/cJ mice, and 89% of SCID/NCr mice sustained infections for 13 weeks, if inoculated before 5 weeks of age. Infection rates were lower in all mouse strains inoculated at 7 weeks of age, although BALB/cAnNCr mice were significantly more susceptible than BALB/cJ or SCID/NCr mice. Vaginal bacterial flora did not account for the variation in mouse-strain susceptibility, although coagulase-negative staphylococci in vaginal flora were associated with failure of T. foetus to infect. As with infected cattle, T. foetus-specific vaginal immunoglobulin (Ig) G and IgA antibodies were elevated after infection. The number and viability of day-10 fetuses were reduced in mice infected at 5 weeks of age and bred 12 weeks after infection. Lesions in pregnant and nonpregnant infected mice, including suppurative and eosinophilic vaginitis; cervicitis; endometritis with distension of the uterine lumen; endometrial ulceration; and glandular ectasia, with neutrophils in the glandular lumen and loss of gland epithelium, were similar to those in cattle. The decidua and placenta were multifocally necrotic. Immunohistochemistry demonstrated trichomonads in vaginal folds and uterine glands, and adjacent to fetal tissues. In summary, experimentally infected BALB/cAnNCr mice showed many pathologic similarities to cattle and may serve as a model to study host-trichomonad interactions.
Tritrichomonas foetus is a sexually transmitted obligate protozoan pathogen of cattle and is responsible for causing occult pregnancy losses where natural service of cattle is practiced. 8 Trichomoniasis is an insidious disease, which results in loss of a significant portion of a year's calf crop before there is any suspicion of infection. Infection usually leads to undetected vaginitis, endometritis, pregnancy loss, and transient infertility, but occasionally may progress to pyometra and permanent endometrial damage. 8, 46 Cows are typically infected by subclinical carrier bulls at coitus and clear the infection within a few months. 9 Over the last 100 years, there has been considerable research on the parasite, its virulence factors, and the host response. This work, however, has been hampered by the logistic and economic difficulties of working with the natural host, the cow.
The rapid reproductive rate, low cost of maintenance, defined genetic traits, and growing number of molecular tools, including genetic knockouts, make the mouse a desirable candidate for a trichomoniasis model. 6 Previous efforts to create such a model have been only partially successful. Models that use intraperitoneal, subcutaneous, or intramuscular infection have been developed, but their relevance for the study of a venereal pathogen is limited. 30, 35 More recently, mice were successfully intravaginally infected with T. foetus. In some studies, infection only persisted if the mice were also treated with supraphysiologic doses of estrogen. 32, 62, 64 These hormonally supplemented models were useful in assessing virulence of strains and in examining trichomonad pathogenic mechanisms in vivo, but infection was maintained in only 80% of the animals beyond 2 weeks postinfection (PI), and dropped to 20% after 6 weeks PI. In addition, the use of estrogen may mask much of the important interplay between host and parasite. Estrogen has been shown to have marked immune-modulating effects, both systemically and in the reproductive tract. 29, 39, 47, 48, 55, 61, 62 A recent mouse model demonstrated marked suppression of vaginal immunoglobulin (Ig) A, uterine IgG, and serum IgG in estrogenized CD1 mice compared with nonestrogenized mice, although a similar effect was not seen in BALB/c mice. 42 Estrogen's modulatory effect on eosinophils is particularly noteworthy 47 in light of the large number of eosinophils seen in T. foetus infections. 1, 18, 40 Loss of eosinophil function has resulted in greater susceptibility to helminth infections in mouse models. 60 In addition, the use of supraphysiologic levels of estrogen precludes examination of the immune system and the host-parasite interaction during the important process of conception and gestation, because successful pregnancy depends on physiologically appropriate hormonal levels, which control tubular transport of gametes, secretory activity, and uterine motility. 23 More recently, mouse models that did not require estrogen supplementation were used to examine antibody responses to T. foetus infection in mice and adherence to reproductive mucosa in vivo. 40, 42 Another mouse study examined the effects of stress and estrogens on infection, 55 but the effects on pregnancy were not examined in these models.
Some models for T. foetus (and Tritrichomonas vaginalis, a related human parasite) have relied on Lactobacillus sp. to enhance infection rates, 37 but the role Lactobacillus sp. and normal flora might have in the success of intravaginal infection is not clear. 3, 25, 59 The mouse estrous cycle has a marked effect on the intravaginal flora, 12, 43 but, in one in vivo mouse study, the stage of the estrous cycle at the time of T. foetus inoculation had no effect on the success of infection. 42
Because the pregnant uterus is markedly different from the nonpregnant uterus, physically, physiologically, hormonally, and immunologically, 66 a pregnant mouse model would be invaluable to study the effects of trichomoniasis. Thus, the objectives of this study were to develop and characterize a pregnant mouse model that would provide insight into the pathogenesis of pregnancy loss in trichomoniasis.
Materials and Methods
Mice
Three strains of inbred laboratory mice were used in these studies: 1) BALB/cJ mice (Jackson Laboratories, West Sacramento, CA), 2) BALB/cAnNCr mice (Frederick Cancer Research Center, Frederick, MD), and 3) BALB/c SCID mice (Frederick Cancer Research Center, Frederick, MD). Hybrid 7- to 8-week-old B6D2F1 male mice (Frederick Cancer Research Center, Frederick, MD) were used for mating. BALB/c SCID mice were used only for initial studies to determine the effect of age and mouse-strain variation on susceptibility. All mice were maintained in isolator cages in groups of 3 or 4 within an infectious-disease containment room and were fed commercial mouse diet and water ad libitum.
Parasites
T. foetus strain D1 was originally obtained from a cow with severe postcoital pyometra. 58 This strain, at a dose of 1 × 106 T. foetus organisms intravaginally instilled, was used in several studies to intravaginally infect 100% of experimental heifers at estrus. 10, 16 Parasites were maintained in culture by using trypticase-yeast extract-maltose (TYM) medium that contained 10% fetal calf serum until inoculation. 22
A mouse-adapted strain of T. foetus (MuTF 52100V) was developed by first intravaginally infecting BALB/cAnNCr mice with strain D1. Cultures from vaginal lavages of infected mice were then pooled, grown in culture by using TYM media, transferred to fresh media every 2 to 3 days, and subsequently inoculated into susceptible mice. A second-generation mouse-adapted strain (MuTF 10N) was developed by selecting positive cultures from mice infected with strain MuTF 52100V.
Experimental protocol
After ear-notching and initial blood sampling, female mice were allowed to acclimate for 2 days before an experiment. Females in each group were intravaginally inoculated with 104 motile T. foetus strain MuTF10N organisms suspended in 10 µl trypticase-yeast extract-iron serum (TYI) media by using an Eppendorf pipette; control animals were sham inoculated with sterile TYI media. 21 A second inoculation of T. foetus or TYI at the same dose and volume was given the following day. Successful T. foetus infection was confirmed by vaginal lavage and culture (as described below) 1 week after inoculation and at weekly intervals as needed to determine the strain and age susceptibility, as well as the interactions with normal flora with T. foetus in mice. Age susceptibility of the mice was examined by comparing T. foetus culture results in 5-week-old and 7- to 8-week-old female mice.
To assess the effect of T. foetus on pregnancy in mice, female mice were preconditioned, acclimated, and inoculated as described above. After confirmation of successful infection 1 week PI, females were maintained in groups of 2–3 for either approximately 3 weeks or approximately 12 weeks before the introduction of males. Male mice were introduced for mating at a ratio of 1 : 2 or 1 : 3 male : female. Females were examined once daily in the morning for vaginal plugs that indicated successful mating. Males were removed 4 days after introduction. The first day a plug was noted was considered gestation day (gd) 0.5. Mice were humanely killed by using pentobarbital injection (Nembutal, Abbott Laboratories, North Chicago, IL) followed by exsanguination via cardiac puncture and cervical dislocation. A necropsy was performed on gd 10–11, at which time both regressing moles and midterm dying embryos could still be detected. Age- and strain-matched control mice were similarly preconditioned, inoculated with an identical volume of sterile TYI, bred to B6D2F1 males, and killed and examined at gd 11.
Sample collection
Vaginal washes were performed by 4 replicates of 10 back-and-forth flushes of 50 µl phosphate buffered saline solution (PBS). These replicates were pooled to make approximately 200 µl of sample. Fifty microliters from each wash were cultured in TYM, and the remainder were frozen at −70°C for later analyses. In some experiments, the stage of estrous cycle was confirmed by vaginal cytology as follows: approximately 10 µl of fresh wash sample was placed on a glass slide, a drop of new methylene blue stain was added, and a coverslip was applied. The stage of the estrous cycle was determined by subjectively assessing the relative numbers of neutrophils; small nucleated epithelial cells; and large, often anuclear, squamous epithelial cells according to established standards. 13 The presence of trichomonad organisms was also noted. At the beginning of each experiment, approximately 100–150 µl of blood was collected into capillary serum separator tubes with gel (Monoject, St. Louis, MO) via the saphenous vein and was centrifuged after clotting, and the serum was frozen at −20°C for later analysis.
At the termination of each experiment, all mice were humanely killed. Uterus, oviduct, ovaries, and vagina were dissected from the mice, and these were examined, photographed, and incised. The number of fetuses was noted, and the uterus and ovaries were weighed. The uterus was bisected at the cervix, and 1 horn was flushed from the proximal end with 1.0 ml PBS. Fifty microliters of this flush was cultured in TYM and the remainder was frozen at −70°C. All reproductive organs were fixed in 4% paraformaldehyde for 12–24 hours, washed for 24 hours in PBS, dehydrated by transferring, every 24 hours, from 30% ethanol to 50% ethanol and to 70% ethanol. The remaining tissues were preserved in 10% neutral buffered formalin. The uterine horn that was flushed was not used for any subsequent histologic examinations.
Preserved tissues were paraffin embedded and 5-µm sections were mounted on slides and routinely stained with HE, as well as Congo red to assess eosinophil numbers. 2, 28 Selected slides were stained with Bodian's stain to highlight the presence of trichomonads. 31, 34 Additional sections were stained by using standard immunohistochemical methods with trypsin antigen retrieval and a rabbit polyclonal antibody against whole T. foetus provided by California Animal Health and Food Safety Laboratory. 50, 52 After deparaffinization and hydration, endogenous peroxidase was quenched with 3% hydrogen peroxide for 5–10 minutes. Trypsin antigen retrieval was performed at 37°C for 10–20 minutes. The specific primary antibody was diluted 1 : 1500 and applied for 30–60 minutes. Horseradish peroxidase–labeled anti-rabbit polymer (DakoCytomation, DakoUSA, Carpenteria, CA) was used as a secondary detection solution and was applied for 30–60 minutes according to the manufacturer's instructions, and AEC substrate chromogen solution (DakoCytomation, DakoUSA, Carpenteria, CA) was used for color development. Mayer's hematoxylin was applied for 15–90 minutes as a counterstain. Aqueous mounting medium was applied, allowed to dry, and slides were coverslipped.
Vaginal bacterial culture
An aseptically handled 100-µl aliquot of the vaginal washes described above was cultured to assess the microflora of the vagina before and after experimental T. foetus infection in 10 BALB/cAnNCr and 10 BALB/cJ. Based on previous studies, the most abundant bacterial genera expected were Streptococcus, Staphylococcus, Enterococcus, members of the Enterobacteriaceae, and other Gram-negative bacteria. 43, 63 Therefore, vaginal washes were inoculated on defibrinated sheep blood, MacConkey, chocolate, and Rose agars (Hardy Diagnostics, Santa Maria, CA). Individual colonies were identified by using standard biochemical microbiologic methods. 41 Identification of enteric bacteria used API 20E test strips (Biomerieux, Hazelwood, MO) and for Enterococcus sp. API 20Strep test strips (Biomerieux, Hazelwood, MO) were used. Relative bacterial numbers were estimated by colonies in streaked plate quadrants, where + represents growth in the first quadrant (inoculum), ++ represents growth in the first and second quadrant, and +++ represents growth in 3 quadrants. 49 Partial 16S rRNA gene sequencing was used to confirm the identity of Lactobacillus spp., Acinetobacter spp., and nonfermentative bacteria. 14 Briefly, DNA was extracted by using the boiling lysis method with 2% sodium dodecyl sulfate and 10% Triton X-100 (Sigma, St. Louis, MO) in Tris-EDTA buffer (pH 8.0) and heating at 98°C for 5–15 min., cooling at 4°C for 1 min. followed by centrifugation to pellet bacterial debris and collection of supernatent. 33 The 16S rDNA was amplified by using FideliTaq PCR Master Mix (USB Corp. Cleveland, OH, USA) and the universal 16S rDNA primers, 8FPL (5′-CTGCAGAGTTTGATCCTGGCTCAG-3′) and 1492RPL (5′-CGGGTTACCTTGTTACGACTT-3′), as previously described by others. 51 The PCR product was purified and concentrated by ultrafiltration by using a Microcon YM100 Centrifugal Filtration Unit (Millipore, Billerica, MA). Amplified DNA was sequenced in one direction with one of the above primers by using dye terminator cycle sequencing on an ABI 3730 DNA sequencer (Applied Biosystems, Foster City, CA; Davis Sequencing, Davis, CA). The resulting sequence was compared with the GenBank database (http://www.ncbi.nlm.nih.gov/Genbank/) for identification of bacterial genus.
Determination of antibody levels
Isotype- and antigen-specific antibody levels in serum, vaginal washes, and uterine washes were determined by enzyme-linked immunosorbent assay (ELISA) by using methods similar to those previously described. 10, 17, 57 Briefly, 96-well 1/2-well microtiter plates (CoStar, Cambridge, MA) were coated with 50 µl whole T. foetus organisms that were taken from a 48-hour culture, washed, and diluted to 5 × 106/ml in PBS. Plates were centrifuged at 500 × g for 5 minutes and then organisms were fixed by carefully adding 50 µl 0.1% glutaraldehyde to each well. Plates were left overnight at room temperature (RT), uncovered in a protected area, and then the contents were carefully removed. Plates were then dried overnight in a fume hood, covered, and frozen at −80°C to allow simultaneous assay of samples from multiple experiments.
After thawing, the plates were washed 3 times with PBS that contained 0.05% Tween 20 (PBS-Tw). Nonspecific binding was blocked by adding 180 µl PBS with 3% gelatin (Difco Laboratories, Detroit, MI) and 0.02% sodium azide, and were incubated at 37°C overnight. Plates were then emptied and washed 3 times with PBS-Tw. Mouse vaginal washes were diluted 1 : 10 in PBS that contained 0.05% Tween 20 and 0.2% gelatin (PBS-Tw-G). Uterine flushes were diluted 1 : 10 in PBS-Tw-G, and serum was diluted 1 : 100. All samples were run in duplicate. Fifty microliters of diluted sample was added to each well, the plates were shaken, and then incubated in a humid chamber for 90 minutes at 37°C. The plates were emptied, washed 3 times with PBS-Tw-G, and 50 µl of secondary horseradish peroxidase conjugated antibodies (rabbit anti-mouse IgG and anti-mouse IgA) were diluted 1 : 10,000 (Zymed Laboratories, Inc., San Francisco, CA). The plates were incubated at 37°C for 30 minutes again, before being emptied and washed 3 times, then 50 µl TMB substrate (SureBlue, Kirkegaard and Perry Laboratories, Inc., Gaithersburg, MD) was added to each well, and the plates were incubated at RT. The color reaction was stopped after precisely 30 minutes by adding 50 µl 1 N hydrochloric acid. The optical density was read on a dual-wavelength ELISA microplate reader (Benchmark, Bio-Rad Laboratories, Hercules, CA) at 450 and 650 nm.
Each plate contained positive controls composed of serum from a single mouse that had been exposed to intravaginal T. foetus after vaccination with a dose of whole killed T. foetus organisms in Quil A adjuvant. 15 The negative control was serum from a male mouse unexposed to T. foetus. Appropriate negative controls where a sample or secondary antibody and conjugate were excluded were also used.
Statistical analysis
The relative susceptibility of mouse strains and age groups to T. foetus intravaginal infection was analyzed by a chi-square test, and associations between vaginal microflora and T. foetus infection were analyzed by the Fisher exact test. The numbers of fetuses found per pregnant mouse in each treatment group were analyzed by analysis of variance. A comparison of mean serologic responses in infected and uninfected mice was made by using the Student's t-test.
Results
Mouse strain and age susceptibility to vaginal infection
Experimental intravaginal infections in nonpregnant mice showed that, after 3 weeks PI, all BALB/cJ or BALB/cAnNCr mice were still infected if they were inoculated at 4–5 weeks of age (Table 1). In BALB/cAnNCr mice, T. foetus was cultured from 100% of mice (6/6) even after 13 weeks when inoculated at 4–5 weeks of age. Typically, if an individual mouse was still infected after 1 week PI, then it remained infected; i.e., those mice that cleared the infection appeared to do so within the first week. A significantly lower percentage of mice were infected in all strains, and fewer BALB/cJ were infected than BALB/cAnNCr if they were inoculated at or after 7 weeks of age. Infections with T. foetus of SCID/NCr on a BALB/c background produced similar results, in that most of the mice were still infected after 5 weeks PI, if the mice were 4–5 weeks of age at the time of inoculation, but only a few mice retained the infection if they were inoculated after 7 weeks of age.
Effect of age and mouse strain on persistence of Tritrichomonas foetus infection for at least 3 weeks in BALB/c mice inoculated intravaginally with a mouse-adapted strain of T. foetus. ∗

Percentages indicate the proportion of those initially inoculated that were still infected on the day of sampling.
Indicates that results within strains with the same superscript are significantly different from each other (P < .0001).
Indicates that results within time groups are significantly different from each other (P = .03).
Indicates that results within strains with the same superscript are significantly different from each other (P < .0001).
Indicates that results within strains with the same superscript are significantly different from each other (P < .0001).
Uterine infection
At necropsy, 77% of mice (37/48) (both BALB/cJ and BALB/cAnNCr) with T. foetus–positive vaginal cultures also had positive uterine cultures. Of those mice infected for 13 weeks before necropsy, 100% of mice with positive vaginal cultures (7/7) also had positive uterine cultures, whereas in those mice infected for only 3 weeks or less, 73% of mice with positive vaginal cultures had positive uterine cultures (30/41).
Mouse strain and bacterial associations with T. foetus infection
The results of microbiologic cultures of vaginal washes from 10 BALB/cAnNCr and 10 BALB/cJ mice before and after inoculation with T. foetus, as well as vaginal cytology results, are provided in Table 2. The stage of estrous cycle was estimated by external examination of the vulva and by vaginal cytology. In this study, there was no significant association between the presence or absence of bacteria in general, any particular species of bacteria, or the relative numbers of bacteria found and the estrous cycle stage when a sample was taken. There was a significant association between the presence of Lactobacillus sp. in the vagina after inoculation and successful infection with T. foetus (P = .05), but this association became insignificant if the presence of Lactobacillus sp. before infection was also included in the analysis. In addition, the presence of any Staphylococcus sp. preinfection or PI in either mouse strain was negatively associated with the presence of T. foetus infection (P = .008). Many of the post–T. foetus inoculation bacterial cultures revealed nonfermentative bacteria (i.e., did not ferment glucose, lactose, or sucrose, and had an alkaline reaction on a triple sugar-iron [TSI] slant). These Gram-negative rod bacteria could not be specifically identified, but the majority were 99% homologous to Sphingomonas sp. by partial 16S rRNA gene sequencing, and one isolate was similar to Acinetobacter sp. Bacteriologic culture of the T. foetus inoculum yielded no growth.
Isolation of bacteria and T. foetus from the vagina before and after intravaginal inoculation of BALB/c mice with T. foetus. ∗
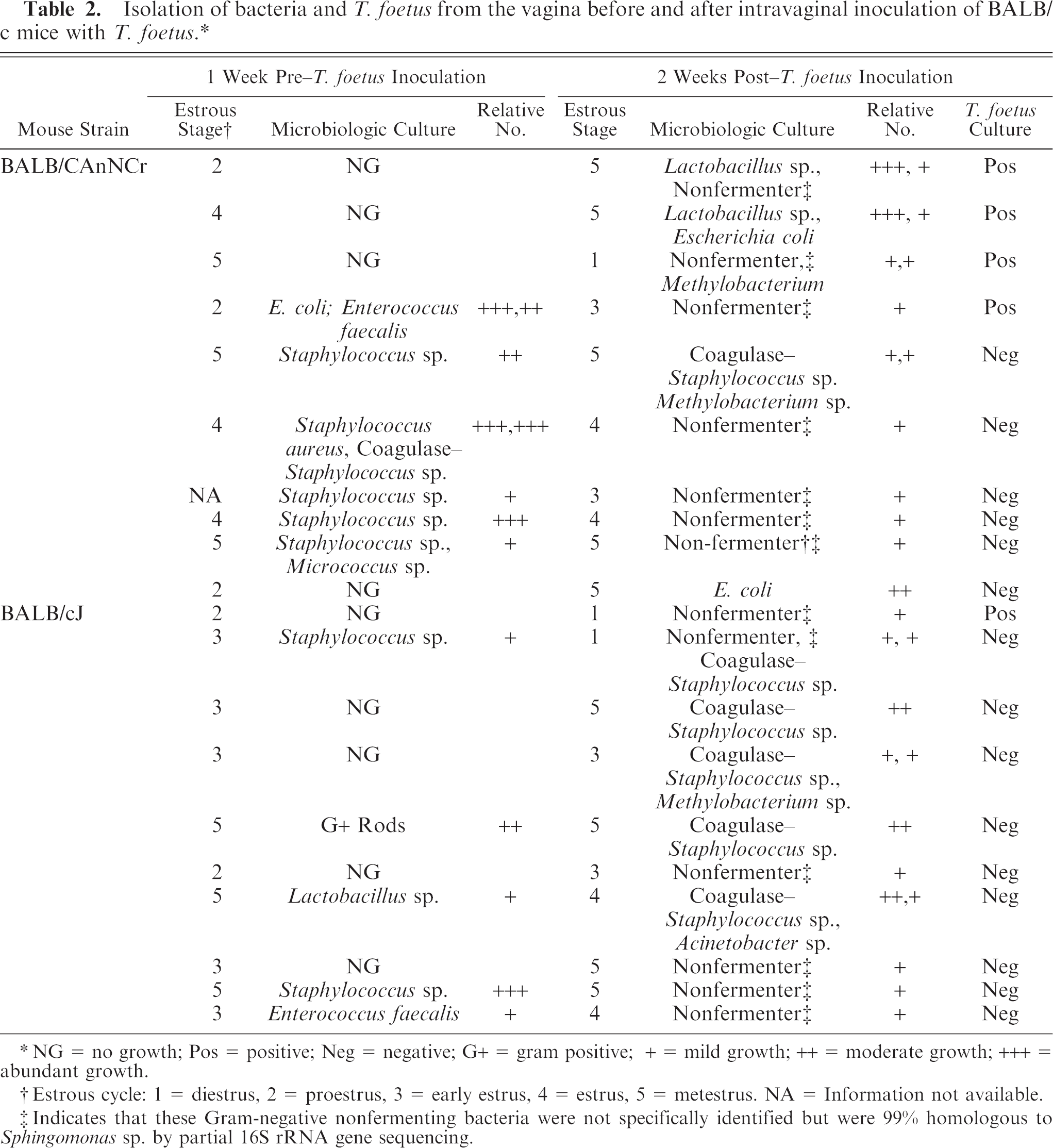
NG = no growth; Pos = positive; Neg = negative; G+ = gram positive; + = mild growth; ++ = moderate growth; +++ = abundant growth.
Estrous cycle: 1 = diestrus, 2 = proestrus, 3 = early estrus, 4 = estrus, 5 = metestrus. NA = Information not available.
Indicates that these Gram-negative nonfermenting bacteria were not specifically identified but were 99% homologous to Sphingomonas sp. by partial 16S rRNA gene sequencing.
Antibody responses to T. foetus infection
After 13 weeks of reproductive infection, there was a strong serum and vaginal IgG response in BALB/cAnNCr and BALB/cJ mice that was significantly higher than uninfected animals (P = .003 for serum and P = .03 for vaginal fluid) (Fig. 1). There was also a marked IgA response in the vagina of infected mice that was greater than uninfected mice (P = .003) but little serum IgA response (Fig. 2). There was only negligible uterine IgG or IgA antibody activity noted in this experiment after 13 weeks of infection. In mice infected for only 2–3 weeks, serum, vaginal, and uterine IgG and IgA responses did not differ significantly from uninfected mice.
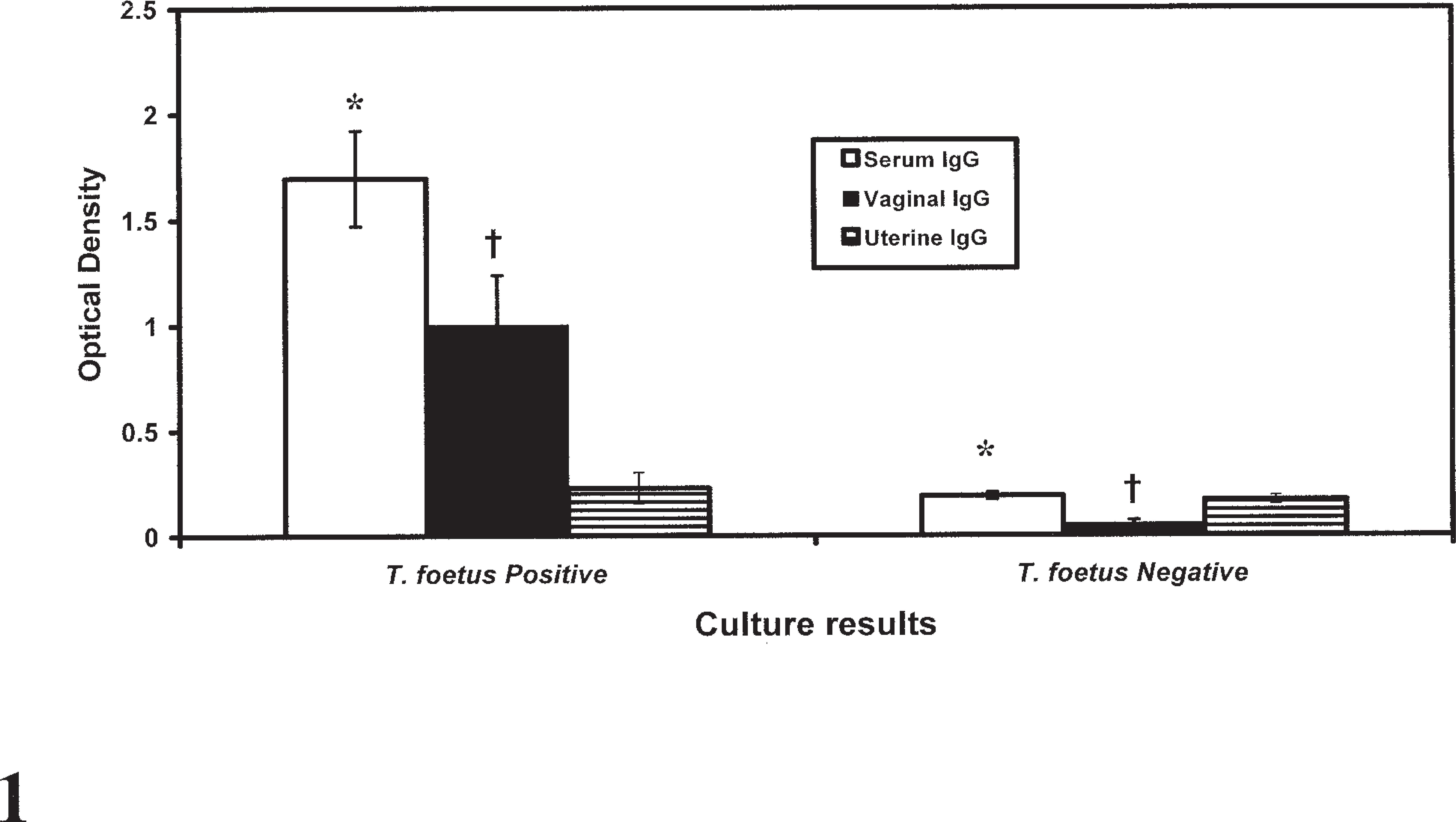
Pooled serum, vaginal, and uterine immunoglobulin (Ig) G responses in 7 BALB/cAnNCr and BALB/cJ mice infected for 13 weeks with Tritrichomonas foetus in the reproductive tract compared with 5 uninfected mice. Bars represent mean ± SEM. ∗ = column values are significantly different (P = .003); † = column values are significantly different (P = .03).
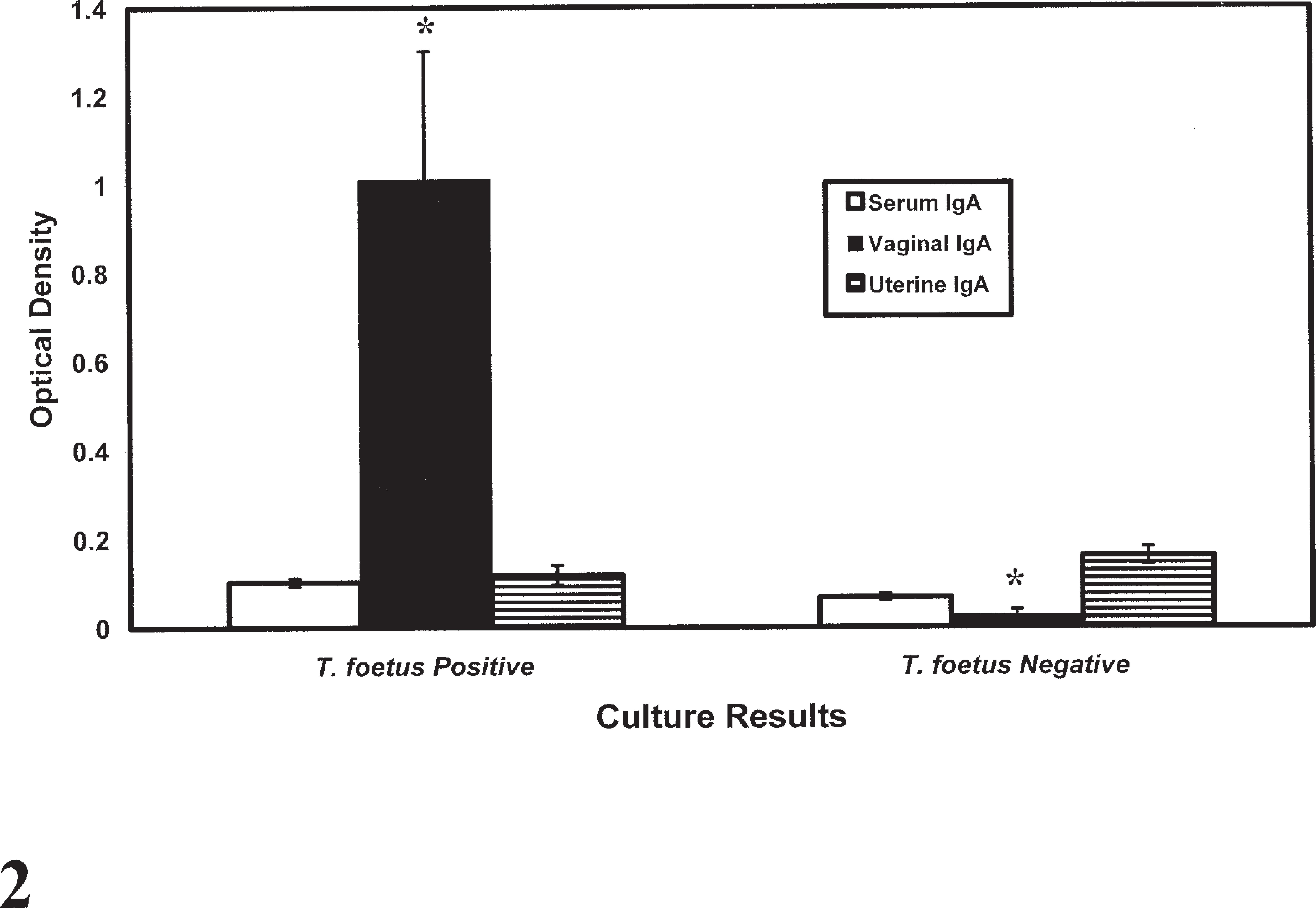
Pooled serum, vaginal, and uterine IgA responses in 7 BALB/cAnNCr and BALB/cJ mice infected for 13 weeks with T. foetus in the reproductive tract compared with 5 uninfected mice. Bars represent mean ± SEM. ∗ = column values are significantly different between groups (P = .003).
Pregnancy results
The pregnancy results are summarized for BALB/cAnN mice infected for approximately 3 weeks or for 12 weeks before mating and are compared with uninfected age- and strain-matched controls (Table 3). Of the BALB/cAnN mice infected for 12 weeks that were bred and had a vaginal plug, only 50% were pregnant at the time of necropsy (3/6), whereas 86% of controls were pregnant (12/14). Mice infected for 3 weeks had a 55% pregnancy rate (5/9). The mice infected for 12 weeks had fewer fetuses per mouse than either the mice infected for 3 weeks or uninfected controls. The difference was significant (P = .04) only for fetuses per mouse in 12-week infected mice versus controls.
The conception rate and number of viable fetuses per mouse in BALB/cAnN mice infected vaginally with T. foetus for approximately 3 weeks or for approximately 12 weeks before mating with B6D2F1 male mice are compared with uninfected age- and strain-matched controls.
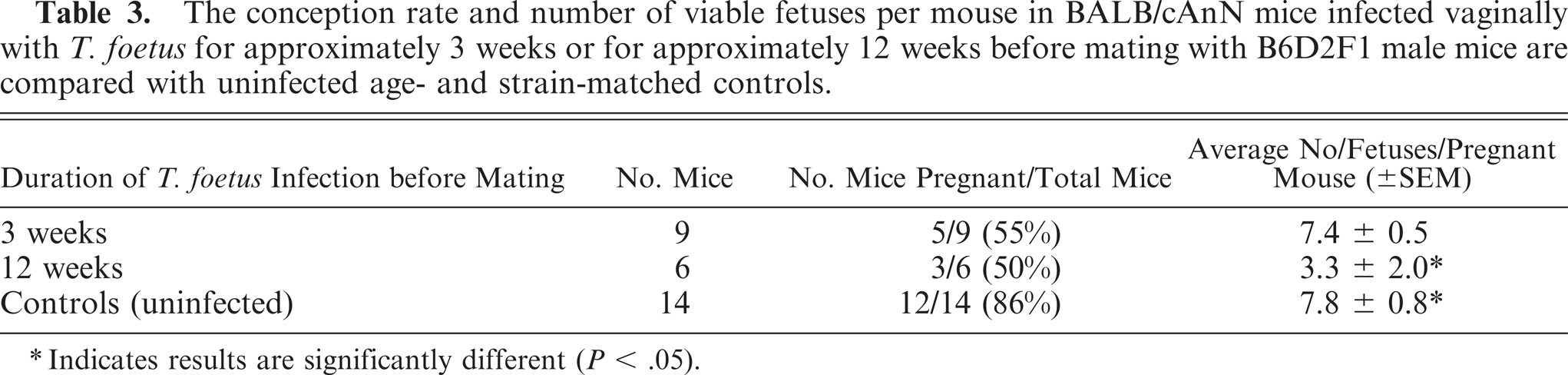
Indicates results are significantly different (P < .05).
Gross lesions
Throughout the experiments, no mice were overtly affected, based on clinical signs, feed intake, grooming behavior, or appearance. Gross examination of internal organs revealed no abnormalities beyond the reproductive tract.
Nonpregnant mouse lesions
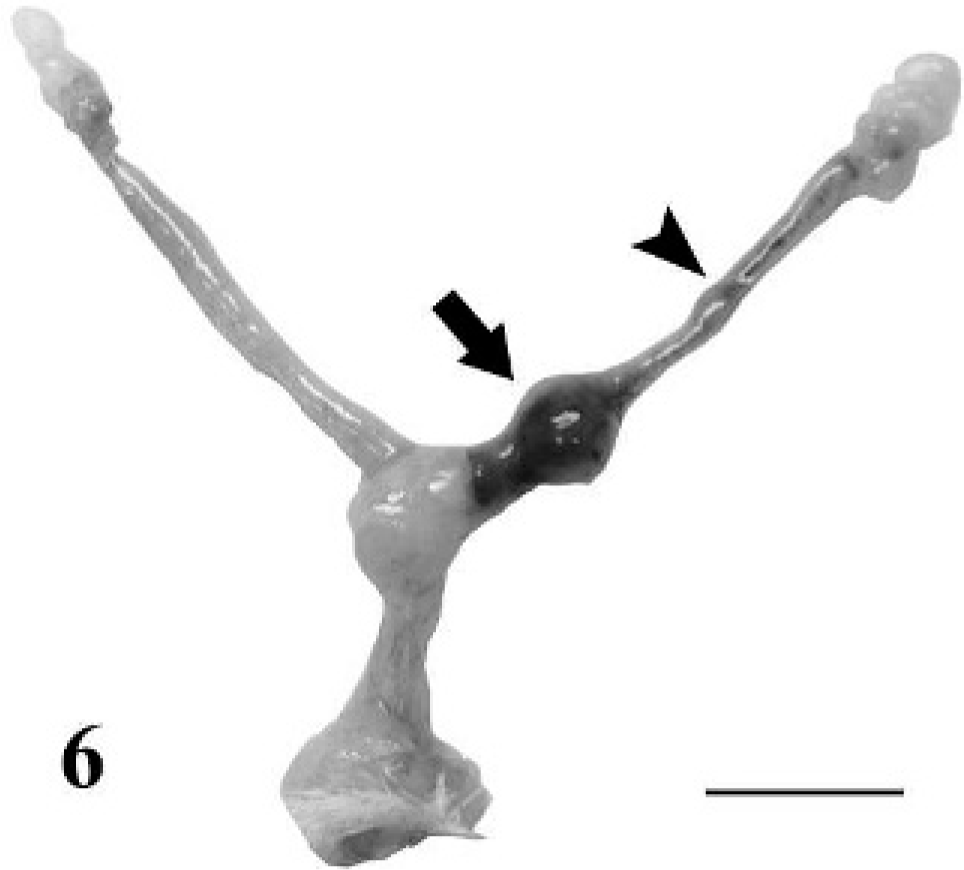
Infected nonpregnant mice frequently had red flocculent vaginal discharge. On gross examination, the reproductive organs of uninfected mice and mice infected for 3 weeks or less were unremarkable, whereas the uteri of mice infected for 13 weeks were all dilated and fluid-filled, with thickened reddened walls (Figs. 3, 4).
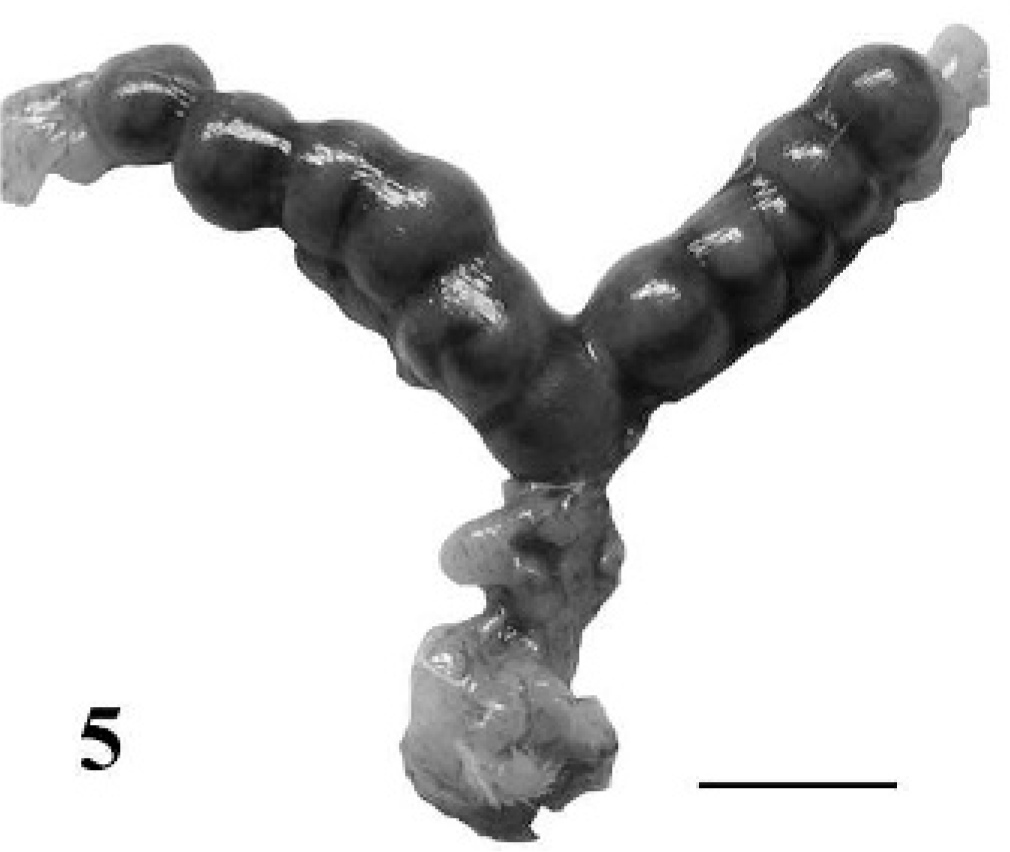
Reproductive tract; normal uninfected nonpregnant BALB/cAnNCr mouse gestation. Bar = 1 cm.
Reproductive tract; nonpregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. Note the thickened uterine horns. Bar = 1 cm.
Pregnant mouse lesions
In pregnant mice, bloody mucus plugs were often present in the anterior vagina, and the fetuses of infected mice were subjectively smaller and more variable in size than uninfected pregnant mice (Figs. 5, 6). In addition, at least 1 blood mole (hemorrhagic and necrotic remnants of a previous implantation site) was present within the uterus of the majority of infected pregnant mice (Fig. 6).
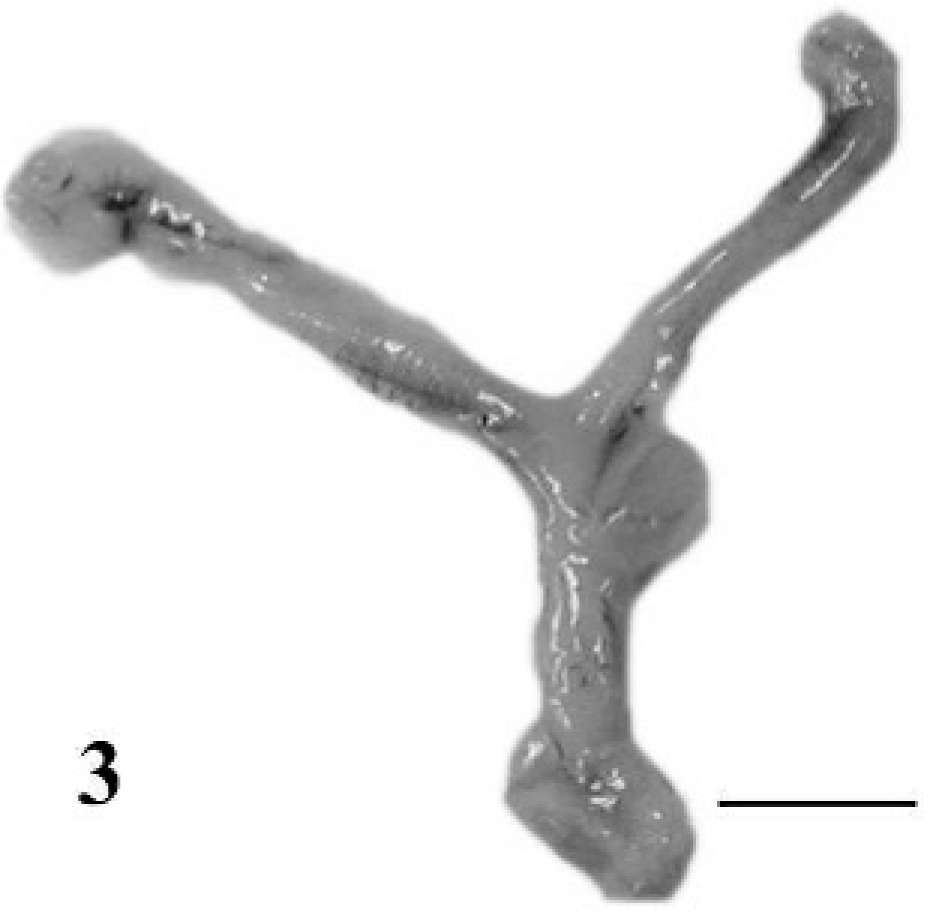
Reproductive tract; normal uninfected pregnant BALB/cAnNCr mouse at 10 days of gestation containing 11 fetuses. Bar = 1 cm.
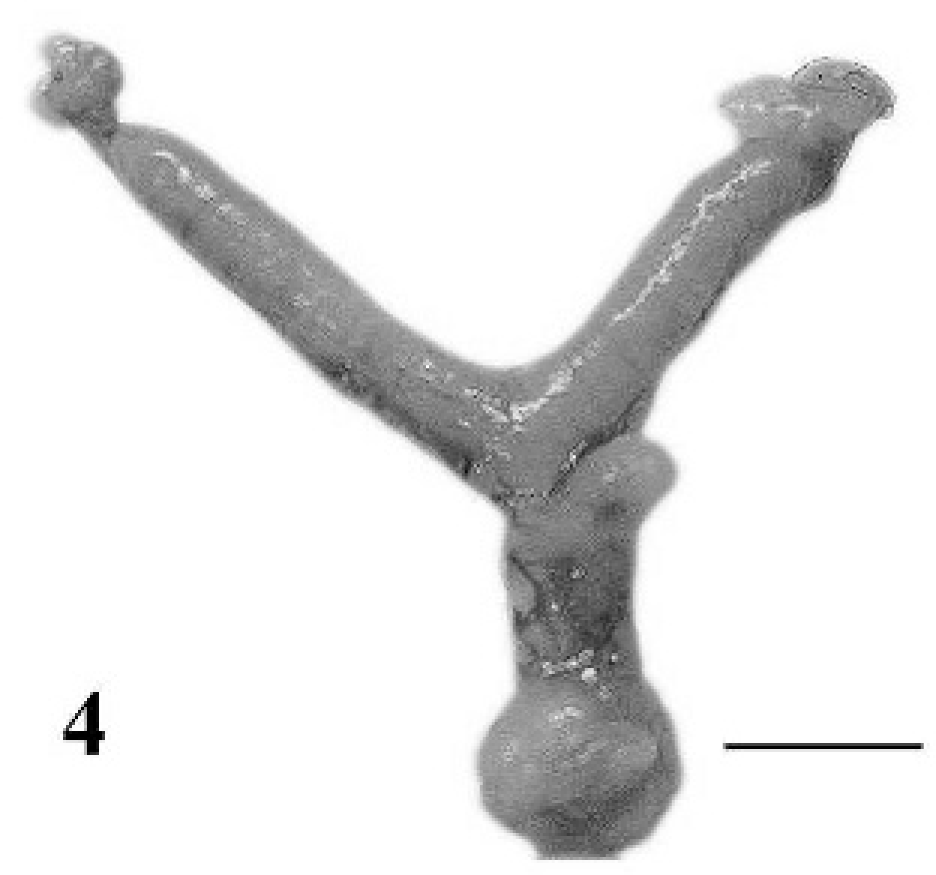
Reproductive tract; pregnant BALB/cAnNCr mouse at 10 days of gestation containing 1 fetus. This mouse was infected with T. foetus 13 weeks earlier. Arrowhead shows a mole and arrow shows a small dying fetus. Bar = 1 cm.
Histopathology
Lesions were similar in the uterus and vagina of all mice, with some variation in the severity.
Nonpregnant mouse uterine lesions
The uterine lumen and many glands were markedly distended by clear fluid in nonpregnant mice infected for approximately 12 weeks (Fig. 7). In some areas, the glands were distended by cellular debris, proteinaceous fluid, and neutrophils (Fig. 8). The endometrial epithelium was occasionally ulcerated or infiltrated by neutrophils and eosinophils. In some areas, the glandular epithelium was markedly disrupted or absent, replaced by cellular debris and neutrophils. The endometrial stratum compactum was multifocally infiltrated by abundant neutrophils, eosinophils, lymphocytes, plasma cells, and macrophages that aggregated around glands. Lymphocytes also formed follicle-like structures in the endometrium of mice infected for 12 weeks (Figs. 7, 8). Eosinophils were commonly found in the endometrial epithelial infiltrates or diffusely scattered in the stratum compactum and spongiosum (Fig. 9). Bodian stain and immunohistochemistry demonstrated numerous trichomonads deep in glands, often amid necrotic debris but rarely in the lumen of the uterus (Fig. 10). In some instances, organisms were noted within the lamina propria adjacent to glands but only in regions where the epithelium was disrupted. In mice infected for 4 weeks or less, there was some dilation of the lumen and a mild diffuse inflammatory-cell infiltrate within the endometrium but no lymphoid aggregates, gland disruption or neutrophil infiltration, or detectable organisms in the glands by immunohistochemistry.
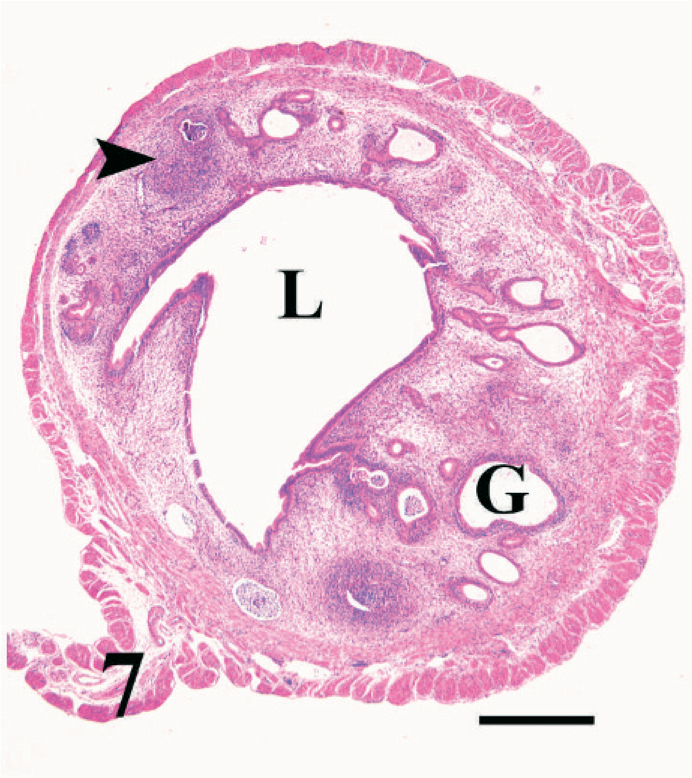
Cross section of uterus; nonpregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. Note the dilated lumen (L) and gland (G). Arrowhead points to a large lymphoplasmacytic aggregate. HE. Bar = 400 µm.
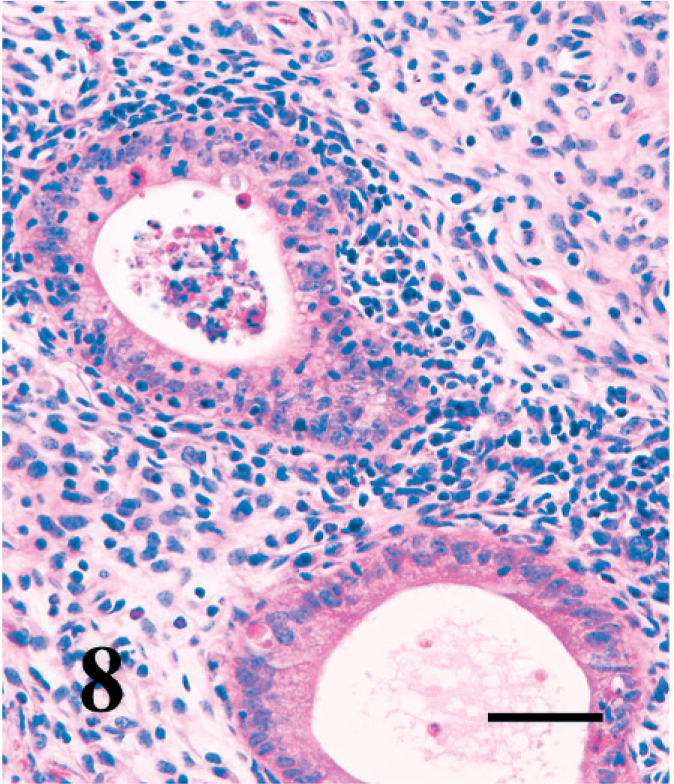
Higher magnification of uterus; nonpregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. There are dense aggregates of lymphocytes, plasma cells, neutrophils, and eosinophils surrounding glands. Glandular epithelium is degenerating and infiltrated by neutrophils and the lumen contains cellular debris. HE. Bar = 60 µm.
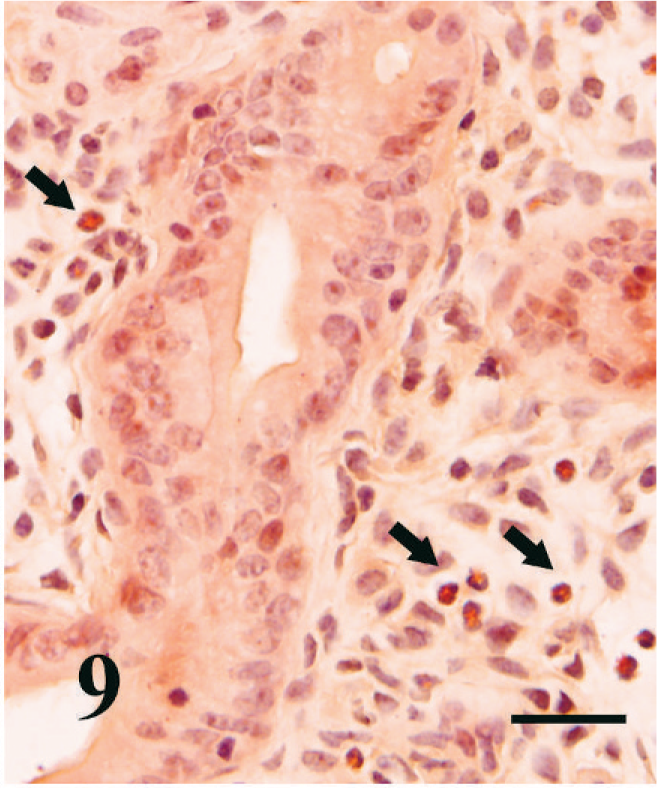
Uterus; nonpregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. Abundant eosinophils (arrows) are scattered throughout the edematous stroma surrounding endometrial glands. Congo Red stain. Bar = 100 µm.
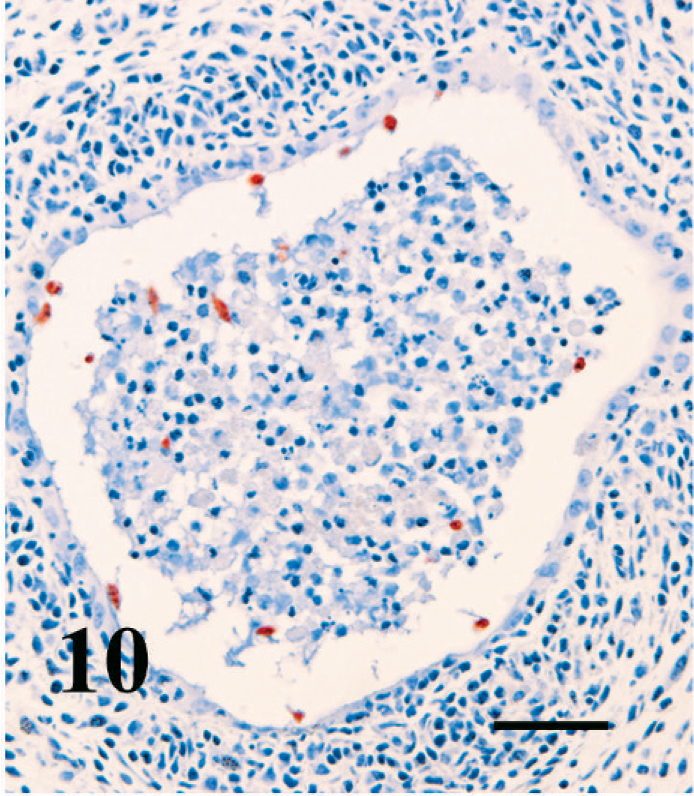
Uterus; pregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. Many T. foetus organisms (stained red-brown) are within the necrotic debris and adherent to the lining of a partially degenerate uterine gland. T. foetus immunohistochemical stain, AEC chromogen, hematoxylin counterstain. Bar = 40 µm.
Pregnant mouse lesions in the uterus
Placental necrosis was present in many of the pregnant mice infected for 12 weeks before breeding. These necrotic moles consisted of retained decidua, rafts of viable and degenerate trophoblasts, occasional neutrophils, homogenous proteinaceous debris, edema, and hemorrhage. Eosinophils were occasionally found in the immediate vicinity of necrotic implantation sites but were more plentiful in other areas of the infected pregnant uteri. Eosinophils were only rarely identified in any of the age- or strain-matched pregnant controls. Organisms were identified by immunohistochemistry in the uterine lumen in proximity to implantation sites, within disrupted glands, and often within the areas of necrosis. Overall lesions were similar in the nonpregnant regions of the uterus to those seen in the nonpregnant mice, although eosinophils appeared less commonly in the endometrium near surviving fetal placental structures.
Most implantation sites in pregnant mice infected for 4 weeks or less were histologically normal; however, in 1 mouse, a mole similar to those seen in 13 weeks infections was present, with marked necrosis of the decidua, neutrophils, disrupted rafts of viable trophoblast cells, fibrin, and hemorrhage (Fig. 11). Amid the debris and necrotic decidua, there were many trichomonads identified by immunohistochemistry (Fig. 11, inset). The adjacent endometrium was also infiltrated by eosinophils. Adjacent to implantation sites in other mice infected for 4 weeks or less, the endometrium and the decidua parietalis were ulcerated and infiltrated by eosinophils, neutrophils, and mononuclear cells that formed an adherent plaque of necrotic cellular debris mixed with numerous trichomonads identified by immunohistochemistry (Figs. 12, 13, and inset). The immediately adjacent delicate fetal membranes were unaffected.
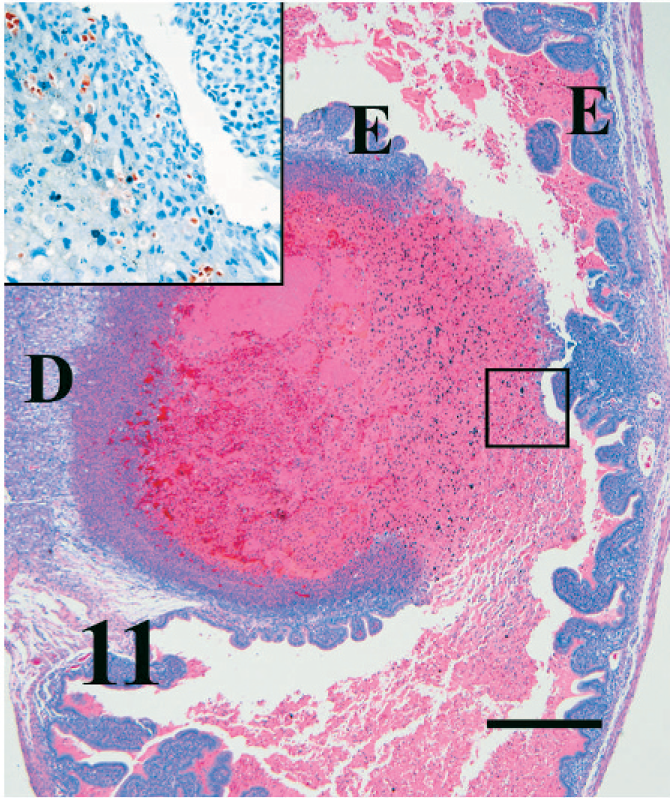
Uterus; pregnant BALB/cAnNCr mouse infected with T. foetus for 4 weeks. A dying fetal placental unit or “blood mole” composed of hemorrhage, necrotic debris, and rafts of inflammatory cells is attached to the remaining decidua (D) and the endometrium (E). Inset shows immunohistochemically stained T. foetus organisms within the necrotic debris (black square). HE. Bar = 300 µm. (Inset bar = 60 µm.)
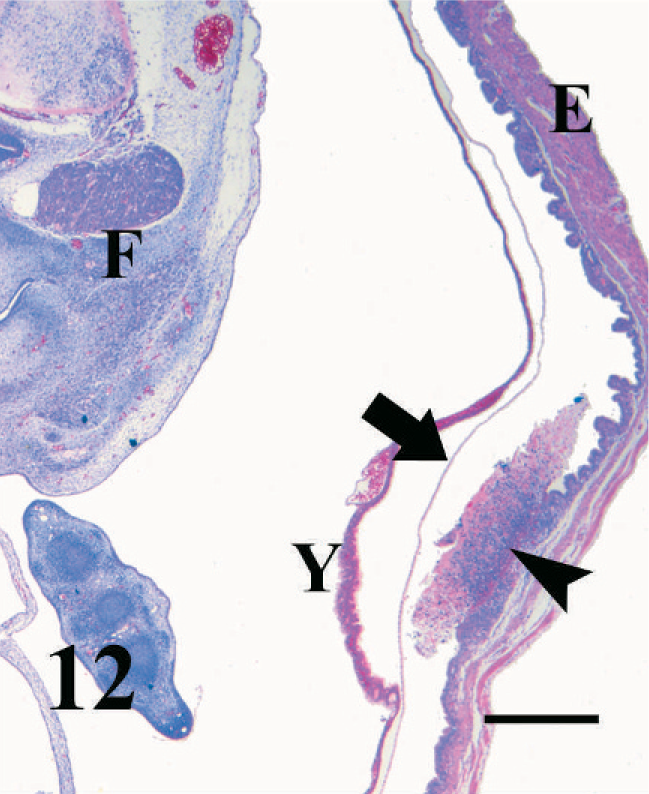
Uterus; pregnant BALB/cAnNCr mouse infected with T. foetus for 4 weeks. The necrotic decidua parietalis (arrowhead) and the underlying endometrium (E) are infiltrated with neutrophils and mononuclear cells while the immediately adjacent chorion (arrow), yolk sac (Y), and fetus (F) are intact. HE. Bar = 200 µm.
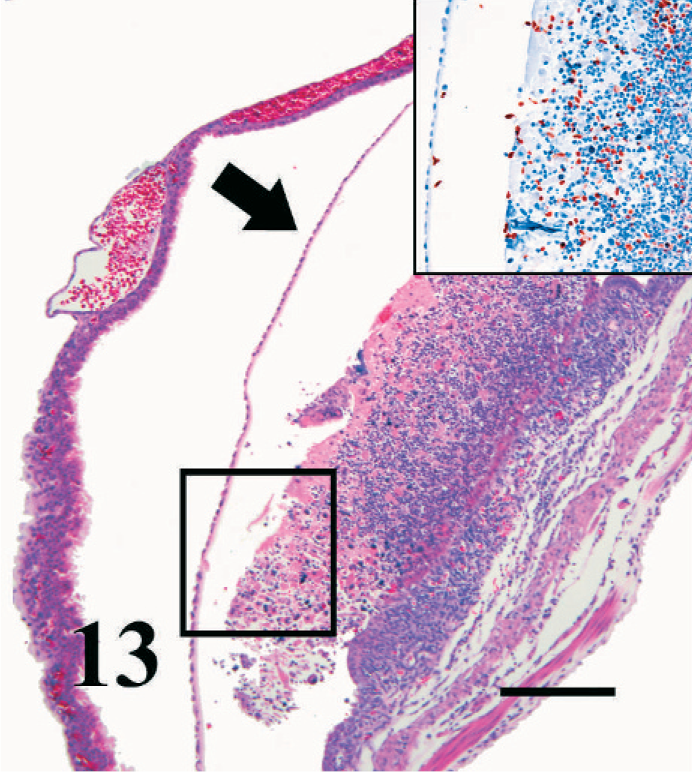
Uterus; pregnant BALB/cAnNCr mouse infected with T. foetus for 4 weeks. A higher magnification of Fig. 12 demonstrates the marked infiltration of inflammatory cells into the decidua parietalis and endometrium, while the adjacent delicate chorionic membrane is intact (arrow). Inset demonstrates by immunohistochemistry the presence of myriad T. foetus organisms within the necrotic decidua and endometrium, within the uterine lumen but not within the fetal placenta. HE. Bar = 80 µm. (Inset bar = 60 µm.)
Vaginal histopathology (pregnant and nonpregnant)
In mice infected for 13 weeks, there was a diffuse increase in cellularity in the submucosa and often in the serosa and associated adventitia, including lymphocytes, plasma cells, and eosinophils (Fig. 14). Occasional regions of epithelial erosion or ulceration were present, associated with neutrophil infiltration. Bodian stain and immunohistochemistry demonstrated trichomonads in the lumen amid mucus, as well deep within folds of vaginal epithelium (Fig. 15). No transepithelial invasion of the mucosa by organisms was noted. No vaginal lesions were observed in mice infected for less than 4 weeks.
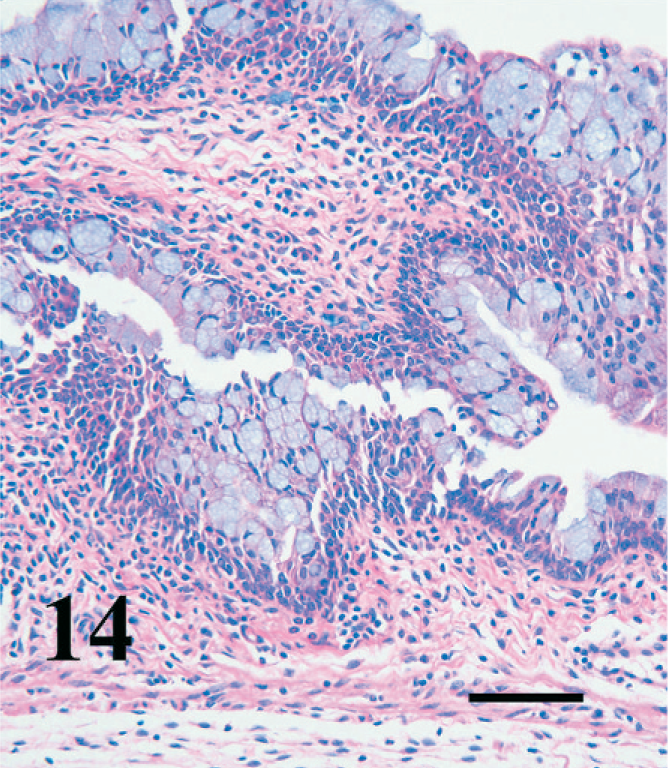
Longitudinal section of vagina; pregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. Note the diffuse hypercellularity of the submucosa and infiltration of the epithelium by inflammatory cells. HE. Bar = 100 µm.
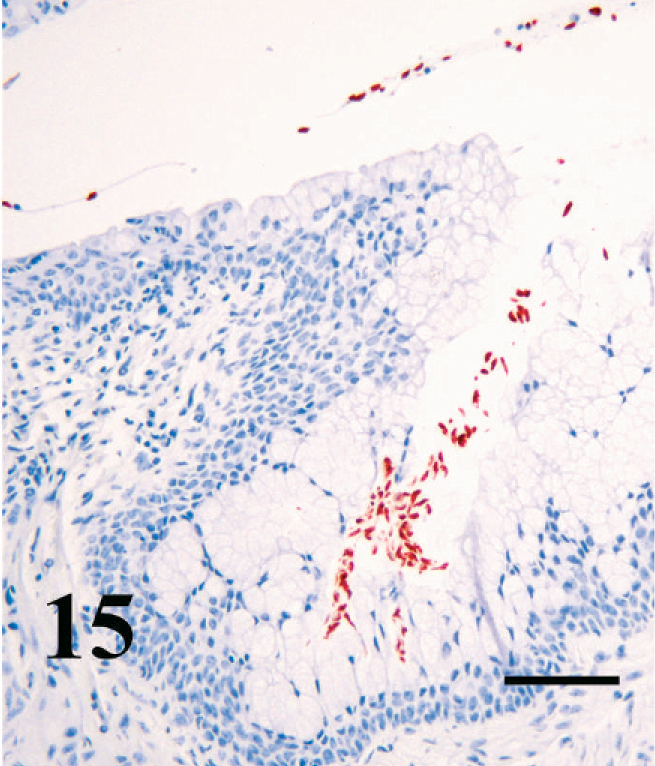
Vagina; pregnant BALB/cAnNCr mouse infected with T. foetus for 13 weeks. Trichomonads are free in the vaginal lumen and deep in vaginal folds. T. foetus immunohistochemical stain, AEC Chromagen, hematoxylin counterstain. Bar = 50 µm.
Discussion
This study shows that, in BALB/c mice, of either BALB/cAnNCr, BALB/cJ, or SCID/NCr strains, intravaginally infecting with a mouse-adapted strain of T. foetus before puberty at 4–5 weeks of age results in a chronic infection in up to 100% of the mice. This infection can persist for at least 13 weeks. These infected mice develop an antibody response and can be successfully bred and pregnancy established. In addition, both pregnant and nonpregnant infected mice have vaginal and uterine lesions similar to the pregnant and nonpregnant cow, respectively. The pregnant mouse model developed in these studies provides a useful tool to study bovine trichomoniasis.
In cattle, there are no breeds that are known to be more or less susceptible to T. foetus. 11 Although breed susceptibility has not been thoroughly tested, variations in susceptibility are thought to be because of environmental or management factors, such as shared grazing, frequent changing of breeding stock, poor fencing, or the use of bulls over 5 years of age. 8, 11 Interestingly, in this study, postpubertal BALB/cJ were more resistant to infection than postpubertal BALB/cAnNCr mice, although the reasons for this were not clear. Possible genetic differences in susceptibility and/or different vaginal microflora are 2 possible explanations for this difference. There is likely considerable genetic difference between these 2 strains, which have been separated genetically since 1940 (MGI, Mouse Genome Informatics, Jackson Laboratories, Bar Harbor, ME), but the possible character of these differences was not addressed in this study. The decreased susceptibility of SCID mice suggests that this variation in susceptibility involves the innate response to parasite infection, because SCID mice retain an intact innate immune system and their defect lies in the acquired immune system. Exploration of the mechanisms involved in the relative susceptibility of mouse strains might prove helpful in developing potential intervention strategies.
The potential role of normal flora in individual cow susceptibility to T. foetus infection has not been evaluated. A few studies have been conducted, however, that evaluated the normal flora in healthy cattle. 4, 19, 44, 45, 65 These studies demonstrated that the dominant bacteria were Streptococcus sp., followed by coagulase-negative Staphylococcus sp. 4, 44, 65 Enterobacteriaceae (in particular, Escherichia coli); members of the genus Lactobacillus were consistently present but in much lower numbers. 44 The number of Staphylococcus sp. bacteria and enterococci was static, from 6 months of age through puberty to 23 months of age, whereas the number of Enterobacteriaceae and lactobacilli increased in numbers over the same period. 44 Similar to cows, the mice in this study frequently had coagulase-negative Staphylococcus sp. in their vaginal flora, with lesser numbers of enterococci and lactobacilli. Unlike cattle, many of the mice had Gram-negative bacteria in their vaginal flora, and streptococci were only occasionally identified.
The bacterial species identified in initial microbiologic culture from the vaginas of mice in this report were similar to those of previous studies, 12, 38, 43, 63 although initial cultures had no growth in almost 50% of the mice, which reflected either 1) improved hygiene in suppliers' breeding facilities since the time these earlier studies were conducted, or 2) the young age of the mice in the current study. After infection with T. foetus, however, there was an increase in the number of bacteria cultured, and many mice acquired Gram-negative nonfermenting bacteria. This alteration in vaginal flora is different from a study in which T. vaginalis was inoculated into 22–24 g adult BALB/c mice, and there was little change in microflora. 38 The inoculum used in the current study was bacteriologically sterile, which suggests that the media, the inoculation process, or the presence of T. foetus changed the vaginal environment that favored the growth of Gram-negative bacteria. Lactobacillus spp. were cultured after T. foetus inoculation from 2 of the 5 mice that maintained T. foetus infection to the end of the experiment and from 1 mouse before inoculation of T. foetus. Because the latter mouse did not maintain the T. foetus infection, the interaction of Lactobacillus sp. and trichomonads is not clear and may not exist. However, all 11 mice that were positive for Staphylococcus sp. either before or after inoculation were negative for T. foetus, whereas in all 5 mice positive for T. foetus, no Staphylococcus sp. were identified in their vaginal flora, which suggests that staphylococci may inhibit T. foetus infection.
Cattle are only naturally infected with T. foetus after puberty because of the parasite's status as an obligate venereal pathogen. The susceptibility of prepubertal cows to experimental infection has not been tested; however, the mice described in this study are only effectively infected if inoculated before puberty. The effect of estrogen on susceptibility is unclear and appears to vary with the study and strain. One previous study demonstrated that high doses of estrogen can make BALB/c mice more susceptible to T. foetus infection, 62 whereas another study showed that estrogen made no difference in the susceptibility of BALB/c mice and even inhibited infection in CD1 mice. 42 A third study showed similar colonization rates in estrogen-treated, stressed, and control BALB/cAnN mice, although the infection was not as persistent in the estrogen-treated mice at 14 and 21 days PI. It is not clear from these earlier studies why in the current study mice less than 5 weeks of age were more susceptible to T. foetus infection when these mice presumably had lowered prepubertal levels of estrogen. 27 Certainly, the interactions between the sex hormones and the reproductive immune system are complex. 66 In fact, these interactions can be contradictory, as illustrated by a recent study of the effects of estrogen treatment on the immune response of T. foetus–infected mice, which showed that estrogen enhanced the inflammatory response and tissue damage but decreased the ability of T. foetus to establish infection. 55 Immune function and resistance to T. foetus infection may be compromised when estrogen levels are either too high or too low. It is also possible that, in these prepubertal mice, the vagina was infected and T. foetus organisms were established deep in the epithelial folds before the first diestrus resulted in a massive influx of neutrophils into the lumen. Trichomonads intravaginally inoculated into postpubertal cycling mice (i.e., older than 7 weeks) would have been exposed to this neutrophilic wave within 4 days or less, the length of a typical murine estrous cycle. Indeed, neutrophil numbers were increased in estrogen-treated and stressed T. foetus–infected mice over controls in a recent study. 55 Cows do not have a similar neutrophilic wave, although they do have a brief postestrual period of endometrial hemorrhage. Presumably, T. foetus organisms may be exposed to neutrophils from the systemic circulation during the flow of a negligible volume of blood during this time, but the relative numbers are likely less than those seen in the vaginal infiltrate of mice. The thickness of the vaginal epithelium is a third factor to consider. The vaginal epithelium in both mice, and to a lesser extent, cows, thickens and keratinizes in response to the estrogen wave of estrus. 36 The epithelium of prepubertal animals and animals in diestrus is usually thin, with only a scant keratin layer, whereas animals under the influence of estrogen have a very thick epithelium. No explanation for the requirement for estrogen for T. foetus infection in some mouse strains has been offered, but it may be that excess keratin protects the organisms from the immune response, provides nutrients for the parasites, or alters the vaginal flora to enhance infection. In vitro studies with bovine vaginal epithelial cells showed that T. foetus adhered at a significantly higher rate to squamous cells than to round, less-mature epithelial cells. 17 Alternatively, estrogen may mute some aspects of the immune response systemically or locally. 66
The antibody results in this mouse study mirror some of the results described in earlier bovine studies. 5, 10 The vaginal antibody response was characterized by elevated IgA levels in vaginal secretions in both cows and mice compared with controls. 10, 18 There was no significant IgG or IgA uterine response in mice in the current study, unlike the previous work by Mutwiri and Corbeil. 42 However, this may be because of the high dilution that resulted from flushing with 1 ml PBS and then diluting 1 : 10 in this study, compared with a 0.1-ml flush per uterine horn in the previous study. Vaginal IgG results in mice reflected IgG1 results from cattle when measured by a similar ELISA method, 10 although the marked serum IgG response in mice differed from cattle. 10 Similar to an earlier study in which BALB/cJ and CD1 mice (acquired from Harlan Sprague Dawley, Inc., Indianapolis, IN) were vaginally infected with the T. foetus D1 strain, 42 there was no antibody response in the serum or vaginal secretions at 2–4 weeks. A strong vaginal IgA response was detected at 13 weeks in this study and at 16 weeks in the CD1 mice but not the BALB/c in the study by Mutwiri et al. 42 The antibody response, however, did not clear the infection in this study. In fact, mice with active infections in the present study were the only animals to show an antibody response, which reflected the need for a prolonged antigenic stimulus to induce an antibody response. Whether the response would have eventually cleared the infection is not known. The fact that the mice that do clear the infection do so within the first week before acquired immune responses are induced, suggests that the innate immune system plays the more important role in parasite clearance during the early stages of infection in this model.
In naturally infected cattle, the most prevalent sign is return to estrus after natural service, i.e., multiple services per successful conception. 11 In 1 study, most pregnancy loss was detected during the first 2 to 3 months of infection, which suggests that most of the pregnancy loss occurs at the end of the first trimester. 11, 46 Later T. foetus–induced abortions occur but are less common. This is similar to the mice in this study in which there was a marked decrease in the percentage of dams pregnant and the number of fetuses per dam. Some dying mid gestational fetuses alongside viable fetuses suggested that pregnancy loss occurred at early or mid gestation.
The gross and histologic lesions of trichomoniasis in both naturally and experimentally infected cattle have been described. In 2 studies, cows that were simultaneously infected and bred did not develop endometrial lesions until approximately 8–9 weeks PI. 5, 46 Another study examined experimentally infected nonpregnant heifers at 10 weeks PI and found lesions similar to the previous study. 5, 18 An additional study looked at field cases at various times in the disease course. 24 The endometrial lesions were characterized by lymphoplasmacytic and eosinophilic endometritis; lymphoid aggregates surrounding glands; and glandular dilation, neutrophils infiltration, and necrosis; and, in very severe cases, pyometra. 5, 24, 46 Placentas were examined in 2 other studies of field cases from a diagnostic laboratory. In these cases, the fetuses and their placentas were aborted, and the study found the placentas to be variably affected, with mixed inflammatory infiltrates and only mild-to-moderate focal necrosis of chorionic epithelium. 53, 54 Trichomonads were rarely seen in the placental epithelium but were identified along with inflammatory cells in the chorionic stroma. 53 In another study that involved experimental infections, trichomonads and inflammatory cells were identified within the placentome. 46
In the present mouse study, vaginal lesions were minimal to mild, with only occasional areas of suppuration, erosion, and ulceration. Endometrial lesions in nonpregnant and pregnant mice after 13 weeks PI consisted of severe lymphoplasmacytic endometritis, with mononuclear-cell aggregation around glands, formation of lymphocytic nodules in the stratum spongiosum and along the serosa, and glandular abscesses and obliteration similar to the cow. Similar, but less striking lesions were seen in the nonpregnant and pregnant mice infected for 4 weeks, which suggests that there is a progression of severity over time, much like that seen in cattle in which lesions are not seen until after 8–9 weeks PI. 5, 46 Future studies should be able to better pinpoint the timing of lesion development in mice between 4 and 13 weeks PI to allow better modeling of the disease in cattle. The gross and histologic lesions associated with chronic intravaginal and intrauterine T. foetus infection seen in the nonpregnant animals in this study were also similar to those described in earlier reports that used an estrogenized mouse model. 37, 64 Any study that involves mice must, however, carefully interpret inflammatory changes in light of the normal and expected cyclic flux of neutrophils and eosinophils in the vagina and, to a lesser extent, the uterus.
It is particularly interesting to note the apparent predilection of T. foetus organisms for endometrial tissue in the current study. In several implantation sites, abundant T. foetus organisms are found amid necrotic debris and adhered to ulcerated endometrium. In many areas of the uteri of pregnant mice infected for both 4 and 13 weeks, delicate fetal membranes were adjacent to myriad trichomonads, endometrial necrosis, and inflammatory debris, and yet remained histologically normal. Trichomonads were only identified within placental or fetal tissue when the implantation site was necrotic, which suggests that T. foetus may not be invasive before tissue damage in this model. However, it could be that trichomonads were only detectable in tissue on histologic examination when the numbers were high and that this high concentration of trichomonads in tissue would secondarily result in tissue damage. There have not been any studies to determine how and at what speed T. foetus infection progresses through the uterus; this could also potentially explain regional differences in uterine damage. It is possible that the bovine placentas in the previous studies were infiltrated by trichomonads after their integrity had already been compromised by direct cytotoxicity or secondary to endometrial damage. 53, 54 Further investigation of these observations in both mice and cattle may offer more insight into the precise cause of fetal death in cattle.
Eosinophils may play a role in the pathogenesis of trichomoniasis, as well as in the protection of the uterus from infection. Their prominence in T. foetus infection in cattle has been noted in several studies. 1, 5, 16, 18, 46 Their tissue damaging granules can have bystander effects on local tissues and marked vascular effects that lead to edema, although a specific role of eosinophils in trichomoniasis has not been determined. 26 However, their vascular effects could well contribute to the transport of antibodies to the uterine and vaginal lumen, and provide some protection from infection and later clearance of trichomonads. In vitro studies demonstrated that eosinophils may be involved in killing T. foetus. 20 Another recent study examined histologic and ultrastructural lesions in nonestrogenized but nonpregnant mice. 40 The latter study demonstrated endometrial gland disruption, neutrophil infiltration, edema, and infiltration of macrophages and eosinophils, as in the current study. Ultrastructural examination demonstrated phagocytosis of T. foetus and granule release by eosinophils. 40 This study did not, however, account for the eosinophilic fluctuations characteristic of the normal mouse estrous cycle. Eosinophils are normally found in abundance in the mucosa of the vagina and uterus of mice throughout the estrous cycle, but they are notably absent in the stroma of normal pregnant uteri and are found only in the myometrium. 56 In the current study, most of the pregnant infected mice had eosinophils throughout the endometrium and myometrium, and in the serosa, although there were fewer in proximity to viable implantation sites, which suggests the powerful local control the fetus can have over eosinophil migration. Whether increased numbers of eosinophils in the infected pregnant uterus are pathogenic or protective is not known. Many knockout mouse strains with compromised eosinophil function are available to further investigate the role of eosinophils in trichomoniasis.
One previous study in cows suggests that T. foetus does not interfere with conception or blastocyst development, at least not when fertilization, early development, and T. foetus exposure occur in vitro. 7 In the study reported here, there was a reduction in the number of implantation sites in mice infected for 12 weeks, but it could not be determined whether this was pre- or postimplantation pregnancy loss. The current report is the first to document lesions associated with T. foetus infection in the pregnant mouse and demonstrated that experimental T. foetus infection can cause pregnancy loss similar to the cow, in spite of differences in placental structures in mice and cattle. This pregnancy loss in mice appears to be associated with interference at early to mid gestational effects, because there are fewer pregnant animals, fewer fetuses per breeding, and evidence of dead and dying fetuses in infected dams.
Historically, it has been difficult to study the relative roles of the parasite and host response in pregnancy loss, the most economically important outcome of natural infection with T. foetus. Research has been hampered because of the logistic and economic challenges of working with the natural host and the inability to successfully create a laboratory animal model that includes pregnancy. Long-term infection of prepubertal BALB/cAnNCr mice before mating allows the development of lesions and antibody responses. This provides a model for study of these factors in pathogenesis. The model should also be useful for investigation of potential intervention strategies, including vaccines.
Footnotes
Acknowledgements
We thank Karen Sverlow of California Animal Health and Food Safety laboratory for her expertise in histochemical and immunohistochemical staining. In addition, we thank Spencer Jang for culturing mouse vaginal specimens, Karen Van Hoosear and Alex Friend for assistance in running antibody ELISAs, and the animal care staff at the Center for Laboratory Animal Care at UC Davis for maintaining the mouse colonies. This work was supported in part by the Center for Food Animal Health, under the Formula Funds program of the California Agricultural Experiment Station. In addition, this publication was made possible by grant number T32 RR07038 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of NCRR or NIH.
