Abstract
Digital dermatitis is an infectious cause of lameness primarily affecting cattle but also described in sheep, goats, and wild elk. Digital dermatitis is a polymicrobial infection, involving several Treponema species and other anaerobic bacteria. Although the exact etiology has not been demonstrated, a number of bacterial, host, and environmental factors are thought to contribute to disease development. To study host–bacterial interactions, a reproducible laboratory model of infection is required. The objective of this study was to demonstrate key aspects of bovine digital dermatitis lesions in an easy-to-handle sheep model. Crossbred sheep were obtained from a flock free of hoof disease. Skin between the heel bulb and dewclaw was abraded before wrapping to emulate a moist, anaerobic environment. After 3 days, abraded areas were inoculated with macerated lesion material from active bovine digital dermatitis and remained wrapped. By 2 weeks postinoculation, experimentally inoculated feet developed erosive, erythematous lesions. At 4 weeks postinoculation, microscopic changes in the dermis and epidermis were consistent with those described for bovine digital dermatitis, including erosion, ulceration, hyperkeratosis, ballooning degeneration of keratinocytes, and the presence of neutrophilic infiltrates. Silver staining of lesion biopsy sections confirmed that spirochetes had penetrated the host epidermis. The model was then perpetuated by passaging lesion material from experimentally infected sheep into naïve sheep. This model of bovine digital dermatitis will allow for future novel insights into pathogenic mechanisms of infection, as well as the development of improved diagnostic methods and therapeutics for all affected ruminants.
Bovine digital dermatitis (BDD) is the leading cause of infectious lameness in cattle worldwide. A major concern in dairy cattle, it is also an increasing problem in beef cattle. Digital dermatitis causes significant economic losses through decreased production (lost weight gain or milk yield), decreased reproductive efficiency, increased culling rate, and costs associated with treatment or prevention. 3,7,17,27,36,49 Additionally, consumer interest in the welfare of production animals raises concerns with respect to the occurrence and prevention of lameness in production systems that source their food. 20
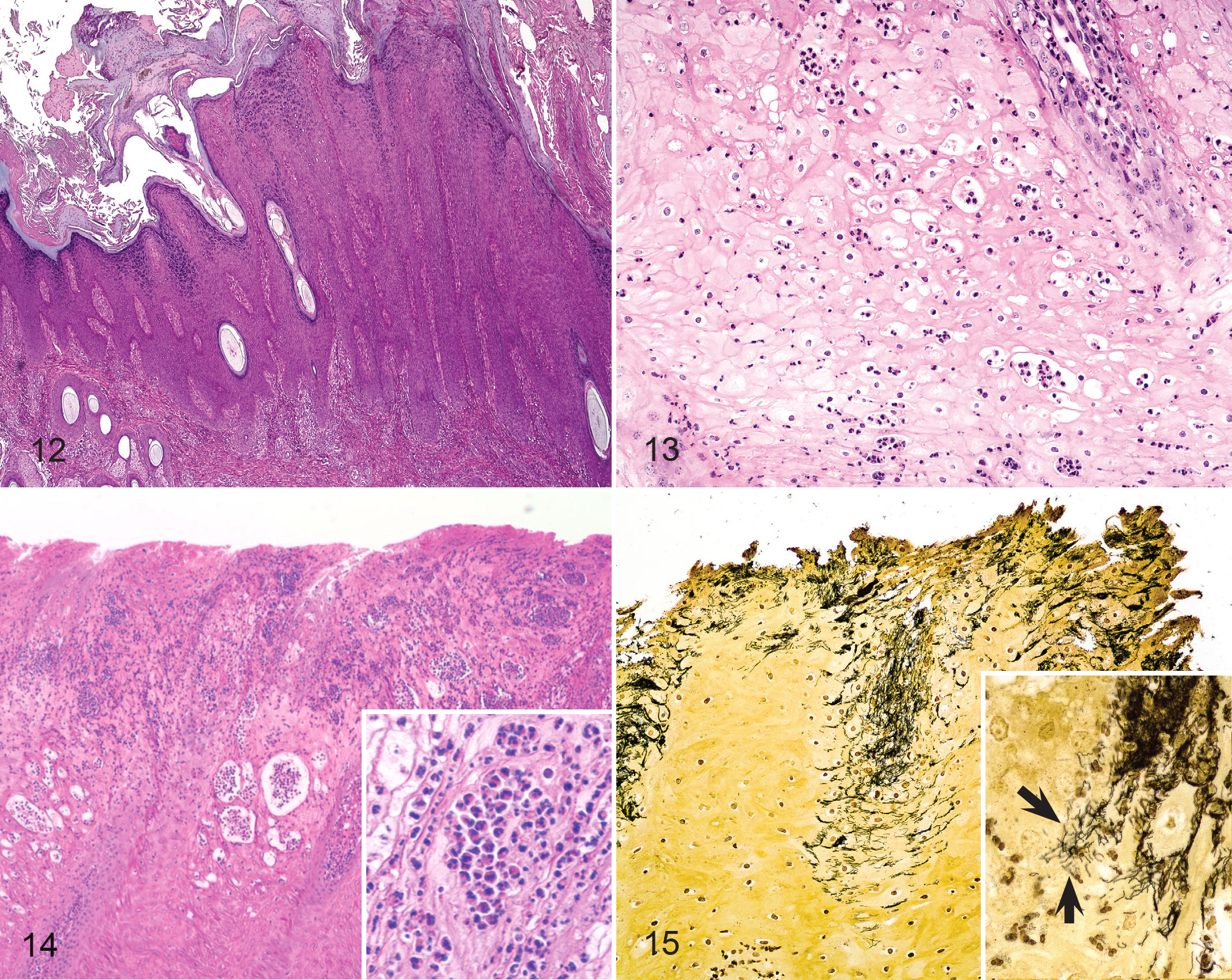
Bovine digital dermatitis has been defined as a circumscribed plaque of eroded acanthotic epidermis with parakeratotic proliferation. 35 Characteristic histologic lesions include dermal infiltration of neutrophils, plasma cells, lymphocytes, and eosinophils and show epidermal invasion of the stratum spinosum by spirochete bacteria. More developed lesions are marked by localized necrosis and hyperplasia of the epidermis, ballooning degeneration of epithelial cells, necrotizing vasculitis, and intralesional bacteria including spirochetes. 45
Considered a polymicrobial bacterial disease, the exact etiology of digital dermatitis remains unclear. Many studies have identified a large number of species from the genus Treponema in BDD lesions including Treponema phagedenis, Treponema pedis, and Treponema medium, contributing to lesion persistence. 22,26,31,37,51 The high incidence of BDD in first-calf heifers following introduction into the herd also suggests that digital dermatitis is highly contagious in nature, and other host susceptibility factors are potentially involved. 6 Genomic analysis suggests that between 3 and 14 phylotypes or genomogroups of Treponema can be found within BDD lesions; however, only a few of these phylotypes or species have been isolated and maintained in culture, potentially because of difficulty in culture and isolation of anaerobic Treponema. 9,47
Digital dermatitis diseases have been described in other ruminants: sheep and goats in the United Kingdom and Ireland, buffalo (Bubalus arnee bubalis) in Italy, and recently, in wild North American elk (Cervus elaphus). 2,10,18,19,38 Although somewhat different gross clinical presentations were observed, key aspects of digital dermatitis were present in each host species including histopathologic changes, presence of multiple Treponema species, and presence of a robust serum antibody response to Treponema antigens.
Only 2 descriptions of experimental BDD induction models have been published in the peer-reviewed literature. 16,25 Gomez et al 16 utilized lengthy wrapping of calves’ feet and repeated exposure to macerated lesion material. Krull et al 25 utilized large numbers of Holstein calves with a simplified wrapping procedure, abrasion of the tissue, and single inoculation with macerated lesion material. Several attempts to reproduce BDD with individual Treponema strains have met with little (Gomez), questionable (Krull), or no (D. P. Alt, unpublished data) success. 16,25,34 Thus, there remains a need to develop a model of BDD that would allow investigation of the role of different bacterial species, the interaction among bacteria during infection, and the complex host and environmental factors that contribute to BDD.
As research subjects, cattle require specialized equipment for safe restraint and lifting to inspect their feet and are expensive to procure, house, and feed. Rodent models have inherent limitations and differ greatly from cattle in their anatomy and physiology. The ruminant immune system is unique because of large populations of circulating γδ T cells, which have various innate immune, regulatory, and memory functions. 42 Sheep, as an alternate ruminant, provide advantages with respect to housing, handling, and restraint and allow study of the ruminant immunologic response during disease development. As digital dermatitis has already been described in sheep, we hypothesized that sheep would be susceptible to BDD organisms, and histopathology, bacterial persistence, and immune induction to BDD bacteria could be observed in sheep. Our objective was to develop a reproducible infection model of BDD induction that demonstrates key aspects (histopathology, presence of spirochetes, and induction of systemic immune response) of the bovine disease in a feasible animal model. Herein, we describe induction and perpetuation of lesions with hallmarks consistent with BDD in an ovine experimental infection model.
Methods
Animals
White-faced mixed-breed and crossbred sheep (Ovis aries), ranging in age from 6 months to 2 years, were obtained from a herd free of hoof disease. The majority of the animals were less than 1 year of age and in growing phase. Sheep were both castrated males and nonpregnant, nonlactating females. Before initiation of the study, hooves were trimmed and examined for signs of disease, abnormalities, and soundness. Animals with any disease or abnormalities (ie, orf, hoof-rot, etc) were removed from the study. Sheep were housed in an outdoor concrete paddock with deep straw or corn-stover bedding under the covered half of the paddock (Supplemental Fig. S1), with free access to water and grass hay. To emulate the low energy balance of high production dairy animals in the early lactation cycle, no concentrate or grain was fed during the study. Body condition was monitored by visual inspection or by palpation of the backbone and assessed using a 5-point scale (http://smallfarms.oregonstate.edu; accessed 12/28/15). Animals were maintained at body condition scores of 2. Sheep were nonrandomly assigned to 2 treatment groups; to evenly distribute variances in body size and ages across treatment groups, sheep were sorted largest to smallest and every third sheep was selected for the mock-infected group. Size assortment was repeated with a new assortment of sheep for each experiment. All experimental groups (inoculated and mock-inoculated) were housed in the same pen.
All animal procedures were approved by the National Animal Disease Center Institutional Animal Care and Use Committee (Animal Use Protocol No. ARS2790) in accordance with the standards established by Public Health Service Policy “US Government Principles for the Utilization and Care of Vertebrate Animals Used in Testing, Research, and Training,” the Guide for the Care and Use of Laboratory Animals (National Research Council), the Animal Welfare Act (1966), and the Guide for the Care and Use of Agricultural Animals in Research and Teaching (USDA, Federation of Animal Science Societies).
Experimental Overview
The experiment was performed in 2 phases, with mock-inoculated animals included in each phase. The first phase comprised induction of lesions in sheep with bovine-derived digital dermatitis lesion material (8 inoculated sheep and 4 mock-inoculated controls). Two feet (hind limbs) were used on each sheep, with observations being made at both the sheep and foot levels. These animals were inoculated 3 days after being prepared and wrapped as described below. One foot of each animal was unwrapped and observed at 2 weeks postinoculation (PI), and the wrap was replaced. Animals in the first phase were euthanized at 4 weeks PI.
The second phase used lesion material derived from experimentally infected sheep in the first phase as the source of inoculum (4 inoculated sheep and 2 mock-inoculated controls). One foot from each of these animals was observed at 2 weeks PI and the wrap replaced. The opposite foot was unwrapped and observed at 4 weeks PI. At 4 weeks PI, a biopsy was taken of the lesion (described below) and the wrap replaced. At 6 weeks PI, wraps were removed from both feet for observation of lesion formation, and a biopsy taken from 1 foot. Wraps were not replaced in order to observe natural progression or healing of the lesions without any direct treatment. All animals were euthanized at 10 weeks PI. This demonstrated the ability to perpetuate lesion induction using the experimental model. Group sizes and infectivity time (weeks PI) were based on the high infectivity rate observed in a calf induction model of digital dermatitis with a < 80% infectivity rate and average time of lesion development of less than 4 weeks PI. 25
Preparation and Wrapping of Inoculation Site
Three days prior to inoculation, hair and wool on both hind legs was clipped to above the hock. Sheep were restrained using a sheep-specific tilt-table (Sydell Inc., Burbank, SD), inverting the animal and allowing access to feet and legs. Two regions, each approximately 1.5 cm2, on each side of the pastern area, midway between the dewclaw and heel bulb and just above the coronary band, were abraded using a tungsten disk. 25 The intent was to remove the stratum corneum and epidermis without causing bleeding or disrupting the dermis (Fig. 1). Cotton cast padding, approximately 40 × 40 × 10 mm, was saturated with a mixture of 50% phosphate buffered saline (PBS) (pH 7.2) and 50% clarified sterile bovine rumen fluid (collected and processed in-house) and placed over the abraded area and between the heel bulbs (Fig. 2). 40 A piece of soft tubing (butterfly catheter with needle portion removed) was sandwiched in duct tape and placed so that the tip was over the cotton padding (Fig. 2). Cotton and tubing were secured with a layer of brown “cling” gauze (Jorgensen Laboratories, Loveland, CO), followed by plastic film (Fig. 3) applied to retain moisture and exclude air. Next, a layer of Vetwrap® (3M Animal Care Products, St Paul, MN) (Fig. 4) was applied for cushioning followed by a layer of Gorilla Tape® (Gorilla Glue, Cincinnati, OH) (Fig. 5). An additional round of Vetwrap was used to secure and cover the injection port of the catheter (Fig. 6). Wraps were checked twice per week for integrity. Gorilla Tape was removed and reapplied as needed. Every 7 ± 2 days, 5 ml of anaerobic medium mixture (50% fastidious anaerobe broth [Difco, Livonia, MI] supplemented with 0.01% L-cysteine, 25% clarified bovine rumen fluid, 25% PBS) was added via the catheter tubing to maintain the wet, anaerobic environment in the pastern region.
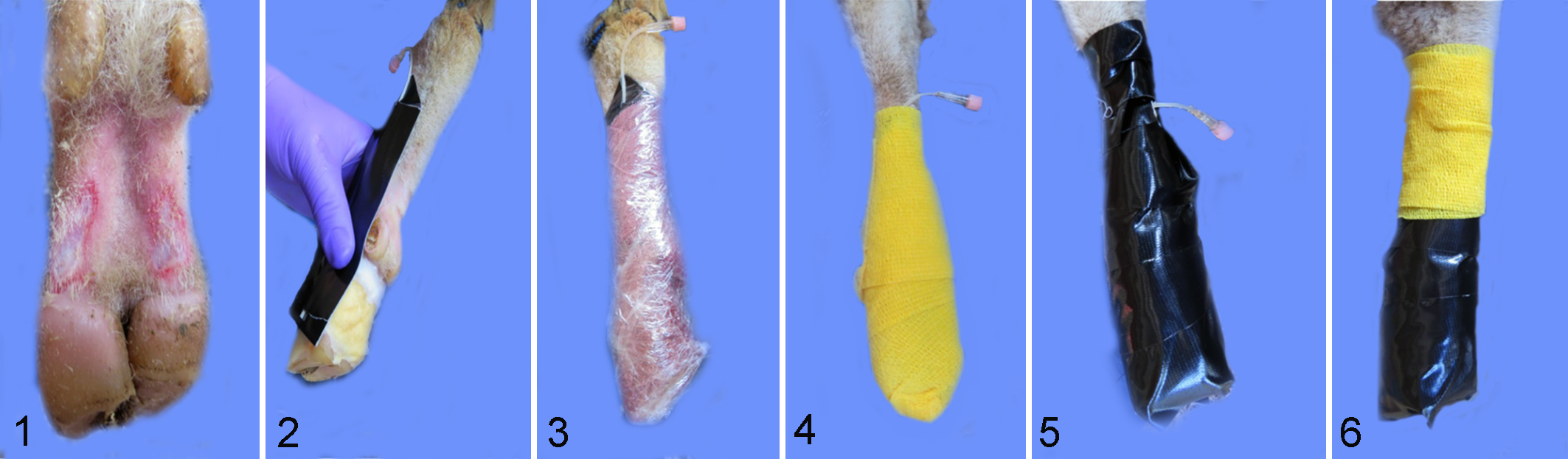
Inocula Preparation
An inoculum from active BDD lesions was prepared according to published methods. 25 Briefly, lesion material was collected from dairy cattle housed at the Iowa State University Dairy Research and Teaching Facility exhibiting lesions with scores of 1 to 4 (Iowa scoring system 25 ), placed in an anaerobic environment, and minced finely in the presence of a sterile growth medium (40% MTGE [Anaerobe Systems, Morgan Hill, CA], 30% brain heart infusion broth [BD and Company, Sparks, MD], 15% trypticase arginine serine broth, and 15% Mueller Hinton broth [BD and Company]) containing 20% fetal bovine serum. 25,26 The resulting suspension was strained through a 70-μm mesh filter and stored at –80°C. Prior to use, the inoculum was thawed in an anaerobic environment and placed in 1-ml syringes for inoculation. The bovine-derived lesion inoculum contained T. phagedenis, T. medium, T. pedis, Fusobacterium necrophorum, and Dichelobacter nodosus but not T. denticola or T. vincentii as detected by polymerase chain reaction (PCR) (details below).
Inoculum prepared from induced sheep lesions was prepared in a similar manner. Sheep with experimentally induced lesions (first experiment phase) were euthanized by intravenous administration of sodium pentobarbital. After euthanasia, both hind limbs were severed at or below the hock to include the inoculated area, placed in a plastic bag, held on ice, and transported to the laboratory. All bandages and dressings were removed, hair clipped if needed, and feet examined. Tissue with gross lesions (approximately 2 × 3 cm2) was removed and transferred to an anaerobic chamber (Coy Laboratories, Grass Lake, MI). Tissues from several animals were pooled and minced with a scalpel in the presence of oral treponeme enrichment broth (OTEB; Anaerobe Systems) (1 ml broth/g tissue). The resulting suspension was pressed through a 70-μm mesh filter and fetal bovine serum was added to a final concentration of 20%. The resulting inoculum was examined using dark-field microscopy and the total spirochete numbers were estimated. Adjustments to the final suspension volume were made to obtain approximately 1 × 108 spirochetes/ml. Aliquots of inocula were loaded into 1-ml syringes in the anaerobic environment before transport to the animal housing area. The sheep lesion inoculum contained T. phagedenis, T. medium, T. pedis, and F. necrophorum but not T. denticola, T. vincentii, or D. nodosus as detected by PCR.
Inoculation was performed through a small incision made at the bottom of the wrap. The incision provided access to the abraded region without removal of the wrap, maintaining the wet, anaerobic environment. One ml of prepared inoculum was placed under the cotton padding via syringe fitted with a teat cannula (Jorgensen Laboratories) and the wrap was resealed with tape.
Clinical Observations
Sheep were monitored no less than twice each week for development of clinical signs, including lameness (altered gait or non-weight-bearing posture), heat penetrating the bandage, tenderness on palpation, and the presence of atypical odor associated with the bandaged foot. Frequency of lameness observations increased as clinical signs developed. Flunixin meglumine (1 mg/kg, intramuscular, daily) or meloxicam (1 mg/kg, orally, every 24 to 48 hours) was given for analgesia once animals became non-weight bearing in at least 1 hind limb and continued as long as lameness was observed or until euthanasia. Personnel assessing clinical signs and administering analgesics were not blinded to the treatment group of the individual animals.
One foot from each animal was unwrapped at 2 weeks PI and observed for lesion development. Lesion development observations were made on the foot level, with the caveat that 2 feet on the same animal were not independent. Wrap was reapplied. Additional observations were made at 4 weeks PI either at necropsy for phase 1 (bovine-derived lesion inoculum) or by unwrapping 1 foot for phase 2 (sheep lesion inoculum). At 4 weeks PI in phase 2, the observed foot was biopsied and rewrapped with fresh bandages. Both feet were unwrapped and observed at 6 weeks PI for phase 2, and then 1 foot was biopsied (opposite foot from the one that was biopsied at 4 weeks PI) and bandages were left off to allow for progression or healing of the lesions without further intervention other than biweekly cleaning until 10 weeks PI.
Biopsy Procedure
While the foot was unwrapped, a superficial cutaneous lidocaine ring block was applied to 3 to 4 sites intradermally around the lesion or area to be biopsied using a 25 g needle. When the animal was no longer responsive to a probe (tip of blunt scissors), a 5-mm disposable biopsy punch was used to sample a region at the edge of the lesion. Biopsies were placed in 10% neutral buffered formalin and processed as described below.
Histology
Samples of lesions and adjacent healthy skin were preserved in 10% neutral buffered formalin. After fixation, hoof/tissue samples were decalcified with Kristensen’s decalcifying formic acid solution (50% 1 N sodium formate, 50% 8 N formic acid). Decalcification was monitored through chemical testing. 39 Briefly, decalcifying solution was tested each day for completion as follows. The decalcifying solution (5 ml) was neutralized with 0.5 N sodium hydroxide, and 1 ml of 5% ammonium oxalate was added. If the resulting solution became turbid, decalcification was not complete and tissues were placed in fresh Kristensen’s solution. This was repeated each day until the chemical test was no longer turbid, indicating that decalcification was complete. Once decalcification was complete, hoof/tissues were soaked in fibrous tissue soaking solution (100 ml Tween 80 in 1000 ml 1 N hydrochloric acid). 5 Tissues were tested daily for softness (pliability). Depending on the size of the tissues, this process took between 3 and 10 days. Then, tissues were routinely processed and paraffin embedded, and 5-μm-thick sections were stained with hematoxylin and eosin. Adjacent 5-μm sections were stained using Steiner and Steiner silver stain for visualization of bacteria. Slides were examined by a pathologist blinded to the identity of the study groups. Observations of individual feet were collated to give an aggregate observation per animal.
Polymerase Chain Reaction
To determine the presence of known BDD-associated treponemes, 1 ml of inocula was centrifuged to pellet any bacteria material. Pellets were processed for DNA extraction using Qiagen DNeasy Blood and Tissue Kit (Qiagen, Germantown, MD) following the manufacturer’s instructions for Gram-negative bacteria. To test lesions for presence of treponemes, swabs of lesion sites were collected at 2 and 4 weeks PI, and additionally in the second phase at 6 weeks PI. After removal of all bandages, a sterile, dry cotton swab (Puritan Medical Products Company, Guilford, ME) was rubbed over the abrasion or lesion for 15 seconds. Resulting swabs were placed inside individual sterile polystyrene tubes and transported to the laboratory. The tip of the cotton swab was cut off using sterile scissors and DNA extracted using Qiagen DNeasy Blood and Tissue Kit following the manufacturer’s instructions. Due to the absorption by the cotton, an additional 200 μl of lysing buffer (ATL) and 20 μl of proteinase K were added during the initial lysis step in the first incubation. DNA from both feet of the same animal was pooled and analyzed on the animal level. DNA was quantified using Qubit™ Fluorometric Quantitation (Thermo Fisher Scientific, Waltham, MA).
PCR for Treponema spp. and D. nodosus was performed in a 2-stage nested PCR assay. The initial PCR step was performed utilizing a universal bacterial primer set (primers listed in Supplemental Table S1), encompassing the majority of the 16 S rDNA gene, resulting in a 1526 bp product. Universal PCR reactions contained 1 µl forward primer (10 pmol; IDT, Coralville, IA), 1 µl reverse primer (10 pmol; IDT), 2.5 μl Roche 10× PCR buffer with magnesium (Roche, Indianapolis, IN), 100 µM dNTPs (Qiagen), 0.5 U Taq (Roche), DNA template to give 5 ng/DNA, and PCR grade water to give a final volume of 25 μl. PCR conditions were 95°C for 5 minutes, 25 cycles of 94°C for 1 minute, 55°C for 3 minutes, and 72°C for 3 minutes, with a final extension step at 72°C for 7 minutes. The nested PCR reaction used 1 µl of the initial PCR reaction as a template and contained as above but included primers specific for the 16 S rDNA gene for Treponema phylotypes T. phagedenis, T. medium, T. pedis, and D. nodosus. 13,14,50 PCR for detecting F. nodosus was performed using a primer pair directed toward the 16 S rDNA conserved segment specific for Fusobacterium spp. 29 The reaction mixture was prepared as above for universal PCR reaction. PCR primers, conditions, and expected amplicon size for each phylotype-specific reaction are given in Supplemental Table S1. Positive controls were genomic DNA from each of the respective phylotypes; negative control was the reaction mixture with sterile water. PCR reaction products were visualized by electrophoresis on 1.5% agarose gel stained with SYBR Safe DNA Gel Stain (Invitrogen, Waltham, MA).
Bacterial Observation and Culture
Microscopic observations
At 2, 4, or 6 weeks PI, sterile cotton swabs of the lesion sites as described above were placed in sterile 5-ml polystyrene tubes and transported to the laboratory. Swabs from each foot were maintained separately and evaluated on the individual foot level. Approximately 0.5 ml sterile PBS or OTEB was added to each tube and incubated at room temperature with periodic vortexing for 1 hour, and then 10 µl was placed on a clean glass slide and viewed using dark-field microscopy for the presence of spirochete-shaped bacteria. Some slides were viewed using a phase-contrast equipped imager (Cytation 5; BioTek, Winooski, VT). Tubes were then placed into the anaerobic atmosphere chamber. One ml of OTEB enriched with additional 10% bovine serum, 25 µg/ml rifampin (Sigma, St Louis, MO), and 5 µg/ml enrofloxacin (Baytril; Bayer HealthCare, Shawnee Mission, KS) was added to each tube and incubated overnight at room temperature. Tubes were checked for spirochetes by dark-field or phase-contrast microscopy.
Enzyme-Linked Immunosorbent Assay
Whole blood (8-10 ml) was collected via jugular venipuncture into serum separator vacutainer tubes (BD Vacutainer SST; BD and Company) prior to inoculation (week 0), at 4 weeks PI, and in phase 2, at 6 and 10 weeks PI. Blood was allowed to clot, and serum was separated by centrifugation (Beckman Coulter Avanti J-E with JS-5.3 rotor, 700 × g, 20 minutes, 4°C) and then stored at –20°C. T. phagedenis, T. denticola, T. vincentii, T. medium, and T. pedis were grown in OTEB to confluence, bacterial cells harvested and washed by centrifugation, and antigen prepared from whole-cell sonicates as described previously. 21,47 Treponeme antigen (10-20 µg protein/ml in PBS) was bound overnight to a 96-well plate (Nunc Maxisorp; Thermo Fisher Scientific). Serum was serially diluted, added to plates, and incubated for 1 hour at 37°C and then at 4°C overnight. Plates were washed 3 times with PBS with 0.05% (v/v) Tween 20 (Sigma). Bound antibody was detected by horseradish peroxidase-conjugated rabbit anti-sheep IgG (γ-chain) (KPL, Gaithersburg, MD) and incubated 1 hour at 37°C. Substrate was SureBlue Reserve TMB Microwell Peroxidase Substrate (KPL) and reaction was stopped with TMB stop solution (R&D Systems, Minneapolis, MN). Plates were read at 455 nm. Titer reported is the lowest dilution with optical density equal to or greater than mean plus 2 standard deviations of the wells containing only PBS. Assay was performed in duplicate by a technician blinded to the treatment groups and lesion outcome of the individual animals. Enzyme-linked immunosorbent assay results for phase 1 (bovine-derived inoculum) were analyzed using GraphPad Prism 6 statistical software fitting a 1-way analysis of variance (ANOVA) with Tukey’s multiple comparisons. Enzyme-linked immunosorbent assay results for phase 2 (sheep-passaged inoculum) were analyzed using GraphPad Prism 6 statistical software fitting a 2-way ANOVA with repeated measures and Tukey’s multiple comparisons for simple effects within columns.
Results
Two phases of experimental infection were performed. The first phase comprised the induction of lesions in sheep using tissue homogenates derived from BDD lesions (bovine-derived inoculum). The second phase perpetuated the lesions in an experimental model by using lesion material from experimentally infected sheep (phase 1) to induce lesions in naïve sheep (sheep-passaged inoculum, phase 2). There was no detectable soreness, swelling, or distress attributed to the wraps in inoculated or mock-inoculated animals. Warmth, malodor, and tenderness assessed by palpation through the wrap were evident in inoculated feet at approximately 10 days PI, with visual evidence of lameness beginning at approximately day 18 PI and continuing until the end of the experiment in both phases.
Phase 1: Lesion Induction in Sheep With Bovine-Derived Inoculum
Gross observations
At 2 weeks PI, the wrap on 1 foot of each animal was removed and feet inspected for signs of lesion development. Gross lesions were observed in 7 of 8 unwrapped feet (Table 1) in the inoculated group. Gross lesions were characterized by malodorous, multifocal, variably sized (0.5 to 2.0 cm2), well-demarcated areas of redness, erosion, and ulceration. The appearance of the ulcerated areas was consistent with granulation tissue (Fig. 8). Affected regions were painful upon palpation and bled easily. Some ulcerated lesions were circumscribed by pale, rounded fibrous tissue, consistent with second intention wound healing. In mock-inoculated animals, the abraded area had completely healed (4/4 feet observed). Observed feet were rewrapped and lesion development was allowed to continue.
Summary of Experimental Observations (gross lesions and spirochete presence) by Group and Time Point.
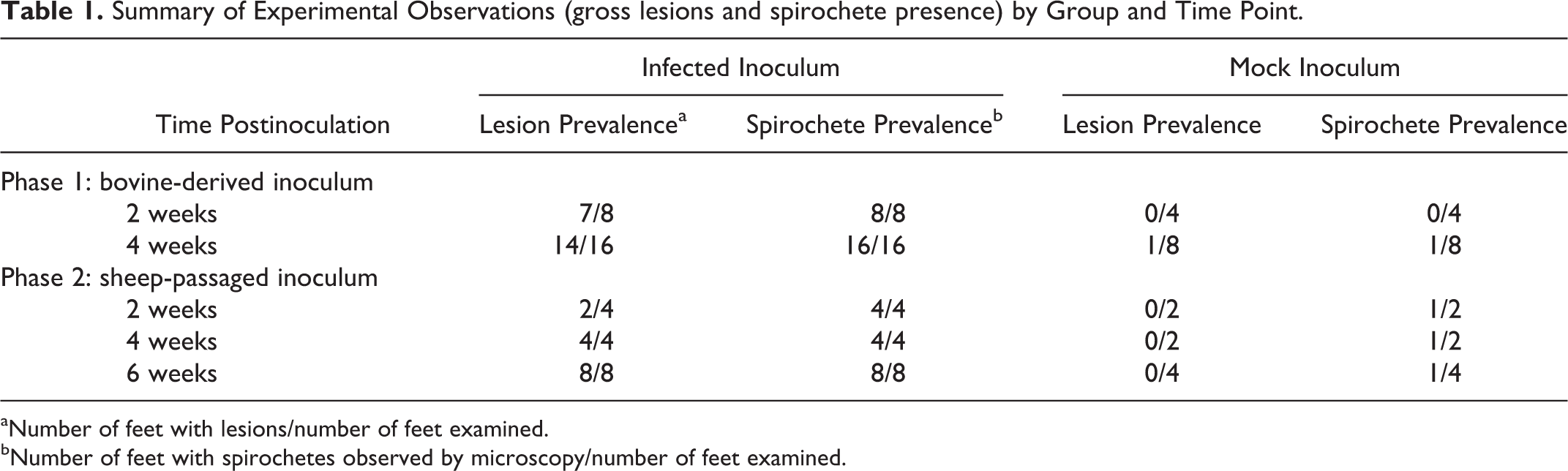
aNumber of feet with lesions/number of feet examined.
bNumber of feet with spirochetes observed by microscopy/number of feet examined.
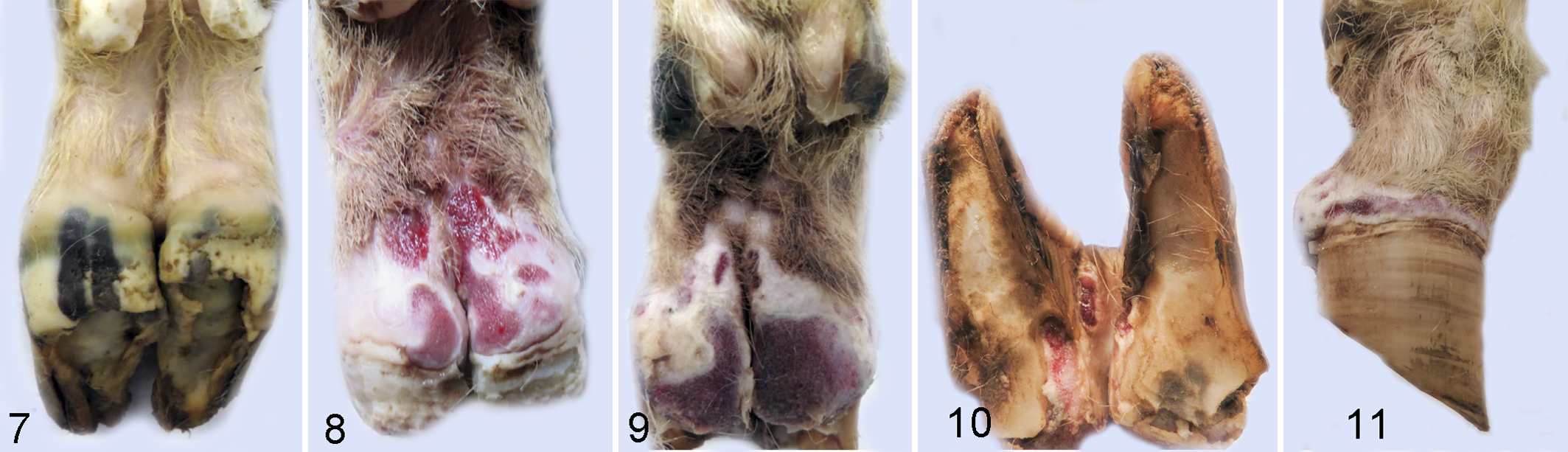
At 4 weeks PI, lesions were observed in 14 of 16 inoculated feet. The 2 feet that did not develop lesions belonged to the same animal. Lesions progressed from 2 weeks PI to 4 weeks PI (Fig. 9), encompassing a larger area of the heel and sole. In some animals, the lesions extended from the coronary band to the axial interdigital regions (Fig. 10) and the abaxial surface of both claws (Fig. 11).
In the 4 uninfected mock-wrapped control animals, 1 foot developed lesions at 4 weeks PI (mock), but the other 7 feet appeared grossly normal at both 2 and 4 weeks PI (mock) (Table 1, Fig. 7).
Microscopic observations
At 4 weeks PI, lesions were well developed (Table 2). Major histologic features were epidermal erosion, ulceration, hyperplasia, orthokeratotic hyperplasia (Fig. 12), and ballooning degeneration (Fig. 13) with neutrophilic infiltrates within the epidermis (Fig. 14). In some cases, neutrophils formed intraepithelial pustules. When spirochetes were found within the epidermis (Fig. 15), they were generally present in large numbers and sometimes colocalized, with bacteria morphologically dissimilar to spirochetes. Consistent with findings from BDD, clear spirochetal forms were observed deep within all layers of the epidermis, many times at the leading edge of erosions or ulcerations.
Histologic Lesions at 4 or 6 Weeks After Inoculation With Material From Digital Dermatitis Lesions.
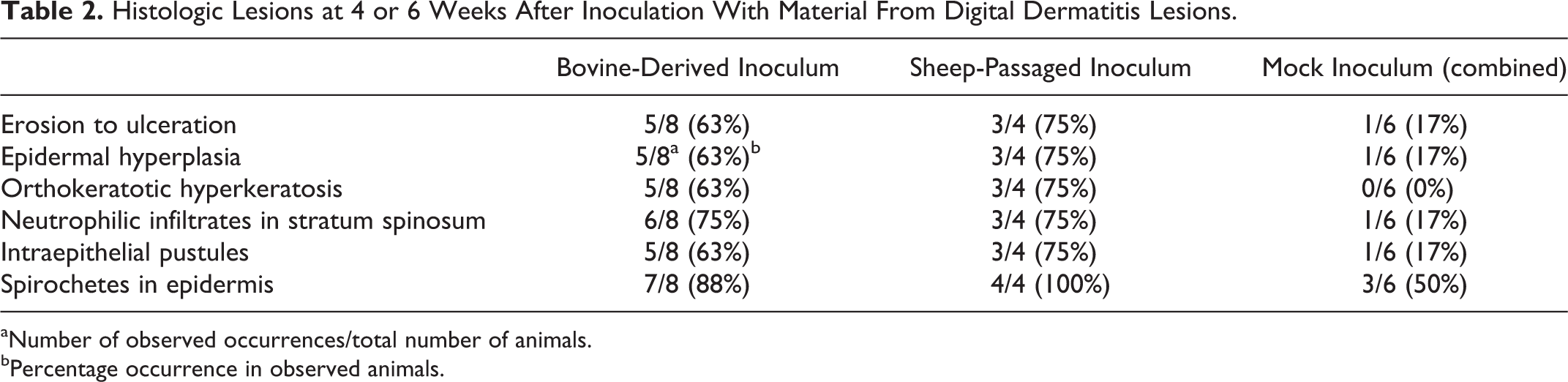
aNumber of observed occurrences/total number of animals.
bPercentage occurrence in observed animals.
Detection of Treponema spp
At both 2 and 4 weeks, swab samples from the abrasions or lesions on all inoculated feet sampled contained multiple morphologies of spirochetes as visualized by dark-field or phase-contrast microscopy (Table 1, Supplemental Fig. S2). The mock-inoculated control foot that was suspected visually of lesion development at 4 weeks also had numerous spirochetes present. By nested PCR, swabs of the abrasions or lesions were positive for T. phagedenis, T. medium, and T. pedis in 8 of the 8 inoculated sheep at 4 weeks PI. T. phagedenis and T. medium were detected in 2 and 1 of the 4 mock-inoculated controls, respectively (Table 3).
Identification of Treponema spp. by Polymerase Chain Reaction in Skin Swabs, 4 Weeks Postinoculation of Sheep With Digital Dermatitis Lesion Homogenate.
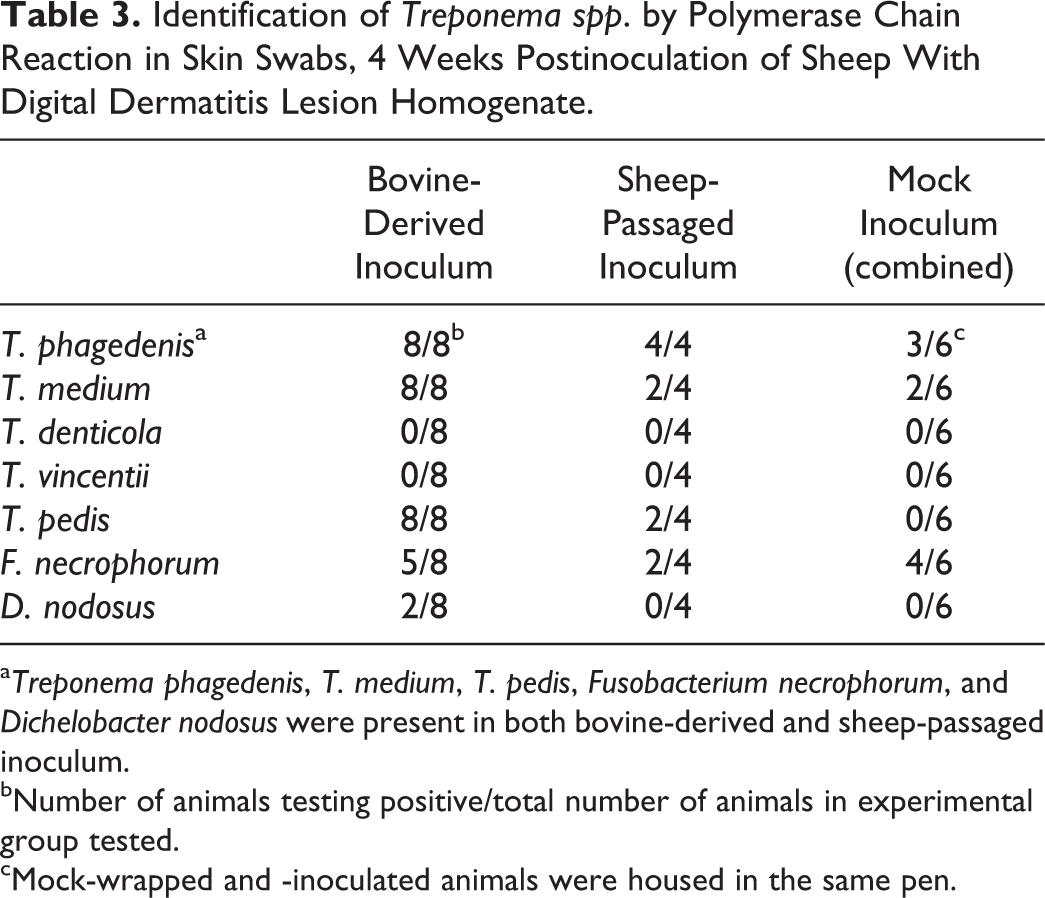
a Treponema phagedenis, T. medium, T. pedis, Fusobacterium necrophorum, and Dichelobacter nodosus were present in both bovine-derived and sheep-passaged inoculum.
bNumber of animals testing positive/total number of animals in experimental group tested.
cMock-wrapped and -inoculated animals were housed in the same pen.
Serum antibody response
Serum was collected from experimentally infected and mock-inoculated sheep prior to and at 4 weeks PI, and antibody titers to treponeme antigens were determined by endpoint dilution. Serum samples taken 4 weeks PI had significantly higher anti-treponemal antibody titers (median value = 800, P = .0023) than preinoculation serum samples (week 0, median value = 50) and those from mock-inoculated controls (median value = 50) (Fig. 16).
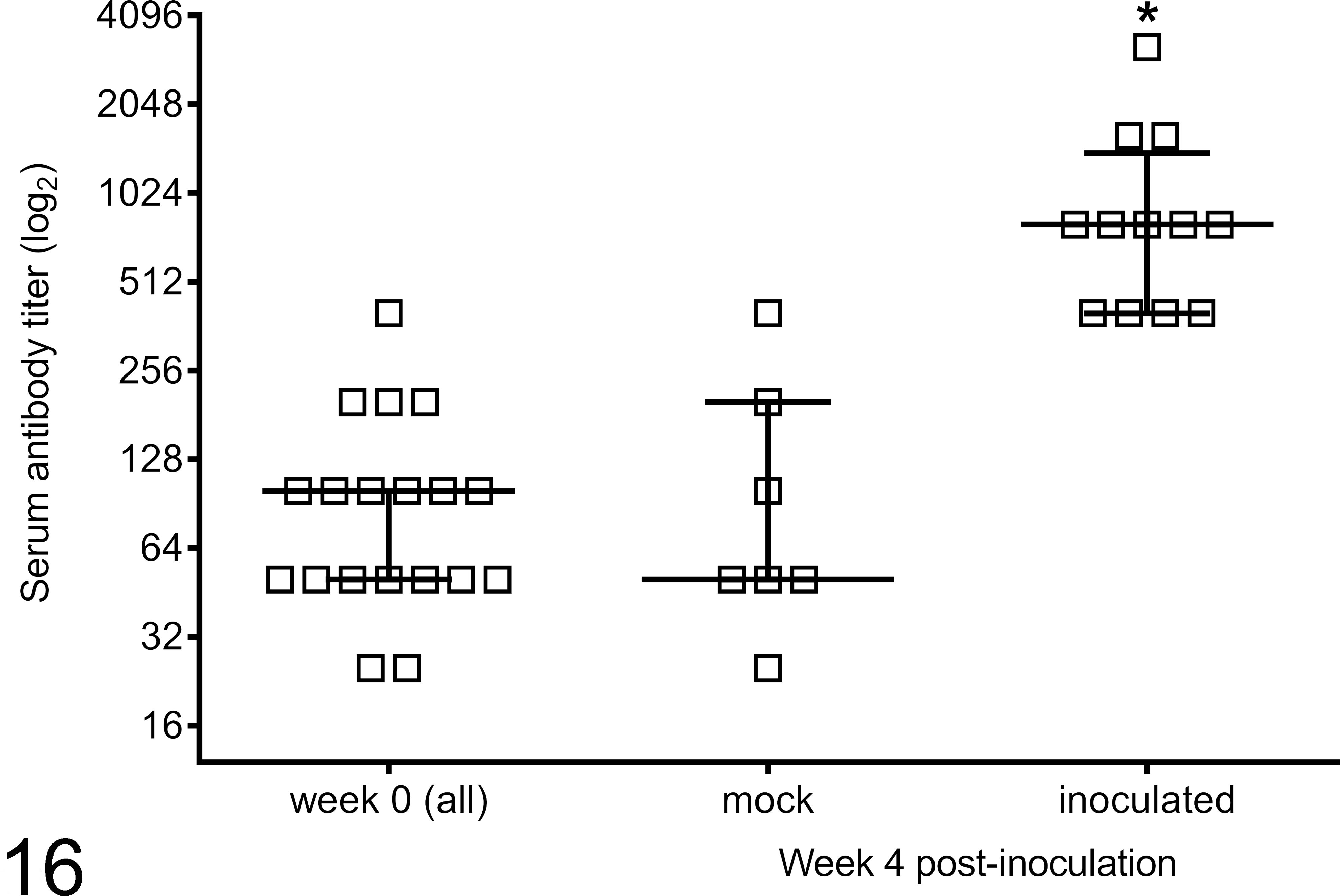
Serum antibody specific for mixed treponeme antigens as detected by enzyme-linked immunosorbent assay 4 weeks postinoculation with material derived from bovine digital dermatitis lesions. Median and interquartile range (horizontal bar and error bars) are depicted along with individual animal values. *P = .0023; 4-week postinoculation values are statistically different from week 0 and from 4 weeks mock-inoculated.
Phase 2: Perpetuation of Lesions With Sheep-Passaged Inoculum
Gross observations
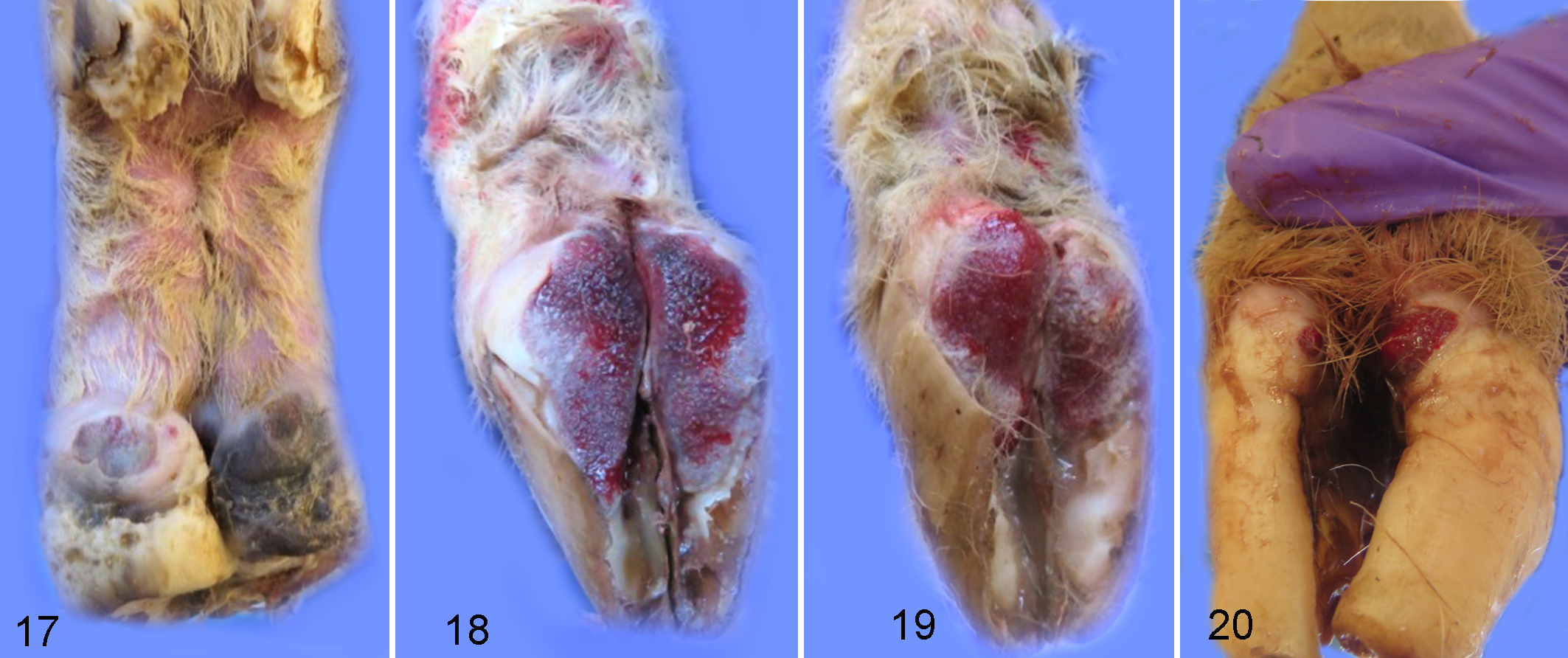
Homogenate was prepared from lesions of experimentally infected sheep and used to inoculate wrapped and abraded feet in a group of naïve sheep. This sheep-passaged inoculum resulted in fewer lesions at 2 weeks PI compared to sheep inoculated with non-passaged bovine-derived inoculum (Table 1, Fig. 17). All inoculated feet developed lesions by 4 weeks PI (Fig. 18), and these lesions were more severe when the wraps were removed at 6 weeks PI (Fig. 19). Lesions at 6 weeks PI in sheep inoculated with sheep-passaged inoculum were similar to those seen at 4 weeks PI using the bovine-derived inoculum. Multifocal lesions of redness, erosion, and ulceration varied in size (0.5 to 3 cm2) (Fig. 19) and extended from the axial interdigital regions to the abaxial aspects of each claw (Fig. 20). Ulcerative lesions extended distally, leaving ulcerated or undermined regions of the posterior sole. Similar to lesions induced by bovine-derived inoculum, ulcerated regions were filled with granulated tissue, were painful upon palpation, and bled easily. In mock-inoculated sheep, the abraded area had healed in observed feet at 2 weeks PI (mock) with no gross evidence of lesion development (2 feet observed), and observed mock-inoculated feet remained grossly normal at 4 and 6 weeks PI (mock).
Microscopic observations
The inoculum created from experimentally induced digital dermatitis in sheep induced similar microscopic lesions to lesions induced from bovine-derived lesion material, that is, epidermal erosion, ulceration, irregular acanthosis, orthokeratotic hyperkeratosis, and ballooning degeneration with neutrophilic infiltrates within the epidermis (Figs. 21–24, Table 2). Multiple morphologies of spiral-shaped bacteria (presumably treponemes) were present within the epidermis as observed by silver staining (Fig. 24).
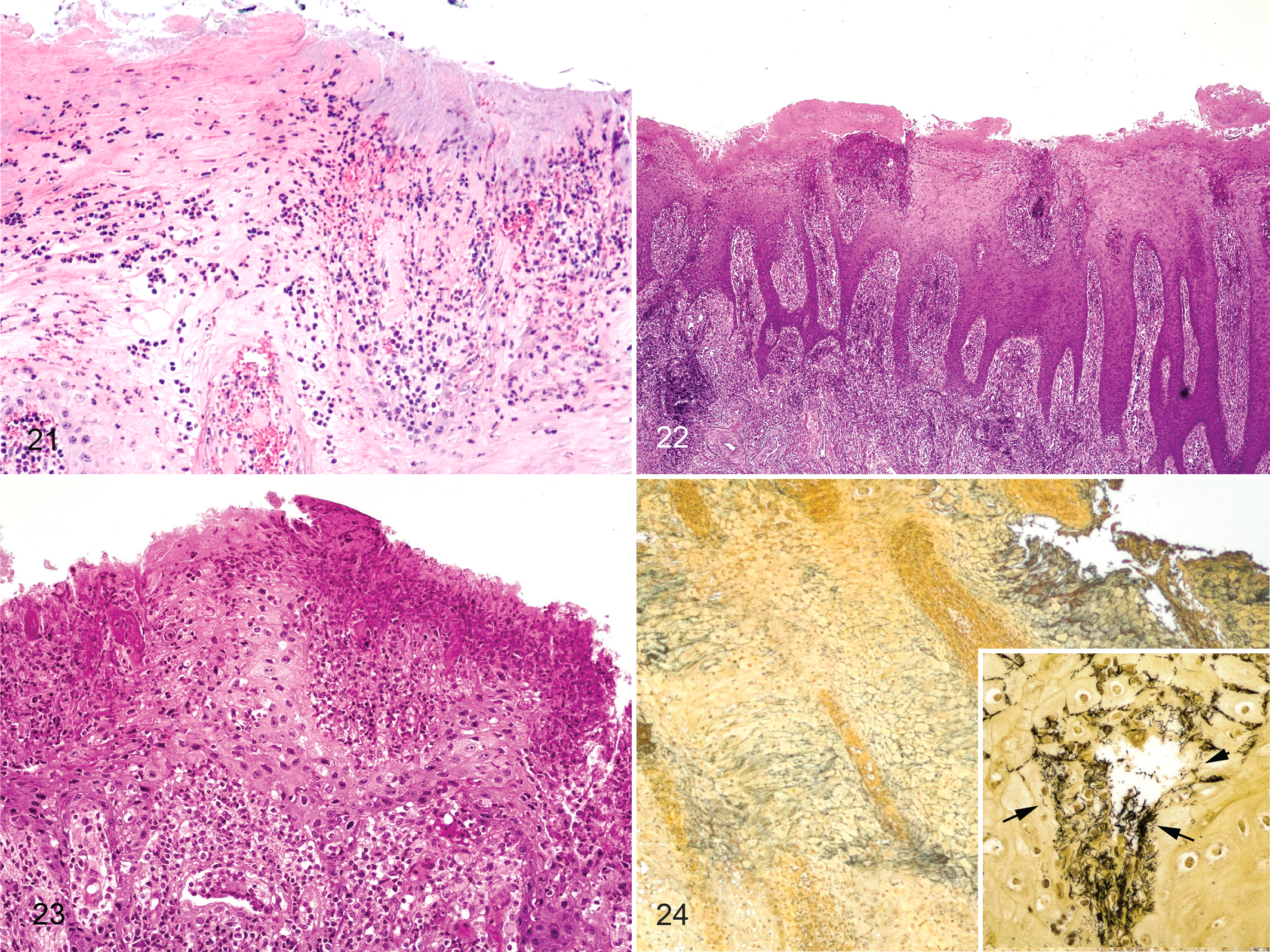
Detection of Treponema spp
Swabs were taken of the skin at the abrasion site or lesions at 2, 4, and 6 weeks PI and were observed for the presence of spiral-shaped bacteria. Even though few lesions were present at 2 weeks PI, all inoculated feet had multiple morphologies of spirochetes detected by dark-field microscopy (Table 1). These spirochetes appeared to increase in number relative to other bacteria present by 4 and 6 weeks PI. When wraps were removed, spirochetes remained visibly present until lesions were completely healed. This varied in the individual animals from 7 to 10 weeks PI (1 to 4 weeks PI after wraps removed), with 2 feet not resolving at the time of euthanasia (Supplemental Figs. S7, S8). The presence of Treponema species associated with BDD was tested using a nested PCR assay on swabs taken of the skin at the abrasion site or lesions of the sheep inoculated with sheep-passaged inoculum at 6 weeks PI. Swabs were positive for T. phagedenis, T. medium, and T. pedis in 4, 2, and 2 of the 4 inoculated sheep and 1, 1, and 0 of the 2 mock-inoculated sheep, respectively (Table 3).
Serum antibody response
Sheep inoculated with sheep-passaged inoculum developed median antibody titers to mixed treponemal antigens of 75 before inoculation, 800 at 4 weeks PI, 1200 at 6 weeks PI, and 1600 at 10 weeks PI (Fig. 25). The magnitude of response was similar to that seen in sheep inoculated with bovine-derived inoculum and was significantly greater at 6 and 10 weeks than preinoculation (week 0) titers (P = 0.047 and P = 0.030, respectively).
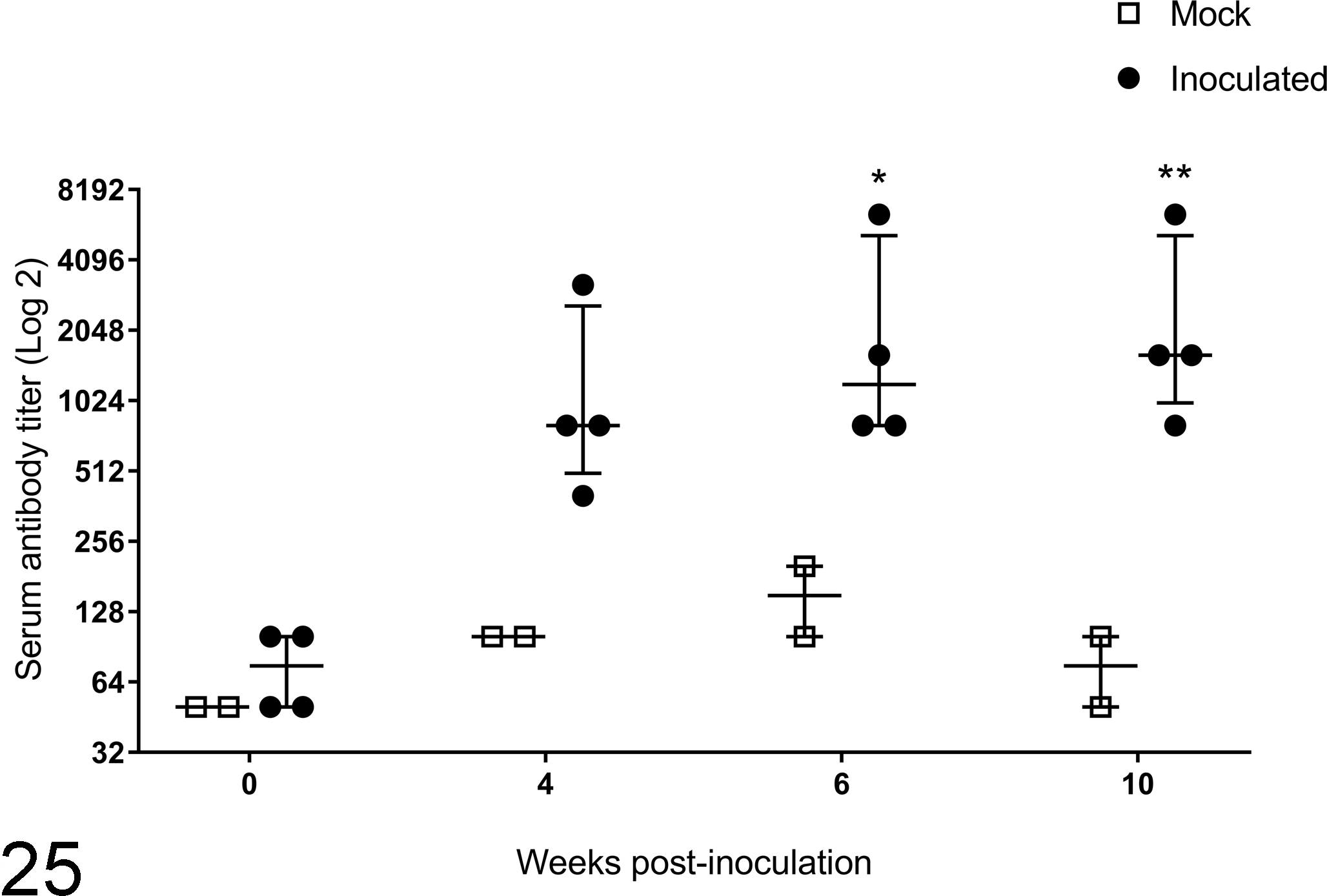
Serum antibody specific for mixed treponeme antigens as detected by enzyme-linked immunosorbent assay at 0, 4, 6, and 10 weeks postinoculation with material derived from experimentally induced sheep digital dermatitis lesions. Median and interquartile range (horizontal bar and error bars) are depicted along with individual animal values. *P = 0.0467; week 6 inoculated values are statistically different from week 0. **P = 0.0297; week 10 inoculated values are statistically different from week 0.
Discussion
The goal of this work was to develop a reproducible animal model of BDD using sheep. There is an advantage to using smaller ruminants with respect to housing, handling, and restraint, and it also allows study of the ruminant immunologic response during disease development. Additionally, we demonstrated the highly infectious nature of digital dermatitis and the susceptibility of compromised hoof to this disease. We were able to use bovine-sourced lesion material to create lesions in sheep, showing cross-species transmissibility. Using relatively small numbers of animals, a high rate of gross lesion induction was observed; specifically, lesions developed within 4 weeks PI in 7 of 8 sheep infected with material derived from BDD lesions and within 6 weeks PI in 4 of 4 sheep infected with material that had been passaged through sheep. This time frame is consistent with the time from inoculation to lesion development in previously published bovine models. 16,25 As with the model published by Krull et al, scarification or abrasion of the skin as well as maintaining a moist environment were critical for successful lesion development. Dilute rumen fluid in a nutrient broth was added weekly to approximate dairy slurry, a proposed factor contributing to BDD. 32
These lesions were confirmed microscopically to have features consistent with BDD, including the presence of spirochetes within the epidermis. The closed wrap system described in this model allowed for greater containment of the pathogenic organisms/factors, allowing for future study of individual factors (abrasion, wetness) and individual bacterial species with minimal influence of the external environment.
The gross lesions observed in this model system did not exactly match those observed in classical cases of BDD. In BDD, lesions are found on the skin in the pastern area or adjacent to the coronary band or heel bulb. This model also differs from described lesions of naturally occurring contagious ovine digital dermatitis (CODD) in sheep, where the lesions run along the coronary band, progressing to under the hoof wall, leading to hoof capsule sloughing. 1 The lesions in this model grossly affect the heel bulb, extending from the interdigital space to the abaxial aspects of each claw, and from the coronary band distally to the posterior sole, with large areas of erosion, ulceration, and granular tissue. We hypothesize that anatomic and physical differences (size, mechanical stresses on hoof structures) potentially are the reason for differences in the appearance of lesions in the natural host (cattle) and the experimental host (sheep model). It is speculated that shifting or pooling of materials at the bottom of the wraps contributed to the predilection for lesions to form on the heel bulb or sole rather than higher on the pastern area. On a microscopic level, findings matched BDD with areas of erosion, acanthosis, hyperkeratosis, neutrophilic infiltrates, intraepithelial pustules, and spirochetes in the epidermis. These microscopic changes have been used to define cases of BDD. 8,35 Gradations of CODD include mild to severe periadnexal/perifollicular lymphoplasmacytic infiltrate, epidermal hyperplasia, and orthokeratotic hyperkeratosis. 2 Thus, BDD, CODD, and this model in sheep all share common histologic characteristics. Although the model has some differences from the natural disease with respect to the gross presentation, it effectively allows reproduction of acute histologic lesions as well as controlled manipulation of the skin microenvironment and infection with specific pathogens, all in the context of a ruminant immune system. The use of this model allows for manipulation of the host (eg, immune regulation, enhanced cytokine production, etc) and the pathogen (adding or subtracting virulence factors) in a controlled environment for developing and testing hypothesis-driven experiments. Tested hypotheses can then be confirmed in the natural host model with targeted assays.
As digital dermatitis disease is currently understood, this sheep transmission model has shown the persistence of at least 3 phylotypes of Treponema species in addition to other uncharacterized anaerobic bacteria. These bacteria, transferred as a complex homogenate, have shown similar cellular pathogenesis in 2 different species of host animals. A nested PCR assay was used to identify the main 3 phylotypes of Treponema; these same phylotypes have been found in other species with digital dermatitis (cattle, buffalo, goats, sheep, and elk) and also globally in BDD (Europe, South America, United States). 2,3,8 –10,12,13,18,19,22,26,30,31,35,37,38,41,43,48,51 These 3 phylotypes were present in both the bovine-derived lesion material inoculum and the sheep-passaged lesion material inoculum. All sheep (8/8) receiving the bovine-derived lesion inoculum had all 3 phylotypes present 4 weeks PI. Two sheep (2/4) had all 3 phylotypes present 6 weeks PI and 2 sheep (2/4) had 2 of the 3 phylotypes present. D. (Bacteroides) nodosus and F. necrophorum have long been associated with foot-rot, interdigital dermatitis, or interdigital phlegmon in sheep, goats, and cattle. 4,23,24,33 These 2 bacterial species have also been found in some analyses of digital dermatitis (reviewed in Wilson-Welder et al 46 ). D. nodosus was present in the bovine-derived lesion inoculum but absent in the sheep-passaged inoculum, F. necrophorum was present in both inocula. F. necrophorum was present in slightly more than half of the inoculated feet (5/8 for bovine-derived inoculum and 2/4 for sheep-passaged inoculum) and was present in more than half of the wrapped feet of 4 of the 6 mock-inoculated sheep without inducing disease. This indicates that although present in digital dermatitis lesions, Fusobacterium alone is not the solitary inducer of lesions. Similarly, D. nodosus, present in only the bovine inoculum, was not universally detected in the experimentally induced lesions. The lack of this organism in lesions would indicate that it probably has little role in development of digital dermatitis lesions. The exact role of these and other bacteria in digital dermatitis pathogenesis is not understood. We had hypothesized that passage of lesion material in experimental inoculations would alter the microbial composition, possibly eliminating some organisms that were not essential in disease induction. Additional studies will be needed to characterize other bacteria present in the lesions and the effect of in vivo passage on the microbial community.
Cattle with digital dermatitis generally exhibit robust serum antibody responses to antigens from treponemes associated with the lesions. 28,43,44 In naturally occurring BDD and in calf models of infection, a robust antibody response to treponemal antigens is observed, even within a 4-week time frame. 11,28,43,44 Treponeme-specific antibody was present in both phases of the sheep model, indicating that as with the calf-model and natural bovine infections of BDD, a systemic immune response is generated.
It is important to note that all animals, along with some sheep with unwrapped feet (n = 6), were housed in the same pen for the duration of the experiments. The unwrapped and unabraded naïve animals did not develop any signs of disease or antibody responses to treponemal antigens (data not shown). The development of lesions on 1 mock-inoculated animal in phase 1 (bovine to sheep transmission) and the presence of spirochetes in the epidermis of 3 of 6 mock-inoculated (phases 1 and 2) (Table 2) by silver stain and PCR (phases 1 and 2) (Table 3) demonstrate the highly contagious nature of digital dermatitis diseases and possibility of transmission within a closely housed pen of animals. Examination and rewrapping of the feet may have inadvertently transferred the infectious organisms from inoculated to mock-inoculated animals. Transfer of digital dermatitis to naïve animals and naïve farms by tools and hoof trimmers has been theorized. 15 Subsequent studies with sheep were modified to ensure handling of controls or mock-inoculated animals prior to inoculated animals, and no mock-inoculated animals developed lesions or positive PCR reactions occurred in these subsequent experiments. Although other models have shown within-pen transmission resulting in lesions, this appears to be minimized with the current model possibly because of the more extensive wraps used. 25
This study describes the use of a small ruminant model for experimental infection with BDD lesion material. Macerated bovine lesions of digital dermatitis induced lesions resembling digital dermatitis in wrapped sheep feet. Histologic lesions were consistent with descriptions of BDD in cattle and were dominated by treponeme bacteria similar to those detected in BDD. The resulting lesions in sheep were macerated and applied to wrapped feet of naïve sheep, demonstrating intentional transmission of the infection. Thus, the early, acute phase of the disease has been well replicated. For BDD, lack of animal models has hindered progress into deeper understanding of the initiating disease process, thus slowing development of intervention strategies and therapeutics. We acknowledge that this model is not a complete representation of all aspects of on-farm observed digital dermatitis, nor is it a replication of CODD. However, the model does present a controllable, reproducible platform from which to investigate the complex interactions among host, environment, and pathogens in this polymicrobial disease.
Footnotes
Authors’ Note
Mention of trade names or commercial products in this study is solely for providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Acknowledgements
We wish to acknowledge Ami Frank for excellence in laboratory assistance and preparation of this manuscript, James Fosse and the Visual Services Department at the National Center for Animal Health for excellent assistance in preparing the images for this manuscript, and Judith Stasko and the Microscopy Services Unit at the National Center for Animal Health for excellence in developing protocols and methods for preparation of foot and hoof tissues.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. JHWW, JEN, DPA, and MVP are US Department of Agriculture employees, work was completed in the course of their assigned duties in relation to project number 5030-32000-223-00-D. JC and PP were supported in part by Agriculture and Food Research Initiative Competitive Grant no. 11641835 from the USDA National Institute of Food and Agriculture. Work was completed by US Department of Agriculture employees in the course of their assigned duties in relation to project number 5030-32000-223-00-D.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
