Abstract
An epidemic of early fetal loss (EFL), late fetal loss (LFL), fibrinous pericarditis, and unilateral uveitis which occurred during the spring of 2001, are together now known as the mare reproductive loss syndrome (MRLS). A similar epidemic with less intensity was reported during the same period of time from southern Ohio, West Virginia, and Tennessee. The same syndrome with lesser intensity recurred in 2002. The estimated economic loss from the syndrome in 2001 and 2002 together was approximately $500 million. Both EFL and LFL were characterized by the absence of specific clinical signs in aborting mares. Nonhemolytic Streptococcus spp. and Actinobacillus spp. accounted for 65% of the organisms isolated from fetuses submitted for a postmortem during the MRLS period in 2001 and 2002. The pathologic findings in fetoplacental units of LFL included bronchopneumonia and funisitis, and there were no findings in EFL. Epidemiologic studies conducted in 2001 suggested an association between the presences of eastern tent caterpillars (ETC) in pastures with MRLS. Experimental studies in pregnant mares by exposure to ETC, or administration by stomach tube or with feed material, reproduced EFL and LFL. Similar experimental studies in mouse, rats, and goats with ETC were unsuccessful. Currently, 2 hypotheses are proposed for MRLS. One hypothesis proposes that an ETC-related toxin with secondary opportunistic bacterial invasion of the fetus leads to MRLS. The second hypothesis suggests that a breach of gastrointestinal mucosal integrity by hairs of ETC leads to a bacteremia and results in MRLS. In 2004, a similar equine abortion storm was reported from Australia and caterpillar exposure was identified as a risk factor for the abortion. In 2006, the syndrome was observed in Florida and New Jersey.
Definition
During the spring of 2001, horse farms in and around central Kentucky experienced an outbreak of early fetal losses (EFL), late-term abortions, stillbirths, and neonatal foal deaths, together called late fetal losses (LFL). Fewer cases of fibrinous pericarditis (FP) and unilateral uveitis (UU) were also reported. These losses and disease entities came to be known as mare reproductive loss syndrome (MRLS). EFL and LFL occurred roughly at about the same time (the last week of April to the last week of May 2001), peaked at the same time (the first week of May), and declined at a similar rate. More than 1,500 cases of EFL were reported during this period. The FP and UU cases were identified during the same time period. A similar pattern of abortions, FP and UU but, in lesser numbers, was observed during the same period in 2002. 20, 22, 23, 37
Analysis of the data collected from the Livestock Disease Diagnostic Center (LDDC), University of Kentucky, showed that the abortions peaked during the end of April and early May in 2001 and in 2002. A comparison of the number of aborted fetus and/or placenta submissions during the previous 4 years (1996–2000) indicated that increased abortions from MRLS were observed during the last week of April through the last week of May, and, by June 1, the abortion submissions were similar to previous years. Aborted equine fetuses from 32 counties in the state of Kentucky were submitted to the LDDC for a postmortem. 20, 22, 23, 37
Overall, approximately 3,500 Thoroughbread foals (3,000 EFL and 500 LFL) and numerous foals from other breeds (approximately 1,000 Paint horses and Quarter horses) were lost because of MRLS in 2001. 31, 39 Smaller numbers of clinically similar fetal losses, abortions, and stillbirths were reported from southern Ohio, West Virginia, and Tennessee during the same time period, which indicated that MRLS was not limited to Kentucky. 17, 25, 27 The economic impact of MRLS was substantial, and the economic loss from MRLS in 2001 was estimated at $336 million. 55 The combined losses from MRLS 2001 and 2002 were estimated at approximately $500 million. MRLS was one of the most economically devastating acute diseases to strike the animal industry.
Clinical Features of MRLS
EFL
EFL was first identified during routine fetal sexing. On April 26, the lack of a fetal heart beat was observed in 2 fetuses. The same observation was made at multiple farms in central Kentucky by several practitioners during the last week of April. Mares who were undergoing EFL showed minimal systemic clinical signs. Few mares had serosanguineous or purulent vulvar discharge, and some mares had fetal membranes that protruded from their vulva, with fetuses located either in the vagina or the vulva. Approximately 10–15% of mares exhibited mild colic signs, abdominal straining, or low-grade fever (101.0–101.5°F), 1 to 3 days before EFL.
In most EFL cases, rectal palpation was normal. The typical ultrasound presentation of EFL and/or MRLS was a dead fetus (i.e., no heartbeat), surrounded by echogenic allantoic fluid and a more echogenic amniotic fluid (Fig. 1). In cases where the fetus was not dead, an ultrasound examination generally showed a fetus with a slow heart rate and slow movements, suspended in an echogenic amniotic fluid that was described as being either cloudy or flocculent. Some mares aborted markedly autolyzed fetuses, whereas some retained the fetus in the vaginal canal. Most mares with ultrasonographically identified EFL expelled the fetus within 2 days to 2 weeks. 4, 38, 39
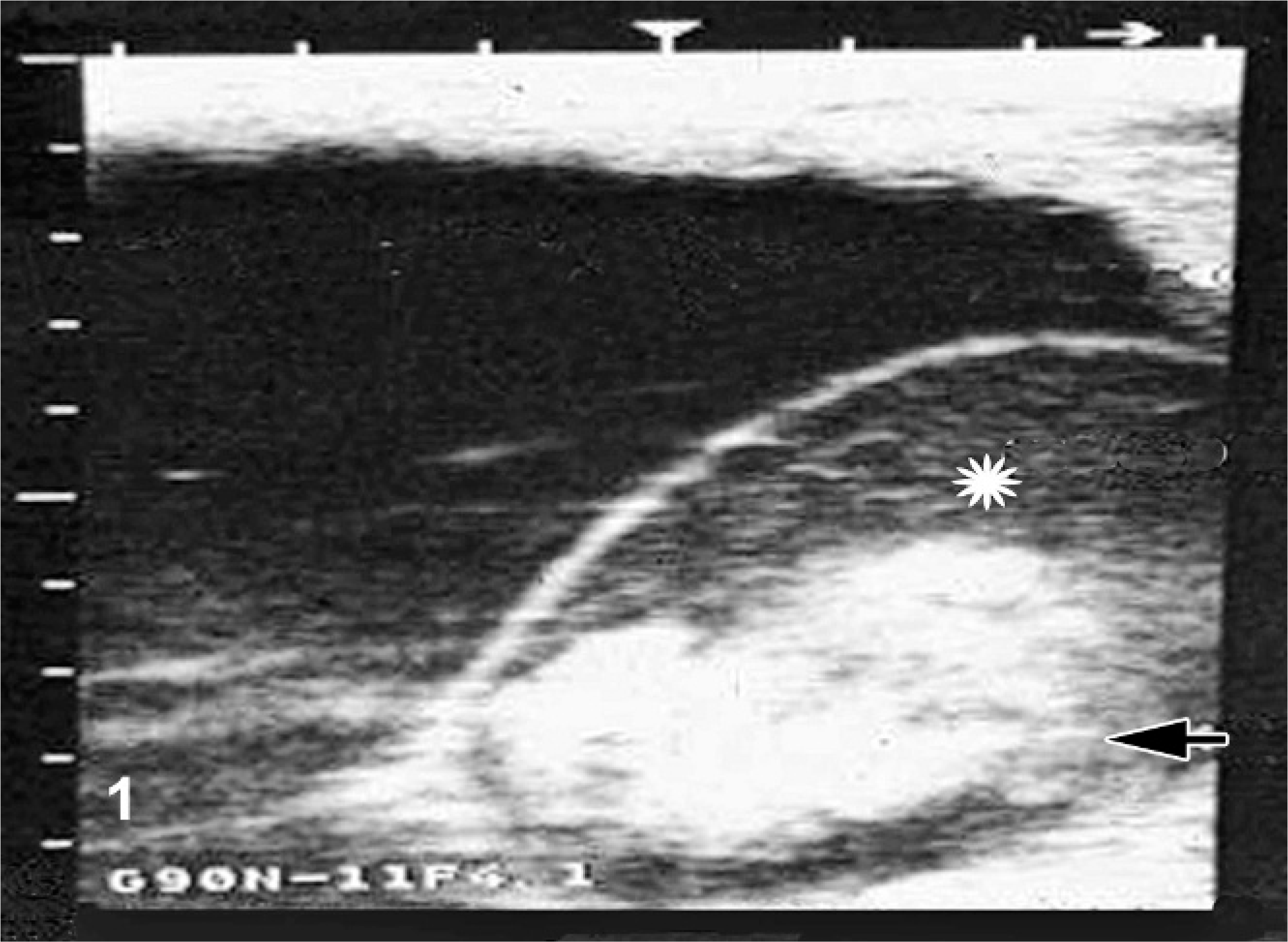
Uterus, equine. Ultrasonographic image of early fetal loss in a pregnant mare on day 78 of gestation. Asterisk = cloudy amniotic fluid. Arrow = fetus in amniotic cavity.
Efforts to breed post-EFL mares were unsuccessful. These mares failed to conceive throughout 2001, apparently because of the presence of equine chronic gonadotrophin produced by the normal development of endometrial cups, which develop by about 37 days of pregnancy. 38, 39 Mares bred in the fall of 2001 in southern hemisphere breeding schedules, after their endometrial cups had regressed, conceived and maintained their pregnancies.62
LFL
LFL was diagnosed on April 28, 2001, 2 days after EFL was observed. LFL was observed during the last trimester of gestation, with increased frequency in the last month of gestation; some were close to or at term. The majority of LFL mares exhibited minimal signs of impending foaling or abortion. Clinical features associated with LFL included explosive parturition, dystocia, foaling while standing, premature placental separation stillbirth, foals born weak, and agalactic mares. 4 Clinical signs associated with foals born by LFL included weakness, with asphyxia, which required resuscitation. Many of the weak foals were admitted to local equine veterinary hospitals and survived for up to 4 days. Blood cultures were rarely positive for bacteria. The most frequently isolated bacteria were alpha Streptococcus and Actinobacillus spp. These foals were dehydrated, hypothermic, and tachycardic, with irregular respiration. Leucopenia, hypoglycemia, and acidosis were also consistent laboratory findings. Bilateral hyphema was observed in some of these foals at birth. 6
FP
The first case of FP epidemic was diagnosed on April 28, 2001, just 2 days after EFL was identified. During the next 2 months, 38 cases of FP were admitted to 2 large central Kentucky referral veterinary clinics. These cases were reported in horses of all ages, breeds, and sexes, and were not restricted to pregnant mares. 54
The clinical presentation of the FP cases included sonographic evidence of effusive FP (100%), tachycardia (90%), muffled heart sounds (69%), hyperfibrinogenemia (64%), lethargy (62%), tachypnea (57%), jugular pulse distention (54%), and pleural effusion and/or ascites (45%). Several FP cases were reported during the same time period in 2002; however, the numbers admitted to veterinary hospitals was markedly fewer. Analysis of the fluid removed from the pericardial sac of most cases demonstrated a low white cell count, with neutrophils and a high protein content. This fluid was mostly sterile exudate; however, Actinobacillus spp. were isolated and were similar to those isolated in EFL and/or LFL cases. Fifteen cases of FP were recorded in 2002, and 2 cases were reported associated with MRLS in 2003. 2, 3, 54
UU
During MRLS 2001, approximately 40 cases of UU were observed in foals, yearlings, and adult horses of all breeds, ages, and sexes. Yearlings and foals appeared overrepresented in the affected population. The majority of affected horses were presented during the first 2 weeks of May. The condition was unilateral in all cases. The onset of clinical signs was peracute, with profound exudative endophthalmitis. Consistent changes included corneal edema and exudates in the anterior and posterior chambers (Fig. 2). The anterior chamber exudates were tan yellow, presumably proteinaceous, and were often accompanied by hemorrhage from the surface of the iris. A mid-range pupil and vitreal turbidity were confirmed by ultrasound. No eyes were enucleated for histopathologic examination. The culture and cytology results were negative (n = 1). Fewer cases were reported in 2002. 28
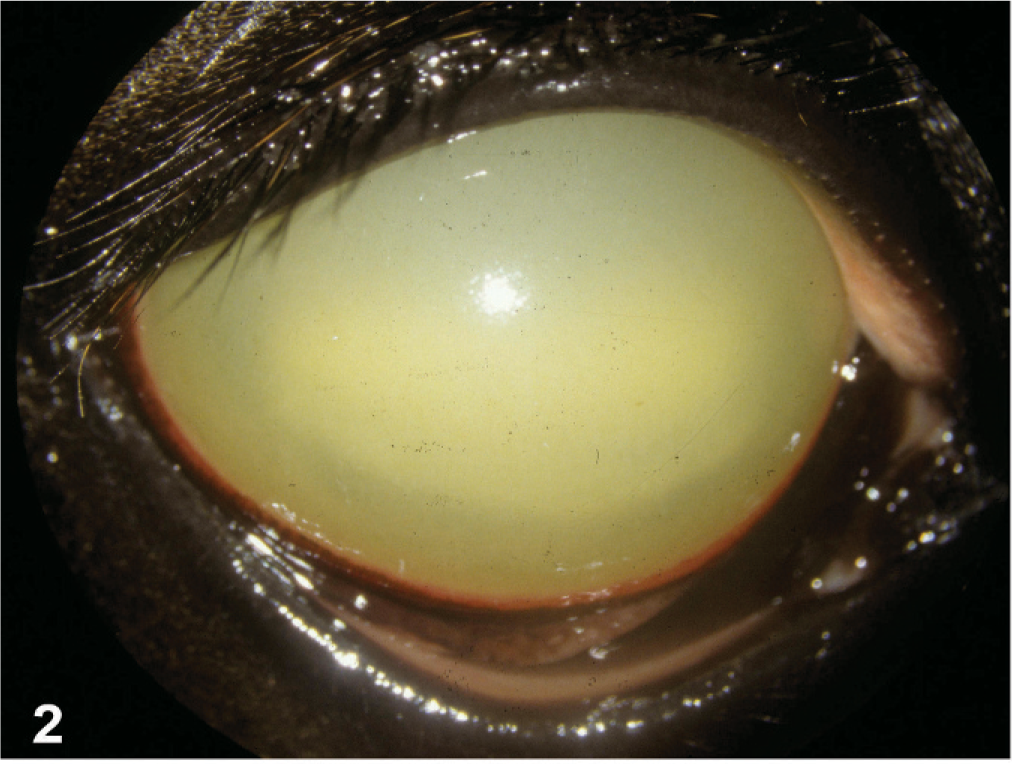
Eye, equine, 2-year-old Thoroughbred colt. Uniform corneal edema, diffuse xanthochromic anterior chamber debris.
Actinobacillus meningoencephalitis
Three cases of Actinobacillus meningoencephalitis (age > 4 years), caused by the same organism associated with the EFL and/or LFL and/or FP cases were diagnosed by a postmortem during MRLS 2001. No cases of Actinobacillus meningoencephalitis were observed during MRLS 2002 in central Kentucky. 44
Pathologic and Microbiologic Findings
EFL
The majority of EFL cases occurred between days 40 and 80 of gestation, with a few cases up to 140 days. A culture of aspirates of allantoic fluid collected from 3 EFL mares in 2001 by using a pipette through the cervix grew alpha Streptococcus bacteria in 2 cases and Escherichia coli in 1 case. Complete blood counts and chemistries from these mares did not show any significant abnormalities. 38, 39
Microscopic examination of uterine biopsy specimens within 7 days of abortion from mares with EFL showed moderate-to-severe inflammation, with a predominance of neutrophils admixed with mononuclear cells. After the abortion, all mares were treated by uterine lavage and were infused with antibiotics based on culture and sensitivity. In subsequent heat cycles, most of the mares had normal uterine cytologic findings and no bacterial growth from the uterus. No significant pathologic changes were observed in the uterine biopsy specimens performed on 10 mares, 1 to 2 months after their abortions in 2001. 38, 39
Most of the EFL fetoplacental units were moderately to markedly autolyzed at the time of submission for a postmortem, which precluded detailed pathologic evaluation and related testing. Most fetoplacental units showed moderate neutrophilic infiltrates in the placental membranes and the amniotic and allantoic fluids. The major finding of EFL was a bacterial placentitis. The fetoplacental units were in a state of moderate-to-severe autolysis, which precluded localization of the inflammation to specific anatomical regions of the placenta, similar to histopathologic findings of LFL, as described later. The same bacteria (the majority were Streptococcus spp. and Actinobacillus spp.) isolated from LFL were isolated from the EFL aborted fetuses. No funisitis was observed in these EFL fetuses. 11, 60, 61
LFL
The majority of fetuses with LFL were between 10 months of gestation and term. Grossly visible lesions included a pale brown, thick, edematous placenta, with intact cervical star, a thick, edematous, yellowish umbilical cord (Fig. 3), and, in some fetuses, pulmonary edema. Characteristic histopathologic changes associated with LFL included funisitis (Figs. 3, 4), placentitis, and perinatal pneumonia. The alveoli contained free bacterial colonies, with and without inflammatory response, and free squamous epithelial cells. 7, 47, 60, 61 Funisitis was a characteristic lesion of LFL and/or MRLS and was mostly confined to the amniotic segment of the umbilical cord. Umbilical cord lesions were generally confined to the surface and consisted of mild-to-dense infiltrations of inflammatory cells, mostly neutrophils. Microscopic lesions observed in the placenta were mostly confined to the coelomic space in the chorioallantoic stroma and were usually perivascular, with a few cases that infiltrated the blood vessel walls (Fig. 5). The inflammatory cells were predominantly composed of neutrophils, with lesser numbers of mononuclear cells. The chorionic villi were not infiltrated by inflammatory cells as observed in classical ascending placentitis. 47, 49 No single lesion was pathognomonic for LFL. The diagnosis depended on the overall evaluation of the case and of correlation with the history. Standard bacteriologic isolation yielded up to 10 different bacterial species from lung, stomach fluid, allantochorion, and placental membranes of LFL 2001 cases. Non-beta hemolytic Streptococcus spp. (51%) and/or Actinobacillus spp. (13%) accounted for the majority of the isolates. 11
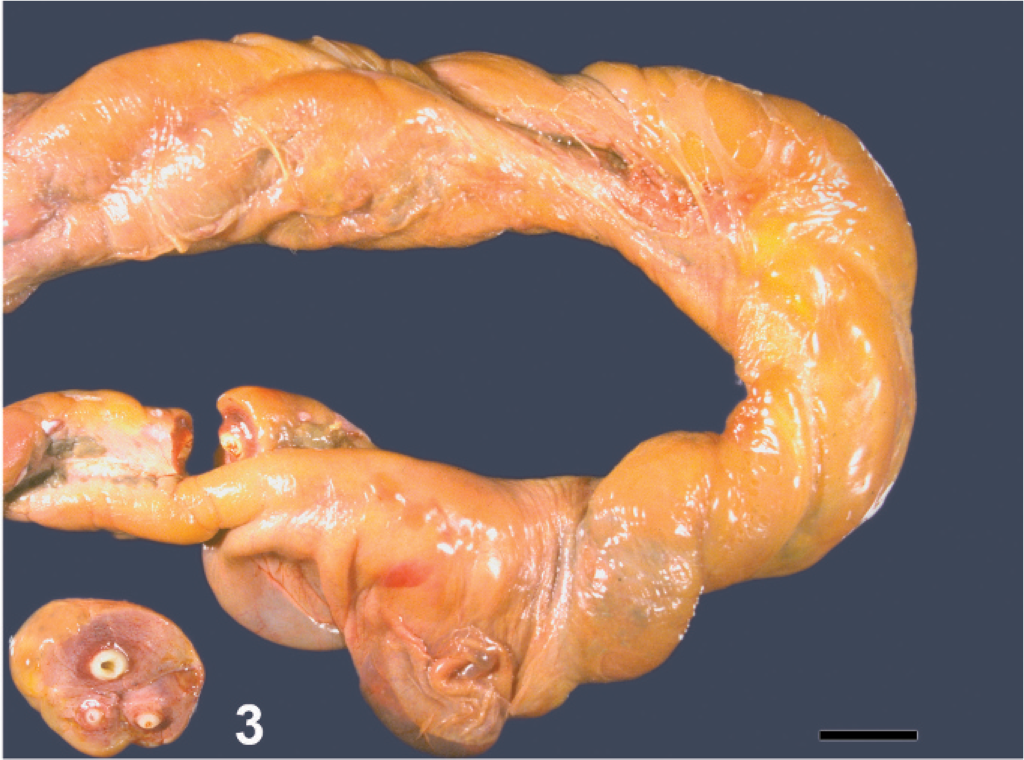
Umbilical cord, equine aborted fetus. Longitudinal view of cord with yellow discoloration on surface. Cross sections, circumferential yellow discoloration on surface. Bar = 2 cm.
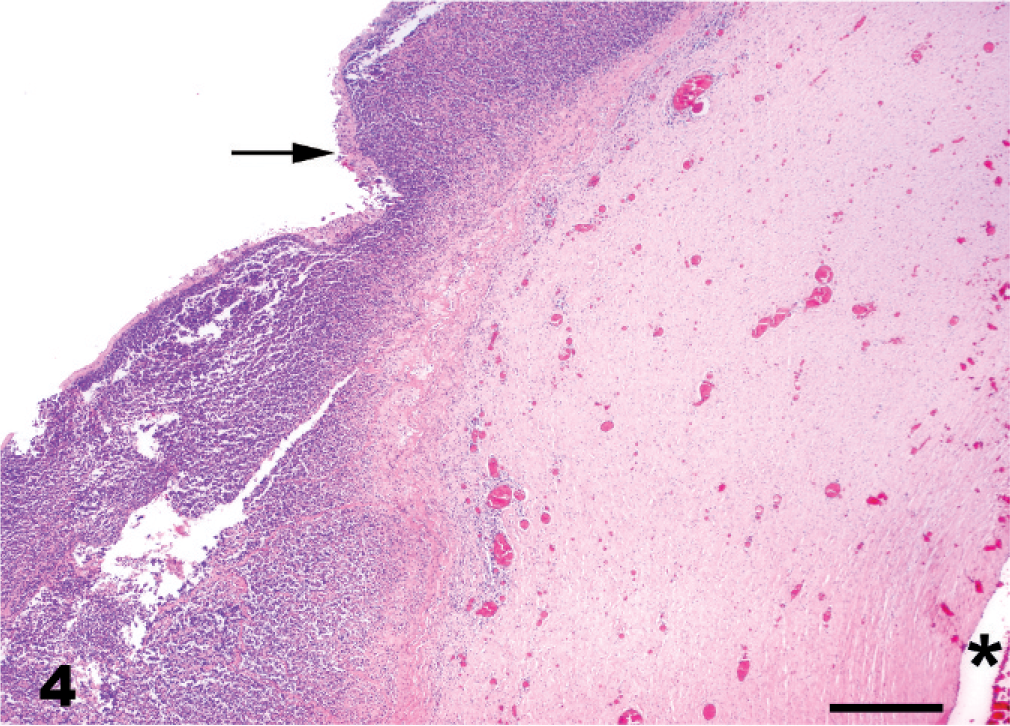
Umbilical cord, aborted equine fetus. Histologic appearance of the yellowish material on the surface of umbilical cord in Fig. 1. Surface of the umbilical cord with inflammatory exudate comprised of neutrophils (arrow). Asterisk = umbilical cord vessel. HE. Bar = 100 µm.
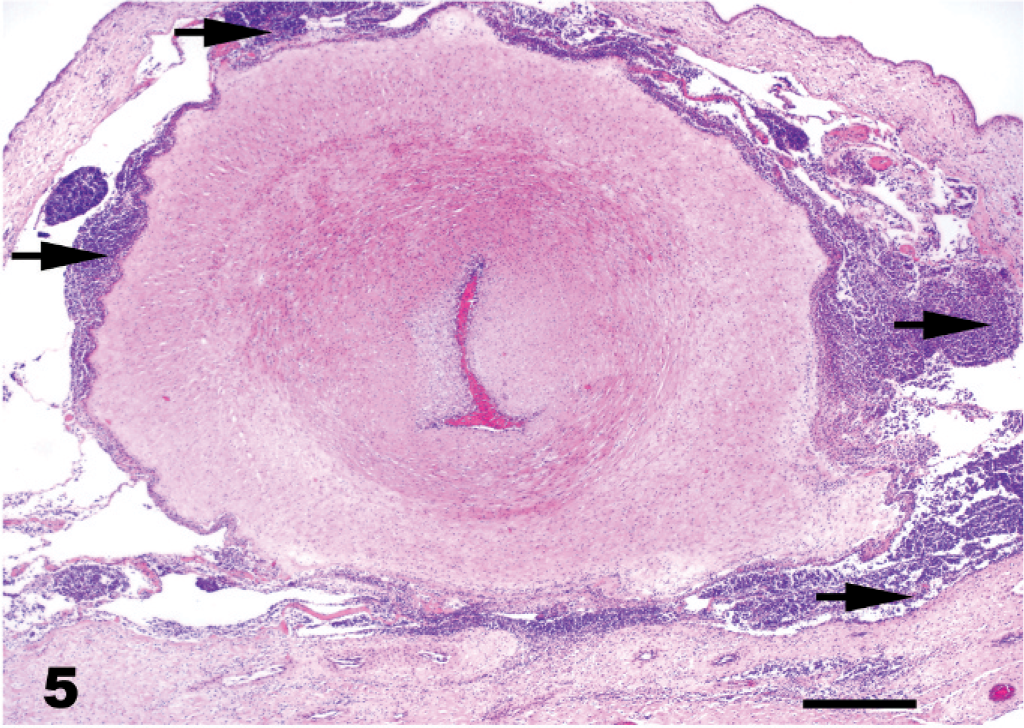
Placenta, equine. Allantochorion with inflammation around a large vessel in stroma/coelomic space of allantochorion. Arrows show inflammatory cells HE. Bar = 100 µm.
FP
The gross findings of typical FP cases included an enlarged heart, with large quantities of fluid, up to 3 liters of serofibrinous fluid in the pericardial cavity (Fig. 6). Histopathologic findings consisted of the visceral pericardium covered with fibrin, admixed with inflammatory cells, predominantly neutrophils, and lesser numbers of lymphocytes and histiocytes. Fibrin tags were observed on the parietal surface of the pericardium. In many cases, the inflammation extended through the epicardium into the myocardium and formed a thick uniform layer of fibrinous-to-fibrinopurulent exudate composed of neutrophils and mononuclear cells admixed with proliferating fibroblasts and large quantities of fibrin. A number of patients with FP failed to survive, in spite of vigorous treatment, and were submitted for a postmortem. Actinobacillus spp. were isolated from pericardial fluid and tissue samples from 8 of 10 nontreated horses of 32 cases submitted for a postmortem during MRLS 2001. Many cases of FP had no bacterial growth probably because of extensive antibiotic administration for this condition. 2, 3
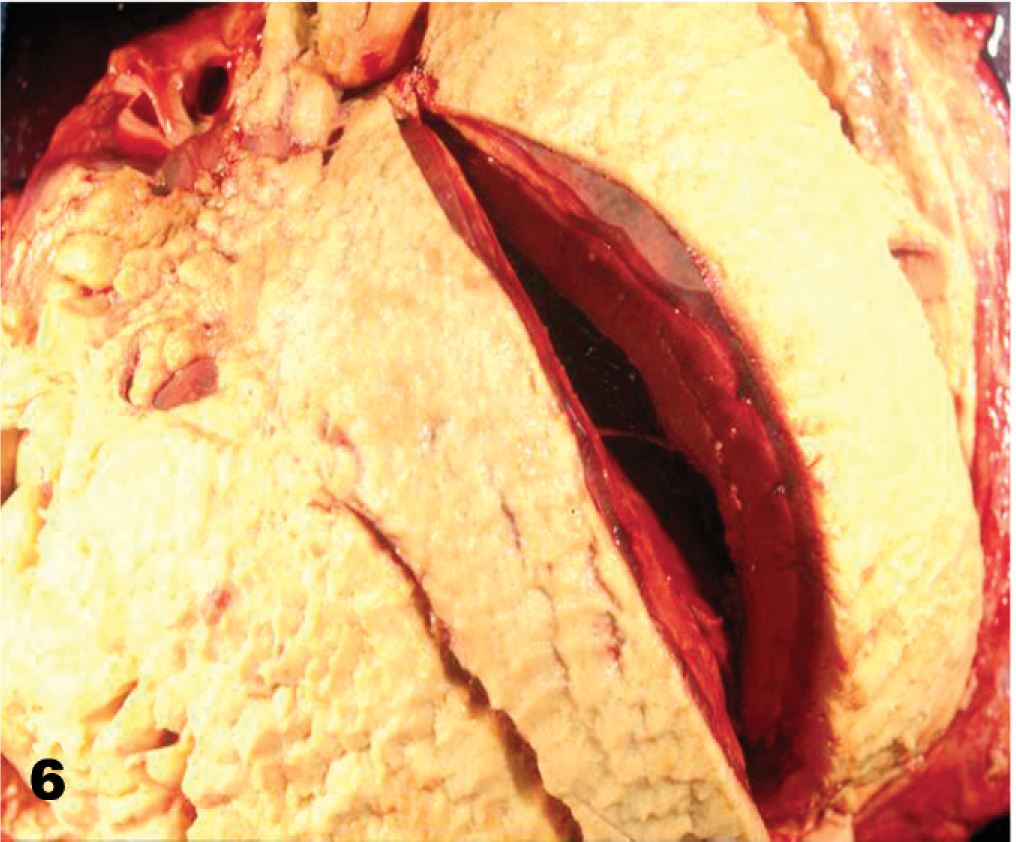
Heart, equine, yearling Thoroughbred colt. Fibrinous pericarditis, surface of epicardium coated with fibrin. The left ventricle is cut open.
No eye tissue from the UU cases was submitted for histopathologic examination, because none of the eyes affected were enucleated. Affected eyes appeared refractive to treatment, and blindness ensued, followed by various degrees of global atrophy. 28 In the 3 cases of meningoencephalitis (multifocal inflammation in the neuropil and meninges of cerebrum and cerebellum) that occurred during the same period, Actinobacillus spp. was isolated from the brain. The isolates were similar to the EFL and/or LFL cases. 44 Detailed virologic and serologic examinations were conducted on the fetal and placental samples, and no viral agents were identified and thus ruled out a primary infectious cause.
Epidemiology and risk factors of MRLS
Multiple epidemiologic investigations were conducted to identify risk factors and the period of exposure to the causative agent associated with MRLS. Risk factors identified included exposure to moderate-to-high concentrations of eastern tent caterpillar (ETC) (Malacosoma americanum) in pastures (Fig. 7), an increased number of ETC in pastures, abortion during a previous pregnancy, the presence of cherry trees, ≥50 brood mares and/or farm, mares being bred during February 2001, drinking from a water trough, or not drinking from water trough, access to water buckets or automatic water taps, etc. Feeding hay to mares on the ground in pasture was associated with a decreased risk of EFL. Pasture composition and management factors were not significantly different between horses in affected and nonaffected pastures. 8, 9, 13 Factors identified as being associated with FP include being from a farm with mares and foals affected by MRLS, exposure to ETC in or around the pastures, younger age, shorter duration of residence in Kentucky and at the farm of current residence, access to orchard grass for grazing, being fed hay grown outside Kentucky, a lack of access to pond water, and a lack of direct contact with cattle. 41 In another study to evaluate the time period of exposure to the etiologic agent of MRLS, it was found that mares in central Kentucky bred between mid February and early March were observed to be at greatest risk of early term abortion. Those mares that were bred after April 1, 2001, appeared to be at less risk of aborting, indicating that exposure to the cause of MRLS (not unidentified) likely occurred before this date. 10 In a retrospective study conducted to evaluate the EFL on 4 equine farms in central Kentucky, it was found that 90% of the fetal losses occurred between 48 and 147 days of gestation and the median age of EFL was 71 days. In the same study, it was observed that 1 farm had fewer fetal losses, which suggested that management or environmental factors may have an effect. The study also ruled out the association of any specific sire used for mating at the stud farm. 32

Eastern tent caterpillars, late instar stage in their nest.
In a study to estimate spatial risks among horses associated with MRLS during 2001 in a specific study population, it was observed that there was considerable data to support an ecologic cause and potential ecologic risk factors for MRLS. 56
Preliminary Investigation of Etiologic Agents
When MRLS was first observed in 2001, a variety of possible causes were considered. A primary infectious cause was initially ruled out because of the lack of clinical symptoms in the aborting mares. Investigations were mostly directed toward an environmental toxin, because the syndrome appeared to be a point-source onset.
Nitrate and/or nitrite toxicosis was ruled out by evaluating fetal fluid samples. Similarly, pasture samples, collected around May 1, 2001, some from heavily fertilized fields, had less than toxic levels of nitrate and/or nitrite. Affected mares did not have prolonged gestations, as seen with fescue toxicosis, and, unlike fescue toxicosis, the placentas from mares with MRLS showed severe inflammatory changes. Additional testing conducted at the University of Kentucky ruled out classic ergot alkaloid and/or fescue toxicosis. 40 Phytoestrogens, ergot, estrogenic, and other mycotoxins were evaluated and ruled out by the first few weeks of the outbreak. 29, 33
During MRLS 2001, there were exceptionally large numbers of late instar ETC dispersing in the horse pastures. Preliminary field surveys confirmed time and place associations between dispersing ETC and MRLS cases. These observations led to a search for an ETC-associated toxin that might cause MRLS. Cyanide is a known toxin associated with the black cherry tree (Prunus serotina), and cherry tree leaves are the primary food source of ETC. 14 Several experiments were conducted in pregnant mares to rule out the role of free cyanide or its precursors in MRLS. None of the experiments resulted in abortions. 21
Black cherry tree (leaf) is the food source of ETC. Prunasin, the primary organic cyanide in cherry tree leaves, is metabolized to mandelonitrile. Mandelonitrile, which spontaneously hydrolyses to cyanide, is also present in the regurgitated materials of ETC and is used by the ETC as a defensive chemical against predators. 15 Experiments that investigated the possible role of the mandelonitrile or free cyanide in inducing abortions in pregnant mares were performed. In early 2002, 6 pregnant mares were challenged with mandelonitrile. In these experiments, mandelonitrile was administered at a dose of 2.0 mg/kg per horse in feed, twice a day, for 14 days to 6 late-term pregnant mares. Each administration yielded a sharp increase in the plasma concentration of cyanide, peaking at about 600 ng/nl, followed by an 8- to 12-hour increase in plasma thiocyanate. Mild diarrhea was observed in the treatment mares. No abortions were observed in any treated mare, which eliminated the role of mandelonitrite in MRLS. 47
Experimental studies with tent caterpillar
When live ETC were available in spring of 2002, experiments were designed to study the natural exposure of pregnant mares to ETC. In these experiments, pregnant mares in small pasture plots were exposed to live ETC or their frass (fecal material). Mares were exposed to ETC and frass (2 treatment groups) for 6 hours each day for a maximum of 10 days or until they aborted. EFL was experimentally reproduced in 7 of 10 mares maintained in 16 × 16-ft experimental plots by exposing the mares to live ETC and frass. 58, 59 However, 3 of 10 control mares in the experiment also aborted, but it was observed that ETCs escaped from treatment plots and reached control plots. These results provided the first experimental evidence that exposure of early term pregnant mares to ETC can cause fetal loss and possibly MRLS.
In a second study with the same protocol, 3 of 7 pregnant mares exposed to ETC aborted; however, no abortions occurred in the frass-exposed group or in the negative control group. The fetoplacental units recovered showed no significant gross findings, but bacterial pathogens similar to field cases of MRLS were cultured from most of the fetal and/or placental tissues. The second experiment established that ETC exposure can cause EFL abortions and suggested that the abortifacient has a greater association with ETC than its frass. 58 Shortly thereafter, pregnant mares (40–80 days of gestation) were challenged with frozen ETC and frass. This experiment was composed of 5 mares administered saline solution (controls), 5 mares administered 2.5 g frass, which was stored at −80°C, by a nasogastric tube, and 5 mares were administered by nasogastric tube 50 g (per day) homogenized refrigerated ETCs that were collected from cherry trees and were maintained without food at 50°C until use (2–4 weeks). All the 3 groups were treated for 10 days. All mares were confined in stalls in a fully enclosed barn during the experiment and were fed hay from Nevada. Four of 5 mares treated with ETCs aborted during days 8–13 of the experiment. No abortions were observed in control group mares and mares administered frass for 1 month after the end of the experiment. This study confirmed the relationship of ETC exposure to fetal losses in early stages of gestation, consistent with MRLS, and ruled out involvement of Kentucky hay or pastures. 1
The first LFL study was conducted by nasogastric administration of homogenized ETC collected from the upper peninsula of Michigan (ETCs were collected from black cherry trees, Prunus serotina) to late term (7–11 months) pregnant mares. Six mares were challenged with 50 g of fresh ETC via nasogastric intubation for 9 days. The ETC were fed fresh Kentucky black cherry leaves. Five mares served as saline solution controls. All mares were confined in individual stalls in a fully enclosed barn for the duration of the experiment. Pregnancies were confirmed by ultrasonographic observation before the experiment. Placental thicknesses and fetal heart rates were recorded during the experiment. Fetal heart rates increased in 5 of the 6 fetuses of the ETC-exposed mares. These same 5 fetuses aborted at 62, 69, 95 142, and 166 hours after the first treatment. The sixth mare aborted 6 days after receiving the last dose of ETC. All placentas had intact cervical stars. No control mare aborted. Abnormal clinical signs or blood chemistries were not observed. Bacterial culture of mare's blood was negative. The intact cervical stars and the light brown–yellow appearance of the allantochorions in this experiment suggested that placental detachment happened in utero. 42, 47 It is well known that placental detachment leads to hypoxia, which results in fetal death and abortion. So this experiment suggested that the abortifacient component of ETC may be acting on the uteroplacental interface and leading to placental detachment, which may be the probable pathogenesis. This experiment confirmed that ETC exposure was abortigenic for mares and also demonstrated that ETCs from a region other than central Kentucky were capable of causing abortions. 42, 47
The potential of different anatomical components of ETC to cause abortion were evaluated in pregnant mares. The integument of ETC was separated from their digestive tracts. One hundred grams of integument or digestive tract was administered orally to 6 mares (early to mid gestation stage) for 5 days. Two of the 5 mares administered the ETC integument fraction aborted, whereas, the 5 mares administered the digestive-tract contents did not abort. The results of this experiment suggested that ETC abortifacient property was associated with the ETC integument and not the digestive-tract components. This experiment did not identify the factors or anatomic structures of the ETC integument that are abortigenic. 24, 52
Later, the ability of frozen ETCs, autoclaved ETCs, and gypsy moth caterpillars (GC) (Lymantria dispar), a hirsute caterpillar, to induce MRLS was investigated in detail. Fourteen mares in the early stage of gestation were divided into 3 treatment groups. These three groups were administered 50 g of previously frozen ETC, 50 g ETC that were frozen and then autoclaved, or 50 g of frozen GC. The ETC and GC had been preserved at –80°C for approximately 4 months. Three of 5 mares administered frozen ETC aborted. None of the 5 mares administered ETC that was autoclaved before administration aborted. One of the 4 GC-treated mares aborted; however, the abortion was excluded from the analysis. The signs of pregnancy loss in this mare was not consistent with those of mares fed with ETC or MRLS field cases based on the observation that this fetus failed to grow at a normal rate and died, and the allantoic fluid volume appeared to decrease before fetal death, without a marked increase in echogenicity, an increase in echogenicity being a common observation in EFL abortions. This experiment suggested that the abortifacient property of ETC was not lost during freezing, being stable for at least 4 months at –80°C but that the abortigenic factor was inactivated by autoclaving. 59
Another study was conducted to determine the potential of alcohol-sterilized ETCs to induce MRLS. Fifteen healthy mares in mid-gestation was divided into 3 treatment groups: group A, the test group received ethanol-treated ETC; group B, positive controls received untreated ETC; and group C, negative controls received 10% ethanol diluted in deionized water. Fetal viability in mares was evaluated by ultrasonography. Each day, 250 g of frozen ETC and 500 ml of 95% ethanol were blended in a mechanical blender for 5 minutes and allowed to sit at room temperature for 2 hours. This homogenate was cultured to assure the absence of viable bacteria. The homogenate was then divided into 5 aliquots, yielding a 50-g dose, which were diluted by adding deionized water, which brought the final ethanol concentration to 10%. A 50-g dose was administered to each mare for 10 consecutive days via a nasogastric tube. In the group B mares, saline solution replaced the ethanol, whereas, the negative-control mares received 200 ml of 10% ethanol. Two of the 5 mares in the positive untreated-ETC control aborted, and 2 of the 5 test mares that received the ethanol and/or ETC homogenate aborted. These abortions were considered typical of MRLS. No abortions were observed in the ethanol sham-control groups. This experiment suggested that the causative agent was not an ETC-associated bacterium. Although ethanol treatment was bactericidal, it may not have been potent enough to destroy viruses, and, hence, the possibility of a viral agent as the cause of MRLS was excluded. 51, 52
Later in 2002, the abortifacient potential of a closely related Forest Tent Caterpillars (FTC) (Malacosoma distria) to induce equine abortion was evaluated. These FTC were reared in a laboratory, and the FTC homogenates were shipped frozen overnight to Kentucky. Four mares in the early stages of gestation were administered 100 g of FTC homogenate each for 5 days via a nasogastric tube. A second group of 4 mares administered normal saline solution by a nasogastric tube served as controls. There were no abortions in the FTC challenged or the control groups. This experiment suggested that the abortifacient responsible for MRLS is not uniformly present in all Malacosoma and/or tent caterpillar species. 51
In 2003, the abortifacient potential of different anatomical components and filtrates of ETC were evaluated in detail. This experiment consisted of 7 groups of 5 pregnant mares in early stages of pregnancy, and the ETC used in the experiments had been preserved at –80°C. Group 1, the positive controls, was fed 50 g ETC homogenate mixed in sweet feed. Group 2, the negative controls, were fed saline solution mixed sweet feed. Group 3 was fed 50 g exoskeleton, group 4 was fed 50 g gut, and group 5 was fed 50 g of the remaining internal body parts. Group 6 were fed filtrates of a phosphate-buffered saline solution homogenate of ETC that had been filtered (<45 microns), and group 7 was fed the retentate from this preparation. All 5 mares in group 1 positive-control group aborted. Three of 5 mares in group 3 fed with exoskeleton aborted. One mare in the group 7, fed the retentate aborted at day 31. None of the mares in group 2 (negative control), group 4 (fed with gut), and group 5 (remaining internal body) aborted. This experiment suggested that the abortifacient is associated with ETC exoskeleton, 59, 60 which supported the findings of an earlier study. 24 This study also suggested that filtration of the homogenates appear to have removed the abortifacient component of ETC.
In another experiment in 2003, the effects of irradiation on the abortigenic factor in ETC were evaluated. Homogenates of ETC prepared in normal saline solution were exposed to 30 kGy units of cobalt irradiation, a level demonstrated to destroy and/or inactivate marker bacteria and viruses while not significantly altering the activity of enzymatic or chemical markers. The ETC used for this experiment were collected from central Kentucky and were frozen at –80°C until required. Eighteen late-term mares were randomly divided into 3 groups of 6: group A, the positive-control mares were administered 100 g ETCs; group B, the test mares, were administered 100 g of irradiated ETCs; and group C, the negative controls, were administered normal saline solution for 10 days. All test material and saline solution were administered via a nasogastric tube. All 6 mares in group A aborted within 32 to 120 hours after the first dose. Three of 6 mares in group B aborted at day 12, 14, and 24 after the first dose. These aborted fetuses had gross and histopathologic changes that closely resembled those observed in the MRLS field cases, which included funisitis, placentitis, and bronchopneumonia. This was the first experiment in which the typical field MRLS funisitis was observed in experimental MRLS cases, funisitis being a typical and to some extent defining lesion in field MRLS cases of Actinobacillus spp. (1 fetus) and Streptococcus spp. (2 fetuses) were cultured from these fetoplacental units. 45, 50 The results of this experiment suggested that frozen ETCs collected in 2003, preserved at –80°C, will induce abortion in late-term gestation. The study also suggested that irradiation might reduce the abortifacient potency of 2003 ETCs.
The immune status of horses experimentally exposed to ETC was also evaluated. Blood samples collected before and 7 days after oral administration of irradiated and nonirradiated ETC were tested for lymphocyte subpopulation distribution, lymphocyte proliferation, phagocytosis and oxidative burst capacity, opsonic capacity of serum, and serum immunoglobulin isotype levels. No definitive evidence of an immunosuppressive condition was observed in the study. 16
Laboratory Animal Models of MRLS
Attempts to identify a laboratory model of MRLS by using mice have been unsuccessful. Various preparations of fresh and frozen ETCs, ETC homogenates, ETC frass, and ETC setae (hair) fragments were administered orally to pregnant mice, ranging from 5–12 days of gestation, and the effects were evaluated on pregnancies and litter sizes. No statistically significant fetal resorption or abortion was observed. 43
Experimental studies in pregnant rats fed finely chopped ETC mixed in feed from day 4 of gestation through the end of gestation did not yield any statistically significant fetal resorption or abortion, which indicated that ETC do not induce abortions in rats. Histopathologic examination of the gastrointestinal tissue revealed granulomas with setal fragments in their center. 47
The abortifacient potential of ETC was investigated in pregnant pigs. Five mid gestational gilts were fed 40 g of fresh frozen ETCs per day, mixed in their concentrate ration for 10 days. Five gilts fed with concentrate ration served as controls. Two of the 5 gilts fed ETCs aborted their litters, whereas none of the controls aborted. A postmortem was performed on the remaining gilts before parturition. Streptococci spp. were isolated from fetuses of all the gilts fed ETCs, aborted or not, and from the fetuses of 1 control gilt. Numerous caterpillar setae were observed in granulomas located in the mucosa and submucosa of the intestinal tracts of gilts fed with ETCs. This experiment suggested that species other than equine can be affected by the ETC-related abortifacient. 30
Discussion
Neither the experimental mares in experiments that were administered substantial doses of ETC nor the field cases of EFL and LFL demonstrated significant clinical signs of illness despite abortion. Consistent with the absence of clinical signs, no abnormalities in clinical chemistry panels or complete blood counts were observed in the natural or experimental cases. Blood cultures from mares in field or experimental cases of MRLS were also negative. MRLS is also unique in specific location of lesions in fetoplacental units, pericardium, and eye. There is no definite explanation for these specific locations. The LFL-MRLS fetoplacental units had unique histopathologic findings. The histopathologic studies conducted on placenta showed no indications, suggestive of either an ascending infection via the cervix, or hematogenous routes. Ascending infection via the cervix was ruled out by the absence of inflammation in the cervical star areas. The intact cervical star in placentas of aborted experimental late-term fetuses also supports this inference. The hematogenous route of infection is unlikely, because there was no significant evidence of inflammation on the chorionic surface of the allantochorion. In LFL-MRLS, histopathologic findings consisted of inflammatory cells, primarily neutrophils, that infiltrated the perivascular areas of the chorioallantoic stroma with very rare inflammatory cells in the villi. Hence, the microscopic lesions observed in the allantochorion suggest a primary vascular involvement. 47, 49 In LFL fetuses submitted to the LDDC, no single pathologic finding was identified as being specific for MRLS, although a characteristic finding was funisitis. Funisitis has rarely been reported in the veterinary literature as a primary lesion. 36, 48 The specific pathogenesis of the characteristic funisitis of MRLS is unknown. The inflammatory cells observed on the superficial layers of the umbilical cord appear to have emigrated from the umbilical cord vessels in response to a chemotactic factor in the amniotic fluid. This chemotactic factor may have stimulated leukocytes to diffuse through the Wharton's jelly from the umbilical vessels to the amniotic fluid on the surface of the umbilical cord. 19 As mentioned earlier, no single pathologic finding was pathognomonic for MRLS-associated late-term abortions. 7 A pathologist can only rule out MRLS-associated placentitis based on the history of exposure to caterpillars and, to a degree, the absence of inflammation on the chorionic villi.
Before 2001, FP was a rare clinical or pathologic condition in the equine population of central Kentucky. The incidence of equine FP in central Kentucky from May through September of 2001 represents a substantial increase of this condition when compared with the previous year's prevalence of 4 cases/year. 3 The incidence of FP during MRLS 2001 was 8- to 10-fold above the mean local prevalence, therefore, warranting the inclusion of this condition in the case definition of MRLS. One case of FP was observed in a mare that aborted as part of EFL. FP and UU together had not been identified clinically or at a postmortem in any mares that aborted during the MRLS period. Similar to the FP cases, the occurrence of UU cases coincident with MRLS 2001 and 2002 indicates that the UU cases were highly correlated with the MRLS but is a distinctive clinical condition of MRLS. UU was not diagnosed in any mare affected by EFL or LFL. Neither FP nor UU was reproduced in any of the ETC-exposure experiments. It may be possible that different routes or duration of exposure to the etiologic agent may increase the probability of these relatively rare MRLS events.
Before MRLS, it was rare for the bacterial species Actinobacillus spp. and Streptococcus spp. isolated from fetuses and placental membranes to be associated with equine abortion and/or placentitis. In retrospective studies conducted on equine abortions in Kentucky, these bacteria were uncommon causes of equine abortion. 18, 26 Actinobacillus spp. isolates recovered from fetuses and pericardial fluid samples of horses affected by MRLS in 2001 to 2003 were found to be identical to Actinobacillus spp. found in the oral cavity and alimentary tracts of healthy horses. 12 The bacteriologic findings associated with MRLS are remarkably different than those previously reported as being associated with the abortion and FP in horses submitted to LDDC.
Increased echogenicity of fetal fluid was one of the clinical parameters for identification of EFL. Before MRLS, there was no established baseline for echogenicity of fetal fluids at varying stages of normal gestation. An ultrasonographic study was conducted in non-MRLS mares in which hyperechogenicity was not observed before day 85 of gestation, but hyperechogenic fetal fluids were observed after day 85 of gestation. 57 This study demonstrated the remarkable clinical diagnostic correlation of fetal-fluid hyperechogenicity and EFL-MRLS. Cytologic examination of allantoic fluid aspirate from 3 mares affected with EFL showed sheets of squamous epithelial cells, coccoid bacteria in chains, and occasional and/or rare neutrophils, which suggested a poor host cellular response. 39 Based on these findings, the hyperechoic material observed during an ultrasonographic examination is mostly sheets of squamous epithelial cells, presumably sloughed allantoic epithelium.
The effect of MRLS on future fertility status was also investigated. The EFL-MRLS affected short-term fertility but not long-term fertility. The LFL-MRLS did not affect either short- or long-term fertility. To analyze the effect of MRLS on fertility, breeding histories of 31 mares that aborted during MRLS 2001 were evaluated. Thirty mares conceived the following year, which indicated that MRLS did not have a chronic negative impact on fertility. 39 Furthermore, it was reported that 2001 MRLS-affected mares bred in 2002, conceived and maintained their pregnancies at rates consistent with prior years. There was no substantial evidence that MRLS had a long-term effect on fertility.
Many therapeutic approaches to EFL and LFL were attempted, and many were unsuccessful. Prevention of ETC exposure to horses included cutting down cherry trees and spraying of ETC nests with insecticides and avoiding direct exposure to ETC by muzzling mares while in pasture. Those farms that adopted measures for ETC control had a lower incidence of MRLS in 2002.
Epidemiologic studies conducted immediately after MRLS 2001 identified ETC as one of the risk factors that resulted in designing and conducting experiments with ETC. Black cherry leaves, the primary food source of ETC, is known to contain cyanide. Cyanide is known to produce tissue hypoxia and was initially suspected to be directly linked to the pathogenesis of MRLS. The experiments conducted during the summer and fall of 2001 ruled out the role of free cyanide as an abortifacient agent in MRLS. 21 The experiments with live ETC closely replicated the environmental conditions that established a direct association between ETC exposure and MRLS. 58, 59 In one of the experiments conducted by using live ETC, few mares aborted after being exposed to frass, which raised a question about the role of ETC and frass in MRLS. The experiments conducted later suggested that ETC but not the frass caused MRLS. 1, 58 Experiments conducted during the fall of 2002 demonstrated that ETC from regions other than central Kentucky can cause abortion and MRLS in pregnant mares. 42, 47
Two similar experiments that separated anatomical components of ETC yielded results that suggested that MRLS may have a specific association to the integument and/or exoskeleton of ETC. 24, 59 In these experiments, pregnant mares exposed to integument and/or exoskeleton fractions aborted. These observations suggest that the specific abortifacient may be either a structural component or a material in the integument of ETC. Experiments conducted by using autoclaved ETC did not cause abortion suggests that the abortifacient agent is heat labile. 59 The experiment conducted by using ETC homogenate mixed in ethanol resulted in abortion, which suggested that bacteria associated with ETC is not the cause of MRLS; however, this experiment did not rule out the involvement of a virus in the ETC. In studies that used irradiated ETC, abortions, albeit fewer, occurred. The bacteria isolated from the aborted fetuses and their membranes did not originate from the ETCs, because the ETCs were “sterile” after irradiation. Results of this experiment strongly suggest that an ETC resident bacteria or virus is not the cause of MRLS. Hence, the abortifacient appears to be a factor other than bacteria and virus in the ETC. 45
The only Malacosoma species other than ETC that is present in Kentucky is FTC. 14 The studies with laboratory-raised FTC suggest that the abortifacient property may not be present in the entire genus of Malacosoma, but the abortifacient potential of wild FTC cannot be ruled out, because the laboratory-raised caterpillars were maintained on an artificial diet compared with the natural diet of FTC. Analysis of the time course of abortions observed after experimental administration of different doses of ETC via a nasogastric tube to late-term mares showed that the time course of these abortions well fitted an unusual statistical model called accelerated failure time analysis. This analysis showed that, when the dose of and/or exposure to the caterpillars is large, as occurred in central Kentucky in 2001, exposed pregnant mares are likely to abort rapidly, within a period of 1–7 days of initial exposure. When the dose of caterpillars is smaller, the lag time is longer, the time to peak abortions is delayed, and abortions can occur after dispersion of the caterpillars has stopped, obscuring both the diagnosis and the role of caterpillars in this syndrome. 46
Currently, 2 hypotheses are being investigated. The first is that MRLS is caused by an unidentified toxin carried and/or deposited in the environment by the ETC that is either toxic to the placenta or the fetus The second hypothesis is that bacteria in the oral or gastrointestinal tract enter blood stream of the mare, after a breach of the normal intestinal mucosal barrier by the hairs of ETC and eventually cross the placenta, reach the fetus, and result in the death of the fetus by septicemia. A comparison of the first abortion time between 50 g and 100 g dosing of ETC suggests that, when the dose was doubled, the first abortion happened in approximately half the time, which suggests that abortion appears to be dose related, which supports the toxin hypothesis. 42, 45, 50 The absence of abortion in mares exposed to GCs and FTCs, which do have numerous hairs similar to ETC, also suggests that the hairs in ETC are not capable of inducing abortions by themselves, thereby supporting the toxin theory. 51, 52
An abortion storm similar to MRLS is believed to have occurred previously in Kentucky, during 1981 and 1982, when estimated losses of 256 and 162 foals, respectively, were reported in central Kentucky. However, a definite cause or etiologic agent was not identified in either year. 5 Equine abortions similar to MRLS were reported recently from Florida and New Jersey. The mares that aborted were also exposed to ETC. Recently, an equine abortion storm similar to MRLS occurred in Australia (2004–2005), and current studies show that the exposure to processionary caterpillars (Ochragaster lunifer) was considered as an important risk factor for abortion. 35 A recent study conducted in pregnant goats to evaluate the abortifacient potential of ETC showed that ETC are not abortigenic in pregnant goats. An histopathologic examination of the gastrointestinal tract of the goats exposed to ETC showed numerous granulomas with setae (hair of caterpillar). 53 This experiment suggests that ruminants are resistant to the abortigenic potential of ETC, thereby underscoring that the abortifacient property of ETC is species specific.
Future studies to identify the pathogenesis are ongoing, and the primary limitation for defining the specific pathogenesis is the failure to identify a laboratory animal model as of yet. Currently, the only method to test a hypothesis is to conduct experimental studies in pregnant mares, which is expensive, time consuming, and is limited to a small number of animals. These factors appear to be the most important hurdle for future studies. Future studies on pathogenesis should be concentrated toward evaluating the various anatomical components of the ETC and also chemical evaluation of ETC with subsequent animal trials in pregnant mares.
Footnotes
Acknowledgements
The authors thank Dr. N. Cox, Associate Dean for Research, College of Agriculture, University of Kentucky; Department of Veterinary Science, University of Kentucky; Kentucky Thoroughbred Owners and Breeders Association; Keeneland Association Inc.; and the College of Veterinary Medicine, University of Georgia, for the research funds and support.
