Abstract
Porcine circovirus 2 (PCV2) is the cause of postweaning multisystemic wasting syndrome (PMWS). The most common lesions of PMWS are lymphohistiocytic to granulomatous lymphadenitis, interstitial pneumonia and interstitial nephritis, with intracytoplasmic amphophilic botryoid inclusion bodies in macrophages. In addition to these typical changes, intracytoplasmic botryoid inclusion bodies were observed in bronchial, bronchial glandular, and renal tubular epithelium of several pigs from 4 different farms in Western and Eastern Canada. PCV2 inclusion bodies were demonstrated to be located in the cytoplasm of epithelial cells by immunohistochemical staining for PCV2 and cytokeratin antigens and by ultrastructural demonstration of viral particles in the inclusion bodies within renal tubular epithelium.
Keywords
Porcine circovirus 2 (PCV2) is associated with several porcine disease entities, the first of which was initially described in Western Canada and named postweaning multisystemic wasting syndrome (PMWS). 4– 6, 11, 12 The most common lesions of PMWS are (1) generalized lymphoid depletion with lymphohistiocytic to granulomatous lymphadenitis, often with intracytoplasmic amphophilic botryoid inclusion bodies in macrophages, (2) lymphoplasmacytic and histiocytic interstitial pneumonia, and (3) lymphoplasmacytic and histiocytic interstitial nephritis. 4– 6 Typical amphophilic botryoid inclusion bodies were initially reported only in the cytoplasm of mononuclear/histiocytic cells of pigs with natural PCV2 infection. 4, 5, 15 In 2003, Hirai et al. reported the presence of typical PCV2 intracytoplasmic inclusions in hepatocytes of 1 out of 8 colostrum-deprived piglets experimentally infected with tissue homogenates obtained from pigs with PMWS. 13 The following year, Bastianello reported the presence of typical PCV2 inclusions in hematoxylin and eosin (HE)-stained renal tubular epithelium of 9% of pigs (n = 37) affected with PMWS in New Zealand. 2 In 2006, Imai et al. confirmed by immunohistochemistry and electron microscopy the presence of intracytoplasmic PCV2 inclusions in the renal epithelium of a pig kept as pet. 14 This study describes the presence of PCV2 inclusion bodies in the cytoplasm of bronchial, bronchial glandular, and renal tubular epithelial cells.
Twelve 7-to-14-week old pigs from 4 different farms (2 in Saskatchewan and 2 in Prince Edward Island) were examined. PMWS was diagnosed in pigs on 3 of these farms based on weight loss, typical microscopic lesions, and demonstration of PCV2 antigen and DNA within lesions (Table 1). Presence of PCV2 viral antigen and DNA was demonstrated by standard diagnostic polymerase chain reaction (PCR), immunohistochemistry, or fluorescent antibody test (FAT) performed by Prairie Diagnostic Services, Inc., at the University of Saskatchewan, or by the Regional Diagnostic Virology Laboratory at the University of Prince Edward Island (Table 1). In addition to previously reported typical lesions associated with PMWS, intracytoplasmic epithelial inclusion bodies were observed in lungs and kidneys (Figs. 1 and 2, respectively) processed routinely for histologic examination and stained with HE in 7 pigs (Pig Nos. 1–7). Immunohistochemistry and electron microscopy were used to further characterize these intracytoplasmic epithelial inclusion bodies.
Summary of pathologic lesions and Porcine circovirus 2 (PCV2) test results for pigs with intraepithelial cytoplasmic inclusion bodies.∗

SK = Saskatchewan, Canada; PEI = Prince Edward Island, Canada; N/A = Not available or not done; Y = Yes; N = No; + = Positive; — = Negative.
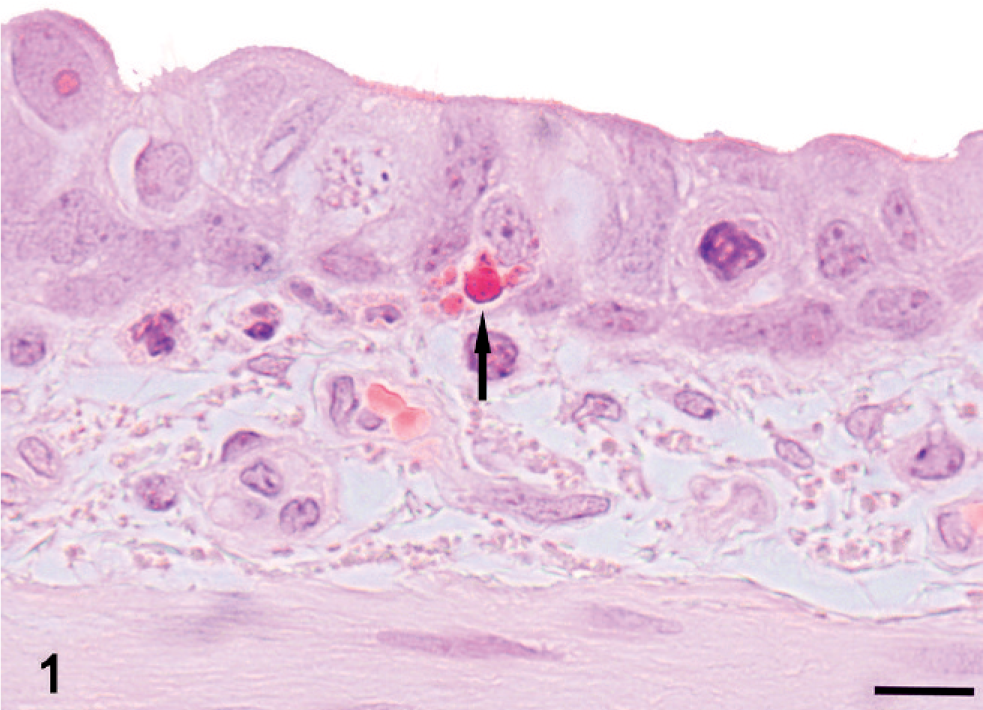
Lung and kidney; Pig No. 3 and Pig No. 6, respectively. Basophilic inclusion bodies (arrows) in cytoplasm of a bronchial ciliated epithelial cell (Fig. 1, Bar = 10 µm) and renal tubular epithelial cells (Fig. 2, Bar = 25 µm). HE.
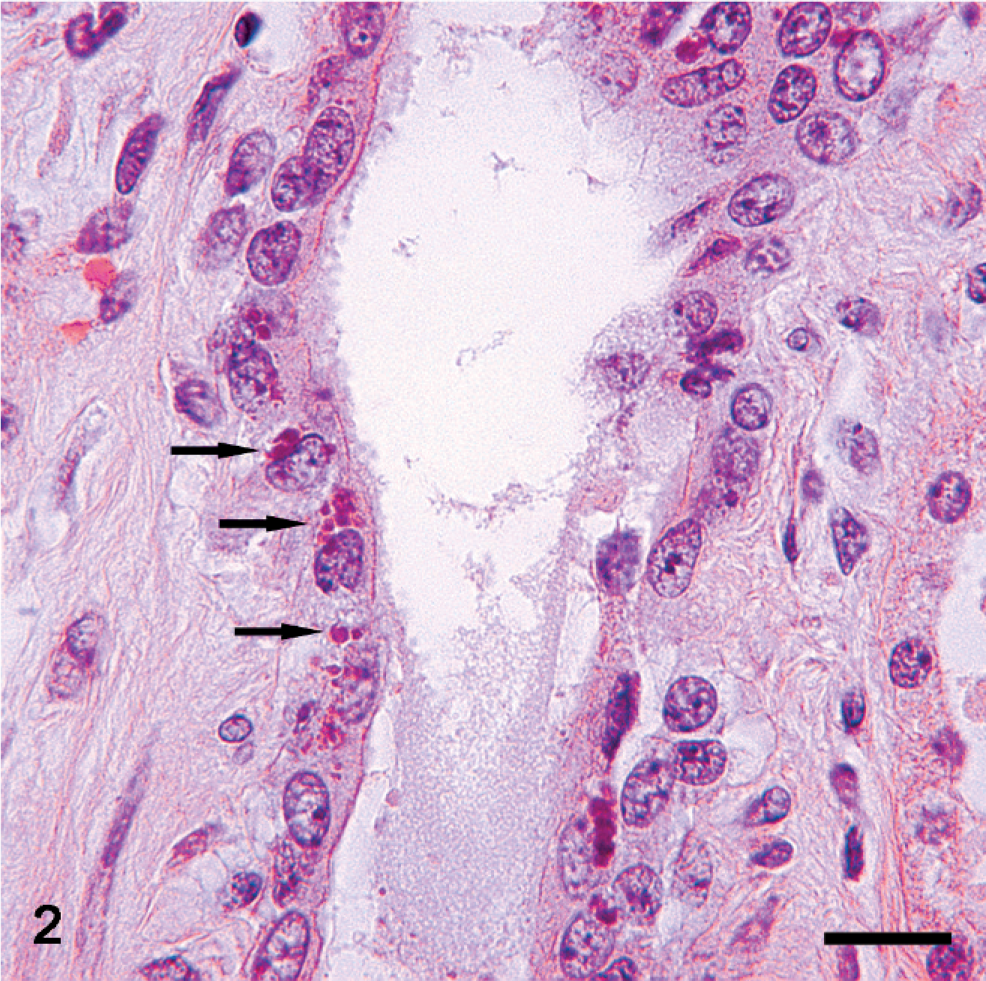
Lung and kidney; Pig No. 3 and Pig No. 6, respectively. Basophilic inclusion bodies (arrows) in cytoplasm of a bronchial ciliated epithelial cell (Fig. 1, Bar = 10 µm) and renal tubular epithelial cells (Fig. 2, Bar = 25 µm). HE.
Immunohistochemical staining for PCV2 antigen was performed on formalin-fixed, paraffin-embedded tissues using a streptavidin-biotin complex technique adapted for an automated slide stainer as previously described. 8, 10 The primary chicken anti-PCV2 antibody (1 : 1000 dilution) (donated by Dr. G. M. Allan, Department of Agriculture for Northern Ireland, Belfast, UK) was used for detection of PCV2 antigens in the inclusion bodies observed within the cytoplasm of epithelial cells. To demonstrate that PCV2 inclusion bodies were located in the epithelial cells, serial histologic sections were subjected to antigen retrieval by protease treatment and subsequently to immunohistochemical detection of cytokeratin using rabbit antibody raised against a wide spectrum of cytokeratin (1 : 2000, Dako Canada, Inc., Mississauga, ON, Canada). Transmission electron microscopy was performed on paraffin-embedded, HE-stained tissue sections according to the previously published protocol. 3
In routine HE histologic sections, multiple amphophilic botryoid intracytoplasmic inclusion bodies were observed in bronchial epithelium of 5 pigs (Fig. 1), bronchial glandular epithelium of 6 pigs (Fig. 4A), and in renal tubular epithelium and necrotic cells within renal tubules of 3 pigs (Table 1, Figs. 2, 3A). Immunohistochemical staining of serial sections of lung and kidney from pig No. 2 revealed the presence of PCV2 antigen in intracytoplasmic inclusion bodies in bronchial glandular and renal tubular epithelial cells (Figs. 4B and 3B, respectively) and confirmed that these PCV2 positive inclusions were within cytoplasm of bronchial glandular and renal tubular epithelial cells based on positive cytokeratin staining (Figs. 4C and 3C, respectively). In addition, electron microscopy demonstrated that intracytoplasmic inclusion bodies observed in HE-stained renal tubular epithelium of Pig No. 2 were located within renal tubular epithelial cells exhibiting microvilli on the luminal plasma membrane, and that the inclusions consist of a paracrystalline array of icosohedral viral particles (Figs. 5A, B). Similar viral particles were also present in the nuclei of renal tubular epithelial cells (Fig. 5C).
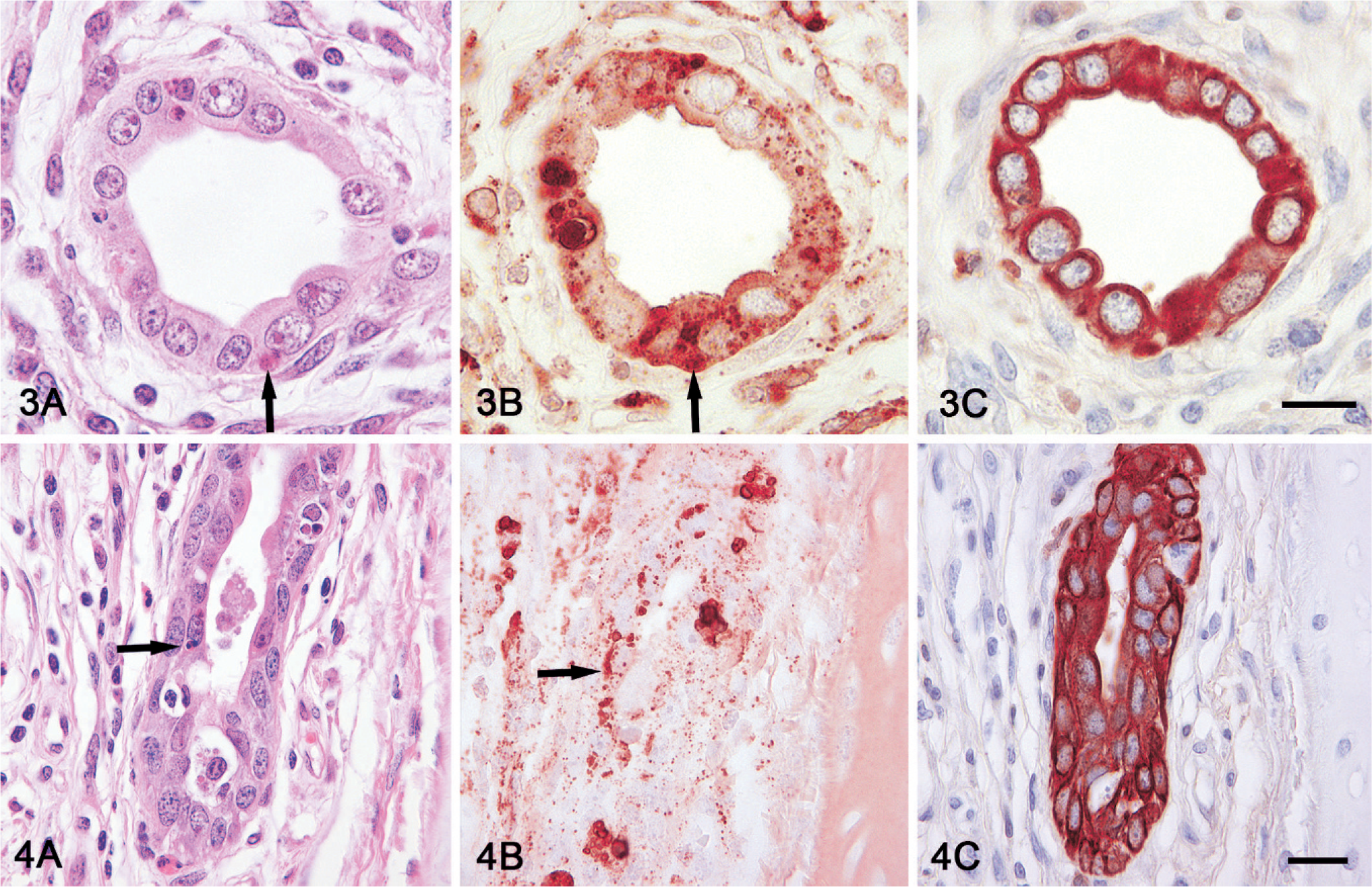
Serial histologic sections of kidney and lung; Pig No. 2. Basophilic inclusion bodies (arrows) in cytoplasm of renal tubular (3A) and bronchial glandular epithelial cells (4A) stained with HE. Positive immunohistochemical staining of inclusions with Porcine circovirus 2 (PCV2) antibodies (3B and 4B) and epithelial cells containing inclusions with cytokeratin antibodies (3C and 4C). Bar = 25 µm. Serial histologic sections of kidney and lung; Pig No. 2. Basophilic inclusion bodies (arrows) in cytoplasm of renal tubular (3A) and bronchial glandular epithelial cells (4A) stained with HE. Positive immunohistochemical staining of inclusions with Porcine circovirus 2 (PCV2) antibodies (3B and 4B) and epithelial cells containing inclusions with cytokeratin antibodies (3C and 4C). Bar = 25 µm.
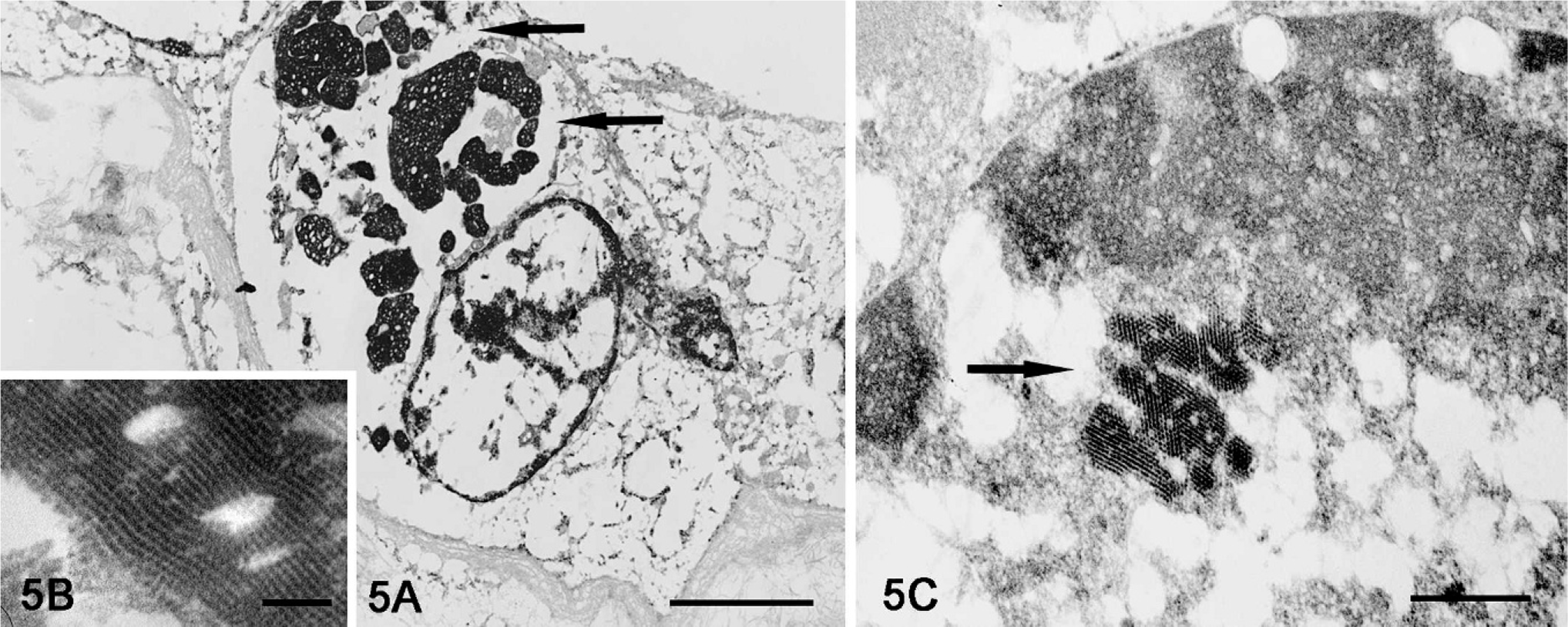
Kidney; Pig No. 2. Electron dense inclusion bodies (arrows) in cytoplasm of a renal tubular epithelial cell (5A, Bar = 2500 nm) are composed of paracrystalline array of viral particles (5B, Bar = 95 nm). Similar viral particles are also present in the nucleus of a renal tubular epithelial cell (5C, Bar = 210 nm). Lead citrate, uranyl acetate. Processing artifact is due to prior histologic processing and staining of specimen (Fig. 2).
Demonstration of intraepithelial PCV2 antigen by immunohistochemistry has been reported by several researchers, 1, 6– 8, 16 and the presence of characteristic intraepithelial PCV2 inclusion bodies in renal tubular epithelium of pigs affected by natural PMWS was also reported recently in New Zealand 2 and the United States. 14 Intracytoplasmic PCV2 inclusion bodies in bronchial and bronchial glandular epithelium have not been reported previously.
The pathogenesis of PCV2 infection and intracytoplasmic inclusion bodies is not completely understood. PCV2, a DNA virus, replicates in the nuclei of host cells as the virus itself is thought to lack DNA polymerase. 9 The presence of intracytoplasmic PCV2 antigen in macrophages is thought to be owing to phagocytosis of PCV2 infected cells. 7 The mechanism behind the formation of intracytoplasmic PCV2 inclusion bodies in epithelial cells is not known; however, it is possible that epithelial cells engulf adjacent apoptotic cells or apoptotic cellular fragments containing viral particles, which subsequently are observed as typical PCV2 intracytoplasmic inclusions.
The severity of characteristic PMWS lesions within tissues and the presence or absence of inclusion bodies in lymphoid tissues was not correlated with the presence or absence of intracytoplasmic epithelial inclusions. The gross and histologic lesions in pigs from Farm 1 (Pig Nos. 1–4) were mild to moderate, whereas in pigs from Farm 2 (Pig No. 5) and Farm 3 (Pig No. 6) lesions were severe. In addition, Pig No. 7, although not diagnosed with PMWS, did have intracytoplasmic inclusions in the epithelium of bronchial glands and PCV2 antigen and DNA were demonstrated in the lungs by immunohistochemical staining, FAT, and PCR, respectively. Accordingly, this pig may have been in the early stages of PCV2 infection. Only the lungs and heart of Pig No. 7 were submitted from Farm 4, which was reportedly without problems associated with PMWS. The lungs of Pig No. 7 exhibited a mild chronic lymphoplasmacytic and histiocytic interstitial pneumonia superimposed by an acute interstitial pneumonia characterized by moderate fibrinous alveolar exudation, neutrophilic infiltration, and disseminated capillary thrombosis consistent with bacterial septicemia.
The clinical significance and pathogenetic relevance of intracytoplasmic epithelial PCV2 inclusion bodies are not clear. The respiratory epithelial infection might be important in the early stages of disease development, whereas renal tubular infection might contribute to secretion and eventual transmission of PCV2 among pigs.
Footnotes
Acknowledgements
We thank personnel at their respective diagnostic laboratories (Prairie Diagnostic Services and Atlantic Veterinary College Diagnostic Services) for all their help and effort in this investigation. We are also thankful to Ian Shirley and Jennifer Cowell for their help and expertise with electron microscopy performed on histologic sections.
